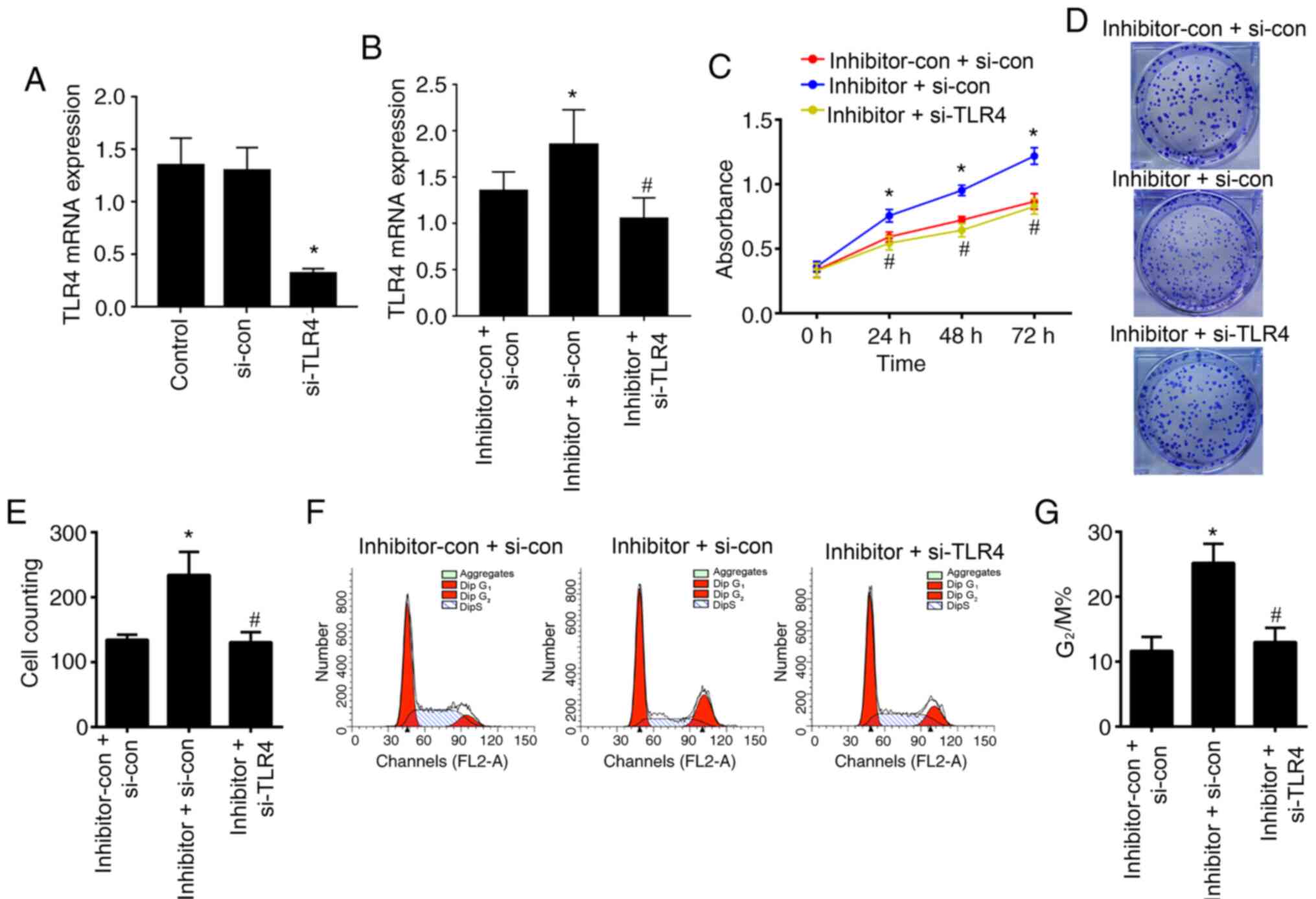
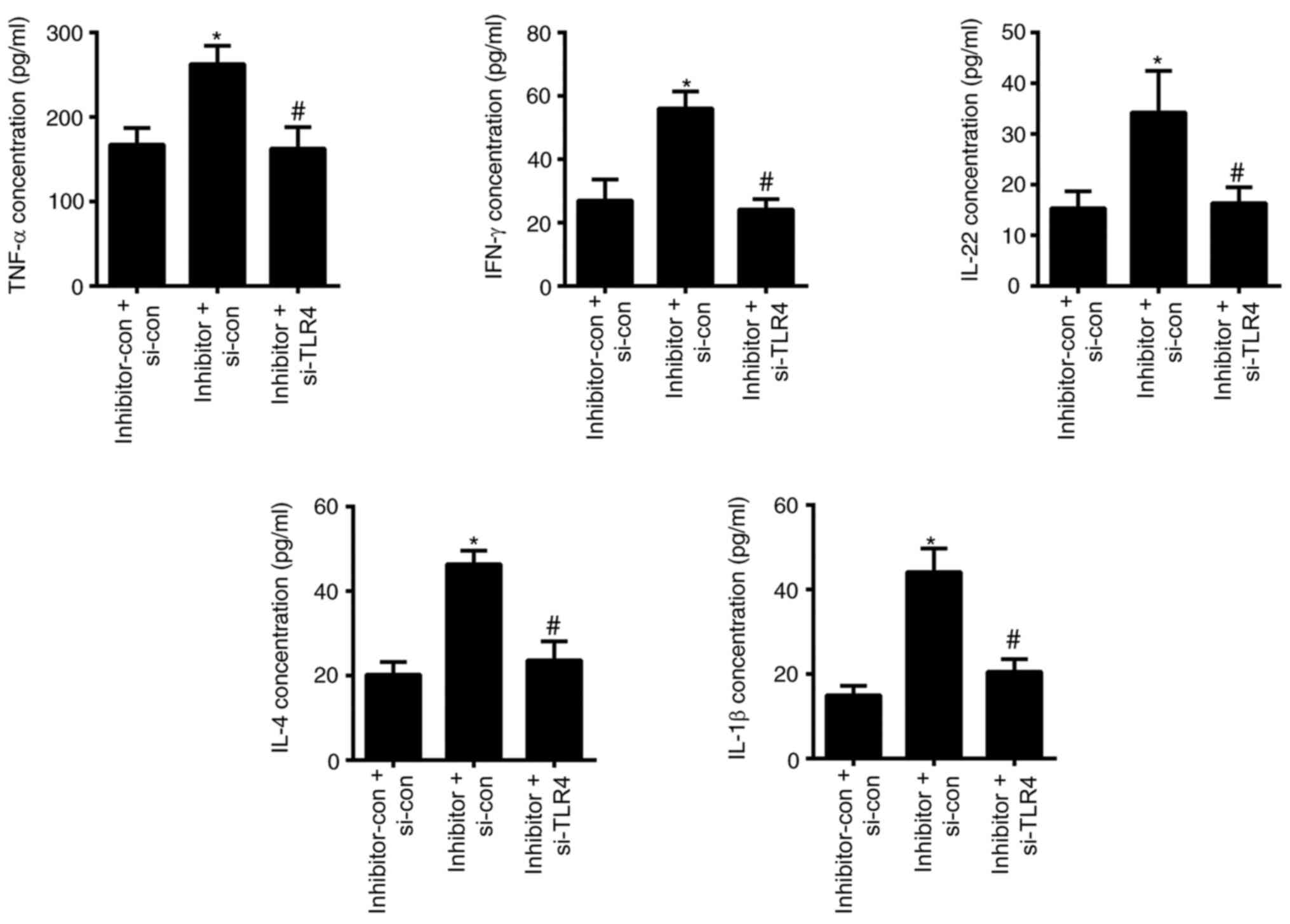
|
1
|
Langley RG, Krueger GG and Griffiths CE:
Psoriasis: Epidemiology, clinical features, and quality of life.
Ann Rheum Dis. 64 (Suppl 2):ii18–ii25. 2005.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Zhang K and Shen ZA: Research advances on
immunological properties and gene regulation of skin keratinocytes.
Zhonghua Shao Shang Za Zhi. 37:89–92. 2021.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
3
|
Lowes MA, Bowcock AM and Krueger JG:
Pathogenesis and therapy of psoriasis. Nature. 445:866–873.
2007.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Qureshi AA, Choi HK, Setty AR and Curhan
GC: Psoriasis and the risk of diabetes and hypertension: A
prospective study of US female nurses. Arch Dermatol. 145:379–382.
