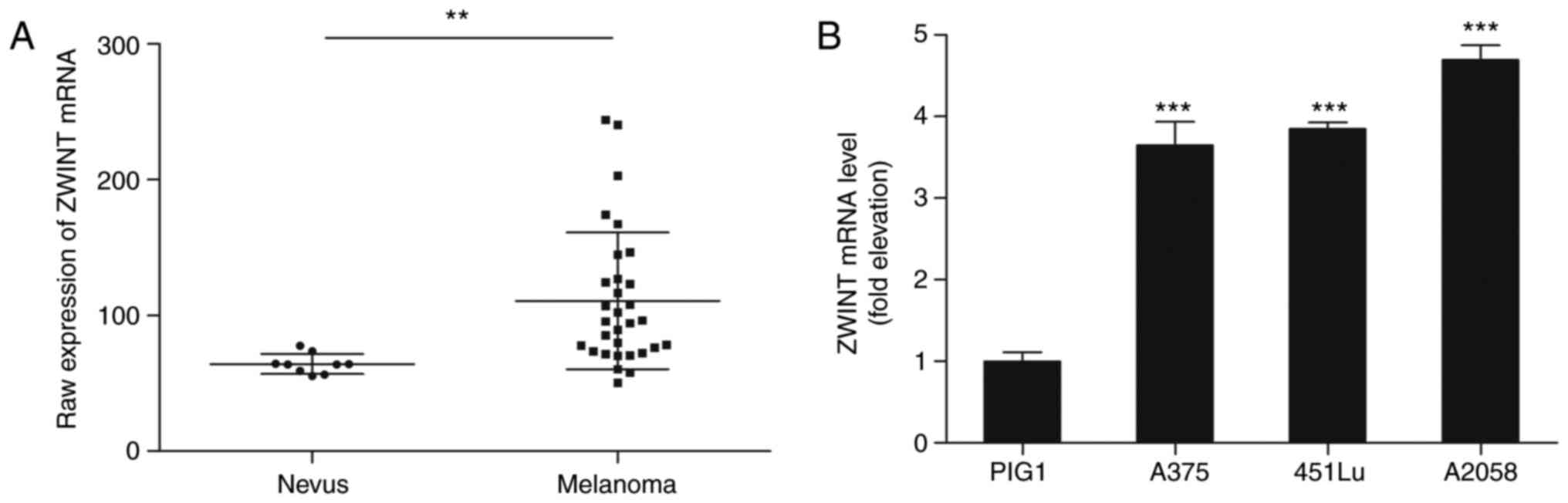
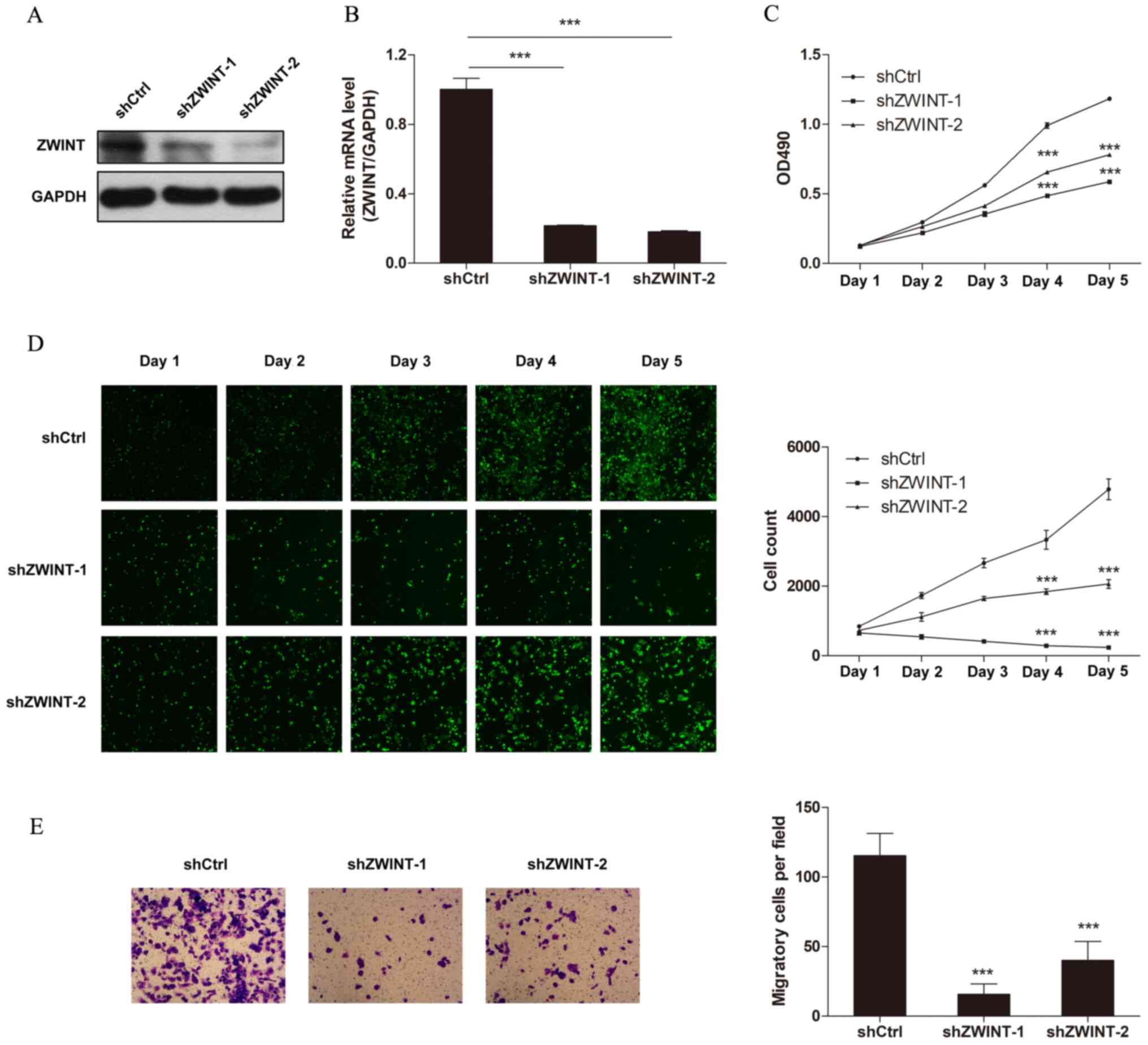
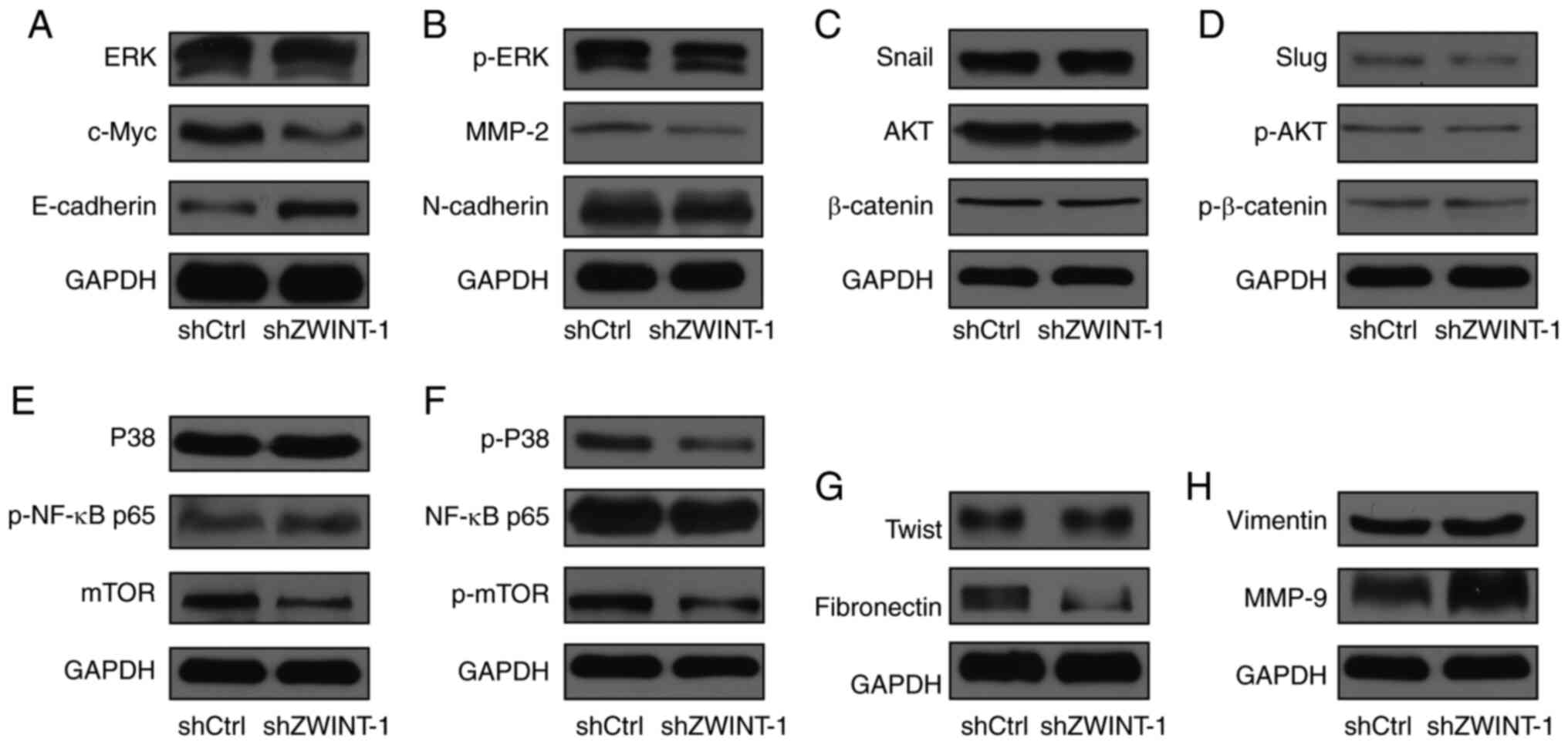
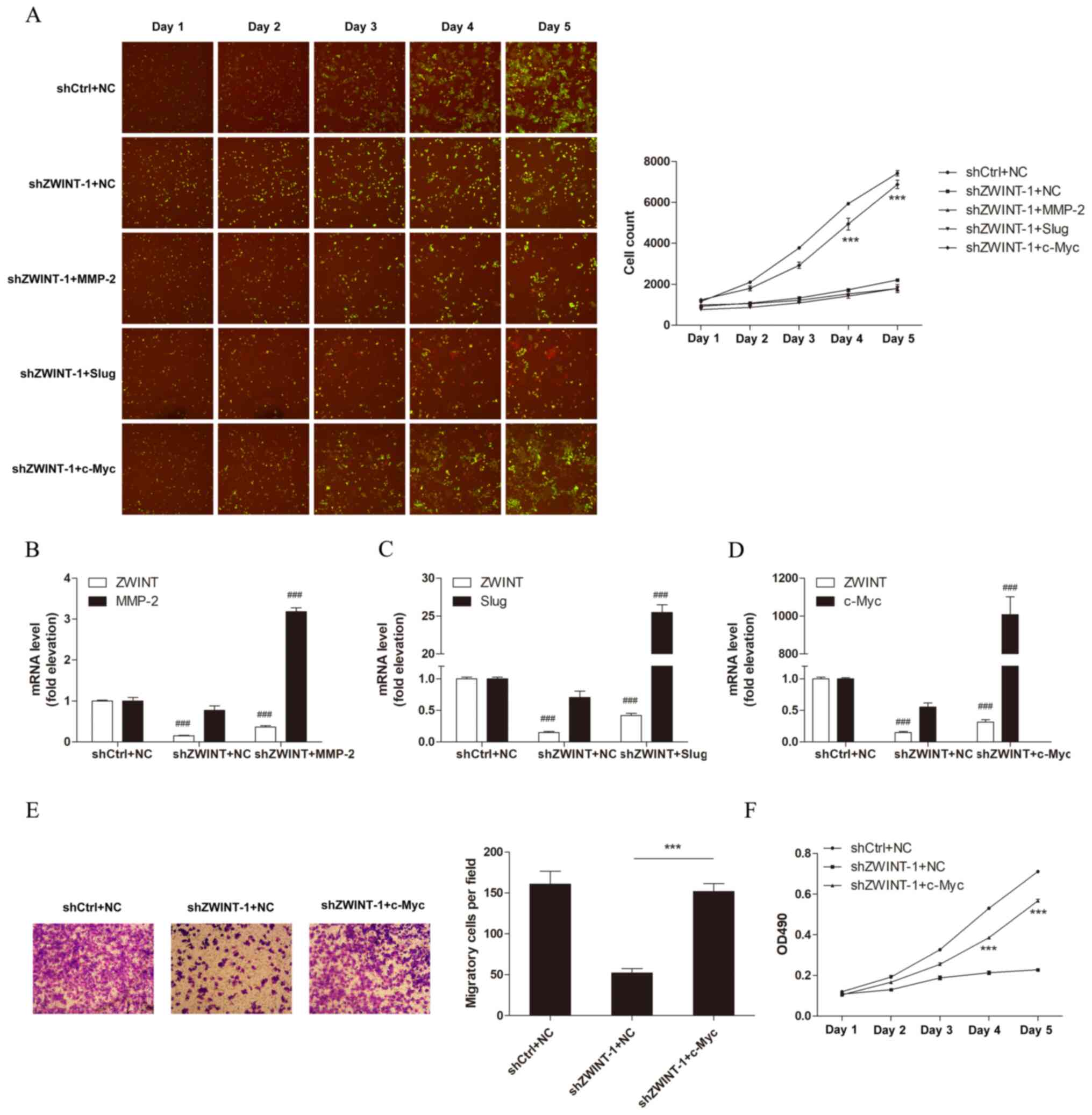
|
1
|
Coricovac D, Dehelean C, Moaca EA, Pinzaru
I, Bratu T, Navolan D and Boruga O: Cutaneous melanoma-a long road
from experimental models to clinical outcome: A review. Int J Mol
Sci. 19(1566)2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2020. CA Cancer J Clin. 70:7–30. 2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Rozeman EA, Dekker T, Haanen J and Blank
CU: Advanced melanoma: Current treatment options, biomarkers, and
future perspectives. Am J Clin Dermatol. 19:303–317.
2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Eggermont AM, Spatz A and Robert C:
Cutaneous melanoma. Lancet. 383:816–827. 2014.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Woo SD, Yeop YS, Chung WJ, Cho DH, Kim JS
