According to the World Health Organization (WHO),
the number of hepatitis B virus (HBV) and hepatitis C virus (HCV)
infections worldwide in 2015 was 257 million and 71 million,
respectively (1,2). It is predicted that the number of
patients who develop complications such as liver cirrhosis and
hepatocellular carcinoma (HCC), due to persistent infections with
HBV and HCV, will peak in 2030-2035. Thus, the goal is to diagnose
90% and treat 80% of the HBV and HCV patients by 2030 in order to
eliminate this disease (3). HCC,
which accounts for the majority of primary liver cancers, is one of
the most common cancers worldwide and a leading cause of
cancer-related death (4). HCC rates
are the highest in East and South-East Asia and Northern and
Western Africa and lowest in South-Central Asia and Northern,
Central, and Eastern Europe (4).
Furthermore, HCC is the leading cause of death in patients with HCV
infection (5,6). Approximately 80% of HCCs result from
infections with HBV and HCV in Japan (7-10).
Hepatitis C and its complications are the leading causes of
HCC.
An interferon (IFN)-free oral agent called direct
antiviral agent (DAA) is the first choice of treatment for
effectively eliminating HCV (11).
DAA treatment has enabled many patients to achieve sustained
virologic response (SVR) with few side effects (12-15).
In addition, it reduces the incidence of chronic hepatitis C
(16) and HCC (17), and mortality (18). Only nine countries (Australia,
France, Iceland, Italy, Japan, South Korea, Spain, Switzerland, and
the United Kingdom) are on track toward eliminating HCV by 2030,
based on their current rates of diagnosis and treatment (19). Nucleotide analog therapy suppresses
HBV replication (20), and leads to
a reduction in both cirrhosis decompensation and HCC (21-23).
However, it is estimated that approximately 3
million people are infected with HBV and HCV in Japan; moreover,
approximately 780,000 people have latent infections without their
knowledge. Among the HCV-infected people, an estimated 470,000
people visit medical institutions, 300,000 people are unaware of
the fact that they are infected, and 250,000 to 750,000 people are
aware of the infection but not receiving treatment in the country
(24). In order to eradicate HCC,
it is necessary to improve the examination rate of the hepatitis
virus and encourage infected patients to receive treatment.
However, discrimination and prejudice against people
infected with hepatitis virus exist. A national survey involving
1,705 people with viral hepatitis revealed that dental clinics were
the most common places for discrimination against infected patients
(25). In a previous study, we
reported the prevalence of prejudice and discrimination among
HBV/HCV-infected individuals; of the 69 patients with viral liver
disease, 76.8% revealed a psychological burden and 20.3%
experienced prejudice and discrimination by healthcare workers
(26). Prejudice was most prevalent
within the dental clinic setting (50%). The presence or absence of
prejudice experienced by the patient from a healthcare worker was
found to be significantly associated with the psychological burden
(P=0.0255).
The dental clinic is often visited by patients with
underlying diseases. In Japan, the number of elderly patients who
undergo dental examination is increasing with the increase in age,
and at least one in three patients who undergo dental examinations
is over 65 years old (27).
Furthermore, a significant number of patients with HCV infection in
Japan are over the age of 65(28),
and a large proportion of them present with complications such as
cirrhosis or HCC (29).
Dentists need to have in-depth knowledge about liver
disease from the viewpoints of both nosocomial infection control
and treatment of oral diseases. Dentists often come in contact with
the patient's saliva and blood. Unlike a general medical office, a
dental office is often polluted by suspended matter from teeth and
prostheses. In addition, dentists frequently have accidents due to
punctures from sharp dental instruments and injection needles;
70.3% of dentists and 77.2% of dental hygienists and assistants are
reported to have been exposed to needle stick injuries in Japan
(30). Dentists are known to have
significantly higher HBV infection rates than other healthcare
workers (31). According to the
results of our survey in 2007, the positive rates of hepatitis B
core antibody (anti-HBc) among Japanese dentists increased with age
(85.7% in their 60s and 100% in their 70s; average rate, 12.1%)
(32). The coverage of the
hepatitis B vaccine was only 48.2%, and 25.4% of those who did not
receive the vaccine were positive for anti-HBc (32). These findings indicate that dentists
may be routinely exposed to HBV.
In 2017, an online survey involving members of the
Japanese Society of Dental Practice Administration found that 26.8%
of dentists were not vaccinated against hepatitis B (33). Statistical analyses showed that male
dentists had significantly higher scores for risk behavior of
infection control (P=0.002) and knowledge deficit scores of
hepatitis (P=0.031) than female dentists. Furthermore, general
dentists had significantly higher risk scores for infection control
(P<0.001) and knowledge deficit scores (P=0.014) than those
working in university hospitals.
It is impossible to detect the presence or absence
of infectious diseases in all patients in a general dental
practice. Therefore, it is important for the dentist to obtain
detailed and accurate medical histories from patients and take
appropriate precautions. We have previously reported that only
59.8% of patients with viral liver disease self-disclose their
infection at the dental clinic (38). The most common reason for not
disclosing was because the dentist did not enquire about the
condition of the liver. It is vital to pay attention to bleeding
tendencies associated with liver cirrhosis (39), drug administration, and
hypoalbuminemia (40,41) while providing dental treatment to
patients with liver disease.
