|
1
|
Betjemann JP and Lowenstein DH: Status
epilepticus in adults. Lancet Neurol. 14:615–624. 2015.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Trinka E, Cock H, Hesdorffer D, Rossetti
AO, Scheffer IE, Shinnar S, Shorvon S and Lowenstein DH: A
definition and classification of status epilepticus-report of the
ILAE task force on classification of status epilepticus. Epilepsia.
56:1515–1523. 2015.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Wong M: Mammalian target of rapamycin
(mTOR) inhibition as a potential antiepileptogenic therapy: From
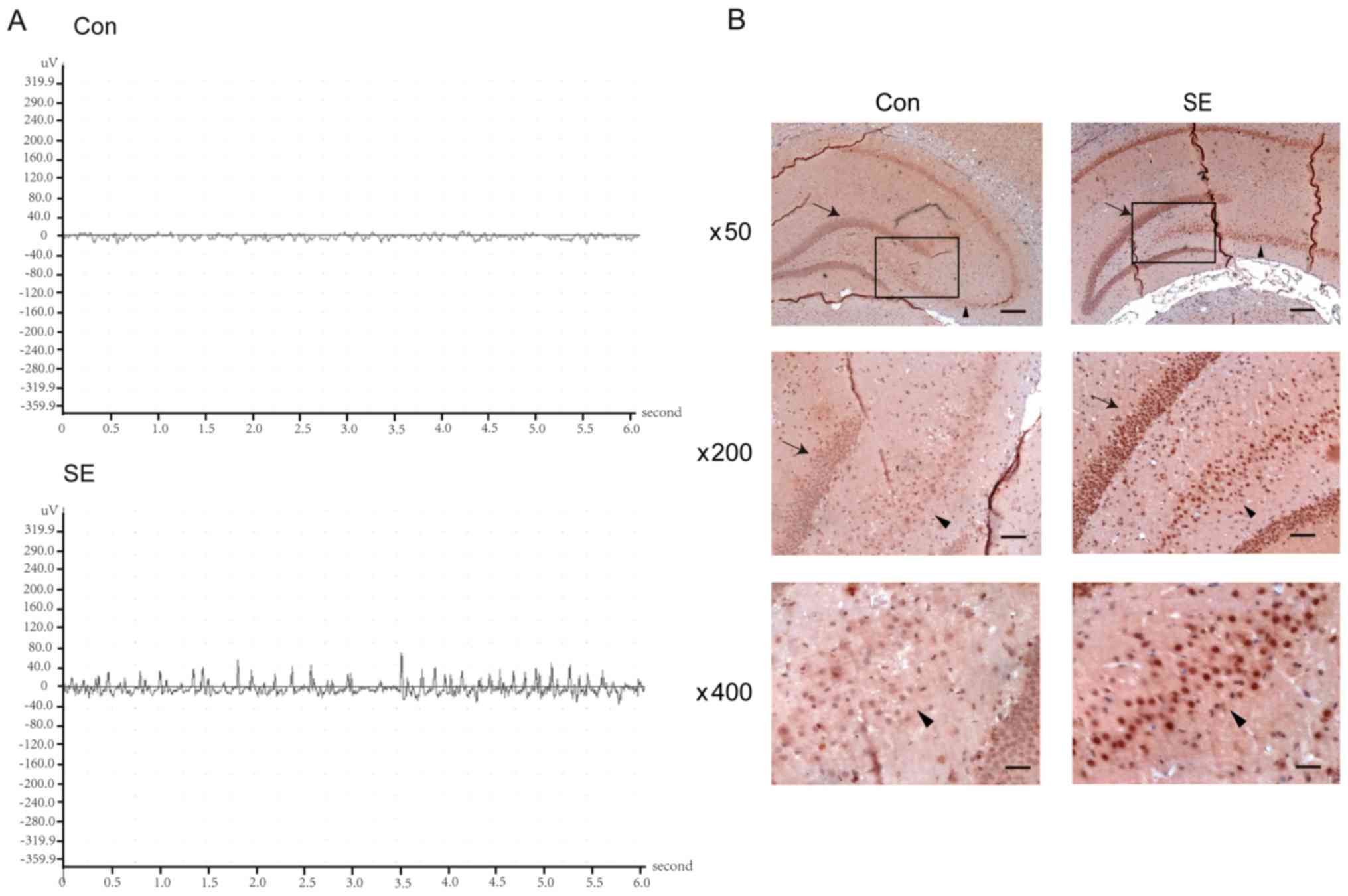
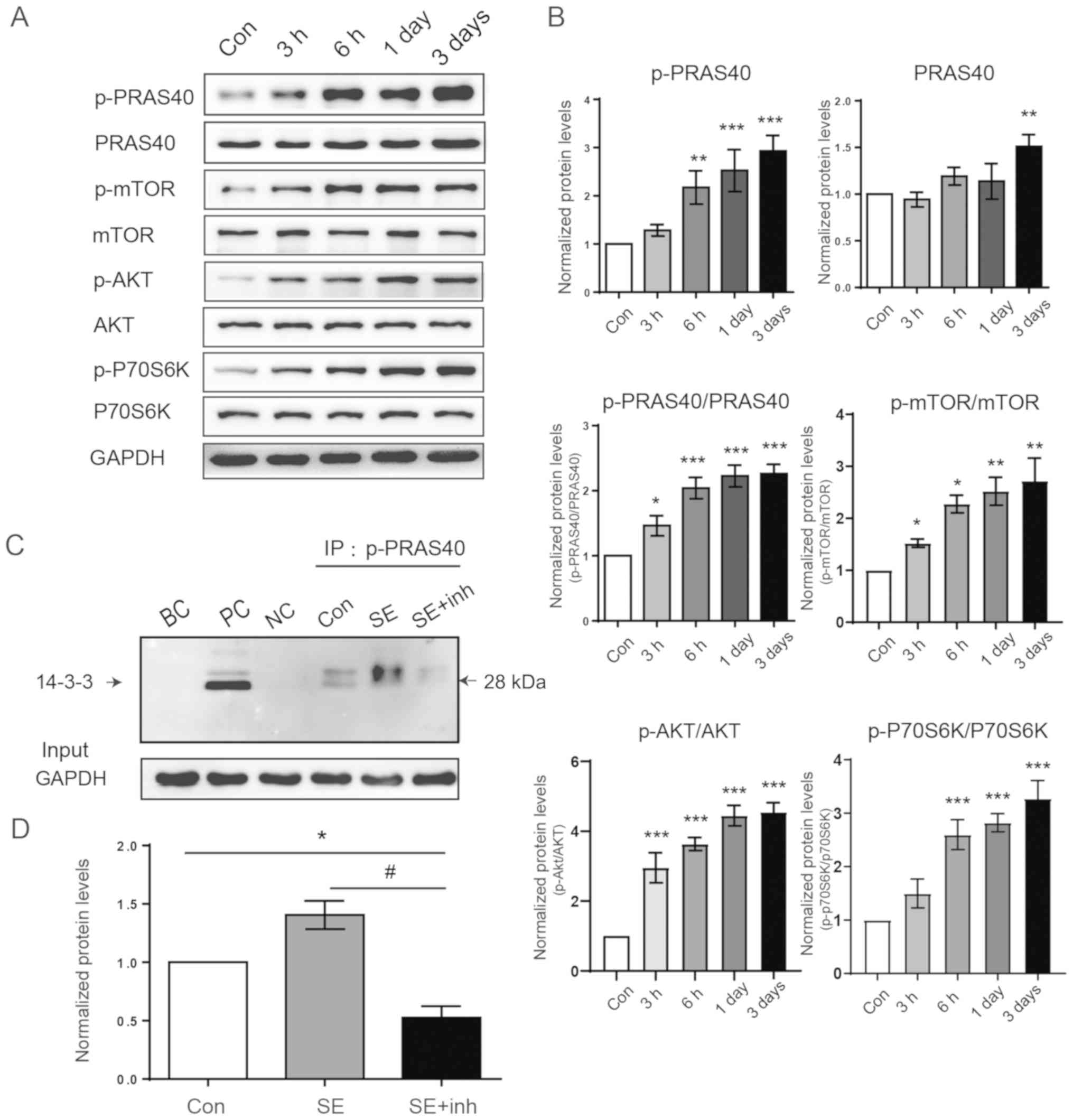
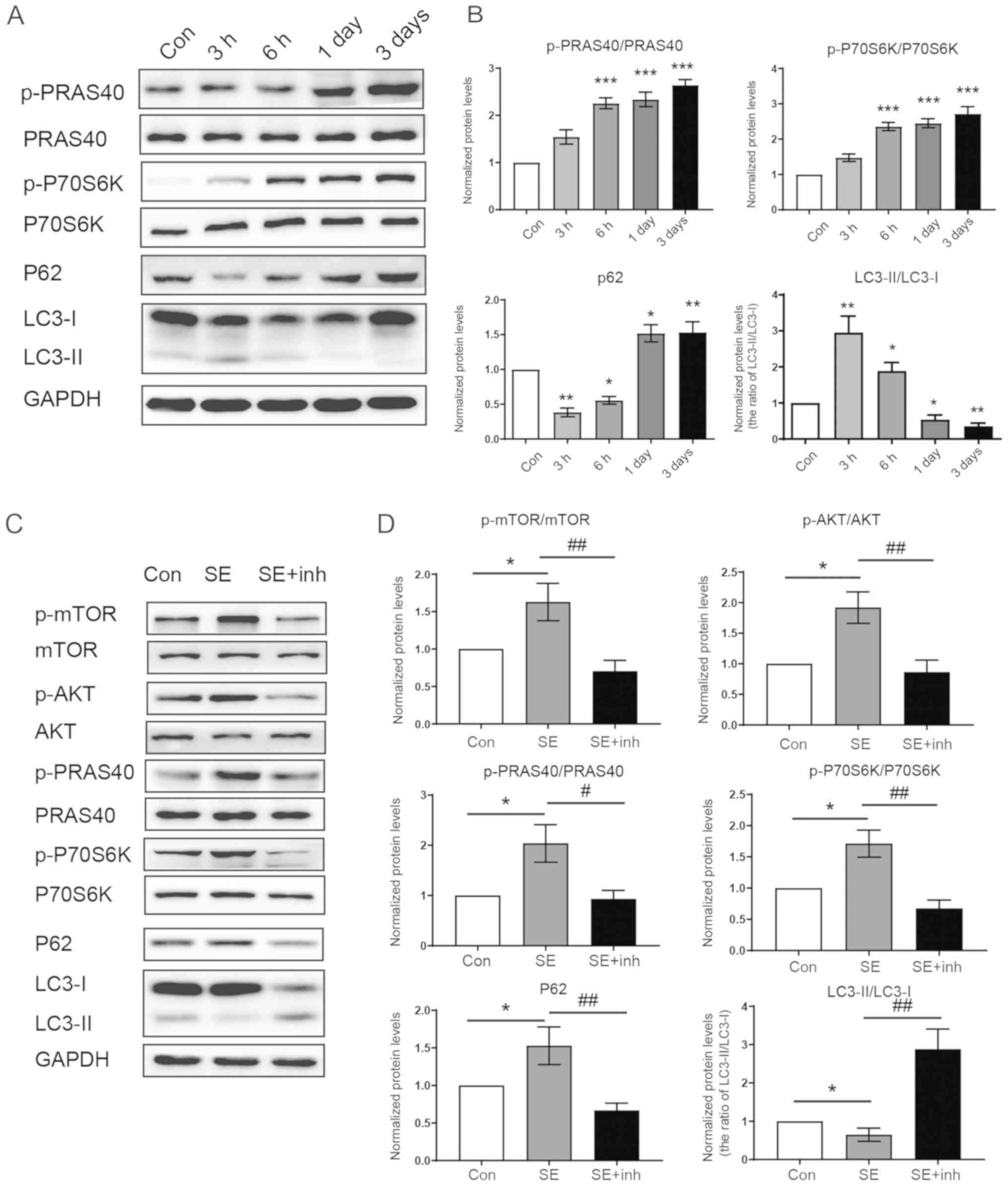
tuberous sclerosis to common acquired epilepsies. Epilepsia.
51:27–36. 2010.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Kovacina KS, Park GY, Bae SS, Guzzetta AW,
Schaefer E, Birnbaum MJ and Roth RA: Identification of a
proline-rich Akt substrate as a 14-3-3 binding partner. J Biol
Chem. 278:10189–10194. 2003.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Nascimento EB, Snel M, Guigas B, van der
Zon GCM, Kriek J, Maassen JA, Jazet IM, Diamant M and Ouwens DM:
Phosphorylation of PRAS40 on Thr246 by PKB/AKT facilitates
efficient phosphorylation of Ser183 by mTORC1. Cell Signal.
22:961–967. 2010.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Thedieck K, Polak P, Kim ML, Molle KD,
Cohen A, Jenö P, Arrieumerlou C and Hall MN: PRAS40 and PRR5-like
protein are new mTOR interactors that regulate apoptosis. PLoS One.
2(e1217)2007.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Kazi AA and Lang CH: PRAS40 regulates
protein synthesis and cell cycle in C2C12 myoblasts. Mol Med.
