Introduction
Lung cancer is the most common malignant tumor with
the highest morbidity and mortality rates, and is one of the most
life-threatening cancers worldwide (1). Based on histological type, lung cancer
is classified as small cell lung cancer and non-small cell lung
cancer (NSCLC); of these subtypes, NSCLC accounts for ~80-85% of
all lung cancer cases (2). At
present, the effects of NSCLC treatment are not satisfactory, and
no major breakthrough in survival outcome has been achieved. In
addition, most patients are in the advanced stages of disease at
initial diagnosis, have missed the opportunity for surgery, and
cannot tolerate systemic chemotherapy; thus, radiotherapy has
become an alternative method for the treatment of NSCLC (3). However, recurrence and distant
metastasis remain major obstacles in the implementation of this
therapeutic method. Thus, it is essential to identify specific
molecular markers to predict the response to radiotherapy in
patients with invasive disease; such markers may increase the
efficacy of radiotherapy for NSCLC.
High mobility group box 1 (HMGB1), located in the
eukaryotic nucleus, is a nonhistone DNA-binding protein involved in
various cellular functions, such as inflammation, proliferation,
differentiation and metastasis (4-6).
A number of studies have demonstrated that as a chromatin-binding
nuclear protein, HMGB1 is highly expressed in multiple tumor types,
which is correlated with tumor development and progression
(6,7). The expression levels of HMGB1 have
also been associated with the chemosensitivity or radiosensitivity
of several types of cancer (8-10).
In lung cancer, HMGB1 expression is upregulated, promotes tumor
cell proliferation, and is associated with tumor cell
chemosensitivity (11-13).
However, the function of HMGB1 in the radioresistance of NSCLC, as
well as the underlying mechanism, remains unclear.
MicroRNAs (miRNAs) are a group of small endogenous
noncoding RNA molecules that downregulate gene expression by
repressing or degrading their target mRNAs. miRNAs are implicated
in various pathological and physiological processes through the
regulation of a wide array of cellular functions, such as
differentiation, apoptosis, proliferation and metastasis (14,15).
Numerous studies have indicated that the abnormal expression of
specific miRNAs may be involved in the acquisition of
radioresistance in numerous cancer types, particularly NSCLC
(16-18).
miR-107 is aberrantly expressed in various malignancies and can
function as a tumor suppressor or oncogene by regulating cellular
proliferation and invasiveness. In NSCLC, miR-107 inhibits tumor
growth and metastasis by regulating the brain-derived neurotrophic
factor (BDNF)-mediated PI3K/AKT pathway, demonstrating that the
miR-107/BDNF axis may contribute to NSCLC tumorigenesis (19). Additionally, targeting CDK8 via
miR-107 may promote the reversal of chemoresistance, and provide a
biomarker for cisplatin responsiveness in patients with NSCLC
(20). However, the level of
miR-107 in the serum of patients with NSCLC, and its role in
radiotherapy, require further investigation.
In the present study, the expression levels of HMGB1
were detected in patients with NSCLC who had received radiotherapy,
and its diagnostic value for distinguishing radiotherapy-sensitive
from radiotherapy-resistant patients was analyzed. Furthermore, the
effect of HMGB1 on the radiosensitivity of A549 and H1299 cells was
investigated in vitro. Specifically, a direct interaction
between miR-107 and the 3' untranslated region (UTR) of HMGB1 was
observed, suggesting a novel mechanism by which HMGB1 and miR-107
regulate NSCLC cell proliferation and radiosensitivity.
Materials and methods
Patients and serum samples
The present study was approved by the Ethics
Committee of the Affiliated Hospital of Chengde Medical University,
and all the patients provided written informed consent. A total of
73 patients (31 female and 42 male; mean age, 55.24 years; age
range, 26-75 years) who were pathologically diagnosed with NSCLC
were enrolled into the present study between January 2017 and
December 2018, among. The patients were recruited based on the
following inclusion criteria: i) Aged <70 years old, and with
NSCLC, confirmed by histopathology; ii) had an expected survival
time >3 months; iii) had received chest radiotherapy for the
first time, without concurrent chemotherapy or immunomodulator
treatment; iv) had a Karnofsky performance score >70; and v)
exhibited normal heart, liver and kidney function. All patients
received radiotherapy with intensity-modulated radiation therapy on
an Elekta Synergy linear accelerator, and the total radiation dose
was 60 Gy (2 fractions at 30 Gy).
Fasting venous blood was collected from the patients
1 week prior to, and 2, 4 and 6 weeks after radiotherapy. The blood
samples were centrifuged at 700 x g for 10 min at -4˚C to isolate
the serum, which was then stored at -80˚C for subsequent
analysis.
ELISA
The HMGB1 concentrations in the blood samples were
determined using an HMGB1 ELISA kit (cat. no. ARG81351; arigo
Biolaboratories Corp.) according to the manufacturer's
instructions.
Cell culture and X-ray
irradiation
The NSCLC cell lines, A549 and H1299, were obtained
from the Shanghai Institute of Cell Biochemistry and Cell Biology
(Shanghai, China). The cells were cultured in DMEM supplemented
with 10% FBS (both Invitrogen; Thermo Fisher Scientific, Inc.) at
37˚C and a CO2 concentration of 5%.
NSCLC cell irradiation was conducted at room
temperature using a 6-MV linear accelerator (Siemens AG), with a
source skin distance of 100 cm and an irradiation dose rate of 200
cGy/min.
Transfection
The miR-107 mimics (5'-AGCAGCAUUGUACA GGGCUAUCA-3'),
mimic negative control (NC; 5'-UUCUCC GAACGUGUCACGUTT-3'), miR-107
inhibitor (5'-UGAUAG CCCUGUACAAUGCUGCU-3') and inhibitor NC
(5'-CAGUA CUUUUGUGUAGUACA-3') were synthesized and purchased from
Shanghai GenePharma Co., Ltd. Preliminary experiments were carried
out using A549 and H1299 cells transfected with 50 nM miR-107
mimics, inhibitor or the relevant NCs.
To knockdown HMGB1, shRNA HMGB1 or a scrambled shRNA
sequence was sub-cloned into pGPU6/Neo plasmids (Shanghai
GenePharma Co., Ltd.), referred to as sh-HMGB1 or scrambled
control. A549 and H1299 cells were transfected with sh-HMGB1 or the
associated scrambled control.
All transfections were performed using
Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's protocol. After
transfection for 24 h at 37˚C, the transfection efficiency was
verified using reverse transcription-quantitative PCR
(RT-qPCR).
