Introduction
With the change in people's quality of life, the
incidence of cerebral infarction increases, leading to an increase
of mortality in patients with cardiovascular and cerebrovascular
diseases (1). For stroke patients,
conventional treatment mainly includes anticoagulation,
thrombolysis and other supportive treatments (2). In recent years, cerebrovascular stent
implantation has been applied in the interventional therapy of
cerebral infarction, which has achieved great success and has been
widely applied in clinical practice (3). However, cerebrovascular restenosis is
often complicated during the later operation of stent implantation,
which is one of the difficulties that hinder the recovery of
patients after surgery. The disease deteriorates severely, and the
mortality is high (4,5). Therefore, it is urgent to study
predictors of restenosis after cerebrovascular stent implantation
to save patients' lives and assist their recovery.
It has been found that serum complement Clq tumor
necrosis factor-related protein 9 (CTRP9) is one of the adipocyte
factors synthesized and released by adipose tissue. It is highly
homologous with adiponectin during evolution, has the functions of
regulating inflammatory response, anti-endothelial dysfunction,
protecting myocardium and the metabolism of lipolysaccharide, and
plays a role in regulating the stability of the body (6-10).
Stromal interaction molecule 1 (STIM1) is closely related to
endothelial cell function and homing of endothelial progenitor
cells. The increased content may lead to injury of endothelial
cells and delay of stent reendothelialization after cardiac stent
implantation (11,12). However, there are relatively few
studies on the effect of serum CTRP9 and STIM1 on restenosis after
cerebrovascular stent implantation. Therefore, this study
investigated the predictive value of serum CTRP9 and STIM1 for
restenosis after cerebrovascular stent implantation, and their
relationship with the vasoactive substances nitric oxide (NO),
tumor necrosis factor α (TNF-α), and interleukin-6 (IL-6) to
validate biological indicators of early restenosis in
cerebrovascular stent implantation.
Patients and methods
Baseline data
In this prospective study, 128 cases of cerebral
infarction treated with cerebrovascular stent implantation in
Yantaishan Hospital (Yantai, China) from March 2013 to January 2016
were selected as experimental subjects. A total of 66 cases with
restenosis after cerebrovascular stent implantation were included
in group A, while 62 cases without stenosis were included in group
B. There were 75 males and 53 females, with an average age of
54.82±7.74. There was no significant difference in gender, age,
etc. between the two groups (all P>0.05; Table I).
 | Table IBaseline data of patients. |
Table I
Baseline data of patients.
| Characteristics | Group A (n=66) | Group B (n=62) | χ2
test | P-value |
|---|
| Age (years) | 53.58±7.36 | 54.36±7.86 | 0.580 | 0.563 |
| Sex [n (%)] | | | 0.058 | 0.809 |
|
Male | 38 (57.58) | 37 (59.68) | | |
|
Female | 28 (42.42) | 25 (40.32) | | |
| Height (cm) | 163.53±7.45 | 164.35±7.14 | 0.635 | 0.527 |
| BMI
(kg/m2) | 25.64±2.34 | 25.57±2.28 | 0.171 | 0.864 |
| Smoking history [n
(%)] | 48 (72.73) | 44 (70.97) | 0.049 | 0.825 |
| Drinking history [n
(%)] | 51 (77.27) | 49 (79.03) | 0.058 | 0.810 |
| Past history [n
(%)] | | | | |
|
Hypertension | 25 (37.88) | 22 (35.48) | 0.079 | 0.779 |
|
Coronary
heart disease | 14 (21.21) | 11 (17.74) | 0.245 | 0.621 |
|
Diabetes
mellitus | 17 (25.76) | 15 (24.19) | 0.042 | 0.838 |
|
Hyperlipidemia | 16 (24.24) | 17 (27.42) | 0.169 | 0.681 |
Inclusion and exclusion criteria
Inclusion criteria: Patients diagnosed with cerebral
infarction by cerebral angiography technology.
Exclusion criteria: Patients with abnormal function
of previous coagulation; patients with contraindications of stent
implantation; patients complicated with endocrine system diseases;
patients with severe hepatic and kidney function obstacle; patients
with cognitive impairment or communication barrier; patients with
poor compliance.
