1. Introduction
The hantavirus infection is part of a rare group of
zoonoses with increasing frequency and geographic spread in the
last years. The Hantavirus genus includes various human
pathogenic serotypes, whose hosts are mainly wild rodents (1). Hantaviruses are classified in two
large groups, according to their geographic distribution: The Old
World hantaviruses and The New World hantaviruses (1). The Old World hantaviruses cause
hemorrhagic fever with renal syndrome (HFRS), while the New World
hantaviruses are associated with hantavirus cardiopulmonary
syndrome (CPS) (1).
Onset of HFRS is similar to a flu-like syndrome,
with fever and prospects to evolve into an acute kidney injury
(AKI) with severe thrombocytopenia, anemia and coagulation
dysfunction (2), mimicking the
thrombotic microangiopathy (TMA) from the hemolytic uremic syndrome
(HUS), with which it is frequently confused. The renal dysfunction
is manifested through decrease in the glomerular filtration rate,
hematuria and transient proteinuria, often in the nephrotic range,
which indicates a lesion of the glomerular filtration barrier
(2). The typical histological
aspect is that of an acute tubulointerstitial nephritis with
hemorrhage in the outer medulla, but glomerular endothelial lesions
are also present (3).
This review emphasizes the importance of the renal
biopsy in the diagnosis of hantavirus hemorrhagic nephritis and
draws attention on this zoonosis that may be highly underdiagnosed
in the Balkan Peninsula.
2. Epidemiology
Hantavirus infection belongs to a group of rare
zoonotic diseases but with increasing incidence and geographical
range in the last years. The genus Hantavirus is a member of
the Bunyaviridae family and includes over 28 serotypes that
cause infections in humans, rodents being regarded as the main
reservoir for the pathogen. Hantaviruses have a diameter of 80-120
nm and are enveloped viruses; the lipoprotein envelope being easily
inactivated by disinfectants. They are characterized by a
negative-sensed trisegmented single-stranded RNA genome (2). The envelope has glycoprotein
projections with hemagglutinin activity (Gn and Gc). The genome
comprises three segments (small, S; medium, M; and large, L) coding
the viral nucleocapsid protein (N), two envelope glycoproteins and
the genome associated RNA-dependent RNA-polymerase (4). The viruses replicate within the
cytoplasm of the infected cells and are released in the bloodstream
by virus budding.
The history of hantaviruses initiated between 1951
and 1953, during the Korean war, when the first cases of
hemorrhagic fever (Korean hemorrhagic fever) were reported in an
area along the Hantaan River, after which they were named (1). The virus was isolated in 1978 by Ho
Wang Lee from lung tissue belonging to the striped field mouse
Apodemus agrarius that was experimentally inoculated with
the pathogen of the Korean hemorrhagic fever (5). Hantaviruses are spread worldwide in
endemic areas and their classification is performed according to
the geographical distribution: The Old World hantaviruses which
cause HFRS and The New World hantaviruses which cause CPS; these
diseases are responsible for the hospitalization of 150.000-200.000
individuals worldwide each year (2,6).
Within the Old World hantaviruses, which are usually found in
Europe and Asia, the best known species are Puumala
orthohantavirus (PUUV), Hantaan orthohantavirus (HTNV),
Seoul orthohantavirus (SEOV) and Dobrava-Belgrade
orthohantavirus (DOBV), the latter being spread mostly in the
Balkan Peninsula, but also the Saaremaa virus (SAAV) which is
genetically related to DOBV, and the Tula virus (TULV) (2). DOBV, the most prevalent serotype
observed in the Balkan Peninsula, is responsible for a 12%
mortality rate associated with infection (7). There are four rodent species
constituting the virus host: Apodemus, Microtus,
Myodes and Rattus; the rodents do not develop the
disease, but their growth may be affected as a consequence of the
infection (8,9) and they can also spread the virus to
other animals: Moose, red fox, bat, domestic cat and dog (10-13).
The New World hantaviruses are found in America; the best known are
Sin Nombre virus and Andes virus, the latter being the only
Hantavirus for which person-to-person transmission may take place
(14,15).
The European Center for Disease Prevention and
Control published a list with 29 countries reporting 1,826 cases of
hantavirus infection for the year 2018, with most of the cases
(97%) involving PUUV (16).
However, there is the possibility that most HFRS cases, being mild
and moderate forms of disease, are underdiagnosed and thus, the
incidence of the disease in Europe may be underestimated. The
Dobrava strain was confirmed as the causative agent in endemic
nephropathy cases in Central and South Eastern Europe (17).
Humans do not belong to the natural host range of
hantaviruses and infection generally occurs by inhalation of
virus-containing aerosols from rodent faeces, urine and saliva, but
also by rodent bite and skin lesions (18,19).
People at increased risk of infection are those who live or work in
rural areas and come into close contact with virus reservoirs:
Forestry workers, farmers, hunters, and military personnel (who can
be easily infected during military exercises) (2). The existence of a genetic
susceptibility involved in the pathogenesis of the disease was
recently revealed: The HLA-B8 DR3 and DQ2 haplotypes are associated
with a severe clinical course of the infection, while HLA-B27 is
associated with a benign clinical course of the infection (20-23).
There are two peaks of incidence over the course of one year: The
first peak is in August, during the vacation season that starts in
July, and the second peak is in November and December when the
onset of cold weather causes mice to search for shelter in people's
homes (24).
3. Pathogenesis
In patients with endemic nephropathy, hantaviruses
infect renal endothelial cells, podocytes and tubular epithelial
cells. The pathogenesis of the infection is complex and not yet
fully understood and it is characterized by endothelial damage with
enhanced capillary permeability, disruption of tight junctions of
tubular epithelial cells and alterations of the podocyte
cytoskeleton (25-29).
Endothelial integrity is the result of a complex
interaction between endothelial cells, thrombocytes and immune
cells (29).
