Introduction
Brain abscess is a serious and life-threatening
infection, although advances in radiology, diagnostics and
treatment have led to a reduction in associated mortality from 40
to 10% over the last 60 years (1).
The diagnosis of brain abscess is most commonly validated by
craniocerebral MRI, particularly using diffusion-weighted imaging
(2) and the reported incidence of
brain abscess ranges from 0.3 to 1.8 cases per 100,000 individuals
(3-6).
Although the overall incidence of brain abscess has declined, the
incidence remains high in specific populations, including those
with human immunodeficiency virus, a history of treatment with
immunosuppressive drugs, disruption of the natural protective
barriers surrounding the brain and those with a systemic source of
infection (1,7-10).
Most patients with brain abscess have predisposing factors
(4,10-12).
For instance, bacterial transmission may be caused by penetrating
trauma, neurosurgery or otorhinolaryngological infection (10,11).
A brain abscess is a focal suppurative process in
the brain parenchyma. The first stage of brain-abscess formation
may lead to perivascular inflammation (13). Subsequently, a capsule is formed,
which evolves over approximately two weeks into a well-vascularized
wall surrounding the necrosis (9,14). A
post-contrast MRI scan displays obvious ring-shaped enhancement of
the abscess wall. While the formation of an abscess wall is
important for limiting pathogen dissemination during the
development of a brain abscess, the thickness of a brain abscess
wall may increase the difficulty of stereotactic aspiration and
also decrease the curative effect of antimicrobial therapy
(15,16). However, studies regarding the wall
of brain abscesses are currently limited.
The formation of a brain-abscess wall refers to the
formation of a fibrotic wall, and the extensive proliferation and
activation of fibroblasts (17,18).
Activated macrophages regulate fibrogenesis by providing cytokines
and growth factors that modulate the proliferation and collagen
synthesis of fibroblasts (19).
Research on fibrotic walls in the central nervous system (CNS) has
identified that alternatively activated M2 macrophages and
microglia accumulate during the course of brain-abscess development
(20).
The distinction between deep and superficial
abscesses is not clearly defined. In the present study, abscesses
of the basal ganglia region and thalamus were defined as deep
abscesses according to the division of midline gliomas (21).
The aim of the present study was to explore
differences in the thickness of deep and superficial brain
abscesses and investigate the factors that affect the capsule of
brain abscesses.
Materials and methods
Patients
Information on patients with brain abscess treated
at Sanbo Brain Hospital, Capital Medical University (Beijing,
China) between December 2008 and December 2018 was retrospectively
reviewed. Inclusion criteria for this group were adult and
pediatric patients diagnosed with a brain abscess and admitted to
the above hospital. The diagnosis of brain abscess was mainly based
on symptomatology, laboratory examination and imaging findings
(2,11). In total, 95 patients with complete
imaging information were selected for inclusion in the study.
Unfortunately, due to the limited samples for immunohistochemical
examination for macrophages, only 37 patients underwent surgical
resection and had sufficient tissue specimens for relevant tests.
The study cohort was divided into those with deep abscesses
(thalamus and basal ganglia region) and superficial abscesses
(frontal lobe, parietal lobe, temporal lobe, occipital lobe and
cerebellum).
The enrolled cases were diagnosed by CT and/or MRI
and pathology. Age, sex, clinical presentation, duration of
symptoms/signs in patients and localization of the abscess were
identified.