This study was approved by the Ethics Committee of
Yantaishan Hospital (approval no. YTSH20130301). All the patients
and their families were well informed, and a signed informed
consent was obtained.
Experimental reagents and
materials
CTRP9 kit was purchased from Aviscera Bioscience
Co., (XY-RD191180200R). STIM1 kit was purchased from Shanghai
Renjie Biotechnology Co., Ltd., (RJ12740). NO kit and IL-6 kit were
purchased from Wuhan Mskbio Biotechnology Co., Ltd. (KT76381,
KT63251). TNF-α kit was purchased from Nanjing Camilo
Bioengineering Co., Ltd. (H-KMLJ37541).
Detection of serum CTRP9 and
STIM1
Serum expression levels of CTRP9 and STIM1 of study
subjects were measured by ELISA. After 1 month of cerebrovascular
stent implantation, 5 ml of patient's venous blood was collected
and centrifuged at room temperature for 8 min. The serum was
separated at 1,500 x g, sealed and stored at -80˚C for testing. A
total of 100 µl of serum sample was added to the orifice coated
with anti-CTRP9 antibody. A blank well and a standard well were set
up. The CTRP9 antibody was added at 37˚C for reaction for 90 min,
and the liquid was discarded, then dried and washed repeatedly
three times. A total of 150 µl of the chromogenic reagent was added
at 37˚C for reaction for 30 min. A total of 50 µl of the mixture
was taken out, mixed and put into enzyme-labelling measuring
instrument to measure the average optical density of each well by
500 nm wavelength. STIM1, NO, TNF-α and IL-6 levels were detected
as above in strict accordance with the ELISA kit instructions.
Observation indexes
Comparison of serum CTRP9 and STIM1 levels of
patients in the two groups; comparison of NO, TNF-α and IL-6 levels
of patients in the two groups; comparison of diagnostic efficacy of
serum CTRP9, STIM1 and their combination; analysis of the
correlation between serum CTRP9, STIM1 levels and NO, TNF-α, IL-6
levels.
Statistical methods
SPSS 19.0 statistical software (Shanghai Kabei
Information Technology Co., Ltd.) was used for statistical analysis
of experimental data. The counting data were tested by chi-square
test. Measurement data were expressed as mean ± standard deviation.
T test was used for comparison between two groups. The diagnostic
value of CTRP9, STIM1 and their combination in restenosis after
cerebrovascular stent implantation was analyzed by ROC. Correlation
analysis was performed by Pearson's correlation coefficient.
Graphpad Prism8 was used for image rendering. P<0.05 was
considered to indicate a statistically significant difference.
Results
Comparison of serum CTRP9 and STIM1
levels of patients in the two groups
Serum CTRP9 level in group A was significantly lower
than that in group B. Serum STIM1 level in group A was
significantly higher than that in group B. The differences were
statistically significant (both P<0.001). More details are shown
in Table II.
 | Table IIComparison of serum CTRP9 and STIM1
levels in the two groups. |
Table II
Comparison of serum CTRP9 and STIM1
levels in the two groups.
| Factors | Group A (n=66) | Group B (n=62) | t | P-value |
|---|
| CTRP9 (ng/ml) | 159.64±32.55 | 184.53±34.53 | 4.190 | <0.001 |
| STIM1 (U/l) | 12.15±1.44 | 11.31±0.92 | 3.905 | <0.001 |
Comparison of NO, TNF-α, IL-6 levels
of patients in the two groups
Serum NO level in group A was significantly lower
than that in group B. Serum TNF-α and IL-6 levels in group A were
significantly higher than those in group B. The differences were
statistically significant (all P<0.001). More details are shown
in Table III.
 | Table IIIComparison of NO, TNF-α, IL-6 levels
of patients in the two groups. |
Table III
Comparison of NO, TNF-α, IL-6 levels
of patients in the two groups.