Hantaviruses infect the susceptible cells
(endothelial cells, tubular epithelial cells and glomerular cells)
by interaction of the viral envelope glycoproteins (Gn and Gc) with
β-integrin surface molecules. Both pathogenic and nonpathogenic
hantavirus strains infect the human cells, but the receptors they
use for cellular entry are different: αvβ1 and αvβ3, respectively
(26); these surface receptors,
usually involved in matrix adhesion, are being blocked in the
process of virus attachment. Thus, the virus affects the renal
tubular epithelium, the podocytes and the capillary endothelial
cells by disturbing the integrity of cell-to-cell contacts and also
by stimulating the immune system. Evidence has revealed that
immature dendritic cells are involved in the dissemination of
hantaviruses, as they express on their surface the αvβ3 integrin
and can be found in the proximity of epithelial cells (30). Hantavirus-infected dendritic cells
help spread the virus by migrating through lymphatic vessels to the
regional lymph nodes and then to the endothelial cells (31), as they exert their role of
antigen-presenting cells and induce the immune activation of T
lymphocytes. High levels of cytotoxic CD8+ T lymphocytes
were detected in the serum of patients with HFRS and the evidence
revealed that the endothelial damage was not caused by direct
cellular cytotoxicity of the hantavirus, but by pro-inflammatory
cytokines produced by the virus-specific T lymphocytes that attack
the endothelial cells expressing viral antigens on their surface
(32), thus contributing to the
capillary leakage. Infection of the endothelial cells initiates an
inflammatory response involving the activation of the complement
system and the release of pro-inflammatory cytokines such as TGF,
interferons, interleukins (IL-6 and IL-10) and TNF (33). Increased levels of IFNγ, IL-10 and
TNFα were revealed in the serum of patients with PUUV and DOBV and
also in patients with severe clinical course of disease (34). Several signaling cascades that use
vascular endothelial growth factor (VEGF), IL-6 or urokinase
plasminogen activator receptor (uPAR) as mediators have been
described to be involved in the regulation of podocyte function;
these mediators are also known to be upregulated in endemic
nephropathy (35), thus supporting
the involvement of podocyte alteration in the hantavirus infection
pathogenesis.
4. Pathological findings
The most frequent pattern of injury observed in HFRS
kidney biopsy specimens is acute tubulointerstitial nephritis
(3).
The kidney biopsy performed in the acute phase of
the disease course can raise the suspicion of hantavirus infection
immediately after removal of tissue, as it reveals macroscopic
features of hemorrhage in the renal medulla (3).
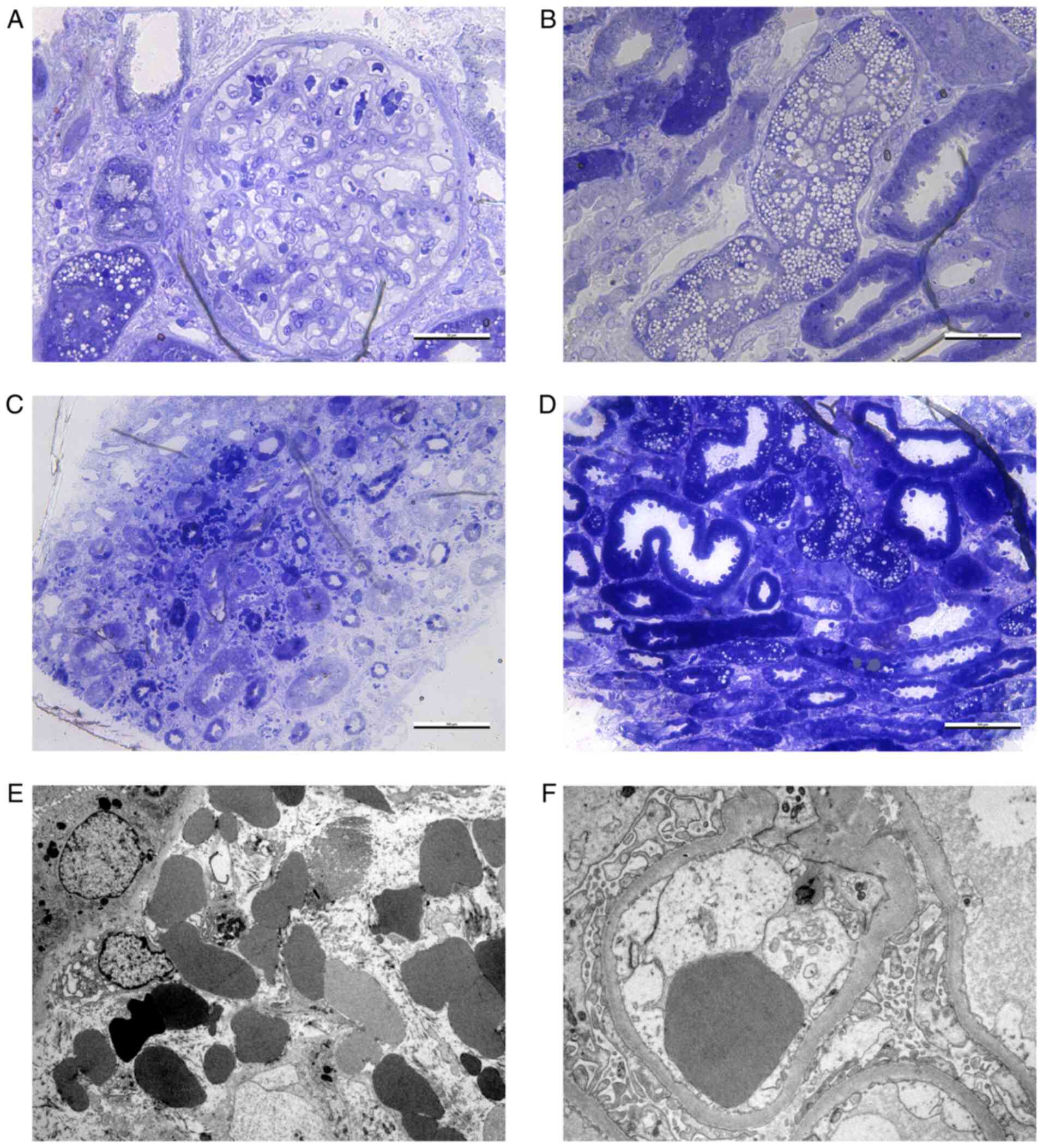
Light microscopy (LM) can reveal normal glomeruli or
glomeruli with increased mesangial cellularity. A few studies have
described cases of mesangiocapillary glomerulonephritis in
hantavirus infection (3,36). The pathognomonic histopathological
finding is the interstitial hemorrhage (with both isomorphic and
dysmorphic erythrocytes), most commonly found in the outer medulla,
that can compress the renal tubules (Fig. 1). The interstitial hemorrhage is
present in 20-60% of the kidney biopsy specimens in the acute phase
of hantavirus infection (17). LM
also reveals interstitial and periglomerular lymphoplasmacytic cell
infiltrates, tubulitis and arterial endothelial swelling.
