1. Introduction
Rheumatoid arthritis (RA) is a multisystem
inflammatory autoimmune disease that destroys the surrounding
joints. The basic pathological change that occurs in RA is chronic
synovitis, which is accompanied by synovial cell proliferation,
inflammatory cell infiltration and the formation of vasospasms that
invade the cartilage and bone tissue of the subsynovial layer,
causing joint destruction (1-4).
When the disease progresses to an advanced stage, the joint tissue
is severely damaged and all joint function is lost. Furthermore, to
a certain extent, the lungs (5),
cardiovascular system (6,7), nervous system (8) and other organs (9,10) are
selectively affected, which seriously impairs the patients' quality
of life. RA is a disease that occurs globally. It may occur in
adults of any age but is mainly diagnosed in middle-aged females
(11,12). For the treatment of RA, timely and
effective control of disease progression are urgently required.
Early diagnosis and treatment may prevent bone and joint damage, as
well as reduce disability and suffering, which lays a foundation
for improving the quality of life of affected patients (13).
To date, the etiology and pathogenesis of RA have
remained to be fully elucidated. Due to its complex pathogenesis,
there is currently no ideal drug that is able to completely cure
RA. Accordingly, animal models of RA are an important resource for
studying and exploring the pathogenesis of RA, as well as
developing effective anti-inflammatory drugs. These animal models
may be classified in several ways according to species (mainly rat
and mouse), disease type (genetically engineered, induced or
spontaneous) and inciting agent (chemicals, collagen or exogenous
polysaccharides/proteins/proteoglycans) (14). Commonly used models include
collagen-induced arthritis (CIA) (15), proteoglycan-induced arthritis
(16), Staphylococcus
aureus-induced arthritis (17,18)
and genetically engineered arthritis mice (such as K/BxN mice)
(19,20). In addition, chimera models are
frequently used to test drugs with specific targets by transferring
corresponding human tissue samples to nonobese diabetes
(NOD)/severe combined immunodeficiency (SCID) mice. For instance,
the human RA synovium-cartilage-NOD/SCID mouse chimera model
(arthritis/SCID mouse chimera model) (21,22)
may be used to test the mechanism of synovial invasion of cartilage
and bone and the efficacy of related drugs. General practical
considerations in the use of various rodent disease models have
been reviewed by Bolon et al (14) and Williams (23). Caplazi et al (24) discussed mouse models of RA,
providing a wider perspective regarding systemically induced mouse
models of RA as well as the value of polyarthritis and
spontaneously occurring and genetically engineered models of
RA.
The present review mainly focused on providing a
detailed introduction to the CIA mouse model. This model is always
the first choice and the most widely used, as it may be generated
rapidly and inexpensively and is similar in pathogenesis to human
RA (25). However, this model has
several critical features that are frequently overlooked by
researchers. For instance, the use of the model has a limited scope
and the arthritis induction rate varies depending on the genetic
background of the mice (26).
Furthermore, there are several controversies regarding the modeling
process, such as when drugs should ideally be administered and how
to choose the best administration method. The present review
provides detailed knowledge related to these topics. With this
information, it is expected that researchers who are new to the
field or unfamiliar with this knowledge are able to avoid
unnecessary errors and select the appropriate model to obtain
reliable results.
2. Currently, the CIA model best reproduces
the clinical symptoms of RA
In 1977, Trentham et al (27) reported for the first time that the
immunization of rats with a human, chicken or rat type II collagen
(CII) emulsion in complete Freund's adjuvant (CFA) led to the
development of erosive polyarthritis, accompanied by an autoimmune
response to cartilage. This CIA model was reproduced in mice and
monkeys in 1980 and 1986, respectively (28,29).
Since CII is the main protein in articular cartilage, the immune
response generated by CII mainly targets the joints. The production
of CII-specific antibodies in mice is an important feature that has
also been reported in RA (30). The
clinical and histological appearance of CII-induced arthritis in
mice indicates that this is an ideal animal model for the
investigation of various immunogenetic traits in RA (31). For instance, mice immunized with CII
also produce rheumatoid factors (26,32).
Furthermore, the pathological features of both RA and CIA consist
of marked synovitis with cartilage degradation and bone erosion
(32).
Susceptibility to RA is closely related to certain
allelic subtypes of human leukocyte antigen (HLA)-DR and HLA-DQ
(23,33). The DR and DQ molecules expressed by
these subtypes are able to present autoantigen peptides such as CII
260-273 and may be recognized by a specific T-cell receptor (TCR),
thereby activating T cells (34).
The susceptibility of mice to CIA is also closely related to major
histocompatibility complex (MHC) class II molecules. The mouse
histocompatibility 2, class II antigen A, beta 1 (IA) gene is
homologous to HLA-DQ, and the mouse histocompatibility 2, class II
antigen E alpha (IE) gene is homologous to the HLA-DR gene
(35). It has been indicated that
polymorphisms of the β1 chain of the IA molecule determine
susceptibility to CIA and that the IE molecule is also involved in
the regulation of CIA-related pathological processes, which are
related to the incidence and severity of CIA (36). Sequence analysis of IA, IE, DQ and
DR indicated that in humans and mice, the MHC class II molecule β1
chain expressed by the RA/CIA allelic subtypes frequently had the
conserved sequence
glutamine-lysine/arginine-arginine-alanine-alanine, known as the
shared epitope. The spatial structure formed by the side chain
molecules of these adjacent amino acid residues is the key binding
site of the antigenic peptide. The MHC II molecules expressed by
H-2q mice are IAq and IEq, and the MHC class II molecule that
recognizes the presented CII antigen is IAq; therefore, IAq mice
(such as DBA/1 and B10.Q mice) are frequently used to generate CIA
animal models (31,37).
Accordingly, the establishment of CIA requires
heterogeneous (bovine or chicken) CII to immunize susceptible mice
two times. The disease-related CII antigenic peptide must contain a
core peptide. CII is a homotrimer composed of three A1 chains
containing 1,018 amino acids each. Within these chains, the CII
260-270 segment contains functional amino acids that are able to
bind to MHC II molecules and be recognized by the TCR; they are
known as the core antigen peptides and are related to the
occurrence of RA and CIA. The core peptide is easily degraded at
room temperature, which is why it is necessary to work at 4˚C when
emulsifying collagen. As the H-2q haplotype has strong
susceptibility to CII core peptides, it is able to induce strong
T-cell proliferation and CIA production (38). Excessive activation of T cells,
increased secretion of cytokines and production of CII-specific
antibodies are the major factors involved in the pathogenesis of
CIA (39). The CIA mouse model is
mainly induced by CD4+ T cells and MHC class
II-restricted T cells (40). In
this disease model, T helper type 1 (Th1) cytokines are secreted
and damage is mediated by the cellular immune response. T cells
interact with antigen-presenting cells, activate lymphocytes and
stimulate monocytes/macrophages to release numerous inflammatory
factors, such as IL-1β and TNF-α, leading to CIA inflammation,
hyperplasia of the synovial lining, neoangiogenesis, pannus
formation and the destruction of cartilage and bone (41,42).
Of note, Bolon et al (14) and Williams (23), among others, reported that
immunization of mice with heterologous type II collagen usually
leads to a relatively acute and self-remitting form of arthritis.
By contrast, immunization with autologous collagen results in more
severe and prolonged arthritis that is probably more reminiscent of
human RA. However, due to the low affinity of a specific epitope of
murine collagen (CII256-270) for IAq, autologous type II collagen
is less arthritogenic than heterologous collagen, resulting in a
low level of CII-specific T-cell activation. The use of
heterologous CII protein is still recommended as the first choice
from a comprehensive perspective (43). In addition, CII-responsive T cells
can only be induced when the amount of heterologous CII is large
enough. An appropriate amount of CII should be used when immunizing
mice; the optimal CII concentration is specified further below.