| Factors | Group A (n=66) | Group B (n=62) | t | P-value |
|---|
| NO (mol/l) | 156.57±12.58 | 172.73±13.83 | 6.901 | <0.001 |
| TNF-α (ng/l) | 10.52±1.68 | 7.78±0.93 | 11.310 | <0.001 |
| IL-6 (ng/l) | 98.75±13.53 | 83.48±9.84 | 7.263 | <0.001 |
Comparison of serum CTRP9, STIM1
levels and combined diagnostic efficacy by ROC curve analysis
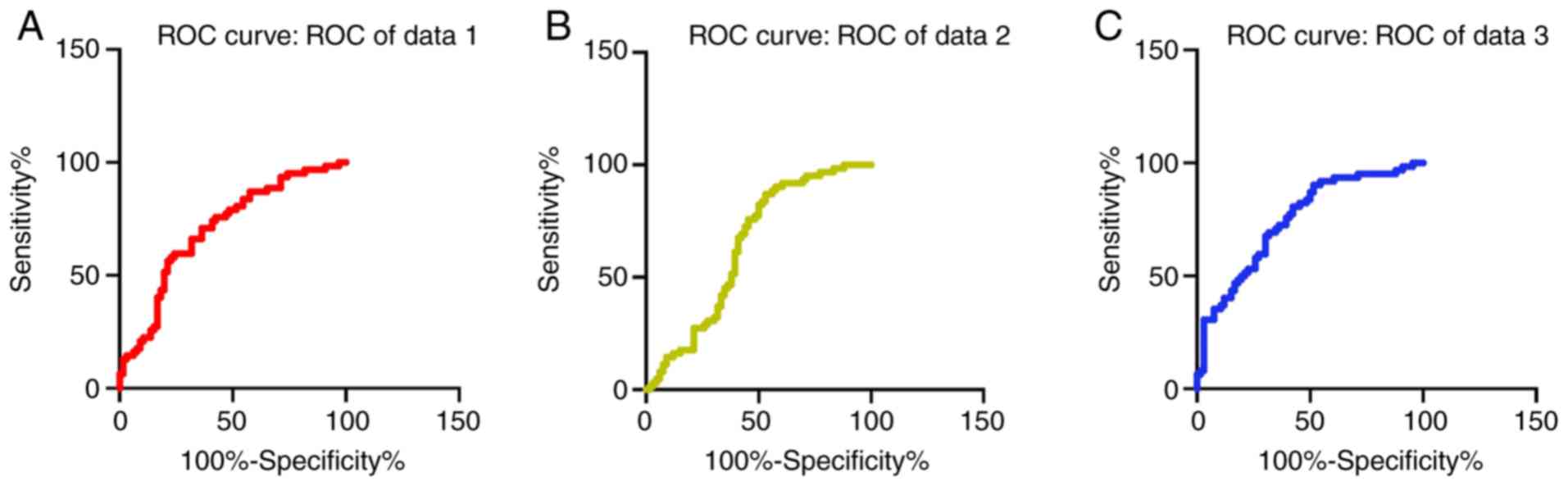
The sensitivity, specificity, AUC and cut-off of
serum CTRP9 level in the diagnosis of restenosis after
cerebrovascular stent implantation were 59.68, 75.76, 0.705 and
180.6%, respectively. The sensitivity, specificity, AUC and cut-off
of serum STIM1 level in diagnosis of restenosis after
cerebrovascular stent implantation were 87.10, 46.97, 0.637 and
12.24%, respectively. The sensitivity, specificity, AUC and cut-off
of the combination of serum CTRP9 and STIM1 levels in the combined
diagnosis of restenosis after cerebrovascular stent implantation
were 90.32, 48.48, 0.747 and 0.484%, respectively (Fig. 1).
Analysis of the correlation between
serum CTRP9, STIM1 levels and NO, TNF-α, IL-6 levels
After cerebrovascular stent implantation, the level
of CTRP9 was positively correlated with NO (r=0.711, P<0.001),
and negatively correlated with TNF-α (r=-0.761, P<0.001) and
IL-6 (r=-0.751, P<0.001). The level of STIM1 was negatively
correlated with NO (r=-0.761, P<0.001), and positively
correlated with TNF-α (r=0.776, P<0.001) and IL-6 (r=0.709,
P<0.001) (Fig. 2).