RNA extraction and RT-qPCR
Total RNA was extracted from blood using the
miRNeasy Mini Kit (Qiagen GmbH), and from NSCLC cells using
TRIzol® reagent (Invitrogen; Thermo Fisher Scientific,
Inc.) per the manufacturers' instructions. Then, the total RNA was
reverse transcribed into cDNA using the miScript II RT Kit (Qiagen
GmbH) or PrimeScript™ RT Master Mix (Takara Bio, Inc.) according to
the manufacturers' instructions. qPCR was performed using the SYBR
Premix Ex Taq II PCR kit (Takara Bio, Inc.) according to the
manufacturer's protocol. The thermocycling conditions were as
follows: 95˚C for 5 min, followed by 35 cycles of 95˚C for 20 sec,
60˚C for 30 sec, and 72˚C for 20 sec, with a final extension at
72˚C for 5 min. The relative levels of miR-107 and HMGB1 expression
were calculated using the 2-ΔΔCq
method (21), and U6 and GAPDH
served as the internal controls for miRNA and mRNA expression,
respectively. The qPCR primer sequences were as follows: miR-107
forward, 5'-GCCGAGAGCAGCAUUGUACA-3', and reverse,
5'-CTCAACTGGTGTCGTGGA-3'; HMGB1 forward, 5'-CCA
GGGGCTTTTCTACCAGG-3' and reverse, 5'-AACCCCAAC AAACTGCTGGA-3'; U6
forward, 5'-GTGCTCGCTTCGGCA GCACATATAC-3' and reverse,
5'-AAAAATATGGAACGC TCACGAATTTG-3'; and GAPDH forward, 5'-ACCCAGAAG
ACTGTGGATGG-3' and reverse, 5'-CACATTGGGGGTAG GAACAC-3'.
Cell Counting Kit 8 (CCK-8) assay
A549 and H1299 cells were seeded into 96-well plates
at a density of 5x103 cells/well and incubated at 37˚C.
A total of 10 µl CCK-8 solution (Dojindo) was added to each well at
0, 24, 48 and 72 h. After incubation for 2 h, the optical density
value was measured with a microplate reader at a wavelength of 450
nm.
Clonogenic survival assay
NSCLC cells were irradiated with a set of graded
doses (0, 2, 4, 6 and 8 Gy) and then cultured in 6-well plates for
14 days. The medium was replaced with fresh medium every 3 days.
After the formation of cell clones (colonies containing ≥50 cells),
the cells were stained with 0.5% crystal violet solution
(Sigma-Aldrich; Merck KGaA) at room temperature for 20 min, and
counted under a light microscope (Olympus Corporation).
Transwell invasion assay
Transwell invasion assays were conducted to
determine the invasive ability of radioresistant cells, using
precoated Matrigel Transwell chambers (BD Bioscience) with 8-µm
porous membranes (Corning, Inc.). Briefly, A549 cells were seeded
into 24-well plates and then exposed to a 2-Gy dose of radiation.
After irradiation, 5x104 A549 cells resuspended in
serum-free medium were seeded into the top chamber, and complete
medium was added to the lower chamber as a chemoattractant. After
48 h, invasive cells were fixed with 4% paraformaldehyde for 10 min
at room temperature, and then stained with 0.1% crystal violet at
room temperature for 20 min. The number of invaded cells was
counted in five random independent fields per well using a light
microscope (Olympus Corporation).
Luciferase reporter assay
A computer-based algorithm, the TargetScan database
(www.targetscan.org), was used to predict
potential miRNAs that could target HMGB1. In the present study,
miR-107 was searched for in humans following the entry of the HMGB1
gene, after which a luciferase reporter assay was conducted.
Briefly, partial sequences of the HMGB1 3'-UTR with wild-type (WT)
or mutant (MT) miR-107-binding sites were cloned into the pmirGLO
dual-luciferase vector (Promega Corporation) to generate a WT
(HMGB1 3'-UTR-WT) or an MT (HMGB1 3'-UTR-MT) reporter plasmid.
Then, A549 cells were transiently transfected with the reporter
plasmids along with the miR-107 mimics, mimic NC, miR-107 inhibitor
or the inhibitor NC using Lipofectamine® 2000
(Invitrogen; Thermo Fisher Scientific, Inc.); firefly luciferase
activity was evaluated ~48 h later using a Dual-Luciferase Reporter
Assay System (Promega Corporation) and normalized to that of
Renilla luciferase.
Western blot analysis
Proteins were extracted from A549 cells using
radioimmunoprecipitation assay buffer with protease inhibitor
cocktail (both Cell Signaling Technology, Inc.). Protein
concentration was determined using a BCA kit (CoWin Biosciences).
The proteins (15 µg/lane) were separated using 10% SDS-PAGE and
transferred to nitrocellulose membranes (Amersham; Cytiva). The
membranes were subsequently blocked with 5% skim milk at room
temperature for 2 h. Proteins were detected by western blotting
using rabbit anti-HMGB1 (1:400; cat. no. ab18256; Abcam), rabbit
anti-Toll-like receptor 4 (TLR4; 1:400; cat. no. ab13556; Abcam),
rabbit anti-NF-κB p65(1:400; cat. no. ab207297; Abcam), and rabbit
anti-GAPDH antibodies (1:1,000; cat. no. ab8245; Abcam) and were
incubated at 4˚C overnight. The membranes were then incubated with
the relevant goat anti-rabbit HRP-conjugated secondary antibodies
(1:5,000; cat. no. ab7090; Abcam) at room temperature for 1 h, and
the protein bands were visualized using ECL reagents (Amersham;
Cytiva) and quantified by densitometry (ImageJ software; National
Institutes of Health; http://rsbweb.nih.gov).
Statistical analysis
Statistical analyses were performed using GraphPad
Prism 5.0 (GraphPad Software, Ltd.) and SPSS 23.0 (IBM Corp.). The
data are presented as the mean ± SD. Patient sample data were
non-normally distributed and analyzed by Friedman analysis followed
by Nemenyi's post hoc test. Differences between in vitro
experimental groups were analyzed by one-way ANOVA followed by
Tukey's post hoc test. Receiver operating characteristic (ROC)
curve analysis was performed to evaluate the diagnostic value of
HMGB1 in identifying radiotherapy-sensitive patients with NSCLC.
P<0.05 was considered to indicate a statistically significant
difference.
