Introduction
Lung cancer is one of the most common causes of
cancer-associated mortality worldwide, which affects ~1.6 million
individuals every year (1).
Non-small cell lung cancer (NSCLC) is the major subgroup of lung
cancer that results in a large number of cancer-associated
mortalities worldwide, which places substantial burden on patients,
their families and society (1,2).
Despite improvements in multiple therapeutic approaches for NSCLC
made over recent decades, including surgery, chemotherapy and
radiotherapy, the survival rate of NSCLC remains low due to a high
rate of distant metastasis and recurrence (3,4). Since
the underlying mechanism of tumorigenesis and distant metastasis
remain poorly understood, progress in the development of targeted
therapies to improve the NSCLC survival rate has been limited.
Osteoprotegerin (OPG) is a member of the tumor
necrosis factor receptor superfamily that suppresses
osteoclastogenesis by interrupting cell-to-cell interactions
(5). OPG is primarily expressed as
a circulating glycoprotein that is 401 amino acids long that was
initially been identified as a bone-remodeling agent, which
prevents osteolysis by inhibiting osteoclast differentiation and
bone resorption (2,6). However, a role of OPG in regulating
tumor metastasis has been previously revealed (7). Holen and Shipman reported that OPG can
bind to tumor necrosis factor-related apoptosis-inducing ligand
(TRAIL) to block the activation of death receptors 4 and 5(8). Therefore, OPG potentially serves an
anti-apoptotic role in OPG-expressing cells by harnessing the
mechanism of TRAIL induction (8,9).
Supporting this notion, previous studies demonstrated that OPG may
be released into the serum of the patients with colorectal or
myeloma to exert an anti-apoptotic effect on colorectal or myeloma
cancer cells (9,10). Weichhaus et al (11) also reported that suppression of OPG
has an important suppressive role in breast cancer cell metastasis.
Mechanistically, downregulation of OPG in breast cancer cells
resulted in reducing metastasis by inhibiting the expression of
proteases cathepsin D and matrix metalloproteinase 2(11). Yu et al (2) indicated that OPG levels are observably
upregulated in human lung cancer tissues compared with those in
normal tissues. In addition, overexpression of OPG in lung cancer
cells has been reported to contribute to cell invasion in
vitro (2). In particular, an
underlying regulatory interaction between OPG and microRNAs
(miRNAs/miRs) in tumor progression has been previously revealed
(12).
miRNAs are a class of small non-coding RNAs that
regulate gene expression by targeting mRNA (13). miRNAs may serve as effective
regulators of cell proliferation, apoptosis and carcinogenesis
(14-16).
Previous studies have demonstrated that compared with those in
healthy individuals, expression of miRNAs are frequently
dysregulated in patients with lung cancer, such that miRNAs may
serve as biomarkers with predictive diagnostic and prognostic
significance (17). Kuo et
al (18) reported that miR-33a
is an effective inhibitor of bone metastasis in lung cancer by
repressing the expression of parathyroid hormone-related protein.
In another study, Jia et al (12) suggested that estrogen regulates the
expression of OPG after transcription by inhibiting the expression
of miR-145 in human osteosarcoma cells.
Based on these previous observations aforementioned,
the present study aimed to assess OPG expression in lung cancer
tissues with or without distant metastasis and normal tissues. In
addition, the present study also investigated the potential
interaction between OPG and miRNAs in the regulation of lung cancer
cell invasion.
Materials and methods
Patients and specimens
The protocols of the present study were approved by
the Ethics Committee of Jiangxi Cancer Hospital (Nanchang, China).
All volunteers had provided written informed consent prior to
sample collection. Serum samples from healthy volunteers (sex, 24
males and 16 females; age, 54.45±7.27; age range, 42-73 years) and
patients with NSCLC (sex, 34 males and 13 females; age, 60.7±10.9;
age range, 42-83 years) was obtained at Jiangxi Cancer Hospital
(Nanchang, China) from Jan 2017 to May 2019. The
clinicopathological data of the patients with NSCLC are provided in
Table I. Bone metastasis of NSCLC
were diagnosed using computed tomography. The inclusion criteria
for patients with NSCLC were as follows: i) Aged <85 years; and
ii) imaging and pathological diagnosis of NSCLC stages I-IV
according to TNM staging system (19). No patients received chemo- or
radiotherapy when they were recruited. Exclusion criteria: i)
Patients with type I or II diabetes or cardiovascular disease; ii)
body mass index >30; and iii) patients with respiratory failure
(PaO2 <60 mmHg (20),
breathing room air at rest). The inclusion criteria for healthy
volunteers were: i) Aged <85 years; and ii) non-tumor
population. Exclusion criteria were: i) Patients with type I or II
diabetes or cardiovascular disease; ii) body mass index >30; and
iii) with respiratory failure (PaO2 <60 mmHg,
breathing room air at rest). All samples were collected, followed
by ELISA and reverse transcription-quantitative PCR (RT-qPCR)
analysis.
 | Table IClinical characteristics of patients
with primary non-small cell lung cancer. |
Table I
Clinical characteristics of patients
with primary non-small cell lung cancer.
| | Osteoprotegerin
expressiona | |
|---|
| Patient
characteristics | N | Low | High |
P-valueb |
|---|
| Sex | | | | 0.306 |
|
Female | 13 | 6 | 7 | |
|
Male | 34 | 15 | 18 | |
| Age (years) | | | | |
|
≤60 | 24 | 10 | 14 | 0.412 |
|
>60 | 23 | 11 | 12 | |
| Histological
type | | | | |
|
Lung
adenocarcinoma | 35 | 20 | 15 | 0.165 |
|
Lung
squamous cell carcinoma | 12 | 7 | 5 | |
| History of
smoking | | | | |
|
Yes | 27 | 12 | 15 | 0.081 |
|
No | 20 | 9 | 11 | |
| TNM stage | | | | |
|
I | 2 | 0 | 2 | 0.227 |
|
II | 3 | 1 | 2 | |
|
IIIA/IIIB/IIIC | 6 | 3 | 3 | |
|
IV | 36 | 15 | 21 | |
| Bone
metastasis | | | | 0.008 |
|
Yes | 16 | 5 | 11 | |
|
No | 31 | 13 | 18 | |
Cell lines
The human NSCLC cell lines A549 and H3122 were
obtained from the American Type Culture Collection, and grown in
DMEM (Gibco; Thermo Fisher Scientific, Inc.) supplemented with 10%
FBS (Gibco; Thermo Fisher Scientific, Inc.), 1% streptomycin and
penicillin (Gibco; Thermo Fisher Scientific, Inc.) in a cell
incubator at 37˚C with 5% CO2.
