Introduction
Thyroid-associated ophthalmopathy (TAO) is one of
the most common extra-thyroidal manifestations of Graves' disease
(GD). It is frequently mild and self-limiting and only requires
observation or local treatment. However, 3-5% of cases of TAO
progress and require more intensive therapy, including
glucocorticoid therapy, orbital radiotherapy and orbital
decompression surgery (1). As one
of the most important non-surgical approaches, glucocorticoid
therapy may be administered via the oral, retrobulbar,
subconjunctival or intravenous (i.v.) route. Intravenous
glucocorticoids have been confirmed to be more effective and much
safer than administration via oral and local routes (2). Therefore, the preferred first-line
treatment for active moderate-to-severe TAO, as recommended by the
European Group on Graves' Ophthalmopathy (EUGOGO), is i.v.
glucocorticoid therapy (3). There
are two major regimens for i.v. glucocorticoid therapy: one is a
weekly protocol and the other is a daily scheme. A recent study has
indicated that a weekly protocol was more effective and safer than
a daily scheme in a cumulative dose of 4.5 g i.v. glucocorticoid
therapy for moderate-to-severe active TAO (4). However, the details of the treatment
schedule are still debatable. More data and further studies are
required. On the other hand, the first and most important step
prior to starting therapy is to establish whether TAO is severe and
whether there is active inflammation (5). To date, no biomarker has been
confirmed that is able to definitely assess the activity and
severity of TAO and the effect of treatment. Therefore, the primary
objective of the present study was to compare the treatment effects
between the two regimens. The secondary objective was to
investigate associated cytokines.
Patients and methods
Patients
The present study was a single center, prospective,
randomized, open-label trial performed at the Third Affiliated
Hospital of Sun Yat-sen University (Guangzhou, China) between
January 2012 and June 2017. A total of 160 consecutive patients
with TAO (including 54 inactive, 16 active mild and 90 active
moderate-to-severe), 60 patients with isolated GD without
ophthalmopathy and 60 normal controls (NC) were enrolled. The
diagnoses of TAO were based on the EUGOGO consensus (3). Patients were excluded if they had
infectious diseases, allergies, autoimmune diseases, coronary
artery disease, diabetes mellitus, hypertension, liver disease,
kidney disease or a history of alcoholism. None of the patients had
received any previous steroid treatment, orbital radiotherapy or
orbital surgery.
Study design
A total of 90 patients with active
moderate-to-severe TAO were randomized at a ratio of 1:1 to receive
glucocorticoid therapy for 12 weeks on a weekly protocol or daily
scheme. Sealed opaque envelopes, which were arranged in a
computer-generated random order prepared by a statistician prior to
the study, were opened to determine the patients' treatment
assignments. The weekly protocol was as follows: 0.5 g
methylprednisolone i.v. weekly for 6 weeks and then the dose was
tapered by 0.25 g/week over the following 6 weeks. The daily scheme
was as follows: 0.5 g methylprednisolone i.v. daily for 5
consecutive days, followed by oral methylprednisolone for 3 months.
Oral methylprednisolone started at 32 mg/day for 2 weeks and then
the dose was tapered by 4 mg/day every 2 weeks. As a result, the
cumulative doses of glucocorticoid were 4.5 g in the weekly
protocol and 4.3 g in the daily scheme (P>0.05). All patients
were required to quit smoking, to take oral anti-thyroid drugs and
received local measures for relieving symptoms (including
sunglasses, artificial tears, lubricant ointments and prisms)
according to advice from specialists. Within the cohort, two
patients treated under the weekly protocol and four on the daily
scheme were infected with hepatitis B. All of them received
antiviral treatments.
Measurement of serum cytokines
Blood samples from all 289 subjects, which consisted
of 69 NCs, 60 patients with isolated GD and 160 with TAO, were
collected at baseline. At the 12-week follow-up visit, blood
samples from all 160 subjects with TAO were collected, which
comprised 54 patients with inactive TAO, 16 patients with active
mild TAO and 90 patients with active moderate-to-severe TAO. Serum
samples were centrifuged and stored at -80˚C for future analysis.
Interleukin (IL)-2, IL-6 and IL-17 concentrations were measured
using a solid-phase sandwich ELISA (Neobioscience Technology Co.).
The lowest detectable limits of the assays for IL-2, IL-6 and IL-17
were 2.23, 0.7 and 1.10 pg/ml, respectively.
Ophthalmic assessment
Ophthalmic assessments were performed prior to
therapy and at the 12-week follow-up visit. The ophthalmic
assessments included physical eye examinations (eye symptoms,
swelling, conjunctival congestion, diplopia, visual acuity and
proptosis) and orbital CT examinations (thickness of the
extraocular muscles and orbital fat density). Physical eye
examinations were performed by one single ophthalmologist and
orbital CT examinations were determined by a single radiologist.
Proptosis was measured by a Hertel exophthalmometer. The upper
limit of normal for Chinese subjects in the present study was 18.6
mm (6). Soft tissue involvement was
assessed using a color atlas. The thickness of the extraocular
muscles was defined as the maximum coronal section area of the most
hypertrophic rectus muscle in each eye, as measured by orbital CT.
Activity of TAO was assessed via the clinical activity score (CAS)
(7). The severity of TAO was
assessed according to the EUGOGO (3).
Clinical outcome assessment
The primary clinical outcome was the overall
response rate. ‘Response’ was defined as an improvement in at least
two major criteria and one minor criterion. Otherwise, the outcome
was considered ‘non-response’. The major criteria included the
following: i) Improvement in CAS by ≥2 points, ii) improvement in
diplopia(disappearance or reduction in degree), iii) improvement in
visual acuity by ≥1/10 and i.v.) reduction in proptosis or lid
width of ≥2 mm. The minor criteria were favorable changes in soft
tissues and improvement in the patient's self-assessment (8).