Results
Serum HMGB1 levels in patients with
NSCLC after radiotherapy
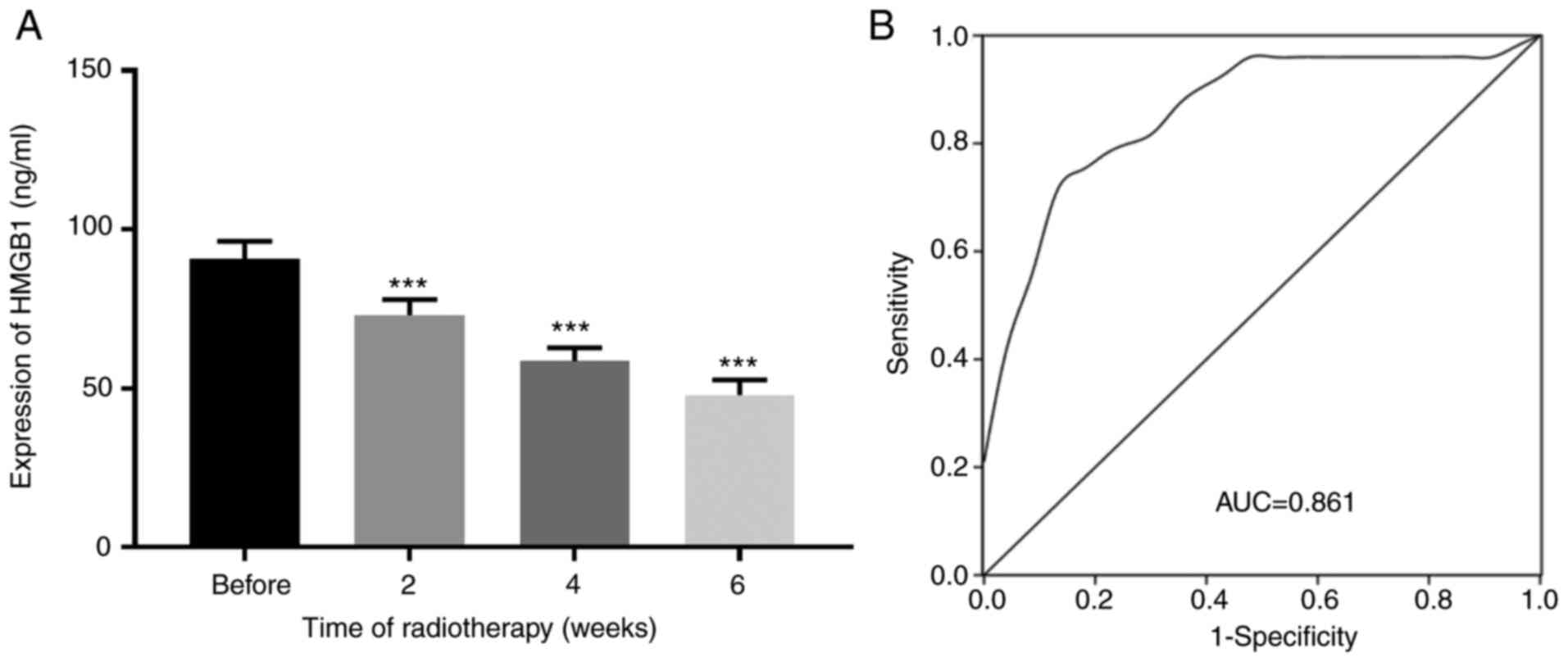
The levels of HMGB1 were quantified in the serum of
patients with NSCLC who had received radiotherapy. As shown in
Fig. 1A, the expression levels of
HMGB1 in the serum gradually declined at 2-, 4- and 6-weeks
post-radiotherapy compared with those 1 week before radiotherapy
(P<0.001).
Diagnostic value of HMGB1 for
determining the radiosensitivity of patients with NSCLC
Then, the expression levels of HMGB1 before
radiotherapy were evaluated for the capacity to distinguish between
radiotherapy-sensitive and -resistant patients with NSCLC. As shown
in Fig. 1B, ROC curve analysis
revealed an area under the curve of 0.861 (sensitivity, 74%;
specificity, 87%; cutoff value, 91.68 ng/ml), suggesting that the
expression of HMGB1 had the diagnostic accuracy for distinguishing
between radiotherapy-sensitive and -resistant patients with
NSCLC.
HMGB1-knockdown inhibits the
proliferation and increases the radiosensitivity of NSCLC cells in
vitro
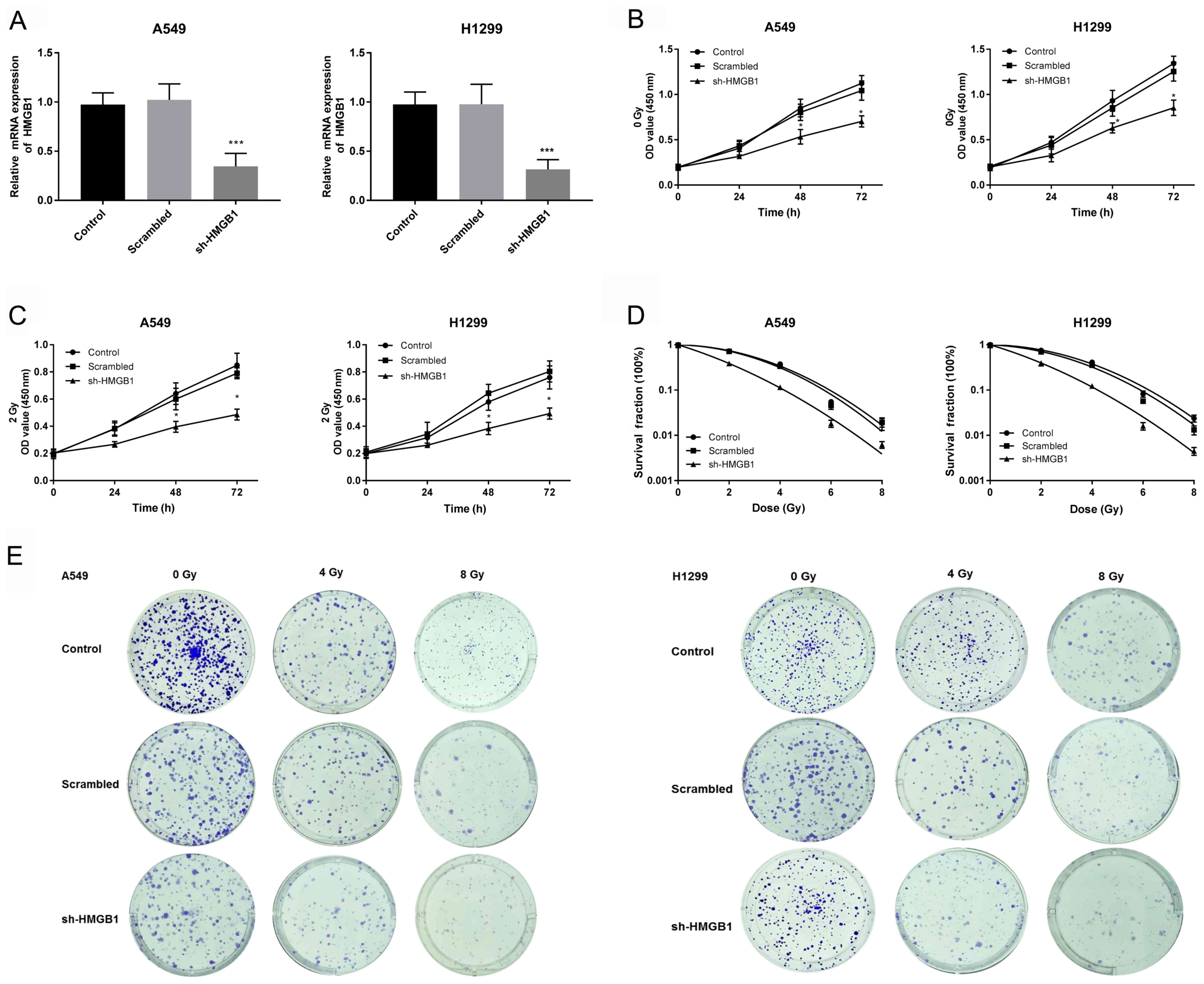
To investigate the biological function of HMGB1 in
NSCLC cell viability, A549 and H1299 cells were transfected with
HMGB1 shRNA (sh-HMGB1) to knock down HMGB1expression, and cells
treated under different conditions were harvested following
transfection and irradiation. RT-qPCR analysis was used to evaluate
transfection efficiency. The results indicated that HMGB1 mRNA
expression was significantly downregulated in HMGB1-knockdown A549
and H1299 cells compared with that of the control group
(P<0.001, Fig. 2A). The results
of the CCK-8 assay revealed that cellular proliferation following
sh-HMGB1 transfection was significantly lower than that of the
corresponding control cells, both without (Fig. 2B) and with (Fig. 2C) irradiation (P<0.05). These
results suggested that radiation significantly suppressed tumor
cell proliferation, and that this inhibitory effect was enhanced
when combined with HMGB1-knockdown.