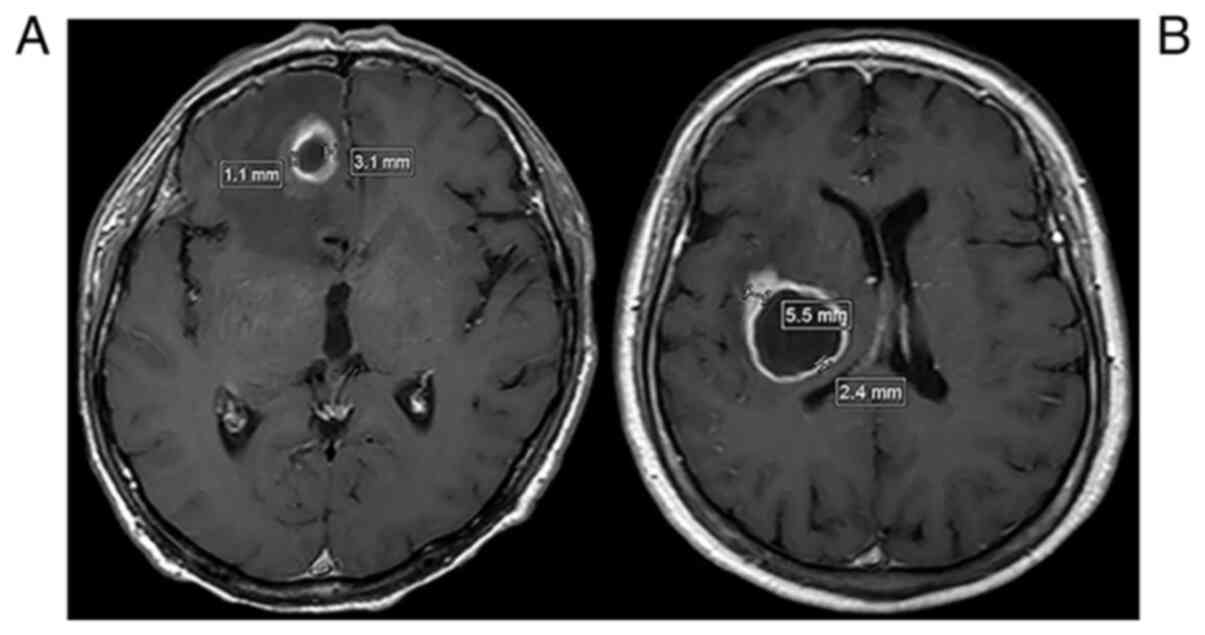
Measurements of the thickness of the
brain abscess wall
All enrolled patients underwent post-contrast MRI
and/or CT scanning prior to treatment. The abscess wall thickness
was measured using miPlatform Viewer version 2.0 (Hinacom Software
and Technology, Ltd.). The thickness of the abscess wall was
measured at the thickest and thinnest places in different axial
slices (≥3 slices; layer thickness, 5 mm). The median thickness in
each layer was calculated and the mean values of the medians were
defined as the thickness of the abscess wall.
Culture and identification of
pathogenic microorganisms
According to our laboratory's standard operation
procedure, all samples of the pus or wall of brain abscesses were
sent to the microbiology laboratory at Sanbo Brain Hospital,
Capital Medical University (Beijing, China) for urgent microscopy,
culture and sensitivity testing. The pus and wall of brain
abscesses were collected in a sterilized vial and snap-frozen in
liquid nitrogen immediately after surgery/aspiration.
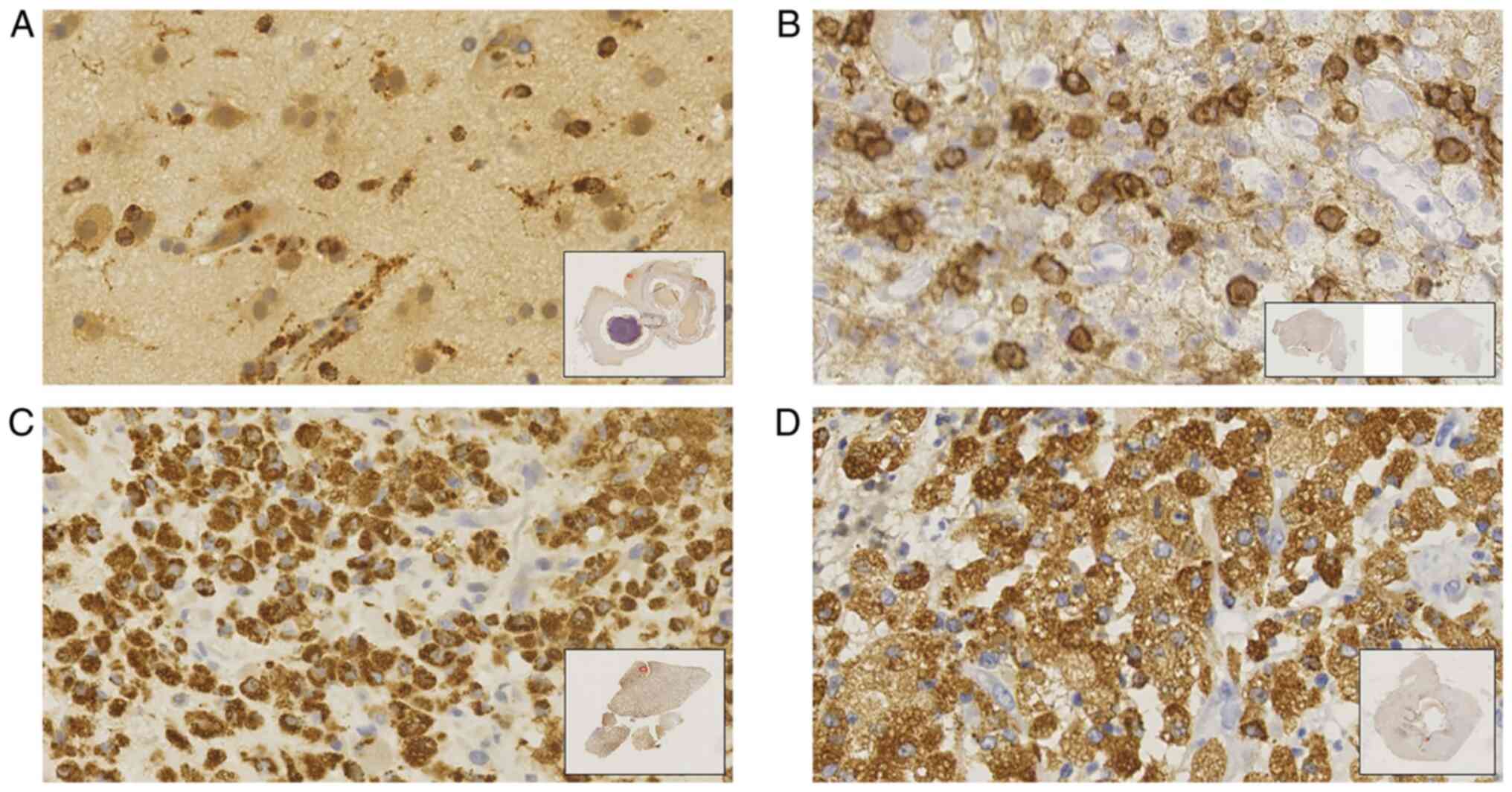
Immunohistochemistry
The group of patients with abscesses included 10
cases with abscess in the deep brain and 27 in the superficial
brain. The samples were fixed with 10% neutral buffered formalin
and further embedded in paraffin. Antigen retrieval was facilitated
by heat and endogenous peroxidases were neutralized with 3%
hydrogen peroxide in a routine fashion. Primary antibodies against
CD68 (anti-CD68 antibody; 1:100; cat. no. ab125212; Abcam) were
applied overnight at 4˚C. According to the manufacturer's protocol,