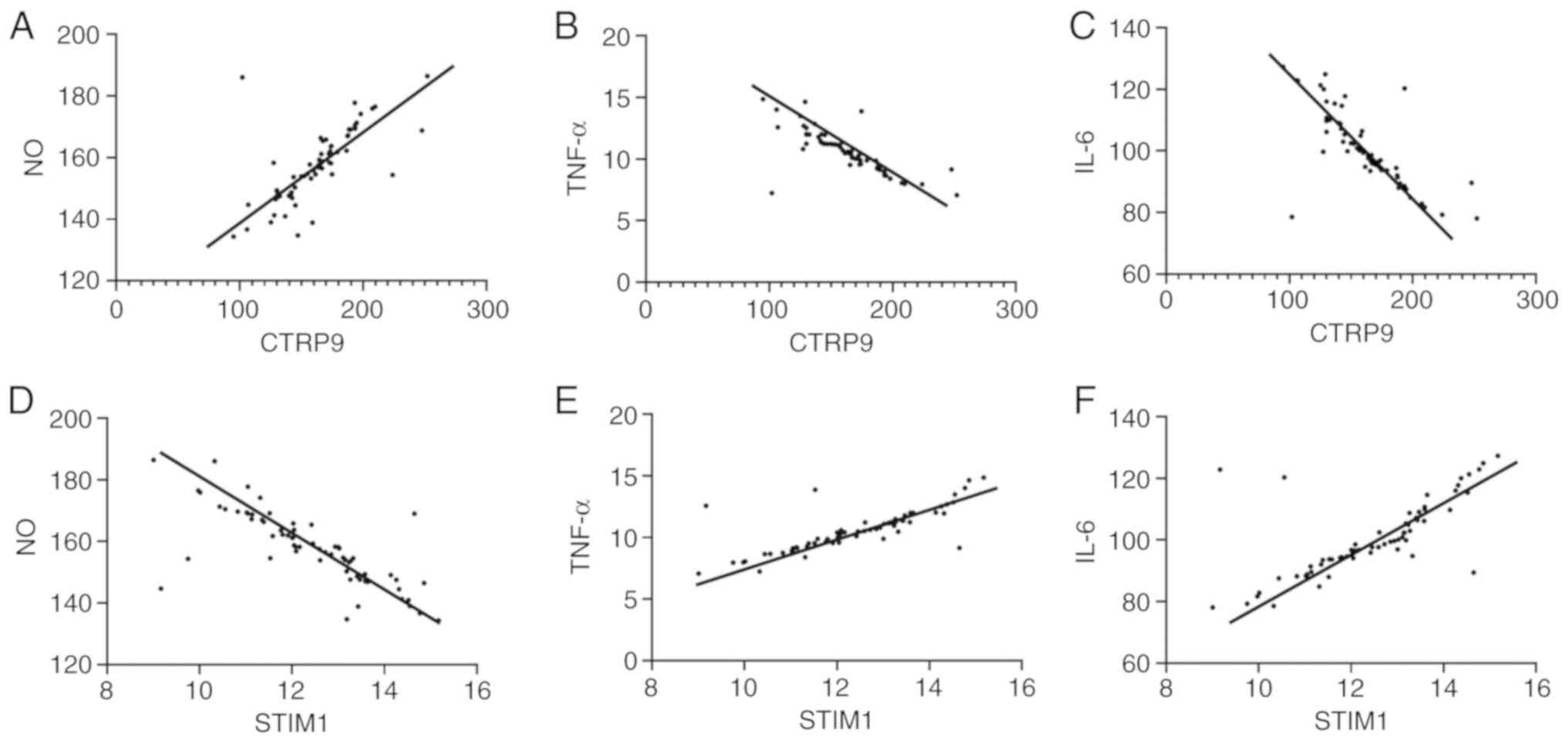 | Figure 2Analysis of the correlation between
serum CTRP9, STIM1 levels and NO, TNF-α, IL-6 levels after
cerebrovascular stent implantation. (A) Analysis of the correlation
between serum CTRP9 levels and NO levels after cerebrovascular
stent implantation. Pearson's analysis showed that the expression
of CTRP9 and NO was positively correlated (r=0.711, P<0.001).