2009.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Guttman-Yassky E, Nograles KE and Krueger
JG: Contrasting pathogenesis of atopic dermatitis and
psoriasis-part I: Clinical and pathologic concepts. J Allergy Clin
Immunol. 127:1110–1118. 2011.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Rizwan M and Khan A: Association of
psoriasis and serum uric acid levels: A case control study.
Pakistan Armed Forces Med J. 69:408–412. 2019.
|
|
7
|
Pradyuth S, Rapalli VK, Gorantla S,
Waghule T, Dubey SK and Singhvi G: Insightful exploring of
microRNAs in psoriasis and its targeted topical delivery. Dermatol
Ther. 33(e14221)2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Valencia-Sanchez MA, Liu J, Hannon GJ and
Parker R: Control of translation and mRNA degradation by miRNAs and
siRNAs. Genes Dev. 20:515–524. 2006.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Rebane A and Akdis CA: MicroRNAs:
Essential players in the regulation of inflammation. J Allergy Clin
Immunol. 132:15–26. 2013.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Chang L, Liang J, Xia X and Chen X:
miRNA-126 enhances viability, colony formation, and migration of
keratinocytes HaCaT cells by regulating PI3 K/AKT signaling
pathway. Cell Biol Int. 43:182–191. 2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Covert MW, Leung TH, Gaston JE and
Baltimore D: Achieving stability of lipopolysaccharide-induced
NF-kappaB activation. Science. 309:1854–1857. 2005.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Kyriakis JM and Avruch J: Mammalian
mitogen-activated protein kinase signal transduction pathways
activated by stress and inflammation. Physiol Rev. 81:807–869.
2001.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wei W, Dejie L, Xiaojing S, Tiancheng W,
Yongguo C, Zhengtao Y and Naisheng Z: Magnolol inhibits the
inflammatory response in mouse mammary epithelial cells and a mouse
mastitis model. Inflammation. 38:16–26. 2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Lowes MA, Suárez-Fariñas M and Krueger JG:
Immunology of psoriasis. Annu Rev Immunol. 32:227–255.
2014.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Zhang C, Xiao C, Dang E, Cao J, Zhu Z, Fu
M, Yao X, Liu Y, Jin B, Wang G and Li W: CD100-Plexin-B2 promotes
the inflammation in psoriasis by activating NF-κB and the
inflammasome in keratinocytes. J Invest Dermatol. 138:375–383.
2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Wu AG, Conway J, Barazani L, Roy B, Cline
A and Pereira F: Is clear always clear? Comparison of psoriasis
area and severity index (PASI) and the Physician's global
assessment (PGA) in psoriasis clearance. Dermatol Ther (Heidelb).
10:1155–1163. 2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Cai Y, Fleming C and Yan J: New insights
of T cells in the pathogenesis of psoriasis. Cell Mol Immunol.
9:302–309. 2012.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Griffiths CE and Barker JN: Pathogenesis
and clinical features of psoriasis. Lancet. 370:263–271.
2007.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Han G, Williams CA, Salter K, Garl PJ, Li
AG and Wang XJ: A role for TGFbeta signaling in the pathogenesis of
psoriasis. J Invest Dermatol. 130:371–377. 2010.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Tattersall D, Scott CA, Gray C, Zicha D
and Kelsell DP: EKV mutant connexin 31 associated cell death is
mediated by ER stress. Hum Mol Genet. 18:4734–4745. 2009.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Kalali BN, Köllisch G, Mages J, Müller T,
Bauer S, Wagner H, Ring J, Lang R, Mempel M and Ollert M:
Double-stranded RNA induces an antiviral defense status in
epidermal keratinocytes through TLR3-, PKR-, and
MDA5/RIG-I-mediated differential signaling. J Immunol.
181:2694–2704. 2008.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Aldehlawi H, Usman S, Lalli A, Ahmad F,
Williams G, Teh MT and Waseem A: Serum lipids, retinoic acid and
phenol red differentially regulate expression of keratins K1, K10
and K2 in cultured keratinocytes. Sci Rep. 10(4829)2020.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Ni X and Lai Y: Keratinocyte: A trigger or
an executor of psoriasis? J Leukoc Biol. 108:485–491.