the Poly-HRP Anti-Mouse/Rabbit IgG Detection System (cat. no.
PV-9000; Zhongshan Goldenbridge-Bio) was employed with incubation
for 30 min at 37˚C. The cutoff value was 10% for CD68. This
approach was identical to that used in a previous study by our
group (22).
The samples were examined using a Nanozoomer Digital
Pathology (NDP; Hamamatsu Photonics; magnification, x100), which
was used to identify five regions (hot spots) with the highest
density of stained CD68+ macrophages or microglia. The
number of stained macrophages or microglia was counted in every
area at a higher magnification (magnification, x400). The average
of the three highest values was used as the final value (23,24).
Statistical analysis
Statistical analyses were performed using the
statistical analysis software package SPSS (version 25.0; IBM
Corp.). Values are expressed as the mean ± standard errors or n
(%). Unpaired student's t-test and Mann-Whitney U test were
utilized. The thickness of the brain-abscess wall and the extent of
macrophage infiltration among different pathogenic microorganisms
were determined using ANOVA. Scheffe's test was used for post-hoc
multiple-comparison testing, with a Scheffe-corrected alpha of 0.05
considered to indicate statistical significance. Correlations were
assessed using Spearman test. Overall survival (OS) was measured
from the date of treatment until death or the date of the last
follow-up. Survival analysis results were performed using
Kaplan-Meier (K-M) survival curves with log rank testing. P<0.05
was considered to indicate statistical significance.
