Introduction
Diabetic mellitus (DM), which describes a series of
metabolic disorders affecting protein, fat and electrolytes, is
caused by the absolute or relative deficiency of insulin secretion
or decreased insulin sensitivity in target tissue cells, which is
mainly characterized by abnormally elevated blood sugar content
(1,2). The metabolic disorder caused by DM
directly affects cardiac myocytes and cardiac fibroblasts, changing
the function of cardiac myocytes and the deposition of collagen in
the cardiac interstitium, leading to decreases in myocardial
compliance and diastolic function (3). As the disease progresses, the systolic
function of the heart also decreases, causing congestive heart
failure, cardiogenic shock and sudden mortality, which seriously
affect the life and health of patients with DM (4). Myocardial fibrosis (MF) is an
important factor in cardiac diastolic, contractile function
impairment and congestive heart failure during the progression of
diabetic cardiomyopathy (DCM) (5-7).
Previous studies have shown that transforming growth
factor β1 (TGFβ1) regulates the transcription of related
extracellular matrix (ECM) components by activating downstream Smad
proteins and serves an important regulatory role in fibrosis in
various tissues and organs (8-10).
In the condition of diabetic pathological hyperglycaemia, the renin
angiotensin aldosterone system (RAAS) is activated, which increases
blood circulation and angiotensin II (Ang II) content in the
myocardium (11). Furthermore, Ang
II increases the expression of a variety of fibrotic factors,
including transforming growth factor β (TGF-β), connective tissue
growth factor (CTGF) and collagen in the myocardial interstitium
whilst reducing the degradation of collagen, thus promoting the
development of diabetic MF (12,13).
Irbesartan is an Ang II receptor antagonist that can effectively
block the binding of Ang II to its receptor and inhibit its
downstream biological functions (14). Currently, irbesartan is widely used
in the clinic, mainly for the treatment of hypertension and type II
diabetic nephropathy with hypertension (15). In addition, previous studies have
reported that irbesartan can improve MF induced by hypertension
(16-18);
however, to the best of our knowledge, there have been an
insufficient number of studies on the effects of irbesartan on MF
in patients with DM. Therefore, the aim of the present study was to
investigate whether irbesartan serves a role in improving MF in a
diabetic rat model.
Materials and methods
Ethical statement
The present study was approved by the Animal Ethics
Committee and was conducted in accordance to the relevant
agreements with the Beijing Chaoyang Hospital of the Capital
Medical University (approval no. 201705231). All procedures were
performed in accordance with the Guidance Suggestions for the Care
and Use of Laboratory Animals (19).
Main reagents and materials
DMEM, FBS and penicillin-streptomycin were purchased
from Gibco (Thermo Fisher Scientific, Inc.), and a hydroxyproline
assay kit (cat. no. A030-1-1) was purchased from Nanjing Jiancheng
Bioengineering Institute. Irbesartan was purchased from Sanofi
(Hangzhou) Pharmaceutical Co., Ltd. Healthy adult male Sprague
Dawley (SD) rats (n=30; age, 6 weeks; weight, 220-240 g) were
acquired from the Guangdong Medical Laboratory Animal Center. A
high-fat and high-sugar diet was purchased from Jiangsu Synergy
Pharmaceutical Bioengineering Co., Ltd. (https://www.jsxtsw.com/) and streptozotocin (STZ)
citrate buffer was purchased from Sigma-Aldrich (Merck KGaA). A rat
collagen I ELISA kit (cat. no. 20180302A) was purchased from Suzhou
Calvin Biotechnology Ltd. (https://biocalvin.biogo.net/) and rat cardiac
fibroblasts (RCFs) were purchased from Cloud-Clone Corp. (cat. no.
CSI095Ra01). The TGFβ1 inhibitor pirfenidone was purchased from
Selleck Chemicals. Rabbit anti-human TGFβ1 (cat. no. ab92486;
1:2,000), anti-phosphorylated (p)-Smad2/3 (cat. no. ab272332;
1:2,000), Smad2/3 (cat. no. ab63672, 1:2,000), and anti-collagen
type I α1 (COLIA1; cat. no. ab34710; 1:2,000) polyclonal antibodies
were purchased from Abcam. ECL chemiluminescence solutionand
bicinchoninic acid protein quantitative detection kits were
purchased from Beyotime Institute of Biotechnology. Rat type I
procollagen carboxyl-terminal peptide (PICP; cat. no. CSB-E08081r)
and Type III procollagen amino terminal pro-peptide (PIIINP) ELISA
kits (cat. no. CSB-E08096r) were purchased from CusabioTechnology
LLC. Chloral hydrate and PBS were obtained from Sangon Biotech Co.,
Ltd. Biological function was assessed with a BL-420L biological
signal collection system (Chengdu Taimeng Software Co., Ltd.), and
flow cytometry was performed using a Beckman Coulter FC500 MCL flow
cytometer (Beckman Coulter, Inc.).
Establishment of the DCM rat
model
Rats were allowed free access to water and food
under 22±2˚C, 55±10% humidity and 12-h light/dark cycle. A rat
model of type 2 DM was established by feeding freely with a
high-fat and high-sugar diet combined with an intraperitoneal
injection of a low dose of STZ (40 mg/kg). After 1 week of adaptive
feeding of SD rats, the model was established. DM model rats were
fed a high-fat and high-sugar diet for 2 months. After the rats
were fasted for 12 h, 1% STZ citrate buffer was injected into the
abdominal cavity (40 mg/kg). The model was considered successful
when the fasting blood glucose (FBG) concentration was >16.7
mmol/l for 3 consecutive days. Blood was obtained by using the
blood glucose meter blood sampling needle to pierce the tip of the
tail of the rat and then squeezed a drop of blood onto the blood
glucose test paper to test the blood glucose level. Food intake,
drinking water intake and urine output were significantly higher in
model rats compared with control rats, indicating that the DM model
was successfully established. The rats remained on the high-fat and
high-sugar diet for a further 10 weeks.
The eight rats in the control group were fed on a
standard diet, given intraperitoneal injections of citrate buffer
(10 ml/kg, no STZ) after 2 months of feeding and continued on the
standard feed until the end of the experiment.
Irbesartan treatment of DCM rats
In total, 30 rats were randomly divided into the
blank control group (n=8) and the DCM1 group (n=22). The remaining
rats in which the DCM model was successfully established were
divided into two groups: DCM group (n=6) and the DCM + irbesartan
group (n=6). Irbesartan was intragastrically administered at 50
mg/kg (20) once daily for 10
weeks. In the DCM and blank control groups, a corresponding volume
of saline (10 ml/kg) was administered by gavage. DCM rats were fed
the high-fat and high-sugar diet, whilst rats in the blank control
group were fed on the standard maintenance diet for 10 weeks.
