Introduction
Coronavirus disease 2019 (COVID-19) has spread
worldwide and has seriously threatened human health and the global
social and economic order. Although China has taken effective
measures to control the epidemic, the increasing number of imported
cases and asymptomatic carriers make prevention and control
measures, such as some air travel restrictions, even more necessary
(1,2). In general, the clinical manifestations
of COVID-19 include fever (3),
fatigue (4), cough (5), sore throat (6), headache (7) and joint pain (8). In severe cases, breathing
difficulties, chest pain and even acute heart injury may occur. The
absolute number of red blood cells (RBC), white blood cells (WBC),
lymphocytes (LYM) and neutrophils (NEU), as well as the percentage
of NEU (NEU%) and LYM (LYM%) and the serum levels of interleukin-6
(IL-6) and C-reactive protein (CRP) are considered as important
indicators of inflammation, reflecting the inflammatory state of
patients with COVID-19. In addition, lactate dehydrogenase (LDH),
creatine kinase (CK) and CK isoenzyme (CK-Mb) are crucial
biochemical markers of heart damage. Therefore, increased levels of
myocardial enzymes indicate that the patient's condition is severe.
Patients with COVID-19 may also exhibit impaired liver function,
which may be indicated by the levels of alkaline phosphatase (ALP).
Therefore, at Tianjin Haihe Hospital (Tianjin, China), the
aforementioned indicators are mainly detected.
In the present study, blood parameters, IL-6 levels,
neutrophil-to-lymphocyte ratio (NLR), CRP and kinase levels, liver
function and other routine biochemical test results from patients
with COVID-19 were recorded in order to evaluate their diagnostic
efficacy and association with COVID-19. Thereby, the present study
aimed to provide a reference for the early clinical diagnosis and
treatment of patients with COVID-19.
Materials and methods
Patients
The present study retrospectively analyzed 84 cases
of confirmed COVID-19, with 30 healthy individuals included as the
control group. Patients with COVID-19 tested positive for severe
acute respiratory syndrome coronavirus 2 (SARS-CoV-2) nucleic acid
at Tianjin Haihe Hospital (Tianjin, China) between January and
February 2020, according to real-time fluorescent quantitative PCR
detection of throat swab specimens. The diagnostic criteria and
clinical classification were based on the ‘Diagnosis and Treatment
Plan for COVID-19’ (fifth trial version) (9). Newly diagnosed and untreated patients
were included in the patient group, while cured patients with no
recurrence of symptoms at the time-point of discharge were included
in the cured group. The baseline characteristics of patients and
healthy individuals are listed in Table
I. In brief, 37 male and 47 female patients, aged 21-91 years
(median age, 51 years) were enrolled in the patient group.
According to the clinical classification of COVID-19, 61 cases were
of the common type and 23 were severe. In the voluntary healthy
group, 14 males and 16 females aged 22-80 years (median age, 51
years) were included. There were no statistically significant
differences in the clinical characteristics between the healthy and
patient groups. This study was approved by the Ethics Committee of
the Tianjin Haihe Hospital (Tianjin, China) and patients provided
their written informed consent for participating in the current
study.
 | Table IBaseline characteristics of patients
and healthy individuals (). |
Table I
Baseline characteristics of patients
and healthy individuals ().
| Variable | Patients with
COVID-19 (n=84) | Healthy controls
(n=30) |
|---|
| Age (median,
years) | 51 (21-91) | 51 (22-80) |
| Sex, n (%) | | |
|
Males | 37(44) | 14(47) |
|
Females | 47(56) | 16(53) |
Reagents and instruments
The automatic biochemical immunoanalyzer VITROS 5600
with clinical chemistry rapid quantitative multilayer film
technology and the CK (cat. no. 8749396), CK-Mb (cat. no. 8058232),
LDH (cat. no. 8384489) and ALP detection kits (cat. no. 65240528)
were purchased from Ortho-Clinical Diagnostics, Inc. The Pylon3D
cycle enhanced fluorescence immunoassay analyzer with cyclic
enhanced fluorescence immunoassay technology and the CRP (cat. no.
200057) and IL-6 (cat. no. 200107) detection kits were obtained
from Xingtong Medical Technology Co., Ltd. The XT-1800i Automated
Hematology Analyzer with electrical impedance method was purchased
from Sysmex Co.
Laboratory examination
Venous blood samples (5 ml) were collected from all
subjects and centrifuged at 2,000 x g for 10 min at room
temperature. Following centrifugation, the serum was collected and
used for the detection of IL-6, CK, CK-Mb, CRP, ALP and LDH, while
whole-blood samples were used for blood analyses and detection of
LYM, LYM%, NEU and NEU%. All specimens were detected within 2 h of
acquisition.