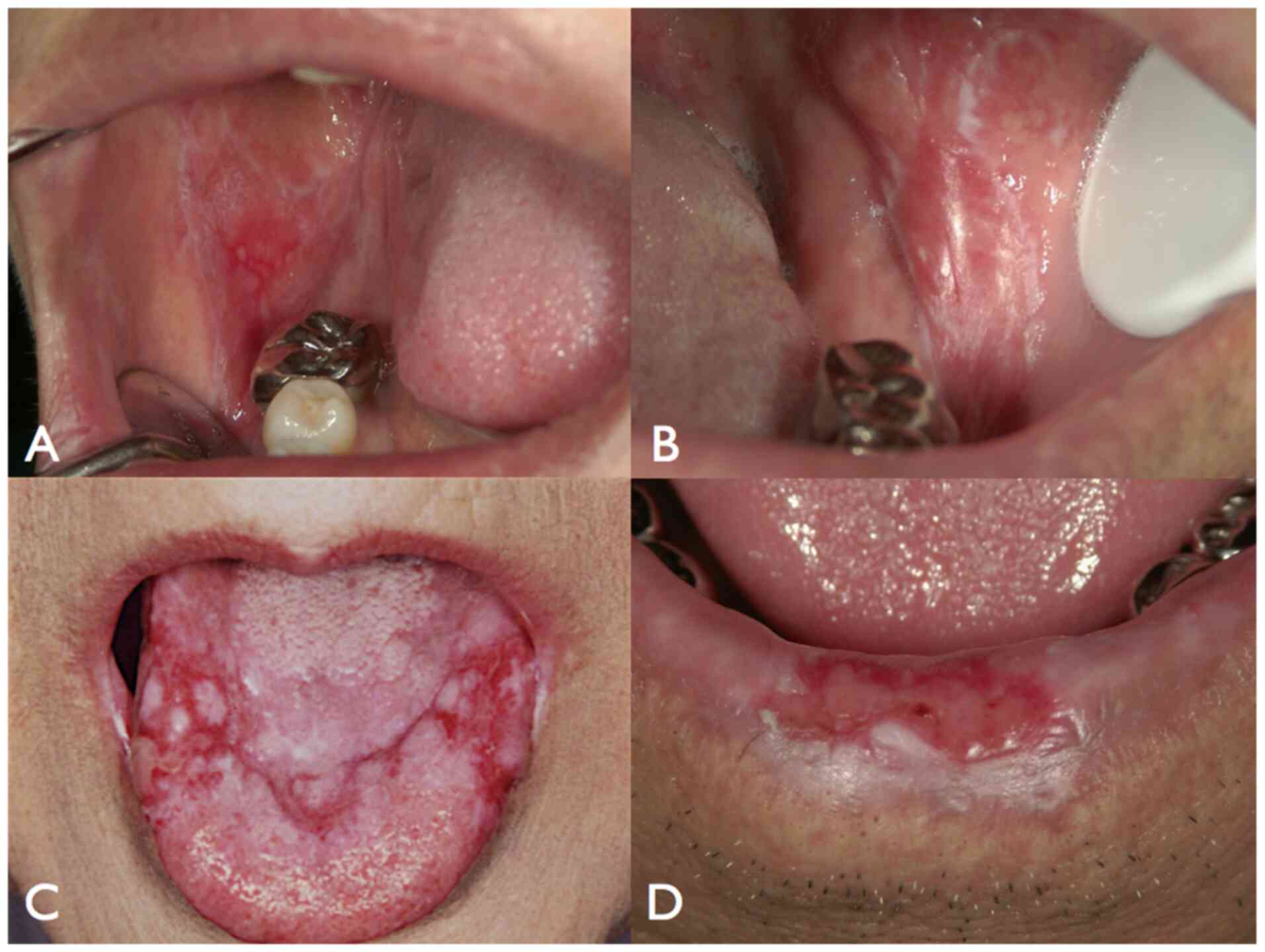
Oral lichen planus (OLP) is a T-cell mediated
chronic inflammatory mucosal disorder with different clinical
characteristics, and can be classified into six types as follows:
Reticular, popular, plaque, atrophic, erosive, and bullous
(42). It occurs predominantly in
adults over 40 years of age, and is characterized by the presence
of long-term, painful areas on the oral mucous membrane (Fig. 1).
In a previous study, we showed the histopathological
disappearance of OLP when HCV was eliminated by IFN therapy
(64). On the other hand, IFN
therapy may trigger the onset of OLP (65-67)
or sometimes act as an aggravating factor (66,68,69).
Recent studies have demonstrated improvements in OLP after the
elimination of HCV via DAA treatment (70-75).
Su et al (76) reported that
patients with chronic HCV infection had a significantly higher risk
of oral cancer than patients without HCV infection, and that
antiviral therapy with pegylated IFN (PegIFN)/ribavirin (RBV)
significantly reduced the risk of oral cancer. In addition to OLP
and oral cancer, SVR leads to substantial reductions in
extrahepatic manifestations such as cryoglobulinemia vasculitis,
malignant B-cell lymphoproliferative diseases, insulin resistance,
diabetes, mixed cryoglobulinemia, and glomerulonephritis (77,78).
The detection and proliferation of HCV in oral
tissues, including the normal oral mucosa and those affected by OLP
and oral cancer, have been reported previously (79-81).
However, virus detection is not a direct factor in the development
of OLP. The onset of OLP due to HCV depends on three factors: Viral
(82-85),
host (86), and drugs (69,70).
In Japan, various measures have been taken to
improve the coverage rate of the hepatitis virus test. According to
the Basic Guidelines on Hepatitis Measures, all citizens are
required to undergo hepatitis virus testing once in a lifetime.
However, the national testing rate for these viruses is only ~60%
(87). Despite the increase in the
eradication rate of HCV following the introduction of DAAs, issues
concerning the establishment of an efficient pick-up and follow-up
system for people infected with hepatitis virus remain unsolved. It
is challenging to improve the screening rate of the hepatitis virus
and build a follow-up system for hepatitis virus-positive
persons.