Results
Patients
All patients were diagnosed with a brain abscess
between December 2008 and December 2018. The mean age of the
participants was 41.29 years (range, 5-71 years) and the
male-to-female ratio was 1:0.44 (Table
I).
 | Table ICharacteristics of the patients
(n=95). |
Table I
Characteristics of the patients
(n=95).
| Characteristic | Value |
|---|
| Sex | |
|
Male | 66 (69.47) |
|
Female | 29 (30.52) |
| Age (years) | 41.29±16.76
(5-71) |
| Region of
abscess | |
|
Midline deep
brain (thalamus and basal ganglia region) | 22 (30.14) |
|
Shallow
brain (frontal lobe, parietal lobe, temporal lobe, occipital lobe
and cerebellum) | 73 (76.84) |
| Signs/symptoms in
the deep brain | |
|
Focal
neurological deficits | 16 (72.73) |
|
Fever | 14 (63.64) |
|
Headache | 7 (31.82) |
|
Nausea/vomiting | 5 (22.73) |
|
Impaired
consciousness | 5 (22.73) |
|
Seizures | 2 (9.09) |
| Signs/symptoms in
the shallow regions | |
|
Headache | 40 (54.79) |
|
Focal
neurological deficits | 30 (41.10) |
|
Fever | 20 (27.40) |
|
Headache | 40 (54.79) |
|
Nausea/vomiting | 16 (21.92) |
|
Seizures | 12 (20.27) |
|
Impaired
consciousness | 7 (9.89) |
| Predisposing
conditions in the deep brain | |
|
Diabetes | 4 (18.18) |
|
Surgery | 5 (22.73) |
|
None | 13 (59.09) |
| Predisposing
conditions in the shallow regions | |
|
Surgery | 19 (26.03) |
|
Congenital
heart disease | 6 (8.22) |
|
Intracranial
tumors | 5 (6.85) |
|
Cerebral
hemorrhage | 4 (5.48) |
|
Diabetes | 3 (4.11) |
|
Injury | 3 (4.11) |
|
Leukemia/bone-marrow
transplantation | 2 (2.74) |
|
None | 31 (42.47) |
| Sequelae in the
deep brain | |
|
Limb
dyskinesia | 5 (22.73) |
|
Seizures | 1 (4.55) |
|
None | 16 (72.7) |
| Sequelae in the
shallow brain | |
|
Limb
dyskinesia | 6 (8.22) |
|
Seizures | 5 (6.85) |
|
Headache | 3 (41.10) |
|
Hypaesthesia | 1 (1.37) |
|
None | 58 (79.45) |
Clinical findings
In patients with deep-brain abscess, the prevalent
symptoms were impaired consciousness and focal neurological
deficits. However, these symptoms were less common in patients with
superficial-brain abscess, who were more likely to experience
symptoms such as nausea, vomiting and fever (Table I).
Thickness of the abscess wall and
macrophages in brain abscesses
In total, 95 patients with brain abscess were
reviewed. The location of the brain abscesses was as follows: A
total of 22 patients had deep brain abscess (thalamus and basal
ganglia region) and 73 patients had superficial brain abscess
(frontal lobe, parietal lobe, temporal lobe, occipital lobe and
cerebellum). According to the independent-samples t-test, the
thickness of the wall of the brain abscesses was significantly
different between the deep (4.65±2.66 mm) and the superficial
(3.16±1.92 mm) groups (95% CI: 0.48-2.51; P=0.004; Fig. 1 and Table II). To study the infiltration of
macrophages or microglia in the brain abscesses, CD68-stained
slides were collected from 37 patients and the number of
macrophages/microglia was counted. This group included 10 patients
with deep-brain abscess and 27 patients with superficial-brain
abscess. There was a significant difference between the number of
macrophages/microglia in the deep-brain abscess (41.40±11.44) and
the superficial-brain abscess walls (19.67±15.77; 95% CI:
0.63-22.84; P=0.04; Fig. 2 and
Table II). However, there was no
statistically significant linear correlation (P=0.99; Table II) between the thickness of the
brain abscess wall and the number of macrophage infiltrations, as
calculated with a bivariate correlation.