Furthermore, a colony formation assay was conducted
to determine the effects of HMGB1 on the radiosensitivity of A549
and H1299 cells. Survival curves were plotted based on the
formation of clones and the radiobiological parameters of each
group, and the results indicated that compared with the control,
HMGB1-knockdown enhanced radiosensitivity compared with the control
(Fig. 2D and E). These results indicated that
transfection with sh-HMGB1 inhibited cellular proliferation with or
without irradiation, and increased the radiosensitivity of A549 and
H1299 cells in vitro.
HMGB1 is negatively regulated by
miR-107 in NSCLC
Previous studies have shown that miRNA dysregulation
plays a crucial role in tumor progression by negatively regulating
target genes via binding to their 3'-UTRs. Thus in the present
study, bioinformatics analysis was performed using the TargetScan
database to predict potential upstream regulators of HMGB1. Among
the conserved candidates (miR-107, miR-103 and miR-142-3p), miR-107
was selected as a putative regulator of HMGB1, which was based on
the results of a study indicating that miR-107 plays a tumor
suppressor role by inhibiting tumor growth and metastasis in NSCLC
(19). As shown in Fig. 3A, the HMGB1 3'-UTR contains a
putative binding sequence for miR-107. The serum expression levels
of miR-107 were also measured by RT-qPCR in patients with NSCLC
before radiotherapy, and the correlation between miR-107 expression
and HMGB1 levels was assessed. The results showed that HMGB1 level
was negatively associated with miR-107 expression in the serum of
patients with NSCLC (r=-0.5266 and P<0.001, Fig. 3B). The results also indicated that
miR-107 mimics promoted the expression of miR-107, while inhibitors
suppressed expression (P<0.001, Fig.
3C). Subsequently, dual-luciferase reporter assays revealed
that when A549 cells were transfected with HMGB1 3'-UTR-WT,
enhanced expression of miR-107 inhibited, while miR-107-knockdown
increased luciferase activity (P<0.05, Fig. 3D). However, in the cells transfected
with HMGB1 3'-UTR-MT, overexpression or downregulation of miR-107
had no significant effect on luciferase activity. Furthermore, the
mRNA and protein levels of HMGB1 were determined in A549 cells
transfected with miR-107 mimics or inhibitor. The results indicated
that miR-107 overexpression decreased the mRNA and protein levels
of HMGB1, and that miR-107-knockdown promoted HMGB1 protein
(P<0.05, Fig. 3E) and mRNA
(P<0.001, Fig. 3F)
expression.
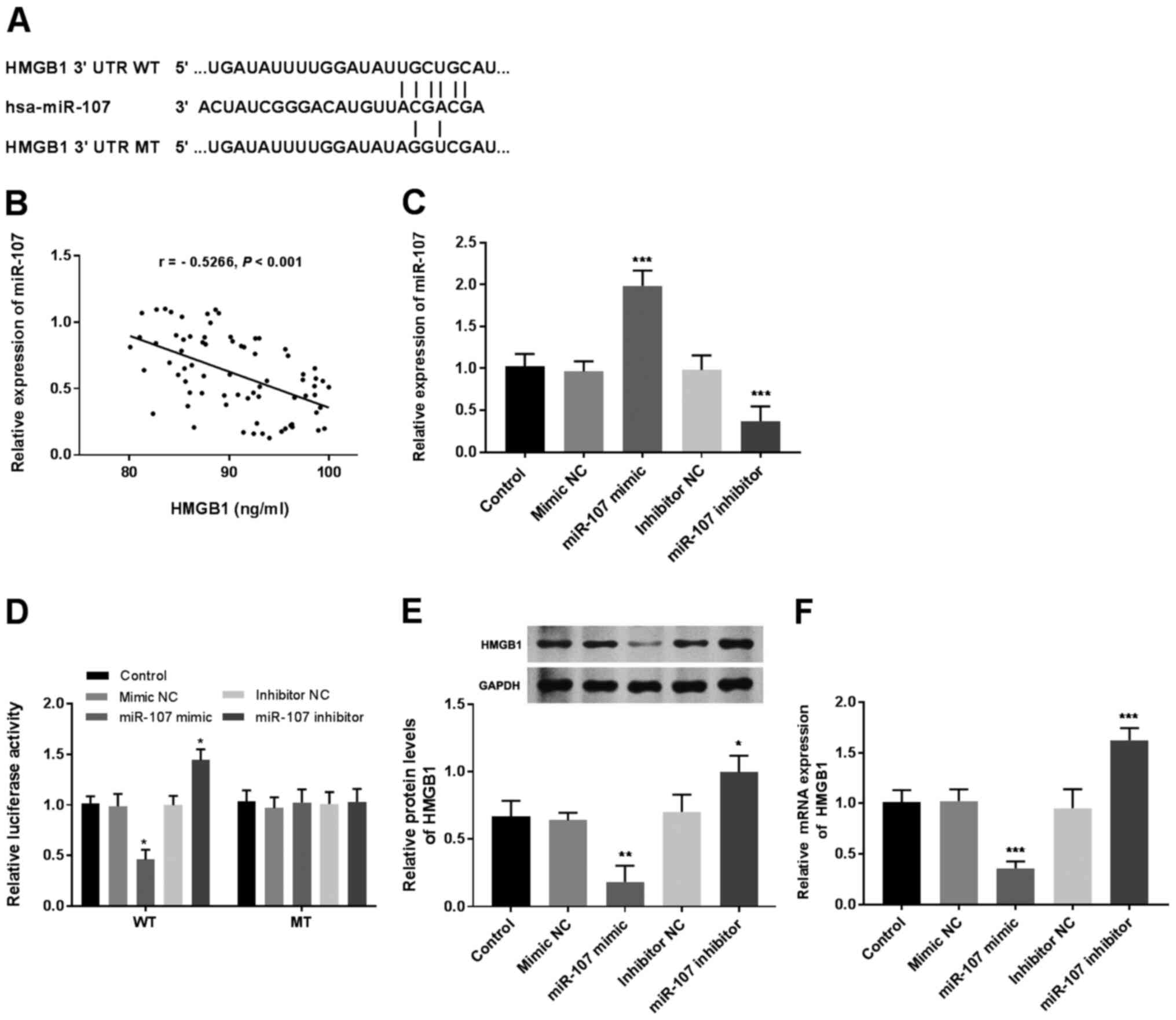 | Figure 3miR-107 negatively regulates HMGB1
expression. (A) Putative miR-107-binding sequences in the HMGB1
3'UTR were predicted using TargetScan. (B) Negative correlation
between miR-107 and HMGB1 expression in the serum of patients with
NSCLC, determined using Spearman's correlation analysis (r=-0.5266
and P<0.001). (C) Transfection efficiency was detected using
reverse transcription-quantitative analysis.