The CIA mouse model also has certain drawbacks. The
joints of mice are small and CIA in mice exhibits a variable
disease pattern. By contrast, rat arthritis models offer much
larger specimen sizes and the distribution and extent of
inflammatory changes in rat CIA joints are more reproducible
(14). Therefore, modeling with
mice requires more standardized modeling protocols and operations
to ensure the replicability of the model as much as possible. Mice
have the advantage of costing less than rats (44); thus, it is prudent to use a CIA
mouse model in the initial screening of anti-RA drugs unless the
purpose of the experiment has specific requirements for specimen
size or other restrictions that would preclude mice.
In general, among the various mouse models, the CIA
model is most similar to RA in terms of pathogenesis and clinical
characteristics. It is relatively stable and is an ideal and
internationally recognized arthritis model for studying the
pathogenesis of RA and screening drugs for RA treatment.
3. Establishment of the CIA mouse model
Selecting the sex and age of
animals
Both females and males may be used for the CIA model
(42,45,46).
However, it is reported that in mice, CIA tends to be more severe
in males than in females (45,47).
Overall, the choice of sex in mice depends on the purpose of the
experiment. If the study requires to exclude estrogen-related
factors, male mice must be selected. If there are no special
requirements in the experiment, the use of female mice is also
possible. Additionally, there is currently no report clearly
stating that female mice cannot be used. In addition, the
prevalence of RA is significantly higher in females than in males
(48). In the CIA model, the
incidence of arthritis may reach 100% in male mice (49) and >80% in female mice.
Mice older than 7-8 weeks may be used to construct
the CIA model. The immune system of mice does not mature until the
mice reach 7-8 weeks of age. It has been reported that 8-12 weeks
of age is the optimal age for starting mouse experiments (42). However, certain studies suggested
that the use of older mice (10-14 weeks of age) is important, as
the incidence of CIA is higher in older mice (36,50).
Of note, the incidence and severity of disease in aged mice also
decreases when the mice are too old (36). The ideal compromise should be to use
mice aged approximately 10-12 weeks for these experiments.
Preparation of related materials
The main reagents required are as follows: Complete
Freund's adjuvant (CFA; 5 ml); incomplete Freund's adjuvant (IFA; 5
ml); and immunization-grade chick type II collagen (10 mg),
lyophilized. In the first immunization, it is recommended to use an
emulsion of CFA and chicken CII. In the second immunization, an
emulsion of IFA and chicken CII is recommended.
Freund used a water-in-mineral oil emulsion
containing Mycobacterium tuberculosis (Mtb) cells; this was
termed CFA. CFA is primarily used to help activate immune cells and
produce antibodies that target the desired antigen. A possible
adverse reaction to CFA is the formation of an epithelioid
granuloma at the injection site. Conversely, the use of IFA, which
lacks the mycobacterial component, does not cause such acute
granulomatous adverse reactions (51). It is the CII antigen, not Mtb, that
must be constantly present after activation of the immune cells.
CFA is not necessary after the first immunization because the
immune cells are already active at this point. Thus, it is better
to use IFA in the second immunization.
Glacial acetic acid (0.1 M) should be prepared one
day in advance, filtered and sterilized. Glacial acetic acid should
be added to the chicken CII reagent bottle and a pipette should be
used to gently and evenly mix the reagents (final concentration, 4
g/l). The mixed solution should be stored at 4˚C overnight to fully
dissolve the chicken CII. In addition, the following supplies
should be prepared in advance: A shaver, mouse holders, a
homogenizer, 1-ml syringes, an ice box, a bottle of sterile saline,
medical cotton balls, 6-well plates and marker pens. In order to
produce the immunizing emulsion, CII should be combined with an
equal volume of CFA or IFA (the final concentration of CII is 2 g/l
in 0.05 M glacial acetic acid solution) by mixing them in short
bursts using an homogenizer. Specific instructions for
emulsification are as follows: Freund's adjuvant (2.5 ml) is added
to the syringe, followed by the addition of an equal volume of
collagen solution and stirring at a low speed while adding the
collagen solution in a dropwise manner. Subsequently, mixing at
full power (~1,000 g) for 2-3 min at a time with intervals for 0.5
min is performed until a stable emulsion is obtained. The whole
emulsification process requires to be performed on ice to prevent
heating. The final emulsion should be sufficiently thick not to
drip out of the vessel when it is inverted (36). A good way to test its quality is to
place a drop onto a surface of water. If the emulsion does not
disperse, it has the correct consistency [for more specific
directions, please refer to this published protocol (36)].
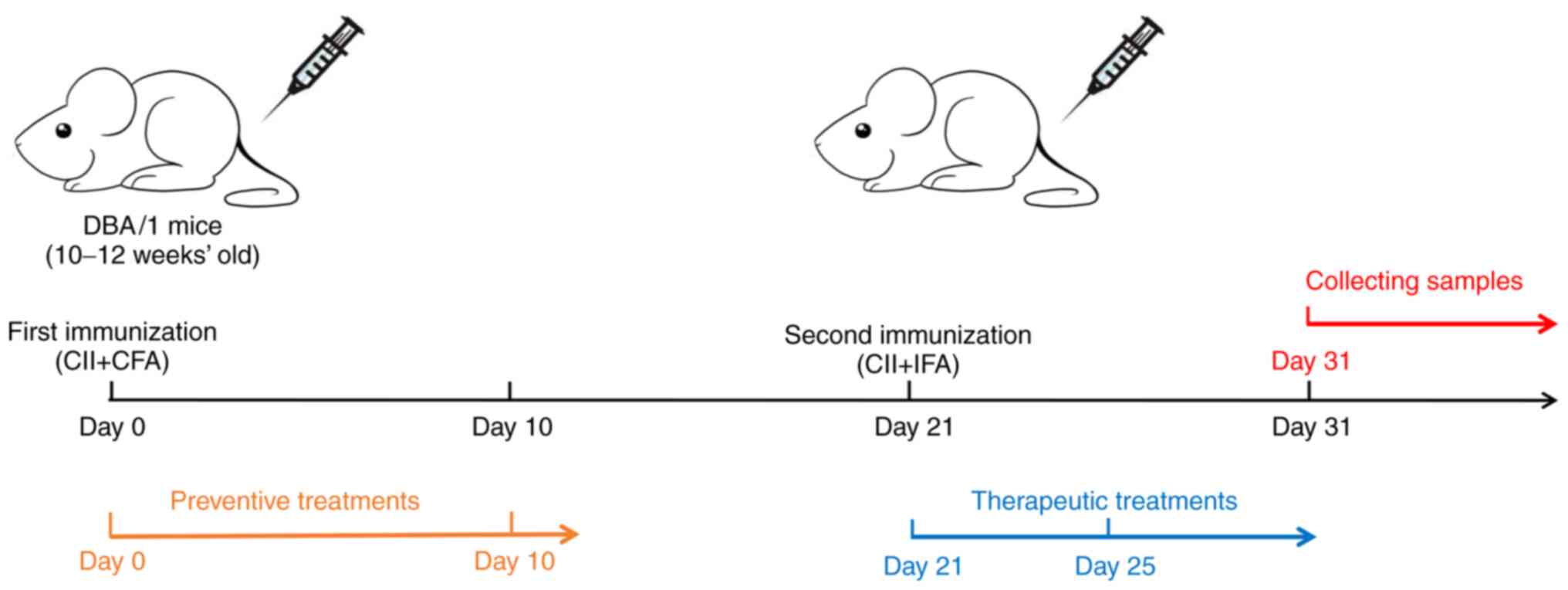
Induction of the mouse CIA model
As mentioned above, equal volumes of CFA (4 mg/ml)
and chicken CII should be mixed and fully emulsified at low
temperatures with a homogenizer until the obtained liquid has a
milky white appearance and is insoluble in water. Subsequently, the
emulsifier should be dispensed into a 1-ml syringe (the dispensed
emulsifier requires to be kept at 4˚C and should be used within 6
h). The mouse should be placed in the holder and the hair should be
removed from the base of the tail with a shaver or depilatory
cream. The tail of the mouse should be disinfected with alcohol on
a cotton ball. Each mouse should be subcutaneously injected with
100 µl of the emulsion. After the injection, the injection site of
the mouse should be disinfected with alcohol on a cotton ball. A
bulge should form at the base of the tail due to the accumulation
of the drug and the operator must gently rub the bulge with their
fingers until the bulge is no longer present; this step promotes
the complete absorption of the emulsifier. The mouse is then
removed from the holder and placed back in the cage. On the 21st
day after the first immunization, the same method is used to inject
the emulsion of IFA and chicken CII (100 µl per mouse). The mental
state, activity, food/water intake and body weight of the mice, as
well as the presence of redness and swelling of their paws, should
be recorded everyday starting on day 0 (D0) (36). A flowchart for the generation of CIA
model mice is provided in Fig.
