Introduction
Myomas or fibroids are benign smooth muscle tumors
of the uterus (1,2). Uterine leiomyomas are the most common
benign uterine tumors in women of reproductive age, with an
incidence of 70% (3,4). The majority are asymptomatic,
and only 30-40% of women with myomas present with symptoms (such as
heavy menstrual bleeding, infertility, recurrent pregnancy loss and
pressure symptoms) (3,5). For these women the treatment remains
the surgical removal of these fibroids (1,2). For
the women who wish to preserve their uterus, the gold standard
treatment is myomectomy. Abdominal myomectomy, transvaginal
myomectomy, laparoscopic myomectomy (LM), laparoscopically-assisted
myomectomy via minilaparotomy incision, and robot-assisted LM are
the techniques used in gynecological myomectomy practice. The
recent technological advances in endoscopy have allowed
gynecological surgeons to implement LM in practice (1,6-8).
The major problem associated with myomectomy is the
excessive bleeding from multiple uterine blood supply (9). Myomectomy is an extremely hemorrhagic
intervention. Numerous measures have been assessed in an attempt to
reduce blood loss during LM (10).
The more commonly used interventions are uterine
artery embolization/ligation, pericervical tourniquet placement,
intramyometrial vasopressin/epinephrine administration, use of
uterotonics (including ergometrine, oxytocin and misoprostol), and
antifibrinolytic agents (such as tranexamic acid and recombinant
factor VIIa) (2,3,11).
A skillful surgeon may prevent excessive operative
blood loss, the results being better when combined with other
techniques for reducing the hemorrhage. Vasopressin and oxytocin
are usually used to prevent excessive blood loss during LM
(1,12,13).
The antidiuretic hormone vasopressin is injected intramyometrially
to reduce blood loss during surgery (1,14).
Vasopressin is a direct vasoconstrictor that has been revealed to
reduce bleeding during myomectomy. Local administration of
vasopressin may cause severe or even lethal complications in spite
of the rarity of its side effects (9).
Laparoscopy became popular among gynecologists and
patients due to a faster recovery, shorter hospital stay, improved
esthetic results, and excellent long-term results in terms of the
resolution of symptoms and reproductive outcomes (8,10,15-17).
Vasopressin is a synthetic antidiuretic hormone
analogue. It has been approved in several countries being used as a
vasoconstrictor agent in numerous surgical procedures, LM included
(10,18,19).
Vasopressin produces a local vasoconstriction that lasts ~30 min
(10).
Vasopressin appears to affect the blood vessels,
however, the exact mechanism remains unknown. There are only a few
studies regarding the interesting phenomenon of the effect of
vasopressin on blood vessels.
Clinicians have followed the uterine blood flow
after vasopressin administration and reported that its effect
extends to the uterine artery. During laparoscopic surgery the
effect of vasopressin extends to both local and general circulation
(14).
In Europe, vasopressin is usually used as a
vasoconstrictor during surgery, but in some of these countries
vasopressin is not available, and thus, terlipressin (a synthetic
long-acting vasopressin analogue) is used. In Romania, a synthetic
form of vasopressin, glypressin (terlipressin acetate) 0.2 mg/ml
solution is used.
The aim of the present study was to assess the
efficacy and safety of intramyometrial glypressin administration in
reducing blood loss during LM.
Materials and methods
Patients
An observational study was conducted at the Iasi
‘Cuza Voda’ University Hospital of Obstetrics and Gynecology
(Romania). Our unit is a tertiary referral center for advanced
laparoscopic gynecological procedures in Northeastern Romania. The
present study was conducted to investigate whether the use of
glypressin played an important role in LM, comparing cases in which
LM was performed with the use of this agent and without the use of
this or any other vasoconstrictor agent.
Data was collected from 188 patients, between 23 and
54 years old, who underwent LM at our department from January 2013
to December 2019. Of these, in 64 patients glypressin was
administrated intraoperatively (group 1) and in 124 patients no
vasoconstrictor was administered (group 2). All procedures were
performed by senior surgeons at our department. All patients signed
an informed consent, including permission to use glypressin and
power morcellation to extract myomas. Ethical approval was obtained
from the Ethics Committee of ‘Cuza Voda’ Obstetrics and Gynecology
Clinical Hospital (Iasi, Romania).