In immunofluorescence (IF), the staining is usually
negative, but a few studies have described mild granular C1q, C3,
IgG and IgM deposits along the glomerular capillary walls, thus
supporting the theory of classical complement pathway activation in
hantaviral infection (11,36-38).
Electron microscopy (EM) can reveal diffuse foot
process effacement, denudation of the tubular epithelial cells,
resorption droplets in the tubules (in cases of high levels of 24-h
proteinuria) and endothelial swelling in the glomerular or
peritubular capillaries (Fig. 1);
the infected cells can display hypertrophy of the Golgi apparatus
(3). The foot process effacement
and detachment of podocytes from the glomerular basement membranes
explains the nephrotic range proteinuria observed in some cases of
AKI and strengthens the role of podocyte cytoskeletal alterations
in the pathogenicity of HSRF (27,28,39).
5. Clinical manifestations
Hantavirus infection causes two types of clinical
syndromes in humans: HFRS and CPS; the difference consisting in the
implicated strains and the affected vascular circulation:
Capillaries from renal medulla and pulmonary capillaries,
respectively. The common lesion is the increased capillary
permeability and both forms of disease begin with fever syndrome
and flu-like symptoms and have in common thrombocytopenia,
left-shifted leukocytosis and arterial hypotension (1,2,29). In
the following sections, detailed aspects of HFRS, also known as
endemic nephropathy, are presented. Clinical presentation can range
from mild to severe forms of disease, depending on the causing
strain. Therefore, strains such as HTNV and DOBV can cause severe
forms of disease with mortality ranging from 5 to 15% and strains
such as SEOV, PUUV and SAAV in general cause moderate or mild forms
of disease with a mortality below 1% (18). The disease evolution has five
chronological phases including:
i) Febrile phase
After an incubation period of 2-4 weeks, the disease
begins with flu-like syndrome including high grade fever, myalgia,
low back pain, headache, abdominal pain, nausea and vomiting. At
this stage most clinicians will first look for a cause of the fever
and the following basic laboratory tests will be necessary:
Erythrocyte sedimentation rate or C reactive protein, hemoglobin,
platelet count and differential, electrolytes, creatinine,
abdominal ultrasonography and other imaging studies (40). This phase lasts 3-7 days and it can
be accompanied by ophthalmological disorder; in PUUV infection, a
decrease of intraocular pressure was reported in 88% of the cases,
myopia in 78% of the cases and visual acuity reduction in 87%
(41).
ii) Hypotensive phase
This phase lasts between a few hours and 2 days. It
can appear as severe arterial hypotension and irreversible shock.
This is the phase when thrombocytopenia, leukocytosis, haemorrhagic
manifestations such as skin and mucous petechiae, hematemesis,
epistaxis, melena, hematuria or even intracerebral haemorrhage
appear; in addition, the production of urine decreases in this
phase.
iii) Oliguric phase
The duration of this phase is between 3 and 7 days
and it can be accompanied by abdominal or low back pain. Glomerular
filtration rate diminishes and oliguria or even anuria appear,
nitrogen waste products increase and microscopic hematuria and
proteinuria occur. This is the phase when arterial hypotension
disappears and patients can even develop arterial hypertension
through hydrosaline retention.
iv) Polyuric phase
The duration of this phase ranges from a few days to
a few weeks. The recovery of renal function begins and diuresis
increases to a few liters per day, thus, important
hydroelectrolytic disorders may occur.
v) Convalescence
This phase lasts from 2 to 6 months. This is the
period when renal function and laboratory abnormalities are
completely recovering; however, there are cases which can develop
long-term complications and persistent complications such as
arterial hypertension and chronic kidney disease (42-44).
The most frequent laboratory abnormalities are
thrombocytopenia, leukocytosis, increase of C-reactive protein,
increase of creatinine, proteinuria and hematuria. Increased levels
of procalcitonin, IFNγ, TNFα and IL-10, especially in patients with
DOBV infection have also been revealed (34,45).
In PUUV infection, IL-6 serum level has been revealed to be
correlated with thrombocytopenia and severity of renal function
damage and it is used as a disease severity marker (46); the urinary level of IL-6 has been
demonstrated to be correlated with proteinuria, suggesting local
production of this cytokine (47).
It is already recognized that alternative pathway
complement activation contributes to pathogenesis of PUUV infection
(48,49) and there is evidence that increased
serum levels of the C5b-C9 complex and reduced C3 level from acute
phases appear, especially in patients with pulmonary radiologic
abnormalities (24).
Thrombocytopenia is recognized as a severity marker
of hantavirus infection (46) and
is caused by peripheral consumption related to endothelial
activation (24,50). It has been demonstrated that
platelet adhesion to infected endothelial cells is directed by
hantavirus pathogenic strains through β3 integrin receptor
(25). This results in alteration
of platelet activation with reduction of their count and loss of
vascular integrity, so that the endothelial lesions may promote
coagulation activation and fibrinolysis, effects observed through
increased prothrombin and D-dimers (50). Literature data has revealed that
platelet count is a predictor of severity and progression of
disease (51,52).
