Introduction
Collagen type III glomerulopathy (CG) is an
idiopathic glomerular disease, characterized by massive
accumulation of collagen type III within the glomerular mesangial
regions and basement membrane. This rare renal disease has been
previously reported in Japan and occasionally in China (1). CG may be classified into two subtypes,
designated as collagenofibrotic glomerulopathy and nail patella
syndrome (NPS), according to the location of collagen deposition.
In CG, deposits containing type III collagen fibrils are mainly
located in the subendothelium and mesangium, while those in NPS are
located at the basement membrane. Furthermore, the clinical
symptoms of CG are confined to the kidney, while patients with NPS
usually have a range of extra-renal manifestations (2). CG is a rare renal disease that may
occur at any age in humans and animals. Although its pathogenesis
remains elusive, cases with childhood onset have certain genetic
components, whereas males and females are equally likely to be
affected (3).
The clinical manifestations of CG are not
sufficiently specific for diagnosis uninformed by pathological
findings. The major clinical manifestations of CG are proteinuria,
hypertension and subsequent slowly progressive chronic kidney
disease (1,4,5).
Electron microscopic examination of glomerular basement membrane
(GBM) biopsy specimens has revealed massive deposition of banded
type III collagen fibrils (diameter, 50-60 nM) (2). In settings such as developing
countries without ready access to electron microscopy,
immunohistochemical (IHC) examination may be useful. Since its
etiology and pathogenesis remain unknown, there are no specific
treatments for CG, which has a poor prognosis and frequently leads
to end-stage kidney failure calling for renal transplant (6).
Treatment with angiotensin II receptor blockers,
including telmisartan, may decrease the glomerular pressure and a
previous report indicated that telmisartan is able to attenuate
kidney cell apoptosis and autophagy-associated protein expression
in kidney diseases (7).
Furthermore, telmisartan has anti-fibrotic effects in human
mesangial cells and decreased the expression levels of the
peroxisome transcription factor peroxisome proliferator-activated
receptor (PPAR)γ and the L-arginine-dependent neuronal nitric oxide
synthase (nNOS) to delay the progression of renal failure (8-11).
To the best of our knowledge, the present study was the first to
report a case of nephrotic syndrome (NS) with CG in northeastern
China. Diagnosis was confirmed by IHC and electron microscopic
examination of a biopsy specimen. This patient was followed up for
3 years of therapy with telmisartan, which effectively relieved the
clinical symptoms and delayed the progression and loss of renal
function. From this experience and a literature search, a
mini-review of CG was derived, aiming to make clinicians aware of
this condition and telmisartan as an efficient treatment
modality.
Case report
A 59-year-old male patient was admitted to the
Second Hospital of Jilin University (Changchun, China) for the
treatment of intermittent edema without any other complaints in May
2016. The patient had suffered from intermittent edema of the lower
limbs for one month. The patient did not report other history of
disease or heredopathia. On examination, the patient was revealed
to be hypertensive (150/95 mmHg), edematous and to have
albuminuria; however, the systemic examination was otherwise
unremarkable. The laboratory data indicated the following levels:
Proteinuria, 3+; serum total protein, 55.3 g/l; blood albumin, 27.1
g/l; blood urea nitrogen, 8.97 mM; blood uric acid, 597 µM; blood
creatinine, 1.233 mg/dl; blood triglyceride, 2.64 mM; blood
cholesterol, 6.81 mM; and 24-h urine protein output, 5.44 g, which
were higher compared with normal levels. The patient's clinical and
laboratory data at initial presentation are presented in Table I. According to the medical history
and laboratory findings, the patient was first diagnosed with NS
and consequently, renal biopsy was performed. The patient signed an
informed consent agreement prior to biopsy that allowed the use of
the clinical and pathological data for scientific research.