and Su OJ: Zwint-1 is required for spindle assembly checkpoint
function and kinetochore-microtubule attachment during oocyte
meiosis. Sci Rep. 5(15431)2015.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Starr DA, Saffery R, Li Z, Simpson AE,
Choo KH, Yen TJ and Goldberg ML: HZwint-1, a novel human
kinetochore component that interacts with HZW10. J Cell Sci.
113:1939–1950. 2000.PubMed/NCBI
|
|
7
|
Vos LJ, Famulski JK and Chan GK: hZwint-1
bridges the inner and outer kinetochore: identification of the
kinetochore localization domain and the hZw10-interaction domain.
Biochem J. 436:157–168. 2011.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Famulski JK, Vos L, Sun X and Chan G:
Stable hZW10 kinetochore residency, mediated by hZwint-1
interaction, is essential for the mitotic checkpoint. J Cell Biol.
180:507–520. 2008.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Faesen AC, Thanasoula M, Maffini S, Breit
C, Müller F, van Gerwen S, Bange T and Musacchio A: Basis of
catalytic assembly of the mitotic checkpoint complex. Nature.
542:498–502. 2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Li HN, Zheng WH, Du YY, Wang G, Dong ML,
Yang ZF and Li XR: ZW10 interacting kinetochore protein may serve
as a prognostic biomarker for human breast cancer: An integrated
bioinformatics analysis. Oncol Lett. 19:2163–2174. 2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Ying H, Xu Z, Chen M, Zhou S, Liang X and
Cai X: Overexpression of Zwint predicts poor prognosis and promotes
the proliferation of hepatocellular carcinoma by regulating
cell-cycle-related proteins. Onco Targets Ther. 11:689–702.
2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Xu Z, Zhou Y, Cao Y, Dinh TL, Wan J and
Zhao M: Identification of candidate biomarkers and analysis of
prognostic values in ovarian cancer by integrated bioinformatics
analysis. Med Oncol. 33(130)2016.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Peng F, Li Q, Niu SQ, Shen GP, Luo Y, Chen
M and Bao Y: ZWINT is the next potential target for lung cancer
therapy. J Cancer Res Clin Oncol. 145:661–673. 2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Kabbarah O, Nogueira C, Feng B, Nazarian
RM, Bosenberg M, Wu M, Scott KL, Kwong LN, Xiao Y, Cordon-Cardo C,
et al: Integrative genome comparison of primary and metastatic
melanomas. PLoS One. 5(e10770)2010.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Ge R, Liu L, Dai W, Zhang W, Yang Y, Wang
H, Shi Q, Guo S, Yi X, Wang G, et al: Xeroderma pigmentosum group A
promotes autophagy to facilitate cisplatin resistance in melanoma
cells through the activation of PARP1. J Invest Dermatol.