1.
4. Evaluation indices for the CIA model
Success rate of model
establishment
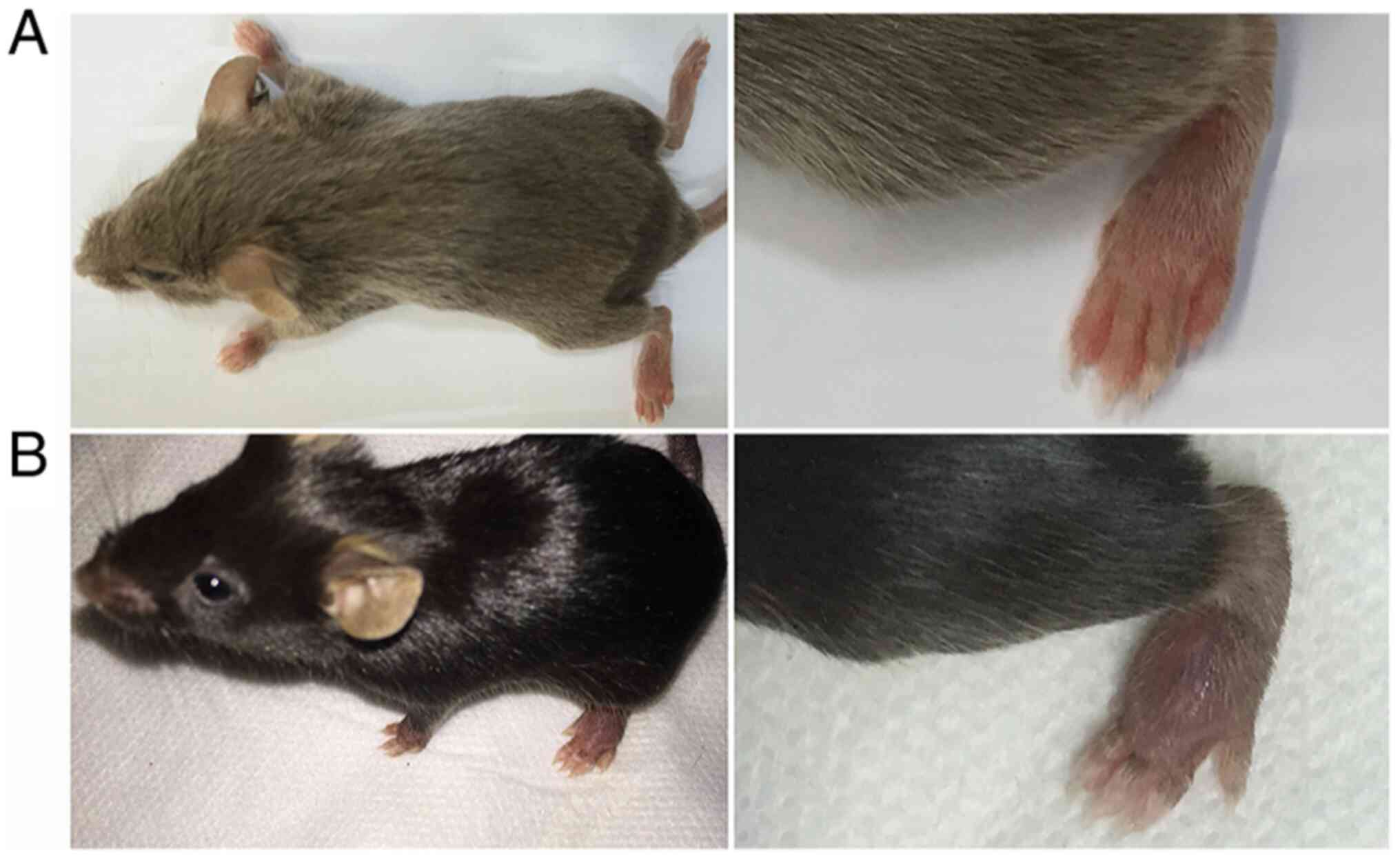
The development of CIA depends on the mouse strain.
The DBA/1 strain (H-2q) haplotypes exhibit the greatest
susceptibility (52) and are the
most commonly used strain for the CIA model in the preclinical
testing of potential antiarthritic drugs (42,53).
Mice with a C57BL/6 (H-2b) background may also be
used for the CIA model (54). For
example, certain studies may use transgenic mice to assess the
impact of a specific gene on the pathogenesis of arthritis. As most
transgenic mice are C57BL/6, those that are C57BL/6 may be used to
construct CIA models. However, the C57BL/6 (H-2b) strain is
relatively resistant to CIA. After repeated trials, it was verified
that the induction rate of mice with a C57BL/6 background was low
(approximately 15-65%), while that of mice with a DBA/1 background
reached 80-100% (Fig. 2).
Therefore, the use of mice with a DBA/1 background is recommended
for experiments without special requirements.
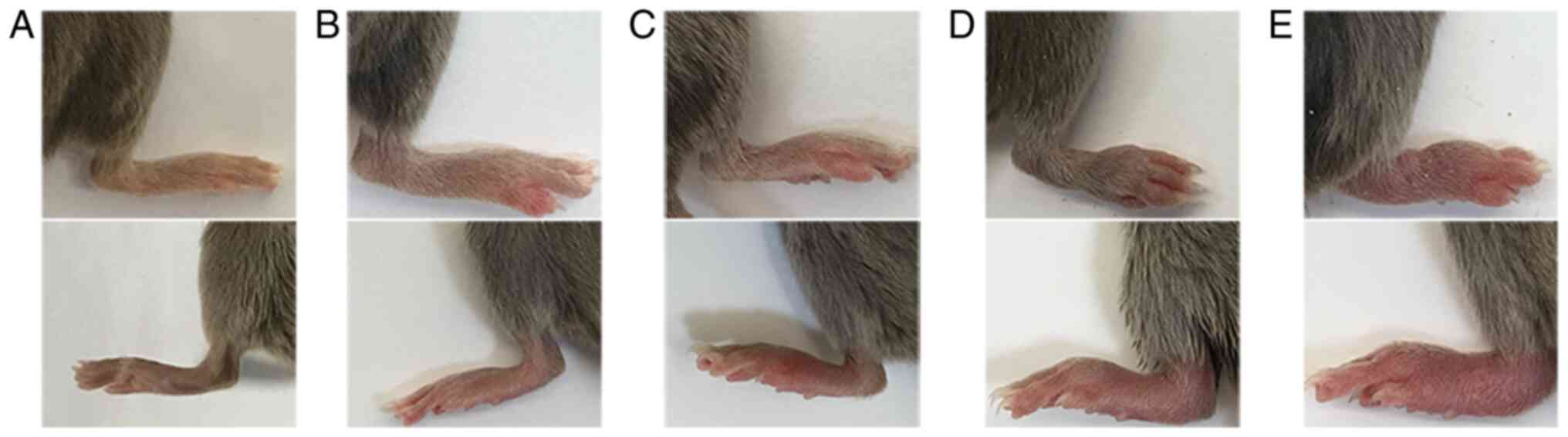
CIA mouse arthritis score
Arthritis scores are determined on the extremities
of each mouse and the score for each mouse is the sum of the limb
scores (range, 0-16 points, with a maximum score in each mouse of
16). The scoring criteria were adapted from multiple references
(55,56), as presented in Table I. Representative images
demonstrating the specific changes in the joints for each score are
presented in Fig. 3. The mean
arthritis index of each group of mice is calculated as follows:
Mean arthritis index = (total arthritis score of all mice in the
group)/(the number of mice in the group).