Blood glucose and blood lipid
detection
Rats were anaesthetized with an intraperitoneal
injection of chloral hydrate (300 mg/kg), before 2 ml blood was
obtained from the orbital venous plexus and a sterile cotton ball
was used to stop the bleeding immediately after pressing the orbit,
following which the rats resumed normal diet and activities. FBG,
total cholesterol (TC), triglyceride (TG), high-density lipoprotein
cholesterol (HDL-C) and low-density lipoprotein cholesterol (LDL-C)
levels were measured with a 7180 Automatic Biochemical Analyzer
(Hitachi, Ltd.).
Heart weight index (HWI) and left
ventricular weight index (LVWI) determination
After the rats were weighed, they were anaesthetized
with an intraperitoneal injection of chloral hydrate (300 mg/kg)
and the hearts were removed. After the heart tissue was washed with
saline and filter paper was used to absorb excess water, each heart
was weighed. Subsequently, the left and right atrium and the right
ventricle were removed, and the left ventricle was weighed to
provide the LVW. HWI and LVWI were calculated using the following
equations: HWI=HW/body weight (BW; mg/g) and LVWI=LVW/BW (mg/g).
The heart tissue was then stored at -80˚C for further
experimentation.
Cardiac function evaluation
Cardiac haemodynamic indexes of each group were
measured by cannulation of the carotid artery, and cardiac function
was evaluated after intraperitoneal injection of 10% chloral
hydrate (300 mg/kg). The right cervical artery was isolated,
exposed and cannulated. The left ventricle was cannulated through
the right common carotid artery. The left ventricular systolic
pressure (LVSP) and left ventricular end-diastolic pressure (LVEDP)
were detected using the BL-420L biological signal collection
system.
Detection of myocardial
hydroxyproline
In total, 100 mg myocardial tissue and 400 µl
pre-cooled PBS were added to a manual glass homogenizer, which was
then evenly grinded it in an ice box and poured into a 1.5 ml tube.
This mixture was centrifuged at 4,000 x g 5 min at 4˚C, before the
supernatant was collected. The hydroxyproline content was detected
by ELISA according to manufacturer's protocol. The plates included
blank, standard and sample wells. A myocardial hydroxyproline
standard sample (10 µl) was first diluted in a 40 µl solution. The
plate was sealed and incubated at 37˚C for 30 min. The sealing film
was then carefully removed, the liquid was discarded and the plate
was dried and washed with PBS five times for 30 sec each.
Enzyme-labelling reagent (50 µl) was added to each well (except the
blank well), and the plate was sealed and incubated at 37˚C for 30
min. The sealing membrane was carefully removed from the plate, and
the wells were filled with washing liquid. This process was
repeated five times. Chromogenic agent A (50 µl) was added to each
well and then 50 µl colour agent B was added. Subsequently, the
plates were lightly shaken at 37˚C for 15 min in the dark.
Terminating solution (50 µl) was added to each well to terminate
the reaction. With the blank well set to zero, the absorbance of
each well was measured at 450 nm.
RCF cell culture and treatment
RCF cells were inoculated into culture dishes
(1x106/well), and DMEM supplemented with 10% FBS and 1%
penicillin-streptomycin was added. Cells were cultured in a
CO2 culture box with 5% CO2 at 37˚C. After
the cells were grown to 80-90% confluence, cells were then cultured
at a 1:5 ratio and tested within four generations.
According to the different treatment concentrations
of D-glucose (Dalian Meilun Biotechnology Co., Ltd.), the RCF cells
were divided into the normal-glucose concentration group (5 mM
D-glucose; control group) and the high-glucose (HG) treatment group
(25 mM D-glucose). RCF cells treated with HG were further divided
into three groups: HG group, the HG + irbesartan (10 µM) group and
the HG + irbesartan (10 µM) + pirfenidone (5 mM) group. Cells in
each treatment group were cultured (5% CO2, 37˚C) for 48
h in vitro and collected for mRNA and protein detection.
EdU detection of cell
proliferation
After RCF cells were incubated in complete culture
medium for 2 h at 37˚C with 10 µl EdU, the cells were cultured for
an additional 48 h in accordance with the aforementioned groups and
digested with 0.2% trypsin at 25˚C for 60 sec. After PBS washing
and centrifugation (500 x g, 25˚C, 5 min), cells were incubated in
10% neutral formaldehyde at room temperature for 15 min. PBS was
then added to the cells, which were then centrifuged at 300 x g for
5 min at 25˚C. Subsequently, 100 µl 0.5% Triton X-100 in PBS was
added to the cells at room temperature and 500 µl reaction liquid
was added for detection. Cell proliferation was detected after
incubation at room temperature for 30 min, followed by the addition
of 3 ml washing liquid and centrifugation of the washing liquid.
The cell pellets were then resuspended in 500 µl washing liquid and
flow cytometry was performed using a Beckman Coulter FC 500 MCL
flow cytometer, which was analyzed using the FlowJo software
(FlowJo v7.6.5; FlowJo LLC).
ELISA detection of type I collagen in
rat tissues and PICP and PIIINP in RCF cell culture medium
PBS was added to the myocardial tissue at 1:5
(myocardial tissue:PBS), which was then homogenized and centrifuged
at 1,000 x g 10 min at 4˚C before the supernatant was obtained.
Type I collagen content was then measured in the supernatant
according to the manufacturer's instructions.
RCF cells from each treatment group were cultured
for 48 h. The supernatant of the culture medium was collected and
the levels of PICP and PIIINP in the culture medium were detected
by ELISA, according to the manufacturer's instructions.
Western blotting of protein
expression
After SDS lysis buffer (cat. no. P0013G; Beyotime
Institute of Biotechnology) was added to the cells or myocardial
homogenate, each sample was boiled for 5 min and the bicinchoninic
acid method was used to detect the protein concentration. In total,
60 µg sample was then separated by 5% SDS-PAGE and transferred to a
PVDF membrane. The membrane was blocked with 5% skimmed milk powder
for 1 h at room temperature and then incubated with primary
antibodies TGFβ1 (1:2,000), p-Smad2/3 (1:2,000), Smad2/3 (1:2,000),
COLIA1 (1:2,000), β-actin (1:500; cat. no. ab179467; Abcam)
overnight at 4˚C. After the membrane was washed with PBS + 10%
Tween-20 for three times, the membranes were incubated with an
appropriate horseradish peroxidase-labelled goat anti-rabbit IgG
secondary antibody (1:2,500; cat. no. G-21234; Thermo Fisher
Scientific, Inc.) for 1 h at room temperature. The membranes were
subsequently detected with ECLand analyzed using the Quantity One
image analysis software (version 4.5.0; Bio-Rad Laboratories,
Inc.).