***P<0.001 vs. the associated control. (D) Luciferase
reporters containing WT or MT miR-107 binding sites were
transfected into A549 cells along with the miR-107 mimics, mimic
NC, miR-107 inhibitor or inhibitor NC. Luciferase activity was
evaluated by dual-luciferase reporter assay. *P<0.05.
(E) miR-107 overexpression decreased the protein levels of HMGB1,
while knockdown of miR-107 promoted the HMGB1 protein levels.
*P<0.05 and **P<0.01 vs. the associated
control. (F) miR-107 overexpression decreased the mRNA levels of
HMGB1, while miR-107-knockdown promoted HMGB1 mRNA expression.
***P<0.001 vs. the associated control. miR, microRNA;
HMGB1, high mobility group box 1; UTR, untranslated region; WT,
wild-type; MT, mutant-type; NC, negative control. |
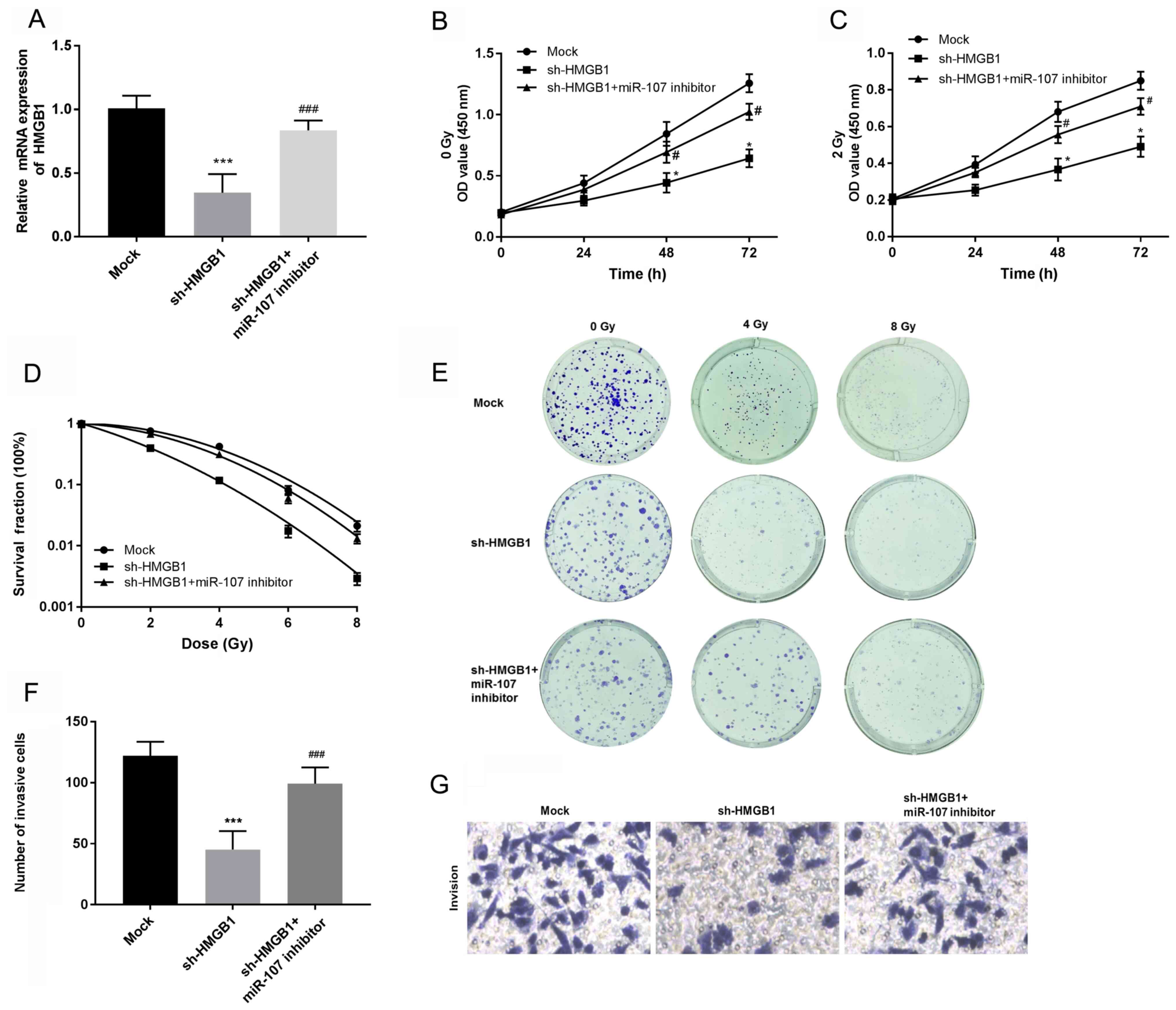
Downregulation of miR-107 relieves the
enhanced radiosensitivity of A549 cells caused by sh-HMGB1
To further investigate whether the reduction in
HMGB1-enhanced radiosensitivity was regulated by miR-107, A549
cells transfected with sh-HMGB1 or a combination of sh-HMGB1 and
the miR-107 inhibitor were treated with or without radiation. The
RT-qPCR results showed that sh-HMGB1 downregulated the expression
levels of HMGB1, and that knocking down miR-107 reversed the
sh-HMGB1-mediated decrease in HMGB1 expression (P<0.001,
Fig. 4A). Knocking down miR-107
with an miR-107 inhibitor reversed the inhibition in cellular
proliferation without (Fig. 4B) or
with (Fig. 4C) irradiation, and
reduced the survival fractions (Fig.
4D and E) induced by sh-HMGB1
(P<0.05). Furthermore, Transwell invasion assay results showed
that HMGB1-knockdown suppressed A549 cell invasiveness, and that
downregulating miR-107 reversed the sh-HMGB1-mediated decrease in
the invasion abilities of A549 cells after irradiation (P<0.001,
Fig. 4F and G). In summary, these results revealed that
the knockdown of HMGB1 enhanced the radiosensitivity of A549 cells
regulated by miR-107 expression.