OPG overexpression
OPG was overexpressed using pcDNA3.1 plasmid vector
(Thermo Fisher Scientific, Inc.). In brief, total RNA were
extracted from A549 cells using TRIzol® kit (Thermo
Fisher Scientific, Inc.) and cDNA was synthesized by reverse
transcription PCR using iScripe™ cDNA Synthesis kit (Bio-Rad
Laboratories, Inc.) according to the manufacturer's protocols. The
full-length coding sequence of OPG (GenBank no. U94332.1) was
amplified from the cDNA using PCR and then inserted into the vector
at the restriction sites BamHI and XhaI to form the
recombinant plasmid pcDNA3-OPG. The specific primer sequences used
for amplifying OPG cDNA were: Forward,
5'-cgggatcccgATGAACAAGTTGCTGTGCTGC-3', reverse,
5'-gctctagagcTTATAAGCAGCTTATTTTTACTGATTGG-3'. pcDNA3-OPG plasmids
(5 µg) were transfected into A594 cells using
Lipofectamine® 3000 (Invitrogen; Thermo Fisher
Scientific, Inc.) for 48 h to overexpress OPG. pcDNA3 was used as
the negative control.
ELISA
OPG protein levels in serum samples from patients
with NSCLC with or without metastasis and healthy individuals were
assessed using an ELISA kit (cat. no. EHTNFRSF11B; Invitrogen;
Thermo Fisher Scientific, Inc.) as per the manufacturer's protocol.
The absorbance of samples was measured at 450 nm with a microplate
reader (Thermo Fisher Scientific, Inc.).
RT-qPCR
A total of 13 NSCLC-associated miRNAs were assessed
using RT-qPCR. TRIzol® kit (Thermo Fisher Scientific,
Inc.) was applied to isolate total RNA from A549 cells. Moloney's
murine leukemiavirus reverse transcriptase (cat. no. 2641A; Takara
Biotechnology Co., Ltd.) was used to perform reverse-transcription
reaction using Oligo(dT) primers (Vazyme Biotech Co., Ltd.) and
dNTPs (cat. no. 4035; Takara Bio, Inc.). The temperature protocol
for reverse transcription was 70˚C for 5 min, followed
by ice bath for 2 min and then 42˚C for 60 min. The specific
reverse transcription primers for miR-20a were synthesized as
previously described (21). qPCR
was performed using TB Green® Premix Ex Taq™ (cat. no.
RR420L; Takara Bio, Inc.) on a real-time PCR system (CFX96 Touch
system; Bio-Rad Laboratories, Inc.). The thermocycling protocol for
qPCR was 95˚C for 10 min, followed by 35 cycles of 95˚C for 15 sec
and 58˚C for 20 sec. The expression of OPG mRNA and 13 miRNAs was
normalized to those of β-actin and U6, respectively. The primer
sequences involved were listed in Table II. The qPCR results were analyzed
and calculated using the 2-∆∆Cq method (22).
 | Table IIPrimer sequences used in the present
study. |
Table II
Primer sequences used in the present
study.
| Name | Sequences
(5'→3') |
|---|
| OPG | F:
5'-TGGACATGCTAACCTCACCTTCG-3' |
| | R:
5'-GCCATTTTTTATTCGCCACAAAC-3' |
| β-actin | F:
5'-GTTGCCCTGAGGCTCTTTTCC-3' |
| | R:
5'-CCACCAGACAGCACTGTGTTG-3' |
| U6 | F:
5'-CTCGCTTCGGCAGCACA-3' |
| | R:
5'-AACGCTTCACGAATTTGCGT-3' |
| microRNA-20a | F:
5'-CGGCTAAAGTGCTTATAGTGCA-3' |
| | R:
5'-GTGCAGGGTCCGAGGT-3' |
| microRNA-200 | F:
5'-CGGCTAACACTGTCTGGTAACG-3' |
| | R:
5'-GTGCAGGGTCCGAGGT-3' |
| microRNA-34 | F:
5'-GCCGCAATCAGCAAGTATACT-3' |
| | R:
5'-GTGCAGGGTCCGAGGT-3' |
| microRNA-31 | F:
5'-CCGGAGGCAAGAUGCUGGC-3' |
| | R:
5'-GTGCAGGGTCCGAGGT-3' |
| microRNA-221 | F:
5'-CGGCAGCTACATTGTCTGCTGG-3' |
| | R:
5'-GTGCAGGGTCCGAGGT-3' |
| microRNA-222 | F:
5'-CCGGCTCAGTAGCCAGTGTAG-3' |
| | R:
5'-GTGCAGGGTCCGAGGT-3' |
| Let-7 | F:
5'-GCCTGAGGTAGTAAGTTGTA-3' |
| | R:
5'-GTGCAGGGTCCGAGGT-3' |
| microRNA-126 | F:
5'-CGGCTCGTACCGTGAGTAAT-3' |
| | R:
5'-GTGCAGGGTCCGAGGT-3' |
| microRNA-125 | F:
5'-CGGCCCTGAGACCCTTTAACC-3' |
| | R:
5'-GTGCAGGGTCCGAGGT-3' |
| microRNA-145 | F:
5'-GGCGTCCAGTTTTCCCAGGAAT-3' |
| | R:
5'-GTGCAGGGTCCGAGGT-3' |
| microRNA-21 | F:
5'-CCGGTAGCTTATCAGACTGAT-3' |
| | R:
5'-GTGCAGGGTCCGAGGT-3' |
| microRNA-146a | F:
5'-CGGCCTCTGAAATTCAGTTC-3' |
| | R:
5'-GTGCAGGGTCCGAGGT-3' |
| microRNA-141 | F:
5'-GCCTAACACTGTCTGGTAAAG-3' |
| | R:
5'-GTGCAGGGTCCGAGGT-3' |
miR-20a mimics and inhibitor
Human miR-20a mimics (5'-UAAAGUGCUUAUAGUGCAGGUAG-3')
and miR-20a inhibitor (5'-2'-O-methyl-CUACCUGCACUAUAAGCACUUUA-3')
were synthesized by Invitrogen (Thermo Fisher Scientific, Inc.),
and applied to overexpress or inhibit miR-20a expression in A549
cells using Lipofectamine® 3000 (Invitrogen; Thermo
Fisher Scientific, Inc.) following the manufacturer's protocol. The
overexpression/inhibition was identified using RT-qPCR analysis
after transfection (80 ng miR-20a mimics or inhibitor) for 48 h
(23). miRNA mimic Control
(5'-UUCUCCGAACGUGUCACGUUU-3'; Invitrogen; Thermo Fisher Scientific,
Inc.) and miRNA inhibitor control
(5'-2'-O-methyl-AAACGUGACACGUUCGGAGAA-3'; Invitrogen; Thermo Fisher
Scientific, Inc.) were used as control after transfection (80 ng)
for 48 h.