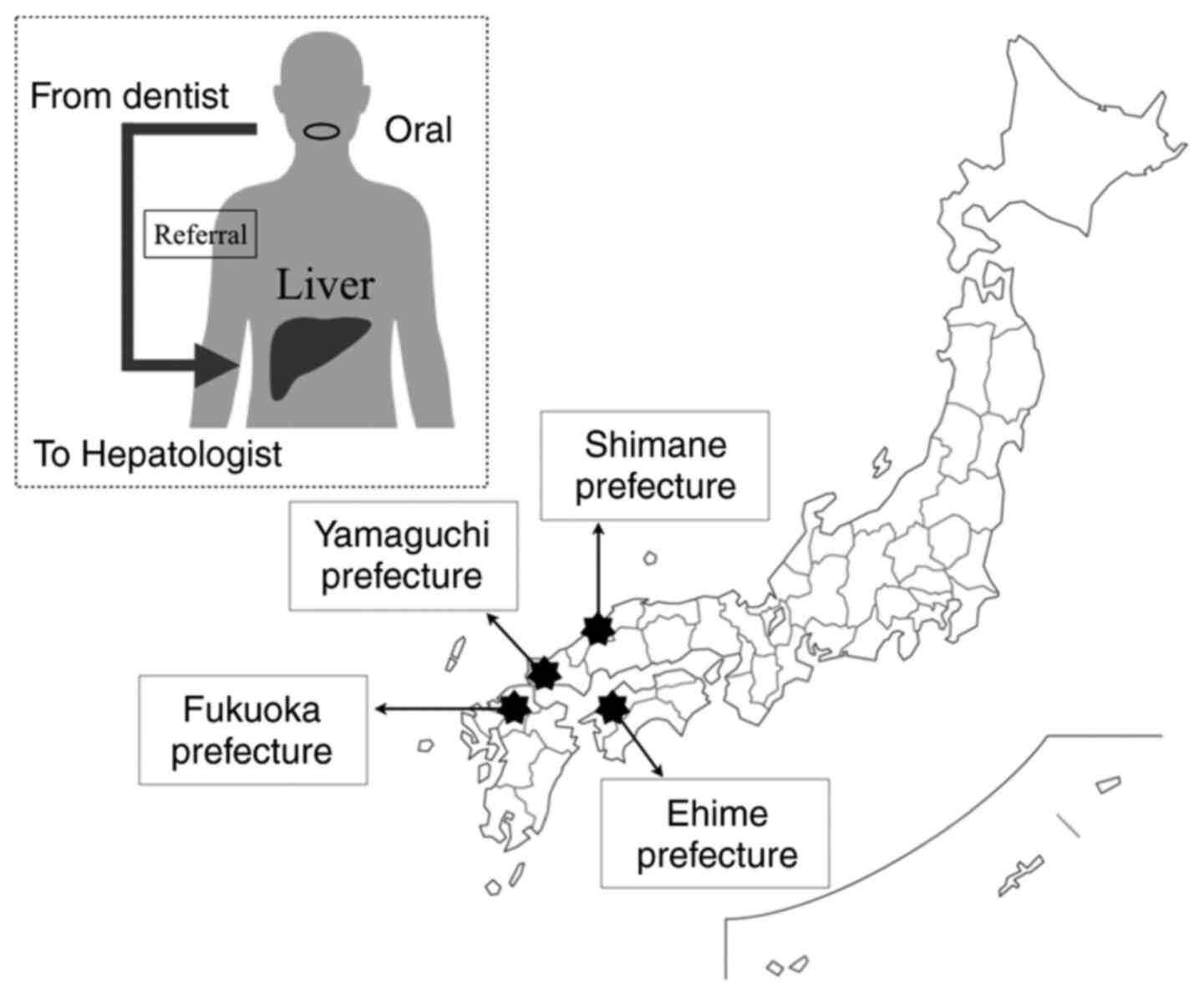
Several untreated hepatitis patients have been
identified during routine dental treatment in several parts of
Japan in retrospective and prospective studies (Fig. 2 and Table I) (74,88,89).
Ninety new patients who visited the T Dental Clinic
in Fukuoka prefecture from May 2015 to February 2017 for the
treatment of oral mucosal diseases (average age, 68.9 years ± 13.1
years) were included in a retrospective study (74). The main oral diseases were OLP (39
cases), leukoplakia (7 cases), head and neck cancer (4 cases), oral
candidiasis (8 cases) Sjögren's syndrome (4 cases), and taste
disorders (3 cases). Fifty-one (56.7%) of the 90 subjects were
referred to a hepatologist or a family doctor, by an oral surgeon,
to evaluate the liver disease for the presence or absence of HCV or
HBV infection. The prevalence of anti-HCV was 29.4% (15/51). The
oral surgeon was able to encourage 12 (80%) out of 15
anti-HCV-positive patients to undergo treatment for hepatitis. All
patients with HCV infection (and OLP-complications) who received
DAA treatment following referral from a dental clinic achieved SVR.
In Japan, general dentists do not perform blood tests due to issues
with medical insurance. Nonetheless, dentists can detect untreated
viral hepatitis patients among those with oral mucosal diseases or
a history of hepatitis in cooperation with the medical doctors.
We conducted prospective studies to determine
whether a general dentist could recommend hepatitis testing to
patients who visited a dental clinic (88,89).