 | Table IScoring criteria for collagen-induced
arthritis in mice. |
Table I
Scoring criteria for collagen-induced
arthritis in mice.
| Severity score | Degree of
inflammation |
|---|
| 0 | No erythema or
swelling |
| 1 | Erythema and mild
swelling confined to the tarsals, ankle, or paw joint, with mild
swelling at single limb |
| 2 | Erythema and mild
swelling extending from the ankle to the tarsals or erythema and
mild swelling of more than one toe |
| 3 | Erythema and
moderate swelling extending from the ankle to the metatarsal joints
or the whole paw with swelling and obvious erythema |
| 4 | Erythema and the
whole paw with severe swelling encompass the ankle, foot and
digits, or ankylosis of the limb, and dysfunction of the above
joints |
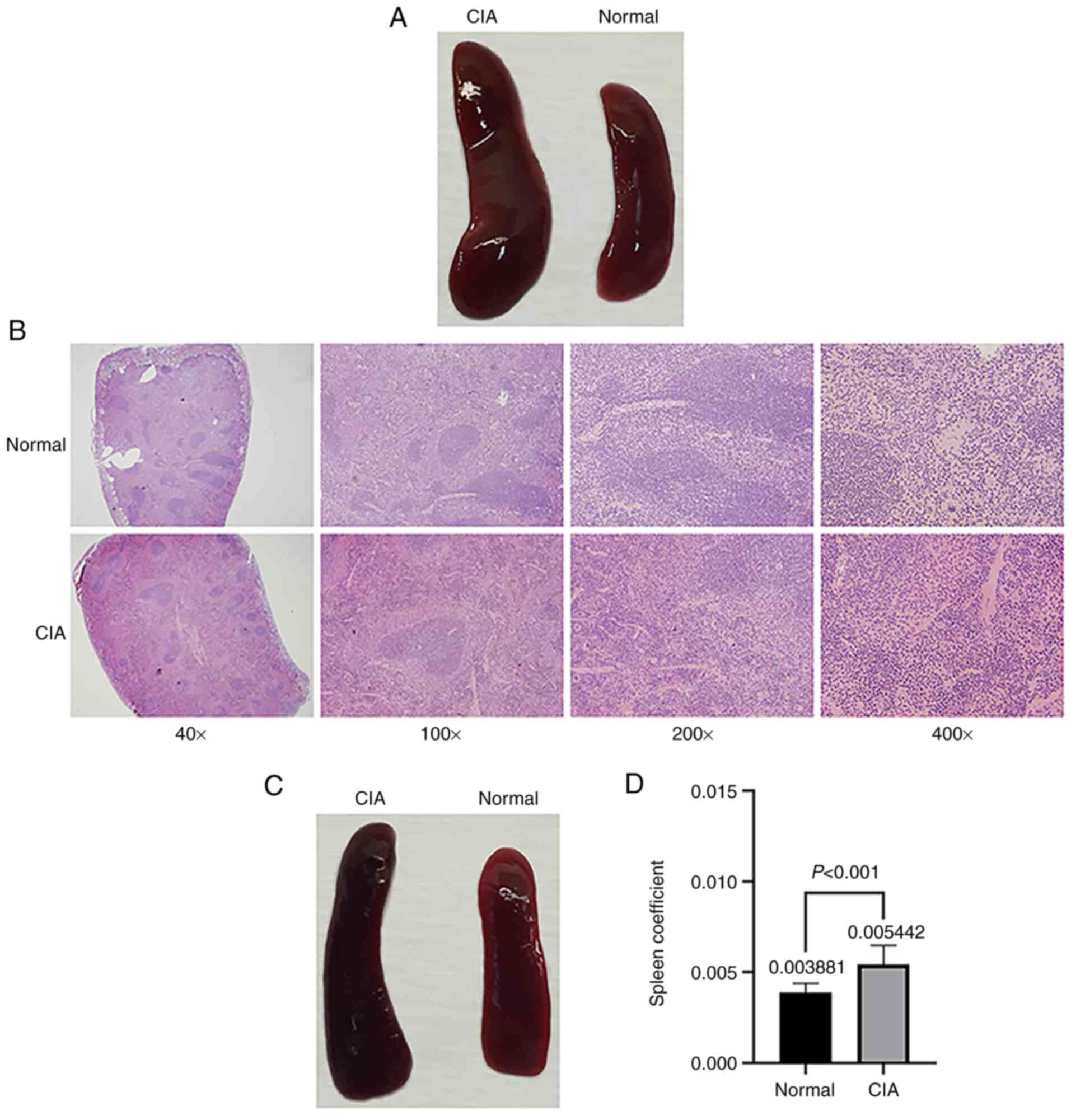
Sample collection
Samples and data are collected to detect and
evaluate the severity of arthritis in mice with CIA. In general,
mouse body weight, joint tissues, serum, peripheral blood
lymphocytes and spleen samples are routinely collected (57). The following results are very clear
and have been verified in published studies (36,57).
On D31, the spleens of CIA mice are significantly larger than those
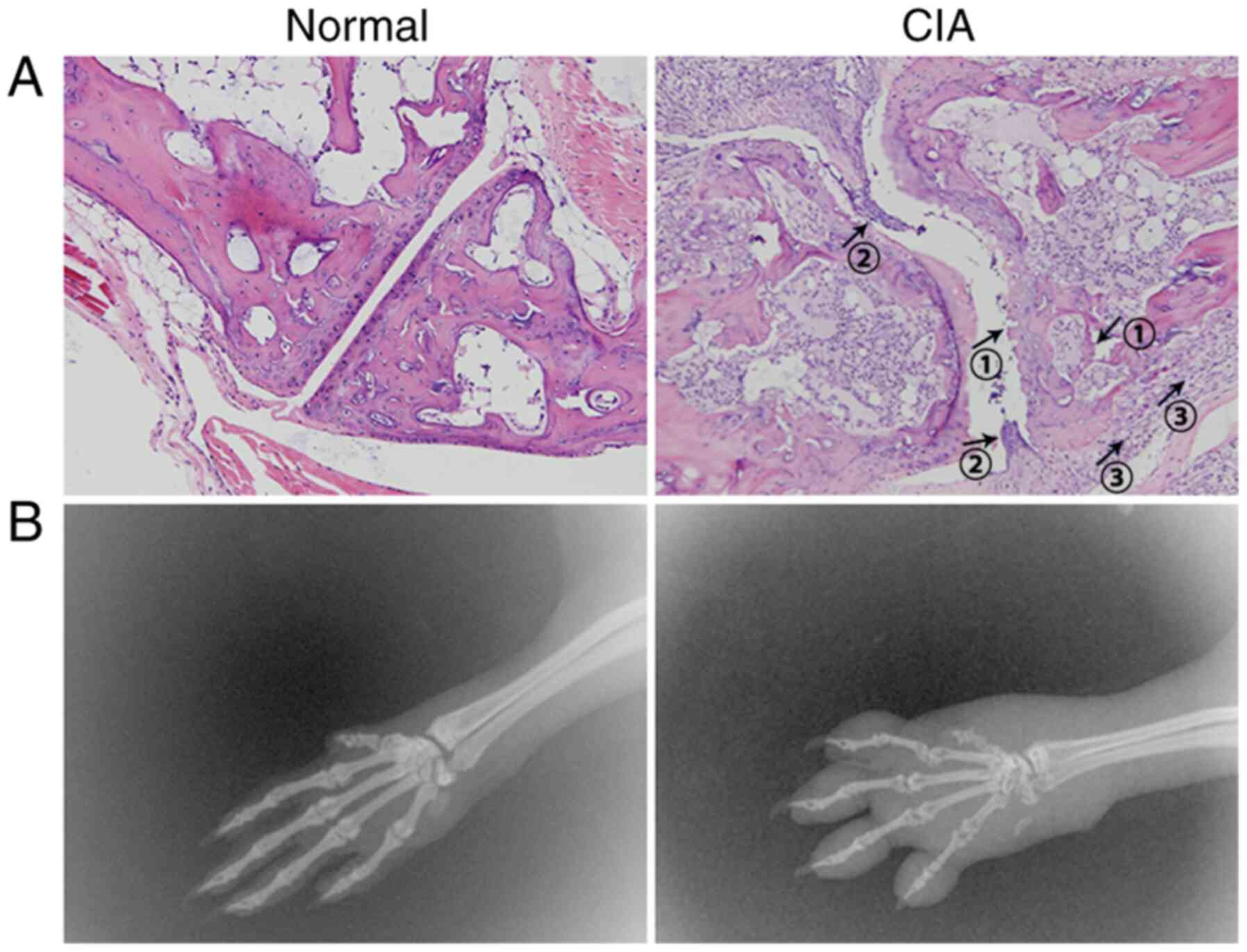
of normal mice and the color of the spleens is dark red (Fig. 4A and B). It is worth noting that even in the
absence of arthritis symptoms in CIA mice, obvious spleen
enlargement may be observed (Fig.