 | Table IISummary of statistical results on the
related factors in 95 patients. |
Table II
Summary of statistical results on the
related factors in 95 patients.
| Index | Statistical
method | P-value |
|---|
| Thickness of
abscess wall | t-test | 0.004 |
|
Deep-brain
(mm) | 4.65±2.66 | |
|
Superficial
brain (mm) | 3.16±1.92 | |
| Number of
macrophages/microglia | t-test | 0.04 |
|
Deep
brain(n) | 41.40±11.44 | |
|
Superficial
brain(n) | 19.67±15.77 | |
| Thickness of
abscess wall | Spearman test | 0.99 |
| Number of
macrophages/microglia | | |
| Duration of
symptoms | Mann-Whitney | 0.65 |
|
Deep
brain(day) | 15 | |
|
Superficial
brain(day) | 14 | |
| Thickness of
abscess wall | Spearman test | 0.21 |
| Duration of
symptoms | | |
Duration of the symptoms or signs
The different symptoms or signs between the two
types of brain abscesses were present for 41.88±90.15 days (ranging
from 1 to 730 days; median, 15 days). The median duration of
symptoms for patients with deep-brain abscess (23.58±30.63 days;
median, 15 days) was shorter than for those with superficial
abscess (47.00±90.41 days; median, 14 days), but there was no
statistically significant difference according to nonparametric
testing (P=0.654; Table II);
furthermore, there was no correlation between the thickness of the
brain abscess wall and symptom duration (P=0.21; Table II) according to bivariate
correlation analysis.
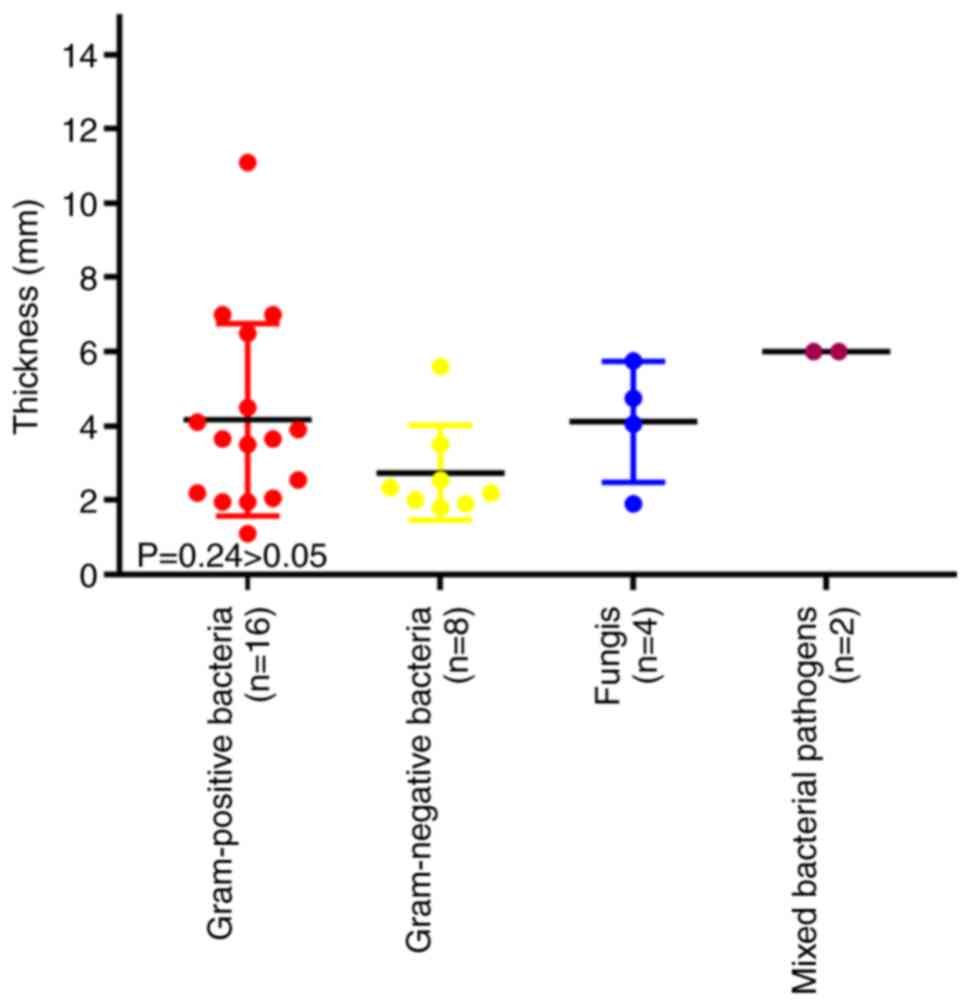
Macrophages and microorganisms
Taken from the pus and wall of the abscess, abscess
cultures were positive in 34 of 71 patients who received surgery or
drainage of the abscess (Table
III). The most common species were Gram-positive bacteria,
followed by Gram-negative bacteria and fungi (Table III). The thickness of the abscess
wall exhibited no significant difference between different
underlying organisms according to one-way ANOVA (F=2.00, P=0.24;
Fig. 3).