Statistical analysis
Statistical analysis was performed using SPSS 18.0
software (SPSS, Inc.). Data are presented as the mean ± standard
deviation from three experimental repeats. A t-test was used to
compare data between two groups. Differences among >2
experimental groups were evaluated by one-way ANOVA followed by
Tukey's test. P<0.05 considered to indicate a statistically
significant difference.
Results
Abnormal blood glucose and blood lipid
content in DCM model rats
The blood glucose test results indicated that the
FBG content in the DCM group was significantly higher compared with
the control group (P<0.001). Moreover, the levels of TC, TG and
LDL-C in the DCM group were significantly higher compared with the
control group (P<0.05), while the levels of HDL-C were
significantly lower (P<0.05), as presented in Table I.
 | Table IBlood glucose and blood lipid levels
in two groups of rats. |
Table I
Blood glucose and blood lipid levels
in two groups of rats.
| Index | FBG (mmol/l) | TC (mmol/l) | TG (mmol/l) | HDL-C (mmol/l) | LDL-C (mmol/l) |
|---|
| Control group
(n=8) | 4.69±0.58 | 1.23±0.19 | 1.33±0.21 | 1.03±0.11 | 0.88±0.09 |
| DCM1 group
(n=6) | 23.61±3.89 | 2.71±0.39 | 2.59±0.42 | 0.84±0.08 | 1.06±0.15 |
| t | 11.816 | 9.431 | 7.407 | 3.568 | 2.807 |
| P-value | <0.001 | <0.001 | <0.001 | 0.002 | 0.008 |
DCM model rats exhibit notable MF
The cardiac test results demonstrated that HWI and
LVWI in the DCM group were significantly higher compared with the
control group (P<0.001; Table
II). ELISA results suggested that the myocardial concentrations
of type I collagen and hydroxyproline in the DCM group were
significantly increased compared with that in the control group
(P<0.001; Table II). The LVSP
were significantly decreased (P<0.05; Table II) and LVEDP were significantly
increased (P<0.001; Table II)
in the DCM group compared with those in the control group
(P<0.001; Table II).
 | Table IIComparison of HWI, LVWI and
myocardial fibrosis indexes in two groups of rats. |
Table II
Comparison of HWI, LVWI and
myocardial fibrosis indexes in two groups of rats.
| Index | HWI (mg/g) | LVWI (mg/g) | Collagen I
(ng/g) | Hydroxyproline
(µg/mg) | LVSP (mmHg) | LVEDP (mmHg) |
|---|
| Control group
(n=8) | 2.81±0.33 | 2.09±0.22 | 115.63±13.77 | 33.66±6.29 | 127.9±13.8 | 3.1±0.4 |
| DCM1 group
(n=6) | 4.05±0.38 | 3.14±0.28 | 238.51±34.65 | 76.58±11.34 | 97.4±10.9 | 7.9±0.9 |
| t | 6.528 | 7.878 | 8.214 | 9.077 | 4.2 | 11.9 |
| P-value | <0.001 | <0.001 | <0.001 | <0.001 | 0.002 | <0.001 |
Irbesartan inhibits the TGFβ1/Smad2/3
pathway and reduces MF in DCM rats
The blood glucose test indicated that there was no
significant difference in FBG content between the DCM group and the
irbesartan group. Cardiac measurements identified that, compared
with the DCM group, the DCM + irbesartan group had a significantly
lower HWI and LVWI (P<0.001). Furthermore, compared with the DCM
group, the DCM + irbesartan group had significantly reduced levels
of collagen I and hydroxyproline in myocardial tissue (P<0.001),
as presented in Table III.
 | Table IIIComparison of HWI, LVWI and
myocardial fibrosis indexes in two groups of rats. |
Table III
Comparison of HWI, LVWI and
myocardial fibrosis indexes in two groups of rats.
| Index | FBG (mmol/l) | HWI (mg/g) | LVWI (mg/g) | Collagen I
(ng/g) | Hydroxyproline
(µg/mg) |
|---|
| DCM group
(n=6) | 22.85±3.77 | 4.04±0.36 | 3.16±0.26 | 229.67±31.92 | 79.23±13.31 |
| DCM + Irbesartan
group (n=6) | 21.17±4.12 | 3.18±0.31 | 2.75±0.25 | 158.72±29.63 | 52.86±10.77 |
| t | 0.737 | 4.434 | 2.784 | 3.990 | 3.773 |
| P-value | 0.478 | 0.001 | 0.019 | 0.003 | 0.004 |
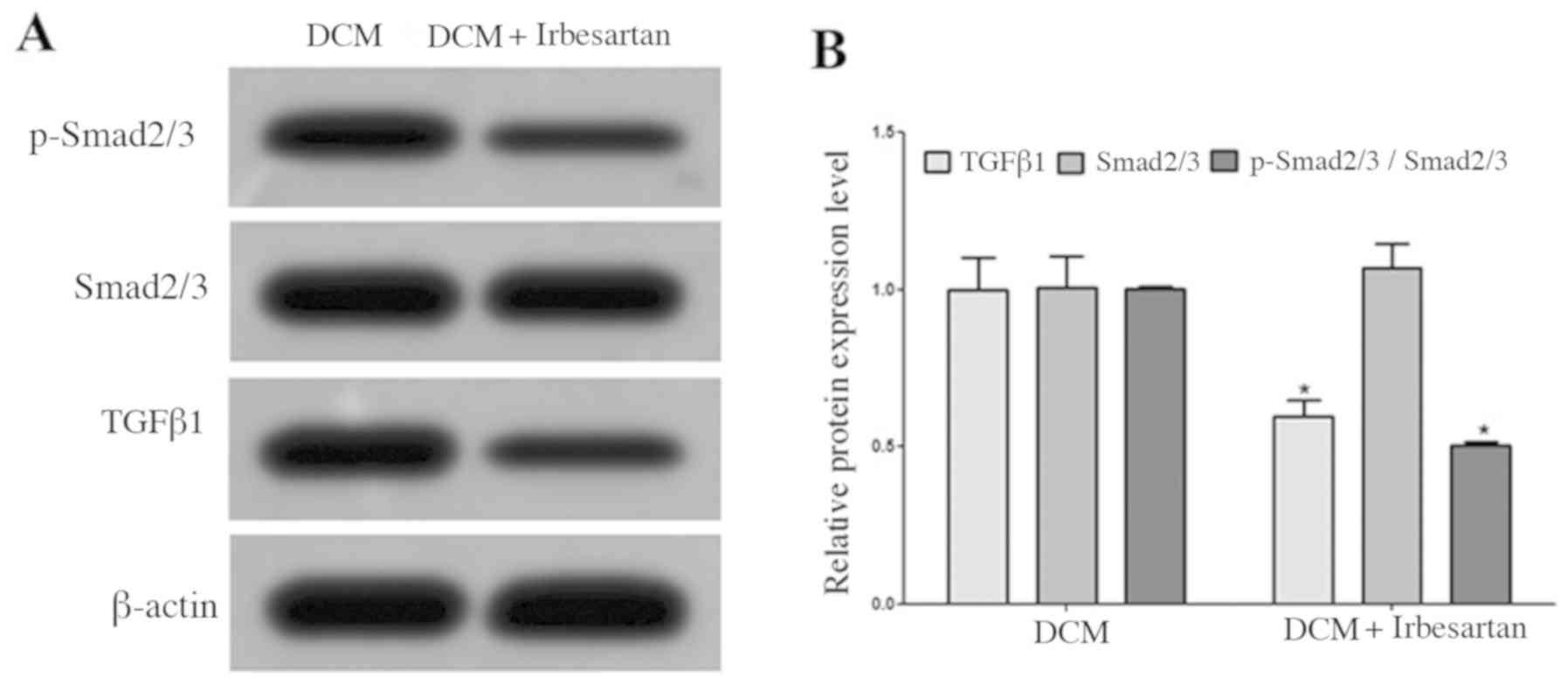
Western blotting results demonstrated that the
myocardial expression of TGFβ1 protein in the DCM + irbesartan
group was significantly lower compared with the DCM group, and the
protein expression of p-Smad2/3 was also significantly decreased
(Fig. 1).