4C), and the spleen coefficient of mice in the CIA group is
significantly higher than that of the mice in the normal control
group (Fig. 4D). Compared with
normal mice, CIA mice were also reported to have an increased
thymus index (58). Severe synovial
hyperplasia, inflammatory cell infiltration and destruction of bone
and cartilage were observed in the joint space and bone tissue of
CIA mice (Fig. 5A and B). The levels of the cytokines TNF-α,
IFN-γ, IL-1β, IL-6, IL-17A and IL-18 in the serum of CIA mice were
significantly increased; by contrast, the levels of the cytokines
IL-4 and IL-10 were significantly decreased (59). Accordingly, the Th1/Th2 and
Th17/T-regulatory cell ratios exhibited significant increases
(60).
5. Choice of intervention time and
administration method
There are three commonly used methods for
administration: Intraperitoneal administration (61), intravenous administration (62) and oral administration (63). The medication route is selected
according to the purpose of the experiment and the characteristics
of the drug. In general, Traditional Chinese Medicine is
administered orally by dissolving the medicine in drinking water or
by gavage (64).
The time of administration should be given special
consideration and depends on whether the researcher aims to detect
the preventive or therapeutic effect of the drug. Normally, the
time of the first immunization is referred to as D0 and the time of
the second immunization of DBA/1 mice is D21. In most studies, mice
are administered injections as preventive treatments on D0-10 and
as therapeutic treatments on D21-25 (65-69).
Based on the pathogenesis of the CIA model as described above, the
specific development of RA pathology began after the first
immunization. The second immunization only enhanced the symptoms
(63). Approximately 10% of CIA
mice develop symptoms of joint swelling, which can be visually
observed between the first and second immunizations. It further
verifies that the onset of pathological signs occurs during this
period. Accordingly, the other 90% were observed 4-5 days after the
second immunization (D25-D26 of the experiment) (70). Consequently, it is recommended that
mice are administered preventive treatments from D0 and therapeutic
treatments from D21; regardless of the drug's preventive or
therapeutic effects, earlier administration of the drug is more
efficient. Furthermore, the observation time should not be too
long. If the lymphocyte reaction is being monitored, the best
time-point is D31. If joint damage and deformation are being
monitored, D31-D41 is an appropriate range. When the observation
time exceeds 42-60 days, the mouse heals and symptoms of arthritis,
such as redness and swelling, disappear (26).
6. Should the CIA mouse model be used?
Study of RA pathogenesis
The CIA mouse model may be used to study the
mechanisms by which cytokines and lymphocytes, including T cells
and particularly CD4+ T cells and B cells, are involved
in the pathogenesis of RA. In addition, it may be used to study the
pathogenesis of bone and cartilage destruction in RA (23,71).
Typically, mouse CIA is characterized by symmetrical joint
involvement and the peripheral joints are affected (38). Infiltration of cells (T cells, B
cells, macrophages and neutrophils) into the joint space leading to
pannus formation, hyperplastic synoviocyte membranes, marginal
erosion, and bone and cartilage destruction are similar to the
processes observed in human RA (26).
Study of the efficacy of anti-RA
drugs
For small-molecule drugs, the mouse CIA model is
preferred. For antibodies, the mouse arthritis model is not ideal
due to issues with species specificity. With the development of
antibody technology, humanized chimeric antibodies and fully
humanized antibodies are gradually becoming available. However, the
technical development of a corresponding humanized animal model has
not kept up. Therefore, in studies on the efficacy of such antibody
drugs, the absence of a suitable animal model will lead to
limitations. The existing solutions are not perfect. If the antigen
targeted by the antibody is homologous and highly conserved between
humans and mice, the animal model may be used to preliminarily
verify the efficacy of the antibody (72,73).
Although largely similar, RA and murine CIA also
differ in certain aspects. For instance, the joint pathology of RA
is chronic and symmetrical, whereas that of CIA in mice is
transient and asymmetrical. In addition, CIA is self-resolving
(after ~60 days in mice). The similarities and differences between
human RA and CIA are summarized in Table II (26,74,75).
Overall, the CIA mouse model is currently the closest to the
pathogenesis of human RA, however, researchers must consider their
individual experimental aims when deciding whether this model
should be used.
 | Table IINon-exhaustive list of similarities
and differences in clinical disease course and pathologic
mechanisms between RA and CIA. |
Table II
Non-exhaustive list of similarities
and differences in clinical disease course and pathologic
mechanisms between RA and CIA.
| Parameter | RA | CIA |
|---|
| Type | Polyarticular | Polyarticular |
| Predisposition
association | With certain MHC-II
haplotypes | With certain H-2
complexes |
| Joint
pathology | Similar | Similar |
| Hyperplasia of
synovial membrane | Present | Present |
| Synovial immune
infiltration | Present | Present |
| Neutrophils in
synovial fluid | Present | Present |
| Pannus
formation | Present | Present |
| Bone destruction by
osteoclasts | Present | Present |
| Serological
markers | Present | Present |
| Rheumatoid
factor | Present | Present |
| Anti-CII
antibodies | Present | Present |
| Anticyclic
citrullinated peptide antibodies | Present | Present |
| Changes in body
composition and increased resting energy expenditure found in
cachexia | Present | Present |
| Joint
involvement | Symmetric | Nonsymmetric |
| Disease
progression | Chronic, almost
impossible to self-heal | Transient, able to
self-heal |
| Systemic
manifestations |
Systemic/extra-articular manifestations
reported | None reported |
7. Additional considerations
Consideration should be given to the extent of
animal suffering (76). In the
process of CIA modeling, mice exhibit ulceration and inflammatory
reactions in the tail skin due to stimulation from the emulsion at
the base of the tail. Therefore, it is necessary to perform the
injections as slowly as possible and the depth of the needle should
be appropriate. If the needle is placed too superficially, it may
penetrate the skin. This leads to leakage of the emulsion, which
may increase the possibility of ulceration in the tail of the
mouse. In general, mice experience substantial pain due to the
process of CIA model generation and the symptoms of inflammation
itself. Therefore, the researcher should be as gentle as possible
with the experimental animals, provide stable keeping conditions
and minimize errors and pain during the operations. Humane
end-points with severity limits should be incorporated into
protocols to limit suffering (76).