Institutional Review Board approval (approval no. 2018198) was
provided prior to study onset.
 | Table ILaboratory data of the patient at
initial presentation. |
Table I
Laboratory data of the patient at
initial presentation.
| Parameter | Value | Normal range |
|---|
| White blood cells
(x109/l) | 9.0 | 3.5-9.5 |
| Hemoglobin (g/l) | 120 | 115-150 |
| Platelet count
(x109/l) | 242 | 125-350 |
| Creatinine
(µmol/l) | 102 | 57-111 |
| 24-h urinary protein
(g) | 5.44 | <0.15 |
| Triglyceride
(mmol/l) | 2.64 | 0.56-1.71 |
| Cholesterol
(mmol/l) | 6.81 | 2.90-5.17 |
| Albumin (g/l) | 27.3 | 40.0-55.0 |
| HBsAg | Negative | 0.00-0.05 |
| Anti-HBs | Negative | 0.00-10.00 |
| Anti-HBc | Negative | 0.00-1.00 |
| Rheumatoid factor
(IU/ml) | <20.0 | <20.0 |
| Complement C4
(mg/dl) | 21.2 | 16-38 |
| Complement C3
(mg/dl) | 99.5 | 79-152 |
| ANA | Negative | <1:100 |
| HIV | Negative | 0-1.000 |
| Proteinuria | 3+ | Negative |
| IgG (g/l) | 5.25 | 7.51-15.60 |
| IgM (g/l) | 0.787 | 0.46-3.04 |
| IgA (g/l) | 1.68 | 0.82-4.53 |
| ESR (mm) | 89 | <20 |
| ANCA (RU/ml) | Negative | 0-20 |
Immunofluorescence (IF) microscopy of the biopsy
specimen revealed IgA(-), IgM(-), IgG(-), complement (C)3(-),
C4(-), C1q(-), fibronectin(-), κ(-) and λ(-). Under light
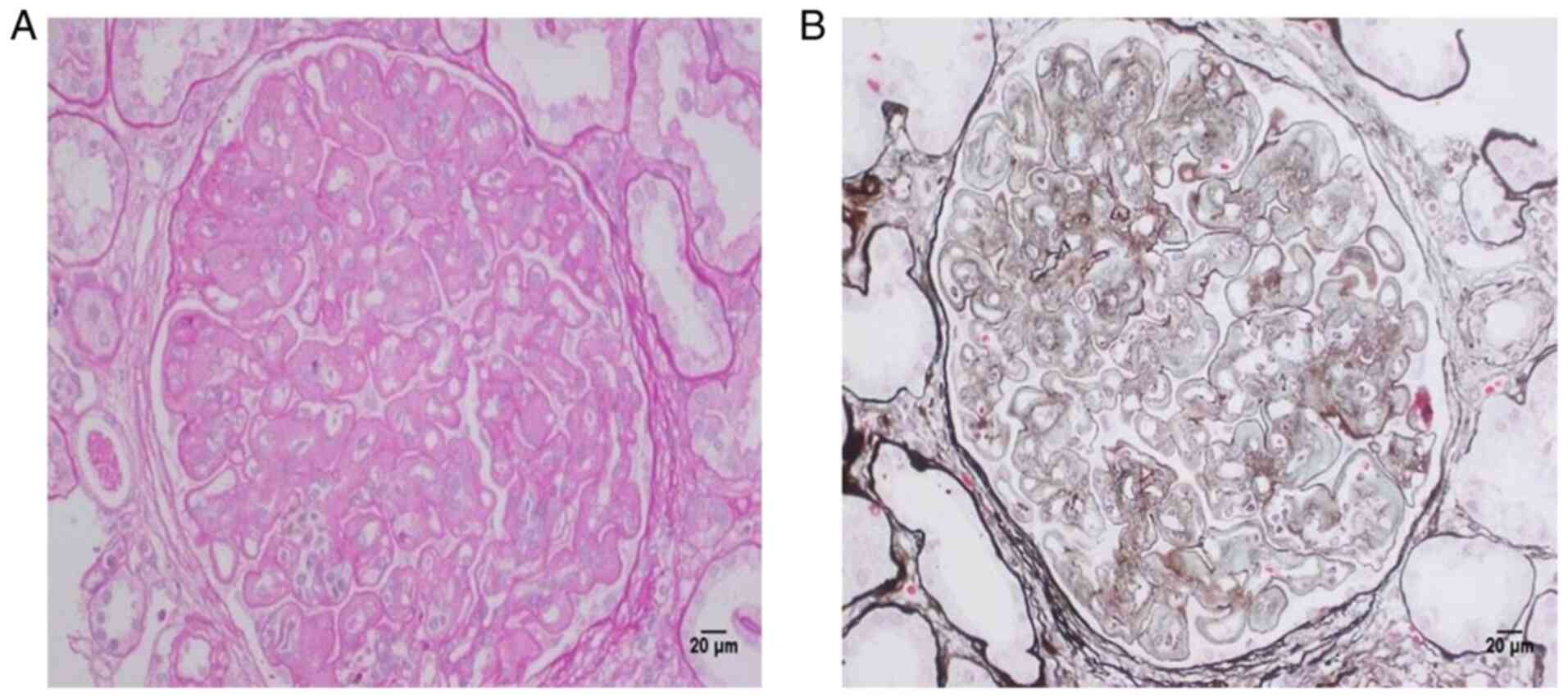
microscopy, the sample contained 33 complete glomeruli, most of
which were lobulated in shape, with occasional enlargement.