Western blot analysis
Total protein was extracted from A549 cells using
RIPA Buffer (Beijing Solarbio Science & Technology Co., Ltd.)
and protein concentration was quantified using a bicinchoninic acid
protein assay kit (Beijing Solarbio Science & Technology Co.,
Ltd.). In total, ~80 µg total protein was separated by 12% SDS-PAGE
and transferred onto PVDF membranes (Thermo Fisher Scientific,
Inc.) as previously described (24). To measure the protein expression
levels of OPG, the membranes were incubated with anti-OPG (cat. no.
ab11994; 1:1,000) and anti-β-actin (cat. no. ab179467, 1:5,000
dilution) primary antibodies (both from Abcam) for 2 h at room
temperature. HRP-conjugated goat anti-mouse IgG Fc (cat. no.
ab205719; 1:20,000 dilution; Abcam) or goat anti-rabbit IgG Fc
(cat. no. ab97200; 1:20,000 dilution; Abcam) was used to as the
secondary antibody to incubate the membranes for 2 h at room
temperature. An ECL chemiluminescence kit (EMD Millipore) was used
to visualize the specific blots and autoradiograms were quantified
by densitometry with Quantity One® software (version
4.6.9; Bio-Rad Laboratories. Inc.) after normalizing to
β-actin.
Transwell invasion assay
Transwell chambers (8-µm pore size) coated with
Matrigel were purchased from BD Biosciences (cat. no. 354481) and
applied to evaluate the cell invasion capacity (25). Briefly, after transfection with
pcDNA-OPG in the presence or absence of miR-20a inhibitor for 48 h,
3.0x104 A549 cells suspended in serum-free DMEM medium
were seeded onto the upper chambers. Complete DMEM medium with 10%
FBS was added into the lower chambers. Cells that had invaded into
the lower chamber 48 h later were fixed with 4% formaldehyde for 15
min and stained with crystal violet (0.1%) for 10 min at room
temperature. The number of invaded cells was counted from three
visual fields using an Olympus light microscope (Olympus
Corporation) under x200 magnification.
Experimental lung metastasis
assay
In total, 24 BALB/c nude female mice (weight,
20.6±2.04 g; age, 4-6 weeks; randomly divided into three groups of
eight mice) were obtained from Shanghai Model Organisms Center,
Inc. Mice were maintained in sterile conditions (temperature,
22-25˚C, humidity, 45-65%) under a 12-h light/dark cycle with free
access to water and food. All experimental protocols were approved
by the Animal Committee of Jiangxi Cancer Hospital (Nanchang,
China). A549 cells transfected with pcDNA-OPG with or without
miR-20a inhibitor for 48h (5x106/200 µl PBS) were
injected through the tail vein of the nude mice (26,27).
Lung tissues were collected 10 weeks later for analysis of lung
metastasis by gross anatomy after euthanasia. Euthanasia was
performed by CO2 inhalation at 20% V/min for 5 min
followed by cervical dislocation. Humane endpoints included
dehydration, weight loss >15%, hunched appearance and severe
lameness. None of mice exhibited these humane endpoint
criteria.
H&E (hematoxylin and eosin)
staining
Murine lung tissues were fixed with 4%
paraformaldehyde for 30 min at room temperature, dehydrated with an
ascending gradient of ethanol and embedded in paraffin. The tissues
were then cut into 5-µm sections, deparaffinized with xylene for 20
min and stained with hematoxylin for 1 h and then with eosin for 1
h (Beyotime Institute of Biotechnology) at room temperature. The
sections were photographed using a light microscope (BX41; Olympus
Corporation) and observed under x200 magnification.
Statistical analysis
The SPSS 16.0 statistical analysis software (SPSS,
Inc.) was used for data analysis. Values are expressed as the mean
± standard deviation from ≥ three separate experiments. Unpaired
Student's t-test (Fig. 1, Fig. 2 and Fig.
3) and one-way analysis of variance followed by Scheffé test
(Figs. 4 and 5) were applied to analyze differences
between two groups and those among ≥ three groups, respectively.
Data in Table I was analyzed using
Fisher's exact test. P<0.05 was considered to indicate a
statistically significant difference.