The secondary clinical outcomes were the remission
rate of major ophthalmic manifestations, average changes in the CAS
and adverse events.
Adverse events
To assess the treatment-associated side effects,
body weight, blood pressure, routine biochemical analysis and
electrocardiogram were measured at baseline and at the 12-week
follow-up visit. The measurements were also performed whenever the
patient felt uncomfortable. Major adverse events were defined as
life-threatening liver failure and cardio- or cerebrovascular
complications (5). Minor adverse
events included weight gain (an increase of at least 2 kg at the
12-week follow-up visit compared to baseline), hyperglycemia
(fasting blood glucose ≥7 mmol/l or 2-h postprandial blood glucose
≥11.1 mmol/l), hypertension (systolic blood pressure ≥140 mmHg or
diastolic blood pressure ≥90 mmHg), renal dysfunction (estimated
glomerular filtration rate <60 ml/min/1.73 m2),
hepatic dysfunction (alanine aminotransferase or aspartate
aminotransferase elevated by 2.5-fold above the upper normal
limits), gastrointestinal discomfort, sleeplessness and
infection.
Statistical analysis
All statistical analyses were performed with SPSS
18.0 (SPSS, Inc.). Continuous variables are expressed as the mean ±
standard deviation or median (interquartile range,
25th-75th percentile). Categorical variables
are expressed as frequencies and percentages. Differences in
continuous variables between groups were evaluated by Student's
t-test, while differences in categorical variables between groups
were analyzed by the χ2 test. Paired Student's
t-tests were used to compare continuous variables prior to
and after treatment. One-way analysis of variance was applied for
comparison of continuous variables among >2 groups, which was
followed by Fisher's Least Significant Difference post-hoc test in
the case of statistical significance. P<0.05 was considered to
indicate a statistically significant difference.
Results
Clinical characteristics at
baseline
A total of 280 individuals (158 men and 122 women;
age, 35.1±9.8 years) were enrolled. A total of 82 (29.3%)
individuals were smokers and 198 (70.7%) were non-smokers. The
detailed clinical characteristics at baseline are summarized in
Table I. No significant differences
were observed in terms of age, sex, or smoking status among all
groups. Patients with isolated GD and TAO had similar levels of
thyroid hormones (P>0.05). Free triiodothyronine and free
thyroxine levels in these two groups were significantly higher than
those in the NC group, whereas thyroid stimulating hormone was
significantly lower (P<0.01).
 | Table IBaseline characteristics of all
groups. |
Table I
Baseline characteristics of all
groups.
| | | | | | Active
moderate-to-severe TAO |
|---|
| Variable | NC (n=60) | Isolated GD
(n=60) | Inactive TAO
(n=54) | Active mild TAO
(n=16) | Weekly protocol
(n=46) | Daily scheme
(n=44) | Overall (n=90) |
|---|
| Age (years) | 32.5±7.4 | 35.1±8.2 | 38.7±10.7 | 34.6±1.8 | 35.2±7.8 | 34.8±10.4 | 35.1±11.8 |
| Sex
(female/male) | 32/28 | 38/22 | 28/26 | 8/8 | 28/18 | 24/20 | 52/38 |
| Disease course of
TAO (months) | NA | NA | 18.60±6.80 |
4.10±0.60a | 12.64±7.73 | 6.56±7.23 |
9.65±7.49a |
| Smoking | 16 (26.7) | 16 (20.0) | 16 (22.2) | 4 (25.0) | 16 (34.8) | 14 (31.8) | 30 (33.3) |
| Ocular
symptoms | | | | | | | |
|
Photophobia | NA | NA | 30 (55.6) | 14 (87.5) | 26 (56.5) | 30 (68.2) | 56 (62.2) |
|
Epiphora | NA | NA | 30 (55.6) | 14 (87.5) | 26 (56.5) | 34 (77.3) | 60 (66.7) |
|
Swelling | NA | NA | 12 (22.2) | 8 (50.0) | 26 (56.5) | 34 (77.3) | 60
(66.7)a |
|
Grittiness | NA | NA | 26 (48.1) | 12 (75.0) | 34 (73.9) | 24 (54.5) | 58 (64.4) |
|
Diplopia | NA | NA | 18 (33.3) | 0 (0)a | 16 (34.8) | 16 (36.4) | 32
(35.6)b |
|
Vision
loss | NA | NA | 18 (33.3) | 0 (0)a | 26 (56.5) | 30 (68.2) | 56
(62.2)b |
|
Pain | NA | NA | 0 (0) | 4
(25.0)a | 26 (56.5) | 30 (68.2) | 56
(62.2)a |
| Proptosis (mm) | NA | NA | 20.01±2.41 | 16.09±3.10 | 17.09±3.20 | 19.33±3.24 | 18.19±3.10 |
| EUGOGO
(mild/moderate/severe) | NA | NA | 16/24/14 | 16/0/0 | 0/36/10 | 0/28/16 | 0/64/26 |
| CAS | NA | NA | 1.0 (1.0) | 4.0
(2.0)a | 4.0 (1.0) | 4.0 (3.0) | 4.0
(3.0)a |
| Thyroid
function | | | | | | | |
|
TRAb
(+) | 0 | 36 (60.0) | 16 (29.6) | 8 (50.0) | 14 (30.4) | 24 (54.5) | 38 (42.2) |
|
TPOAb
(+) | 0 | 8 (13.3) | 28 (51.9) | 8 (50.0) | 20 (43.5) | 20 (45.5) | 40 (44.4) |
| TSH (µIU/ml) | 1.64±0.86 |
0.19±0.15c |
0.36±0.87c |
1.03±6.13d | 0.26±0.67 | 0.38±0.41 |
0.32±0.54c |
| FT4 (pmol/l) | 15.80±2.90 |
31.14±29.47c |
26.27±19.34c |
26.58±20.76c | 31.08±30.44 | 29.80±33.26 |
30.45±31.25c |
| FT3 (pmol/l) | 4.90±1.10 |
11.54±10.06c |
8.35±7.15c |
8.00±5.71c | 12.39±11.15 | 10.24±11.23 |
11.34±11.10c |
| CT features | | | | | | | |
| Thickness of EOM
(mm) | NA | NA | 8.7±2.6 | 6.8±2.1 | 8.9±3.7 | 9.7±3.5 | 9.3±3.6 |
| Orbital fat
(HU) | NA | NA | -94.91±10.19 | -92.25±10.18 | -93.45±11.48 | -94.81±7.13 | -94.11±10.32 |
The duration of eye disease in the subjects with
active TAO (either mild or moderate-to-severe) was much shorter
than that in the subjects with inactive TAO (P<0.01). The
subjects with active moderate-to-severe TAO were more inclined to
experience epiphora and pain in the eyes than the subjects with
inactive TAO (P<0.01). Numerous subjects with active
moderate-to-severe TAO suffered from diplopia and vision loss (35.6
and 62.2%, respectively). However, none of the patients with active
mild TAO complained of diplopia and decreased vision. Proptosis,
thyroid function and ocular CT features were similar among patients
with inactive TAO, active mild TAO and active moderate-to-severe
TAO (P>0.05).