Irbesartan alleviates DM-induced
cardiac dysfunction
Compared with the DCM group, the DCM + irbesartan
group had a significantly higher LVSP (P=0.016) but significantly
lower LVEDP (P<0.001; Table
IV). Thus, these data indicated that irbesartan improved
myocardial contractile function and compliance in DM rats.
 | Table IVComparison of LVSP and LVEDP test
results in two groups of rats. |
Table IV
Comparison of LVSP and LVEDP test
results in two groups of rats.
| Index | LVSP (mmHg) | LVEDP (mmHg) |
|---|
| DCM group
(n=6) | 96.7±11.3 | 9.4±1.3 |
| DCM + Irbesartan
group (n=6) | 116.9±12.8 | 5.6±0.8 |
| t | 2.898 | 6.098 |
| P-value | 0.016 | <0.001 |
Irbesartan inhibits the TGFβ1/Smad2/3
pathway and HG-induced fibrosis in RCF cells
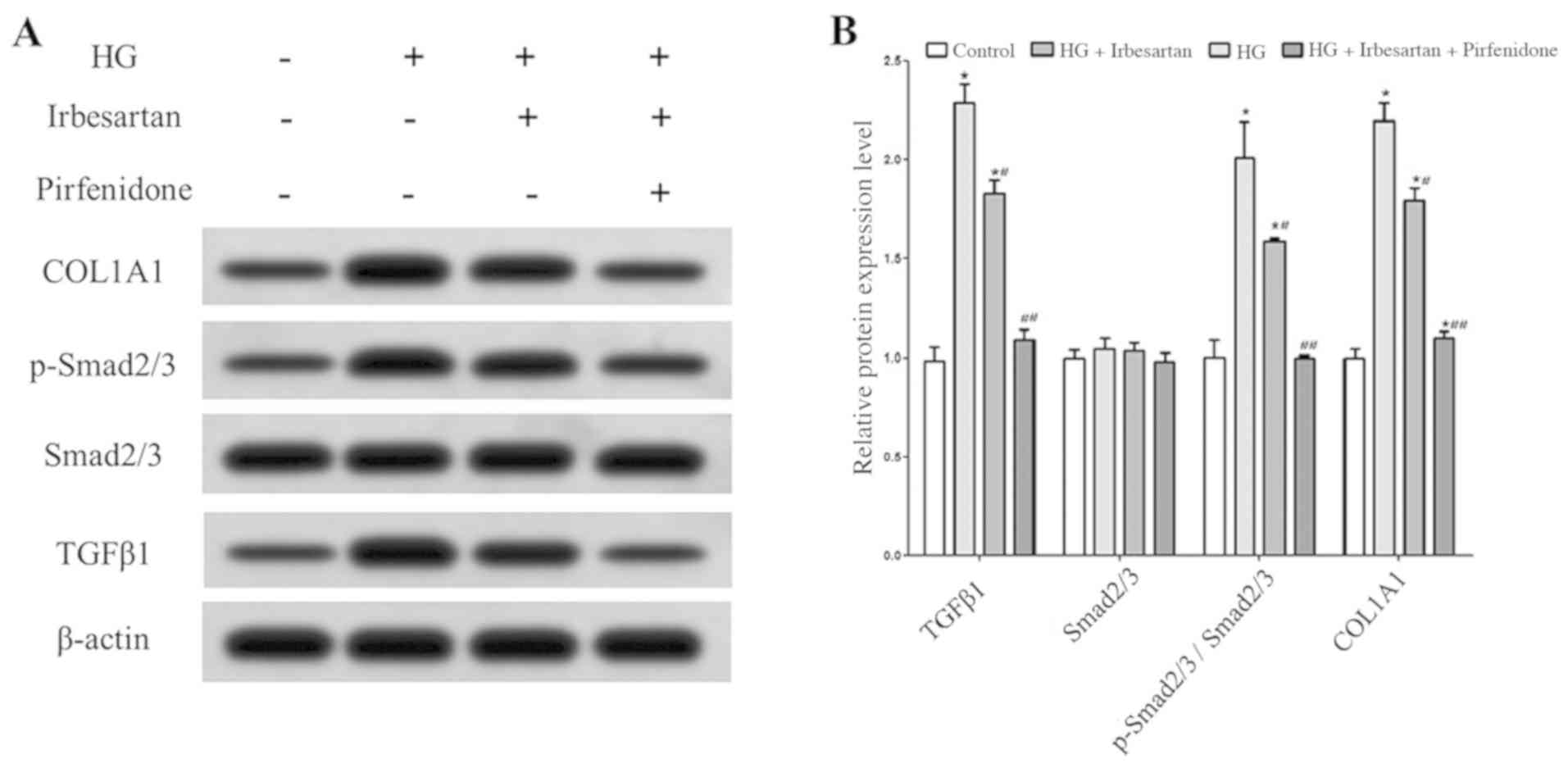
Western blot analysis results demonstrated that the
expression levels of TGFβ1 and p-Smad2/3 in RCF cells were
significantly higher in the HG treatment group compared with the
control group. In addition, HG treatment significantly increased
the expression of COL1A1 in RCF cells, indicating that the ability
of RCF cells to synthesize collagen was enhanced and that fibrosis
was significantly changed.
After treatment of HG-exposed RCF cells with
irbesartan, TGFβ1 expression was significantly decreased,
TGFβ1/Smad2/3 pathway activity was significantly inhibited, COL1A1
expression was reduced. When combined with irbesartan, treatment
with the TGFβ1 inhibitor pirfenidone reduced TGFβ1/Smad2/3 pathway
activity, COL1A1 expression further in RCF cells under HG treatment
conditions (Fig. 2).