Most frequently the recovery of hantavirus infection
is complete, but there are cases which evolve with acute or chronic
complications. The majority of the acute complications that can be
life-threatening are caused by hydroelectrolytic disorders, shock
with ischemia, haemorrhage or necrosis (24). Thus, in PUUV and DOBV strain
infections, pituitary gland haemorrhage and necrosis,
meningoencephalitis, AKI with renal replacement therapy, pulmonary
edema, pericarditis, myocarditis, disseminated intravascular
coagulation have been observed (6,53-66)
(Table I).
 | Table IAcute complications of hantavirus
infection [adapted from Vaheri et al (24)]. |
Table I
Acute complications of hantavirus
infection [adapted from Vaheri et al (24)].
| Acute complications
in hantavirus infection with PUUV and DOBV serotypes |
|---|
| Renal |
|
Acute kidney
injury and renal replacement therapy required (53,54) |
|
Cardiopulmonary |
|
Pulmonary
edema (55) |
|
Arterial
hypotension and shock (6) |
|
Pericarditis,
myocarditis (56) |
| Neurological |
|
Meningoencephalitis
(57,58) |
|
Seizures
(59) |
|
Pituitary
haemorrhage (60) |
|
Guillain-Barre
syndrome (61) |
|
Neurogenic
bladder and hemiparesis (62,63) |
| Hematological |
|
Disseminated
intravascular coagulation (50) |
| Digestive |
|
Pancreatitis
(64,65) |
| Others |
|
Multiple
organ dysfunction syndrome (66) |
Arterial hypotension is one of the most common acute
complications of HFRS, being caused by dehydration secondary to
nausea and vomiting and by vascular injury from complement
activation (33,48,49),
thrombocytes (24,46,50)
and immune effector cell activation that synthetize vasoactive
mediators, interleukins and TNF. Patients can also develop arterial
hypertension based on decreased glomerular filtration rate in the
oliguric phase of disease, but also on capillary leak phenomenon
with high protein extravasation to third space leading to weight
gain (revealed by severe hypoalbuminemia), acute pulmonary edema
and renal interstitial edema.
The most frequent chronic complications of
hantavirus infection are hormone deficiency (hypopituitarism and
hypothyroidism) and tubulointerstitial nephritis with tubular
proteinuria developed after years from infection, as well as
membranoproliferative glomerulonephritis and arterial hypertension
(67-72)
(Table II).
 | Table IIChronic complications of Hantavirus
infection [adapted from Vaheri et al (24)]. |
Table II
Chronic complications of Hantavirus
infection [adapted from Vaheri et al (24)].
| Chronic
complications of hantavirus infection |
|---|
| Renal |
|
Chronic
tubulointerstitial nephritis (67) |
|
Membranoproliferative
glomerulonephritis (36,68) |
| Cardiovascular |
|
Arterial
hypertension (69) |
| Endocrine |
|
Testicular
insufficiency (70) |
|
Hypopituitarism
(70) |
|
Hypothyroidism
(71,72) |
One of the proposed mechanisms for arterial
hypertension in hantavirus infection is the renal microvascular
impairment along with significant tubulointerstitial inflammation.
Every factor inducing vasoconstriction in renal medulla and the
adjacent cortex can cause arterial hypertension. Hantavirus
infection constitutes an initiating factor for renal
vasoconstriction, a theory supported by the histological appearance
with congestion and haemorrhage around the vessels in the outer
medulla and corticomedullary junction and also by the endothelial
injury (73).
6. Diagnosis and treatment
Diagnosis is performed based on epidemiological,
clinical and laboratory findings. However, it has to be suspected
whenever a patient presents with fever, headache, abdominal or low
back pain, AKI and thrombocytopenia and confirmed with serological
testing. At hospitalization, patients usually already have IgM and
IgG anti-hantavirus antibodies because they become positive along
with the occurrence of symptoms. The most commonly used method of
serological detection of antibodies is by indirect ELISA for IgM
and IgG, however indirect IF and rapid immunochromatographic tests
are also used. Hantavirus infection can also be revealed through
detection of the viral genome from the serum of patients using
RT-PCR and the level of viremia has been revealed to be correlated
with disease severity (74-77).
Due to the specificity of interstitial hemorrhage on the kidney
biopsy specimens of patients with HFRS as well as the frequency of
chronic tubulointerstitial nephritis as a complication of this
pathology, a kidney biopsy should be considered as a diagnostic
tool. It is important to estimate the risk/benefit ratio of kidney
biosy in these patients, as thrombocytopenia associated to HFSR
increases the risk of bleeding. Careful attention should be paid to
the medical history of patients regarding the treatments that could
interfere with the level of thrombocytes and coagulation tests such
as oral anticoagulation (OA) recognizing that both traditional OA
and new oral anticoagulants are used at an increasing scale
(78).
Considering that the hantavirus typically causes a
self-limiting infection and it resolves in 2-3 weeks, treatment is
mainly supportive. Occasionally renal replacement therapy is
required (in <5% of the cases) (24,79)
and this is mostly due to hypervolemia. Thus, an effective option
of supportive treatment is the proper monitoring of
hydroelectrolytic balance and avoidance of fluid retention,
especially for patients with anuria (2). Platelet transfusions may be provided
in cases of severe thrombocytopenia with bleeding risk (2,43).
Among the antiviral drugs, anti-hantavirus activity
was demonstrated with ribavirin in vitro and in vivo
and there are several Chinese studies on patients with HFRS that
exhibited an important reduction of mortality and morbidity when it
was administered in the first 5 days after appearance of symptoms
(80,81). Another study revealed that early
intravenous administration of ribavirin in HFRS reduced the
severity of AKI and the occurrence of oliguria (82).
Treatment with icatibant, an antagonist of
bradykinin B2 receptor, may be beneficial in severe forms of HFRS
with important capillary leakage phenomenon (83,84)
due to the role of bradykinin in vascular permeability.
Corticosteroids are not part of the routine viral
infection therapy, however, considering the damage of the
hantavirus on endothelial cells and the cytokine storm with
increased capillary permeability, it appears that
methylprednisolone has beneficial effects on the pathogenesis of
the disease, controlling the immune response. A retrospective study
from Chile that included 22 patients with CPS treated with
high-dose methylprednisolone demonstrated its efficacy in
decreasing mortality (85), and
therefore this treatment was implemented for hantavirus infection
in various medical centers (86).
