Introduction
Neural stem cells (NSCs) are self-renewing,
multipotent cells that are located in two vital neurogenic niches:
The subventricular zone (SVZ) and the subgranular zone of the adult
brain (1). Following ischaemic
insult, NSCs in the SVZ proliferate, divert from their
physiological migration path and move directly to the lesioned
brain area, where they differentiate, mature and functionally
integrate into the neural circuits (2). However, this neuroregeneration process
is typically inadequate and transient, leading to ineffective
neurological function recovery (3).
A number of studies have shown that promoting endogenous stem cell
migration could improve the neurological recovery of rats with MCAO
(4,5). Furthermore, increased endogenous stem
cell migration could be evidenced by the presence of recently
divided (5-bromo-20-deoxyuridine (BrdU)-labeled) and immature
neuronal (doublecortin (DCX)-positive) cells in the injured area in
cerebral ischaemic rats (4,5). In addition, another study has reported
that specific ablation of DCX-expressing cells suppressed
neuromigration in adult mice and decreased neurological recovery in
mouse stroke models (6). Therefore,
these previous results suggest that the migration of NSCs is
critical to stroke recovery.
Repetitive transcranial magnetic stimulation (rTMS)
is a non-invasive technique that delivers magnetic pulses through
the skull to induce electrical or electromagnetic currents in
selective cortical tissues, leading to changes in cortical
excitability (7). It has been
widely applied for the functional treatment of patients with stroke
(8,9), though the mechanisms underlying its
beneficial effects remain poorly elucidated. DCX is a
microtubule-associated protein that is expressed almost exclusively
in cytoplasm of the newly formed neural cells and in migrating
neuroblasts in the adult brain (10,11).
Using an intracerebral haemorrhage mouse model, a previous study
(12) demonstrated that rTMS
significantly increased the number of DCX-positive cells in the
injured basal ganglia, suggesting its influence on the migration of
endogenous stem cells. Additionally, electromagnetic field
stimulation has been shown to increase the migration of NSCs from
the SVZ into demyelinated lesions in rats with multiple sclerosis
(13). However, further studies are
required to explore whether rTMS can promote the migration of NSCs
in vivo in the SVZ and to explore the mechanism underlying
the response following cerebral infarction in rats.
Stromal cell-derived factor 1α (SDF-1α) and its
affinity receptor CXC chemokine receptor 4 (CXCR4) are highly
expressed in the central nervous system during the developmental
process (14). Previous studies
revealed that mice deficient in either the expression of SDF-1α or
CXCR4 receptors exhibited ectopic navigation of neuronal precursor
cells, resulting in the abnormal development of the cortex in the
embryonic brain (15-18).
During the generation of the hippocampal dentate gyrus (DG), a
primary dentate neuroepithelium gives rise to a proliferative cell
population that streams along the ventral surface of developing
hippocampus to take up a position in the hilus of the DG (14,16).
Without functional CXCR4, the number of proliferating cells in the
migratory stream is reduced. Therefore, cells fail to reach the
forming DG in sufficient numbers, resulting in the failure of DG
morphogenesis (17). Normally,
during the development of cerebellum, granule cell progenitors are
generated in the rhombic lip and then migrate rostromedially along
the surface of the cerebellar anlage, forming the external granule
cell layer (18). In SDF-1α- or
CXCR4-deficient brains of mouse embryos, cells appear to migrate
incorrectly from the proliferative external granule cell layer,
resulting in the ectopic positioning of granule cells in the
Purkinje cell layer, leading to a disorganized cerebellar (19). Following cerebral ischaemia, SDF-1α,
which is secreted by astrocytes and endothelial cells surrounding
the infarct area, attracts stem cells. CXCR4 is mainly expressed on
neural progenitor cells and stroke-induced neuroblasts (20). CXCR4-positive cells migrate along
the chemokine gradient of SDF-1α, scaffolded by vessels, corpus
callosum and glial cells and are involved in the regulation of
axonal outgrowth and patterning, synaptic function and remodelling
(21). Some rehabilitation measures
have been demonstrated to increase neurological function and
augment the migration of endogenous NSCs via the SDF-1α/CXCR4 axis
in rats stroke models (4,5). The protein expression levels of CXCR4
are significantly increased in bone marrow mesenchymal stem cells,
which enhance cell migration in vitro after external
electric current stimulation (22,23).
In addition, electrical stimulation increases SDF-1α concentrations
and enhances the migration of transplanted bone marrow stromal
cells to the infarcted areas of rats with stroke (24). However, the effects of rTMS on the
expression levels of SDF-1α and CXCR4 in rats after stroke remain
unknown.
Cerebral infraction results in neuronal death and
therefore, protective approaches against neuronal loss in the
penumbra are sorted after. The SDF-1α/CXCR4 axis has conflicting
reported functions in neuronal survival, depending on the
conditions. An in vitro study previously demonstrated that
SDF-1α can promote the survival of cortical neurones (25). By contrast, the CXCR4 antagonist
AMD3100 has been shown to attenuate inflammatory responses and
reduce brain damage in mice following acute ischaemic stroke
(26). In addition, another CXCR4
antagonist, CX549, effectively suppresses the inflammatory response
and increases neuronal survival, contributing to post-stroke
behavioural recovery (27).
Previous studies indicated that rTMS not only promotes cell
proliferation and inhibits apoptosis in oxygen- and
glucose-deprived neuronal cells (28), but also suppresses neuronal
degeneration in the peri-infarct region in rats following stroke
(29). However, the role of
SDF-1α/CXCR4 signalling in the rTMS-mediated protection of neurones
in cerebral ischaemic rats remains unknown.
The present study explored whether rTMS could rescue
middle cerebral artery occlusion (MCAO)-induced functional
impairment by producing a more favourable microenvironment for NSC
migration and neuronal survival. Moreover, the role of the
SDF-1α/CXCR4 axis in this process was explored.
Materials and methods
Animals and groups
A total of 135 adult male Sprague-Dawley rats (age,
8 weeks; Jingda Bioengineering Co., Ltd., Hunan, China) weighing
210-230 g, were used. The rats were housed in controlled conditions
(environment temperature of 25±2˚С, ambient humidity of 50±10%,
12-h light/dark cycle) and were provided with ad libitum
access to food and water. Randomization was enforced by assigning a
number to each rat, and a random number generator was implemented
to divide animals into the experimental groups. Rats were divided
into sham (S), MCAO (M), rTMS + MCAO (R), rTMS + MCAO + AMD3100
(RA) and MCAO + AMD3100 (MA) groups. These five groups were
subdivided further into subsets at two time points, namely 7 and 14
days after MCAO. The present animal study was approved by The
Animal Experimentation Ethics Committee of Tongji Hospital
(approval. no. 2017609; Wuhan, China) and all treatments conformed
to the National Institutes of Health Guide for the Care and Use of
Laboratory Animals (Publication no. 80-23; revised 1996). The rats
were sacrificed at 7 days (n=11 for each subgroup) or 14 days (n=16
for each subgroup) after MCAO depending on the experimental
schedule. In total, six rats in each subgroup were used for
immunofluorescence staining and five were used for western blot
analysis for both 7 and 14 days. Additionally, Nissl staining was
performed in rats (n=5) in the 14-day subgroups.
Focal ischaemic stroke model of
rat
A stroke model was established by intraluminally
occluding the right middle cerebral artery in rats according to the
procedure used by Longa et al (30). After being anaesthetized with 5%
isoflurane inhalation for induction and 2.5% for maintenance (RWD
Life Science Co., Ltd.), a nylon suture thread was inserted into
the right internal carotid artery to block blood supply for 90 min
as described in a previous study (31). The sham group was subjected to
external carotid artery ligation only. During the surgery and
recovery period, the rat body temperature was maintained at
37±0.5˚C by a heat lamp. The neurological deficit score (0, no
deficit; 1, failure to stretch the left forepaw fully; 2, circling
to the left; 3, falling to the left; and 4, no spontaneous gaits
with abnormal consciousness) was assessed 4 h after the operation
and rats were selected based on a score of 2-3(30).
Modified neurological severity score
(mNSS)
The mNSS (32)
evaluation was performed at 1, 7 and 14 days after surgery, where
the parameters assessed included movement, sense of touch, reflexes
and balance indications. The measures were rated on a scale of 0 to
18(32), with normal marked as 0
and the most severe deficits marked as 18. Behavioural tests were
performed by blinded observers (FG and XHH).
Grip strength metric
On days 1, 7 and 14 post-ischaemia, the forepaw
strength of rats was measured using a grip strength meter (Ugo
Basile SRL). The rats were trained to grasp the grid with their
forelimbs whilst being tugged backwards from the base of tail.