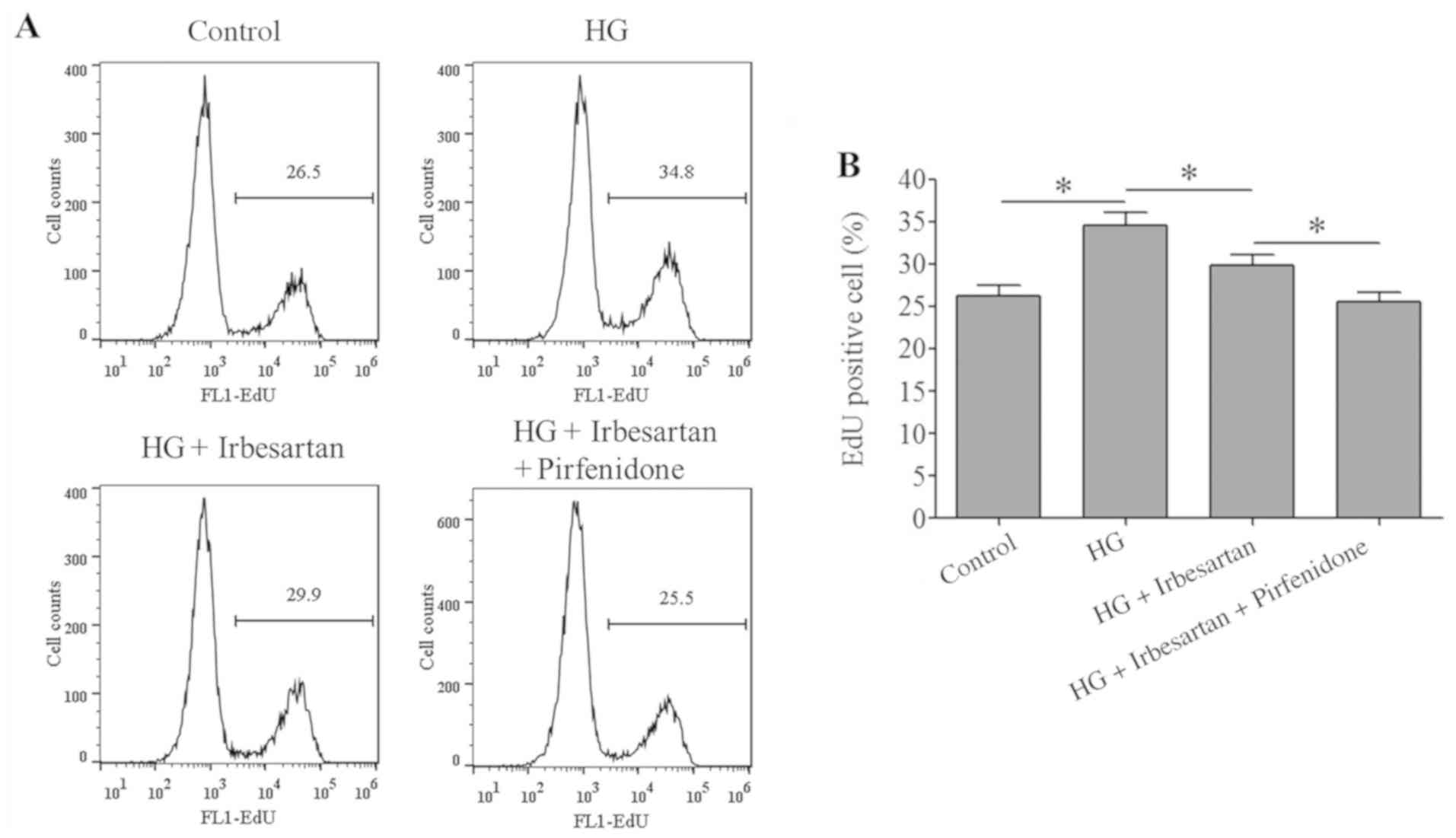
Irbesartan inhibits RCF cell
proliferation and collagen synthesis
The proliferative ability of RCF cells in the HG
treatment group was significantly increased compared with the
control group. However, irbesartan treatment reduced the RCF
proliferative ability, which was further decreased by combined
treatment with irbesartan + pirfenidone (Fig. 3).
The ELISA results demonstrated that PICP and PIIINP
contents in HG RCF cell culture conditions were significantly
higher compared with the control culture conditions. It was found
that irbesartan treatment significantly reduced the PICP and PIIINP
content in the culture solution, while combined treatment with
irbesartan + pirfenidone resulted in the lowest PICP and PIIINP
content in the culture solution (Table
V).
 | Table VPICP and PIIINP levels in RCF cell
culture medium from each treatment group. |
Table V
PICP and PIIINP levels in RCF cell
culture medium from each treatment group.
| Group | Control | HG | HG +
Irbesartan | HG + Irbesartan +
Pirfenidone |
|---|
| PICP (pg/ml) | 615.8±31.9 |
1186.6±43.8a |
926.5±36.9a |
811.8±35.1a |
| PIIINP (pg/ml) | 236.5±18.7 |
458.4±37.5a |
364.3±26.5a |
306.7±24.7a |
Discussion
Previous studies have reported that TGFβ1 plays an
important role in regulating fibrosis in various tissues and
organs, such as cardiac (21),
liver (22), lung (23) and kidney (24) tissue, by activating downstream Smad
proteins. TGFβ1 must bind with specific cell-surface receptors to
exert its corresponding biological effects. In mammalian cells,
three types of TGFβ1 receptors (TβRs) are expressed: TβR-I, TβR-II
and TβR-III (25). Moreover,
TβR-III lacks intrinsic activity and has no signal transduction
function itself. The active forms of TβR-II and TβR-III respond to
stimuli in the body and function in signal transduction (26). Smads have signal transduction
functions and act in the cytoplasm with TβR-Ⅰ kinase, which plays a
key role in the regulation of the TGFβ1/Smad pathway; within this
pathway, Smad2 and Smad3 mediate TGFβ1 protein signal transduction
(27,28). Under the stimulation of various
factors, TGFβ1 combines with TβR-II and TβR-II binds to TβR-I in
response to phosphorylation (25,26).
Phosphorylation-activated TβR-I then combines with Smad2/3.
Moreover, phosphorylation-activated Smad2/3 combines with Smad4 to
form a protein polymer, which is transferred from the cytoplasm to
the nucleus and regulates the transcription and expression of a
variety of downstream target genes, including Col-I, Col-III and
α-SMA (29-31).
In a positive feedback mechanism, the protein complex acts on the
promoter region of the TGFβ1 gene to promote its expression and
TGFβ1 autocrine signalling. In addition, TGFβ1 plays a positive
feedback role by promoting the expression of the TβR-I and TβR-II
receptor genes and amplifying the TGFβ1/Smad2/3 signalling pathway
(29,30).