(B) Analysis of the correlation between serum CTRP9 levels and
TNF-α levels after cerebrovascular stent implantation. Pearson
analysis showed that the expression of CTRP9 and TNF-α was
negatively correlated (r=-0.761, P<0.001). (C) Analysis of the
correlation between serum CTRP9 levels and IL-6 levels after
cerebrovascular stent implantation. Pearson's analysis showed that
the expression of CTRP9 and IL-6 was negatively correlated
(r=-0.751, P<0.001). (D) Analysis of the correlation between
serum STIM1 levels and NO levels after cerebrovascular stent
implantation. Pearson's analysis showed that the expression of
STIM1 and NO was negatively correlated (r=-0.761, P<0.001). (E)
Analysis of the correlation between serum STIM1 levels and TNF-α
levels after cerebrovascular stent implantation. Pearson's analysis
showed that the expression of STIM1 and TNF-α was positively
correlated (r=0.776, P<0.001). (F) Analysis of the correlation
between serum STIM1 levels and IL-6 levels after cerebrovascular
stent implantation. Pearson's analysis showed that the expression
of STIM1 and IL-6 was positively correlated (r=0.709, P<0.001).
NO, nitric oxide; TNF-α, tumor necrosis factor α; IL-6;
interleukin-6. |
Discussion
Many studies have shown that the incidence of
postoperative in-stent restenosis is higher than that of
non-operative surgery (13,14). However, the mechanism of restenosis
after cerebrovascular stent implantation remain unclear. Most
scholars agree that it is related to vascular remodeling, intima
abnormal growth, polymer absorption hindering inflammatory response
and postoperative vascular elastic retraction and other factors
(15,16). It is of practical significance to
find out the cause of restenosis after cerebrovascular stent
implantation. It has been reported (17) that STIM1 is a sensory receptor for
calcium channel of calcium store controllability in vivo.
The biological effects of cells are affected by controlling
intracellular calcium concentration to regulate vasoconstriction.
In vitro experiments have shown that CTRP9 can inhibit the
proliferation of smooth muscle cells and angiogenesis and the
formation of vascular new intima through Cyclic AMP
(cAMP)-independent mechanism after injury of vascular (18). The increase of nitric oxide (NO)
production promotes vasodilation and greatly improves endothelial
cell function (19). These effects
may be related to STIM1, CTRP9 and restenosis after cerebrovascular
stenting. Therefore, this study investigated the effects of serum
CTRP9 and STIM1 on restenosis after cerebrovascular stent
implantation, and explored their relationship with vasoactive
substances and inflammatory cytokines.
Based on the results of this study, serum CTRP9
level in group A was significantly lower than that in group B, and
serum STIM1 level in group A was significantly higher than that in
group B (all P<0.001), suggesting that serum CTRP9 low
expression and STIM1 over-expression after cerebrovascular stent
implantation may increase the risk of in-stent restenosis. When
STIM1 expression is released, it inhibits the biological and homing
functions of endothelial progenitor cells, directly damaging the
repair of local vascular injury after stent implantation (20). According to a previous study
(21), the increase of free fatty
acids during tissue ischemia/reperfusion injury promotes the
oxidative stress and increase of expression of CTRP9, and improves
the metabolism function of the body to protect damaged myocardial
cells, which is consistent with our results. The levels of serum
NO, TNF-α and IL-6 in group A were significantly higher than those
in group B (all P<0.001). Pearson results showed that serum
CTRP9 level was positively correlated with NO and negatively
correlated with TNF-α and IL-6. STIM1 was positively correlated
with TNF-α and IL-6 and negatively correlated with NO (all
P<0.001). The sustainable or controlled release of NO has
diastolic effect on the endothelium and can repair and accelerate
the vascular regeneration (22).
TNF-α can regulate the expression of vasomotion substances and
cause vasoconstriction after the decrease of the levels of
vasorelaxation factors (23,24). It has been reported that IL-6 is
involved in tissue fibrosis and induces the release of a large
amount of inflammatory mediators under the effect of vascular
endothelial tissue injury (25). In
summary, serum TNF-α and IL-6 levels increase with vascular
stenosis, while NO expression decreases. Combined with the results
of CTRP9 and STIM1, the results of correlation between serum CTRP9,
STIM1 levels and NO, TNF-α and IL-6 were inferred and confirmed,
which further confirmed the predictive value of serum CTRP9 and
STIM1 levels on restenosis after cerebrovascular stent
implantation. The sensitivity of serum CTRP9 level in the diagnosis
of restenosis after cerebrovascular stent implantation was 59.68%,
and the specificity was 75.76%. The sensitivity of serum STIM1
level in diagnosis of restenosis after cerebrovascular stent
implantation was 87.10% and the specificity was 46.97%. The
sensitivity of serum CTRP9 and STIM1 levels in the combined
diagnosis of restenosis after cerebrovascular stent implantation
was 90.32% and the specificity was 48.48%. It indicated that both
CTRP9 and STIM1 may be involved in the disease process of
pathophysiological mechanism of restenosis after cerebrovascular
stent implantation. The regulation and protection of CTRP9 and
STIM1 on cerebrovascular function result in abnormal changes of
intravascular components after reversal. There is substantial
heterogeneity in the influence of signaling pathways and action
sites, and the emphasis indicates that metabolic regulation is also
different. Synergistic combined detection may improve the
diagnostic efficacy of cardiovascular and cerebrovascular
diseases.