Statistical analysis
All statistical analyses were performed using SPSS
23.0 software (IBM Corp.). Continuous variables are expressed as
the mean ± standard deviation. Comparisons between two groups were
performed using unpaired Student's t-tests, while those among
multiple groups were determined with one-way ANOVA. In addition,
multiple comparisons between two groups were carried out using
Tukey's post hoc test. Count data are expressed as n (%) and
comparisons of count data between two groups were performed with a
χ2 test. Non-normally distributed data are presented as
the median and interquartile range and the Wilcoxon rank-sum test
was utilized for comparisons between groups. Furthermore, receiver
operating characteristic curves (ROC) were constructed and the area
under the curve (AUC) and the best cut-off values were calculated
using GraphPad Prism 6.0 software (GraphPad Software, Inc.).
Pearson's correlation analysis was used to analyze the correlation
between variables, with |r|>0.3 indicating a significant
correlation. Finally, P<0.05 was considered to indicate a
statistically significant difference. The cut-off value for common
COVID-19 was 0.42, according to the binary regression equation
logistic (P)=-5.92 x LYM + 0.004 x CK + 10.46; and the cut-off
value for the severe type was 0.50, according to P=-145.78 x LYM +
2.16 x CK + 109.77.
Results
Clinical features of patients with
COVID-19
Patients with COVID-19 were admitted to Tianjin
Haihe Hospital (Tianjin, China) and underwent routine hematological
and biochemical tests. In addition, the serum levels of IL-6 were
determined. The values of the aforementioned parameters in patients
with common and severe COVID-19 are presented in Table II. There were different normal
reference values for red blood cells (RBC) and CK between males and
females.
 | Table IIChanges in clinical parameters in
patients with coronavirus disease 2019. |
Table II
Changes in clinical parameters in
patients with coronavirus disease 2019.
| | Common group
(n=61) | Severe group
(n=23) | |
|---|
| Variable | Increased | Normal | Decreased | Increased | Normal | Decreased | Normal reference
value range |
|---|
| RBC
(1012/l) |
|
Males | 1 (4.35) | 18 (78.26) | 4 (17.39) | 0 (0) | 11 (78.57) | 3 (21.43) | 4.0-5.5 |
|
Females | 0 (0) | 36 (94.74) | 2 (5.26) | 1 (11.11) | 8 (88.89) | 0 (0) | 3.5-5.0 |
| WBC
(109/l) | 2 (3.28) | 40 (65.57) | 19 (31.15) | 1 (4.35) | 13 (56.52) | 9 (39.13) | 4-10 |
| LYM
(109/l) | 0 (0) | 45 (73.77) | 16 (26.23) | 0 (0) | 11 (47.83) | 12 (52.17) | 0.8-4.0 |
| LYM% | 2 (3.28) | 36 (59.02) | 23 (37.7) | 0 (0) | 9 (39.13) | 14 (60.87) | 0.2-0.4 |
| NEU
(109/l) | 4 (6.56) | 48 (78.69) | 9 (14.75) | 1 (4.35) | 18 (78.26) | 4 (17.39) | 2-7.5 |
| NEU% | 12 (19.67) | 45 (73.77) | 4 (6.56) | 10 (43.48) | 13 (56.52) | 0 (0) | 0.5-0.75 |
| IL-6 (pg/ml) | 22 (36.07) | 38 (62.3) | 1 (1.64) | 19 (82.61) | 4 (17.39) | 0 (0) | 1-10 |
| CRP (mg/l) | 22 (36.07) | 32 (52.46) | 7 (11.48) | 17 (73.91) | 6 (26.09) | 0 (0) | 1-10 |
| CK (U/l) |
|
Males | 3 (13.04) | 14 (60.87) | 6 (26.09) | 4 (28.57) | 9 (64.29) | 1 (7.14) | 55-170 |
|
Females | 5 (13.16) | 28 (73.68) | 5 (13.16) | 3 (33.33) | 6 (66.67) | 0 (0) | 30-135 |
| CK-Mb (U/l) | 1 (1.64) | 60 (98.36) | 0 (0) | 2 (8.7) | 21 (91.3) | 0 (0) | 0-25 |
| ALP (U/l) | 4 (6.56) | 55 (90.16) | 2 (3.28) | 5 (21.74) | 17 (73.91) | 1 (4.35) | 38-126 |
| LDH (U/l) | 16 (26.23) | 41 (67.21) | 4 (6.56) | 11 (47.83) | 8 (34.78) | 4 (17.39) | 313-618 |
Comparison of clinical parameters
between different types of COVID-19
The results demonstrated that compared with the
healthy group, NEU% was significantly increased, while WBC, LYM and
LYM% were notably decreased in the patient group. The serum levels
of CK-Mb were significantly reduced after treatment. However,
increased ALP levels were observed in the cured group compared with
the healthy group, indicating COVID-19-induced hysteretic liver
injury (Table III). RBC (female),
NEU% and NLR, as well as IL-6 and CRP levels in patients with
severe disease were significantly higher compared with those in
patients with the common type of COVID-19. In addition, LYM and
LYM% in the severe group were significantly lower than those in the
group with the common type. The age and LDH levels in patients with
severe COVID-19 were higher compared with those with common
COVID-19; however, the differences were not statistically
significant (P>0.05; Table
IV).