TGFβ1 is the regulatory factor most closely related
to MF (21). Increased expression
and activation of TGFβ1 occurs within a common pathway and acts as
an intermediate bridge for MF caused by several factors. Thus,
TGFβ1 is considered to be a direct MF factor (32). Previous studies have revealed that
Ang II signalling can lead to collagen deposition via a variety of
pathways, including the 5'adenosine monophosphate-activated protein
kinase-angiotensin converting enzyme 2 and CTGF-fractalkine
pathways, leading to fibrosis (33,34).
Irbesartan, an Ang II receptor antagonist, affects the biological
function of Ang II by blocking the binding of Ang II to its
receptor (14). In a previous study
examining ventricular fibrosis induced by hypertension, irbesartan
regulated the degradation and synthesis of ECM by downregulating
the expression of matrix metalloproteinase-2 and TIMP
metallopeptidase inhibitor 2 to reduce the degree of ventricular MF
(35). Irbesartan is widely used in
the treatment of hypertension and type II diabetic nephropathy with
hypertension and has been shown to alleviate MF caused by renal
vascular hypertension (15).
However, to the best of our knowledge, there have been few reports
in China or elsewhere on whether irbesartan can reverse DM-induced
MF. Therefore, the aim of the present study was to investigate
whether irbesartan can improve MF in DM model rats.
Lipid metabolism dysfunction is an important factor
in type 2 DM pathogenesis (36).
Therefore, the present study examined the serum TC level in
STZ-induced DM model to determine whether the model was
successfully established. Compared with the control group, the DCM
group had significantly higher blood glucose levels and abnormal
blood lipid levels, indicating the successful establishment of the
DM rat model. Furthermore, the HWI and LVWI of DCM rats were
significantly increased. The myocardial content of type I collagen
and hydroxyproline in DCM rats was significantly higher compared
with control rats, suggesting that the myocardial tissue of DCM
rats had obvious fibrotic changes. In line with other studies, such
as that by Shen et al (37),
significantly increased levels of the type I collagen COL1A1 have
been shown in cardiac muscle tissue in DM rats compared with
control rats.
In the present study, irbesartan-treated DCM rats
exhibited significantly milder fibrosis and lower TGFβ1 and
p-Smad2/3 expression in the myocardium compared with DCM rats,
suggesting that irbesartan may reduce TGFβ1/Smad2/3 pathway
activation to alleviate MF in DM rats by reducing the expression of
TGFβ1. Lv et al (38)
investigated irbesartan and visceral fibrosis in diabetic
nephropathy model animals, and found that irbesartan significantly
reduced the expression of TGFβ1 in the glomerular tissue of
diabetic nephropathy rats. Moreover, Tuncdemir et al
(39) revealed that irbesartan
treatment significantly reduced TGFβ1 expression in the glomeruli
of diabetic nephropathy rats. Furthermore, irbesartan has been
shown to significantly inhibit remodelling and fibrosis of the
myocardium in hypertensive rats by downregulating the expression of
TGFβ1(40). In a study by Kataoka
et al (41), irbesartan was
revealed to serve a role in downregulating the expression of TGFβ1
and inhibiting MF in atrial tachycardia models. In addition, Tanaka
et al (42) demonstrated
that irbesartan significantly inhibited bleomycin-induced pulmonary
fibrosis and reduced the expression of TGFβ1 in lung tissue. It has
also been shown that the degree of renal tubulointerstitial
fibrosis is significantly lower in irbesartan-treated animals
compared with untreated animals, and that the expression of TGFβ1
and the phosphorylation of Smad2 are also significantly inhibited
in irbesartan-treated animals (43). Using a rat model of hepatic fibrosis
induced by bile duct ligation, Kim et al (44) reported that irbesartan significantly
inhibited the expression of TGFβ1 in the liver and reduced the
severity of liver fibrosis. Thus, these previous findings suggest
that irbesartan can inhibit fibrosis of the viscera by regulating
the expression of TGFβ1 and downregulating the activity of the
TGFβ1/Smad2/3 pathway, which is consistent with the present
results.
Through further in vitro experiments, the
present study also demonstrated that irbesartan decreased TGFβ1
expression in RCF cells, inhibiting Smad2/3 activity. Enhancement
and overactivation of cardiac fibroblast proliferation play
important roles in the development of MF (45). Cardiac fibroblast activation mainly
manifests as increased amounts of ECM around cardiac fibroblasts
(46). Moreover, ratio of type I
collagen to type III collagen synthesis is an important metric for
cardiac fibroblast activation (47). Therefore, detection of the amounts
of the PICP before the formation of type I collagen and PIIINP
before the formation of type III collagen has become a common
method that reflects the content of type I and type III collagen
proteins (48). The present study
found that irbesartan significantly inhibited the proliferation of
RCF cells under HG conditions and reduced the content of PICP and
PIIINP. Combined treatment with pirfenidone further enhanced the
inhibitory effect of irbesartan on RCF cell proliferation, collagen
synthesis and fibroblast activation. As there is no RAAS in
cultured cells in vitro, irbesartan may regulate the
expression of TGFβ1 directly or indirectly via other mechanisms and
affect the activity of the TGFβ1/Smad2/3 pathway, thus inhibiting
cardiac myocyte fibrosis. Similarly, in vitro studies by Lv
et al (38) have revealed
that irbesartan significantly downregulated the expression of
TGFβ1, collagen IV and fibronectin in HG-treated glomerular
mesangial cells and inhibited fibrosis of HG-treated mesangial
cells, indicating that irbesartan can inhibit fibrosis by directly
downregulating TGFβ1 without relying on the RAAS system. Thus, it
was indicated that lowering the expression of TGFβ1 and the
activity of the TGFβ1/Smad2/3 pathway may be one of the mechanisms
via which irbesartan reduces MF in DM rats. However, irbesartan may
also regulate MF in DM rats via other mechanisms; for instance, Liu
et al (49) have shown that
irbesartan alleviated MF in DM rats by regulating protein kinase D
and the endoplasmic reticulum stress system. As this study was
limited in its examination of other possible mechanisms, future
studies will investigate the other possible mechanisms, including
those of oxidative stressor immunomodulation, via which irbesartan
affects diabetic MF.
In conclusion, the present results suggested that
irbesartan may downregulate TGFβ1 expression, inhibit TGFβ1/Smad2/3
pathway activity and promote collagen synthesis to reduce MF
lesions and improve myocardial function.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
MZ and HZ designed the experiments. QL, YL and JZ
performed the experiments. YL and JZ collected and analyzed the
data. MZ and HZ drafted the manuscript. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Animal Ethics
Committee and was conducted in accordance to the relevant
agreements with the Beijing Chaoyang Hospital of the Capital
Medical University (approval no. 201705231). All procedures were
performed in accordance with the Guidance Suggestions for the Care
and Use of Laboratory Animals, formulated by the Ministry of
Science and Technology of China.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Chen J, Wang J, Zhang X and Zhu H: Inverse
relationship between serum bilirubin levels and diabetic foot in
Chinese patients with type 2 diabetes mellitus. Med Sci Monit.