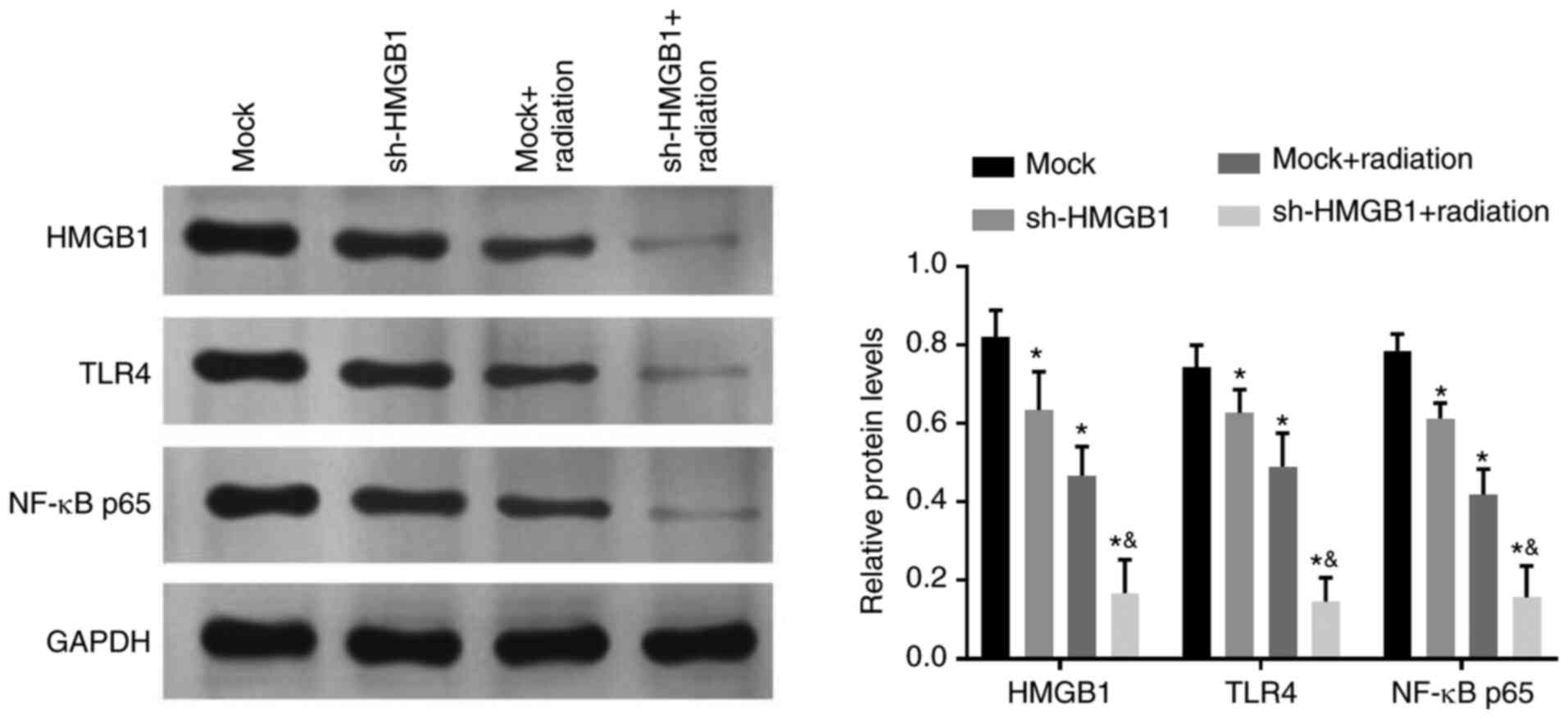
Expression of TLR4/NF-κB signaling
pathway related-proteins in A549 cells
Compared with the mock group, HMGB1-knockdown and
irradiation downregulated the expression of TLR4/NF-κB signaling
pathway related-proteins (P<0.05, Fig. 5). Compared with the mock-radiation
group, the downregulation of HMGB1 enhanced radiation-mediated
decreased expression of TLR4/NF-κB signaling pathway
related-proteins in A549 cells (P<0.05).
Discussion
Radiotherapy is an important treatment option for
patients with locally advanced NSCLC. However, patients frequently
exhibit radioresistance, which in clinical practice, affects
treatment efficacy and survival outcome. Thus, increasing
radiosensitivity and decreasing radioresistance is crucial for
improving patient prognosis. HMGB1 has been reported to be
upregulated in various types of cancer, including NSCLC (4,22,23).
However, the effects of HMGB1 on the radiosensitivity of NSCLC
remain to be clarified.
HMGB1 is involved in regulating various biological
activities, but can also promote tumor development by inducing the
formation of tumor neovascularization, and trigger the protective
antitumor T lymphocyte response to inhibit tumor growth (24). In esophageal carcinoma,
HMGB1-knockdown decreased the proliferation of tumor cells after
irradiation and increased the radiosensitivity of tumor cells
(25).
Previous studies have reported that HMGB1 is
upregulated in NSCLC and may induce the tumorigenesis and
metastasis of lung cancer (26-28).
In the present study, HMGB1 expression was notably downregulated in
the serum of patients with NSCLC after radiotherapy, suggesting
that conventional radiotherapy reduces HMGB1-associated
radioresistance, thus inhibiting tumor growth. ROC curve analysis
subsequently revealed that the aberrant expression of HMGB1 had a
relatively high diagnostic value for distinguishing between
radiosensitive and radioresistant patients.
Furthermore, functional assays results demonstrated
that HMGB1-knockdown inhibited the proliferation of A549 and H1299
cells, which is consistent with findings in other tumor cell
studies (25,29). In addition, irradiation
significantly suppressed NSCLC cell proliferation, and this
inhibitory effect was enhanced by combining radiotherapy with
HMGB1-knockdown. Furthermore, in vitro experimentation also
showed that decreased expression of HMGB1 resulted in the
heightened radiosensitivity of NSCLC cells. HMGB1-knockdown has
also been reported to enhance radiosensitivity in bladder cancer
(10) and esophageal squamous cell
carcinoma (30), which is
consistent with the findings of the present study.
Numerous studies have demonstrated that miRNAs play
crucial roles in modulating the progression and tumorigenesis of
various cancers by negatively regulating target mRNAs (31,32).
In renal carcinoma, the miR-454-3p-induced downregulation of BTG1
enhanced the radiosensitivity of renal carcinoma cells (33). In the present study, only a
computer-based algorithm (namely TargetScan) was used to identify a
binding association between miR-107 and the HMGB1 3'-UTR. The lack
of RNA-RNA binding or protection assays to confirm this association
is a limitation to the present study. However, a negative
correlation was observed between the levels of HMGB1 and miR-107 in
the serum of patients with NSCLC. Subsequent luciferase reporter
assay results suggested that miR-107-overexpression inhibited the
luciferase activity of A549 cells transfected with the HMGB1 3'-UTR
WT sequence, but not with the mutant 3'-UTR sequence. Furthermore,
miR-107-overexpression decreased the mRNA and protein levels of
HMGB1, while miR-107-knockdown promoted HMGB1 protein and mRNA
expression. These results suggest that HMGB1 may be a direct target
of miR-107.
Previous studies have also suggested an interaction
between miR-107 and HMGB1, including those with human
osteoarthritic chondrocytes (34)
and breast cancer cells (35).
Using A549 cells, the present study reported that knocking down
miR-107 reversed the inhibition in cellular proliferation with or
without irradiation, and reduced the survival fractions induced by
sh-HMGB1. HMGB1-knockdown also suppressed A549 cell invasiveness,
which was reversed by downregulating miR-107 after irradiation.