Glomerular mesangial cells exhibited mildly or moderately diffuse
proliferation along with endothelial cell proliferation. Light
periodic acid Schiff (PAS) polysaccharide staining was detected in
the mesangium and capillary loops. Renal tubular epithelial cells
exhibited vacuolar degeneration and atrophy, and protein casts.
Focal fibrosis and infiltration of inflammatory cells were observed
in the renal interstitium. Part of the arteriole wall was slightly
thickened (Fig. 1A). No significant
fuchsin staining deposition or rete peg epithelial extensions were
evident on periodic acid methenamine silver + Masson staining
(Fig. 1B). This pathology had not
been previously encountered at our hospital; literature
consultation of the results for light microscopy indicated CG
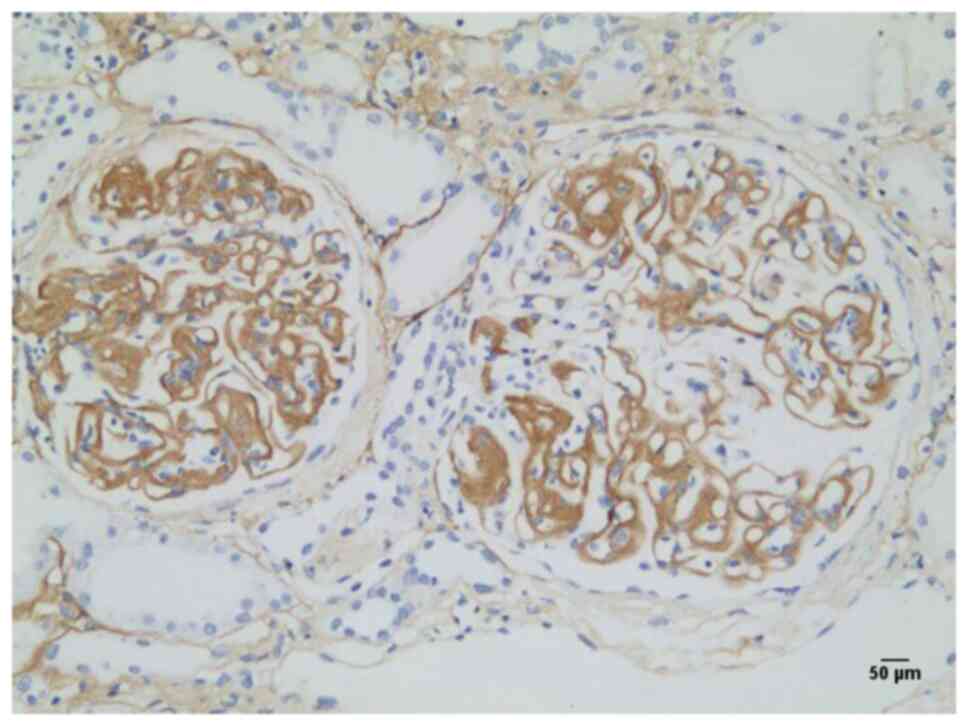
(2). Subsequent IHC examination
suggested deposition of collagen III in subendothelial and
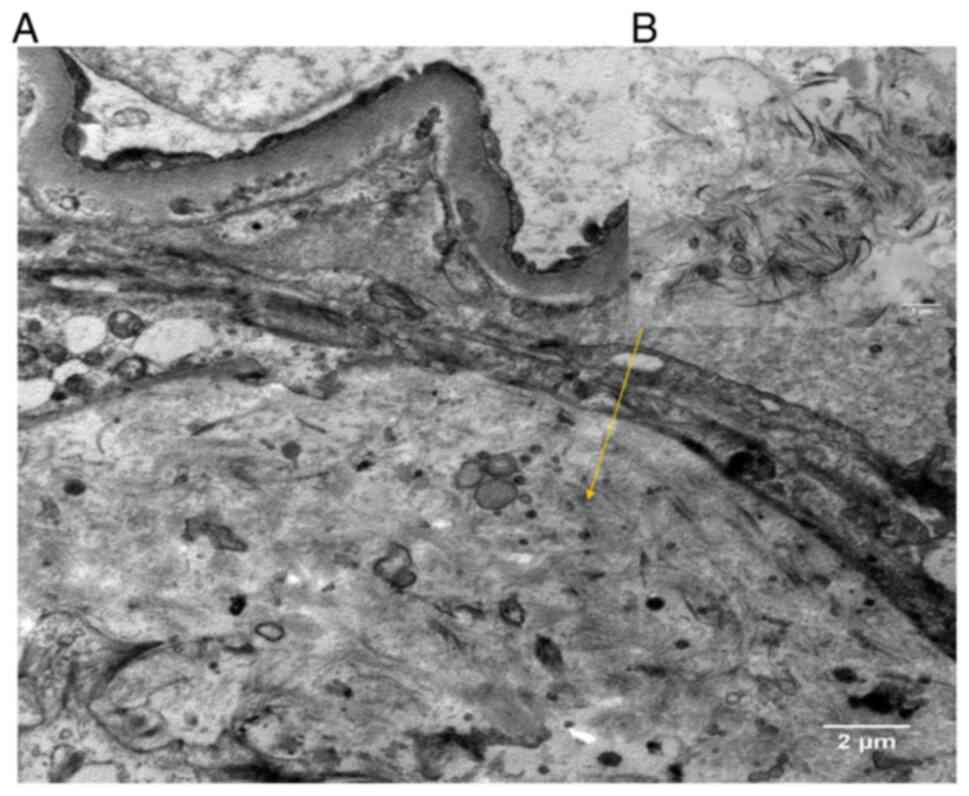
mesangial regions (Fig. 2), which
was confirmed by electron microscopy (Fig. 3) upon pathological diagnosis of CG.
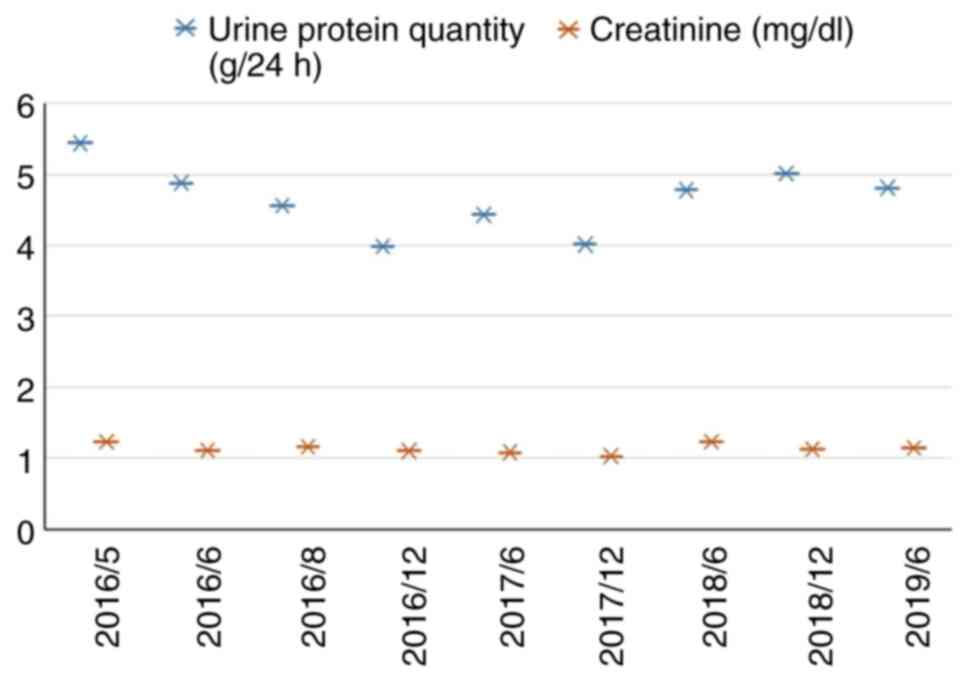
The treatment of the patient with telmisartan (160 mg/d; qd) was
then initiated, which substantially relieved the clinical symptoms
after half a year. Within 3 years of follow-up, the patient had
only two episodes of lower-limb edema and renal function remained
normal despite persistent proteinuria in regular follow-ups
(Fig. 4). The plasma albumin levels
remained at ~30 g/l and uric acid remained persistently slightly
elevated.