Twelve clinics (n=5,091 patients), including 11 dental clinics in
Yamaguchi prefecture and one institution for oral surgery in a
hospital in Shimane prefecture, were included in the study by the
end of October 2017(88). The
number of patients who visited a dentist, the number of HCV-and
HBV-infected patients, the number of OLP patients, the rate of HCV
infection in OLP patients, and the number of patients referred to a
family doctor for consultation regarding liver diseases over a
period of 3 months were recorded. Consequently, 73% of OLP patients
were encouraged to undergo examinations for liver disease and
hepatitis virus infection. One patient was referred to the medical
department for untreated liver disease and achieved SVR with DAA
treatment.
In the same clinical study conducted at 35 dental
clinics in the Ehime prefecture (n=19,077 patients), 42 OLP, 69
HCV-infected, and 76-HBV-infected patients were identified over the
3-month period (89); 66.7% of the
OLP patients were recommended to undergo tests for liver disease
and 47.6% of the OLP patients were identified with the disease.
HCV-related liver disease was the most common finding (70%), and
78.6% of the patients had persistent HCV infection (P=0.0287).
Currently, general dentists are educating their patients about
hepatitis, and are promoting the referral of untreated hepatitis
patients to hepatologists in the Ehime prefecture.
Oral lesions are known to be associated with liver
disease. Thus, the dentist is expected to play a role as a
‘gatekeeper of hepatitis’. The promotion of the examination and
treatment of HBV and HCV infections by dentists aids in providing
appropriate treatment for the oral condition and in the reduction
of the incidence of liver cancer. If the number of people infected
with hepatitis virus decreases, the problems of discrimination and
prejudice will be resolved. Thus, in-depth knowledge about
hepatitis By the dentist and strong cooperation with the medical
department are required to promote these activities. In order to
increase the knowledge about hepatitis and the number of HB
vaccinations among dentists, it is necessary to conduct workshops
on the prevention of nosocomial infections, which should be
attended by dentists. Since April 2018, only those medical
institutions that meet the standards that stipulate nosocomial
infection control are permitted to calculate a new basic medical
examination fee in Japan. If the criteria are not met, the method
of subtraction is adopted. The standard requirements are as
follows: i) the establishment of an adequate system for the
replacement of each patient and the cleaning and sterilization of
the dental equipment using dedicated tools; ii) the establishment
of a system to support the dental treatment for patients with
infectious diseases; iii) the presence of at least one full-time
dentist who can regularly attend the outpatient training for
infection prevention measures every four years; iv) the provision
of in-hospital training, such as standard preventive measures for
the prevention of nosocomial infections, for employees; v) the
provision of information about the measures that need to be taken
to prevent nosocomial infection in the hospital; and vi) The
submission of a report about the implementation status of
nosocomial infection control to the directors of the Regional
Bureau Health and Welfare, once a year.
In Japan, the Dental National Health Association to
which dentists and their employees participate has a ‘Hepatitis B
Vaccination Subsidy Program’ that subsidizes the cost of HB
vaccination. In order to increase the number of HB vaccinations for
dentists, this grant system should be widely announced.
The structure of dental illness in Japan has changed
drastically due to improvements in oral hygiene and the aging
population; hence, there is an increasing need for dental care
depending on the patient's condition. Dentists must disseminate
interventions by acting as coordinators for HCV infection.
Not applicable.
The current study was supported in part by a
Grant-in-Aid for Scientific Research (C) (grant no. 17K12012) from
the Ministry of Education, Culture, Sports, Science and Technology
of Japan.
Not applicable.
YN conceived and designed the review, analyzed the
relevant literature and wrote the manuscript. YN critically revised
the manuscript and produced the figures. The author read and
approved the final manuscript.
Not applicable.
Informed consent for publication of the images was
obtained from the patient.
The author declares that they have no competing
interests.
|
1
|
WHO: Hepatitis B Fact sheet July 2019.
urihttp://www.who.int/mediacentre/factsheets/fs204/en/simplehttp://www.who.int/mediacentre/factsheets/fs204/en/.
|
|
2
|
WHO: Hepatitis C Fact sheet July 2019.
urihttp://www.who.int/mediacentre/factsheets/fs164/en/simplehttp://www.who.int/mediacentre/factsheets/fs164/en/.
|
|
3
|
WHO. Global Health Sector Strategies on
Viral Hepatitis, 2016-2021. urihttp://apps.who.int/gb/ebwha/pdf_files/WHA69/A69_32-en.pdf?ua=1simplehttp://apps.who.int/gb/ebwha/pdf_files/WHA69/A69_32-en.pdf?ua=1.
|
|
4
|
Torre LA, Bray F, Siegel RL, Ferlay J,
Lortet-Tieulent J and Jemal A: Global cancer statistics, 2012. CA
Cancer J Clin. 65:87–108. 2015.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Simonetti RG, Cammà C, Fiorello F, Cottone
M, Rapicetta M, Marino L, Fiorentino G, Craxì A, Ciccaglione A,
Giuseppetti R, et al: Hepatitis C virus infection as a risk factor
for hepatocellular carcinoma in patients with cirrhosis. A
case-control study. Ann Intern Med. 116:97–102. 1992.PubMed/NCBI View Article : Google Scholar
|
|
6
|
El-Serag HB: Epidemiology of viral
hepatitis and hepatocellular carcinoma. Gastroenterology.
142:1264–1273.e1. 2012.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Umemura T, Ichijo T, Yoshizawa K, Tanaka E
and Kiyosawa K: Epidemiology of hepatocellular carcinoma in Japan.
J Gastroenterol. 44 (Suppl 19):102–107. 2009.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Omata M, Kanda T, Wei L, Yu ML, Chuang WL,
Ibrahim A, Lesmana CR, Sollano J, Kumar M, Jindal A, Sharma BC, et
al: APASL consensus statements and recommendations for hepatitis C
prevention, epidemiology, and laboratory testing. Hepatol Int.