 | Table IIIComparison of clinical parameters in
patients with coronavirus disease 2019. |
Table III
Comparison of clinical parameters in
patients with coronavirus disease 2019.
| Variable | Healthy group
(n=30) | Disease group
(n=84) | Cured group
(n=84) | F-value |
|---|
| RBC
(1012/l) |
|
Males | 4.88 (4.57,
4.98) | 4.46 (4.05,
4.71) | 4.08 (3.78,
4.54) | 13.34 |
|
Females | 4.43 (4.24,
4.57) | 3.96 (3.89,
4.24) | 3.86 (3.43,
4.02) | 18.01 |
| WBC
(109/l) | 6 (5.46, 6.93) | 4.61 (3.67,
5.74)a | 5.6 (4.90,
6.55)b | 9.86 |
| LYM
(109/l) | 2.15 (1.93,
2.41) | 0.99 (0.67,
1.28)a | 1.46 (1.18,
1.76)a,b | 63.11 |
| LYM% | 0.36 (0.32,
0.40) | 0.22 (0.16,
0.29)a | 0.27 (0.20,
0.31)a,b | 32.25 |
| NEU
(109/l) | 3.39 (2.74,
4.01) | 2.89 (2.32,
4.00) | 3.4 (3.00,
4.09) | 0.92 |
| NEU% | 0.56 (0.48,
0.58) | 0.67 (0.61,
0.75)a | 0.62 (0.56,
0.69)a,b | 19.54 |
| NLR | 1.61 (1.21,
1.78) | 3.01 (2.10,
4.88) | 2.26 (1.79,
3.42) | 1.181 |
| IL-6 (pg/ml) | 2.00 (1.50,
2.63) | 9.47 (2.81,
29.8)a | 3.9 (1.50,
9.60)a,b | 18.30 |
| CRP (mg/l) | 1.07 (0.52,
1.65) | 9.78 (3.45,
36.92)a | 2.72 (1.18,
5.59)a,b | 15.58 |
| CK (U/l) |
|
Males | 51 (43, 61) | 84 (64,
121)a | 60 (44, 78) | 3.63 |
|
Females | 43 (34, 50) | 50 (36.5,
75)a | 45 (34, 57) | 2.28 |
| CK-Mb (U/l) | 6 (6, 8) | 7.5 (5,
11)a | 6 (4,
9)b | 2.60 |
| ALP (U/l) | 55 (47, 61) | 59 (51,
75)a | 77 (61,
92)a | 4.74 |
| LDH (U/l) | 485.0 (453.1,
542.5) | 505.5 (416.5,
638.5) | 484.5 (411.8,
538.3) | 1.02 |
 | Table IVComparison of clinical parameters in
patients with different types of coronavirus disease 2019
(n=84). |
Table IV
Comparison of clinical parameters in
patients with different types of coronavirus disease 2019
(n=84).