There were no significant differences between the
patients allocated to the weekly protocol and those allocated to
the daily scheme in terms of clinical variables, biochemical
features and ocular CT characteristics (P>0.05).
Clinical outcomes at the 12-week
follow-up visit
The overall response rate was 69.8% in all patients
receiving i.v. glucocorticoid therapy at the 12-week follow-up
visit. The response rate in patients on the daily scheme was higher
than that in those on the weekly protocol, although the difference
was not significant (77.8% vs. 63.6%, P>0.05). In addition,
patients had similar CAS responses, which decreased significantly
from 4 to 2 under the two regimens(P<0.01), but there was no
difference between the two regimens (P>0.05; Table II).
 | Table IIComparison of treatment outcomes
between the two groups. |
Table II
Comparison of treatment outcomes
between the two groups.
| | Weekly protocol
(n=46) | Daily scheme
(n=44) |
|---|
| Item | Baseline | 12 weeks | Baseline | 12 weeks |
|---|
| Eye symptoms | | | | |
|
Photophobia
(+/-) | 26/20 | 4/42 | 30/14 | 10/34 |
|
Epiphora
(+/-) | 26/20 | 8/38 | 34/10 | 10/34 |
|
Swelling
(+/-) | 26/20 | 8/38 | 34/10 | 6/38 |
|
Grittiness
(+/-) | 34/12 | 0/46 | 24/20 | 10/34 |
|
Diplopia
(+/-) | 16/30 | 10/36 (2
worsened) | 16/28 | 6/38 |
|
Vision loss
(+/-) | 26/20 | 10/36 (2
worsened) | 30/14 | 10/34 |
|
Pain
(+/-) | 26/20 | 12/34 | 30/14 | 6/38 |
| Proptosis (mm) | 17.25±1.80 | 17.94±2.08 | 20.42±2.61 | 20.58±2.78 |
| CAS score | 4.0 (1.0) | 2.0
(1.0)a | 4.0 (3.0) | 2.0
(2.0)a |
| Thyroid
function | | | | |
|
FT3
(pmol/l) | 12.39±11.15 |
5.33±1.588b | 10.24±11.23 |
4.82±0.87b |
|
FT4
(pmol/l) | 31.08±30.44 |
15.56±4.36b | 29.80±33.26 |
15.20±2.94b |
|
TSH
(µIU/ml) | 0.26±0.67 |
1.72±1.80b | 0.38±0.41 |
2.82±0.76b |
| CT features | | | | |
|
Thickness of
EOM (mm) | 8.9±3.7 | 8.6±3.4 | 9.7±3.5 | 9.4±3.6 |
|
Orbital fat
(HU) | -91.99±12.49 | -95.23±9.62 | -93.92±7.28 | -104.1±13.15 |
The major ocular symptoms in all patients, which
consisted of photophobia (75%), lacrimation (70%), swelling
(76.7%), grittiness (82.8%), diplopia (62.5%), vision loss (67.9%)
and pain (67.9%), improved significantly (Table III). The two regimens had a
similar remission rate of major ophthalmic manifestations
(P>0.05). Of note, two patients on the weekly protocol acquired
diplopia with vision loss and their conditions worsened. As a
result, they received additional i.v. glucocorticoid therapy and
they improved gradually with the additional treatment.
 | Table IIIRemission of treatment outcomes
between the two groups. |
Table III
Remission of treatment outcomes
between the two groups.