16:359–371. 2010.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Hong-Brown LQ, Brown CR, Kazi AA, Huber
DS, Pruznak AM and Lang CH: Alcohol and PRAS40 knockdown decrease
mTOR activity and protein synthesis via AMPK signaling and changes
in mTORC1 interaction. J Cell Biochem. 109:1172–1184.
2010.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Lipton JO and Sahin M: The neurology of
mTOR. Neuron. 84:275–291. 2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
San YZ, Liu Y, Zhang Y, Shi PP and Zhu YL:
Peroxisome proliferator-activated receptor-γ agonist inhibits the
mammalian target of rapamycin signaling pathway and has a
protective effect in a rat model of status epilepticus. Mol Med
Rep. 12:1877–1883. 2015.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Brewster AL, Lugo JN, Patil VV, Lee WL,
Qian Y, Vanegas F and Anderson AE: Rapamycin reverses status
epilepticus-induced memory deficits and dendritic damage. PLoS One.
8(e57808)2013.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Wang SJ, Bo QY, Zhao XH, Yang X, Chi ZF
and Liu XW: Resveratrol pre-treatment reduces early inflammatory
responses induced by status epilepticus via mTOR signaling. Brain
Res. 1492:122–129. 2013.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Kim KH and Lee MS: Autophagy-a key player
in cellular and body metabolism. Nat Rev Endocrinol. 10:322–337.
2014.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Munson MJ and Ganley IG: MTOR, PIK3C3, and
autophagy: Signaling the beginning from the end. Autophagy.
11:2375–2376. 2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Cao L, Xu J, Lin Y, Zhao X, Liu X and Chi
Z: Autophagy is upregulated in rats with status epilepticus and
partly inhibited by vitamin E. Biochem Biophys Res Commun.
379:949–953. 2009.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Dong Y, Wang S, Zhang T, Zhao X, Liu X,
Cao L and Chi Z: Ascorbic acid ameliorates seizures and brain
damage in rats through inhibiting autophagy. Brain Res.
1535:115–123. 2013.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Benz AP, Niquet J, Wasterlain CG and Rami
A: Status epilepticus in the immature rodent brain alters the
dynamics of autophagy. Curr Neurovasc Res. 11:125–135.
2014.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Rami A, Benz AP, Niquet J and Langhagen A:
Axonal accumulation of lysosomal-associated membrane protein 1
(LAMP1) accompanying alterations of autophagy dynamics in the rat
hippocampus upon seizure-induced injury. Neurochem Res. 41:53–63.
2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Shacka JJ, Lu J, Xie ZL, Uchiyama Y, Roth
KA and Zhang J: Kainic acid induces early and transient autophagic
stress in mouse hippocampus. Neurosci Lett. 414:57–60.
2007.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Racine RJ: Modification of seizure
activity by electrical stimulation. II. Motor seizure.
Electroencephalogr Clin Neurophysiol. 32:281–294. 1972.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Dibble CC and Cantley LC: Regulation of
mTORC1 by PI3K signaling. Trends Cell Biol. 25:545–555.
2015.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Sloviter RS: The functional organization
of the hippocampal dentate gyrus and its relevance to the
pathogenesis of temporal lobe epilepsy. Ann Neurol. 35:640–654.
1994.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Sutula TP and Dudek FE: Unmasking
recurrent excitation generated by mossy fiber sprouting in the
epileptic dentate gyrus: An emergent property of a complex system.
Prog Brain Res. 163:541–563. 2007.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Nascimento EB, Fodor M, van der Zon GC,
Jazet IM, Meinders AE, Voshol PJ, Vlasblom R, Baan B, Eckel J,
Maassen JA, et al: Insulin-mediated phosphorylation of the
proline-rich Akt substrate PRAS40 is impaired in insulin target
tissues of high-fat diet-fed rats. Diabetes. 55:3221–3228.
2006.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Saito A, Hayashi T, Okuno S, Nishi T and
Chan PH: Modulation of proline-rich akt substrate survival
signaling pathways by oxidative stress in mouse brains after
transient focal cerebral ischemia. Stroke. 37:513–517.
2006.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Saito A, Narasimhan P, Hayashi T, Okuno S,
Ferrand-Drake M and Chan PH: Neuroprotective role of a proline-rich
Akt substrate in apoptotic neuronal cell death after stroke:
Relationships with nerve growth factor. J Neurosci. 24:1584–1593.