Conversely, there are phase 2 studies that tested the safety and
efficacy of intravenous administration of methylprednisolone which
did not demonstrate favorable effects (86), therefore treating hantavirus
infection with corticosteroids depends on the decision of the
clinician.
7. Conclusions
Hantavirus endemic nephropathy belongs to a group of
rare zoonoses in the Balkan Peninsula and the most frequently
involved serotypes are Dobrava and Puumala. The disease begins with
flu-like symptoms such as fever and progresses to AKI with severe
thrombocytopenia, anemia and coagulation disorders, symptoms
similar to microangiopathy in hemolytic uremic syndrome with which
it may often be confused. In a geographic region with only sporadic
cases, multiple diagnostic difficulties are encountered, especially
related to the absence of initial diagnosis suspicion. Therefore,
the need to include this pathology in the algorithm of differential
diagnosis of pathologies associated with thrombocytopenia, anemia,
hepatic cytolysis syndrome and renal injury, is emphasized.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
GL, ML, IA, LB, AB, RS and GI contributed to the
conception, writing, review and editing of the manuscript. ML was
responsible for methodology. RS, ML and IA were responsible for
resources; GL and IA contributed to the data curation. GL, ML, IA
and GI contributed to writing of the original draft. GI supervised
the study. All authors have read and approved the final version of
the manuscript to be published.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Zeier M, Handermann M, Bahr U, Rensch B,
Muller S, Kehm R, Muranyi W and Darai G: New ecological aspects of
hantavirus infection: A change of A paradigm and a challenge of
prevention-a review. Virus Genes. 30:157–180. 2005.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Avšič-Županc T, Saksida A and Korva M:
Hantavirus infections. Clin Microbiol Infect. 21S:e6–e16.
2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Satoskar A, Nadasdy T and Silva F: Acute
postinfectious glomerulonephritis and glomerulonephritis caused by
persistent bacterial infection. In Heptinstall's Pathology of the
Kidney, 7th edition. Jannette JG, Olson JL, Silva FG and D'Agati VD
(eds). Lippincott Williams and Wilkins, Philadelphia, pp678-798,
2015.
|
|
4
|
van Regenmortel MHV, Fauquet CM, Bishop
DHL, Carstens EB, Estes MK, Lemon SM, Maniloff J, Mayo MA, McGeoch
DJ, Pringle CR and Wickner RB (eds): Virus Taxonomy. Seventh Report
of the International Committee on Taxonomy of Viruses. 1st edition.
Academic Press, San Diego, San Francisco, New York, Boston, London,
Sydney, Tokyo, 2000.
|
|
5
|
Lee HW, Lee PW and Johnson KM: Isolation
of the etiologic agent of Korean hemorrhagic fever. J Infect Dis.
137:298–308. 1978.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Vapalahti O, Mustonen J, Lundqvist Å,
Henttonen H, Plyusnin A and Vaheri A: Hantavirus infections in
Europe. Lancet Infect Dis. 3:653–661. 2003.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Avsic-Zupanc T, Petrovec M, Furlan P, Kaps
R, Elgh F and Lundkvist A: Hemorrhagic fever with renal syndrome in
the Dolenjska region of Slovenia-a 10-year survey. Clin Infect Dis.
28:860–865. 1999.PubMed/NCBI View
Article : Google Scholar
|
|
8
|
Steer A: Pathogenesis of renal changes in
epidemic hemorrhagic fever. In: The Kidney. IAP Monograph. Mostofi
FK and Smith DE (eds). Williams & Wilkins, Baltimore,
pp476-486, 1996.
|
|
9
|
Jokinen EJ, Lähdevirta J and Collan Y:
Nephropathia epidemica: Immunohistochemical study of pathogenesis.
Clin Nephrol. 9:1–5. 1978.PubMed/NCBI
|
|
10
|
Zaki SR, Greer PW, Coffield LM, Goldsmith
CS, Nolte KB, Foucar K, Feddersen RM, Zumvalt RE, Miller GL, Khan
AS, et al: Hantavirus pulmonary syndrome: Pathogenesis of an
emerging infectious disease. Am J Pathol. 146:552–579.
1995.PubMed/NCBI
|
|
11
|
Kim S, Kang ET, Kim YG, Han JS, Lee JS,
Kim JI, Hall WC, Dalrymple JM and Peters CJ: Localization of
Hantaan viral envelope glycoproteins by monoclonal antibodies in
renal tissues from patients with Korean hemorrhagic fever H. Am J
Clin Pathol. 100:398–403. 1993.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Groen J, Bruijn JA, Gerding MN, Jordans
JG, Moll van Charante AW and Osterhaus AD: Hantavirus antigen
detection in kidney biopsies from patients with nephropathia
epidemica. Clin Nephrol. 46:379–383. 1996.PubMed/NCBI
|
|
13
|
Poljak M and Avšič Županc T:
Immunohistochemical detection of Hantaan virus antigen in renal
tissue from patient with hemorrhagic fever with renal syndrome.
Nephron. 67(252)1994.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Padula PJ, Edelstein A, Miguel SD, López
NM, Rossi CM and Rabinovich RD: Hantavirus pulmonary syndrome
outbreak in Argentina: Molecular evidence for person-to-person
transmission of Andes virus. Virology. 241:323–330. 1998.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Martinez VP, Bellomo C, San Juan J, Pinna
D, Forlenza R, Elder M and Padula PJ: Person-to-person transmission
of Andes virus. Emerg Infect Dis. 11:1848–1853. 2005.PubMed/NCBI View Article : Google Scholar
|
|
16
|
European Centre for Disease Prevention and
Control: https://www.ecdc.europa.eu/en/publications-data/hantavirus-infection-annual-epidemiological-report-2018.