| Item | Total remission
rate (n=90) | Weekly protocol
(n=46) | Daily scheme
(n=44) |
|---|
| Photophobia | 42 (75.0) | 22/26 (84.6) | 20/30 (66.7) |
| Lacrimation | 42 (70.0) | 18/26 (69.2) | 24/34 (70.6) |
| Swelling | 46 (76.7) | 18/26 (69.2) | 28/34 (82.4) |
| Grittiness | 48 (82.8) | 34/34 (100.0) | 14/24 (58.3) |
| Diplopia | 18 (56.2) | 8/16 (50) | 10/16 (62.5) |
| Vision loss | 38 (67.9) | 18/26 (69.2) | 20/30 (66.7) |
| Pain | 38 (67.9) | 14/26 (53.8) | 24/30 (80.0) |
Serum cytokine levels at baseline and
after glucocorticoid therapy
The serum levels of IL-2, IL-6 and IL-17 at baseline
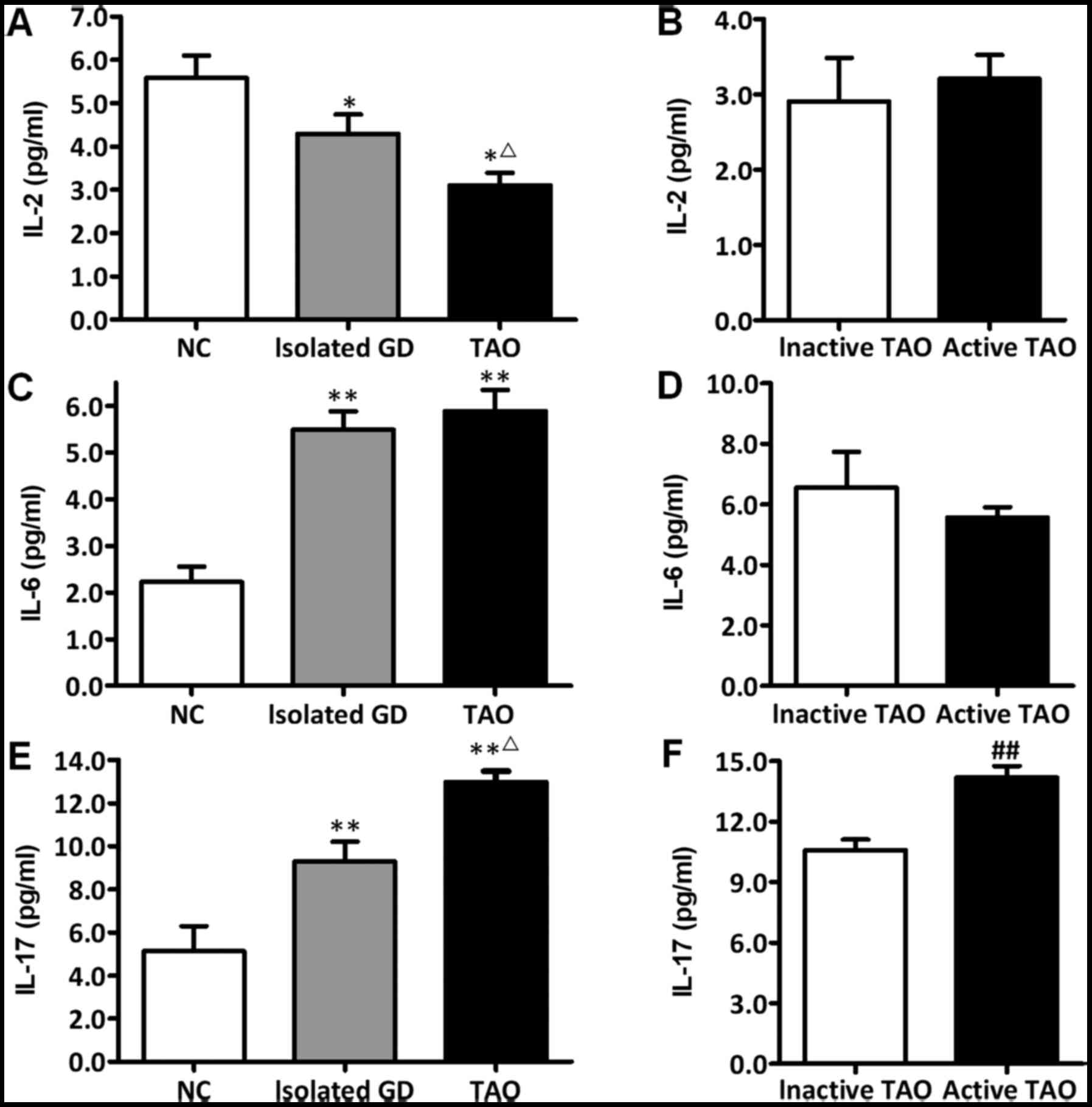
are summarized in Fig. 1. From the
NC group to the isolated GO group followed by the TAO group, the
IL-2 level decreased gradually (P<0.05), while the IL-17 level
increased progressively (P<0.05). The IL-6 level in the isolated
GD group was similar to that in the TAO group (P>0.05), both of
which were significantly higher than those in the NC group
(P<0.05).
Among all subjects with TAO, the IL-17 level in the
active TAO group (either mild or moderate-to-severe) was higher
than that in the inactive TAO group (P<0.01), whereas neither
IL-2 nor IL-6 was different between the active TAO and inactive TAO
groups (P>0.05).