23:5916–5923. 2017.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Xu T, Weng Z, Pei C, Yu S, Chen Y, Guo W,
Wang X, Luo P and Sun J: The relationship between
neutrophil-to-lymphocyte ratio and diabetic peripheral neuropathy
in type 2 diabetes mellitus. Medicine (Baltimore).
96(e8289)2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Russo I and Frangogiannis NG:
Diabetes-associated cardiac fibrosis: Cellular effectors, molecular
mechanisms and therapeutic opportunities. J Mol Cell Cardiol.
90:84–93. 2016.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Dillmann WH: Diabetic cardiomyopathy.
Circu Res. 124:1160–1162. 2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Zhang X, Pan L, Yang K, Fu Y, Liu Y, Chi
J, Zhang X, Hong S, Ma X and Yin X: H3 relaxin protects against
myocardial injury in experimental diabetic cardiomyopathy by
inhibiting myocardial apoptosis, fibrosis and inflammation. Cell
Physiol Biochem. 43:1311–1324. 2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Zou C, Liu X, Xie R, Bao Y, Jin Q, Jia X,
Li L and Liu R: Deferiprone attenuates inflammation and myocardial
fibrosis in diabetic cardiomyopathy rats. Biochem Biophys Res
Commun. 486:930–936. 2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Wang L, Li J and Li D: Losartan reduces
myocardial interstitial fibrosis in diabetic cardiomyopathy rats by
inhibiting JAK/STAT signaling pathway. Int J Clin Exp Pathol.
8:466–473. 2015.PubMed/NCBI
|
|
8
|
He-He H, Chen DQ, Wang YN, Feng YL, Cao G,
Vaziri ND and Zhao YY: New insights into TGF-β/smad signaling in
tissue fibrosis. Chem Biol Interact. 2018:76–83. 2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Meng XM, Nikolic-Paterson DJ and Lan HY:
TGF-β: The master regulator of fibrosis. Nat Rev Nephrol.
12:325–338. 2016.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Xu F, Liu C, Zhou D and Zhang L:
TGF-β/SMAD pathway and its regulation in hepatic fibrosis. J
Histochem Cytochem. 64:157–167. 2016.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Estato V, Obadia N, Carvalho-Tavares J,
Freitas FS, Reis P, Neto HC, Lessa MA and Tibiriçá E: Blockade of
the renin-angiotensin system improves cerebral microcirculatory
perfusion in diabetic hypertensive rats. Microvasc Res. 87:41–49.
2013.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Wong TC, Piehler KM, Kang IA, Kadakkal A,
Kellman P, Schwartzman DS, Mulukutla SR, Simon MA, Shroff SG,
Kuller LH and Schelbert EB: Myocardial extracellular volume
fraction quantified by cardiovascular magnetic resonance is
increased in diabetes and associated with mortality and incident
heart failure admission. Eur Heart J. 35:657–664. 2014.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Patel BM and Mehta AA: Aldosterone and
angiotensin: Role in diabetes and cardiovascular diseases. Eur J
Pharmacol. 697:1–12. 2012.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Parving HH, Lehnert H, Bröchner-Mortensen
J, Gomis R, Andersen S and Arner P: Irbesartan in Patients with
Type 2 Diabetes and Microalbuminuria Study Group. The effect of
irbesartan on the development of diabetic nephropathy in patients
with type 2 diabetes. N Engl J Med. 345:870–878. 2001.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Croom KF, Curran MP, Goa KL and Perry CM:
Irbesartan: A review of its use in hypertension and in the
management of diabetic nephropathy. Drugs. 64:999–1028.
2004.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Nako H, Kataoka K, Koibuchi N, Dong YF,
Toyama K, Yamamoto E, Yasuda O, Ichijo H, Ogawa H and Kim-Mitsuyama
S: Novel mechanism of angiotensin II-induced cardiac injury in
hypertensive rats: The critical role of ASK1 and VEGF. Hypertens
Res. 35:194–200. 2012.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Castoldi G, Di Gioia CR, Bombardi C,
Catalucci D, Corradi B, Gualazzi MG, Leopizzi M, Mancini M, Zerbini
G, Condorelli G and Stella A: MiR-133a regulates collagen 1A1:
Potential role of miR-133a in myocardial fibrosis in angiotensin
II-dependent hypertension. J Cell Physiol. 227:850–856.
2012.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Whaley-Connell A, Habibi J, Cooper SA,
Demarco VG, Hayden MR, Stump CS, Link D, Ferrario CM and Sowers JR:
Effect of renin inhibition and AT1R blockade on myocardial
remodeling in the transgenic ren2 rat. Am J Physiol Endocrinol
Metab. 295:E103–E109. 2008.PubMed/NCBI View Article : Google Scholar
|
|
19
|
National Research Council (US) Committee
for the Update of the Guide for the Care and Use of Laboratory
Animals: Guide for the Care and Use of Laboratory Animals. 8th
edition. National Academies Press, Washington, DC, 2011. Available
from: https://www.ncbi.nlm.nih.gov/books/NBK54050/
doi: 10.17226/12910.
|
|
20
|
Dai H, Zheng M, Tang Rn, Ma Kl, Ni J and
Liu BC: Inhibition of integrin-linked kinase by angiotensin II
receptor antagonist, irbesartan attenuates podocyte injury in
diabetic rats. Chin Med J (Engl). 125:888–893. 2012.PubMed/NCBI
|
|
21
|
Guo Y, Gupte M, Umbarkar P, Singh AP, Sui
JY, Force T and Lal H: Entanglement of GSK-3β, β-catenin and TGF-β1
signaling network to regulate myocardial fibrosis. J Mol Cell
Cardiol. 110:109–120. 2017.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Yang D, Li L, Qian S and Liu L: Evodiamine
ameliorates liver fibrosis in rats via TGF-beta1/smad signaling
pathway. J Natl Med. 72:145–154. 2017.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Yang T, Wang J, Pang Y, Dang X, Ren H, Liu
Y, Chen M and Shang D: Emodin suppresses silica-induced lung
fibrosis by promoting sirt1 signaling via direct contact. Mol Med
Rep. 14:4643–4649. 2016.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Li H, Cai H, Deng J, Tu X, Sun Y, Huang Z,
Ding Z, Dong L, Chen J, Zang Y and Zhang J: TGF-beta-mediated
upregulation of sox9 in fibroblast promotes renal fibrosis. Biochim
Biophys Acta Mol Basis Dis. 1864:520–532. 2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Kajdaniuk D, Marek B, Borgiel-Marek H and
Kos-Kudła B: Transforming growth factor β1 (TGFβ1) in physiology
and pathology. Endokrynol Pol. 64:384–396. 2013.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Chen G, Grotendorst G, Eichholtz T and
Khalil N: GM-CSF increases airway smooth muscle cell connective
tissue expression by inducing TGF-β receptors. Am J Physiol Lung
Cell Mol Physiol. 284:L548–L556. 2003.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Loboda A, Sobczak M, Jozkowicz A and Dulak
J: TGF-beta1/smads and miR-21 in renal fibrosis and inflammation.