After being trained on three successive daily trials before the
surgery, the rats were allowed to grasp the meter grid with their
forepaws and then pulled carefully from the tail until they
released the apparatus. The test was repeated five times per rat
with 1-h intervals in between. The average force exerted by the
bilateral forepaws was determined from the recorded values. The
mean of all five readings was calculated and used for statistical
analyses. The grip strength test process and results analysis were
performed as previously described (33).
rTMS treatment
At 1 day after ischaemic stroke, conscious rats
received rTMS administration by a magnetic stimulator (YRD-CCI,
Wuhan Yiruide Medical Equipment New Technology Co., Ltd.) with a
6-cm diameter figure-eight coil. The stimulation site was placed
over the ipsilateral primary motor cortex. Richter et al
(34) previously observed that 45˚
in relation to the standard lateral orientation is the optimal
direction for stimulation. Therefore, this stimulus orientation was
used. After the rats were fixed on an instrument in a standing
position without resistance, they received rTMS once a day for 7 or
14 consecutive days with the following parameters: 10-Hz frequency;
3-sec duration; 50-sec training interval; 10 successive training
episodes; 300 pulses; and 120% rest motor threshold (RMT)
intensity. The RMT was recorded from the gastrocnemius muscle of
the left hind limb using a TMS (Wuhan Yiruide Medical Equipment New
Technology Co., Ltd.) as previously described (35). The RMT was defined as the lowest
intensity of the TMS able to elicit a motor-evoked potential with
amplitudes ≥15 µv at least 5/10 times (36). The rTMS parameters were set based on
a previous study according to their efficacy and feasibility
(37).
Bromodeoxyuridine and AMD3100
administration
To identify proliferating cells,
5-bromo-20-deoxyuridine [BrdU; 50 mg/kg; intraperitoneal (i.p.)
injection; cat. no. B5002; Sigma-Aldrich; Merck KGaA] was
administered. For the 7-day subgroups, rats were injected with BrdU
every 4 h continuously three times after the last rTMS treatment (7
days after surgery) and then euthanized within 4 h after the last
BrdU injection (38). For the
14-day subgroups, rats were administered with BrdU once daily for 7
consecutive days from 24 h post-surgery (39). The CXCR4 receptor antagonist AMD3100
(cat. no. HY-10046; MedChemExpress) was also used in the MA and RA
groups. Rats in the MA and RA groups received an AMD3100 (medium
saline; 2 mg/kg; i.p.) injection every other day from 2 days after
MCAO until euthanasia according to previous reports (4).
Tissue preparation
The specimens were embedded in Optimum Cutting
Temperature (cat. no. G6059-110ML; Wuhan Servicebio Technology Co.,
Ltd.) compound for immunofluorescence analysis, whilst brain
samples were paraffin-embedded for Nissl staining. At 7 or 14 days
after MCAO, six rats were deeply anaesthetized with 5% isoflurane
inhalation induction and 2.5% maintenance (RWD Life Science Co.,
Ltd.) for 10 min. During this time the rats became unconscious and
were insensitive to tail pinch. They were then perfused
transcardially with saline and 4% paraformaldehyde at 4˚C for 30
min. The brain tissues were then removed and kept in the same
fixative overnight at 4˚C and then incubated in 20 and 30% sucrose
consecutively at 4˚C for 3 days dehydration. Subsequently, the
brains were embedded in Optimum Cutting Temperature and then cut
into 30-µm thick free-floating coronal sections using a cryostat
(CM1900; Leica Microsystems GmbH) for immunofluorescence.
At 14 days after surgery, brain samples (n=5) were
collected by transcardial perfusion, fixed in paraformaldehyde,
dehydrated in sucrose as aforementioned, paraffin-embedded and cut
into 4-µm thick slices in the coronal plane (Leica 2035; Leica
Microsystems GmbH) for Nissl staining. To be more representative,
as referred by a previous report, every sixth slice (40) containing the cerebral infarction was
collected, including the cortex and striatum (0.3-1.2 mm behind the
bregma).
Immunofluorescence staining
To prepare the slices for BrdU labelling, the
free-floating brain sections were washed in PBS three times and
denatured in 2 N HCl for 25 min at 37˚C and neutralized with 0.1 M
borate solution for 15 min. The free-floating sections were blocked
in buffer (10% normal donkey serum and 0.3% Triton X-100 in PBS; pH
7.5; cat. no. ANT051; Wuhan Antejie Biotechnology Co., Ltd. and
cat. no. WGT8200; Wuhan Servicebio Technology Co., Ltd.) for 2 h at
room temperature and then incubated with a mixture of primary
antibodies in 5% donkey serum and PBS overnight at 4˚C. Primary
antibodies included rat anti-BrdU (1:100; cat. no. ab6326; Abcam)
and goat anti-DCX (1:100; cat. no. sc-8066; Santa Cruz
Biotechnology, Inc.). The following day, the primary antibodies
were washed and replaced with the secondary antibodies (1:400 for
both; Thermo Fisher Scientific, Inc.), TRITC 593-conjugated donkey
anti-rat (to conjugate BrdU; cat. no. A18744) and Alexa Fluor
488-labelled donkey anti-goat (to link DCX; cat. no. A-11055;
Thermo Fisher Scientific, Inc.) for 2 h at room temperature before
rinsing with PBS. Slides were mounted with a DAPI-containing
anti-quenching agent (cat. no. AR1177; Wuhan Boster Biological
Technology, Ltd.) and sealed with a coverslip. Images were captured
with a confocal laser-scanning microscope (magnification x200;
Olympus Corporation). Subsequently, two randomly selected regions
in the ipsilateral SVZ were observed per section, where the number
of BrdU/DCX co-labelled positive cells were identified in the
ipsilateral brain area from the SVZ to the ischaemic cortex under a
x20 objective in a blinded manner using the ImageJ version 1.50
(National Institutes of Health) software and the average values per
slide were recorded. The mean value of three sections in each rat
was processed. The results were expressed as the number of labelled
cells.
Western blot analysis
Peri-infarct cortical tissue samples from subset
groups (n=5) at 7 and 14 days were collected for western blot
analysis. Total proteins were extracted with a Teflon-glass
homogenizer in ice-cold homogenization medium containing 1 mM PMSF
(Beyotime Institute of Biotechnology) in chilled RIPA buffer (cat.
no. P0013B; Beyotime Institute of Biotechnology). The bicinchoninic
acid (BCA) protein assay kit (Beyotime Institute of Biotechnology)
was employed to estimate the protein concentrations, as detailed in
a previous study (37). A total of
40 µg protein per lane was separated using 10% SDS-PAGE gels and
then transferred onto PVDF membranes. Subsequently, membranes were
blocked with TBST (0.1% Tween-20 in TBS) containing 5% non-fat milk
powder for 1 h at room temperature and then incubated overnight at
4˚C with the following primary antibodies: Rabbit anti-SDF-1α
(1:300; cat. no. bs-4938R; Bioworld Technology, Inc.) and rabbit
anti-CXCR4 antibody (1:300; cat. no. ab181020; Abcam). Next,
membranes were washed in 0.1% TBST three times, incubated with
associated horseradish peroxidase linked secondary antibodies
(1:5,000; cat. no. BA1054; Wuhan Boster Biological Technology,
Ltd.) for 1 h at room temperature and washed in 0.1% TBST three
times. Blots were visualized using the ECL western blot detection
kit (Thermo Fisher Scientific, Inc.). The bands were examined using
Gel-Pro Analyzer version 4.0 software (Media Cybernetics, Inc.).
The test was repeated in triplicate.
Nissl staining
Nissl staining was performed according to the
standard procedure (41). Briefly,
after being dewaxed in the xylene and rehydrated in the descending
concentration gradient ethanol, the sections were stained with 0.1%
cresyl violet acetate (Sigma-Aldrich; Merck KGaA) for 40 min at
60˚C, before they were rinsed, dehydrated in the ascending
concentration gradient ethanol and cleared in the xylene. Next,
sections were cover slipped and identified under a light microscope
(magnification x400; DM2500; Leica Microsystems GmbH) and the area
of the penumbra was outlined according to the staining colour and
arrangement difference under the x10 objective of the microscope.
Images were captured and cell counting was performed with a x40
objective. A total of five randomly selected fields for each
coverslip were examined, three sections in each rat were taken
before the average values were calculated. Surviving neurones were
calculated using Image-Pro Plus version 6.0 (Media Cybernetics,
Inc.) software in a blinded manner with reference to the method of
Ji et al (42). The Nissl
bodies (blue) staining in neural cells were clearly visible and
distinct. Cells with Nissl staining in the cytoplasm and prominent
nucleoli were identified as surviving neurones, whilst cells with
shrunken shape, cavitation around the nucleus or loss of Nissl
substance were considered damaged (43). Only whole neurones with visible
nuclei were counted. The results are expressed as the number of
viable neurones (Nissl-positive cells) in the penumbra.
Statistical analysis
Values are shown as the means ± standard error of
the mean. All analyses were performed using SPSS version 19 (IBM
Corp.). GraphPad Prism version 6.0 software (GraphPad Software,
Inc.) was used to generate graphics. For multiple comparisons, data
were subjected to a one-way analysis of variance (ANOVA) followed
by Bonferroni post hoc comparison. P<0.05 was considered to
indicate a statistically significant difference.
