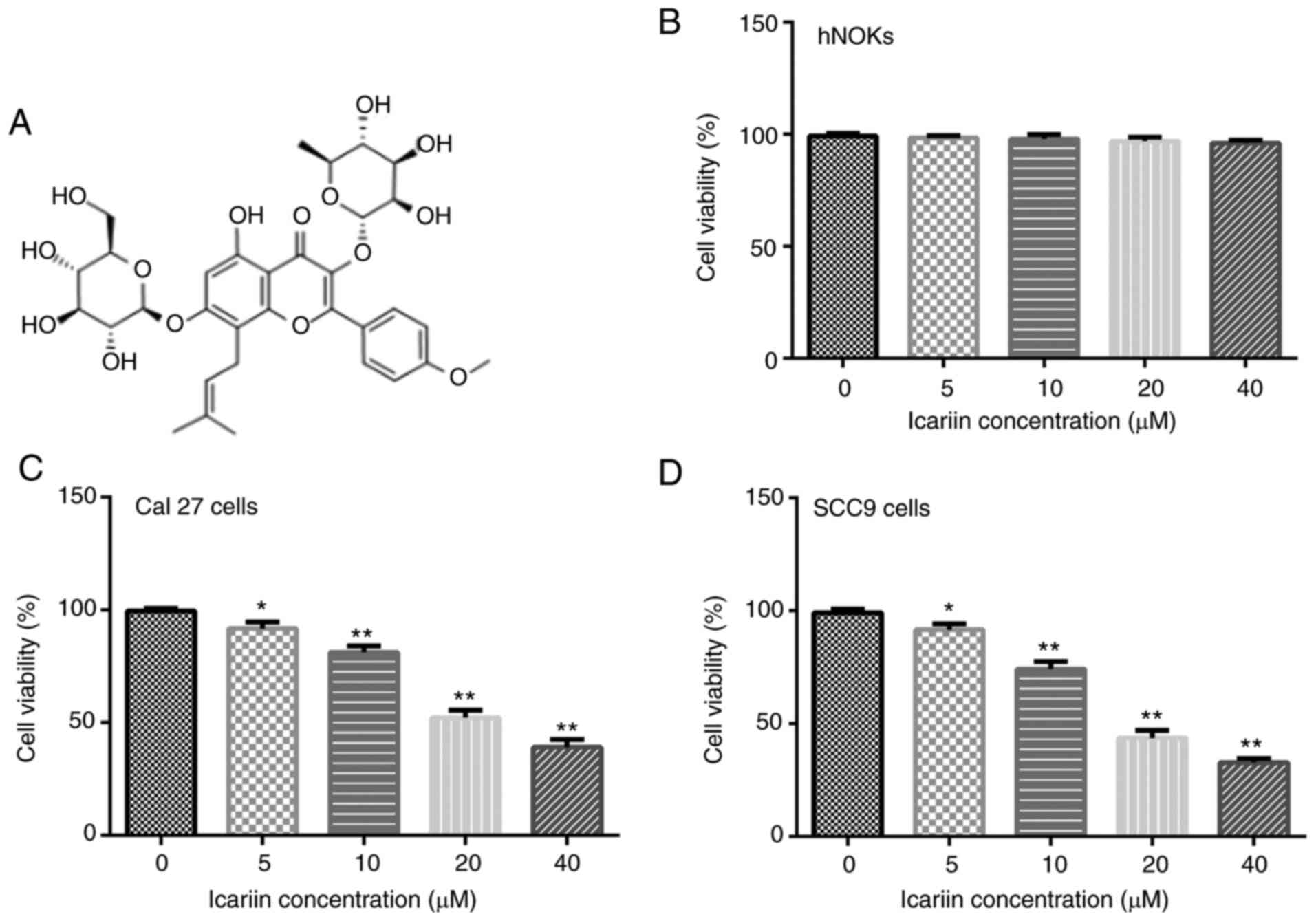
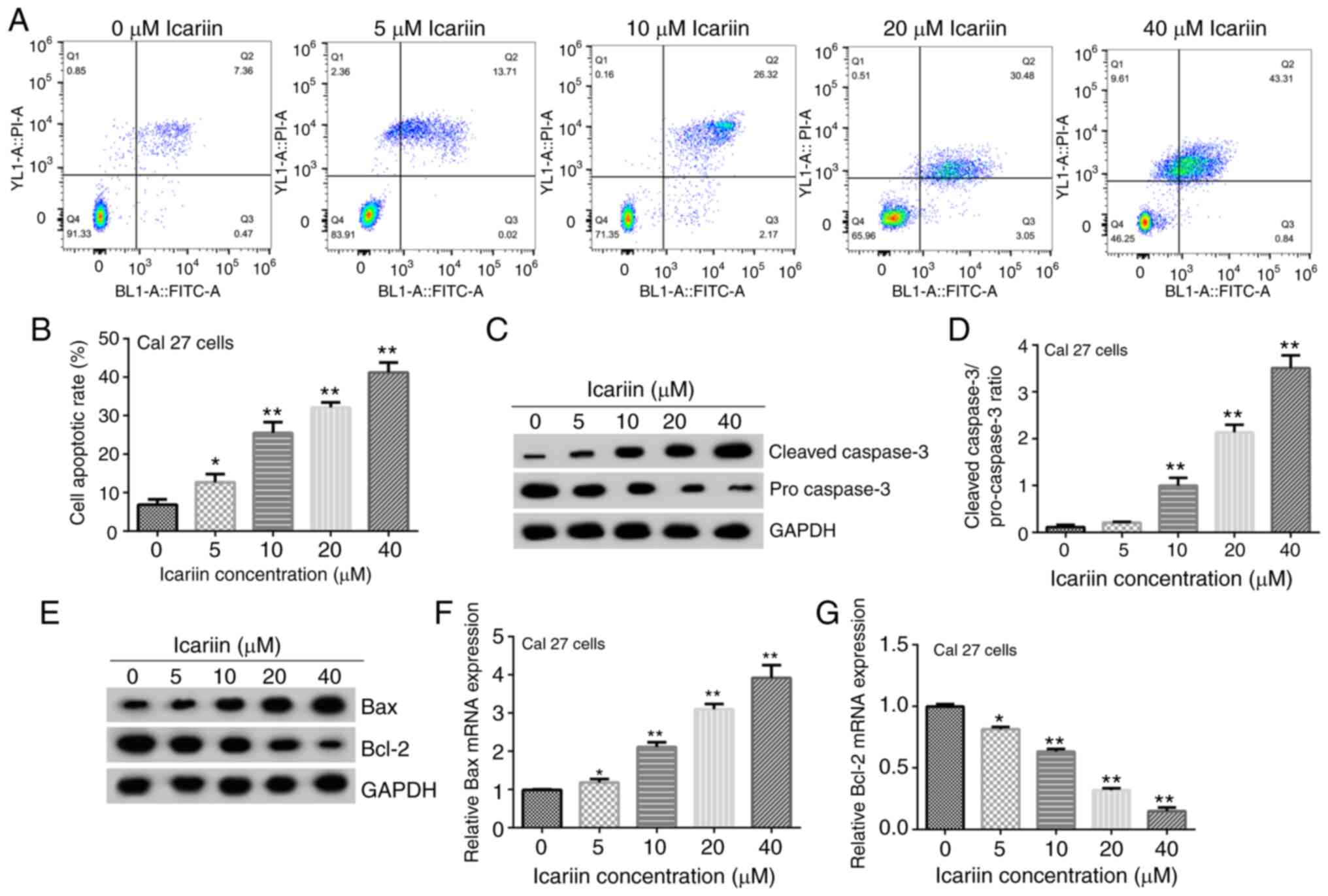
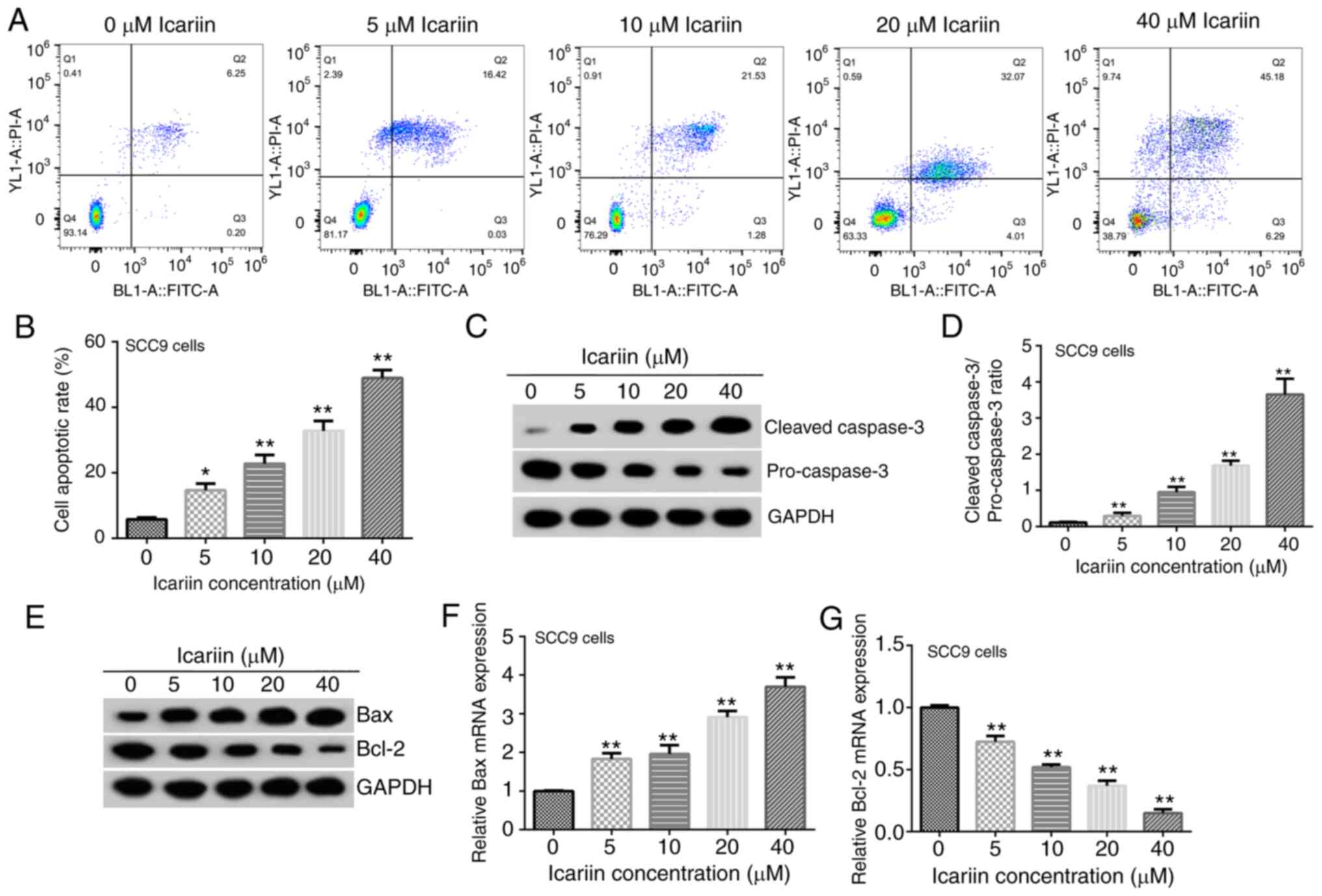
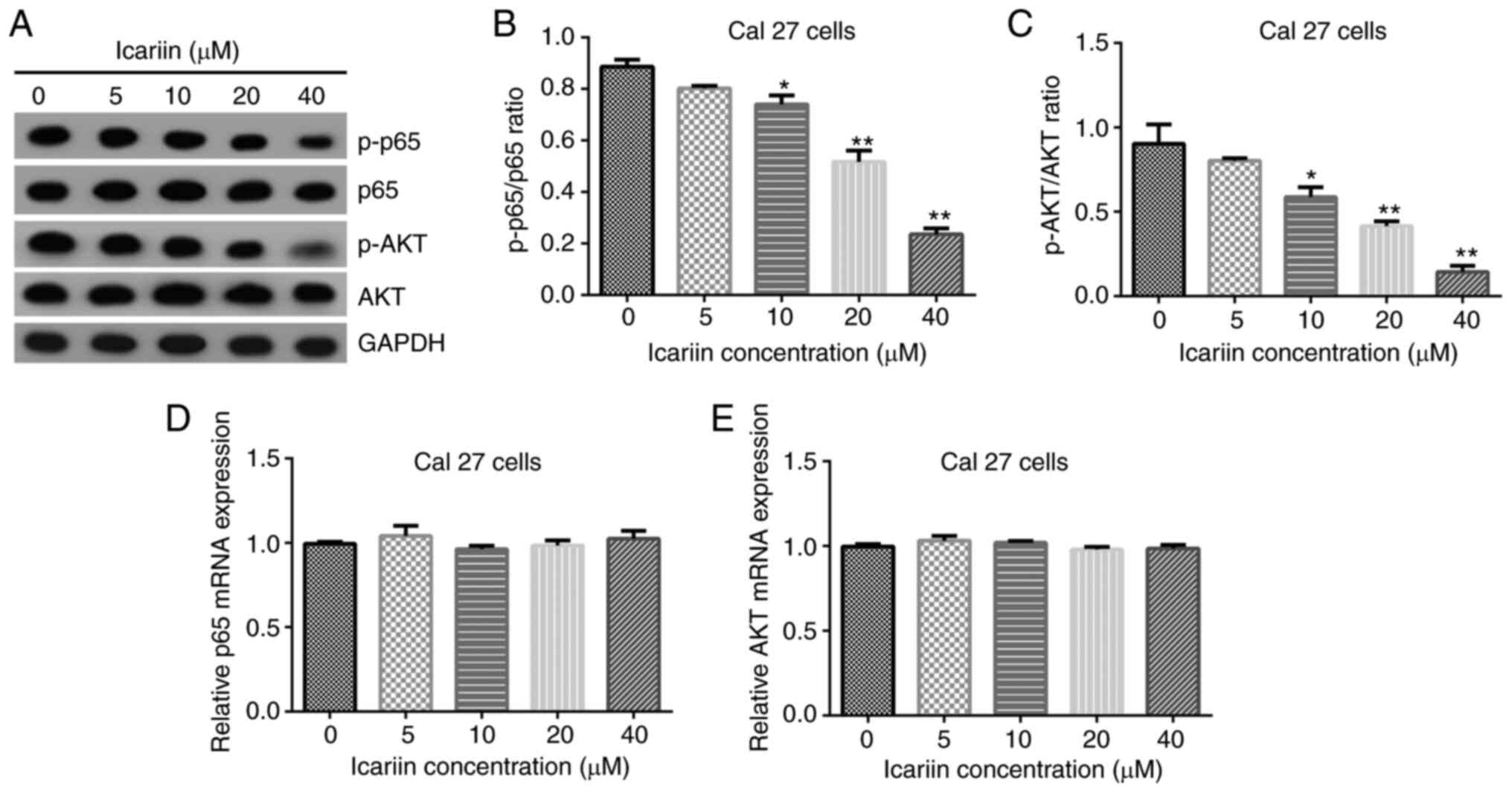
|
1
|
Qin L, Zhang G, Sheng H, Wang XL, Wang YX,
Yeung KW, Griffith JF, Li ZR, Leung KS and Yao XS: Phytoestrogenic
compounds for prevention of steroid-associated osteonecrosis. J
Musculoskelet Neuronal Interact. 8:18–21. 2008.PubMed/NCBI
|
|
2
|
Chen Y, Sun T, Wu J, Kalionis B, Zhang C,
Yuan D, Huang J, Cai W, Fang H and Xia S: Icariin intervenes in
cardiac inflammaging through upregulation of SIRT6 enzyme activity
and inhibition of the NF-kappa B pathway. Biomed Res Int.