 | Table IIICulture of pathogens isolated from 34
patients with brain abscess. |
Table III
Culture of pathogens isolated from 34
patients with brain abscess.
| Organism | Number of
patients |
|---|
| Gram-positive
bacteria | 18 |
|
Streptococcus
species | 6 |
|
Staphylococci | 7 |
|
Corynebacterium
urealyticum | 1 |
|
Gemella
haemolysans | 1 |
|
Other | 1 |
| Gram-negative
bacteria | 10 |
|
Pseudomonas
aeruginosa | 5 |
|
Proteus
mirabilis | 1 |
|
Klebsiella
pneumoniae | 1 |
|
Serratia
glutinosa | 1 |
| Mixed bacterial
pathogensa | 2 |
| Fungi | 4 |
| Negative
culture | 39 |
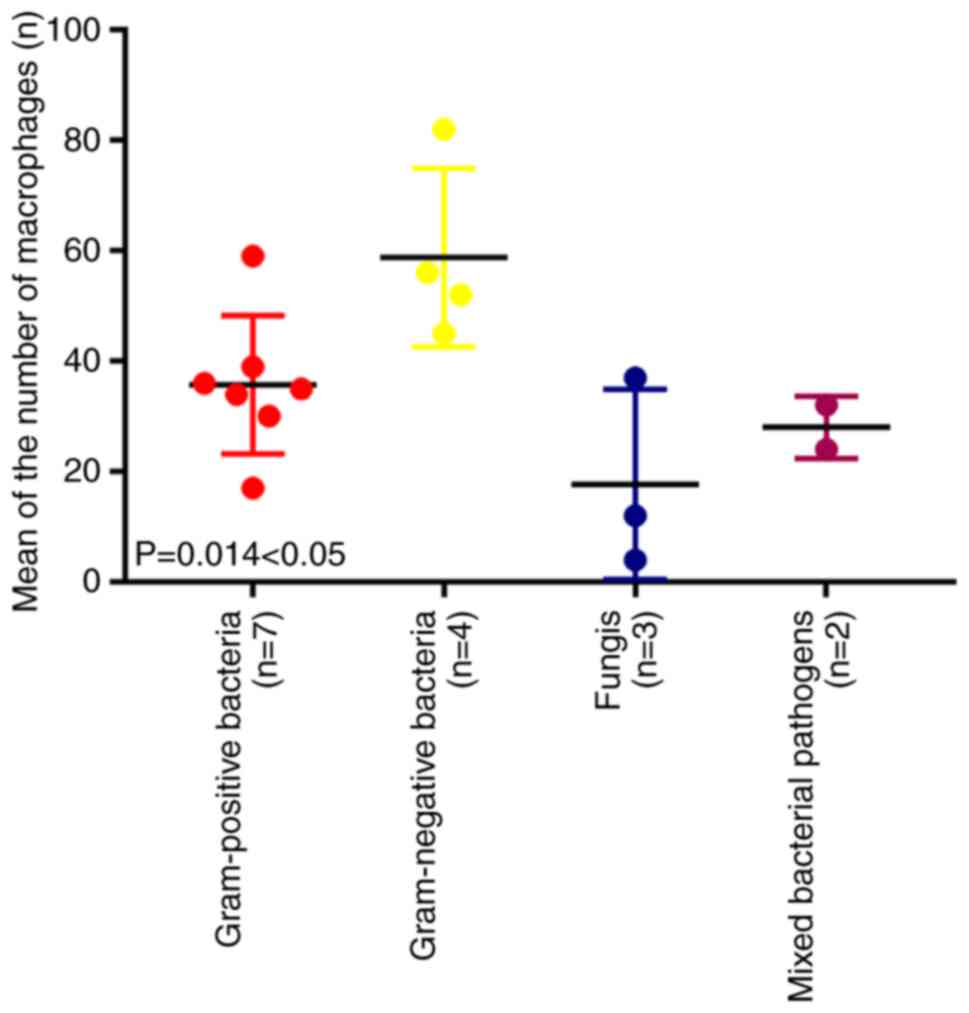
Among the 37 patients with CD68-stained slides, 16
were positive on brain-abscess pus and/or wall bacterial culture
(Table IV). Of note, according to
one-way ANOVA and Scheffe's post-hoc tests, the extent of
macrophage infiltration in brain abscesses (Gram staining-positive
group: n1=35.71±12.51 vs. Gram staining-negative group:
n2=58.75±16.15 vs. fungal group:
n3=17.67±17.20 vs. mixed bacterial group:
n4=28.00±5.67) caused by various microorganisms was
significantly different (F=5.43, P=0.014; Fig. 4) and there was a significant
difference between the gram-negative bacteria group and the fungi
group in macrophage distribution according to Scheffe's post-hoc
test (P=0.019). Among the cases of brain abscess caused by
Staphylococcus, two patients had deep-brain abscesses and