Results
OPG expression is significantly
upregulated in patients with NSCLC
Previous studies have demonstrated that upregulated
OPG expression in lung cancer tissues is associated with cancer
metastasis and worse survival (2,28). In
the present study, OPG levels were detected in the serum of
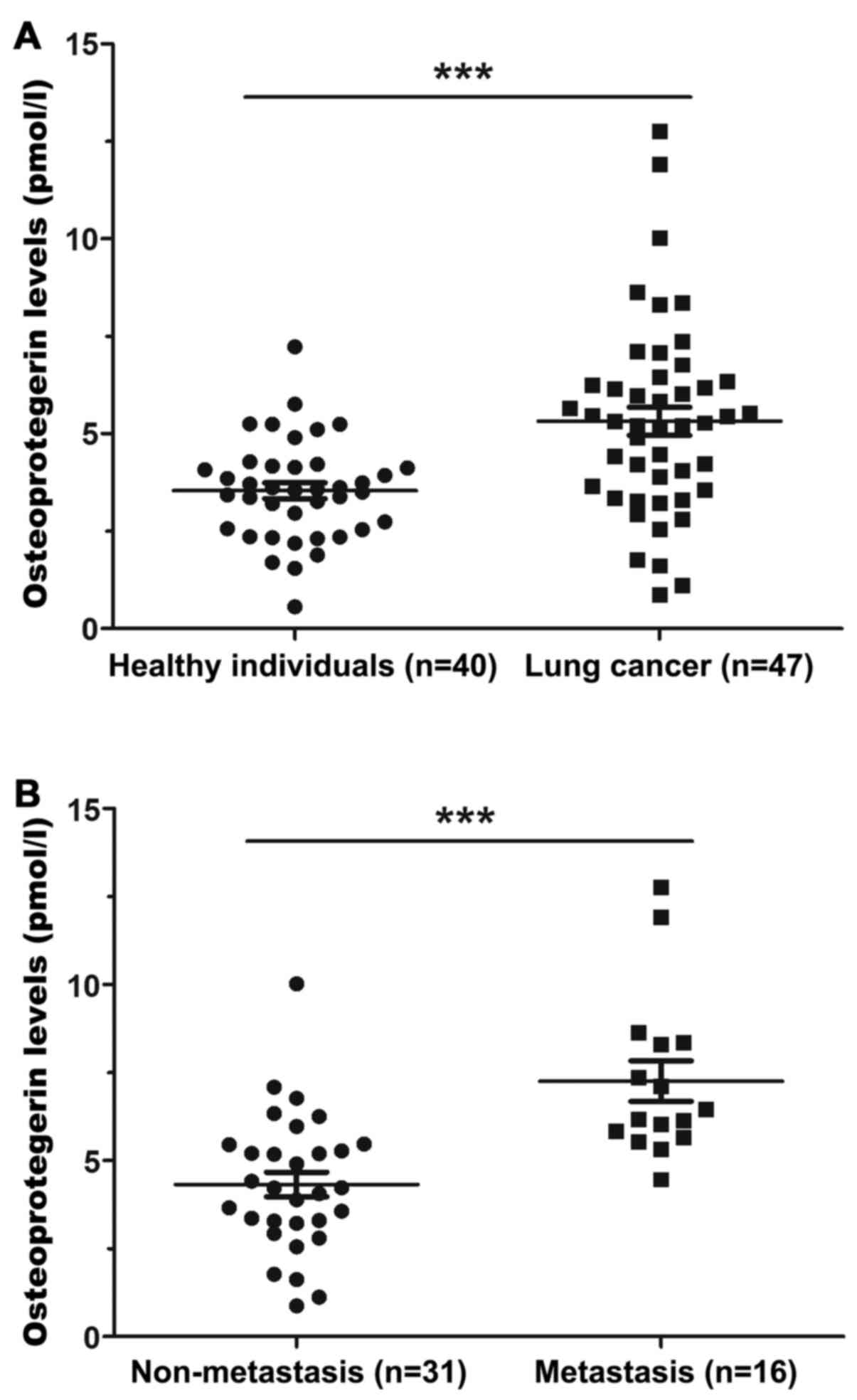
patients with NSCLC. ELISA results suggested that OPG levels were
significantly elevated in the serum of patients compared with those
in healthy individuals (Fig. 1A).
Furthermore, OPG levels were significantly increased in the serum
samples of patients with bone metastasis compared with those in the
serum of patients in non-metastatic group (Fig. 1B). In addition, no association was
found between OPG expression and the clinicopathological features
of patients with NSCLC, specifically sex, age, history of smoking,
histological type and TNM stage (Table
I).
OPG overexpression promotes NSCLC cell
invasion
The biological effects of OPG on NSCLC cell lines
were then explored further. A549 and H3122 cells were first
transfected with the appropriate pcDNA control or pcDNA-OPG
plasmids, before RT-qPCR and western blot analyses were performed
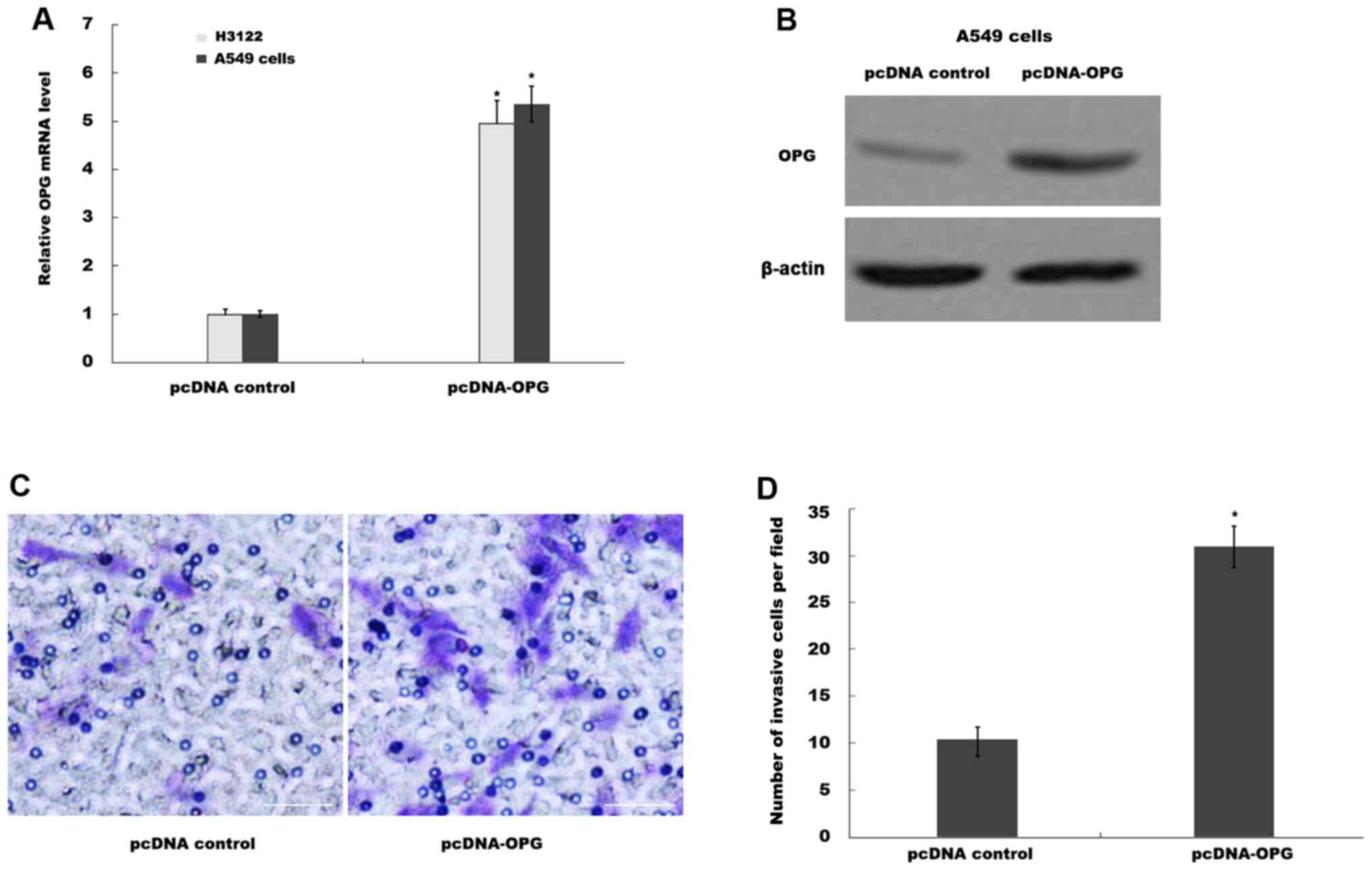
to verify overexpression in the cell lines. The mRNA levels of OPG
were significantly increased in H3122 and A549 cells transfected
with pcDNA-OPG compared with those transfected with pcDNA control
(Fig. 2A). This observation was
confirmed further by western blot analysis in A549 cells (Fig. 2B). Functionally, OPG overexpression
significantly increased the invasive ability of A549 cells compared
with that in the pcDNA control group (Fig. 2C and D).