10:681–701. 2016.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Tanaka J, Akita T, Ohisa M, Sakamune K, Ko
K, Uchida S and Satake M: Trends in the total numbers of HBV and
HCV carriers in Japan from 2000 to 2011. J Viral Hepat. 25:363–372.
2018.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Tanaka J, Akita T, Ko K, Miura Y and
Satake M: Countermeasures against viral hepatitis B and C in Japan:
An epidemiological point of view. Hepatol Res. 49:990–1002.
2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Drafting Committee for Hepatitis
Management Guidelines, the Japan Society of Hepatology. Japan
Society of Hepatology guidelines for the management of hepatitis C
virus infection: 2019 update. Hepatol Res. 50:791–816.
2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Omata M, Nishiguchi S, Ueno Y, Mochizuki
H, Izumi N, Ikeda F, Toyoda H, Yokosuka O, Nirei K, Genda T, et al:
Sofosbuvir plus ribavirin in Japanese patients with chronic
genotype 2 HCV infection: An open-label, phase 3 trial. J Viral
Hepat. 21:762–768. 2014.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Kumada H, Suzuki Y, Ikeda K, Toyota J,
Karino Y, Chayama K, Kawakami Y, Ido A, Yamamoto K, Takaguchi K, et
al: Daclatasvir plus asunaprevir for chronic HCV genotype 1b
infection. Hepatology. 59:2083–2091. 2014.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Mizokami M, Yokosuka O, Takehara T,
Sakamoto N, Korenaga M, Mochizuki H, Nakane K, Enomoto H, Ikeda F,
Yanase M, et al: Ledipasvir and sofosbuvir fixed-dose combination
with and without ribavirin for 12 weeks in treatment-naive and
previously treated Japanese patients with genotype 1 hepatitis C:
An open-label, randomised, phase 3 trial. Lancet Infect Dis.
15:645–653. 2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Chayama K, Suzuki F, Karino Y, Kawakami Y,
Sato K, Atarashi T, Naganuma A, Watanabe T, Eguchi Y, Yoshiji H, et
al: Efficacy and safety of glecaprevir/pibrentasvir in Japanese
patients with chronic genotype 1 hepatitis C virus infection with
and without cirrhosis. J Gastroenterol. 53:557–565. 2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Juanbeltz R, Pérez-García A, Aguinaga A,
Martínez-Baz I, Casado I, Burgui C, Goñi-Esarte S, Repáraz J,
Zozaya JM, San Miguel R, et al: Progress in the elimination of
hepatitis C virus infection: A population-based cohort study in
Spain. PLoS One. 13(e0208554)2018.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Calvaruso V, Cabibbo G, Cacciola I, Petta
S, Madonia S, Bellia A, Tinè F, Distefano M, Licata A,
Giannitrapani L, et al: Incidence of hepatocellular carcinoma in
patients with HCV-associated cirrhosis treated with direct-acting
antiviral agents. Gastroenterology. 155:411–421.e4. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Carrat F, Fontaine H, Dorival C, Simony M,
Diallo A, Hezode C, De Ledinghen V, Larrey D, Haour G, Bronowicki
JP, et al: Clinical outcomes in patients with chronic hepatitis C
after direct-acting antiviral treatment: A prospective cohort
study. Lancet. 393:1453–1464. 2019.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Razavi H, Sanchez Gonzalez Y, Yuen C and
Cornberg M: Global timing of hepatitis C virus elimination in
high-income countries. Liver Int. 40:522–529. 2020.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Aghemo A, Lampertico P and Colombo M:
Assessing long-term treatment efficacy in chronic hepatitis B and
C: Between evidence and common sense. J Hepatol. 57:1326–1335.
2012.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Liaw YF, Sung JJ, Chow WC, Farrell G, Lee
CZ, Yuen H, Tanwandee T, Tao QM, Shue K, Keene ON, et al:
Lamivudine for patients with chronic hepatitis B and advanced liver
disease. N Engl J Med. 351:1521–1531. 2004.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Chang TT, Gish RG, de Man R, Gadano A,
Sollano J, Chao YC, Lok AS, Han KH, Goodman Z, Zhu J, et al: A
comparison of entecavir and lamivudine for HBeAg-positive chronic
hepatitis B. N Engl J Med. 354:1001–1010. 2006.PubMed/NCBI View Article : Google Scholar
|
|
23
|
European Association for the Study of the
Liver. Electronic address: simpleeasloffice@easloffice.eu;
European Association for the Study of the Liver. EASL 2017 Clinical
Practice Guidelines on the management of hepatitis B virus
infection. J Hepatol. 67:370–398. 2017.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Tanaka J: Ministry of Health, Labor and
Welfare 13th Council for Promotion of Measures against Hepatitis,
2015 (In Japanese) urihttps://http://www.mhlw.go.jp/stf/shingi2/0000075691.htmlsimplehttps://http://www.mhlw.go.jp/stf/shingi2/0000075691.html.
|
|
25
|
Tatsuoka S: Ministry of Health, Labor and
Welfare, Research project for practical application of medical
treatment in the field of diseases such as intractable diseases and
cancer (hepatitis-related research field) (In Japanese).
|
|
26
|
Nagao Y, Kawahigashi Y, Kimura K,
Nobayashi H and Sata M: Awareness survey of prejudice and
discrimination in hepatitis B and C virus-infected individuals. Adv
Res Gastroentero Hepatol. 7:6–11. 2017.
|
|
27
|
Ministry of Health, Labor and Welfare,
Study meeting for improving the quality of dentist 2017 (In
Japanese) urihttps://www.mhlw.go.jp/file/05-Shingikai-10801000-Iseikyoku-Soumuka/0000165542.pdfsimplehttps://http://www.mhlw.go.jp/file/05-Shingikai-10801000-Iseikyoku-Soumuka/0000165542.pdf.
|
|
28
|
Yatsuhashi H: Past, present, and future of
viral hepatitis C in Japan. Euroasian J Hepatogastroenterol.