| Variable | Common group | Severe group |
P-valuea |
|---|
| Age (years) | 48 (36, 63) | 56 (44, 64) | 0.3442 |
| RBC
(1012/l) |
|
Males | 4.1 (3.94,
4.54) | 4.27 (4.00,
4.45) | 0.2875 |
|
Females | 4.03 (3.88,
4.47) | 4.43 (4.27,
4.70) | 0.0326 |
| WBC
(109/l) | 4.62 (3.84,
5.78) | 4.29 (3.47,
5.36) | 0.2354 |
| LYM
(109/l) | 1.07 (0.79,
1.41) | 0.77 (0.56,
1.10) | 0.0024 |
| LYM% | 0.24 (0.18,
0.29) | 0.18 (0.13,
0.24) | 0.0440 |
| NEU
(109/l) | 2.89 (2.32,
4.08) | 2.92 (2.35,
3.95) | 0.8130 |
| NEU% | 0.64 (0.59,
0.74) | 0.74 (0.66,
0.77) | 0.0144 |
| NLR | 2.69 (2.07,
4.08) | 4.26 (2.81,
5.89) | 0.0280 |
| IL-6 (pg/ml) | 7.3 (2.00,
18.9) | 30.8 (14.75,
51.90) | 0.0002 |
| CRP (mg/l) | 7.21 (2.84,
21.1) | 40.2 (13.11,
54.90) | <0.0001 |
| CK (U/l) |
|
Males | 75 (54.5, 130) | 84.5 (64.25,
155) | 0.0957 |
|
Females | 50 (32, 71) | 99 (60, 179) | 0.0847 |
| CK-Mb (U/l) | 8 (6, 11) | 6 (4, 10) | 0.2126 |
| ALP (U/l) | 59 (51, 73) | 57(51.5, 98) | 0.6025 |
| LDH (U/l) | 498 (415, 619) | 618 (433, 772) | 0.1635 |
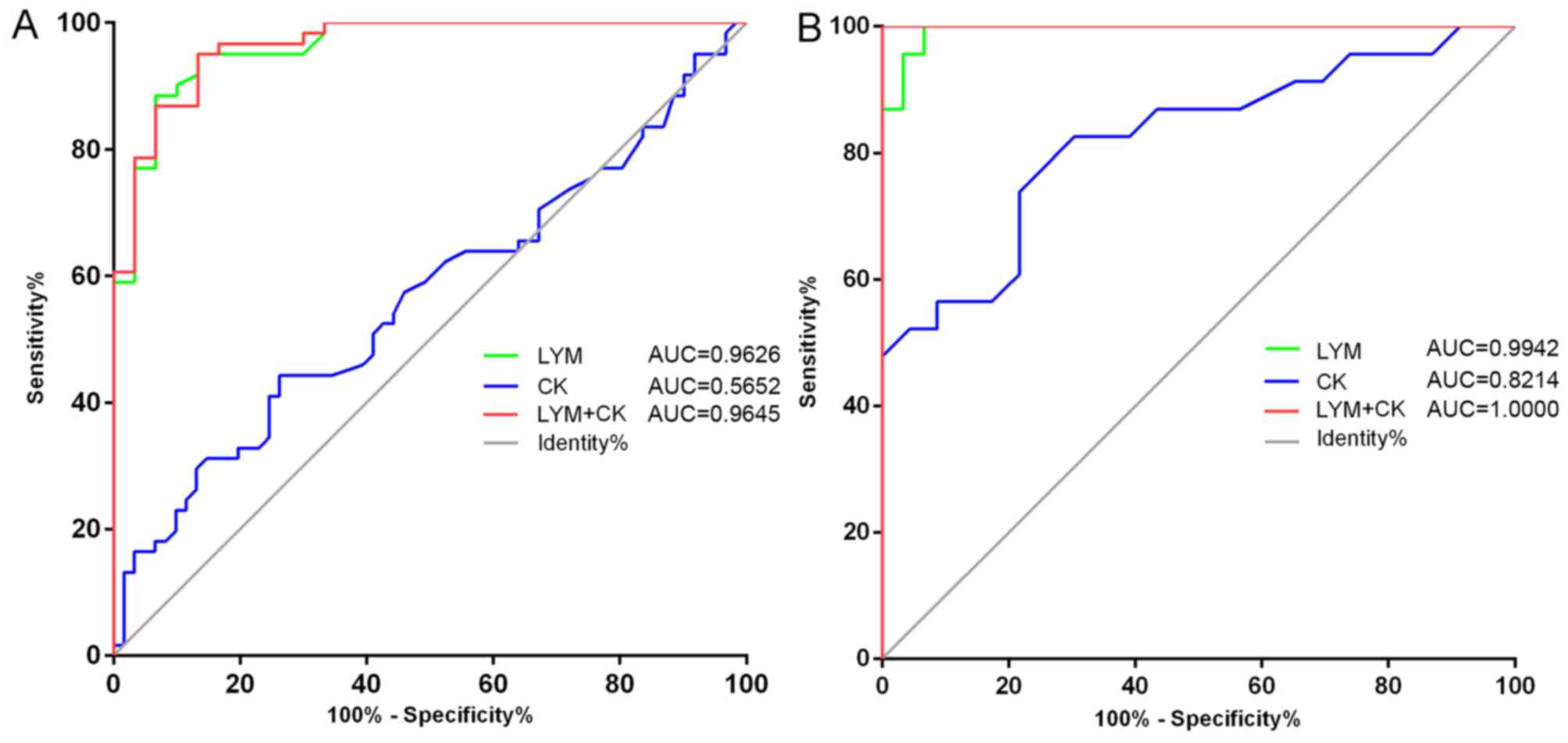
Diagnostic efficacy of clinical
indicators in patients with COVID-19
Hematological and biochemical parameters, as well as
the serum levels of IL-6 and CRP of healthy volunteers and patients
with the common and severe type of COVID-19 were subjected to a ROC
curve analysis to determine their diagnostic value. The AUC,
sensitivity, specificity and cut-off values were calculated. The
analysis revealed that the infection monitoring index, LYM and the
cardiac marker CK had high diagnostic efficacy, sensitivity and
specificity for both common and severe COVID-19. In addition, the
sensitivity of LYM and CK for severe COVID-19 was higher compared
with that for the common type. Of note, the diagnostic efficiency
of LYM combined with CK was higher than that of single factors,
with improved AUC and sensitivity and specificity, particularly for
patients with severe COVID-19, which had a greater AUC (Table V; Fig.