Accessed June 25, 2020.
|
|
17
|
Antoine M, Langlois ME, Bres E, Rabeyrin
M, Reynes JM and Deeb A: Imported haemorrhagic fever with renal
syndrome caused by Dobrava-Belgrade hantavirus in France. Clin
Kidney J. 14:1014–1016. 2020.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Shirai H, Yashima J, Tojimbara T and Honda
K: Thrombotic microangiopathy caused by oral contraceptives in a
kidney transplant recipient. Nephrology (Carlton). 21 (Suppl
1):S41–S43. 2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Douron E, Moriniere B, Matheron S, Girard
PM, Gonzalez JP, Hirsch F and McCormick JB: HFRS after a wild
rodent bite in the Haute-Savoie- and risk of exposure to
Hantaan-like virus in a Paris laboratory. Lancet. 1:676–677.
1984.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Mustonen J, Partanen J, Kanerva M, Pietilä
K, Vapalahti O, Pasternack A and Vaheri A: Genetic susceptibility
to severe course of nephropathia epidemica caused by Puumala
hantavirus. Kidney Int. 49:217–221. 1996.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Mäkelä S, Mustonen J, Ala-Houhala I, Hurme
M, Partanen J, Vapalahti O, Vaheri A and Pasternack A: Human
leukocyte antigen-B8-DR3 is a more important risk factor for severe
Puumala hantavirus infection than the tumor necrosis factor-alpha
(308) G/A polymorphism. J Infect Dis. 186:843–846. 2002.PubMed/NCBI View
Article : Google Scholar
|
|
22
|
Paakkala A, Mäkelä S, Hurme M, Partanen J,
Huhtala H and Mustonen J: Association of chest radiography findings
with hostrelated genetic factors in patients with nephropathia
epidemica. Scand J Infect Dis. 40:254–258. 2008.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Mustonen J, Partanen J, Kanerva M, Pietilä
K, Vapalahti O, Pasternack A and Vaheri A: Association of HLA B27
with benign clinical course of nephropathia epidemica caused by
Puumala hantavirus. Scand J Immunol. 47:277–279. 1998.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Vaheri A, Henttonen H, Voutilainen L,
Mustonen J, Sironen T and Vapalahti O: Hantavirus infections in
Europe and their impact on public health. Rev Med Virol. 23:35–49.
2013.PubMed/NCBI View
Article : Google Scholar
|
|
25
|
Gavrilovskaya IN, Gorbunova EE and Mackow
ER: Pathogenic hantaviruses direct the adherence of quiescent
platelets to infected endothelial cells. J Virol. 84:4832–4839.
2010.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Gavrilovskaya IN, Brown EJ, Ginsberg MH
and Mackow ER: Cellular entry of hantaviruses which cause
hemorrhagic fever with renal syndrome is mediated by beta3
integrins. J Virol. 73:3951–3959. 1999.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Krautkramer E, Grouls S, Stein N, Reiser J
and Zeier M: Pathogenic old word hantaviruses infect renal
glomerular and tubular cells and induce disassembling of
cell-to-cell contacts. J Virol. 85:9811–9823. 2011.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Boehlke C, Hartleben B, Huber T, Hopfer H,
Walz G and Neumann-Haefelin E: Hantavirus infection with severe
proteinuria and podocyte foot-process effacement. Am J Kidney Dis.
64:452–456. 2014.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Krautkramer E, Zeier M and Plyusnin A:
Hantavirus infection: An emerging infectious disease causing acute
renal failure. Kidney Int. 83:23–27. 2013.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Peebles RS Jr and Graham BS: Viruses,
dendritic cells and the lung. Respir Res. 2:245–249.
2001.PubMed/NCBI View
Article : Google Scholar
|
|
31
|
Schonrich G, Rang A, Lutteke N, Raftery
MJ, Charbonnel N and Ulrich RG: Hantavirus-induced immunity in
rodent reservoirs and humans. Immunol Rev. 225:163–189.
2008.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Terajima M and Ennis FA: T cells and
pathogenesis of hantavirus and cardiopulmonary syndrome and
hemorrhagic fever with renal syndrome. Viruses. 3:1059–1073.
2011.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Safronetz D, Zivcec M, Lacasse R, Feldmann
F, Rosenke R, Long D, Haddock E, Brining D, Gardner D, Feldmann H
and Ebihara H: Pathogenesis and host response in Syrian hamsters
following intranasal infection with Andes virus. PLoS Pathog.
7(e1002426)2011.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Saksida A, Wraber B and Avsic-Zupanc T:
Serum levels of inflammatory and regulatory cytokines in patients
with hemorrhagic fever with renal syndrome. BMC Infect Dis.
11(142)2011.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Hägele S, Müller A, Nusshag C, Reiser J,
Zeier M and Krautkrämer E: Motility of human renal cells is
disturbed by infection with pathogenic hantaviruses. BMC Infect
Dis. 18(645)2018.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Mustonen J, Mäkelä S, Heli H, Helanterä A,
Miettinen M, Partanen J and Pasternack A: Mesangiocapillary
glomerulonephritis caused by Puumala hantavirus infection. Nephron.
89:402–407. 2001.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Papadimitriou M: Hantavirus nephropathy.
Kidnei Int. 48:887–902. 1995.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Li XZ, Jin XM and Li H: Semi-thin sections
and electron microscopy in epidemic hemorrhagic fever. Trans Harbin
Med Univ. 3:43–46. 1984.
|
|
39
|
Clement J, Lee APK, Verpooten GA, Laenen
L, Vergote V, De Samblanx H, Berneman ZN, Van Ranst M and Maes P:
Acute hantavirus infection presenting as haemolytic-uraemic
syndrome (HUS): The importance of early clinical diagnosis. Eur J
Clin Microbiol Infect Dis. 37:135–140. 2018.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Gheorghe G, Ceobanu G, Gheorghe F, Bratu
OG, Bacalbasa N, Bungau S and Diaconu CC: Fever of unknown origin.
Rom J Mil Med. 123(213)2020.
|
|
41
|
Hautala N, Kauma H, Vapalahti O, Mähönen
SM, Vainio O, Vaheri A and Hautala T: Prospective study on ocular
findings in acute Puumala hantavirus infection in hospitalised
patients. Br J Ophthalmol. 95:559–562. 2011.PubMed/NCBI View Article : Google Scholar
|
|
42
|
McCaughey C and Hart CA: Hantaviruses. J
Med Microbiol. 49:587–599. 2000.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Linderholm M and Elgh F: Clinical
characteristics of hantavirus infections on the Eurasian continent.