three had superficial-brain abscesses; the extent of
macrophage/microglia infiltration in deep-brain abscesses
(47.00±11.97) was greater than that in the superficial-brain
abscesses (33.33±3.06, P=0.03; Table
V). In addition, the thickness of the brain-abscess wall was
significantly different between the deep (5.60±1.98) and the
superficial (1.95±0.76) abscesses (P=0.04; Table V). The correlation coefficient
between the thickness of an abscess wall and the infiltrating
number of macrophages was 0.09 (P=0.04; Table V), indicating a positive
correlation.
 | Table IVThe number of macrophages in 16 cases
with positive bacterial culture was observed. |
Table IV
The number of macrophages in 16 cases
with positive bacterial culture was observed.
| Organism | Number of
patients |
|---|
| Gram-positive
bacteria | 7 |
|
Staphylococci | 5 |
|
Streptococcus
species | 1 |
|
Other | 1 |
| Gram-negative
bacteria | 4 |
|
Pseudomonas
aeruginosa | 1 |
|
Klebsiella
pneumoniae | 1 |
|
Serratia
glutinosa | 1 |
|
Proteus
mirabilis | 1 |
| Mixed bacterial
pathogensa | 2 |
| Fungi | 3 |
| Negative
culture | 21 |
 | Table VA summary of results on the thickness
of abscess wall and macrophages/microglia in 5 patients with brain
abscess caused by Staphylococcus. |
Table V
A summary of results on the thickness
of abscess wall and macrophages/microglia in 5 patients with brain
abscess caused by Staphylococcus.