2004.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Feliciano DM, Su T, Lopez J, Platel JC and
Bordey A: Single-cell Tsc1 knockout during corticogenesis generates
tuber-like lesions and reduces seizure threshold in mice. J Clin
Invest. 121:1596–1607. 2011.PubMed/NCBI View
Article : Google Scholar
|
|
28
|
Luo CL, Li BX, Li QQ, Chen XP, Sun YX, Bao
HJ, Dai DK, Shen YW, Xu HF, Ni H, et al: Autophagy is involved in
traumatic brain injury-induced cell death and contributes to
functional outcome deficits in mice. Neuroscience. 184:54–63.
2011.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Wang K, Huang J, Xie W, Huang L, Zhong C
and Chen Z: Beclin1 and HMGB1 ameliorate the α-synuclein-mediated
autophagy inhibition in PC12 cells. Diagn Pathol.
11(15)2016.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Shin JY, Park HJ, Kim HN, Oh SH, Bae JS,
Ha HJ and Lee PH: Mesenchymal stem cells enhance autophagy and
increase β-amyloid clearance in Alzheimer disease models.
Autophagy. 10:32–44. 2014.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Aguado C, Sarkar S, Korolchuk VI, Criado
O, Vernia S, Boya P, Sanz P, de Córdoba SR, Knecht E and
Rubinsztein DC: Laforin, the most common protein mutated in Lafora
disease, regulates autophagy. Hum Mol Genet. 19:2867–2876.
2010.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Hosseinzadeh M, Nikseresht S, Khodagholi
F, Naderi N and Maghsoudi N: Cannabidiol post-treatment alleviates
rat epileptic-related behaviors and activates hippocampal cell
autophagy pathway along with antioxidant defense in chronic phase
of pilocarpine-induced seizure. J Mol Neurosci. 58:432–440.
2016.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Ni H, Zhao DJ and Tian T: Ketogenic diet
change cPLA2/clusterin and autophagy related gene expression and
correlate with cognitive deficits and hippocampal MFs sprouting
following neonatal seizures. Epilepsy Res. 120:13–18.
2016.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Jin R, Zhu W, Cao S, Chen R, Jin H, Liu Y,
Wang S, Wang W and Xiao G: Japanese encephalitis virus activates
autophagy as a viral immune evasion strategy. PLoS One.
8(e52909)2013.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Li L, Tian J, Long MK, Chen Y, Lu J, Zhou
C and Wang T: Protection against experimental stroke by ganglioside
GM1 is associated with the inhibition of autophagy. PLoS One.
11(e144219)2016.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Klionsky DJ, Abdalla FC, Abeliovich H,
Abraham RT, Acevedo-Arozena A, Adeli K, Agholme L, Agnello M,
Agostinis P, Aguirre-Ghiso JA, et al: Guidelines for the use and
interpretation of assays for monitoring autophagy. Autophagy.
8:445–544. 2012.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Schaaf MB, Keulers TG, Vooijs MA and
Rouschop KM: LC3/GABARAP family proteins: Autophagy-(un)related
functions. FASEB J. 30:3961–3978. 2016.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Sini P, James D, Chresta C and Guichard S:
Simultaneous inhibition of mTORC1 and mTORC2 by mTOR kinase
inhibitor AZD8055 induces autophagy and cell death in cancer cells.
Autophagy. 6:553–554. 2010.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Lamark T, Svenning S and Johansen T:
Regulation of selective autophagy: The p62/SQSTM1 paradigm. Essays
Biochem. 61:609–624. 2017.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Nezis IP, Simonsen A, Sagona AP, Finley K,
Gaumer S, Contamine D, Rusten TE, Stenmark H and Brech A: Ref(2)P,
the drosophila melanogaster homologue of mammalian p62, is required
for the formation of protein aggregates in adult brain. J Cell
Biol. 180:1065–1071. 2008.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Bartlett BJ, Isakson P, Lewerenz J,
Sanchez H, Kotzebue RW, Cumming RC, Harris GL, Nezis IP, Schubert
DR, Simonsen A and Finley KD: p62, ref(2)P and ubiquitinated
proteins are conserved markers of neuronal aging, aggregate
formation and progressive autophagic defects. Autophagy. 7:572–583.
2011.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Buckmaster PS, Ingram EA and Wen X:
Inhibition of the mammalian target of rapamycin signaling pathway
suppresses dentate granule cell axon sprouting in a rodent model of
temporal lobe epilepsy. J Neurosci. 29:8259–8269. 2009.PubMed/NCBI View Article : Google Scholar
|