Overexpression of OPG results in
increased miR-20a expression
Next, the potential mechanism of the function of OPG
in NSCLC cell invasion were explored. Given the important role of
miRNAs in NSCLC progression (29)
and the previously reported association between OPG and miRNAs
(30), it was investigated whether
OPG promoted NSCLC cell invasion in a miRNA-dependent manner. A
total of 13 NSCLC-associated miRNAs (29,31-33)
were selected as candidate targets that can be regulated by OPG
(Table SI). As presented in
Fig. 3A, the levels of miR-20a and
miR-21 were found to be significantly upregulated (fold change
>2.0 and P<0.05) whereas the levels of miR-200 and miR-34
were significantly downregulated (fold change <0.5 and
P<0.05) after OPG overexpression in A549 cells compared with
pcDNA control. The present study focused on miR-20a and its
possible biological function in A549 cells was investigated because
there was no effect of miR-21 in on OPG-induced cell invasion (data
not shown). Significantly increased expression of miR-20a in A549
cells after OPG overexpression was confirmed compared with those
transfected with the pcDNA control (Fig. 3B). Functionally, miR-20a
overexpression in A549 cells significantly promoted cell invasion
compared with transfected with the mimic control (Figs. 3C, D
and S1).
OPG promotes NSCLC metastasis by
upregulating miR-20a in vitro and in vivo
The results aforementioned suggested that OPG
possessed the potential to activate miR-20a expression in NSCLC
cells and promote cell invasion, whilst that miR-20a overexpression
alone also contributed to NSCLC cell invasion. It was therefore
speculated that OPG may promote NSCLC cell invasion by regulating
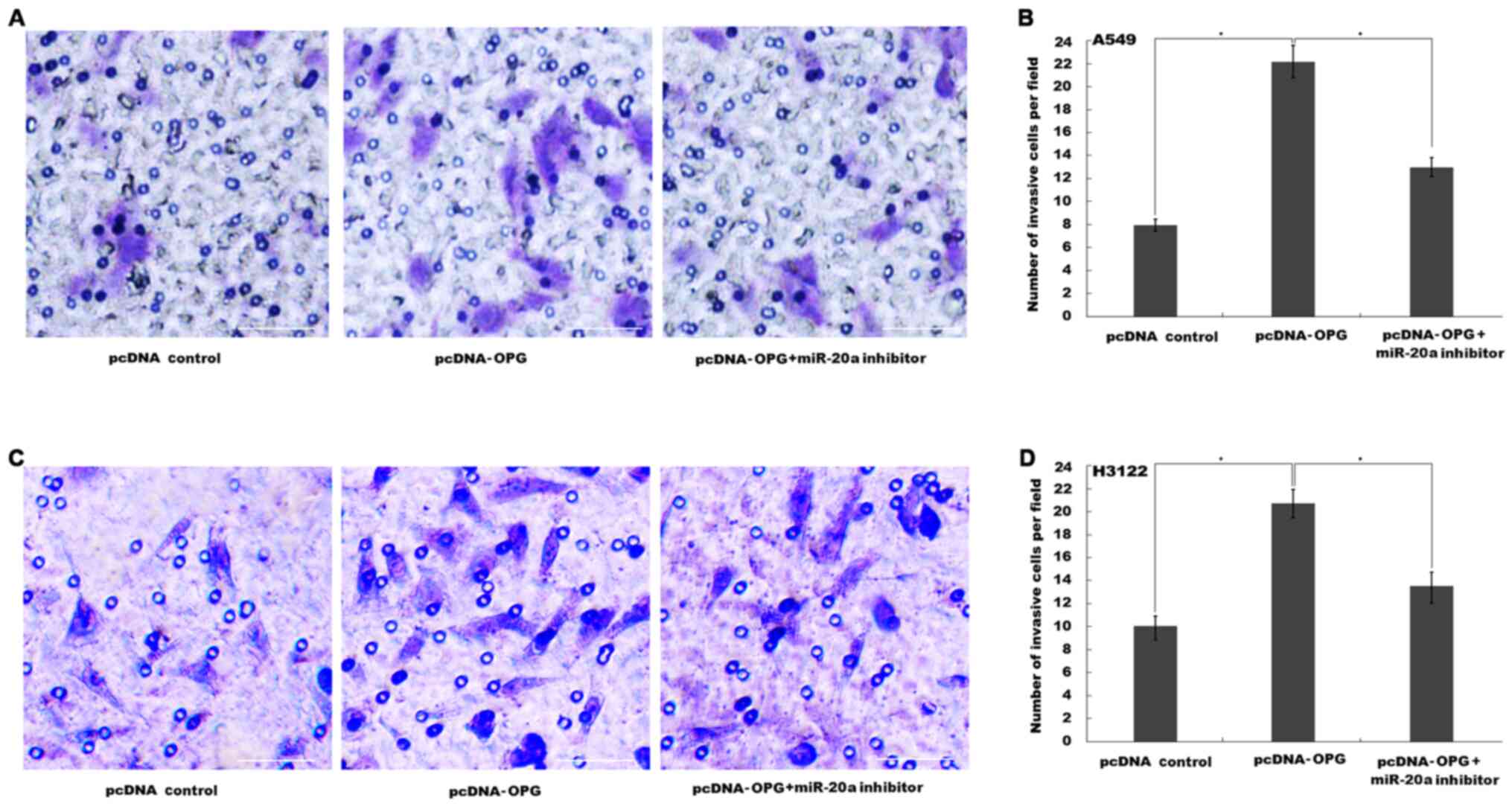
miR-20a expression. OPG overexpression significantly promoted A549
cell invasion, whereas miR-20a inhibition by its specific inhibitor
significantly reversed this effect (Figs. 4A, B
and S1). Similarly, OPG
overexpression also significantly promoted H3122 cells invasion,
which was significantly reversed by miR-20a inhibition (Figs. 4C, D
and S1).