Results
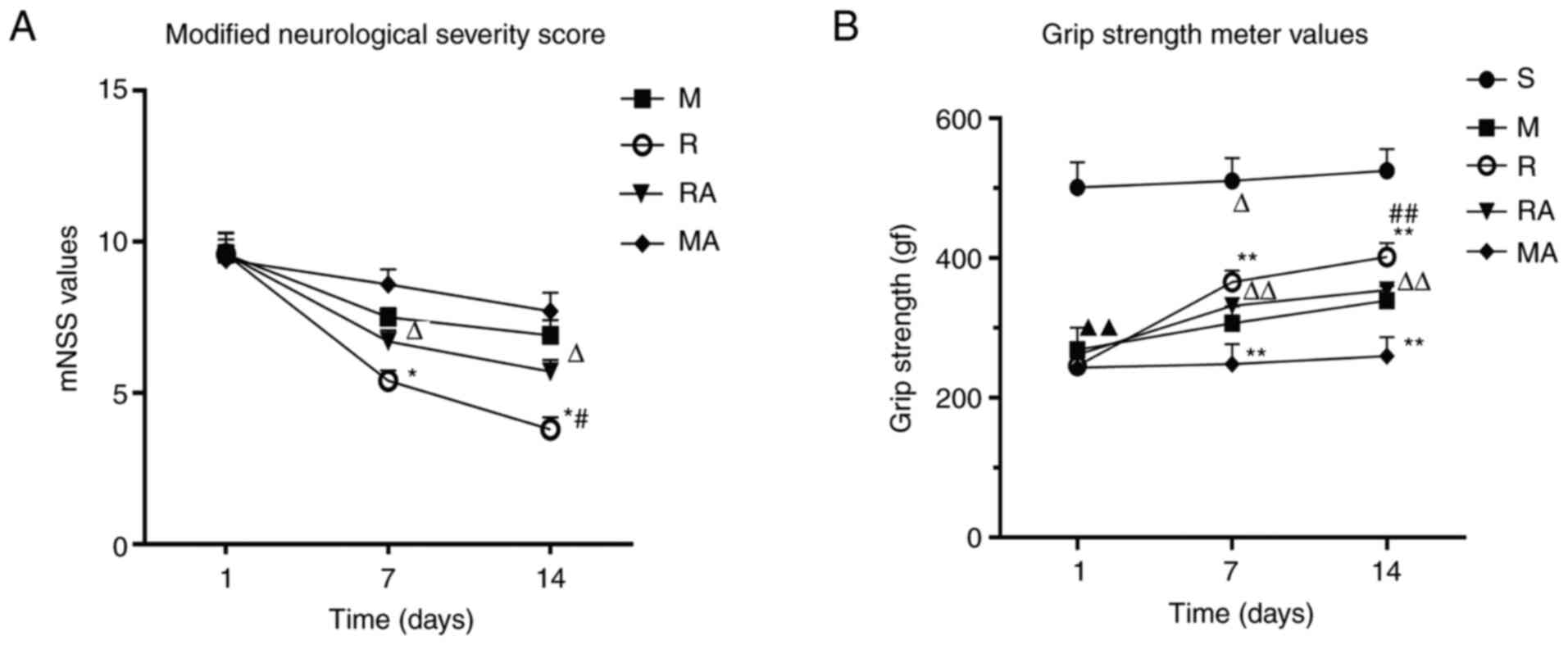
rTMS improves behavioural outcomes of
rats with ischaemic stroke
To assess the neurological function of rats after
MCAO, mNSS scoring was used. The mNSS value of the sham-operated
rats was 0, where significant differences were not observed in the
average scores among the ischaemic rats 1 day after surgery. As
shown in Fig. 1A, compared with
those in the M and MA groups, rats in the R and RA groups
demonstrated significant improvement in the mNSS evaluation at 7
and 14 days after MCAO (P<0.05). Rats in the R group exhibited
no significant decline in the mNSS scores compared with those in
the RA group at 7 days after MCAO. However, a significant increase
was observed in the RA group compared with that in the R group at
14 days (P<0.05). These results suggest that rTMS alleviated the
motor deficiencies in MCAO rats, but AMD3100 abrogated the
rTMS-induced response at 14 days.
The purpose of the grip strength test was to
evaluate muscle strength and neuromuscular integration
corresponding to the forepaw grasping reflex (44). As shown in Fig. 1B, compared with that in
sham-operated rats, the grip strength of the rats in the M group
significantly decreased (P<0.01). Significant differences were
not observed in the grip strength test values among the rats 1 day
after MCAO. At 7 and 14 days after surgery, the strength of the
rats in the R, M and RA groups was increased significantly compared
with that in the M, MA and MA groups, respectively (P<0.01).
Significant differences were not observed between that in the R
group and the RA groups at 7 days after MCAO, whilst a significant
increase in forepaw power occurred in the R group compared with
that in the RA group at 14 days post-ischaemia (P<0.01). These
results revealed that rTMS increased the muscle strength and
neuromuscular integration of MCAO rats. Furthermore, AMD3100
reversed the beneficial effects of rTMS on the forepaw strength of
rats at 14 days. In addition, compared with the rats in MCAO group,
AMD3100 inhibited the forelimb power recovery of MCAO rats at 7
days.
rTMS enhances the migration of NSCs in
rats with ischaemic stroke
To observe migrating SVZ-derived NSCs in the
peri-infarction zone (Fig. 2A and
B), a BrdU/DCX dual-labelling
immunofluorescence test was conducted at different time points of 7
and 14 days. BrdU is a specific maker for cell proliferation,
whilst DCX is expressed in newborns and migrating neuroblasts in
the adult brain (4,5). Therefore, NSC migration can be
quantified by counting the number of BrdU/DCX-immunostained cells
(45). The number of BrdU-positive
cells in the SVZ increased at 7 and 14 days after MCAO compared
with that in the Sham rats (data not shown). Some of the
BrdU-positive cells co-expressed DCX, which represented the NSCs
diverting from the SVZ. A small number of DCX+ cells
were found in the left lateral SVZ of rats in the M group or in the
bilateral SVZ of Sham rats. Thereafter, DCX+ cells were
numerically increased and distributed in the callosum and striatum
of the ipsilateral hemisphere at 7 and 14 days after surgery. At 14
days, the BrdU+/DCX+ cells with elongated
dendrites, formed clusters at the SVZ, arranged in a chain-like
manner along the ventricle lateral wall, and migrated towards the
infarction site (Fig. 2C-2J). As
shown in Fig. 3F at 7 days
post-ischaemia (Figs. 3F and
S1), significantly more
BrdU+/DCX+ cells were observed in the
ipsilateral SVZ of the M group compared with those in the S group
(P<0.05). In addition, rTMS treatment significantly augmented
the number of BrdU+/DCX+ cells in the R group
compared with that in rats in the M group (P<0.05). The number
of dual-positive cells in the RA group was significantly increased
compared with those in the MA group (P<0.05).
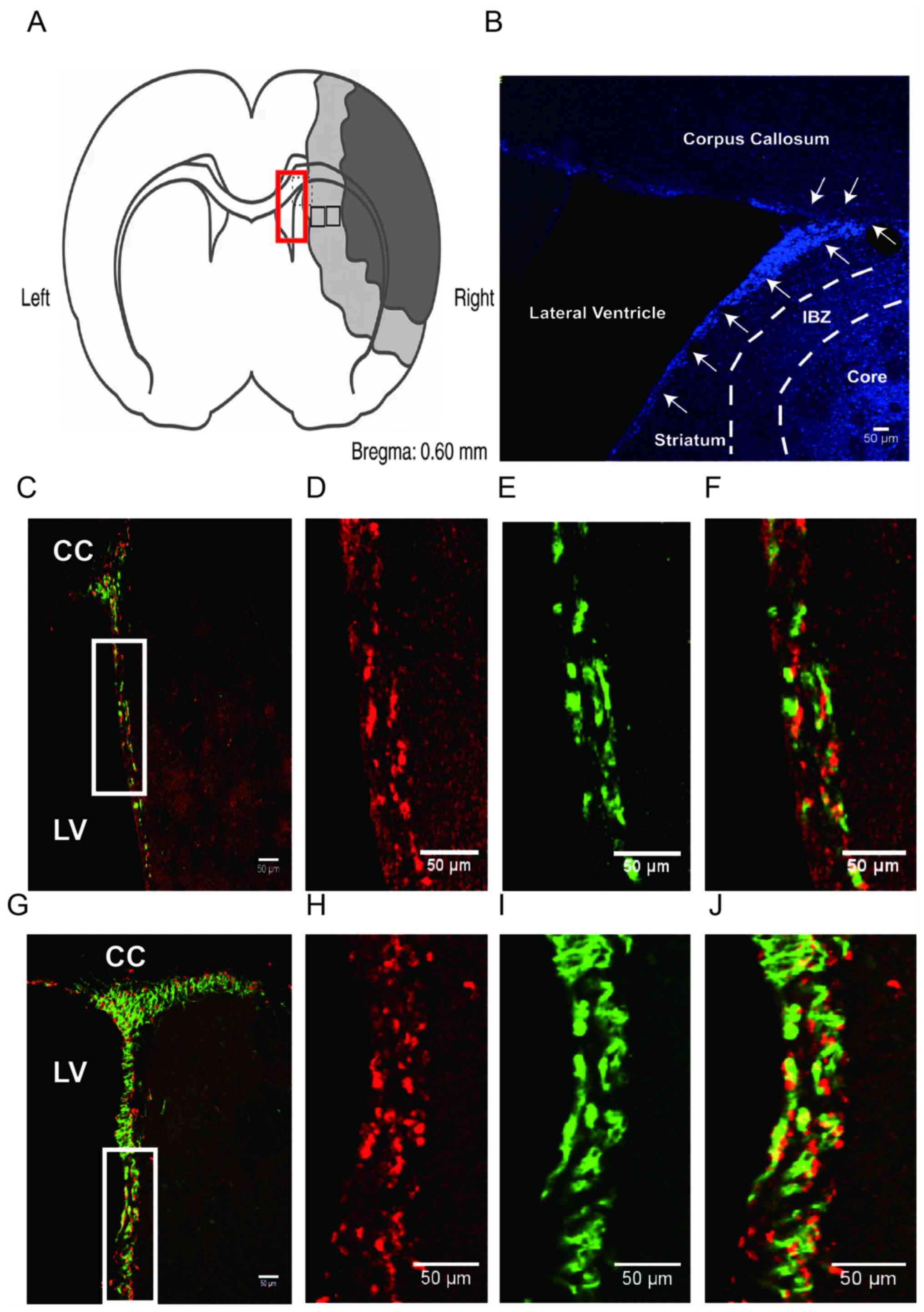 | Figure 2Schematic view of the region of
interest and representative confocal images of immunofluorescence
staining at 14 days post-surgery. (A) Coronal section
representation of an ischaemic rat brain (bregma 0.60 mm). Dark
section denotes the ischaemic core, the grey section indicates the
ischaemic penumbra and white sections indicate non-ischaemic area.