Inclusion and exclusion criteria
The inclusion criteria were as follows: Myomas
50-150 mm and up to 5 fibroids, and no contraindications to the use
of glypressin. The exclusion criteria were as follows: Open
myomectomy, vaginal myomectomy, and the cases that required
conversion to open surgery, history of bleeding diathesis,
concurrent anticoagulation therapy, and endometrial or cervical
malignancies.
Study groups and parameters
assessed
The two study groups were compared in terms of size
and number of fibroids, estimated blood loss (EBL) indirectly by
using pre- and postoperative hemoglobin (Hb) and hematocrit (Ht)
values, rates of glypressin-associated intra- and postoperative
complications, and length of hospital stay.
Preoperative, operative, and postoperative data were
collected from both groups in a prospective manner. The differences
in the age, body weight, and number of myomas of patients were not
significant.
The primary outcome measure was intraoperative blood
loss during surgery, evaluated using the levels of Hb and Ht before
and after the surgery, intra- and postoperative morbidity, and
duration of hospital stay.
In cases treated with glypressin, a solution of 0.2
mg/ml (5 ml) was injected into the myometrium and the subcapsular
space around the myoma in all patients. In the patients with
multiple tumors the injection was repeated before the enucleation
of each myoma. The myometrial defect was sutured using 2-0 V-LOC
(Covidien; Medtronic Limited), barbed-delayed absorbable
sutures.
The mean pulse rate, blood pressure,
electrocardiogram, and temperature (hourly) of patients were
monitored intraoperatively and exhibited no significant changes. Hb
and Ht levels were recorded the day after surgery (postoperative
day 1) and compared with preoperative levels. The need for blood
transfusion, blood pressure, pulse, and temperature as well as the
duration of hospital stay were also recorded.
Statistical analysis
The data were entered and verified for errors in
Microsoft Excel (v. 14.0, 2010; Microsoft Corporation). Data
analysis was performed using SPSS statistical package version 18
(PASW Statistics for Windows; SPSS Inc.).
For numerical data the statistical measures were
presented as the mean ± standard deviation (minimum-maximum) and
median (1st quartile-3rd quartile). For qualitative data absolute
frequencies and percentages for each category from the set were
used.
Standard level of significance for P-value was used
and values ≤0.05 were considered to reject the null hypothesis in
statistical analysis. Normality of data were assessed with the
Shapiro-Wilk test, the differences between normally distributed
data were examined using the t-test (Student's t-test), between
non-normal distributions by Mann-Whitney U test, and for
categorical data the Fisher's exact test was used. The choice of
t-test subtype for independent data was decided based on the result
obtained using the Levene's test for comparing the variances.
Results
Demographic and myoma
characteristics
The demographic data and myoma characteristics of
the study patients are presented in Table I. There were no age differences
between the two study groups (35.9 vs. 36.4 years old; P=0.61).
There was an imbalance in the proportion for area of residence; in
group 1 the percentage of women from an urban area was greater than
in group 2 (79.7 vs. 62.1%; P=0.02). The median and the first and
third quartiles for the number of myomas were similar in the two
groups and no statistical difference was revealed (P=0.394).
However, differences were obtained between the maximum size of the
largest myomas, with higher values in group 1 (median 60 vs. 50;
P=0.01).
 | Table IDemographic characteristics of the
groups. |
Table I
Demographic characteristics of the
groups.
| Demographic
characteristics | Group 1 (glypressin,
n=64) | Group 2 (n=124) | P-value (significance
of two tails) |
|---|
| Age (years) | 35.9±5.7 (23-49) | 36.4±5.6 (23-54) | 0.61a |
| Area of residence U/R
(U%/R %) | 51/13
(79.7/20.3) | 77/47
(62.1/37.9) | 0.02b |
| No. of myomas | | | 0.394c |
|
Median | 1 (1-2) | 1 (1-2) | |
|
Mean | 1.69±0.92 (1-4) | 1.57±0.85 (1-5) | |
| Largest myoma (max.
size in mm) | 60 (50-80) | 50 (50-67.5) | 0.001c |
Studied parameters
The studied parameters are presented in Table II. The Hb levels before LP and
length of hospital stay were similar in the two study groups. Hb
and Ht (as it was presumed) exhibited similar changes in our study.