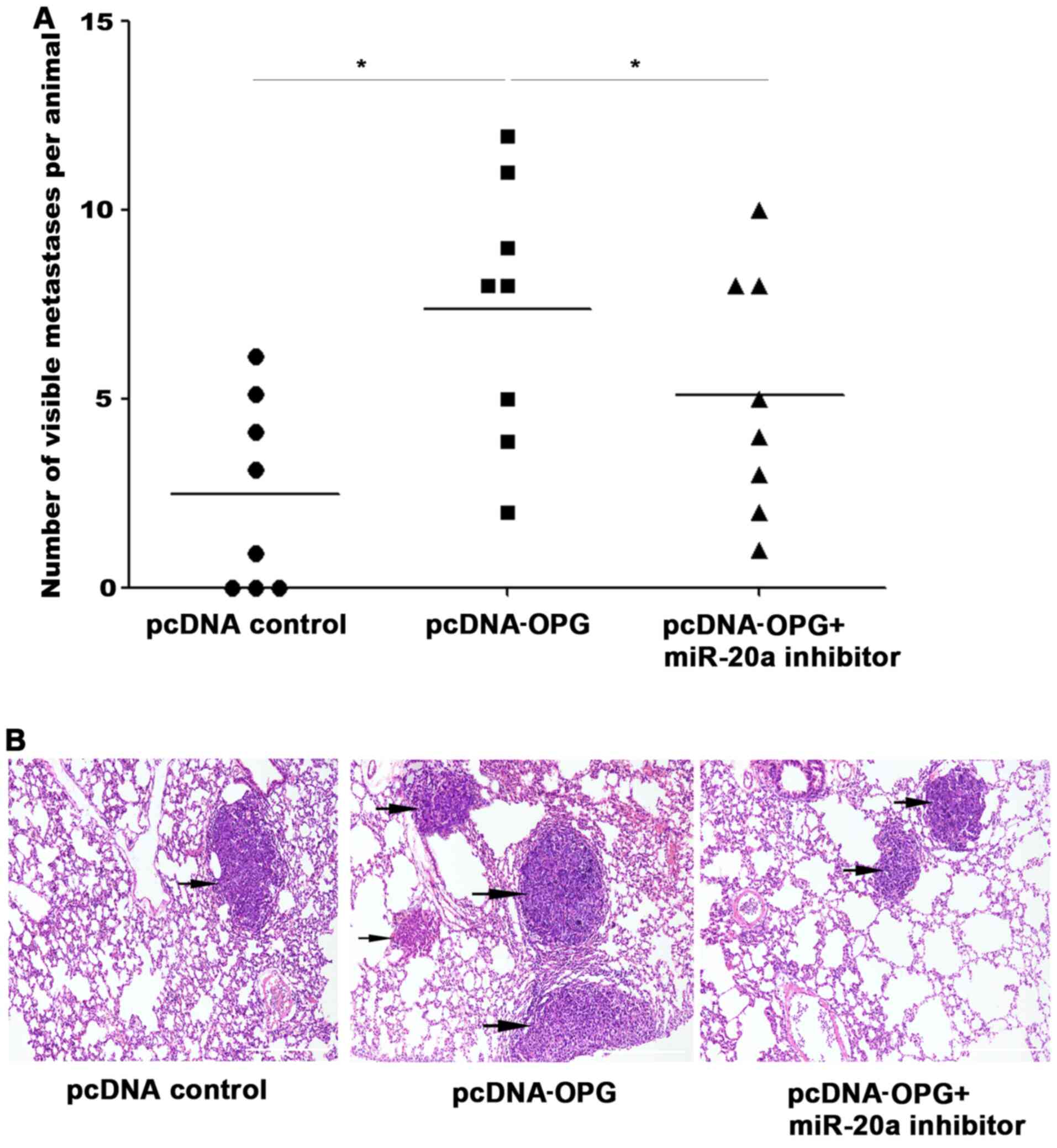
Results from the in vivo experiments appeared
to be consistent with the in vitro data. The number of lung
metastases in mice in the OPG overexpression group was increased
compared with those in the pcDNA control group. Conversely,
co-transfection with the miR-20a inhibitor resulted in the reversal
of this increase (Fig. 5A).
Histological analysis verified that this OPG-induced potentiation
of lung metastasis was partially repressed by inhibiting miR-20a
(Fig. 5B).
Discussion
OPG is normally produced by osteoblasts and stromal
cells and functions as a decoy receptor for RANKL to suppress
osteoclast differentiation and activation (28). This is achieved by blocking the
interaction between RANKL and its receptor RANK (34). OPG was initially defined as a
protein that regulates bone resorption (9,35).
However, over the past decade the role of OPG in the regulation of
carcinogenesis and tumor progression has been gradually revealed.
Yu et al (2) previously
analyzed OPG expression in lung cancer using The Cancer Genome
Atlas (TCGA) project lung cancer database, which revealed that OPG
expression is higher in lung cancer tissues compared. Data from
TCGA seem to exhibit similar trends compared with those that were
previously reported in a wide range of solid tumors types,
including prostate, breast, pancreatic and colorectal (2,36,37).
Yu et al (2) also
investigated the biological function of OPG in H3122 lung cancer
cells, which found that OPG overexpression enhanced the malignant
phenotypes of lung cancer cells in vitro.
Metastasis is a multifactorial and multistep dynamic
process, which are the primary causes of poor prognosis and
mortality associated with cancer (38). Patients with advanced lung cancer
are more prone to bone metastasis, which seriously affects their
daily activities and reduces their quality of life (39). Several bone formation markers,
including OPG, RANKL and osteocalcin exhibit lower levels in tumor
tissues without bone metastasis compared with those in tumor
tissues with lung cancer and bone metastasis (39), indicating that these molecules may
serve a potential role in lung cancer metastasis.
In the present study, OPG expression was first
assessed in the serum of patients with NSCLC, revealing that OPG
was increased in patients with NSCLC compared with those in healthy
individuals. In addition, OPG levels were also significantly
increased in the serum of patients with bone metastases compared
with that in patients with non-metastatic NSCLC. The downstream
biological effects of OPG on NSCLC cells were then investigated.