For image analysis, representative fields (rectangle in red colour)
in the SVZ and representative fields (rectangle in black colour) in
the penumbra were selected. (B) Representative images of
immunofluorescence labelling with DAPI (blue) staining in the M
group at 7 days post-surgery. The SVZ (white arrows) located
between the LV and the striatum. IBZ (penumbra) was defined as an
area surrounded by white dashed lines and ischemic core located
right side from the right broken line. Magnification x100. (C-J)
Representative ipsilateral SVZ zones were subjected to
immunofluorescence labelling with BrdU (red) and DCX (green)
staining. (C) Representative ipsilateral SVZ zone image from the M
group 14 days after surgery. The three columns on the right (D-F)
show higher magnification images of the white boxed areas in (C).
Images of (D) BrdU+, (E) DCX+, (F) BrdU/DCX dual-positive cells in
the M group at 14 days post-surgery. (G) Representative ipsilateral
SVZ zone image from the R group 14 days after surgery. The three
columns (H-J) show higher magnification images of the white boxed
areas in (G). Images of (H) BrdU+, (I) DCX+, (J) BrdU/DCX
dual-positive cells in the R group at 14 days post-surgery. Scale
bars, 50 µm. CC, corpus callosum; LV, lateral ventricle; SVZ,
subventricular zone; IBZ, ischemic boundary zone; Core, infarct
core; MCAO, middle cerebral artery occlusion model; rTMS,
repetitive transcranial magnetic stimulation; S, sham; M, MCAO; R,
rTMS + MCAO; RA, rTMS + MCAO + AMD3100; MA, MCAO + AMD3100; BrdU,
bromodeoxyuridine; DCX, doublecortin. |
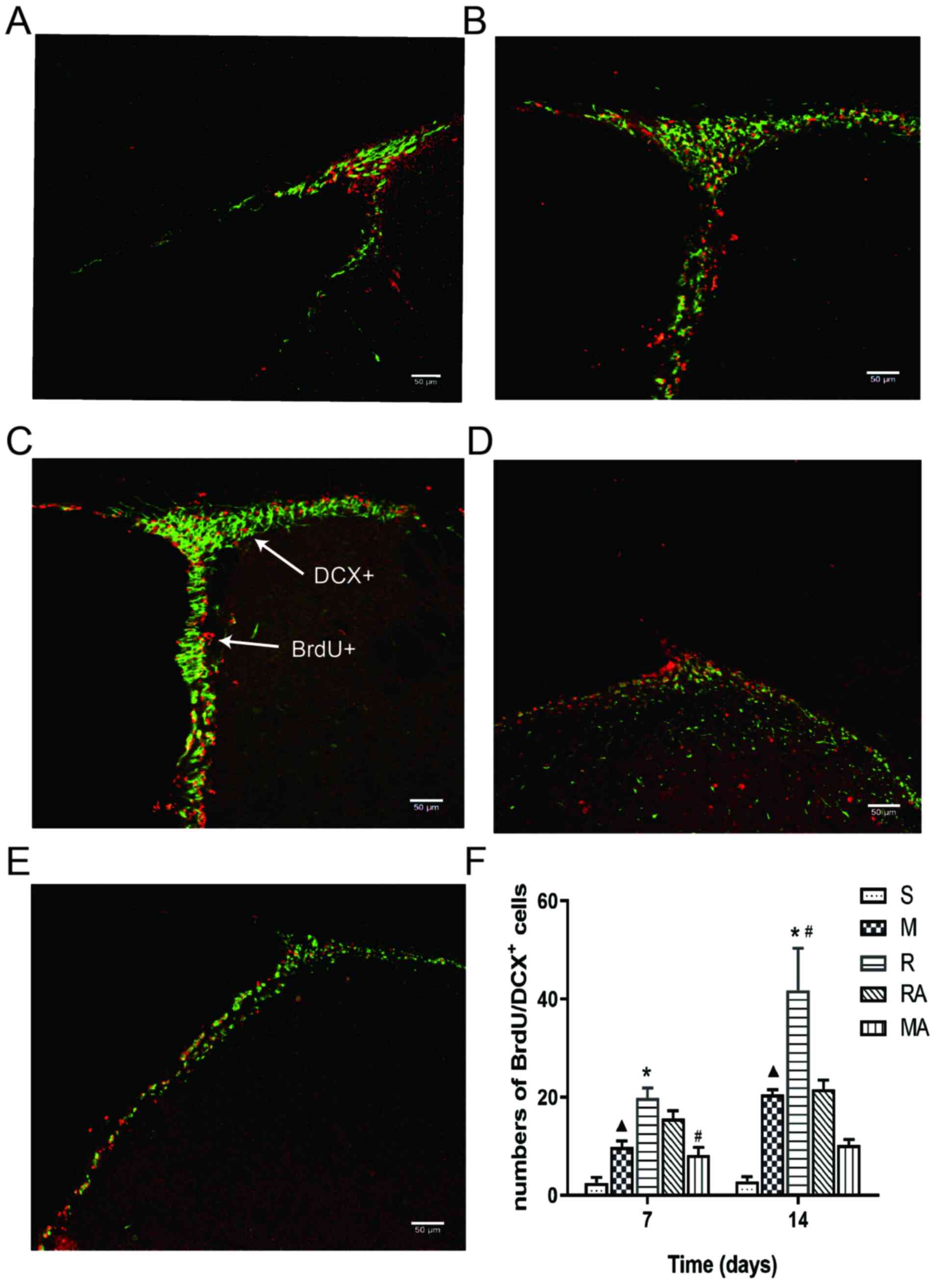 | Figure 3rTMS treatment promotes the migration
of neural stem cells in the ipsilateral SVZ at 7 and 14 days after
MCAO. Representative confocal images of immunofluorescence
staining. Staining in the (A) S group, (B) M group, (C) R group,
(D) RA group and (E) MA group at 14 days after ischaemia onset.
White arrows indicate examples of colocalized cells. Red represents
BrdU and green represents DCX. Magnification, x200; scale bar, 50
µm. (F) Quantification the number of BrdU/DCX dual-positive cells
per section in each group. n=6 rats per group.
▲P<0.05 vs. S; *P<0.05 vs. M;
#P<0.05 vs. RA. MCAO, middle cerebral artery
occlusion model; rTMS, repetitive transcranial magnetic
stimulation; S, sham; M, MCAO; R, rTMS + MCAO; RA, rTMS + MCAO +
AMD3100; MA, MCAO + AMD3100; BrdU, bromodeoxyuridine; DCX,
doublecortin. |
At 14 days post-ischaemia, the numbers of
BrdU+/DCX+ cells increased significantly in
the M group compared with those in the S group (P<0.05), the
same trend was also observed in the R group compared with that in
the M group (P<0.01), in addition to in the R group compared
with that in the RA group (P<0.05). However, significant
differences were not observed between the numbers in the RA and MA
groups (Fig. 3F). These results
suggest that 10 Hz rTMS enhanced the ischaemia-induced increases in
the number of BrdU+/DCX+ co-labelled-cells.
Additionally, AMD3100 inhibited the beneficial effects of rTMS
against MCAO in rats at 14 days but not at 7 days.
rTMS increases the expression levels
of proteins in the SDF-1α/CXCR4 axis in the peri-infarction zone
after MCAO
To examine the effects of rTMS on the SDF-1α/CXCR4
signaling axis in MCAO rats, western blot analysis was used to
observe the protein expression levels of SDF-1α and CXCR4 at 7 and
14 days after MCAO (Fig. 4). The
expression levels of SDF-1α and CXCR4 in rats in the M group were
significantly upregulated compared with those in the sham group
(P<0.05; Fig. 4). Furthermore,
the R group displayed significantly increased SDF-1α and CXCR4
expression levels at both 7 and 14 days compared with those in the
M group (all P<0.05; Fig. 4A).
The expression levels of SDF-1α in the RA group was significantly
increased compared with those in the MA group at 7 and 14 days
(P<0.05; Fig. 4A). In addition,
the expression levels of SDF-1α in the MA group was significantly
decreased compared with that in the M group at 14 days (P<0.05;
Fig. 4A).