Discussion
CG, also known as collagenofibrotic glomerulopathy,
is a rare idiopathic glomerular disease with massive deposition of
abnormal type III collagen protein fibrils in the glomeruli
(12). Persistent massive
proteinuria or NS are among the first complaints in numerous
patients with CG, which then progress to renal damage (6). Type III collagen is normally expressed
in the interstitium and blood vessels throughout the body; however,
it is undetectable in glomeruli. The dysregulated formation of type
III collagen is deposited in the GBM, subendothelium or mesangium,
thus propagating fibrosis. According to the anatomic region of
deposition, CG is divided into collagenofibrotic glomerulopathy and
NPS (2). Deposition is mostly
detected in the subendothelium and mesangium of patients with CG,
while deposition is mainly located at the basement membrane with
extra-renal manifestations in patients with NPS (2). The incidence of CG is low with only
~100 cases reported worldwide to date (Table II) most of them in Asia (2,13-16).
To the best of our knowledge, the patient reported in the present
study was the first case of CG reported in northeastern China and
to be diagnosed by IHC test and further confirmed by electron
microscopy.
 | Table IICases of collagenofibrotic
glomerulopathy reported in previous studies published in the
English language to date. |
Table II
Cases of collagenofibrotic
glomerulopathy reported in previous studies published in the
English language to date.
| Author, year | Region | Cases (n) | Treatment (n/N) | Treatment (n/N) | (Refs.) |
|---|
| Ng et al,
2020; Chen et al, 2017 | China | 33 | 1/33 | 1/33 | (13,15) |
| Anitha et al,
2016; Ng et al, 2020; Matthai et al, 2020; Kurien
et al, 2015 | Other Asia | 57 | 3/57 | 3/57 | (2,13,14,16) |
| Ng et al,
2020 | North/South
America | 6 | - | - | (13) |
| Ng et al,
2020 | Europe | 2 | - | - | (13) |
| Ng et al,
2020 | Not specified | 1 | - | - | (13) |
| Total | | 99 | | | |
The clinical onset of GC typically occurs between
the ages of 30-50, although, recently, a case of a 3-year old
female was reported (17) and there
are occasional cases in older children (18). Massive proteinuria and NS are the
major clinical manifestations; while certain patients have
microscopic hematuria, about half of the patients present with
hypertension only (without extra-renal symptoms) and typically
progress to end-stage renal disease (6,19).
There is no specific indication of CG in laboratory examinations,
although elevated levels of the precursor of procollagen type III
in the circulation may be of a certain, albeit nonspecific,
diagnostic significance (3).
Therefore, obtaining a correct diagnosis may be challenging.
Renal biopsy is the only definitive diagnostic
method for CG and electron microscopy of biopsy material is the
gold standard (2). However,
electron microscopy is not available in a routine setting;
therefore, IHC represents a good alternative for making a positive
diagnosis (15). Previous light
microscopy studies indicated mesangial expansion with weakly
PAS-positive material and negative PAM staining, as well as
negative IF results (3,20). Electron microscopy indicates typical
curved and scattered collagen fibrils in the subendothelial and
mesangial regions with a periodicity of 50-60 nm (21). IHC testing indicates strong
expression of type III collagen within the mesangium and along GBMs
(13). Despite these markers,
differential diagnosis of CG from other collaged III deposition
diseases, including fibrillary glomerulopathy, immunotactoid
glomerulopathy and fibronectin glomerulopathy, can be distinguished
by examination with an electron microscope (15,20).
Although the etiopathogenesis of CG remains elusive,
it appears that genetic factors serve an important role. An
association between inherited deficiency of complement factor H and
CG, such that the absence of factor H may promote the deposition of
type III collagen, has been reported (22). Furthermore, familial lineages with
CG have been reported (23),
implying that genetics may at times contribute to pathogenic
pathways. However, the molecular and genetic mechanisms of the
pathology of CG remain elusive and the typical adult onset may
argue against a common hereditary factor. However, there remains no
specific therapy for CG (24) and
patients have poor prognosis, generally progressing to kidney
failure within a number of years.