2015(895976)2015.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Carlsten M and Childs RW: Genetic
manipulation of NK cells for cancer immunotherapy: Techniques and
clinical implications. Front Inmunol. 6(266)2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Wang Z, Zhang H, Dai L, Song T, Li P, Liu
Y and Wang L: Arsenic trioxide and icariin show synergistic
anti-leukemic activity. Cell Biochem Biophys. 73:213–219.
2015.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Huang S, Xie T and Liu W: Icariin inhibits
the growth of human cervical cancer cells by inducing apoptosis and
autophagy by targeting mTOR/PI3K/AKT signalling pathway. J BUON.
24:990–996. 2019.PubMed/NCBI
|
|
6
|
Wang P, Zhang J, Xiong X, Yuan W, Qin S,
Cao W, Dai L, Xie F, Li A and Liu Z: Icariin suppresses cell cycle
transition and cell migration in ovarian cancer cells. Oncol Rep.
41:2321–2328. 2019.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Kim B, Seo JH, Lee KY and Park B: Icariin
sensitizes human colon cancer cells to TRAIL-induced apoptosis via
ERK-mediated upregulation of death receptors. Int J Oncol.
56:821–834. 2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Song L, Chen X, Mi L, Liu C, Zhu S, Yang
T, Luo X, Zhang Q, Lu H and Liang X: Icariin-induced inhibition of
SIRT6/NF-κB triggers redox mediated apoptosis and enhances
anti-tumor immunity in triple-negative breast cancer. Cancer Sci.
111:4242–4256. 2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Ju ZY, Tang QM, Chen LL, et al: Axl
inhibitor R428 induces autophagy of oral squamous cell carcinoma
Cal27 cells. J Pract Stomatol. 35:2019.
|
|
10
|
Hu XG, Qiu ZL, Zen JC, et al: Differential
expression of microRNAs in human oral squamous cell carcinoma stem
cells. J Pract Stomatol. 3:398–402. 2019.
|
|
11
|
Schneider P, Schon M, Pletz N, Seitz CS,
Liu N, Ziegelbauer K, Zachmann K, Emmert S and Schön MP: The novel
PI3 kinase inhibitor, BAY80-6946, impairs melanoma growth in vivo
and in vitro. Exp Dermatol. 23:579–584. 2014.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Liu YJ, Huang LL, Hao BH, Li H, Zhu S,
Wang Q, Li R, Xu Y and Zhang X: Use of an osteoblast overload
damage model to probe the effect of icariin on the proliferation,
differentiation and mineralization of MC3T3-E1cells through the
Wnt/β-catenin signalling pathway. Cell Physiol Biochem.
41:1605–1615. 2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Li W, Wang M, Wang LY, Ji S, Zhang J and
Zhang C: Icariin synergizes with arsenic trioxide to suppress human
hepatocellular carcinoma. Cell Biochem Biophys. 68:427–436.
2014.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Yang LJ, Wang YX, Guo H and Guo M:
Synergistic anti-cancer effects of icariin and temozolomide in
glioblastoma. Cell Biochem Biophys. 71:1379–1385. 2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Shi DB, Li XX, Zheng HT, Li DW, Cai GX,
Peng JJ, Gu WL, Guan ZQ, Xu Y and Cai SJ: Icariin-mediated
inhibition of NF-κB activity enhances the in vitro and in vivo
antitu-mour effect of 5-fluorouracil in colorectal cancer. Cell
Biochem Biophys. 69:523–530. 2014.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Xu W, Tao H and Zhang Q: Effect of Six1
gene on As2O3-induced oral squamous cell
carcinoma cell apoptosis and ROS levels. J Clin Exp Medicine.