6:49–51. 2016.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Toyoda H, Kumada T, Takaguchi K, Shimada N
and Tanaka J: Changes in hepatitis C virus genotype distribution in
Japan. Epidemiol Infect. 142:2624–2628. 2014.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Kobayashi K: Questionnaire study of
needlestick injures and hepatitis B vaccination in general dental
practices. Japanese J Environmental Inf. 30:348–353. 2015.(In
Japanese).
|
|
31
|
Olubuyide IO, Ola SO, Aliyu B, Dosumu OO,
Arotiba JT, Olaleye OA, Odaibo GN, Odemuyiwa SO and Olawuyi F:
Hepatitis B and C in doctors and dentists in Nigeria. QJM.
90:417–422. 1997.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Nagao Y, Matsuoka H, Kawaguchi T, Ide T
and Sata M: HBV and HCV infection in Japanese dental care workers.
Int J Mol Med. 21:791–799. 2008.PubMed/NCBI
|
|
33
|
Nagao Y, Ozaki T, Kawaguchi A, Chiba I,
Harada K, Saito T and Shiratsuchi S: Survey of dental health care
workers regarding their knowledge of viral liver disease and
prevention of its transmission, using an online questionnaire. Adv
Res Gastroentero Hepatol. 10:95–108. 2018.
|
|
34
|
Ali A, Khan S, Malik SM, Haris Iqbal M and
Aadil M: Comparison of knowledge and attitudes regarding hepatitis
B among healthcare professionals in Pakistan. Cureus.
9(e1049)2017.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Garus-Pakowska A, Górajski M and Szatko F:
Knowledge and attitudes of dentists with respect to the risks of
blood-borne pathogens-A cross-sectional study in Poland. Int J
Environ Res Public Health. 14(69)2017.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Gambhir RS, Kumar M, Singh G, Singh A and
Kaur A: Hepatitis C: Knowledge and awareness of private dental
practitioners of a tricity in India. J Educ Health Promot.
7(7)2018.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Rostamzadeh M, Afkhamzadeh A, Afrooz S,
Mohamadi K and Rasouli MA: Dentists' knowledge, attitudes and
practices regarding Hepatitis B and C and HIV/AIDS in Sanandaj,
Iran. BMC Oral Health. 18(220)2018.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Nagao Y, Kawaguchi T, Ide T and Sata M:
HCV or HBV infection self-disclosure to dentists. Kansenshogaku
Zasshi. 82:213–219. 2008.PubMed/NCBI View Article : Google Scholar : (In Japanese).
|
|
39
|
Medina JB, Andrade NS, de Paula Eduardo F,
Bezinelli L, Franco JB, Gallottini M, Braz-Silva PH and Ortega KL:
Bleeding during and after dental extractions in patients with liver
cirrhosis. Int J Oral Maxillofac Surg. 47:1543–1549.
2018.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Glassman P, Wong C and Gish R: A review of
liver transplantation for the dentist and guidelines for dental
management. Spec Care Dentist. 13:74–80. 1993.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Brigo S, Mancuso E and Pellicano R:
Dentistry and oral and maxillofacial surgery in the patient with
liver disease: Key messages for clinical practice. Minerva
Stomatol. 68:192–199. 2019.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Andreasen JO: Oral lichen planus. 1. A
clinical evaluation of 115 cases. Oral Surg Oral Med Oral Pathol.
25:31–42. 1968.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Pawlotsky JM, Ben Yahia M, Andre C, Voisin
MC, Intrator L, Roudot-Thoraval F, Deforges L, Duvoux C, Zafrani
ES, Duval J, et al: Immunological disorders in C virus chronic
active hepatitis: A prospective case-control study. Hepatology.
19:841–848. 1994.PubMed/NCBI
|
|
44
|
El-Serag HB, Hampel H, Yeh C and Rabeneck
L: Extrahepatic manifestations of hepatitis C among United States
male veterans. Hepatology. 36:1439–1445. 2002.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Negro F, Forton D, Craxì A, Sulkowski MS,
Feld JJ and Manns MP: Extrahepatic morbidity and mortality of
chronic hepatitis C. Gastroenterology. 149:1345–1360.
2015.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Ferri C, Ramos-Casals M, Zignego AL,
Arcaini L, Roccatello D, Antonelli A, Saadoun D, Desbois AC,
Sebastiani M, Casato M, et al: International diagnostic guidelines
for patients with HCV-related extrahepatic manifestations. A
multidisciplinary expert statement. Autoimmun Rev. 15:1145–1160.
2016.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Cacoub P, Poynard T, Ghillani P, Charlotte
F, Olivi M, Piette JC and Opolon P: Extrahepatic manifestations of
chronic hepatitis C. MULTIVIRC Group. Multidepartment Virus C.
Arthritis Rheum. 42:2204–2212. 1999.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Cacoub P, Renou C, Rosenthal E, Cohen P,
Loury I, Loustaud-Ratti V, Yamamoto AM, Camproux AC, Hausfater P,
Musset L, et al: Extrahepatic manifestations associated with
hepatitis C virus infection. A prospective multicenter study of 321
patients. The GERMIVIC. Groupe d'Etude et de Recherche en Medecine
Interne et Maladies Infectieuses sur le Virus de l'Hepatite C.