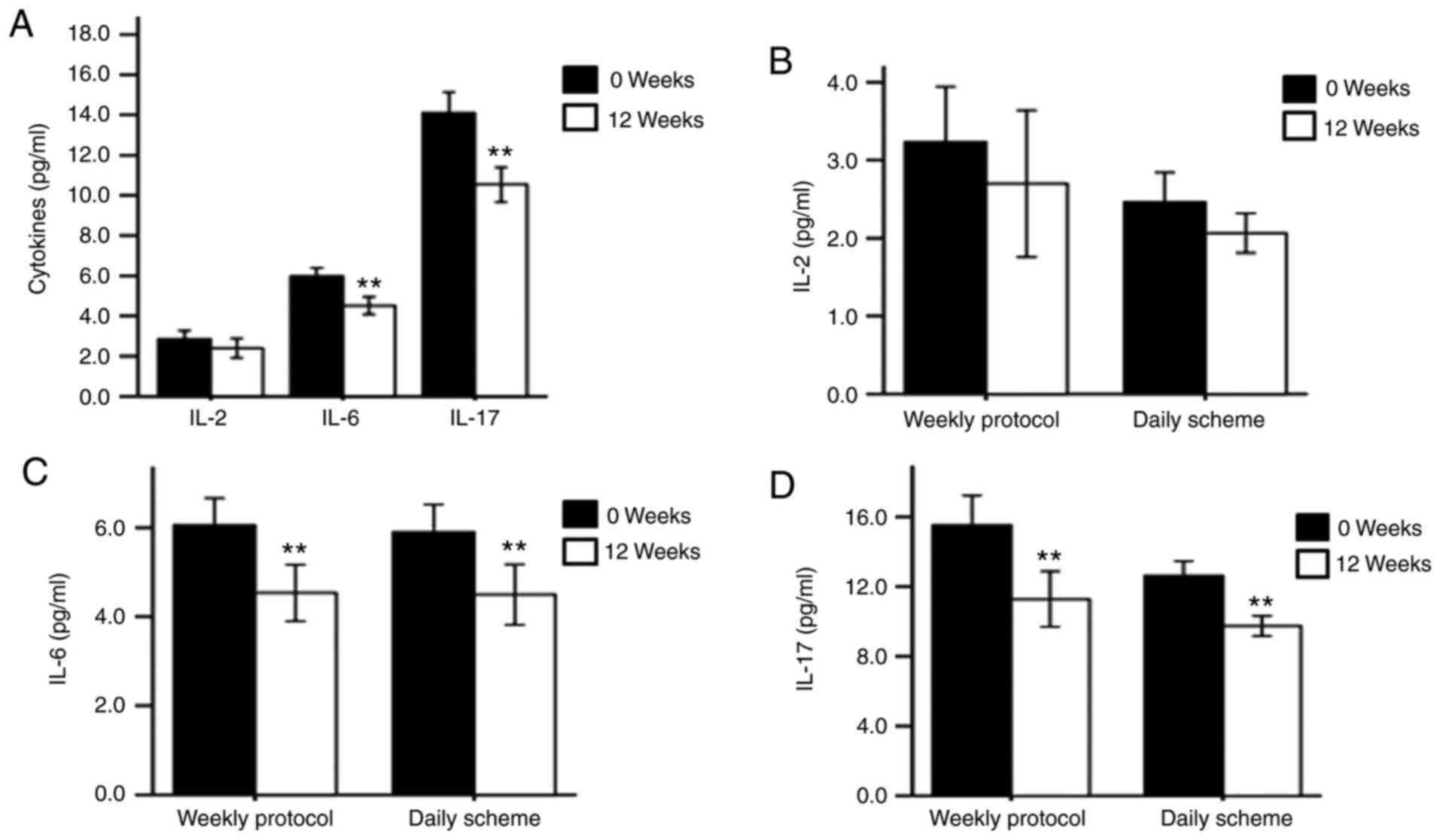
The levels of serum IL-2, IL-6 and IL-17 after
glucocorticoid therapy are summarized in Fig. 2. The IL-2 level in the active
moderate-to-severe TAO group was not significantly changed at the
12-week follow-up visit (P>0.05); however, the IL-6 and IL-17
levels were significantly decreased after treatment (P<0.05).
Notably, the IL-6 and IL-17 levels were markedly declined after
treatment with either of the two regimens (P<0.01). Furthermore,
the changes in IL-6 and IL-17 were similar between the two regimens
(P>0.05; Fig. 2D).
Adverse effects
Intravenous glucocorticoids were fairly well
tolerated in the two regimens without any serious adverse events,
including serious liver function impairment (liver enzymes ≥2.5
times the upper limit) and cardiovascular events. The total number
of patients suffering minor side effects was higher on the daily
scheme than that on the weekly protocol (P<0.05; Table IV). Sleeplessness, gastrointestinal
discomfort and hyperglycemia were the most common adverse events on
the daily scheme, while weight gain was relatively common on the
weekly protocol. Blood pressure and liver and renal function
parameters remained normal under the two regimens.
 | Table IVAdverse events of glucocorticoid
therapy. |
Table IV
Adverse events of glucocorticoid
therapy.
| Item | Weekly
protocol | Daily scheme |
|---|
| Number of adverse
events | 16 (4.35) | 40
(11.36)a |
| Patients with
adverse events | 12/46 (26.1) | 28/44
(63.6)b |
| Weight gain | 8 | 4 |
| Hyperglycemia | 0 | 12 |
| Hypertension | 0 | 0 |
| Gastrointestinal
discomfort | 4 | 8 |
| Sleeplessness | 4 | 12 |
| Hepatic lesion | 0 | 0 |
| Renal
dysfunction | 0 | 0 |
| Infection | 0 | 4 |
Discussion
The present study was a single center prospective
randomized trial in which the efficacy and safety of two regimens
of i.v. glucocorticoid were compared. In this trial, the
improvement in clinical manifestations, including photophobia,
epiphora, swelling, grittiness, diplopia, vision loss, pain,
proptosis and CAS score, and thyroid function and CT features on
extraocular muscle after 12-week treatment were the primary
outcomes and the levels of three cytokines were also investigated
at the same time. The results suggested that the daily scheme had a
higher response rate than the weekly protocol, although the
difference was not significant and that these two regimens yielded
similar improvements in serum cytokines. Numerous minor side
effects were recorded in patients on the daily scheme, whereas two
patients worsened on the weekly protocol.
Although the pathogenesis of TAO is remains to be
fully elucidated, glucocorticoid therapy is the recommended
first-line treatment for TAO (9).
Glucocorticoid therapy may be administered via oral, local
(retrobulbar or subconjunctival) and i.v. routes. Studies have
indicated that high-dose glucocorticoids administered via the i.v.
route are more effective and better tolerated than those taken
orally (10-12).