Overexpression of OPG in A549 cells promoted cell invasion compared
with that in the pcDNA control group. The relationship of OPG with
miRNA expression was next investigated, since aberrant OPG
expression has been reported to result in the dysregulation of
certain miRNA profiles (40).
Although the association between OPG and miRNAs has been described
in previous studies (12,41), the novelty of the present study lied
in the verification that upregulated OPG promotes lung cancer cell
invasion by increasing miR-20a expression. After overexpression of
OPG in A549 cells, the differentially expressed miRNAs associated
with NSCLC progression were examined using qPCR analysis, where the
results suggested that miR-20a levels were observably upregulated
following the overexpression of OPG. Overexpression of miR-20a in
A549 and H1322 cells markedly facilitated cell invasion compared
with that in the mimic control group. Additionally, both in
vitro and in vivo, miR-20a inhibition in A549 and H1322 cells
suppressed OPG-induced cell invasion and A549 metastasis. It is
noteworthy that, although the expression of miR-21, miR-200 and
miR-34 was also found to be dysregulated following the
overexpression of OPG, these miRNAs did not affect the function of
OPG in facilitating cell invasion (data not shown). The present
results suggest that the OPG/miR-20a axis is a functional
mechanistic node in the regulation of lung cancer metastasis, which
provides a novel target for the development of novel targeted lung
cancer therapies.
Supplementary Material
Effect of miR-20a mimics and its
miR-20a inhibitor on miR-20a expression in A549 and H3122 cells.
The expression levels of miR-20a in A549 cells after (A) miR-20a
mimics and (B) miR-20a inhibitor transfection. The expression
levels of miR-20a in H3122 cells after (C) miR-20a mimics and (D)
miR-20a inhibitor transfection. **P<0.01 vs. mimic
control or inhibitor control. Cont, control; miR, microRNA.
Non-small cell lung cancer-associated
miRNAs.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by the project of the
Jiangxi provincial Health commission (grant no. 20175400); the
National Natural Science Foundation of China (grant no. 81660453);
the Youth Foundation of Jiangxi Science and Technology Department
(grant no. 20161BAB215255) and the Six Talent Peaks Project of
Jiangsu Province (grant no. WS-053).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
KW and ZWT participated in the design of the main
research ideas and manuscript correction. ZTL and YC mainly
performed the experiments. YLC and CHL made statistical analysis of
the final data and agreed to publish the paper. All the authors
read and approved the final manuscript. CHL and KW confirm the
authenticity of raw data in the study.
Ethics approval and consent to
participate
The protocols of the present study were approved by
the Ethics Committee of Jiangxi Cancer Hospital (Nanchang, China).
All patients and volunteers provided written informed consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Molina JR, Yang P, Cassivi SD, Schild SE
and Adjei AA: Non-small cell lung cancer: Epidemiology, risk
factors, treatment, and survivorship. Mayo Clin Proc. 83:584–594.
2008.PubMed/NCBI View
Article : Google Scholar
|
|
2
|
Yu Z, Sanders AJ, Owen S, Cheng S, Yang X
and Jiang WG: Expression of osteoprotegrin is enhanced in lung
cancer tissues and promotes aggressive cellular traits in H3122
lung cancer cells. Anticancer Res. 37:4277–4283. 2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Yano T, Okamoto T, Fukuyama S and Maehara
Y: Therapeutic strategy for postoperative recurrence in patients
with non-small cell lung cancer. World J Clin Oncol. 5:1048–1054.
2014.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Coello MC, Luketich JD, Litle VR and
Godfrey TE: Prognostic significance of micrometastasis in
non-small-cell lung cancer. Clin Lung Cancer. 5:214–225.
2004.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Yasuda H, Shima N, Nakagawa N, Yamaguchi
K, Kinosaki M, Mochizuki S, Tomoyasu A, Yano K, Goto M, Murakami A,
et al: Osteoclast differentiation factor is a ligand for
osteoprotegerin/osteoclastogenesis-inhibitory factor and is
identical to TRANCE/RANKL. Proc Natl Acad Sci USA. 95:3597–3602.
1998.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Weichhaus M, Chung ST and Connelly L:
Osteoprotegerin in breast cancer: Beyond bone remodeling. Mol
Cancer. 14(117)2015.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Renema N, Navet B, Heymann MF, Lezot F and
Heymann D: RANK-RANKL signalling in cancer. Biosci Rep.
36(e00366)2016.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Holen I and Shipman CM: Role of
osteoprotegerin (OPG) in cancer. Clin Sci (Lond). 110:279–291.
2006.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Tsukamoto S, Ishikawa T, Iida S, Ishiguro
M, Mogushi K, Mizushima H, Uetake H, Tanaka H and Sugihara K:
Clinical significance of osteoprotegerin expression in human
colorectal cancer. Clin Cancer Res. 17:2444–2450. 2011.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Shipman CM and Croucher PI:
Osteoprotegerin is a soluble decoy receptor for tumor necrosis
factor-related apoptosis-inducing ligand/Apo2 ligand and can
function as a paracrine survival factor for human myeloma cells.
Cancer Res. 63:912–916. 2003.PubMed/NCBI
|
|
11
|
Weichhaus M, Segaran P, Renaud A, Geerts D
and Connelly L: Osteoprotegerin expression in triple-negative
breast cancer cells promotes metastasis. Cancer Med. 3:1112–1125.
2014.PubMed/NCBI View
Article : Google Scholar
|
|
12
|
Jia J, Zhou H, Zeng X and Feng S: Estrogen
stimulates osteoprotegerin expression via the suppression of
miR-145 expression in MG-63 cells. Mol Med Rep. 15:1539–1546.
2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Zhao Q, Li P, Ma J and Yu X: MicroRNAs in
lung cancer and lung cancer bone metastases: Biomarkers for early
diagnosis and targets for treatment. Recent Pat Anticancer Drug
Discov. 10:182–200. 2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Hwang HW and Mendell JT: MicroRNAs in cell
proliferation, cell death, and tumorigenesis. Br J Cancer.
94:776–780. 2006.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Saito Y, Suzuki H, Tsugawa H, Nakagawa I,
Matsuzaki J, Kanai Y and Hibi T: Chromatin remodeling at Alu
repeats by epigenetic treatment activates silenced microRNA-512-5p
with downregulation of Mcl-1 in human gastric cancer cells.