Medicine (Baltimore). 79:47–56. 2000.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Stefanova-Petrova DV, Tzvetanska AH,
Naumova EJ, Mihailova AP, Hadjiev EA, Dikova RP, Vukov MI and
Tchernev KG: Chronic hepatitis C virus infection: Prevalence of
extrahepatic manifestations and association with cryoglobulinemia
in Bulgarian patients. World J Gastroenterol. 13:6518–6528.
2007.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Nagao Y, Sata M, Tanikawa K, Itoh K and
Kameyama T: Lichen planus and hepatitis C virus in the northern
Kyushu region of Japan. Eur J Clin Invest. 25:910–914.
1995.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Carrozzo M, Gandolfo S, Carbone M,
Colombatto P, Broccoletti R, Garzino-Demo P and Ghisetti V:
Hepatitis C virus infection in Italian patients with oral lichen
planus: A prospective case-control study. J Oral Pathol Med.
25:527–533. 1996.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Haddad J, Deny P, Munz-Gotheil C,
Ambrosini JC, Trinchet JC, Pateron D, Mal F, Callard P and
Beaugrand M: Lymphocytic sialadenitis of Sjögren's syndrome
associated with chronic hepatitis C virus liver disease. Lancet.
339:321–323. 1992.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Koike K, Moriya K, Ishibashi K,
Yotsuyanagi H, Shintani Y, Fujie H, Kurokawa K, Matsuura Y and
Miyamura T: Sialadenitis histologically resembling Sjögren syndrome
in mice transgenic for hepatitis C virus envelope genes. Proc Natl
Acad Sci USA. 94:233–236. 1997.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Nagao Y, Hanada S, Shishido S, Ide T,
Kumashiro R, Ueno T and Sata M: Incidence of Sjögren's syndrome in
Japanese patients with hepatitis C virus infection. J Gastroenterol
Hepatol. 18:258–266. 2003.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Nagao Y, Sata M, Tanikawa K, Itoh K and
Kameyama T: High prevalence of hepatitis C virus antibody and RNA
in patients with oral cancer. J Oral Pathol Med. 24:354–360.
1995.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Mahale P, Sturgis EM, Tweardy DJ,
Ariza-Heredia EJ and Torres HA: Association between hepatitis C
virus and head and neck cancers. J Natl Cancer Inst.
108(djw035)2016.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Nagao Y and Sata M: Hepatitis C virus and
lichen planus. J Gastroenterol Hepatol. 19:1101–1113.
2004.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Carrozzo M: Oral diseases associated with
hepatitis C virus infection. Part 2: Lichen planus and other
diseases. Oral Dis. 14:217–228. 2008.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Shengyuan L, Songpo Y, Wen W, Wenjing T,
Haitao Z and Binyou W: Hepatitis C virus and lichen planus: A
reciprocal association determined by a meta-analysis. Arch
Dermatol. 145:1040–1047. 2009.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Lodi G, Pellicano R and Carrozzo M:
Hepatitis C virus infection and lichen planus: A systematic review
with meta-analysis. Oral Dis. 16:601–612. 2010.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Petti S, Rabiei M, De Luca M and Scully C:
The magnitude of the association between hepatitis C virus
infection and oral lichen planus: Meta-analysis and case control
study. Odontology. 99:168–178. 2011.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Alaizari NA, Al-Maweri SA, Al-Shamiri HM,
Tarakji B and Shugaa-Addin B: Hepatitis C virus infections in oral
lichen planus: A systematic review and meta-analysis. Aust Dent J.
61:282–287. 2016.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Gandolfo S, Richiardi L, Carrozzo M,
Broccoletti R, Carbone M, Pagano M, Vestita C, Rosso S and Merletti
F: Risk of oral squamous cell carcinoma in 402 patients with oral
lichen planus: A follow-up study in an Italian population. Oral
Oncol. 40:77–83. 2004.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Nagao Y, Sata M, Suzuki H, Kameyama T and
Ueno T: Histological improvement of oral lichen planus in patients
with chronic hepatitis C treated with interferon. Gastroenterology.
117:283–284. 1999.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Núñez M, Miralles ES, de las Heras ME and
Ledo A: Appearance of oral erosive lichen planus during interferon
alfa-2a therapy for chronic active hepatitis C. J Dermatol.
22:461–462. 1995.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Nagao Y, Sata M, Ide T, Suzuki H, Tanikawa
K, Itoh K and Kameyama T: Development and exacerbation of oral
lichen planus during and after interferon therapy for hepatitis C.
Eur J Clin Invest. 26:1171–1174. 1996.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Varela P, Areias J, Mota F, Canelhas A and
Sanches M: Oral lichen planus induced by interferon-alpha-N1 in a
patient with hepatitis C. Int J Dermatol. 39:239–240.
2000.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Areias J, Velho GC, Cerqueira R, Barbêdo
C, Amaral B, Sanches M, Massa A and Saraiva AM: Lichen planus and
chronic hepatitis C: Exacerbation of the lichen under
interferon-alpha-2a therapy. Eur J Gastroenterol Hepatol.
8:825–828. 1996.PubMed/NCBI
|
|
69
|
Nagao Y, Kawaguchi T, Ide T, Kumashiro R
and Sata M: Exacerbation of oral erosive lichen planus by
combination of interferon and ribavirin therapy for chronic
hepatitis C. Int J Mol Med. 15:237–241. 2005.PubMed/NCBI
|
|
70
|
Nagao Y, Kimura K, Kawahigashi Y and Sata
M: Successful treatment of hepatitis C virus-associated oral lichen
planus by interferon-free therapy with direct-acting antivirals.