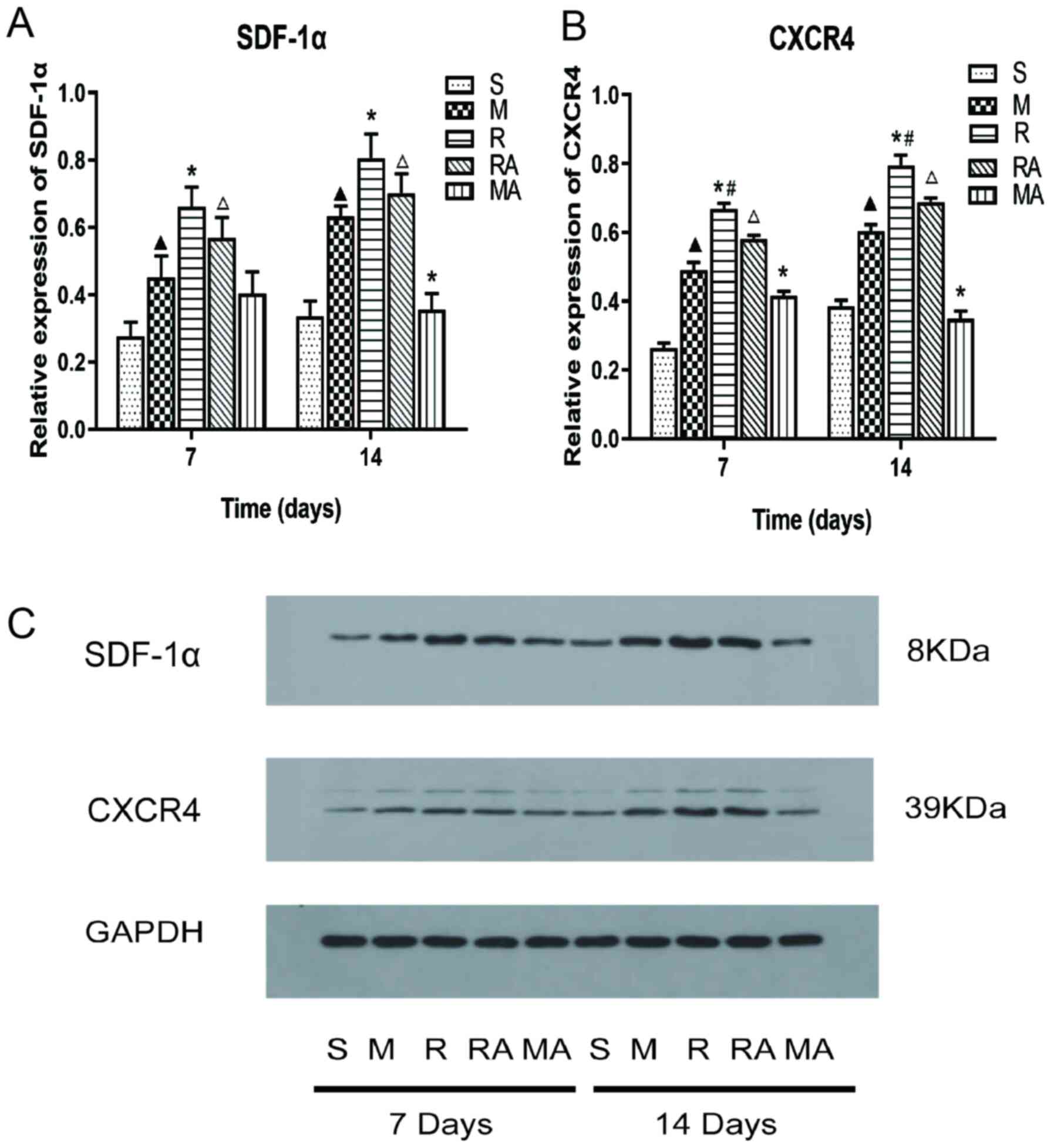 | Figure 4Effects of rTMS on SDF-1α and CXCR4
expression in the ipsilateral tissue at 7 and 14 days after
surgery. Protein expression levels of (A) SDF-1α and (B) CXCR4 in
the ischaemia samples as determined by western blot analysis. (C)
Representative western blotting images of SDF-1α, CXCR4 and GAPDH
on gel. n=5 rats per group. ▲P<0.05 vs. S group;
*P<0.05 vs. M group; #P<0.05 vs. RA
group; ΔP<0.05 vs. MA group. MCAO, middle cerebral
artery occlusion model; rTMS, repetitive transcranial magnetic
stimulation; S, sham; M, MCAO; R, rTMS + MCAO; RA, rTMS + MCAO +
AMD3100; MA, MCAO + AMD3100; SDF-1α, Stromal cell-derived factor
1α; CXCR4, CXC Chemokine receptor 4. |
The expression level of CXCR4 was significantly
increased in the R group compared with that in the RA group, whilst
the same trend was also observed for the M group compared with that
in the MA group, in addition to for that in the RA group compared
with that in the MA group at both timepoints (all P<0.05;
Fig. 4B). These results suggest
that the SDF-1α/CXCR4 axis was inhibited by AMD3100 and that rTMS
could upregulate the expression levels of SDF-1α and CXCR4 in rats
following MCAO.
rTMS increases the number of surviving
neurones in the ischaemic penumbra
Nissl staining was performed to assess neuronal
injury. Since a tendency to behavioural improvement was observed in
rats in the R group after 14 days compared with those in the same
group after 7 days (data not shown), rats in the R group were
selected for Nissl staining after 14 days (Figs. 2A and 5). As shown in Fig. 2A, the injured area (concluded
ischemic core and penumbra) mainly was focused on the cerebral
cortex and the striatum. Damaged neurones would exhibit the absence
of Nissl's bodies, smaller intercellular space and abnormal
staining. Neurones were found to be arranged randomly with
irregular shapes in the M group (Fig.
5B). The number of Nissl+ neurones per high power
field in rats in the M group was significantly decreased compared
with that in sham rats (P<0.05; Fig.
5F). By contrast, rTMS significantly alleviated MCAO-induced
neuronal loss, as shown by the significantly increased numbers of
Nissl+ neurones in the R group compared with those in
the M group (P<0.05; Fig. 5F).
Statistical analysis also demonstrated that the number of surviving
neurones in the MA group was significantly decreased compared with
that in the RA group (P<0.05; Fig.
5F). Additionally, significant differences were observed
between the R group and RA group and between the M group and MA
group (both P<0.05; Fig. 5F).
The number of Nissl+ neurons in the R and M group was
significantly higher than that in the RA and MA groups,
respectively. These results suggest that rTMS can rescue
MCAO-induced neuronal damage in the penumbra, whereas this positive
response was inhibited by AMD3100.
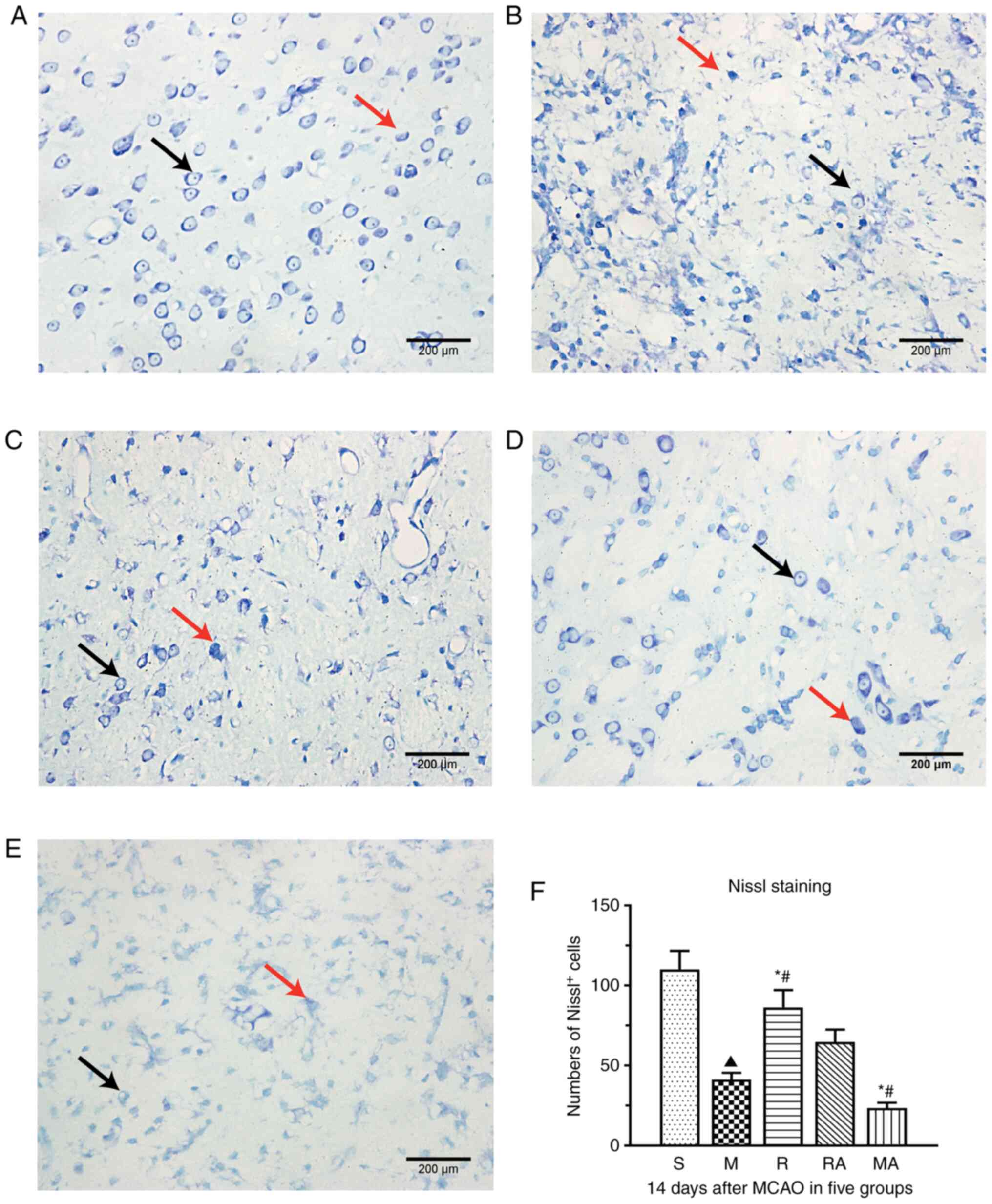 | Figure 5rTMS treatment protects neurones
against ischaemic injury in the penumbra at 14 days after surgery.