These findings suggest that the inhibition in cellular
proliferation and invasiveness, and the reduction in survival
fractions in NSCLC, associated with HMGB1 downregulation, may be
regulated by miR-107.
HMGB1 is an important component of the tumor
microenvironment, which leads to immunosuppression and promotes the
occurrence and development of tumors (36). TLR4, a member of the Toll-like
receptor family, is a crucial component of innate immunity that
enhances the function of HMGB1 in tumors by creating a precancerous
environment through angiogenesis and inflammation (37). As important survival mechanisms in
the response to radiation, various signaling pathways are important
for regulating radiosensitivity, including the TLK4/NF-κB cascade
(38-40).
For instance, HMGB1 and the TLR4 signaling pathway were found to be
overactive in malignant epithelial ovarian cancer, and to be
associated with tumor cell proliferation and invasiveness that may
be mediated via NF-κB signaling (41). Moreover, TLR4 and NF-κB are closely
associated with the radiosensitivity of several cancers, such as
esophageal and liver cancer (42,43).
In the present study, HMGB1-knockdown and irradiation downregulated
the expression of TLR4/NF-κB signaling pathway related-proteins.
Compared with the mock-radiation group, the downregulation of HMGB1
enhanced the radiation-mediated decrease in TLR4/NF-κB signaling
pathway related-protein expression. These findings suggested that
HMGB1-knockdown promoted radiosensitivity, which may be regulated
by miR-107 via the TLR4/NF-κB signaling pathway. Accordingly, the
present study indicates the potential mechanism by which
HMGB1-knockdown results in NSCLC radiosensitivity. In the future,
the expression of additonal radiosensitivity-related molecules will
be detected to elucidate the detailed mechanism of HMGB1-associated
radiosensitivity in NSCLC.
In conclusion, the results of the present study
demonstrated that HMGB1 expression was decreased in the serum of
patients with NSCLC who had received radiotherapy, and that reduced
HMGB1 expression may be a putative diagnostic marker for predicting
radiosensitivity in NSCLC. Furthermore, downregulation of HMGB1
enhanced the radiosensitivity of NSCLC cells (that were regulated
by miR-107) by mediating the TLR4/NF-κB signaling pathway. These
findings revealed that HMGB1 may be a potential therapeutic target
for NSCLC, and that a combination of HMGB1-knockdown and
irradiation may be an effective therapeutic strategy for patients
with radioresistant NSCLC.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by the Chengde
Technology Support Project (grant no. 201701A036).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
LB< JZ, DG, CL, WL and QL made substantial
contributions to conception and design of the study, conducting the
experiments and interpretation of the data. LB, JZ and DG collected
and analyzed the patient data. LB, JZ and CL drafted the
manuscript, and WL and QL revised it critically for important
intellectual content. LB and QL are responsible for the
authenticity of the raw data. All authors approved the final
version of the manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of the Affiliated Hospital of Chengde Medical University,
and all patients provided written informed consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2019. CA Cancer J Clin. 69:7–34. 2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Larsen JE and Minna JD: Molecular biology
of lung cancer: Clinical implications. Clin Chest Med. 32:703–740.
2011.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Zhao L, Wang J, Li H, Che J, Ma N and Cao
B: Safety and Efficacy of Tianfoshen Oral Liquid in Non-Small Cell
Lung Cancer Patients as an Adjuvant Therapy. Evid Based Complement
Alternat Med. 2019(1375439)2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Cheng P, Ma Y, Gao Z and Duan L: High
Mobility Group Box 1 (HMGB1) Predicts Invasion and Poor Prognosis
of Glioblastoma Multiforme via Activating AKT Signaling in an
Autocrine Pathway. Med Sci Monit. 24:8916–8924. 2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Hou C, Chen Y, Huang X, Huang Q, Li M and
Tan X: miR-19 targets PTEN and mediates high mobility group protein
B1(HMGB1)-induced proliferation and migration of human airway
smooth muscle cells. PLoS One. 14(e0219081)2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Yue Y, Zhou T, Gao Y, Zhang Z, Li L, Liu
L, Shi W, Su L and Cheng B: High mobility group box 1/toll-like
receptor 4/myeloid differentiation factor 88 signaling promotes
progression of gastric cancer. Tumour Biol.
39(1010428317694312)2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Chen R, Zhu S, Fan XG, Wang H, Lotze MT,
Zeh HJ III, Billiar TR, Kang R and Tang D: High mobility group
protein B1 controls liver cancer initiation through yes-associated
protein-dependent aerobic glycolysis. Hepatology. 67:1823–1841.
2018.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Huang CY, Chiang SF, Chen WT, Ke TW, Chen
TW, You YS, Lin CY, Chao KSC and Huang CY: HMGB1 promotes
ERK-mediated mitochondrial Drp1 phosphorylation for chemoresistance
through RAGE in colorectal cancer. Cell Death Dis.
9(1004)2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Shi Y and Gong W: Upregulation of
miR-129-5p increases the sensitivity to Taxol through inhibiting
HMGB1-mediated cell autophagy in breast cancer MCF-7 cells. Braz J
Med Biol Res. 52(e8657)2019.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Shrivastava S, Mansure JJ, Almajed W, Cury
F, Ferbeyre G, Popovic M, Seuntjens J and Kassouf W: The Role of
HMGB1 in Radioresistance of Bladder Cancer. Mol Cancer Ther.
15:471–479. 2016.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Ma Y, Kang S, Wu X, Han B, Jin Z and Guo
Z: Up-regulated HMGB1 in the pleural effusion of non-small cell
lung cancer (NSCLC) patients reduces the chemosensitivity of NSCLC
cells. Tumori. 104:338–343. 2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Zheng H, Chen JN, Yu X, Jiang P, Yuan L,
Shen HS, Zhao LH, Chen PF and Yang M: HMGB1 Enhances Drug
Resistance and Promotes In Vivo Tumor Growth of Lung Cancer Cells.
DNA Cell Biol. 35:622–627. 2016.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wu X, Wang W, Chen Y, Liu X, Wang J, Qin
X, Yuan D, Yu T, Chen G, Mi Y, et al: High Mobility Group Box
Protein 1 Serves as a Potential Prognostic Marker of Lung Cancer
and Promotes Its Invasion and Metastasis by Matrix
Metalloproteinase-2 in a Nuclear Factor-κB-Dependent Manner. Biomed
Res Int. 2018(3453706)2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Bai H and Wu S: miR-451: A Novel Biomarker
and Potential Therapeutic Target for Cancer. Onco Targets Ther.
12:11069–11082. 2019.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Chandan K, Gupta M and Sarwat M: Role of
Host and Pathogen-Derived MicroRNAs in Immune Regulation During
Infectious and Inflammatory Diseases. Front Immunol.
10(3081)2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Chen G, Yu L, Dong H, Liu Z and Sun Y:
MiR-182 enhances radioresistance in non-small cell lung cancer
cells by regulating FOXO3. Clin Exp Pharmacol Physiol.
46(2):137–143. 2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Li YH, Xu CL, He CJ, Pu HH, Liu JL and
Wang Y: circMTDH.4/miR-630/AEG-1 axis participates in the
regulation of proliferation, migration, invasion, chemoresistance,
and radioresistance of NSCLC. Mol Carcinog. 59:141–153.