When taking samples from sacrificed mice, researchers should keep
their workspace away from the cages of the remaining animals to
prevent panic and psychological tension caused by the mice
witnessing the suffering of their peers. This panic may even cause
changes in immune cells (77). In
addition, it is worth mentioning that numerous small animals lose
their lives in research and researchers should respect the animals.
Operators should strictly follow the Guide for the Care and Use of
Laboratory Animals from the National Institutes of Health. In fact,
one of the intentions of the present review is to help
experimenters minimize errors and reduce the number of experimental
animals used. The experiments should therefore be carefully planned
and designed together with a primary investigator or supervisor,
ensuring that each individual experiment is ethically approved
beforehand.
8. Conclusion and outlook
The CIA mouse model has been used in numerous
studies on RA etiology, pathogenesis, drug screening, transgenic
mice and immunotherapy worldwide. Research on CIA and the
standardized application of CIA animal models are of great
significance in these fields. However, in certain studies,
irregularities still exist in the establishment and application of
this model. The present review examined the molecular basis and
limitations of the CIA model in detail and aimed to provide a
reference for the investigation and use of CIA. All of the small
and easily overlooked but important details, including the reagents
and protocol required for modeling, are provided in this
report.
It is worth mentioning that scientists are also
exploring humanized RA animal models, which may be used to verify
the efficacy and mechanisms of anti-rheumatic drugs that involve
humanized antibodies. At present, it is thought that the most
stable model animals for humanized RA are humanized bone
marrow/liver/thymus mice. In this model, 6- to 8-week-old
NOD.Cg-Prkdcscid Il2rgtm1wjl/SzJ (NSG)
immunocompromised mice receive a thymus/liver implant, as in the
SCID-hu mouse model, followed by a second human hematopoietic stem
cell transplant (78). The
advantage of this system is the full reconstitution of the human
immune system in the periphery. This model is stable for 12 to 18
weeks (79,80). Accordingly, in theory, it is most
desirable to induce CIA in BLT mice and then assess the efficacy of
humanized antibodies in them. However, several problems were
encountered in the experiment. Frist, as the genetic background of
NSG mice is NOD (MHC II H-2g), the success rate of CIA
establishment is unknown. Second, the emergence of BLT mice is
still relatively novel, meaning that the development of the model
itself is not very mature, and that there are uncertainties
associated with it. In addition, the cost of BLT mice is quite high
right now. Therefore, much work remains to be performed to overcome
the challenges associated with this model.
Acknowledgements
The authors would like to thank Professor Xingchun
Gao from the Youth Innovation Team of Shaanxi Universities for his
suggestions for the writing of the current review.
Funding
Funding: This work was supported by the Research Fund of Shaanxi
Provincial Education Department (grant no. 20JS136), the Xi'an
Weiyang District Science and Technology Information Bureau (grant
no. 201933), the Natural Science Basic Research Plan of Shaanxi
Province in China (grant no. 2020JQ-877) and the Science Research
Program of Xi 'an Medical University (grant no. 2020DOC25).
Availability of data and materials
Not applicable.
Authors' contributions
JL and ZH wrote the manuscript. JL, JC, RZ, PY, HG
and GN performed the experiments, analyzed the data and revised the
manuscript. NG and XG supervised the current study, proofread the
manuscript and confirmed the authenticity of all the raw data. All
authors read and approved the final version.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bird P, Kirkham B, Portek I, Shnier R,
Joshua F, Edmonds J and Lassere M: Documenting damage progression
in a two-year longitudinal study of rheumatoid arthritis patients
with established disease (the DAMAGE study cohort): Is there an
advantage in the use of magnetic resonance imaging as compared with
plain radiography? Arthritis Rheum. 50:1383–1389. 2004.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Døhn UM, Ejbjerg BJ, Hasselquist M,
Narvestad E, Møller J, Thomsen HS and Østergaard M: Detection of
bone erosions in rheumatoid arthritis wrist joints with magnetic
resonance imaging, computed tomography and radiography. Arthritis
Res Ther. 10(R25)2008.PubMed/NCBI View
Article : Google Scholar
|
|
3
|
van der Woude D and van der Helm-van Mil
AHM: Update on the epidemiology, risk factors, and disease outcomes
of rheumatoid arthritis. Best Pract Res Clin Rheumatol. 32:174–187.
2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Ejbjerg BJ, Vestergaard A, Jacobsen S,
Thomsen HS and Østergaard M: The smallest detectable difference and
sensitivity to change of magnetic resonance imaging and
radiographic scoring of structural joint damage in rheumatoid
arthritis finger, wrist, and toe joints: A comparison of the
OMERACT rheumatoid arthritis magnetic resonance imaging score
applied to different joint combinations and the Sharp/van der
Heijde radiographic score. Arthritis Rheum. 52:2300–2306.
2005.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Bendstrup E, Møller J, Kronborg-White S,
Prior TS and Hyldgaard C: Interstitial Lung Disease in Rheumatoid
Arthritis Remains a Challenge for Clinicians. J Clin Med.
8(E2038)2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Bandyopadhyay D, Banerjee U, Hajra A,
Chakraborty S, Amgai B, Ghosh RK, Haddadin FI, Modi VA, Sinha K,
Aronow WS, et al: Trends of Cardiac Complications in Patients With
Rheumatoid Arthritis: Analysis of the United States National
Inpatient Sample; 2005-2014. Curr Probl Cardiol.
46(100455)2021.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Zulfiqar AA, Niazi R, Pennaforte JL and
Andres E: Rheumatoid arthritis and cardiovascular risk factor:
Literature review. Rev Med Liege. 73:634–639. 2018.PubMed/NCBI(In French).
|
|
8
|
Atzeni F, Talotta R, Masala IF, Gerardi
MC, Casale R and Sarzi-Puttini P: Central nervous system
involvement in rheumatoid arthritis patients and the potential
implications of using biological agents. Best Pract Res Clin
Rheumatol. 32:500–510. 2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Nakao E, Mitsunaga A, Hamano T, Shirato M,
Shirato I and Nishino T: Case report of rheumatoid arthritis
associated with type A gastritis and Hashimoto thyroiditis. Nihon
Shokakibyo Gakkai Zasshi. 107:1927–1932. 2010.PubMed/NCBI(In Japanese).
|
|
10
|
Lora V, Cerroni L and Cota C: Skin
manifestations of rheumatoid arthritis. G Ital Dermatol Venereol.
153:243–255. 2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Kurmann RD and Mankad R: Atherosclerotic
Heart Disease in Women With Autoimmune Rheumatologic Inflammatory
Conditions. Can J Cardiol. 34:381–389. 2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Favalli EG, Biggioggero M, Crotti C,
Becciolini A, Raimondo MG and Meroni PL: Sex and Management of
Rheumatoid Arthritis. Clin Rev Allergy Immunol. 56:333–345.
2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Law ST and Taylor PC: Role of biological
agents in treatment of rheumatoid arthritis. Pharmacol Res.
150(104497)2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Bolon B, Stolina M, King C, Middleton S,
Gasser J, Zack D and Feige U: Rodent preclinical models for
developing novel antiarthritic molecules: Comparative biology and
preferred methods for evaluating efficacy. J Biomed Biotechnol.
2011(569068)2011.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Hirose J and Tanaka S: Animal models for
bone and joint disease. CIA, CAIA model. Clin Calcium. 21:253–259.