Representative images of Nissl staining in the (A) S, (B) M, (C) R,
(D) RA and (E) MA groups (magnification, x400; scale bar, 200 µm).
(F) Quantified analysis of Nissl-positive neurones per section in
the five groups. Black arrows represent examples of surviving
neurones (Nissl-positive cells). Red arrows indicate examples of
damaged neurones. n=5 rats per group. ▲P<0.05 vs. S;
*P<0.05 vs. M; #P<0.05 vs. RA. MCAO,
middle cerebral artery occlusion model; rTMS, repetitive
transcranial magnetic stimulation; S, sham; M, MCAO; R, rTMS +
MCAO; RA, rTMS + MCAO + AMD3100; MA, MCAO + AMD3100. |
Discussion
The present study demonstrated that high-frequency
rTMS facilitated functional recovery from ischaemic stroke in a rat
MCAO model, which was associated with enhanced migration of NSCs
and reduced neuronal loss in the peri-infarct area. Together with
these effects, the protein expression levels of SDF-1α and CXCR4
were significantly increased by 10 Hz rTMS treatment. However, the
rTMS-induced changes were significantly inhibited by AMD3100. To
the best of our knowledge, the present study demonstrated for the
first time that the restorative and neuroprotective effects of rTMS
in rats following ischaemic stroke may be dependent on the
activation of SDF-1α/CXCR4 signalling.
Activating endogenous NSCs to remodel the neural
tissue is considered to be an effective therapeutic strategy after
cerebral ischemic injury in rats (46). The infarct area was found to be
enlarged, where the neurological deficits were more severe, in mice
that were depleted of DCX- and BrdU-immunoreactive cells in the SVZ
post-ischaemia, suggesting that boosting NSC migration is a
potential target for stroke treatment (47). The present study used mNSS values
and grip strength to evaluate neurological function, muscular
strength and neuromuscular integration in rats following cerebral
ischaemia. The results indicated that 10 Hz rTMS significantly
restored motor dysfunction and further promoted the migration of
NSCs, as evidenced by the significantly increased number of
BrdU+/DCX+ cells in the ipsilateral SVZ
compared with that in the M group at 7 and 14 days post-ischaemia.
Similar results were found in a previous clinical (9) and preclinical (48) study, indicating that rTMS can
increase overall motor function following ischaemic stroke.
Exogenous electrical stimulation can confer positive effects on NSC
migration in vitro (49).
Additionally, a previous study also demonstrated that
electromagnetic field stimulation can promote the migration of
proliferative cells from the SVZ into demyelinated lesions in the
corpus callosum in a rat model of multiple sclerosis (13). Therefore, the improvement of motor
function observed with rTMS may be associated with the enhancement
of NSC migration in rats with ischaemic stroke.
Chemokines are important factors that regulate cell
migration (50). Using a rat model
of acute ischemic stroke, a previous study has revealed that SDF-1α
and CXCR4 expression levels were upregulated following stroke
(51). Overexpression of SDF-1α in
the boundary of the infarcted area in rats with ischemic stroke
resulted in increased NSCs recruitment in the SVZ (52). Furthermore, SDF-1α/CXCR4 stimulates
outward radial migration of neural precursors from adherent
neurospheres in vitro in a dose-dependent manner after
treatment with CXCL12 (SDF-1α) in the medium (53). When CXCR4 receptor-blocking
antibodies were applied, the migration of NSCs was also
significantly reduced (5).
Therefore, SDF-1α/CXCR4 signalling can potentially manipulate the
trafficking of endogenous NSCs. Results of the present study
demonstrated that rTMS enhanced the expression of SDF-1α and CXCR4,
which was accompanied by an increased number of
BrdU+/DCX+ cells at the ischaemic border, in
a manner that was susceptible to blockade by AMD3100. Similarly, a
previous study observed that electrical stimulation increased
SDF-1α concentrations and enhanced the migration of exogenous bone
marrow stromal cells to the infarction zone in a rat stroke model
(24). In the present study, the
AMD3100-induced reduction in SDF-1α and CXCR4 expression resulted
in decreased neurological recovery in the RA group at 7 and 14 days
after stroke. These results imply that the SDF-1α/CXCR4 axis can
contribute to rTMS-mediated recovery of motor dysfunction after
stroke in rats. These effects may be explained by the significant
reduction in rTMS-induced NSC migration caused by AMD3100 at 14
days post-surgery, thereby significantly attenuating functional
recovery. Moreover, the number of BrdU+/DCX+
cells was significantly increased in the R group compared with that
in the RA group, whereas significant differences were not observed
between the RA group and MA group at 14 days post ischaemia. These
results indicated that AMD3100 inhibited the migration of NSCs
following ischaemic stroke and that rTMS may enhance the migration
of NSCs via other mechanisms. A previous report indicated that
electrical excitation can direct the migration of NSCs via
galvanotaxis independent of SDF-1α chemical gradients (54). Nevertheless, data from the present
study demonstrated that rTMS enhanced NSC migration in rats
following MCAO, possibly by activating the SDF-1α/CXCR4 axis.
The role of SDF-1α in cerebral insults remains under
exploration. SDF-1α-overexpression has been found to protect
neurones in the penumbra following ischaemic brain insult,
resulting in a significantly decreased number of apoptotic cells
(52). A study by Chiazza et
al (55) indicated that
activation of the SDF-1α/CXCR4 axis resulted in reduced tissue
damage, promoted neuronal survival and improved upper-limb function
recovery after post-stroke treatment in MCAO mice. In addition,
using Nissl staining, Sun et al (56) demonstrated that SDF-1α injection
prevents neurones from degenerating in rats following traumatic
brain injuries. Another previous study reported that rTMS can
enhance neuron survival in the perilesional zone in rats after
traumatic brain injury (57). In
the present study, rTMS treatment rescued ischaemia-induced
neuronal damage in the penumbra. Furthermore, AMD3100 treatment
abolished the enhanced survival of neurones in the perilesional
area in the R group. A study has previously revealed that the
content of SDF-1α is significantly higher in the infarcted striatum
of rats after ischaemic stroke (24) and in the anal sphincter of rats
(58) after electrical stimulation.
Although it is possible that rTMS use can protects neural cells in
the injured area in other ways, such activity may be at least in
part attributed to the SDF-1α/CXCR4 axis. The brain is comprised of
an intricate neuronal network where various types of cells interact
with each other (59). The
endogenous neurogenic response is also under the influence of the
surrounding microenvironment, such as the optimization of SDF-1α
expression and neurotrophins in the penumbra (60,61).
Therefore, rTMS may create a favourable microenvironment where
surviving neurones and NSCs can synergistically promote functional
recovery.
The present study has some limitations. An indirect
method was used to evaluate the migration of NSCs instead of
measuring the migration distance using another method. For example,
SVZ neuroblast migration as evaluated by the distance from the SVZ
to the cell cluster nearest to the ischemic area was considered to
reflect the migratory distance, where the maximal distance that the
SVZ cells covered was measured (62). The influence of rTMS on the
proliferation of NSCs also cannot be ruled out. Further studies are
required to validate if the migration of NSCs mediated by rTMS is
associated with the differentiation of new neurones and if they are
fully functional in neural circuits. Although motor-evoked
potential measurements were used for determining optimal
localization (63), poor
localization and the whole brain may be inevitably stimulated
because the stimulator coil (6-cm in diameter) was relatively large
for the rodent brain. In addition, a previous study suggested that
rTMS treatment (10 Hz rTMS stimulation of the ipsilateral motor
cortex) promotes cognitive functional recovery after ischaemic
stroke in rats by inhibiting apoptosis whilst enhancing
neurogenesis in the hippocampus distal to the stimulation site
(64). By contrast, another study
reported that neurogenesis was not significantly increased after 20
Hz rTMS intervention (the stimulation point was set at the left
frontal cortex) in a rat model of chronic psychosocial stress
(65). This discrepancy may be
attributed to the different stimulating frequencies and animal
models used. These results suggest that appropriate parameters for
electrical stimulation are important in clinical settings and
animal models. It is worth assessing the underlying mechanisms and
optimized protocols of rTMS in neurogenesis.
In conclusion, the present study provided new
insights into the effects of rTMS on neurological deficits after
ischaemic stroke, which suggested that the mechanism may be
associated with the enhancement of endogenous NSC migration and
neuroprotection. In addition, SDF-1α/CXCR4 signalling may serve a
notable role in this process.