Based on previous studies and the results of this
study, it can be learned that the decreased level of CTRP9 and
increased level of STIM1 after cerebrovascular stent implantation
are closely related to vascular formation, repair and
re-endothelialization. However, there are some limitations in this
study. The specific differences between the combined diagnosis of
CTRP9 and STIM1 and the separate diagnosis are not discussed in
detail. We did not rule out the interference of other factors on
these indexes, and failed to use the two factors as early
predictors of restenosis after cerebral vascular stenting. These
are the research topics of the future study. However, the results
of this study have preliminarily shown that CTRP9 and STIM1 may
play a role in the early prevention of restenosis after
cerebrovascular stent implantation, providing a new theoretical
direction for the diagnosis and prognostic evaluation of restenosis
after cerebrovascular stent implantation.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
JP, XC and LZ conceived and designed the study. JP,
GW, KX, JH and LZ were responsible for the acquisition, analysis
and interpretation of the data. JP drafted the manuscript. XC and
LZ revised the manuscript critically for important intellectual
content. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
The study was approved by the Ethics Committee of
Yantaishan Hospital (Yantai, China). Signed informed consents were
obtained from the patients and/or guardians.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Losito A, Pittavini L, Ferri C and De
Angelis L: Kidney function and mortality in different
cardiovascular diseases: Relationship with age, sex, diabetes and
hypertension. J Nephrol. 24:322–328. 2011.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Sairanen T, Strbian D, Ruuskanen R,
Silvennoinen H, Salonen O and Lindsberg PJ: Symptomatic
intracranial haemorrhage after thrombolysis with adjuvant
anticoagulation in basilar artery occlusion. Eur J Neurol.
22:493–499. 2015.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Mintz GS: Intravascular ultrasound and
outcomes after drug- eluting stent implantation. Coron Artery Dis.
28:346–352. 2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Guan CL, Shi GX, Li Q, Luo YM and Zeng YC:
Prevention of complications following cerebrovascular stent
implantation. J Clin Rehab Tissue Eng Res. 2 (Suppl)(S52)2010.
|
|
5
|
Dinesch V, Dinesch M and Buruian M:
Drug-eluting stent in-stent restenosis and re-restenosis: A hard
time story. J Saudi Heart Assoc. 29:73–74. 2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Ouchi N, Parker JL, Lugus JJ and Walsh K:
Adipokines in inflammation and metabolic disease. Nat Rev Immunol.
11:85–97. 2011.PubMed/NCBI View
Article : Google Scholar
|
|
7
|
Lago F, Dieguez C, Gómez-Reino J and
Gualillo O: Adipokines as emerging mediators of immune response and
inflammation. Nat Clin Pract Rheumatol. 3:716–724. 2007.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Smith CC and Yellon DM: Adipocytokines,
cardiovascular pathophysiology and myocardial protection. Pharmacol
Ther. 129:206–219. 2011.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Schäffler A and Buechler C: CTRP family:
Linking immunity to metabolism. Trends Endocrinol Metab.
23:194–204. 2012.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Wong GW, Krawczyk SA, Kitidis-Mitrokostas
C, Ge G, Spooner E, Hug C, Gimeno R and Lodish HF: Identification
and characterization of CTRP9, a novel secreted glycoprotein, from
adipose tissue that reduces serum glucose in mice and forms
heterotrimers with adiponectin. FASEB J. 23:241–258.
2009.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Liou J, Fivaz M, Inoue T and Meyer T:
Live-cell imaging reveals sequential oligomerization and local
plasma membrane targeting of stromal interaction molecule 1 after
Ca2+ store depletion. Proc Natl Acad Sci USA.