The values of Hb and Ht before LM were similar in both groups, with
no statistically significant differences (P=0.53 and P=0.97,
respectively). Statistically significant differences were
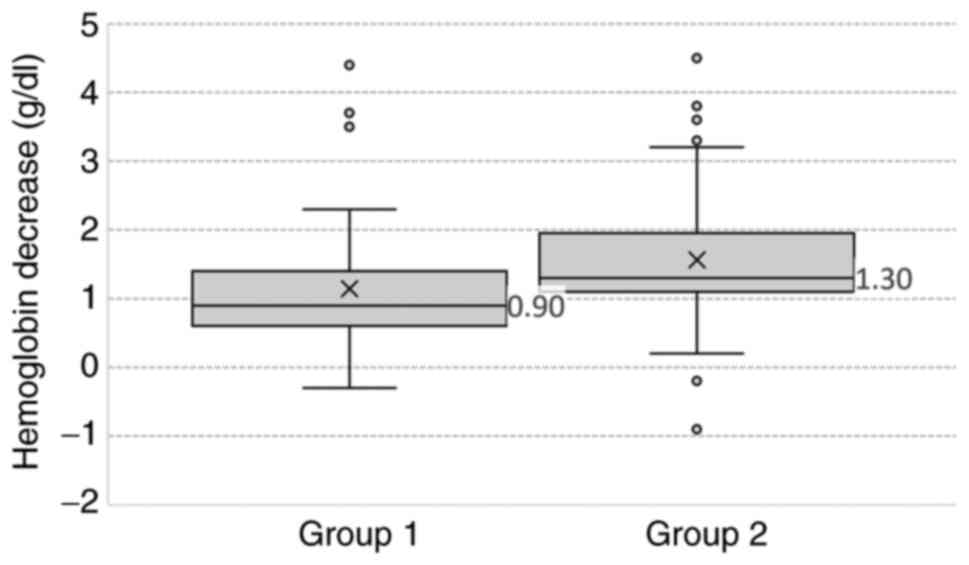
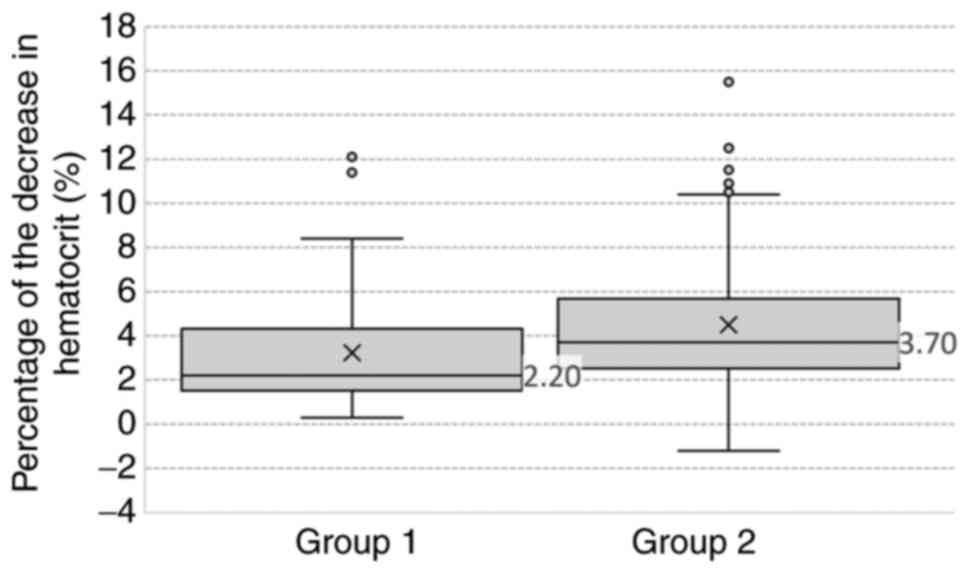
demonstrated (P<0.05) between the two groups, for Hb (Fig. 1) and Ht (Fig. 2), which were decreased after LM. Hb
was lower in group 1 compared with group 2 (medians 0.9 and 1.3,
respectively) and also the Hb after LM was higher in group 1
(P=0.02). Similarly, differences in Ht were also revealed; group 1
(median=2.2) was lower than group 2 (median=3.7) with a P=0.001 and
Ht after LM was also higher in group 1 (P=0.03). Because the values
for hospitalization days were markedly similar, there was no
statistical evidence of a possible difference.