1). The diagnostic efficiency for severe COVID-19 was higher
than for common COVID-19.
 | Table VDiagnostic efficacy of different
clinical indicators in patients with coronavirus disease 2019. |
Table V
Diagnostic efficacy of different
clinical indicators in patients with coronavirus disease 2019.
| Variable/ symptom
type | AUC | Sensitivity
(%) | Specificity
(%) | Cut-off value |
|---|
| Age |
|
Common
group | 0.8348 | 26.23 | 96.67 | 36.50 |
|
Severe
group | 0.5862 | 39.13 | 83.33 | 60.00 |
| RBC |
|
Common
group | 0.7708 | 63.93 | 86.67 | 4.26 |
|
Severe
group | 0.6609 | 69.57 | 66.67 | 4.47 |
| WBC |
|
Common
group | 0.7404 | 65.57 | 83.33 | 5.26 |
|
Severe
group | 0.8348 | 73.91 | 83.33 | 5.34 |
| LYM |
|
Common
group | 0.9626 | 88.52 | 93.33 | 1.75 |
|
Severe
group | 0.9942 | 100.00 | 93.33 | 1.69 |
| LYM% |
|
Common
group | 0.8893 | 77.05 | 90.00 | 0.30 |
|
Severe
group | 0.9514 | 95.65 | 83.33 | 0.31 |
| NEU |
|
Common
group | 0.5740 | 52.46 | 73.33 | 2.90 |
|
Severe
group | 0.5870 | 52.17 | 70.00 | 2.97 |
| NEU% |
|
Common
group | 0.8208 | 70.49 | 86.67 | 0.61 |
|
Severe
group | 0.9232 | 82.61 | 100.00 | 0.66 |
| NLR |
|
Common
group | 0.8787 | 81.97 | 86.67 | 1.95 |
|
Severe
group | 0.9391 | 78.26 | 100.00 | 2.63 |
| IL-6 |
|
Common
group | 0.6259 | 62.30 | 62.30 | 4.60 |
|
Severe
group | 0.8743 | 82.61 | 86.96 | 11.80 |
| CRP |
|
Common
group | 0.7157 | 60.66 | 78.69 | 4.48 |
|
Severe
group | 0.8544 | 73.91 | 86.96 | 14.54 |
| CK |
|
Common
group | 0.5652 | 70.49 | 32.79 | 41.50 |
|
Severe
group | 0.8214 | 82.61 | 69.57 | 570.00 |
| CK-Mb |
|
Common
group | 0.6360 | 65.57 | 54.10 | 6.50 |
|
Severe
group | 0.5208 | 47.83 | 56.52 | 6.50 |
| ALP |
|
Common
group | 0.6732 | 65.57 | 67.21 | 65.50 |
|
Severe
group | 0.6238 | 65.22 | 65.22 | 69.50 |
| LDH |
|
Common
group | 0.5649 | 70.49 | 34.43 | 424.50 |
|
Severe
group | 0.6389 | 65.22 | 43.48 | 475.00 |
| LYM + CK |
|
Common
group | 0.9645 | 95.08 | 86.67 | 0.42 |
|
Severe
group | 1.0000 | 100.00 | 100.00 | 0.50 |
Correlation between clinical
parameters
Correlation analysis between hemocytes, myocardial
enzymes and inflammatory factors in patients with COVID-19
indicated that both lymphocytes and neutrophils (particularly LYM
and LYM%) were significantly correlated with IL-6 and CRP,
suggesting that lymphocytes and neutrophils were related to the
degree of infection in patients with COVID-19. However, CK-Mb and
ALP were not significantly correlated with IL-6 and CRP, suggesting
that myocardial damage may be a secondary symptom after the
inflammatory storm. LDH is a myocardial enzyme, which was
correlated with CRP, lymphocytes and neutrophils, suggesting that
LDH may be an early myocardial damage marker in patients with
COVID-19 (Table VI).