There are various schedules for i.v. glucocorticoid therapy. A
recent study indicated that a weekly protocol (4.5 g of i.v.
glucocorticoid therapy) is safer and more effective than a daily
scheme (4). However, the present
results suggested that the daily scheme and the weekly protocol
were effective for active TAO and that the daily scheme had a
relative higher response rate than that of the weekly protocol,
although the difference was not statistically significant. Why did
the response rate in the present study differ that in the previous
study? This may be due to the different details of the daily
scheme. In a previous study, i.v. glucocorticoid was administered
for 3 consecutive days per week for 4 weeks (4). However, in the present trial, i.v.
glucocorticoid was administered for 5 consecutive days. It is
possible that extending the treatment time of i.v. glucocorticoid
intensified immunosuppression, which leads to favorable effects
(5). The difference in the
selection of patients may be another reason (5). The response rate was 77.8% on the
daily scheme and 63.6% under the weekly protocol in the present
study. Although the difference appeared obvious, it was not
statistically significant. Future studies with larger sample size
and longer study duration would be required to determine whether
there was truly no difference or whether the 12-week follow-up
period was not long enough or whether a total of 90 patients was
not large enough to reach statistical significance in the present
study.
The two regimens were able to improve soft tissue
inflammation. However, neither regimen improved extraocular muscle
dysfunction and proptosis. This may be due to two reasons. One was
the short follow-up time. Improvement in extraocular muscle
dysfunction and proptosis requires more time than amelioration of
soft tissue inflammation. Another reason was probably the fibrosis
in the extraocular muscles, which is more difficult to resolve than
edema. Although certain studies indicated that glucocorticoid
therapy improved extraocular muscle dysfunction and proptosis
(5,13,14),
other studies were in agreement with the present results (10-12,15,16).
A systematic review also indicated that glucocorticoid therapy had
a limited effect on proptosis (5).
Further studies are required to clarify these effects.
Generally, intravenous glucocorticoids are fairly
well tolerated. Cardiovascular risk and hepatotoxicity are the most
severe adverse effects of high-dose glucocorticoids (3-5).
In the present study, no cardiovascular events or impaired liver
function was recorded in the subjects on either the weekly protocol
or daily scheme. The daily scheme had a slightly higher incidence
of total adverse effects than that of the weekly protocol. However,
the deterioration of eye symptoms was only recorded on the weekly
protocol. Thus, the present results indicated that the two regimens
had their own merits.
TAO is regarded as an autoimmune disease (1). Glucocorticoid therapy is a
well-established treatment for TAO owing to its anti-inflammatory
and immunosuppressive actions (9).
Previous studies have focused on T lymphocytes, particularly
CD4+T-helper (Th) cells, which are the principal immune
effector T cells (17,18). Activated CD4+Th cells
differentiate into Th1, Th2, Th17 and Treg subsets, which produce
numerous cytokines (17). These
cytokines have been proven to have an important role in TAO
(3,19,20).
Accordingly, cytokines that regulate inflammation have the
potential to be biomarkers to evaluate the activity of TAO and the
efficacy of treatment options. IL-2 is a characteristic cytokine of
Th1 T cells and IL-6 is one of the key cytokines secreted by Th2 T
cells (19). In addition, IL-17 is
the signature cytokine of Th17 T cell-derived population with
highly pro-inflammatory properties (21). It is considered to have a
significant role in numerous autoimmune diseases, including
rheumatoid arthritis and systemic lupus erythematosus (22,23).
Therefore, IL-2, IL-6 and IL-17 were selected in the present
study.
In the present study, the IL-2 levels in the TAO and
isolated GD groups were lower than those in the NC group. The IL-6
and IL-17 levels in the TAO and isolated GD groups were higher than
those in the NC group. These results implied that all three
cytokines are involved in GD. Previous studies suggested that
cytokines produced by Th1 T cells were dominant at the early stage
and in active TAO, whereas those secreted by Th2 T cells had a
greater role in later stages and in inactive TAO (24,25).
Certain previous studies reported that IL-2 and IL-6 levels in
active TAO were higher than those in inactive TAO (26-29).
However, this result was not supported by other reports (4,30,31).
In the present study, IL-2 and IL-6 levels in active TAO were
similar to those in inactive TAO. Taken together, the present
results indicated that neither IL-2 nor IL-6 was an ideal biomarker
for the evaluation of TAO. Notably, the IL-17 level was
significantly higher in the TAO group than that in the isolated GD
and NC groups. In addition, the IL-17 concentration in the active
TAO group was higher than that in the inactive TAO group, which is
consistent with previous reports (32-34).
Furthermore, the IL-17 level was markedly decreased at the 12-week
follow-up visit after glucocorticoid therapy under the two
regimens. The present results are in line with those of a previous
study (32). Evidence has indicated
that IL-17 has a key role in the pathogenesis of autoimmune
thyroiditis and GD (35-38).
These data suggested that IL-17 may reflect the inflammatory state
and be a potential indicator for the severity and activity of TAO.
However, a recent study indicated that the IL-17 concentration was
elevated in GD with/without TAO but reduced in active and inactive
TAO (31), which was not in
agreement with previous studies. Given that IL-17 has only recently
been studied, more data should be accumulated.
The present study assessedIL-2, IL-6 and IL-17 to
provide novel insight into TAO. Considering the complexity of the
immunopathogenesis and diversity of cytokines, further studies are
required. The sample size should be enlarged in future studies.
Furthermore, other cytokines produced by infiltrating
immunocompetent cells and orbital fibroblasts should be
investigated to help understand the pathogenesis of TAO more
comprehensively.
In conclusion, the present results provide evidence
indicating that: i) For patients with active moderate-to-severe
TAO, the daily scheme of intravenous glucocorticoids had a higher
response rate than the weekly protocol and these two regimens had
their own merits with regard to adverse effects; and ii) IL-17 has
the potential to serve as a marker for evaluating the activity of
TAO and the efficacy of treatment options.