Med Inflamm. 2016(8319283)2016.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Li Y, Yang Y, Yu D and Liang Q: The effect
of tanshinone IIA upon the TGF-beta1/smads signaling pathway in
hypertrophic myocardium of hypertensive rats. J
HuazhongUnivSciTechnolog Med Sci. 29:476–480. 2009.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Yuan X, Gong Z, Wang B, Guo X, Yang L, Li
D and Zhang Y: Astragaloside inhibits hepatic fibrosis by
modulation of TGF-beta1/smad signaling pathway. evidence-based
complementary and alternative medicine. Evid Based Complement
Alternat Med. 2018(3231647)2018.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Wang S, Zhao X, Yan S, Chen B and Shi J:
Knockdown of NLRC5 inhibits renal fibroblast activation via
modulating TGF-beta1/smad signaling pathway. Eur J Pharmacol.
829:38–43. 2018.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Yu B, Li W, Al F and Chen Z: MicroRNA-33a
deficiency inhibits proliferation and fibrosis through inactivation
of TGF-beta/smad pathway in human cardiac fibroblasts. Pharmazie.
72:456–460. 2017.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Zhang Y, Shao L, Ma A, Guan G, Wang J,
Wang Y and Tian G: Telmisartan delays myocardial fibrosis in rats
with hypertensive left ventricular hypertrophy by TGF-beta1/smad
signal pathway. Hypertens Res. 37:43–49. 2014.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Zhang ZZ, Cheng YW, Jin HY, Chang Q, Shang
QH, Xu YL, Chen LX, Xu R, Song B and Zhong JC: The sirtuin 6
prevents angiotensin II-mediated myocardial fibrosis and injury by
targeting AMPK-ACE2 signaling. Oncotarget. 8:72302–72314.
2017.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Roth L, Schrijvers DM, Martinet W and De
Meyer GR: Angiotensin II increases coronary fibrosis, cardiac
hypertrophy and the incidence of myocardial infarctions in
ApoE-/- Fbn1C1039G+/- mice. Acta Cardiol.
71:483–488. 2016.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Chen Q, Pang L, Huang S, Lei W and Huang
D: Effects of emodin and irbesartan on ventricular fibrosis in
goldblatt hypertensive rats. Pharmazie. 69:374–378. 2014.PubMed/NCBI
|
|
36
|
Ritchie RH, Zerenturk EJ, Prakoso D and
Calkin AC: Lipid metabolism and its implications for type 1
diabetes-associated cardiomyopathy. J Mol Endocrinol. 58:R225–R240.
2017.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Shen N, Li X, Zhou T, Bilal MU, Du N, Hu
Y, Qin W, Xie Y, Wang H, Wu J, et al: Shensong Yangxin capsule
prevents diabetic myocardial fibrosis by inhibiting TGF-beta1/smad
signaling. J Ethnopharmacol. 157:161–170. 2014.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Lv J, Wang Z and Wang Y, Sun W, Zhou J,
Wang M, Liu WJ and Wang Y: Renoprotective effect of the
shen-yan-fang-shuai formula by inhibiting TNF-alpha/NF-kappaB
signaling pathway in diabetic rats. J Diabetes Res.
2017(4319057)2017.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Tunçdemir M and Oztürk M: The effects of
angiotensin-II receptor blockers on podocyte damage and glomerular
apoptosis in a rat model of experimental streptozotocin-induced
diabetic nephropathy. Acta Histochem. 113:826–832. 2011.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Chen C, Liang Z, Chen Q and Li ZG:
Irbesartan and emodin on myocardial remodeling in Goldblatt
hypertensive rats. J Cardiovasc Pharm. 60:375–380. 2012.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Kataoka N, Nishida K, Kinoshita K,
Sakamoto T, Nakatani Y, Tsujino Y, Mizumaki K, Inoue H and Kinugawa
K: Effect of irbesartan on development of atrial fibrosis and
atrial fibrillation in a canine atrial tachycardia model with left
ventricular dysfunction, association with p53. Heart Vessels.
31:2053–2060. 2016.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Tanaka J, Tajima S, Asakawa K, Sakagami T,
Moriyama H, Takada T, Suzuki E and Narita I: Preventive effect of
irbesartan on bleomycin-induced lung injury in mice. Respir
Investig. 51:76–83. 2013.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Zhao G, Zhao H, Tu L, Xu X, Zheng C, Jiang
M, Wang P and Wang D: Effects and mechanism of irbesartan on
tubulointerstitial fibrosis in 5/6 nephrectomized rats. J Huazhong
Univ Sci Technol Med Sci. 30:48–54. 2010.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Kim MY, Baik SK, Park DH, Jang YO, Suk KT,
Yea CJ, Lee IY, Kim JW, Kim HS, Kwon SO, et al: Angiotensin
receptor blockers are superior to angiotensin-converting enzyme
inhibitors in the suppression of hepatic fibrosis in a bile
duct-ligated rat model. J Gastroenterol. 43:889–896.
2008.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Pellman J, Zhang J and Sheikh F:
Myocyte-Fibroblast communication in cardiac fibrosis and
arrhythmias: Mechanisms and model systems. J Mol Cell Cardiol.
94:22–31. 2016.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Ivey MJ and Tallquist MD: Defining the
cardiac fibroblast. Circ J. 80:2269–2276. 2016.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Jin-Cheng L, Zhou L, Wang F, Cheng ZQ and
Rong C: Osthole decreases collagen Ⅰ/III contents and their ratio
in TGF-β1-overexpressed mouse cardiac fibroblasts through
regulating the TGF-β/smad signaling pathway. Chin J Nat Med.
16:321–329. 2018.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Eriksen HA, Satta J, Risteli J, Veijola M,
Väre P and Soini Y: Type I and type III collagen synthesis and
composition in the valve matrix in aortic valve stenosis.
Atherosclerosis. 189:91–98. 2006.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Liu X, Xu Q, Wang X, Zhao Z, Zhang L,
Zhong L, Li L, Kang W, Zhang Y and Ge Z: Irbesartan ameliorates
diabetic cardiomyopathy by regulating protein kinase D and ER
stress activation in a type 2 diabetes rat model. Pharmacol Res.
93:43–51. 2015.PubMed/NCBI View Article : Google Scholar
|