 | Table IIStudied parameters. |
Table II
Studied parameters.
| Studied
parameters | Group 1
(glypressin) | Group 2 | Difference (95%
CI) | P-value |
|---|
| Decrease in
hemoglobin values | | | | 0.001a |
|
Median | 0.9 (0.6-1.4) | 1.3 (1.1-1.95) | | |
|
Mean | 1.14±0.83
(-0.3-4.4) | 1.56±0.79
(-0.9-4.5) | -0.67- -0.18 | |
| Hemoglobin before
LM | | | | 0.53 |
|
Median | 13.25
(12.62-13.9) | 13.15
(12.33-13.7) | | |
|
Mean | 13.1±1.09
(9.8-15.8) | 12.9±1.25
(8.1-15.6) | -0.21-0.52 | |
| Hemoglobin after
LM | | | | 0.02a |
|
Median | 12.1
(11.23-12.88) | 11.65
(10.8-12.18) | | |
|
Mean | 11.96±1.38
(8.6-14.8) | 11.39±1.22
(7.5-13.7) | 0.19-0.97 | |
| Decrease in
hematocrit values | | | | 0.001a |
|
Median | 2.2
(1.53-4.33) | 3.7
(2.52-5.67) | | |
|
Mean | 3.23±2.65
(0.3-12.4) | 4.49±2.82
(-1.2-15.5) | -2.1- -0.42 | |
| Hematocrit before
LM | | | | 0.97 |
|
Median | 39.25
(37.1-40.75) | 39.15
(37.23-40.95) | | |
|
Mean | 38.83±3.22
(28.4-47.4) | 38.67±3.58
(25.9-45.2) | -0.88-1.22 | |
| Hematocrit after
LM | | | | 0.03a |
|
Median | 35.6 (33-39.2) | 34.7
(31.9-36.9) | | |
|
Mean | 35.62±4.18
(25.1-45.2) | 34.18±3.96
(22.3-41) | 0.2-2.65 | |
| Days of
hospitalization | | | | 0.23 |
|
Median | 3 (2-4) | 3 (2-4) | | |
|
Mean | 3.39±1.41
(2-9) | 3.18±1.48
(1-8) | -0.23-0.65 | |
Discussion
A number of researchers have reported that,
especially in cases with multiple myomas, myomectomies (abdominal
or laparoscopic) are associated with excessive blood loss,
prolonged operating time, postoperative complications, and longer
hospital stay (20,21). Therefore, some physicians have
created their own particular operating methods trying to decrease
intraoperative blood loss (20).
LM remains a topic of serious debate. The procedure
is technically difficult and involves a high risk of conversion to
laparotomy. Hemorrhage is often abundant and the uterine defect can
be difficult to close due to its length, location, or important
bleeding. Because the conversion to open surgery is more time
consuming and has higher costs than laparotomy, these risks tend to
be an obstacle for the utilization of this procedure (20,22).
In Europe, vasopressin is usually used as a
vasoconstrictor during surgery, but in some of these countries
vasopressin is not available and, thus, terlipressin (a synthetic
long-acting analogue of vasopressin) is used. In Romania, a
synthetic form of vasopressin, glypressin (terlipressin acetate)
0.2 mg/ml solution. Numerous studies have indicated that
intramyometrial injection of vasopressin is effective in reducing
blood loss during LM (2,3,10,23,24).