Acknowledgements
The authors thank the statistician Dr Lian-Xiong
Yuan from the Third Affiliated Hospital of Sun Yat-sen university
for his help with the statistical analysis.
Funding
The current study was funded by National Key R&D
Program of China (grant no. 2017YFA0105803), the General Program of
National Natural Science Foundation of China (grant no. 81770826),
the 5010 Clinical Research Projects of Sun Yat-sen University
(grant no. 2015015), the Science and Technology Plan Projects of
Guangdong Province (grant no. 2016A050502010), the Key Special
Projects of Medical and Health Collaborative Innovation of
Guangzhou City (grant no. 201604020016) and the Special Scientific
Research Project of Guangzhou City (grant no. 2060404). The funding
bodies had no role in the design of the study, the collection,
analysis and interpretation of data or the writing of the
manuscript.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
YC conceived the study, designed the research plan,
supervised and supported the study, verified data and statistical
analysis results, provided guidance with the writing, had full
access to all data in the study and took responsibility for the
integrity of the data and the accuracy of the data analysis. PM and
XT performed the study, collected and analyzed data, wrote and
revised the manuscript and contributed to the interpretation of
results. YW, SL, MW, QY, JS, BZ, JL, LiZ and LoZ followed up with
the participants, collected data and participated in data analysis
and interpretation. Additionally, they revised the manuscript. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
The current study was approved by the Ethics
Committee of the Third Affiliated Hospital of Sun Yat-sen
University, Guangzhou, P.R. China and written informed consent was
obtained from all subjects.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bartalena L, Pinchera A and Marcocci C:
Management of Graves' ophthalmopathy: Reality and perspectives.
Endocr Rev. 21:168–199. 2000.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Gao G, Dai J, Qian Y and Ma F:
Meta-analysis of methylprednisolone pulse therapy for Graves'
ophthalmopathy. Clin Exp Ophthalmol. 42:769–777. 2014.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Bartalena L, Baldeschi L, Dickinson AJ,
Eckstein A, Kendall-Taylor P, Marcocci C, Mourits MP, Perros P,
Boboridis K, Boschi A, et al: Consensus statement of the European
group on Graves' orbitopathy (EUGOGO) on management of Graves'
orbitopathy. Thyroid. 18:333–346. 2008.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Zhu W, Ye L, Shen L, Jiao Q, Huang F, Han
R, Zhang X, Wang S, Wang W and Ning G: A prospective, randomized
trial of intravenous glucocorticoids therapy with different
protocols for patients with Graves' ophthalmopathy. J Clin
Endocrinol Metab. 99:1999–2007. 2014.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Zang S, Ponto KA and Kahaly GJ: Clinical
review: Intravenous glucocorticoids for Graves' orbitopathy:
Efficacy and morbidity. J Clin Endocrinol Metab. 96:320–332.
2011.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Tsai CC, Kau HC, Kao SC and Hsu WM:
Exophthalmos of patients with Graves' disease in Chinese of Taiwan.
Eye (Lond). 20:569–573. 2006.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Mourits MP, Koornneef L, Wiersinga WM,
Prummel MF, Berghout A and van der Gaag R: Clinical criteria for
the assessment of disease activity in Graves' ophthalmopathy: A
novel approach. Br J Ophthalmol. 73:639–644. 1989.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Bartalena L, Marcocci C, Bogazzi F,
Manetti L, Tanda ML, Dell'Unto E, Bruno-Bossio G, Nardi M,
Bartolomei MP, Lepri A, et al: Relation between therapy for
hyperthyroidism and the course of Graves' ophthalmopathy. N Engl J
Med. 338:73–78. 1989.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Modjtahedi SP, Modjtahedi BS, Mansury AM,
Selva D, Douglas RS, Goldberg RA and Leibovitch I: Pharmacological
treatments for thyroid eye disease. Drugs. 66:1685–1700.
2006.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Marcocci C, Bartalena L, Tanda ML, Manetti
L, Dell'Unto E, Rocchi R, Barbesino G, Mazzi B, Bartolomei MP,
Lepri P, et al: Comparison of the effectiveness and tolerability of
intravenous or oral glucocorticoids associated with orbital
radiotherapy in the management of severe Graves' ophthalmopathy:
Results of a prospective, single-blind, randomized study. J Clin
Endocrinol Metab. 86:3562–3567. 2001.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Macchia PE, Bagattini M, Lupoli G, Vitale
M, Vitale G and Fenzi G: High-dose intravenous corticosteroid
therapy for Graves' ophthalmopathy. J Endocrinol Invest.
24:152–158. 2001.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Kauppinen-Makelin R, Karma A, Leinonen E,
Loyttyniemi E, Salonen O, Sane T, Setala K, Viikari J, Heufelder A
and Valimaki M: High dose intravenous methylprednisolone pulse
therapy versus oral prednisone for thyroid-associated
ophthalmopathy. Acta Ophthalmol Scand. 80:316–321. 2002.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Kahaly GJ, Pitz S, Hommel G and Dittmar M:
Randomized, single blind trial of intravenous versus oral steroid
monotherapy in Graves' orbitopathy. J Clin Endocrinol Metab.
90:5234–5240. 2005.PubMed/NCBI View Article : Google Scholar
|
|
14
|
van Geest RJ, Sasim IV, Koppeschaar HP,
Kalmann R, Stravers SN, Bijlsma WR and Mourits MP:
Methylprednisolone pulse therapy for patients with moderately
severe Graves' orbitopathy: A prospective, randomized,
placebo-controlled study. Eur J Endocrinol. 158:229–237.
2008.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Ng CM, Yuen HK, Choi KL, Chan MK, Yuen KT,
Ng YW and Tiu SC: Combined orbital irradiation and systemic
steroids compared with systemic steroids alone in the management of
moderate-to-severe Graves' ophthalmopathy: A preliminary study.