Supplementary Material
Representative confocal images of
immunofluorescence staining. Staining in the (A) S group, (B) M
group, (C) R group, (D) RA group and (E) and MA group at 7 days
after ischemia onset. White arrows indicate examples of colocalized
cells. Red represents BrdU and Green represents DCX. (F)
BrdU/DCX/DAPI co-staining in (C). Magnification x200; scale bar, 50
μm. MCAO, middle cerebral artery occlusion model; rTMS, repetitive
Transcranial Magnetic Stimulation; S, sham; M, MCAO; R, rTMS +
MCAO; RA, rTMS + MCAO + AMD3100; MA, MCAO + AMD3100; BrdU,
bromodeoxyuridine; DCX, doublecortin.
Acknowledgements
The authors would like to thank Professor Wang Wei
(Department of Neurology, Tongji Hospital, Tongji Medical College,
Huazhong University of Science and Technology), for his assistance
with the usage of confocal laser-scanning microscope.
Funding
Funding: The present study was supported by The National Natural
Science Foundation of China (grant. nos. 81702231, 81572238 and
81071601).
Availability of data and materials
The datasets used and/or analysed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
YD performed the majority of the experiments. FG
constructed the animal models and analysed the majority of the
data. XHa analysed the immunofluorescence staining data and revised
the manuscript critically for important intellectual content. XHu
contributed to the conception, design, data acquisition, analysis,
interpretation and critically revised the manuscript for important
intellectual content. YD and XHu confirm the authenticity of all
the raw data. All authors have read and approved the final
manuscript.
Ethics approval and consent to
participate
This animal study was approved by The Animal
Experimentation Ethics Committee of Tongji Hospital (approval no.
2017609; Wuhan, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Sanai N, Tramontin AD, Quiñones-Hinojosa
A, Barbaro NM, Gupta N, Kunwar S, Lawton MT, McDermott MW, Parsa
AT, Verdugo JM, et al: Unique astrocyte ribbon in adult human brain
contains neural stem cells but lacks chain migration. Nature.
427:740–744. 2004.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Kannangara TS, Carter A, Xue Y, Dhaliwal
JS, Beique JC and Lagace DC: Excitable adult-generated GABAergic
neurones acquire functional innervation in the cortex after stroke.
Stem Cell Rep. 11:1327–1336. 2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Tsintou M, Dalamagkas K and Makris N:
Taking central nervous system regenerative therapies to the clinic:
Curing rodents versus nonhuman primates versus humans. Neural Regen
Res. 15:425–437. 2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Luo J, Hu X, Zhang L, Li L, Zheng H, Li M
and Zhang Q: Physical exercise regulates neural stem cells
proliferation and migration via SDF-1α/CXCR4 pathway in rats after
ischemic stroke. Neurosci Lett. 578:203–208. 2014.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Zhao S, Qu H, Zhao Y, Xiao T, Zhao M, Li
Y, Jolkkonen J, Cao Y and Zhao C: CXCR4 antagonist AMD3100 reverses
the neurogenesis and behavioral recovery promoted by forced
limb-use in stroke rats. Restor Neurol Neurosci. 33:809–821.
2015.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Jin K, Wang X, Xie L, Mao XO and Greenberg
DA: Transgenic ablation of doublecortin-expressing cells suppresses
adult neurogenesis and worsens stroke outcome in mice. Proc Natl
Acad Sci USA. 107:7993–7998. 2010.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Hummel FC and Cohen LG: Non-invasive brain
stimulation: A new strategy to improve neurorehabilitation after
stroke? Lancet Neurol. 5:708–712. 2006.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Yuan X, Yang Y, Cao N and Jiang C:
Promotion of poststroke motor-function recovery with repetitive
transcranial magnetic stimulation by regulating the
interhemispheric imbalance. Brain Sci. 10(648)2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Fisicaro F, Lanza G, Grasso AA, Pennisi G,
Bella R, Paulus W and Pennisi M: Repetitive transcranial magnetic
stimulation in stroke rehabilitation: Review of the current
evidence and pitfalls. Ther Adv Neurol Disord.
12(1278099885)2019.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Gleeson JG, Lin PT, Flanagan LA and Walsh
CA: Doublecortin is a microtubule-associated protein and is
expressed widely by migrating neurons. Neuron. 23:257–271.
1999.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Couillard-Despres S, Winner B, Schaubeck
S, Aigner R, Vroemen M, Weidner N, Bogdahn U, Winkler J, Kuhn HG
and Aigner L: Doublecortin expression levels in adult brain reflect
neurogenesis. Eur J Neurosci. 21:1–14. 2005.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Cui M, Ge H, Zeng H, Yan H, Zhang L, Feng
H and Chen Y: Repetitive transcranial magnetic stimulation promotes
neural stem cell proliferation and differentiation after
intracerebral hemorrhage in mice. Cell Transplant. 28:568–584.
2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Sherafat MA, Heibatollahi M, Mongabadi S,
Moradi F, Javan M and Ahmadiani A: Electromagnetic field
stimulation potentiates endogenous myelin repair by recruiting
subventricular neural stem cells in an experimental model of white
matter demyelination. J Mol Neurosci. 48:144–153. 2012.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Li M and Ransohoff RM: Multiple roles of
chemokine CXCL12 in the central nervous system: A migration from
immunology to neurobiology. Prog Neurobiol. 84:116–131.
2008.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Ho SY, Ling TY, Lin HY, Liou JT, Liu FC,
Chen IC, Lee SW, Hsu Y, Lai DM and Liou HH: SDF-1/CXCR4 signaling
maintains stemness signature in mouse neural stem/progenitor cells.
Stem Cells Int. 2017(2493752)2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Hatami M, Conrad S, Naghsh P,
Alvarez-Bolado G and Skutella T: Cell-biological requirements for
the generation of dentate gyrus granule neurons. Front Cell
Neurosci. 12(402)2018.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Lu M, Grove EA and Miller RJ: Abnormal
development of the hippocampal dentate gyrus in mice lacking the
CXCR4 chemokine receptor. Proc Natl Acad Sci USA. 99:7090–7095.
2002.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Zanin JP, Abercrombie E and Friedman WJ:
Proneurotrophin-3 promotes cell cycle withdrawal of developing
cerebellar granule cell progenitors via the p75 neurotrophin
receptor. Elife. 5(e16654)2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Ma Q, Jones D, Borghesani PR, Segal RA,
Nagasawa T, Kishimoto T, Bronson RT and Springer TA: Impaired
B-lymphopoiesis, myelopoiesis, and derailed cerebellar neuron
migration in CXCR4- and SDF-1-deficient mice. Proc Natl Acad Sci
USA. 95:9448–9453. 1998.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Tsai LK, Wang Z, Munasinghe J, Leng Y,
Leeds P and Chuang DM: Mesenchymal stem cells primed with valproate
and lithium robustly migrate to infarcted regions and facilitate
recovery in a stroke model. Stroke. 42:2932–2939. 2011.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Sanchez AB, Medders KE, Maung R,
Sanchez-Pavon P, Ojeda-Juárez D and Kaul M: CXCL12-induced
neurotoxicity critically depends on NMDA receptor-gated and L-type
Ca2+ channels upstream of p38 MAPK. J Neuroinflammation.
13(252)2016.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Hwang SJ, Song YM, Cho TH, Kim RY, Lee TH,
Kim SJ, Seo YK and Kim IS: The implications of the response of
human mesenchymal stromal cells in three-dimensional culture to
electrical stimulation for tissue regeneration. Tissue Eng Part A.
18:432–445. 2012.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Wang X, Gao Y, Shi H, Liu N, Zhang W and
Li H: Influence of the intensity and loading time of direct current
electric field on the directional migration of rat bone marrow
mesenchymal stem cells. Front Med. 10:286–296. 2016.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Morimoto J, Yasuhara T, Kameda M, Umakoshi
M, Kin I, Kuwahara K, Kin K, Okazaki M, Takeuchi H, Sasaki T, et
al: Electrical stimulation enhances migratory ability of
transplanted bone marrow stromal cells in a rodent ischemic stroke
model. Cell Physiol Biochem. 46:57–68. 2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Nicolai J, Burbassi S, Rubin J and Meucci
O: CXCL12 inhibits expression of the NMDA receptor's NR2B subunit
through a histone deacetylase-dependent pathway contributing to
neuronal survival. Cell Death Dis. 1(e33)2010.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Huang J, Li Y, Tang Y, Tang G, Yang GY and
Wang Y: CXCR4 antagonist AMD3100 protects blood-brain barrier
integrity and reduces inflammatory response after focal ischemia in
mice. Stroke. 44:190–197. 2013.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Wu KJ, Yu SJ, Shia KS, Wu CH, Song JS,
Kuan HH, Yeh KC, Chen CT, Bae E and Wang Y: A novel CXCR4
Antagonist CX549 induces neuroprotection in stroke brain. Cell
Transplant. 26:571–583. 2017.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Baek A, Kim JH, Pyo S, Jung JH, Park EJ,
Kim SH and Cho SR: The differential effects of repetitive magnetic
stimulation in an in vitro neuronal model of ischemia/reperfusion
injury. Front Neurol. 9(50)2018.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Zong X, Dong Y, Li Y, Yang L, Li Y, Yang
B, Tucker L, Zhao N, Brann DW, Yan X, et al: Beneficial effects of
theta-burst transcranial magnetic stimulation on stroke injury via
improving neuronal microenvironment and mitochondrial integrity.