Inhibitors of the renin-angiotensin-aldosterone
system are the first-line clinical treatment, aiming to reduce
blood pressure and urine protein. Medicinal approaches include
inhibitors of angiotensin-converting enzyme (ACE) and of
angiotensin II receptor blockers (ARB) (25). There are certain common adverse
effects of ACE inhibitors, including coughing, renal dysfunction
and hyperkalemia, and, in particular, prominent angioedema
(11). ARB drugs are effective in
reducing the risk of end-stage renal disease in patients with
diabetic nephritis and decreased serum creatinine levels two-fold
compared to those obtained with ACE inhibitors (25). Therefore, due to the potent
renoprotective effects and more favorable side effect profile, ARB
medication was selected to treat the patient of the present study.
Telmisartan was chosen because it is reportedly more effective than
other ARB drugs in alleviating proteinuria (26-28).
Furthermore, telmisartan is able to attenuate renal podocyte
apoptosis and autophagy-associated protein expression levels
(7). Finally, telmisartan may have
anti-fibrotic effects in human mesangial cells and reportedly
decreased the expression levels of PPARγ and nNOS to delay the
progression of renal failure (8-11).
Long term follow-up of the present case included the thorough and
persistent monitoring for disease progression, which may be
recommended for telmisartan as a first-line treatment for CG.
In conclusion, although most cases of CG have been
reported in Asia, to the best of our knowledge, the present study
is the first to report on a case of this very rare idiopathic renal
disease in northeastern China. The present study contributes to a
better awareness of CG as a rare cause of proteinuria and presents
telmisartan as an effective treatment against the progression of
the disease and major symptoms over a follow-up of three years.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
QG and PL designed the current study and revised the
manuscript. QG wrote the manuscript. LL and PN collected data. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
The present study did not involve any animal studies
or human experimentation. The project was approved by the Medical
Ethics Committee of the Second Hospital of Jilin University
(Changchun, China; approval no. 2018198).
Patient consent for publication
Written informed consent was obtained from the
patient and the patient allowed his clinical and pathological data
to be published.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Dong J, Wei H, Han M, Guan Y, Wu Y and Li
H: Collagen type III glomerulopathy: A case report and review of 20
cases. Exp Ther Med. 10:1445–1449. 2015.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Anitha A, Vankalakunti M, Siddini V, Babu
K, Bonu R and Ballal S: Type III collagen disorders: A case report
and review of literature. Indian J Pathol Microbiol. 59:75–77.
2016.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Fogo AB, Lusco MA, Najafian B and Alpers
CE: AJKD atlas of renal pathology: Type III collagen
glomerulopathy. Am J Kidney Dis. 69:e25–e26. 2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Patro KC, Jha R, Sahay M and Swarnalatha
G: Collagenofibrotic glomerulopathy-case report with review of
literature. Indian J Nephrol. 21:52–55. 2011.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Rørtveit R, Eggertsdóttir AV, Thomassen R,
Lingaas F and Jansen JH: A clinical study of canine collagen type
III glomerulopathy. BMC Vet Res. 9(218)2013.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Liu H, Chen J, Zhang Y, Wang S and Zou W:
Clinicopathologic features of collagen III glomerulopathy. Zhonghua
Bing Li Xue Za Zhi. 43:732–735. 2014.(In Chinese). PubMed/NCBI
|
|
7
|
Malik S, Suchal K, Gamad N, Dinda AK, Arya
DS and Bhatia J: Telmisartan ameliorates cisplatin-induced
nephrotoxicity by inhibiting MAPK mediated inflammation and
apoptosis. Eur J Pharmacol. 748:54–60. 2015.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Kwon YJ, Suh GH, Kang SS and Kim HJ:
Successful management of proteinuria and systemic hypertension in a
dog with renal cell carcinoma with surgery, telmisartan, and
amlodipine. Can Vet J. 59:759–762. 2018.PubMed/NCBI
|
|
9
|
Liang W, Chen C, Shi J, Ren Z, Hu F, van
Goor H, Singhal PC and Ding G: Disparate effects of eplerenone,
amlodipine and telmisartan on podocyte injury in
aldosterone-infused rats. Nephrol Dial Transplant. 26:789–799.
2011.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Mikami D, Kimura H, Kamiyama K, Torii K,
Kasuno K, Takahashi N, Yoshida H and Iwano M: Telmisartan activates
endogenous peroxisome proliferator-activated receptor-δ and may
have anti-fibrotic effects in human mesangial cells. Hypertens Res.