(15):2019.
|
|
17
|
Peng M and Pang C: MicroRNA-140-5p
inhibits the tumorigenesis of oral squamous cell carcinoma by
targeting p21-activated kinase 4. Cell Biol Int: Aug 8, 2019 (Epub
ahead of print).
|
|
18
|
Wang XM, Liu CM, Zhang CR, Xu XG and Fu
SB: Functional significance of TGF-beta1 signal transduction
pathway in oral squamous cell carcinoma. Zhonghua Zhong Liu Za Zhi.
31:28–32. 2009.PubMed/NCBI(In Chinese).
|
|
19
|
Chen Z, He J, Xing X, Li P, Zhang W, Tong
Z, Jing X, Li L, Liu D, Wu Q and Ju H: Mn12Ac inhibits the
migration, invasion and epithelial-mesenchymal transition of lung
cancer cells by downregulating the Wnt/β-catenin and PI3K/AKT
signaling pathways. Oncol Lett. 16:3943–3948. 2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Xie J, Lin W, Huang L, Xu N, Xu A, Chen B,
Watanabe M, Liu C and Huang P: Bufalin suppresses the proliferation
and metastasis of renal cell carcinoma by inhibiting the
PI3K/Akt/mTOR signaling pathway. Oncol Lett. 16:3867–3873.
2018.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Choi MS, Moon SM, Lee SA, Park BR, Kim JS,
Kim DK, Kim YH and Kim CS: Adenosine induces intrinsic apoptosis
via the PI3K/Akt/mTOR signaling pathway in human pharyngeal
squamous carcinoma FaDu cells. Oncol Lett. 15:6489–6496.
2018.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Hao Y, Zhang C, Sun Y and Xu H:
Licochalcone A inhibits cell proliferation, migration, and invasion
through regulating the PI3K/AKT signaling pathway in oral squamous
cell carcinoma. Onco Targets Ther. 12:4427–4435. 2019.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Fan QC, Tian H, Wang Y and Liu XB:
Integrin-alpha5 promoted the progression of oral squamous cell
carcinoma and modulated PI3K/AKT signaling pathway. Arch Oral Biol.
101:85–91. 2019.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Velu P, Vijayalakshmi A and Vinothkumar V:
Inhibiting the PI3K/Akt, NF-kB signalling pathways with syringic
acid for attenuating the development of oral squamous cell
carcinoma cells SCC131. J Pharm Pharmacol. 72:1595–1606.
2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: Cancer
incidence and mortality worldwide: Sources, methods and major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E386.
2015.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Galluzzi L and Rudqvist NP: Preface: More
than two decades of modern tumor immunology. Methods Enzymol.
629:xxii–xlii. 2019.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Zhang X, Kang Z, Li Q, Zhang J, Cheng S,
Chang H, Wang S, Cao S, Li T, Li J, et al: Antigen-adjuvant effects
of icariin in enhancing tumor-specific immunity in
mastocytoma-bearing DBA/2J mice. Biomed Pharmacother. 99:810–816.
2018.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Gao M and Li W: In vitro study on the
inhibitory effect of icariin on human bladder cancer T24 cells.
Shijiazhuang: Hebei Medical University 2010.
|
|
29
|
Hao H, Zhang Q, Zhu H, Wen Y, Qiu D, Xiong
J, Fu X, Wu Y, Meng K and Li J: Icaritin promotes tumor T-cell
infiltration and induces antitumor immunity in mice. Eur J Immunol.
49:2235–2244. 2019.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Tan HL, Chan KG, Pusparajah P, Saokaew S,
Duangjai A, Lee LH and Goh BH: Anti-cancer properties of the
naturally occurring aphrodisiacs: Icariin and its derivatives.
Front Pharmacol. 7(191)2016.PubMed/NCBI View Article : Google Scholar
|