 | Table VICorrelation analysis of the clinical
parameters in patients with coronavirus disease 2019. |
Table VI
Correlation analysis of the clinical
parameters in patients with coronavirus disease 2019.
| Variable | RBC | LYM | LYM% | NEU | NEU% | IL-6 | CRP | CK | CK-Mb | ALP | LDH | NLR |
|---|
| RBC | - | 0.045 | 0.106 | -0.097 | -0.073 | -0.214a | -0.113 | 0.182a | 0.220a | 0.006 | -0.054 | -0.180a |
| LYM | 0.045 | - | 0.691a | 0.046 | -0.631a | -0.229a | -0.416a | -0.142 | -0.050 | -0.113 | -0.260a | -0.313a |
| LYM% | 0.106 | 0.691a | - | -0.576a | -0.948a | -0.311a | -0.479a | -0.247a | -0.107 | -0.187a | -0.356a | -0.435a |
| NEU | -0.097 | 0.046 | -0.576a | - | 0.633a | 0.166a | 0.253a | 0.284a | 0.213a | 0.043 | 0.306a | 0.448a |
| NEU% | -0.073 | -0.631a | -0.948a | 0.633a | - | 0.305a | 0.469a | 0.256a | 0.090 | 0.175a | 0.400a | 0.469a |
| IL-6 | -0.214a | -0.229a | -0.311a | 0.166a | 0.305a | - | 0.549a | 0.132 | 0.020 | 0.073 | 0.281a | 0.220a |
| CRP | -0.113 | -0.416a | -0.479a | 0.253a | 0.469a | 0.549a | - | 0.266a | 0.074 | 0.099 | 0.492a | 0.469a |
| CK | 0.182a | -0.142 | -0.247a | 0.284a | 0.256a | 0.132 | 0.266a | - | 0.769a | 0.080 | 0.256a | 0.268a |
| CK-Mb | 0.220a | -0.050 | -0.107 | 0.213a | 0.090 | 0.020 | 0.074 | 0.769a | - | -0.084 | 0.203a | 0.061 |
| ALP | 0.006 | -0.113 | -0.187a | 0.042 | 0.175a | 0.073 | 0.099 | 0.080 | -0.084 | - | -0.268a | -0.006 |
| LDH | -0.054 | -0.26a | -0.356a | 0.306a | 0.400a | 0.281a | 0.492a | 0.256a | 0.203a | -0.268a | - | 0.433a |
| NLR | -0.180a | -0.313a | -0.435a | 0.448a | 0.469a | 0.220a | 0.469a | 0.268a | 0.061 | -0.006 | 0.433a | - |
Discussion
COVID-19 is an infectious, progressive disease,
which invades multiple organs such as the respiratory, nervous and
reproductive systems. Early intervention and treatment may
effectively control the progress of the disease (10-13).
Clinical experience has indicated that laboratory examination is
conducive for the early detection and intervention of COVID-19,
thereby effectively reducing the proportion of severe pneumonia,
improving the cure rate and reducing mortality. The ‘Diagnosis and
Treatment Plan for COVID-19’ (fifth trial version) (9) has also revealed that timely and early
diagnosis and treatment are effective measures for improving the
cure rate of patients with COVID-19. Furthermore, routine blood
examination, routine biochemical blood tests for liver and kidney
function, including the levels of lactic acid, blood glucose,
electrolytes and LDH, myocardial injury markers, and CRP and
cytokine levels are considered as early clinical warning
indicators.