2020.PubMed/NCBI View
Article : Google Scholar
|
|
18
|
Sun C, Zeng X, Guo H, Wang T, Wei L, Zhang
Y, Zhao J, Ma X and Zhang N: MicroRNA-125a-5p modulates
radioresistance in LTEP-a2 non-small cell lung cancer cells by
targeting SIRT7. Cancer Biomark. 27:39–49. 2020.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Xia H, Li Y and Lv X: MicroRNA-107
inhibits tumor growth and metastasis by targeting the BDNF-mediated
PI3K/AKT pathway in human non-small lung cancer. Int J Oncol.
49:1325–1333. 2016.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Zhang Z, Zhang L, Yin ZY, Fan XL, Hu B,
Wang LQ and Zhang D: miR-107 regulates cisplatin chemosensitivity
of A549 non small cell lung cancer cell line by targeting cyclin
dependent kinase 8. Int J Clin Exp Pathol. 7:7236–7241.
2014.PubMed/NCBI
|
|
21
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) Method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Amornsupak K, Jamjuntra P, Warnnissorn M,
O-Charoenrat P, Sa-Nguanraksa D, Thuwajit P, Eccles SA and Thuwajit
C: High ASMA+ Fibroblasts and Low Cytoplasmic
HMGB1+ Breast Cancer Cells Predict Poor Prognosis. Clin
Breast Cancer. 17:441–452.e2. 2017.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Suren D, Arda Gokay A and Sayiner A: High
Mobility Group Box 1 (HMGB1) expression in gastric adenocarcinomas.
J BUON. 23:422–427. 2018.PubMed/NCBI
|
|
24
|
Ohmori H, Luo Y, Fujii K, Sasahira T,
Shimomoto T, Denda A and Kuniyasu H: Dietary linoleic acid and
glucose enhances azoxymethane-induced colon cancer and metastases
via the expression of high-mobility group box 1. Pathobiology.
77:210–217. 2010.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Zhang X, Yang X, Zhu S, Li Q and Zou N:
Radiosensitization of esophageal carcinoma cells by knockdown of
HMGB1 expression. Oncol Rep. 41:1960–1970. 2019.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Liu Q, Huo Y, Zheng H, Zhao J, Jia L and
Wang P: Ethyl pyruvate suppresses the growth, invasion and
migration and induces the apoptosis of non small cell lung cancer
cells via the HMGB1/RAGE axis and the NF κB/STAT3 pathway. Oncol
Rep. 42:817–825. 2019.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Wang K, Shan S, Wang S, Gu X, Zhou X and
Ren T: HMGB1-containing nucleosome mediates chemotherapy-induced
metastasis of human lung cancer. Biochem Biophys Res Commun.
500:758–764. 2018.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Wu L and Yang L: The function and
mechanism of HMGB1 in lung cancer and its potential therapeutic
implications. Oncol Lett. 15:6799–6805. 2018.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Liu K, Huang J, Ni J, Song D, Ding M, Wang
J, Huang X and Li W: MALAT1 promotes osteosarcoma development by
regulation of HMGB1 via miR-142-3p and miR-129-5p. Cell Cycle.
16:578–587. 2017.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Di X, He G, Chen H, Zhu C, Qin Q, Yan J,
Zhang X and Sun X: High-mobility group box 1 protein modulated
proliferation and radioresistance in esophageal squamous cell
carcinoma. J Gastroenterol Hepatol. 34:728–735. 2019.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Liu R, Chen Y, Shou T, Hu J and Qing C:
miRNA-99b-5p targets FZD8 to inhibit non-small cell lung cancer
proliferation, migration and invasion. Onco Targets Ther.
12:2615–2621. 2019.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Wang L, Lv X, Fu X, Su L, Yang T and Xu P:
miR-153 inhibits the resistance of lung cancer to gefitinib via
modulating expression of ABCE1. Cancer Biomark. 25:361–369.
2019.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Wu X, Ding N, Hu W, He J, Xu S, Pei H, Hua
J, Zhou G and Wang J: Down-regulation of BTG1 by miR-454-3p
enhances cellular radiosensitivity in renal carcinoma cells. Radiat
Oncol. 9(179)2014.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Lin SS, Yuan LJ, Niu CC, Tu YK, Yang CY
and Ueng SWN: Hyperbaric oxygen inhibits the HMGB1/RAGE signaling
pathway by upregulating mir-107 expression in human osteoarthritic
chondrocytes. Osteoarthritis Cartilage. 27:1372–1381.
2019.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Ai H, Zhou W, Wang Z, Qiong G, Chen Z and
Deng S: MicroRNAs-107 inhibited autophagy, proliferation, and
migration of breast cancer cells by targeting HMGB1. J Cell
Biochem. Dec 2, 2018 (Epub ahead of print). doi:
10.1002/jcb.28157.
|
|
36
|
Chen G, Ward MF, Sama AE and Wang H:
Extracellular HMGB1 as a proinflammatory cytokine. J Interferon
Cytokine Res. 24:329–333. 2004.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Li Z, Block MS, Vierkant RA, Fogarty ZC,
Winham SJ, Visscher DW, Kalli KR, Wang C and Goode EL: The
inflammatory microenvironment in epithelial ovarian cancer: A role
for TLR4 and MyD88 and related proteins. Tumour Biol.
37:13279–13286. 2016.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Bai M, Ma X, Li X, Wang X, Mei Q, Li X, Wu
Z and Han W: The Accomplices of NF-κB Lead to Radioresistance. Curr
Protein Pept Sci. 16:279–294. 2015.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Jung CH, Han AR, Chung HJ, Ha IH and Um
HD: Linarin inhibits radiation-induced cancer invasion by
downregulating MMP-9 expression via the suppression of NF-κB
activation in human non-small-cell lung cancer A549. Nat Prod Res.
33:3582–3586. 2019.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Liu YC, Chiang IT, Hsu FT and Hwang JJ:
Using NF-κB as a molecular target for theranostics in radiation
oncology research. Expert Rev Mol Diagn. 12:139–146.
2012.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Jiang C, Qu X, Ke H, Gong W, Chen R, Yang
W and Cheng Z: Association between the HMGB1/TLR4 signaling pathway
and the clinicopathological features of ovarian cancer. Mol Med
Rep. 18:3093–3098. 2018.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Tang H, Wang F, Zhou XF, Zhou J, Chen L,
Luo JD and Xu EC: Relationship between TLR4 and NF-κB p65 protein
expressions and clinical radiosensitivity of patients with
esophageal squamous cell carcinoma. Pak J Med Sci. 30:982–985.
2014.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Zhi-Feng W, Le-Yuan Z, Xiao-Hui Z, Ya-Bo
G, Jian-Ying Z, Yong H and Zhao-Chong Z: TLR4-dependent immune
response promotes radiation-induced liver disease by changing the
liver tissue interstitial microenvironment during liver cancer
radiotherapy. Radiat Res. 182:674–682. 2014.PubMed/NCBI View Article : Google Scholar
|