Similar to a 2019 study by Protopapas et al,
our results indicated a significantly lower blood loss in the
glypressin group (10). Another
large clinical trial by Frederick et al (23) compared intramyometrial injection of
vasopressin vs. placebo during LM in terms of blood loss and
postoperative drop in Hb and Ht levels (23). Their results were similar to those
found in our study, namely that vasopressin was associated with a
significant reduction in mean Hb (1.7 vs. 5.3 g/dl; P0.001) and Ht
(5 vs. 13%; P<0.001) (10,23).
The safety of vasopressin has been challenged in the
past due to the reports of serious and potentially lethal
cardiovascular (CV) complications, including bradycardia,
hypotension, arrhythmias, atrioventricular block, pulmonary edema,
and even cardiac arrest (9,10,25,26).
In some European countries, such as France and Italy, vasopressin
has been withdrawn from the pharmaceutical market in response to
safety concerns.
Similar to a clinical trial by Cohen et al,
no vasopressin-related adverse events were recorded in our study
(24). Srivastava et al in
their randomized clinical trial revealed a reduction in blood loss
during LM, revealing the superiority of vasopressin over placebo
and the use of uterine tourniquet (3).
As the incidence of fibroids and childbearing age
are increasing, more women are selecting LM over hysterectomy;
thus, the addition of a vasoconstrictor agent may help in reducing
blood loss and ease the enucleation of myomas in women undergoing
LM (3).
In 2010, Walid and Heaton used pitressin (synthetic
vasopressin) during surgery to decrease bleeding. Pitressin can
contract the blood vessels in the myometrium, but it exhibited no
effect in reducing the bleeding from the pseudocapsule vessels
during LM (27).
Laparoscopic uterine artery ligation is helpful in
decreasing the bleeding during LM. However, the occlusion of these
arteries may also decrease uterine blood supply, and thus, may
represent a disadvantage in terms of the fertility rate of these
patients (20). Therefore, the use
of vasoconstrictor agents becomes extremely important and helpful
in this category of patients.
In our observational study, no side effects were
observed with glypressin administration in the circulatory system,
as the pulse rate and blood pressure were in the same normal ranges
as in the control group.
Glypressin has been demonstrated to be useful in
gynecological practices not only in LM. There are few studies on
terlipressin/glypressin in the literature, however Pirtea et
al used glypressin after clipping the hypogastric arteries to
reduce the bleeding during the laparoscopic resection of a rare
case of cesarean scar ectopic pregnancy. Thus, the surgery was
performed in 85 min with 20 ml of blood loss (28). Through the adjuvant action of
glypressin, surgical management can be improved in terms of surgery
duration and blood loss.
In addition, in the Netherlands, the use of
terlipressin in minimally invasive gynecological interventions has
become a common practice. In 2018, Overdijk et al in a
double-blind randomized trial suggested that glypressin may
decrease the intravasation and gaseous embolism in hysteroscopic
surgery, reducing the need for performing the intervention in two
steps (29).
Although, there is sufficient published evidence
concerning the vasoconstrictor action of terlipressin to support
its use in gynecological surgical interventions for reducing blood
loss, Ryckwaert et al (30)
demonstrated its vasopressor effect when directly applied on the
human uterine arteries and rat aorta (10,23,28-30).
More studies with larger sample sizes are required to accurately
evaluate other advantages, such as shorter duration of surgery and
decreased need for intraoperative blood transfusion (2).
Glypressin has made it possible to perform
beneficial and rapid uterine reconstruction with reduced blood loss
during LM. Glypressin has been revealed to be effective in reducing
blood loss during LM. In the present study the use of a dilute
solution was associated with no side effects. Blood loss remains an
issue in most cases operated without the aid of a vasoconstrictor
agent. In addition, the use of this synthetic analogue helps
preserve fertility without a negative impact on pelvic blood
supply. The addition of intramyometrial glypressin resulted in a
significant reduction in blood loss and in the postoperative drop
in Hb and Ht. The combination also improved the ease of fibroid
enucleation and myometrial reconstruction.
Due to the encouraging results obtained after the
intraoperative administration of glypressin in the management of
uterine leiomyomas, and because the literature on this topic is
scarce, adequate clinical trials on glypressin use in gynecological
hemorrhagic conditions are strongly recommended.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during this study
are available from the corresponding author on reasonable
request.