2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Tsuru Y, Jinnin M, Ichihara A, Fujisawa A,
Moriya C, Sakai K, Fukushima S and Ihn H: miR-424 levels in hair
shaft are increased in psoriatic patients. J Dermatol. 41:382–385.
2014.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Xu N, Brodin P, Wei T, Meisgen F, Eidsmo
L, Nagy N, Kemeny L, Ståhle M, Sonkoly E and Pivarcsi A: MiR-125b,
a microRNA downregulated in psoriasis, modulates keratinocyte
proliferation by targeting FGFR2. J Invest Dermatol. 131:1521–1529.
2011.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Li J, Qu W, Jiang Y, Sun Y, Cheng Y, Zou T
and Du S: miR-489 suppresses proliferation and invasion of human
bladder cancer cells. Oncol Res. 24:391–398. 2016.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Liu Q, Yang G and Qian Y: Loss of
MicroRNA-489-3p promotes osteosarcoma metastasis by activating
PAX3-MET pathway. Mol Carcinog. 56:1312–1321. 2017.PubMed/NCBI View
Article : Google Scholar
|
|
29
|
Jiang R, Zhang C, Gu R and Wu H:
MicroRNA-489-3p inhibits neurite growth by regulating PI3K/AKT
pathway in spinal cord injury. Pharmazie. 72:272–278.
2017.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Banerjee S, Thompson WE and Chowdhury I:
Emerging roles of microRNAs in the regulation of Toll-like receptor
(TLR)-signaling. Front Biosci (Landmark Ed). 26:771–796.
2021.PubMed/NCBI
|
|
31
|
Yamamoto M, Sato S, Hemmi H, Uematsu S,
Hoshino K, Kaisho T, Takeuchi O, Takeda K and Akira S: TRAM is
specifically involved in the Toll-like receptor 4-mediated
MyD88-independent signaling pathway. Nat Immunol. 4:1144–1150.
2003.PubMed/NCBI View
Article : Google Scholar
|
|
32
|
Faure E, Equils O, Sieling PA, Thomas L,
Zhang FX, Kirschning CJ, Polentarutti N, Muzio M and Arditi M:
Bacterial lipopolysaccharide activates NF-kappaB through Toll-like
receptor 4 (TLR-4) in cultured human dermal endothelial cells.
Differential expression of TLR-4 and TLR-2 in endothelial cells. J
Biol Chem. 275:11058–11063. 2000.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Muzio M, Natoli G, Saccani S, Levrero M
and Mantovani A: The human Toll signaling pathway: Divergence of
nuclear factor kappaB and JNK/SAPK activation upstream of tumor
necrosis factor receptor-associated factor 6 (TRAF6). J Exp Med.
187:2097–2101. 1998.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Hayden MS and Ghosh S: . NF-κB, the first
quarter-century: Remarkable progress and outstanding questions.
Genes Dev. 26:203–234. 2012.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Vallabhapurapu S and Karin M: Regulation
and function of NF-kappaB transcription factors in the immune
system. Ann Rev Immunol. 27:693–733. 2009.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Sun W, Gao Y, Yu X, Yuan Y, Yi J, Zhang Z,
Cheng Y, Li Y, Peng X and Cha X: ‘Psoriasis 1’ reduces
psoriasis-like skin inflammation by inhibiting the VDR-mediated
nuclear NF-κB and STAT signaling pathways. Mol Med Rep.
18:2733–2743. 2018.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Pivarcsi A, Meisgen F, Xu N, Ståhle M and
Sonkoly E: Changes in the level of serum microRNAs in patients with
psoriasis after antitumour necrosis factor-α therapy. Br J
Dermatol. 169:563–570. 2013.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Loubaki L, Chabot D, Paré I, Drouin M and
Bazin R: MiR-146a potentially promotes IVIg-mediated inhibition of
TLR4 signaling in LPS-activated human monocytes. Immunol Lett.
185:64–73. 2017.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Liu AH, Wu YT and Wang YP: MicroRNA-129-5p
inhibits the development of autoimmune encephalomyelitis-related
epilepsy by targeting HMGB1 through the TLR4/NF-κB signaling
pathway. Brain Res Bull. 132:139–149. 2017.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Nicolas JF: Psoriasis: How the epithelium
influences the immune response: Keratinocytes, dendritic cells and
T lymphocytes. Bull Acad Natl Med. 198:17–30. 2014.PubMed/NCBI(In French).
|