Curr Top Microbiol Immunol. 256:135–151. 2001.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Kruger DH, Ulrich R and Lundkvist AA:
Hantavirus infections and their prevention. Microbes Infect.
3:1129–1144. 2001.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Jereb M, Lunacek NK, Kotar T, Saksida A,
Petrovec M and Avsic-Zupanc T: Procalcitonin in hantavirus
infections. Scand J Clin Lab Invest. 71:287–291. 2011.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Outinen TK, Mäkelä SM, Ala-Houhala IO,
Huhtala HS, Hurme M, Paakkala AS, Pörsti IH, Syrjänen JT and
Mustonen JT: The severity of Puumala hantavirus induced
nephropathia epidemica can be better evaluated using plasma
interleukin-6 than C-reactive protein determinations. BMC Infect
Dis. 10(132)2010.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Mäkelä S, Mustonen J, Ala-Houhala I, Hurme
M, Koivisto AM, Vaheri A and Pasternack A: Urinary excretion of
interleukin-6 correlates with proteinuria in acute Puumala
hantavirus-induced nephritis. Am J Kidney Dis. 43:809–816.
2004.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Paakkala A, Mustonen J, Viander M, Huhtala
H and Pasternack A: Complement activation in nephropathia epidemica
caused by Puumala hantavirus. Clin Nephrol. 53:424–431.
2000.PubMed/NCBI
|
|
49
|
Sane J, Laine O, Mäkelä S, Paakkala A,
Jarva H, Mustonen J, Vapalahti O, Meri A and Vaheri A: Complement
activation in Puumala hantavirus infection correlates with disease
severity. Ann Med. 44:468–475. 2012.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Laine O, Mäkelä S, Mustonen J, Huhtala H,
Szanto T, Vaheri A, Lassila R and Joutsi-Korhonen L: Enhanced
thrombin formation and fibrinolysis during acute Puumala hantavirus
infection. Thromb Res. 126:154–158. 2010.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Wang M, Wang J, Wang T, Li J, Hui L and Ha
X: Thrombocytopenia as a predictor of severe acute kidney injury in
patients with Hantaan virus infections. PLoS One.
8(e53236)2013.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Rasche FM, Uhel B, Krüger DH, Karges W,
Czock D, Hampl W, Keller F, Meisel H and von Müller L:
Thrombocytopenia and acute renal failure in Puumala hantavirus
infections. Emerg Infect Dis. 10:420–425. 2004.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Braun N, Haap M, Overkamp D, Kimmel M,
Alscher MD, Lehnert H and Haas CS: Characterization and outcome
following Puumala virus infection: A retrospective analysis of 75
cases. Nephrol Dial Transplant. 25:2997–3003. 2010.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Hukic M, Valjevac A, Tulumovic D,
Numanovic F and Heyman P: Pathogenicity and virulence of the
present hantaviruses in Bosnia and Herzegovina: The impact on renal
function. Eur J Clin Microbiol Infect Dis. 30:381–385.
2011.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Clement J, Maes P, Lagrou K, Van Ranst M
and Lameire N: A unifying hypothesis and a single name for a
complex globally emerging infection: Hantavirus disease. Eur J Clin
Microbiol Infect Dis. 31:1–5. 2012.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Mäkelä S, Kokkonen L, Ala-Houhala I,
Groundstroem K, Harmoinen A, Huhtala H, Hurme M, Paakkala A, Porsti
I, Virtanen V, et al: More than half of the patients with acute
Puumala hantavirus infection have abnormal cardiac findings. Scand
J Infect Dis. 41:57–62. 2009.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Mustonen J, Brummer-Korvenkontio M, Hedman
K, Pasternack A, Pietilä K and Vaheri A: Nephropathia epidemica in
Finland: A retrospective study of 126 cases. Scand J Infect Dis.
26:7–13. 1994.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Bergmann F, Krone B, Bleich S, Prange H
and Paulus W: Encephalitis due to a hantavirus infection. J Infect.
45:58–59. 2002.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Ahlm C, Lindén C, Linderholm M, Alexeyev
OA, Billhedent J, Elgh F, Fagerlund M, Zetterlund B and Settergren
B: Central nervous system and ophthalmic involvement in
nephropathia epidemica (European type of haemorrhagic fever with
renal syndrome). J Infect. 36:149–155. 1998.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Hautala T, Mähönen SM, Sironen T, Hautala
N, Pääkkö E, Karttunen A, Salmela PI, Ilonen J, Vainio O, Glumoff
V, et al: Central nervous system-related symptoms and findings are
common in acute Puumala hantavirus infection. Ann Med. 42:344–351.
2010.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Launes J and Hautanen A: Nephropathia
epidemica encephalitis. Acta Neurol Scand. 78:234–235.
1988.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Alexeyev OA and Morozov VG: Neurological
manifestations of hemorrhagic fever with renal syndrome caused by
Puumala virus: Review of 811 cases. Clin Infect Dis. 20:255–258.
1995.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Cerar D, Avsic-Zuapanc T, Jereb M and
Strle F: Case report: Severe neurological manifestation of Dobrava
hantavirus infection. J Med Virol. 79:1841–1843. 2007.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Settergren B, Boman J, Linderholm M,
Wiström J, Hägg E and Arvidsson PA: A case of nephropathia
epidemica associated with panhypopituitarism and nephrotic
syndrome. Nephron. 61:234–235. 1992.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Kilit TP, Kilit C and Erarslan S: A rare
cause of acute pancreatitis: Hantavirus infection. Acta
Gastroenterol Belg. 80:59–61. 2017.PubMed/NCBI
|
|
66
|
Hoier S, Aberle SW, Langner C, Schnedl W,
Högenauer C, Reisinger EC, Krejs GJ and Krause R: Puumala virus RNA
in patient with multiorgan failure. Emerg Infect Dis. 12:356–357.