Authors' contributions
DRM, AU, LH and ID contributed to the conception of
the study, data interpretation and wrote the manuscript. DR, BT and
VLB contributed to data collection and performed the statistical
analysis. DRM and ID revised the manuscript for important
intellectual content. All authors read and approved the final
version of the manuscript.
Ethics approval and consent to
participate
Ethical approval was obtained from the Ethics
Committee of ‘Cuza Voda’ Obstetrics and Gynecology Clinical
Hospital (Iasi, Romania) and written informed consent was obtained
from all participants.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Zhang R, Shi H, Ren F and Yuan Z:
Assessment of carboprost tromethamine for reducing hemorrhage in
laparoscopic intramural myomectomy. Exp Ther Med. 10:1171–1174.
2015.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Kongnyuy EJ and Wiysonge CS: Interventions
to reduce haemorrhage during myomectomy for fibroids. Cochrane
Database Syst Rev. 8(CD005355)2014.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Srivastava S, Mahey R, Kachhawa G, Bhatla
N, Upadhyay AD and Kriplani A: Comparison of intramyometrial
vasopressin plus rectal misoprostol with intramyometrial
vasopressin alone to decrease blood loss during laparoscopic
myomectomy: Randomized clinical trial. Eur J Obstet Gynecol Reprod
Biol. 228:279–283. 2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Baird DD, Dunson DB, Hill MC, Cousins D
and Schectman JM: High cumulative incidence of uterine leiomyoma in
black and white women: Ultrasound evidence. Am J Obstet Gynecol.
188:100–107. 2003.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Seracchioli R, Rossi S, Govoni F, Rossi E,
Venturoli S and Bulletti Flamigni C: Fertility and obstetric
outcome after laparoscopic myomectomy of large myomata: A
randomized comparison with abdominal myomectomy. Hum Reprod.
15:2663–2668. 2000.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Rovio PH and Heinonen PK: Transvaginal
myomectomy with screw traction by colpotomy. Arch Gynecol Obstet.
273:211–215. 2006.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Fukuda M, Tanaka T, Kamada M, Hayashi A,
Yamashita Y, Terai Y and Ohmichi M: Comparison of the perinatal
outcomes after laparoscopic myomectomy versus abdominal myomectomy.
Gynecol Obstet Invest. 76:203–208. 2013.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Soto E, Flyckt R and Falcone T: Endoscopic
management of uterine fibroids: An update. Minerva Ginecol.
64:507–520. 2012.PubMed/NCBI
|
|
9
|
Butala BP, Shah VR, Parikh BK, Jayaprakash
J and Kalo J: Bradycardia and severe vasospasm caused by
intramyometrial injection of vasopressin during myomectomy. Saudi J
Anaesth. 8:396–398. 2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Protopapas A, Giannoulis G, Chatzipapas I,
Athanasiou S, Grigoriadis T, Kathopoulis N, Vlachos DE, Zaharakis D
and Loutradis D: Vasopressin during laparoscopic myomectomy: Does
it really extend its limits? J Minim Invasive Gynecol. 26:441–449.
2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Koh MB and Hunt BJ: The management of
perioperative bleeding. Blood Rev. 17:179–185. 2003.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Trehan N: Laparoscopic myomectomy: Methods
to control bleeding. J Gynecol Endosc Surg. 2:33–35.
2011.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wang CJ, Lee CL, Yuen LT, Kay N, Han CM
and Soong YK: Oxytocin infusion in laparoscopic myomectomy may
decrease operative blood loss. J Minim Invasive Gynecol.
14:184–188. 2007.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Shimanuki H, Takeuchi H, Kitade M, Kikuchi
I, Kumakiri J and Kinoshita K: The effect of vasopressin on local
and general circulation during laparoscopic surgery. J Minim
Invasive Gynecol. 13:190–194. 2006.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Bhave Chittawar P, Franik S, Pouwer AW and
Farquhar C: Minimally invasive surgical techniques versus open
myomectomy for uterine fibroids. Cochrane Database Syst Rev.