136:1219–1228. 2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Zhou G, Shen M and Zhang Z: ZW10 binding
factor (ZWINT), a direct target of Mir-204, predicts poor survival
and promotes proliferation in breast cancer. Med Sci Monit.
26(e921659)2020.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Hoxhaj G and Manning BD: The PI3K-AKT
network at the interface of oncogenic signalling and cancer
metabolism. Nat Rev Cancer. 20:74–88. 2020.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Cargnello M and Roux PP: Activation and
function of the MAPKs and their substrates, the MAPK-activated
protein kinases. Microbiol Mol Biol Rev. 75:50–83. 2011.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Multhaupt HA, Leitinger B, Gullberg D and
Couchman JR: Extracellular matrix component signaling in cancer.
Adv Drug Deliv Rev. 97:28–40. 2016.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Hsieh AL, Walton ZE, Altman BJ, Stine ZE
and Dang CV: MYC and metabolism on the path to cancer. Semin Cell
Dev Biol. 43:11–21. 2015.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Dang CV: MYC on the path to cancer. Cell.
149:22–35. 2012.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Mannava S, Grachtchouk V, Wheeler LJ, Im
M, Zhuang D, Slavina EG, Mathews CK, Shewach DS and Nikiforov MA:
Direct role of nucleotide metabolism in C-MYC-dependent
proliferation of melanoma cells. Cell Cycle. 7:2392–2400.
2008.PubMed/NCBI View
Article : Google Scholar
|
|
24
|
Zhuang D, Mannava S, Grachtchouk V, Tang
WH, Patil S, Wawrzyniak JA, Berman AE, Giordano TJ, Prochownik EV,
Soengas MS and Nikiforov MA: C-MYC overexpression is required for
continuous suppression of oncogene-induced senescence in melanoma
cells. Oncogene. 27:6623–6634. 2008.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Beroukhim R, Mermel CH, Porter D, Wei G,
Raychaudhuri S, Donovan J, Barretina J, Boehm JS, Dobson J,
Urashima M, et al: The landscape of somatic copy-number alteration
across human cancers. Nature. 463:899–905. 2010.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Niitsu N, Okamoto M, Miura I and Hirano M:
Clinical features and prognosis of de novo diffuse large B-cell
lymphoma with t(14;18) and 8q24/c-MYC translocations. Leukemia.
23:777–783. 2009.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Rajan L, Broussard D, Lozano M, Lee CG,
Kozak CA and Dudley JP: The c-myc locus is a common integration
site in type B retrovirus-induced T-cell lymphomas. J Virol.
74:2466–2471. 2000.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Jiang C, Ito M, Piening V, Bruck K, Roeder
RG and Xiao H: TIP30 interacts with an estrogen receptor
alpha-interacting coactivator CIA and regulates c-myc
transcription. J Biol Chem. 279:27781–27789. 2004.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Sachdeva M, Zhu S, Wu F, Wu H, Walia V,
Kumar S, Elble R, Watabe K and Mo YY: p53 represses c-Myc through
induction of the tumor suppressor miR-145. Proc Natl Acad Sci U S
A. 106:3207–3212. 2009.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Wu H, Yang TY, Li Y, Ye WL, Liu F, He XS,
Wang JR, Gan WJ, Li XM, Zhang S, et al: Tumor necrosis factor
receptor-associated factor 6 promotes hepatocarcinogenesis by
interacting with histone deacetylase 3 to enhance c-Myc gene
expression and protein stability. Hepatology. 71:148–163.
2020.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Pan J, Deng Q, Jiang C, Wang X, Niu T, Li
H, Chen T, Jin J, Pan W, Cai X, et al: USP37 directly
deubiquitinates and stabilizes c-Myc in lung cancer. Oncogene.
34:3957–3967. 2015.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Patel JH, Du Y, Ard PG, Phillips C,
Carella B, Chen CJ, Rakowski C, Chatterjee C, Lieberman PM, Lane
WS, et al: The c-MYC oncoprotein is a substrate of the
acetyltransferases hGCN5/PCAF and TIP60. Mol Cell Biol.
24:10826–10834. 2004.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Dang CV, Reddy EP, Shokat KM and Soucek L:
Drugging the ‘undruggable’ cancer targets. Nat Rev Cancer.
17:502–508. 2017.PubMed/NCBI View Article : Google Scholar
|