2006.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Miettinen MH, Makela SM, Ala-Houhala IO,
Huhtala HS, Koobi T, Vaheri AI, Pasternack AI, Porsti IH and
Mustonen JT: Tubular proteinuria and glomerular filtration 6 years
after Puumala hantavirus-induced acute interstitial nephritis.
Nephron Clin Pract. 112:c115–c120. 2009.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Miettinen M, Mäkelä S, Haapala M,
Helanterä A, Helin H, Vänttinen T and Mustonen J:
Glomerulonephritis emerging shortly after Puumala hantavirus
infection: A report of 7 patients. Clin Nephrol. 75:550–556.
2011.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Tulumovic D, Imamovic G, Mesic E, Hukic M,
Tulumovic A, Imamovic A and Zerem E: Comparison of the effects of
Puumala and Dobrava viruses on early and long-term renal outcomes
in patients with haemorrhagic fever with renal syndrome. Nephrology
(Carlton). 15:340–343. 2010.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Mäkelä S, Jaatinen P, Miettinen M, Salmi
J, Ala-Houhala I, Huhtala H, Hurme M, Pörsti I, Vaheri A and
Mustonen J: Hormonal deficiencies during and after Puumala
hantavirus infection. Eur J Clin Microbiol Infect Dis. 29:705–713.
2010.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Pekic S, Cvijovic G, Stojanovic M,
Kendereski A, Micic D and Popovic V: Hypopituitarism as a late
complication of hemorrhagic fever. Endocrine. 26:79–82.
2005.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Stojanovic M, Pekic S, Cvijovic G, Miljic
D, Doknic M, Nikolic-Djurovic M, Micic D, Hrvacevic R, Nesic V and
Popovic V: High risk of hypopituitarism in patients who recovered
from hemorrhagic fever with renal syndrome. J Clin Endocrinol
Metab. 93:2722–2728. 2008.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Miettinen MH, Mäkelä SM, Ala-Houhala IO,
Huhtala HS, Kööbi T, Vaheri AI, Pasternack AI, Pörsti IH and
Mustonen JT: Ten-year prognosis of Puumala hantavirus-induced acute
interstitial nephritis. Kidney Int. 69:2043–2048. 2006.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Plyusnin A, Hörling J, Kanerva M, Mustonen
J, Cheng Y, Partanen J, Vapalahti O, Kukkonen SK, Niemimaa J,
Henttonen H, et al: Puumala hantavirus genome in patients with
nephropathia epidemica: Correlation of PCR positivity with HLA
haplotype and link to viral sequences in local rodents. J Clin
Microbiol. 35:1090–1096. 1997.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Evander M, Eriksson I, Pettersson L, Juto
P, Ahlm C, Olsson GE, Bucht G and Allard A: Puumala hantavirus
viremia diagnosed by real-time reverse transcriptase PCR using
samples from patients with hemorrhagic fever and renal syndrome. J
Clin Microbiol. 45:2491–2497. 2007.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Saksida A, Duh D, Korva M and Avsic-Zupanc
T: Dobrava virus RNA load in patients who have hemorrhagic fever
with renal syndrome. J Infect Dis. 197:681–685. 2008.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Xiao R, Yang S, Koster F, Ye C, Stidley C
and Hjelle B: Sin Nombre viral RNA load in patients with hantavirus
cardiopulmonary syndrome. J Infect Dis. 194:1403–1409.
2006.PubMed/NCBI View
Article : Google Scholar
|
|
78
|
Laslo CL, Bacalbasa N, Ana MAS, Carsote M,
Bungau S, Rus M, Bratu OG and Diaconu CC: New oral
anticoagulants-possible extension to other indications (Review).
Exp Ther Med. 20:2401–2405. 2020.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Outinen TK, Mäkelä S, Clement J, Paakkala
A, Pörsti I and Mustonen J: Community acquired severe acute kidney
injury caused by hantavirus-induced hemorrhagic fever with renal
syndrome has a favorable outcome. Nephron. 130:182–190.
2015.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Huggins JW: Prospects for treatment of
viral hemorrhagic fevers with ribavirin, a broad-spectrum antiviral
drug. Rev Infect Dis. 11 (Suppl 4):S750–S761. 1989.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Huggins JW, Hsiang CM, Cosgriff TM, Guang
MY, Smith JI, Wu ZO, LeDuc JW, Zheng ZM, Meegan JM, Wang QN, et al:
Prospective, double-blind, concurrent, placebo-controlled clinical
trial of intravenous ribavirin therapy of hemorrhagic fever with
renal syndrome. J Infect Dis. 164:1119–1127. 1991.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Rusnak JM, Byrne WR, Chung KN, Gibbs PH,
Kim TT, Boudreau EF, Cosgriff T, Pittman P, Kim KY, Erlichman MS,
et al: Experience with intravenous ribavirin in the treatment of
hemorrhagic fever with renal syndrome in Korea. Antiviral Res.
81:68–76. 2009.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Antonen J, Leppänen I, Tenhunen J, Arvola
P, Mäkelä S, Vaheri A and Mustonen J: A severe case of Puumala
hantavirus infection successfully treated with bradykinin receptor
antagonist icatibant. Scand J Infect Dis. 45:494–496.
2013.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Sironen T, Sane J, Lokki ML, Meri S,
Andersson LC, Hautala T, Kauma H, Vuorinen S, Rasmuson J, Evander
M, et al: Fatal Puumala hantavirus disease: Involvement of
complement activation and vascular leakage in the pathobiology.
Open Forum Infect Dis. 4(ofx229)2017.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Tapia M, Mansilla C, Jose TM and Vera M:
Hantavirus pulmonary syndrome, clinical experience in diagnosis and
treatment. Hospital Coyhaique-Chile. Rev Chil Infect. 17:258–269.
2000.
|
|
86
|
Vial PA, Valdivieso F, Ferres M, Riquelme
R, Rioseco ML, Calvo M, Castillo C, Díaz R, Scholz L, Cuiza A, et
al: High-dose intravenous methylprednisolone for hantavirus
cardiopulmonary syndrome in Chile: A double-blind, randomized
controlled clinical trial. Clin Infect Dis. 57:943–951.
2013.PubMed/NCBI View Article : Google Scholar
|