The ‘Diagnosis and Treatment Plan for COVID-19
(fifth trial version)’ (9) has
suggested that the total number of WBCs in peripheral blood is
normal or decreased in the early stages of COVID-19, while LYM is
decreased. However, in cases of severe COVID-19, the peripheral
blood LYM is progressively decreased. In the present study, the
results of the routine blood test in 84 patients with COVID-19 were
recorded. The results revealed that in patients with common
COVID-19, WBC, LYM and LYM% were significantly decreased,
respectively, while the NEU% was elevated by 19.67%. The rates of
WBC, LYM and LYM% in patients with severe COVID-19 were reduced,
while the NEU% was increased. Compared with those in the healthy
control group, LYM and LYM% were significantly decreased and NEU%
was significantly elevated in the patient group. The levels of all
parameters were restored after treatment. In addition, the values
of the aforementioned parameters and the NLR in patients with
severe COVID-19 were significantly elevated compared with those in
healthy individuals. These results were in line with the
descriptions proposed by Qin et al (14) and the 'Diagnosis and Treatment Plan
for COVID-19 (fifth trial version)' on the role of NLR in the
diagnosis of COVID-19, which stated that typically, the ‘LYM is
normal or decreased and the lymphocyte count is decreased’
(9,15).
IL-6 and CRP are important indicators for the
auxiliary diagnosis of infectious diseases (16-19).
In the present study, the levels of IL-6 and CRP in the patient
group were significantly higher compared with those in the cured
group, which was consistent with the descriptions included in the
‘Diagnosis and Treatment Plan for COVID-19’, stating ‘increased CRP
and erythroid sedimentation rate in most patients’. The
‘inflammatory storm’ caused by COVID-19 may lead to myocardial
injury and abnormal liver function. It is also stated in the
‘Diagnosis and Treatment Plan for COVID-19’ that there may be an
‘increase of lactate dehydrogenase, muscle enzyme and myoglobin in
some patients’ and ‘increase of liver enzymes in some patients’. In
the present study, the respective abnormal increase ratio in the
common and severe groups were as follows: CK, 13.04 and 28.57% for
males and 13.16 and 33.33% for females were increased; CK-Mb, 1.64
and 8.7%; LDH, 26.23 and 47.83% were increased, and ALP, 6.56 and
21.74%; however, no statistically significant differences were
observed. Furthermore, correlation analysis indicated that LDH was
correlated with the infection-related parameters LYM%, NEU, NEU%
and CRP, suggesting that myocardial injury was closely associated
with the progression of inflammation in patients with COVID-19.
These results suggested that the aforementioned parameters may be
considered as monitoring indicators for the treatment and
prognostication of patients infected with SARS-CoV-2.
Yi et al (20) demonstrated that the levels of CK
(93.18% patients) and CK-Mb (95.45% patients) were increased in
patients with COVID-19. This may be due to the fact that all
subjects enrolled suffered from mild COVID-19 and the number of
cases included was small. In the present study, 84 patients with
common and severe COVID-19 were enrolled and the results suggested
that although the levels of CK in the severe type were higher
compared with the common type, the normal CK and CK-Mb range in
patients with COVID-19 was only 60.87% (male) or 73.68% (female)
and 98.36%, respectively. In addition, the rate of the increased CK
levels in the severe group was higher than that in the common
group, suggesting that CK was closely associated with COVID-19 and
may be used as a monitoring indicator for disease course.
The results of the ROC curve analysis revealed that
LYM and CK were superior compared with other parameters in the
diagnosis of common and severe COVID-19 and the sensitivity of the
combined application of LYM + CK in the diagnosis of severe and
common COVID-19 was >95 and 100%, respectively. This result
suggested that the combined application of LYM and CK may be used
as a reference index for the clinical diagnosis and progress
monitoring of COVID-19.
In conclusion, the present study found that the
combined application of LYM and CK may be an indicator for the
evaluation of COVID-19, as well as the serum levels of IL-6 and CRP
in patients with COVID-19.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
LZ and YD designed the study, checked and confirmed
the authenticity of the raw data. ZQ and XW performed the
experiments and analyzed the data. All authors read and approved
the final manuscript.
Ethics approval and consent to
participate
This study was approved by the Ethics Committee of
the Tianjin Haihe Hospital (Tianjin, China). The patients provided
their written informed consent for participating in the current
study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Nishiura H, Kobayashi T, Miyama T, Suzuki
A, Jung SM, Hayashi K, Kinoshita R, Yang Y, Yuan B, Akhmetzhanov
AR, et al: Estimation of the asymptomatic ratio of novel
coronavirus infections (COVID-19). Int J Infect Dis. 94:154–155.
2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Zhang LX, Miao SY, Qin ZH, Wu JP, Chen HY,
Sun HB, Xie Y, Du YQ and Shen J: Preliminary analysis of B- and
T-cell responses to SARS-CoV-2. Mol Diagn Ther. 24:601–609.