104:9301–9306. 2007.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Estrada IA, Donthamsetty R, Debski P, Zhou
MH, Zhang SL, Yuan JX, Han W and Makino A: STIM1 restores coronary
endothelial function in type 1 diabetic mice. Circ Res.
111:1166–1175. 2012.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Edgell RC, Zaidat OO, Gupta R, Abou-Chebl
A, Linfante I, Xavier A, Nogueira R, Alshekhlee A, Kalia J, Etezadi
V, et al: Multicenter study of safety in stenting for symptomatic
vertebral artery origin stenosis: Results from the Society of
Vascular and Interventional Neurology Research Consortium. J
Neuroimaging. 23:170–174. 2013.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Albuquerque FC, Fiorella D, Han P,
Spetzler RF and Mc Dougall CG: A reappraisal of angioplasty and
stenting for the treatment of vertebral origin stenosis.
Neurosurgery. 53:607–616. 2003.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Kang SJ, Mintz GS, Park DW, Lee SW, Kim
YH, Lee CW, Han KH, Kim JJ, Park SW and Park SJ: Mechanisms of
in-stent restenosis after drug-eluting stent implantation:
Intravascular ultrasound analysis. Circ Cardiovasc Interv. 4:9–14.
2011.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Virmani R, Liistro F, Stankovic G, Di
Mario C, Montorfano M, Farb A, Kolodgie FD and Colombo A: Mechanism
of late in-stent restenosis after implantation of a paclitaxel
derivate-eluting polymer stent system in humans. Circulation.
106:2649–2651. 2002.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Peel SE, Liu B and Hall IP: A key role for
STIM1 in store operated calcium channel activation in airway smooth
muscle. Respir Res. 7(119)2006.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Zheng Q, Yuan Y, Yi W, Lau WB, Wang Y,
Wang X, Sun Y, Lopez BL, Christopher TA, Peterson JM, et al:
Clq/TNF-related proteins, a family of novel adipokines, induce
vascular relaxation through the adiponectin receptor-1/AMPK/e
NOS/nitric oxide signaling pathway. Arterioscler Thromb Vasc Biol.
31:2616–2623. 2011.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Takeuchi S, Wada K, Uozumi Y, Otani N,
Osada H, Nagatani K and Mori K: Adiponectin receptor 1 expression
is associated with carotid plaque stability. Neurol India.
61:249–253. 2013.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Jing XD, Wei XM, Deng SB, Du JL, Liu YJ
and She Q: The relationship between the high-density lipoprotein
(HDL)-associated sphingosine-1-phosphate (S1P) and coronary
in-stent restenosis. Clin Chim Acta. 15:248–252. 2015.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Kambara T, Ohashi K, Shibata R, Ogura Y,
Maruyama S, Enomoto T, Uemura Y, Shimizu Y, Yuasa D, Matsuo K, et
al: CTRP9 protein protects against myocardial injury following
ischemia-reperfusion through AMP-activated protein kinase
(AMPK)-dependent mechanism. J Biol Chem. 287:18965–18973.
2012.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Elnaggar MA, Seo SH, Gobaa S, Lim KS, Bae
IH, Jeong MH, Han DK and Joung YK: Nitric oxide releasing coronary
stent: A new approach using layer-by-layer coating and liposomal
encapsulation. Small. 12:6012–6023. 2016.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Zhang H, Park Y, Wu J, Chen XP, Lee S,
Yang J, Dellsperger KC and Zhang C: Role of TNF-alpha in vascular
dysfunction. Clin Sci (Lond). 116:219–230. 2009.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Picchi A, Gao X, Belmadani S, Potter BJ,
Focardi M, Chilian WM and Zhang C: Tumor necrosis factor-alpha
induces endothelial dysfunction in Lepr (dp) mice. Circulation.
115:245–254. 2007.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Booth AJ, Csencsits-Smith K, Wood SC, Lu
G, Lipson KE and Bishop DK: Connective tissue growth factor
promotes fibrosis downstream of TGFbeta and IL-6 in chronic cardiac
allograft rejection. Am J Transplant. 10:220–230. 2010.PubMed/NCBI View Article : Google Scholar
|