Oncogene. 28:2738–2744. 2009.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Bao B, Ali S, Kong D, Sarkar SH, Wang Z,
Banerjee S, Aboukameel A, Padhye S, Philip PA and Sarkar FH:
Anti-tumor activity of a novel compound-CDF is mediated by
regulating miR-21, miR-200, and PTEN in pancreatic cancer. PLoS
One. 6(e17850)2011.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Fanini F, Vannini I, Amadori D and Fabbri
M: Clinical implications of microRNAs in lung cancer. Semin Oncol.
38:776–780. 2011.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Kuo PL, Liao SH, Hung JY, Huang MS and Hsu
YL: MicroRNA-33a functions as a bone metastasis suppressor in lung
cancer by targeting parathyroid hormone related protein. Biochim
Biophys Acta. 1830:3756–3766. 2013.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Salgado RA, Snoeckx A, Spinhoven M, Op de
Beeck B, Corthouts B and Parizel PM: Update in non small-cell lung
cancer staging. JBR-BTR. 96:118–122. 2013.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Magnet FS, Majorski DS, Callegari J,
Schwarz SB, Schmoor C, Windisch W and Storre JH: Capillary
PO2 does not adequately reflect arterial PO2
in hypoxemic COPD patients. Int J Chron Obstruct Pulmon Dis.
12:2647–2653. 2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Chen C, Ridzon DA, Broomer AJ, Zhou Z, Lee
DH, Nguyen JT, Barbisin M, Xu NL, Mahuvakar VR, Andersen MR, et al:
Real-time quantification of microRNAs by stem-loop RT-PCR. Nucleic
Acids Res. 33(e179)2005.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Cao Y, Zhao D, Li P, Wang L, Qiao B, Qin
X, Li L and Wang Y: MicroRNA-181a-5p impedes IL-17-induced nonsmall
cell lung cancer proliferation and migration through targeting
VCAM-1. Cell Physiol Biochem. 42:346–356. 2017.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Kapinas K, Kessler CB, Ricks T, Gronowicz
G and Delany AM: miR-29 modulates WNT signaling in human
osteoblasts through a positive feedback loop. J Biol Chem.
285:25221–25231. 2010.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Connor KM, Hempel N, Nelson KK, Dabiri G,
Gamarra A, Belarmino J, Van De Water L, Mian BM and Melendez JA:
Manganese superoxide dismutase enhances the invasive and migratory
activity of tumor cells. Cancer Res. 67:10260–10267.
2007.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Zhuo W, Liu Y, Li S, Guo D, Sun Q, Jin J,
Rao X, Li M, Sun M, Jiang M, et al: Long noncoding RNA GMAN,
Up-regulated in gastric cancer tissues, is associated with
metastasis in patients and promotes translation of ephrin A1 by
competitively binding GMAN-AS. Gastroenterology. 156:676–691.e11.
2019.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Wang W, Ma J, Jin F and Liao J:
Hyperbranched polymer drug delivery treatment for lung metastasis
of salivary adenoid cystic carcinoma in nude mice. Exp Ther Med.
14:3105–3111. 2017.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Naumnik W, Plonska I, Ossolinska M,
Niklinski J and Naumnik B: Prognostic value of osteoprotegerin and
sRANKL in bronchoalveolar lavage fluid of patients with advanced
non-small cell lung cancer. Adv Exp Med Biol. 1047:1–6.
2018.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Weidle UH, Birzele F and Nopora A:
MicroRNAs as potential targets for therapeutic intervention with
metastasis of non-small cell lung cancer. Cancer Genomics
Proteomics. 16:99–119. 2019.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Kanzaki H, Wada S, Yamaguchi Y, Katsumata
Y, Itohiya K, Fukaya S, Miyamoto Y, Narimiya T, Noda K and Nakamura
Y: Compression and tension variably alter Osteoprotegerin
expression via miR-3198 in periodontal ligament cells. BMC Mol Cell
Biol. 20(6)2019.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Zhong S, Golpon H, Zardo P and Borlak J:
miRNAs in lung cancer. A systematic review identifies predictive
and prognostic miRNA candidates for precision medicine in lung
cancer. Transl Rese. 230:164–196. 2021.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Petrek H and Yu AM: MicroRNAs in non-small
cell lung cancer: Gene regulation, impact on cancer cellular
processes, and therapeutic potential. Pharmacol Res Perspect.
7(e00528)2019.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Xu X, Zhu S, Tao Z and Ye S: High
circulating miR-18a, miR-20a, and miR-92a expression correlates
with poor prognosis in patients with non-small cell lung cancer.
Cancer Med. 7:21–31. 2018.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Sisay M, Mengistu G and Edessa D: The
RANK/RANKL/OPG system in tumorigenesis and metastasis of cancer
stem cell: Potential targets for anticancer therapy. Onco Targets
Ther. 10:3801–3810. 2017.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Theoleyre S, Wittrant Y, Tat SK, Fortun Y,
Redini F and Heymann D: The molecular triad OPG/RANK/RANKL:
Involvement in the orchestration of pathophysiological bone
remodeling. Cytokine Growth Factor Rev. 15:457–475. 2004.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Brown JM, Corey E, Lee ZD, True LD, Yun
TJ, Tondravi M and Vessella RL: Osteoprotegerin and rank ligand
expression in prostate cancer. Urology. 57:611–616. 2001.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Shi W, Qiu W, Wang W, Zhou X, Zhong X,
Tian G and Deng A: Osteoprotegerin is up-regulated in pancreatic
cancers and correlates with cancer-associated new-onset diabetes.
Biosci Trends. 8:322–326. 2014.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Wu S, Pan Y, Mao Y, Chen Y and He Y:
Current progress and mechanisms of bone metastasis in lung cancer:
A narrative review. Transl Lung Cancer Res. 10:439–451.
2021.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Mountzios G, Ramfidis V, Terpos E and
Syrigos KN: Prognostic significance of bone markers in patients
with lung cancer metastatic to the skeleton: A review of published
data. Clin Lung Cancer. 12:341–349. 2011.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Pitari MR, Rossi M, Amodio N, Botta C,
Morelli E, Federico C, Gullà A, Caracciolo D, Di Martino MT,
Arbitrio M, et al: Inhibition of miR-21 restores RANKL/OPG ratio in
multiple myeloma-derived bone marrow stromal cells and impairs the
resorbing activity of mature osteoclasts. Oncotarget.
6:27343–27358. 2015.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Zhan FL, Liu XY and Wang XB: The Role of
MicroRNA-143-5p in the differentiation of dental pulp stem cells
into odontoblasts by targeting Runx2 via the OPG/RANKL signaling
pathway. J Cell Biochem. 119:536–546. 2018.PubMed/NCBI View Article : Google Scholar
|