Transl Stroke Res. 11:450–467. 2020.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Longa EZ, Weinstein PR, Carlson S and
Cummins R: Reversible middle cerebral artery occlusion without
craniectomy in rats. Stroke. 20:84–91. 1989.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Sha R, Zhang B, Han X, Peng J, Zheng C,
Zhang F and Huang X: Electroacupuncture alleviates ischemic brain
injury by inhibiting the miR-223/NLRP3 pathway. Med Sci Monitor.
25:4723–4733. 2019.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Chen J, Sanberg PR, Li Y, Wang L, Lu M,
Willing AE, Sanchez-Ramos J and Chopp M: Intravenous administration
of human umbilical cord blood reduces behavioral deficits after
stroke in rats. Stroke. 32:2682–2688. 2001.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Balkaya M, Krober JM, Rex A and Endres M:
Assessing post-stroke behavior in mouse models of focal ischemia. J
Cereb Blood Flow Metab. 33:330–338. 2013.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Richter L, Neumann G, Oung S, Schweikard A
and Trillenberg P: Optimal coil orientation for transcranial
magnetic stimulation. PLoS One. 8(e60358)2013.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Zhang XY, Sui YF, Guo TC, Wang SH, Hu Y
and Lu YS: Effect of paired associative stimulation on motor cortex
excitability in rats. Curr Med Sci. 38:903–909. 2018.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Shin HI, Han TR and Paik NJ: Effect of
consecutive application of paired associative stimulation on motor
recovery in a rat stroke model: A preliminary study. Int J
Neurosci. 118:807–820. 2008.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Peng JJ, Sha R, Li MX, Chen LT, Han XH,
Guo F, Chen H and Huang XL: Repetitive transcranial magnetic
stimulation promotes functional recovery and differentiation of
human neural stem cells in rats after ischemic stroke. Exp Neurol.
313:1–9. 2019.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Guo F, Han X, Zhang J, Zhao X, Lou J, Chen
H and Huang X: Repetitive transcranial magnetic stimulation
promotes neural stem cell proliferation via the regulation of
MiR-25 in a rat model of focal cerebral ischemia. PLoS One.
9(e109267)2014.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Nygren J, Wieloch T, Pesic J, Brundin P
and Deierborg T: Enriched environment attenuates cell genesis in
subventricular zone after focal ischemia in mice and decreases
migration of newborn cells to the striatum. Stroke. 37:2824–2829.
2006.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Chen H, Qian K, Chen W, Hu B, Blackbourn
LW IV, Du Z, Ma L, Liu H, Knobel KM, Ayala M, et al: Human-derived
neural progenitors functionally replace astrocytes in adult mice. J
Clin Invest. 125:1033–1042. 2015.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Zhan L, Yan H, Zhou H, Sun W, Hou Q and Xu
E: Hypoxic preconditioning attenuates neuronal cell death by
preventing MEK/ERK signaling pathway activation after transient
global cerebral ischemia in adult rats. Mol Neurobiol. 48:109–119.
2013.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Ji Y, Teng L, Zhang R, Sun J and Guo Y:
NRG-1β exerts neuroprotective effects against ischemia
reperfusion-induced injury in rats through the JNK signaling
pathway. Neuroscience. 362:13–24. 2017.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Tan F, Wang J, Liu JX, Wang C, Li M and Gu
Y: Electroacupuncture stimulates the proliferation and
differentiation of endogenous neural stem cells in a rat model of
ischemic stroke. Exp Ther Med. 16:4943–4950. 2018.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Meyer OA, Tilson HA, Byrd WC and Riley MT:
A method for the routine assessment of fore- and hindlimb grip
strength of rats and mice. Neurobehav Toxicol. 1:233–236.
1979.PubMed/NCBI
|
|
45
|
Fu DL, Li JH, Shi YH, Zhang XL, Lin Y and
Zheng GQ: Sanhua decoction, a classic herbal prescription, exerts
neuroprotection through regulating phosphorylated tau level and
promoting adult endogenous neurogenesis after cerebral
ischemia/reperfusion injury. Front Physiol. 11(57)2020.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Jin Y, Raviv N, Barnett A, Bambakidis NC,
Filichia E and Luo Y: The shh signaling pathway is upregulated in
multiple cell types in cortical ischemia and influences the outcome
of stroke in an animal model. PLoS One. 10(e124657)2015.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Wang X, Mao X, Xie L, Sun F, Greenberg DA
and Jin K: Conditional depletion of neurogenesis inhibits long-term
recovery after experimental stroke in mice. PLoS One.
7(e38932)2012.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Gao F, Wang S, Guo Y, Wang J, Lou M, Wu J,
Ding M, Tian M and Zhang H: Protective effects of repetitive
transcranial magnetic stimulation in a rat model of transient
cerebral ischaemia: A microPET study. Eur J Nucl Med Mol Imaging.
37:954–961. 2010.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Li L, El-Hayek YH, Liu B, Chen Y, Gomez E,
Wu X, Ning K, Li L, Chang N, Zhang L, et al: Direct-current
electrical field guides neuronal stem/progenitor cell migration.
Stem Cells. 26:2193–2200. 2008.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Luo Y: Cell-based therapy for stroke. J
Neural Transm (Vienna). 118:61–74. 2011.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Leu S, Lin YC, Yuen CM, Yen CH, Kao YH,
Sun CK and Yip HK: Adipose-derived mesenchymal stem cells markedly
attenuate brain infarct size and improve neurological function in
rats. J Transl Med. 8(63)2010.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Yoo J, Seo JJ, Eom JH and Hwang DY:
Effects of stromal cell-derived factor 1α delivered at different
phases of transient focal ischemia in rats. Neuroscience.
209:171–186. 2012.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Dziembowska M, Tham TN, Lau P, Vitry S,
Lazarini F and Dubois-Dalcq M: A role for CXCR4 signaling in
survival and migration of neural and oligodendrocyte precursors.
Glia. 50:258–269. 2005.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Feng J, Liu J, Zhang X, Zhang L, Jiang J,
Nolta J and Zhao M: Guided migration of neural stem cells derived
from human embryonic stem cells by an electric field. Stem Cells.
30:349–355. 2012.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Chiazza F, Tammen H, Pintana H, Lietzau G,
Collino M, Nyström T, Klein T, Darsalia V and Patrone C: The effect
of DPP-4 inhibition to improve functional outcome after stroke is
mediated by the SDF-1α/CXCR4 pathway. Cardiovasc Diabetol.
17(60)2018.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Sun W, Liu J, Huan Y and Zhang C:
Intracranial injection of recombinant stromal-derived factor-1
alpha (SDF-1α) attenuates traumatic brain injury in rats. Inflamm
Res. 63:287–297. 2014.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Lu X, Bao X, Li J, Zhang G, Guan J, Gao Y,
Wu P, Zhu Z, Huo X and Wang R: High-frequency repetitive
transcranial magnetic stimulation for treating moderate traumatic
brain injury in rats: A pilot study. Exp Ther Med. 13:2247–2254.
2017.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Salcedo L, Lian L, Jiang H, Sopko N, Penn
M, Damaser M and Zutshi M: Low current electrical stimulation
upregulates cytokine expression in the anal sphincter. Int J
Colorectal Dis. 27:221–225. 2012.PubMed/NCBI View Article : Google Scholar
|
|
59
|
George PM and Steinberg GK: Novel stroke
therapeutics: Unraveling stroke pathophysiology and its impact on
clinical treatments. Neuron. 87:297–309. 2015.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Belayev L, Hong S, Menghani H, Marcell SJ,
Obenaus A, Freitas RS, Khoutorova L, Balaszczuk V, Jun B, Oriá RB
and Bazan NG: Docosanoids promote neurogenesis and angiogenesis,
blood-brain barrier integrity, penumbra protection, and
neurobehavioral recovery after experimental ischemic stroke. Mol
Neurobiol. 55:7090–7106. 2018.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Shohayeb B, Diab M, Ahmed M and Ng DCH:
Factors that influence adult neurogenesis as potential therapy.
Transl Neurodegener. 7(4)2018.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Nih LR, Deroide N, Leré-Déan C, Lerouet D,
Soustrat M, Levy BI, Silvestre JS, Merkulova-Rainon T, Pocard M,
Margaill I and Kubis N: Neuroblast survival depends on mature
vascular network formation after mouse stroke: Role of endothelial
and smooth muscle progenitor cell co-administration. Eur J
Neurosci. 35:1208–1217. 2012.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Yoon KJ, Lee Y and Han TR: Mechanism of
functional recovery after repetitive transcranial magnetic
stimulation (rTMS) in the subacute cerebral ischemic rat model:
Neural plasticity or anti-apoptosis? Exp Brain Res. 214:549–556.
2011.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Guo F, Lou J, Han X, Deng Y and Huang X:
Repetitive transcranial magnetic stimulation ameliorates cognitive
impairment by enhancing neurogenesis and suppressing apoptosis in
the hippocampus in rats with ischemic stroke. Front Physiol.
8(559)2017.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Czeh B, Welt T, Fischer AK, Erhardt A,
Schmitt W, Müller MB, Toschi N, Fuchs E and Keck ME: Chronic
psychosocial stress and concomitant repetitive transcranial
magnetic stimulation: effects on stress hormone levels and adult
hippocampal neurogenesis. Biol Psychiatry. 52:1057–1065.
2002.PubMed/NCBI View Article : Google Scholar
|