37:422–431. 2014.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Wang ZK, Liu ZY and Yu HB: Protective
effect of telmisartan on rats with renal failure and its mechanism.
Asian Pac J Trop Med. 8:498–501. 2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Kunitomi M, Wada J, Miyatake N, Hayashi Y,
Ota K and Makino H: Ultrastructure of mesangial type III collagen
deposition in a patient with IgA nephropathy. Am J Kidney Dis.
32:146–152. 1998.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Ng YF, Chow CY, Yang WS, Lye WC and Loh
HL: Collagenofibrotic glomerulopathy-report of a rare renal disease
with serial biopsies. Malays J Pathol. 42:131–135. 2020.PubMed/NCBI
|
|
14
|
Matthai SM, Mohapatra A, Duhli N, David VG
and Varughese S: Collagenofibrotic glomerulopathy-A rare disease
diagnosed with the aid of transmission electron microscopy. Indian
J Pathol Microbiol. 63:S47–S49. 2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Chen X, Wang H, Xu W and Zhu J: Collagen
type III glomerulopathy: Case report and review of the literature.
Clin Nephrol. 87:39–46. 2017.PubMed/NCBI View
Article : Google Scholar
|
|
16
|
Kurien AA, Larsen CP and Cossey LN:
Collagenofibrotic glomerulopathy. Clin Kidney J. 8:543–547.
2015.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Pizzo HP, Haas M and Puliyanda D: Collagen
type III glomerulopathy. Kidney Int. 93(1490)2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Alsaad KO, Edrees B, Rahim KA, Alanazi A,
Ahmad M and Aloudah N: Collagenofibrotic (collagen type III)
glomerulopathy in association with diabetic nephropathy. Saudi J
Kidney Dis Transpl. 28:898–905. 2017.PubMed/NCBI
|
|
19
|
Cohen AH: Collagen type III
glomerulopathies. Adv Chronic Kidney Dis. 19:101–106.
2012.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Li L, Zou WZ, Wang SX, Wang SL, Wang W,
Han ZH, Du J and Bo L: Collagen type III glomerulopathy: A
morphologic study. Zhonghua Bing Li Xue Za Zhi. 34:385–388.
2005.(In Chinese). PubMed/NCBI
|
|
21
|
Bao H, Chen H, Zhu X, Xu F, Zhu M, Zhang
M, He Q, Zeng C and Liu Z: Clinical and morphological features of
collagen type III glomerulopathy: A report of nine cases from a
single institution. Histopathology. 67:568–576. 2015.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Vogt BA, Wyatt RJ, Burke BA, Simonton SC
and Kashtan CE: Inherited factor H deficiency and collagen type III
glomerulopathy. Pediatr Nephrol. 9:11–15. 1995.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Chen N, Pan X, Xu Y, Wang Z, Shi H, Yan F
and Dong X: Two brothers in one Chinese family with collagen type
III glomerulopathy. Am J Kidney Dis. 50:1037–1042. 2007.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Nimmagadda S, Mukku K, Devaraju SR and
Uppin MS: Unusual cause of glomerular deposition disease:
Collagenofibrotic glomerulopathy. Indian J Nephrol. 27:62–65.
2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Wang K, Hu J, Luo T, Wang Y, Yang S, Qing
H, Cheng Q and Li Q: Effects of angiotensin-converting enzyme
inhibitors and angiotensin II receptor blockers on all-cause
mortality and renal outcomes in patients with diabetes and
albuminuria: A systematic review and meta-analysis. Kidney Blood
Press Res. 43:768–779. 2018.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Zheng Z, Lin S and Shi H: A systematic
review and meta-analysis of telmisartan versus valsartan in the
management of essential hypertension. J Clin Hypertens (Greenwich).
12:414–421. 2010.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Bakris G, Burgess E, Weir M, Davidai G and
Koval S: AMADEO Study Investigators: Telmisartan is more effective
than losartan in reducing proteinuria in patients with diabetic
nephropathy. Kidney Int. 74:364–369. 2008.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Naruse M, Koike Y, Kamei N, Sakamoto R,
Yambe Y and Arimitsu M: Effects of azilsartan compared with
telmisartan on insulin resistance in patients with essential
hypertension and type 2 diabetes mellitus: An open-label,
randomized clinical trial. PLoS One. 14(e0214727)2019.PubMed/NCBI View Article : Google Scholar
|