10(CD004638)2014.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Dubuisson JB, Chapron C, Fauconnier A and
Babaki-Fard K: Laparoscopic myomectomy fertility results. Ann N Y
Acad Sci. 943:269–275. 2001.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Buckley VA, Nesbitt-Hawes EM, Atkinson P,
Won HR, Deans R, Burton A, Lyons SD and Abbott JA: Laparoscopic
myomectomy: Clinical outcomes and comparative evidence. J Minim
Invasive Gynecol. 22:11–25. 2015.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Kimura T, Kusui C, Matsumura Y, Ogita K,
Isaka S, Nakajima A, Ohashi K, Koyama M, Azuma C and Murata Y:
Effectiveness of hormonal tourniquet by vasopressin during
myomectomy through vasopressin V1a receptor ubiquitously expressed
in myometrium. Gynecol Obstet Invest. 54:125–131. 2002.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Dubuisson J, Fauconnier A and Chapron C:
Laparoscopic myomectomy. In: Atlas of Operative Laparoscopy and
Hysteroscopy. 2nd edition. Donnez J and Nisolle M (eds). The
Parthenon Publishing Group, New York, NY, pp215-224, 2001.
|
|
20
|
Zhao F, Jiao Y, Guo Z, Hou R and Wang M:
Evaluation of loop ligation of larger myoma pseudocapsule combined
with vasopressin on laparoscopic myomectomy. Fertil Steril.
95:762–766. 2011.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Liu WM, Tzeng CR, Yi-Jen C and Wang PH:
Combining the uterine depletion procedure and myomectomy may be
useful for treating symptomatic fibroids. Fertil Steril.
82:205–210. 2004.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Dubuisson JB, Fauconnier A, Fourchotte V,
Babaki-Fard K, Coste J and Chapron C: Laparoscopic myomectomy:
Predicting the risk of conversion to an open procedure. Hum Reprod.
16:1726–1731. 2001.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Frederick J, Fletcher H, Simeon D,
Mullings A and Hardie M: Intramyometrial vasopressin as a
haemostatic agent during myomectomy. Br J Obstet Gynaecol.
101:435–437. 1994.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Cohen SL, Senapati S, Gargiulo AR, Srouji
SS, Tu FF, Solnik J, Hur HC, Vitonis A, Jonsdottir GM, Wang KC and
Einarsson JI: Dilute versus concentrated vasopressin administration
during laparoscopic myomectomy: A randomised controlled trial.
BJOG. 124:262–268. 2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Riess ML, Ulrichs JG, Pagel PS and Woehlck
HJ: Case report: Severe vasospasm mimics hypotension after
high-dose intrauterine vasopressin. Anesth Analg. 113:1103–1105.
2011.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Hobo R, Netsu S, Koyasu Y and Tsutsumi O:
Bradycardia and cardiac arrest caused by intramyometrial injection
of vasopressin during a laparoscopically assisted myomectomy.
Obstet Gynecol. 113:484–486. 2009.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Walid MS and Heaton RL: Laparoscopic
myomectomy: An intent-to-treat study. Arch Gynecol Obstet.
281:645–649. 2010.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Pirtea L, Balint O, Secosan C, Grigoras D
and Ilina R: Laparoscopic resection of caesarian scar ectopic
pregnancy after unsuccessful systemic methotrexate treatment. J
Minim Invasive Gynecol. 26:399–400. 2019.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Overdijk LE, Rademaker BMP, van Kesteren
PJM, de Haan P, Riezebos RK and Haude OCH: The HYSTER study: The
effect of intracervically administered terlipressin versus placebo
on the number of gaseous emboli and fluid intravasation during
hysteroscopic surgery: Study protocol for a randomized controlled
clinical trial. Trials. 19(107)2018.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Ryckwaert F, Virsolvy A, Fort A, Murat B,
Richard S, Guillon G and Colson PH: Terlipressin, a provasopressin
drug exhibits direct vasoconstrictor properties: Consequences on
heart perfusion and performance. Crit Care Med. 37:876–881.
2009.PubMed/NCBI View Article : Google Scholar
|