Hong Kong Med J. 11:322–330. 2005.PubMed/NCBI
|
|
16
|
Aktaran S, Akarsu E, Erbağci I, Araz M,
Okumus S and Kartal M: Comparison of intravenous methylprednisolone
therapy vs. oral methylprednisolone therapy in patients with
Graves' ophthalmopathy. Int J Clin Pract. 61:45–51. 2007.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Huang Y, Fang S, Li D, Zhou H, Li B and
Fan X: The involvement of T cell pathogenesis in thyroid-associated
ophthalmopathy. Eye (Lond). 33:176–182. 2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Khong JJ, McNab AA, Ebeling PR, Craig JE
and Selva D: Pathogenesis of thyroid eye disease: Review and update
on molecular mechanisms. Br J Ophthalmol. 100:142–150.
2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Gianoukakis AG, Khadavi N and Smith TJ:
Cytokines, Graves' disease, and thyroid-associated ophthalmopathy.
Thyroid. 18:953–958. 2008.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Douglas RS and Gupta S: The
pathophysiology of thyroid eye disease: Implications for
immunotherapy. Curr Opin Ophthalmol. 22:385–390. 2011.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Mills KH: Induction, function and
regulation of IL-17-producing T cells. Eur J Immunol. 38:2636–2649.
2008.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Ziolkowska M, Koc A, Luszczykiewicz G,
Ksiezopolska-Pietrzak K, Klimczak E, Chwalinska-Sadowska H and
Maslinski W: High levels of IL-17 in rheumatoid arthritis patients:
IL-15 triggers in vitro IL-17 production via cyclosporin
A-sensitive mechanism. J Immunol. 164:2832–2838. 2000.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Nalbandian A, Crispin JC and Tsokos GC:
Interleukin-17 and systemic lupus erythematosus: Current concepts.
Clin Exp Immunol. 157:209–215. 2009.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Yang D, Hiromatsu Y, Hoshino T, Inoue Y,
Itoh K and Nonaka K: Dominant infiltration of T(H)1-type CD4+ T
cells at the retrobulbar space of patients with thyroid-associated
ophthalmopathy. Thyroid. 9:305–310. 1999.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Aniszewski JP, Valyasevi RW and Bahn RS:
Relationship between disease duration and predominant orbital T
cell subset in Graves' ophthalmopathy. J Clin Endocrinol Metab.
85:776–780. 2000.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Wakelkamp IM, Bakker O, Baldeschi L,
Wiersinga WM and Prummel MF: TSH-R expression and cytokine profile
in orbital tissue of active vs. inactive Graves' ophthalmopathy
patients. Clin Endocrinol (Oxf). 58:280–287. 2003.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Hiromatsu Y, Kaku H, Miyake I, Murayama S
and Soejima E: Role of cytokines in the pathogenesis of
thyroid-associated ophthalmopathy. Thyroid. 12:217–221.
2002.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Avunduk AM, Avunduk MC, Pazarli H, Oguz V,
Varnell ED, Kaufman HE and Aksoy F: Immunohistochemical analysis of
orbital connective tissue specimens of patients with active Graves
ophthalmopathy. Curr Eye Res. 30:631–638. 2005.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Lacka K, Manuszewska E, Korczowska I and
Lacki JK: The effect of methylprednisolone pulse treatment on
cytokine network in Graves ophthalmopathy. Curr Eye Res.
32:291–297. 2007.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Esfahanian F, Naimi E, Doroodgar F and
Jadali Z: Th1/Th2 cytokines in patients with Graves' disease with
or without ophthalmopathy. Iran J Allergy Asthma Immunol.
12:168–175. 2013.PubMed/NCBI
|
|
31
|
Shen J, Li Z, Li W, Ge Y, Xie M, Lv M, Fan
Y, Chen Z, Zhao D and Han Y: Th1, th2, and th17 cytokine
involvement in thyroid associated ophthalmopathy. Dis Markers.
2015(609593)2015.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Kim SE, Yoon JS, Kim KH and Lee SY:
Increased serum interleukin-17 in Graves' ophthalmopathy. Graefes
Arch Clin Exp Ophthalmol. 250:1521–1526. 2012.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Peng D, Xu B, Wang Y, Guo H and Jiang Y: A
high frequency of circulating th22 and th17 cells in patients with
new onset Graves' disease. PLoS One. 8(e68446)2013.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Li JR, Hong FY, Zeng JY and Huang GL:
Functional interleukin-17 receptor A are present in the thyroid
gland in intractable Graves disease. Cell Immunol. 281:85–90.
2013.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Figueroa-Vega N, Alfonso-Perez M,
Benedicto I, Sanchez-Madrid F, Gonzalez-Amaro R and Marazuela M:
Increased circulating pro-inflammatory cytokines and Th17
lymphocytes in Hashimoto's thyroiditis. J Clin Endocrinol Metab.
95:953–962. 2010.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Horie I, Abiru N, Saitoh O, Ichikawa T,
Iwakura Y, Eguchi K and Nagayama Y: Distinct role of T helper Type
17 immune response for Graves' hyperthyroidism in mice with
different genetic backgrounds. Autoimmunity. 44:159–165.
2011.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Inoue N, Watanabe M, Nanba T, Wada M,
Akamizu T and Iwatani Y: Involvement of functional polymorphisms in
the TNFA gene in the pathogenesis of autoimmune thyroid diseases
and production of anti-thyrotropin receptor antibody. Clin Exp
Immunol. 156:199–204. 2009.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Bedoya SK, Lam B, Lau K and Larkin J III:
Th17 cells in immunity and autoimmunity. Clin Dev Immunol.
2013(986789)2013.PubMed/NCBI View Article : Google Scholar
|