2011.PubMed/NCBI(In Japanese).
|
|
16
|
Hanyecz A, Olasz K, Tarjanyi O, Nemeth P,
Mikecz K, Glant TT and Boldizsar F: Proteoglycan aggrecan
conducting T cell activation and apoptosis in a murine model of
rheumatoid arthritis. BioMed Res Int. 2014(942148)2014.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Corrado A, Donato P, Maccari S, Cecchi R,
Spadafina T, Arcidiacono L, Tavarini S, Sammicheli C, Laera D,
Manetti AG, et al: Staphylococcus aureus-dependent septic
arthritis in murine knee joints: Local immune response and
beneficial effects of vaccination. Sci Rep. 6(38043)2016.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Choudhary N, Bhatt LK and Prabhavalkar KS:
Experimental animal models for rheumatoid arthritis.
Immunopharmacol Immunotoxicol. 40:193–200. 2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Christensen AD, Haase C, Cook AD and
Hamilton JA: K/BxN Serum-Transfer Arthritis as a Model for Human
Inflammatory Arthritis. Front Immunol. 7(213)2016.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Sønderstrup G: Development of humanized
mice as a model of inflammatory arthritis. Springer Semin
Immunopathol. 25:35–45. 2003.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Nakano K, Yamaoka K, Hanami K, Saito K,
Sasaguri Y, Yanagihara N, Tanaka S, Katsuki I, Matsushita S and
Tanaka Y: Dopamine induces IL-6-dependent IL-17 production via
D1-like receptor on CD4 naive T cells and D1-like receptor
antagonist SCH-23390 inhibits cartilage destruction in a human
rheumatoid arthritis/SCID mouse chimera model. J Immunol.
186:3745–3752. 2011.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Liu S, Hasegawa H, Takemasa E, Suzuki Y,
Oka K, Kiyoi T, Takeda H, Ogasawara T, Sawasaki T, Yasukawa M, et
al: Efficiency and Safety of CRAC Inhibitors in Human Rheumatoid
Arthritis Xenograft Models. J Immunol. 199:1584–1595.
2017.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Williams RO: Collagen-induced arthritis in
mice. Methods Mol Med. 136:191–199. 2007.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Caplazi P, Baca M, Barck K, Carano RA,
DeVoss J, Lee WP, Bolon B and Diehl L: Mouse Models of Rheumatoid
Arthritis. Vet Pathol. 52:819–826. 2015.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Cho YG, Cho ML, Min SY and Kim HY: Type II
collagen autoimmunity in a mouse model of human rheumatoid
arthritis. Autoimmun Rev. 7:65–70. 2007.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Schurgers E, Billiau A and Matthys P:
Collagen-induced arthritis as an animal model for rheumatoid
arthritis: Focus on interferon-γ. J Interferon Cytokine Res.
31:917–926. 2011.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Trentham DE, Townes AS and Kang AH:
Autoimmunity to type II collagen an experimental model of
arthritis. J Exp Med. 146:857–868. 1977.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Cathcart ES, Hayes KC, Gonnerman WA,
Lazzari AA and Franzblau C: Experimental arthritis in a nonhuman
primate. I. Induction by bovine type II collagen. Lab Invest.
54:26–31. 1986.PubMed/NCBI
|
|
29
|
Courtenay JS, Dallman MJ, dayan AD, Martin
A and Mosedale B: Immunisation against heterologous type II
collagen induces arthritis in mice. Nature. 283:666–668.
1980.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Nandakumar KS: Pathogenic antibody
recognition of cartilage. Cell Tissue Res. 339:213–220.
2010.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Wooley PH, Luthra HS, Stuart JM and David
CS: Type II collagen-induced arthritis in mice. I. Major
histocompatibility complex (I region) linkage and antibody
correlates. J Exp Med. 154:688–700. 1981.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Tarkowski A, Holmdahl R and Klareskog L:
Rheumatoid factors in mice. Monogr Allergy. 26:214–229.
1989.PubMed/NCBI
|
|
33
|
Benson RA, McInnes IB, Garside P and
Brewer JM: Model answers: Rational application of murine models in
arthritis research. Eur J Immunol. 48:32–38. 2018.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Myers LK, Tang B, Rosloniec EF, Stuart JM,
Chiang TM and Kang AH: Characterization of a peptide analog of a
determinant of type II collagen that suppresses collagen-induced
arthritis. J Immunol. 161:3589–3595. 1998.PubMed/NCBI
|
|
35
|
Brand DD, Kang AH and Rosloniec EF:
Immunopathogenesis of collagen arthritis. Springer Semin
Immunopathol. 25:3–18. 2003.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Inglis JJ, Simelyte E, McCann FE, Criado G
and Williams RO: Protocol for the induction of arthritis in C57BL/6
mice. Nat Protoc. 3:612–618. 2008.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Stuart JM, Townes AS and Kang AH: Collagen
autoimmune arthritis. Annu Rev Immunol. 2:199–218. 1984.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Joe B and Wilder RL: Animal models of
rheumatoid arthritis. Mol Med Today. 5:367–369. 1999.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Kannan K, Ortmann RA and Kimpel D: Animal
models of rheumatoid arthritis and their relevance to human
disease. Pathophysiology. 12:167–181. 2005.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Hegen M, Keith JC Jr, Collins M and
Nickerson-Nutter CL: Utility of animal models for identification of
potential therapeutics for rheumatoid arthritis. Ann Rheum Dis.
67:1505–1515. 2008.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Petersen F and Yu X: A novel preclinical
model for rheumatoid arthritis research. Arthritis Res Ther.
12(148)2010.PubMed/NCBI View
Article : Google Scholar
|
|
42
|
Brand DD, Latham KA and Rosloniec EF:
Collagen-induced arthritis. Nat Protoc. 2:1269–1275.
2007.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Huang JC, Vestberg M, Minguela A, Holmdahl
R and Ward ES: Analysis of autoreactive T cells associated with
murine collagen-induced arthritis using peptide-MHC multimers. Int
Immunol. 16:283–293. 2004.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Buglak NE and Bahnson ESM: A Rat Carotid
Artery Pressure-Controlled Segmental Balloon Injury with
Periadventitial Therapeutic Application. J Vis Exp 161:
10.3791/60473, 2020.
|
|
45
|
Holmdahl R, Jansson L, Larsson E, Rubin K
and Klareskog L: Homologous type II collagen induces chronic and
progressive arthritis in mice. Arthritis Rheum. 29:106–113.
1986.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Boissier MC, Feng XZ, Carlioz A, Roudier R
and Fournier C: Experimental autoimmune arthritis in mice. I.
Homologous type II collagen is responsible for self-perpetuating
chronic polyarthritis. Ann Rheum Dis. 46:691–700. 1987.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Wilder RL: Hormones and autoimmunity:
Animal models of arthritis. Baillieres Clin Rheumatol. 10:259–271.
1996.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Vandenbroucke JP, Valkenburg HA, Boersma
JW, Cats A, Festen JJ, Huber-Bruning O and Rasker JJ: Oral
contraceptives and rheumatoid arthritis: Further evidence for a
preventive effect. Lancet. 2:839–842. 1982.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Lee YK, Choi KH, Kwak HS and Chang YH: The
preventive effects of nanopowdered red ginseng on collagen-induced
arthritic mice. Int J Food Sci Nutr. 69:308–317. 2018.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Holmdahl R, Jansson L, Gullberg D, Rubin
K, Forsberg PO and Klareskog L: Incidence of arthritis and
autoreactivity of anti-collagen antibodies after immunization of
DBA/1 mice with heterologous and autologous collagen II. Clin Exp
Immunol. 62:639–646. 1985.PubMed/NCBI
|
|
51
|
Stewart-Tull DE: Freund's complete and
incomplete adjuvants, preparation, and quality control standards
for experimental laboratory animals use. Methods Mol Biol.
626:59–72. 2010.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Wooley PH, Luthra HS, Griffiths MM, Stuart
JM, Huse A and David CS: Type II collagen-induced arthritis in
mice. IV. Variations in immunogenetic regulation provide evidence
for multiple arthritogenic epitopes on the collagen molecule. J
Immunol. 135:2443–2451. 1985.PubMed/NCBI
|
|
53
|
Chen HH, Chen DY, Chao YH, Chen YM, Wu CL,
Lai KL, Lin CH and Lin CC: Acarbose Decreases the Rheumatoid
Arthritis Risk of Diabetic Patients and Attenuates the Incidence
and Severity of Collagen-induced Arthritis in Mice. Sci Rep.