2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Patel NA: Pediatric COVID-19: Systematic
review of the literature. Am J Otolaryngol.
41(102573)2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Wostyn P: COVID-19 and chronic fatigue
syndrome: Is the worst yet to come? Med Hypotheses.
146(110469)2021.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Paliwal VK, Garg RK, Gupta A and Tejan N:
Neuromuscular presentations in patients with COVID-19. Neurol Sci.
41:3039–3056. 2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Krajewska J, Krajewski W, Zub K and
Zatoński T: COVID-19 in otolaryngologist practice: A review of
current knowledge. Eur Arch Otorhinolaryngol. 277:1885–1897.
2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
MaassenVanDenBrink A, de Vries T and
Danser AH: Headache medication and the COVID-19 pandemic. J
Headache Pain. 1(38)2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Chan KW, Wong VT and Tang SCW: COVID-19:
An update on the epidemiological, clinical, preventive and
therapeutic evidence and guidelines of integrative Chinese-Western
Medicine for the management of 2019 novel coronavirus disease. Am J
Chin Med. 48:737–762. 2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
National Health Commission of the People's
Republic of China Diagnosis and treatment plan for COVID-19 (trial
version 5 revised version) [EB/OL]. (2020-02-08)[2020-02-15].
http://www.nhc.gov.cn/xcs/zhengcwj/202002/d4b895337e19445f8d728fcaf1e3e13a.shtml.
(In Chinese).
|
|
10
|
Chan JF, Zhang AJ, Yuan S, Poon VK, Chan
CC, Lee AC, Chan WM, Fan Z, Tsoi HW, Wen L, et al: Simulation of
the clinical and pathological manifestations of Coronavirus Disease
2019 (COVID-19) in golden Syrian hamster model: Implications for
disease pathogenesis and transmissibility. Clin Infect Dis.
71:2428–2446. 2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Dong L, Tian J, He S, Zhu C, Wang J, Liu C
and Yang J: Possible vertical transmission of SARS-CoV-2 from an
infected mother to her newborn. JAMA. 323:1846–1848.
2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Fu J, Huang PP, Zhang S, Yao QD, Han R,
Liu HF, Yang Y and Zhang DY: The value of serum amyloid A for
predicting the severity and recovery of COVID-19. Exp Ther Med.
20:3571–3577. 2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Herold T, Jurinovic V, Arnreich C,
Lipworth BJ, Hellmuth JC, von Bergwelt-Baildon M, Klein M and
Weinberger T: Elevated levels of IL-6 and CRP predict the need for
mechanical ventilation in COVID-19. J Allergy Clin Immunol.
146:128–136.e4. 2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Qin C, Zhou L, Hu Z, Zhang S, Yang S, Tao
Y, Xie C, Ma K, Shang K, Wang W and Tian DS: Dysregulation of
immune response in patients with COVID-19 in Wuhan, China. Clin
Infect Dis. 71:762–768. 2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Tavakolpour S, Rakhshandehroo T, Wei EX
and Rashidian M: Lymphopenia during the COVID-19 infection: What it
shows and what can be learned. Immunol Lett. 225:31–32.
2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Gao Y, Li T, Han M, Li X, Wu D, Xu Y, Zhu
Y, Liu Y, Wang X and Wang L: Diagnostic utility of clinical
laboratory data determinations for patients with the severe
COVID-19. J Med Virol. 92:791–796. 2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Deng Y, Liu W, Liu K, Fang YY, Shang J,
Zhou L, Wang K, Leng F, Wei S, Chen L, et al: Clinical
characteristics of fatal and recovered cases of coronavirus disease
2019 in Wuhan, China: A retrospective study. Chin Med J (Engl).
133:1261–1267. 2020.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Lin F, Wu YH, Deng W, Wu PB, Chen JY, Tang
SH, Wen JZ, Liu SQ, Lin ZM, Li DF, et al: Clinical analysis and RNA
findings in a family with SARS-CoV-2 infection: Case reports. World
Acad Sci J. 2(17)2020.
|
|
19
|
Liu F, Li L, Xu M, Wu J, Luo D, Zhu Y, Li
B, Song X and Zhou X: Prognostic value of interleukin-6, C-reactive
protein, and procalcitonin in patients with COVID-19. J Clin Virol.
127(104370)2020.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Yi F, Li Z and Li P: Analysis of the
clinical value of IL-6, ESR, CRP and routine biochemical tests for
the diagnosis of COVID-19. Int J Lab Med. 8:902–915. 2020.(In
Chinese).
|