Clin Transl Gastroenterol. 7(e179)2016.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Misaka K, Kishimoto T, Kawahigashi Y, Sata
M and Nagao Y: Use of direct-acting antivirals for the treatment of
hepatitis C virus-associated oral lichen planus: A case report.
Case Rep Gastroenterol. 10:617–622. 2016.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Nagao Y, Nakasone K, Maeshiro T, Nishida
N, Kimura K, Kawahigashi Y, Tanaka Y and Sata M: Successful
treatment of oral lichen planus with direct-acting antiviral agents
after liver transplantation for hepatitis C virus-associated
hepatocellular carcinoma. Case Rep Gastroenterol. 11:701–710.
2017.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Yoshikawa A, Terashita K, Morikawa K,
Matsuda S, Yamamura T, Sarashina K, Nakano S, Kobayashi Y, Sogabe
S, Takahashi K, et al: Interferon-free therapy with sofosbuvir plus
ribavirin for successful treatment of genotype 2 hepatitis C virus
with lichen planus: A case report. Clin J Gastroenterol.
10:270–273. 2017.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Nagao Y and Tsuji M: The discovery through
dentistry of potentially HCV-infected Japanese patients and
intervention with treatment. Adv Res Gastroentero Hepatol. 7:38–44.
2017.
|
|
75
|
Harada K, Nakashima D, Nagao Y, Hidaka I,
Sakaida I and Mishima K: Treatment of refractory oral lichen planus
using direct antiviral agents in a patient with chronic hepatitis
C: A case report. Oral Sci Int. 17:213–217. 2020.
|
|
76
|
Su TH, Tseng TC, Liu CJ, Chou SW, Liu CH,
Yang HC, Chen PJ, Chen DS, Chen CL and Kao JH: Antiviral therapy
against chronic hepatitis C is associated with a reduced risk of
oral cancer. Int J Cancer. 147:901–908. 2020.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Cacoub P, Desbois AC, Comarmond C and
Saadoun D: Impact of sustained virological response on the
extrahepatic manifestations of chronic hepatitis C: A
meta-analysis. Gut. 67:2025–2034. 2018.PubMed/NCBI View Article : Google Scholar
|
|
78
|
El-Serag HB, Christie IC, Puenpatom A,
Castillo D, Kanwal F and Kramer JR: The effects of sustained
virological response to direct-acting anti-viral therapy on the
risk of extrahepatic manifestations of hepatitis C infection.
Aliment Pharmacol Ther. 49:1442–1447. 2019.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Nagao Y, Kameyama T and Sata M: Hepatitis
C virus RNA detection in oral lichen planus tissue. Am J
Gastroenterol. 93(850)1998.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Nagao Y, Sata M, Noguchi S, Seno'o T,
Kinoshita M, Kameyama T and Ueno T: Detection of hepatitis C virus
RNA in oral lichen planus and oral cancer tissues. J Oral Pathol
Med. 29:259–266. 2000.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Arrieta JJ, Rodriguez-Inigo E, Casqueiro
M, Bartolomé J, Manzarbeitia F, Herrero M, Pardo M and Carreno V:
Detection of hepatitis C virus replication by In situ hybridization
in epithelial cells of anti-hepatitis C virus-positive patients
with and without oral lichen planus. Hepatology. 32:97–103.
2000.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Nagao Y, Sata M, Itoh K, Tanikawa K and
Kameyama T: Quantitative analysis of HCV RNA and genotype in
patients with chronic hepatitis C accompanied by oral lichen
planus. Eur J Clin Invest. 26:495–498. 1996.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Lodi G, Carrozzo M, Hallett R, D'Amico E,
Piattelli A, Teo CG, Gandolfo S, Carbone M and Porter SR: HCV
genotypes in Italian patients with HCV-related oral lichen planus.
J Oral Pathol Med. 26:381–384. 1997.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Sánchez-Pérez J, Moreno-Otero R, Borque
MJ, Rios-Buceta L and García-Díez A: Lichen planus and hepatitis C
virus infection: A clinical and virologic study. Acta Derm
Venereol. 78:305–306. 1998.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Nagao Y and Sata M: A retrospective
case-control study of hepatitis C virus infection and oral lichen
planus in Japan: Association study with mutations in the core and
NS5A region of hepatitis C virus. BMC Gastroenterol.
12(31)2012.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Nagao Y, Nishida N, Toyo-Oka L, Kawaguchi
A, Amoroso A, Carrozzo M, Sata M, Mizokami M, Tokunaga K and Tanaka
Y: Genome-wide association study identifies risk variants for
lichen planus in patients with hepatitis C virus infection. Clin
Gastroenterol Hepatol. 15:937–944.e5. 2017.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Kaishima T, Fujii T, Matsuoka T, Sakamune
K, Nagashima S, Yamamoto C, Chuon C, et al: Study of the issues of
receiving hepatitis screening and the rate of consulting
hospitals-The rate of recognized receiving hepatitis screening and
that of the unrecognized. Kanzo. 57:634–648. 2016.(In
Japanese).
|
|
88
|
Nagao Y, Sasaki T, Kuzuyama T, Takasato T,
Yoshikawa S, Konishi H, Oka K, Yamaguchi T, Konishi Y, Hanada H, et
al: Promotion by dentists of treatment of undiagnosed and untreated
HCV-infected patients. Adv Res Gastroentero Hepatol. 9:86–90.
2018.
|
|
89
|
Nagao Y, Nishioka S and Koresawa K:
Prevalence of viral liver disease and oral lichen planus in
patients who visited dental clinics: A study by the Ehime Dental
Association. OBM Hepatology Gastroenterology. 3:1–10. 2019.
|