5(18288)2015.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Inglis JJ, Criado G, Medghalchi M, Andrews
M, Sandison A, Feldmann M and Williams RO: Collagen-induced
arthritis in C57BL/6 mice is associated with a robust and sustained
T-cell response to type II collagen. Arthritis Res Ther.
9(R113)2007.PubMed/NCBI View
Article : Google Scholar
|
|
55
|
Liu L, Yang J, Zu B, Wang J, Sheng K, Zhao
L and Xu W: Acacetin regulated the reciprocal differentiation of
Th17 cells and Treg cells and mitigated the symptoms of
collagen-induced arthritis in mice. Scand J Immunol.
88(e12712)2018.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Deng Y, Luo H, Shu J, Shu H, Lu C, Zhao N,
Geng Y, He X and Lu A: Pien Tze Huang alleviate the joint
inflammation in collagen-induced arthritis mice. Chin Med.
15(30)2020.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Luan J, Zhang K, Yang P, Zhang Y, Feng F,
Zhu YM, Zhu P and Chen ZN: The combination of FK506 and an
anti-CD147 mAb exerts potential therapeutic effects on a mouse
model of collagen-induced arthritis. Mol Immunol. 101:1–9.
2018.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Fu J, Zhang L, Song S, Sheng K, Li Y, Li
P, Song S, Wang Q, Chu J and Wei W: Effect of bone marrow-derived
CD11b(+)F4/80 (+) immature dendritic cells on the balance between
pro-inflammatory and anti-inflammatory cytokines in DBA/1 mice with
collagen-induced arthritis. Inflamm Res. 63:357–367.
2014.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Zhou X, Zhao X, Tang L, Zhang Y, Ruan H,
Pi H, Qiu J and Wu J: Immunomodulatory activity of the rhizomes of
Impatiens pritzellii var. hupehensis on collagen-induced arthritis
mice. J Ethnopharmacol. 109:505–509. 2007.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Wang Z, Zhuo F, Chu P, Yang X and Zhao G:
Germacrone alleviates collagen-induced arthritis via regulating
Th1/Th2 balance and NF-κB activation. Biochem Biophys Res Commun.
518:560–564. 2019.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Jia Q, Wang T, Wang X, Xu H, Liu Y, Wang
Y, Shi Q and Liang Q: Astragalin Suppresses Inflammatory Responses
and Bone Destruction in Mice With Collagen-Induced Arthritis and in
Human Fibroblast-Like Synoviocytes. Front Pharmacol.
10(94)2019.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Svetlicky N, Kivity S, Odeh Q, Shovman O,
Gertel S, Amital H, Gendelman O, Volkov A, Barshack I, Bar-Meir E,
et al: Anti-citrullinated-protein-antibody-specific intravenous
immunoglobulin attenuates collagen-induced arthritis in mice. Clin
Exp Immunol. 182:241–250. 2015.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Yang P, Qian F, Zhang M, Xu AL, Wang X,
Jiang B, Zhou L and Zhou X: Zishen Tongluo formula ameliorates
collagen-induced arthritis in mice by modulation of Th17/Treg
balance. J Ethnopharmacol. 250(112428)2020.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Feng ZT, Yang T, Hou XQ, Wu HY, Feng JT,
Ou BJ, Cai SJ, Li J and Mei ZG: Sinomenine mitigates
collagen-induced arthritis mice by inhibiting angiogenesis. Biomed
Pharmacother. 113(108759)2019.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Fan J, Luo J, Yan C, Hao R, Zhao X, Jia R,
He J, Xu D, Miao M and Li X: Methotrexate, combined with
cyclophosphamide attenuates murine collagen induced arthritis by
modulating the expression level of Breg and DCs. Mol Immunol.
90:106–117. 2017.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Wu S, Li Y, Li Y, Yao L, Lin T, Jiang S,
Shen H, Xia L and Lu J: Interleukin-35 attenuates collagen-induced
arthritis through suppression of vascular endothelial growth factor
and its receptors. Int Immunopharmacol. 34:71–77. 2016.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Gui H, Liu X, Liu LR, Su DF and Dai SM:
Activation of cannabinoid receptor 2 attenuates synovitis and joint
distruction in collagen-induced arthritis. Immunobiology.
220:817–822. 2015.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Xuzhu G, Komai-Koma M, Leung BP, Howe HS,
McSharry C, McInnes IB and Xu D: Resveratrol modulates murine
collagen-induced arthritis by inhibiting Th17 and B-cell function.
Ann Rheum Dis. 71:129–135. 2012.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Guo Y, Xing E, Song H, Feng G, Liang X, An
G, Zhao X and Wang M: Therapeutic effect of dioscin on
collagen-induced arthritis through reduction of Th1/Th2. Int
Immunopharmacol. 39:79–83. 2016.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Suszko A and Obmińska-Mrukowicz B:
Influence of polysaccharide fractions isolated from Caltha
palustris L. on the cellular immune response in
collagen-induced arthritis (CIA) in mice. A comparison with
methotrexate. J Ethnopharmacol. 145:109–117. 2013.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Bessis N, Decker P, Assier E, Semerano L
and Boissier MC: Arthritis models: Usefulness and interpretation.
Semin Immunopathol. 39:469–486. 2017.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Zhong C, Wang J, Li B, Xiang H, Ultsch M,
Coons M, Wong T, Chiang NY, Clark S, Clark R, et al: Development
and preclinical characterization of a humanized antibody targeting
CXCL12. Clin Cancer Res. 19:4433–4445. 2013.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Huang X, He Y, Han J, Zhuang J, He J and
Sun E: Not only anti-inflammation, etanercept abrogates
collagen-induced arthritis by inhibiting dendritic cell migration
and maturation. Cent Eur J Immunol. 44:237–245. 2019.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Alabarse PVG, Lora PS, Silva JMS, Santo
RCE, Freitas EC, de Oliveira MS, Almeida AS, Immig M, Teixeira VON,
Filippin LI, et al: Collagen-induced arthritis as an animal model
of rheumatoid cachexia. J Cachexia Sarcopenia Muscle. 9:603–612.
2018.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Evans WJ, Morley JE, Argilés J, Bales C,
Baracos V, Guttridge D, Jatoi A, Kalantar-Zadeh K, Lochs H,
Mantovani G, et al: Cachexia: A new definition. Clin Nutr.
27:793–799. 2008.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Vincent TL, Williams RO, Maciewicz R,
Silman A and Garside P: Arthritis Research UK animal models working
group. Mapping pathogenesis of arthritis through small animal
models. Rheumatology (Oxford). 51:1931–1941. 2012.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Marazziti D, Ambrogi F, Abelli M, Di Nasso
E, Catena M, Massimetti G, Carlini M and Dell'Osso L: Lymphocyte
subsets, cardiovascular measures and anxiety state before and after
a professional examination. Stress. 10:93–99. 2007.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Melkus MW, Estes JD, Padgett-Thomas A,
Gatlin J, Denton PW, Othieno FA, Wege AK, Haase AT and Garcia JV:
Humanized mice mount specific adaptive and innate immune responses
to EBV and TSST-1. Nat Med. 12:1316–1322. 2006.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Akkina R: Humanized Mice for Studying
Human Immune Responses and Generating Human Monoclonal Antibodies.
Microbiol Spectr. 2:2014.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Vatakis DN, Bristol GC, Kim SG, Levin B,
Liu W, Radu CG, Kitchen SG and Zack JA: Using the BLT humanized
mouse as a stem cell based gene therapy tumor model. J Vis Exp.
70(e4181)2012.PubMed/NCBI View
Article : Google Scholar
|