| Index | Statistical
method | P-value |
|---|
| Thickness of
abscess wall | t-test | 0.04 |
|
Deep-brain
(mm) | 5.60±1.98 | |
|
Superficial
brain (mm) | 1.95±0.76 | |
| Number of
macrophages/microglia | t-test | 0.03 |
|
Deep brain
(n) | 47.00±11.97 | |
|
Superficial
brain (n) | 33.33±3.06 | |
| Thickness of
abscess wall | Spearman test | 0.04 |
| Number of
macrophages/microglia | B=0.09 | |
Survival analysis
Follow-up was performed for 67 patients. Of these,
32 patients received surgery, 20 patients received puncture
drainage and 15 patients were treated with drug therapy. The
follow-up time ranged from 0.7-106 months, with a median follow-up
time of 25.00 months. No recurrence of the brain abscesses was
detected during follow-up. The OS of patients with deep-brain
abscess was not significantly reduced compared with that of
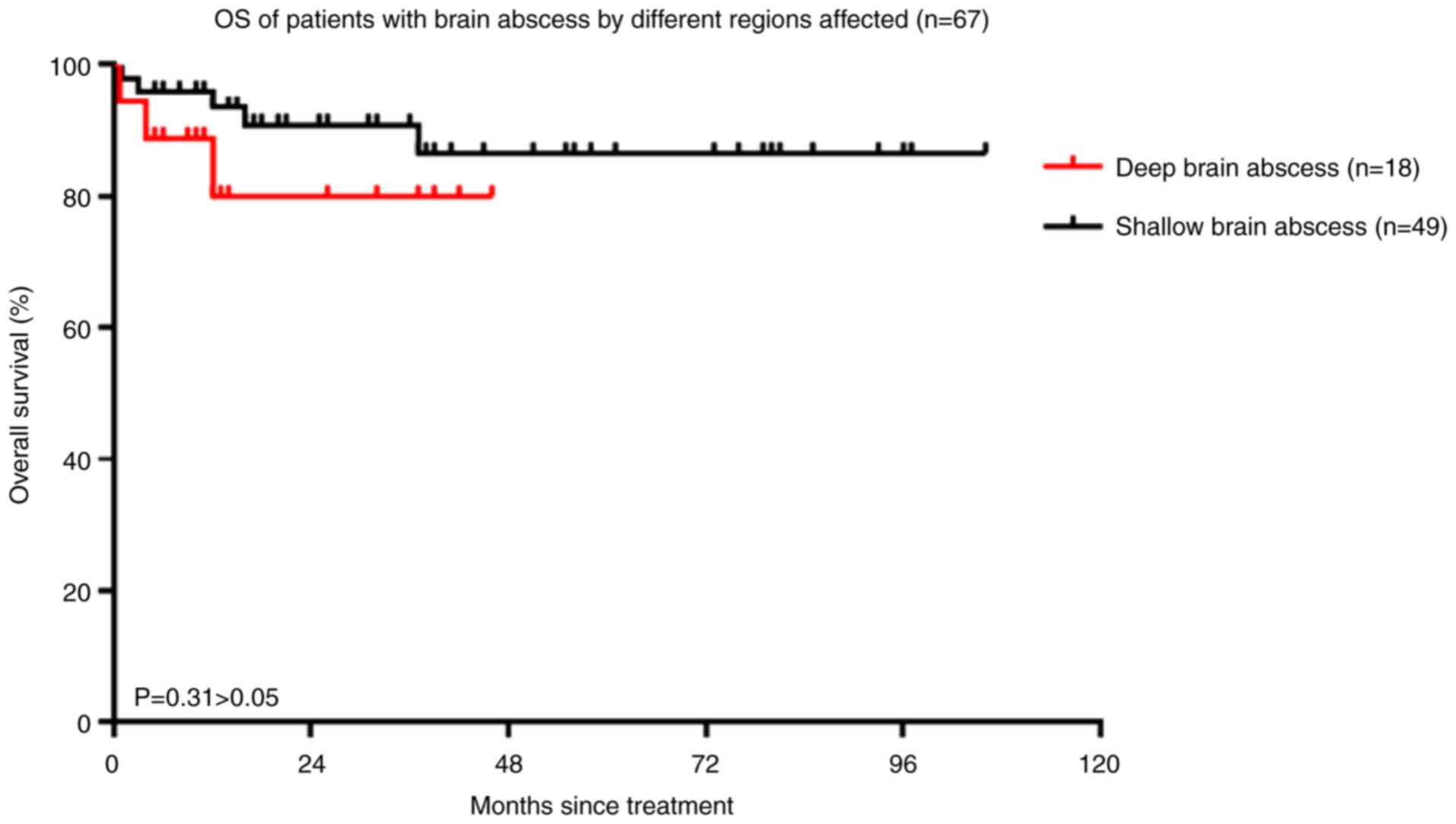
patients with superficial-brain abscess (P=0.31; Fig. 5). Furthermore, there was no
significant difference in OS among the different treatment groups
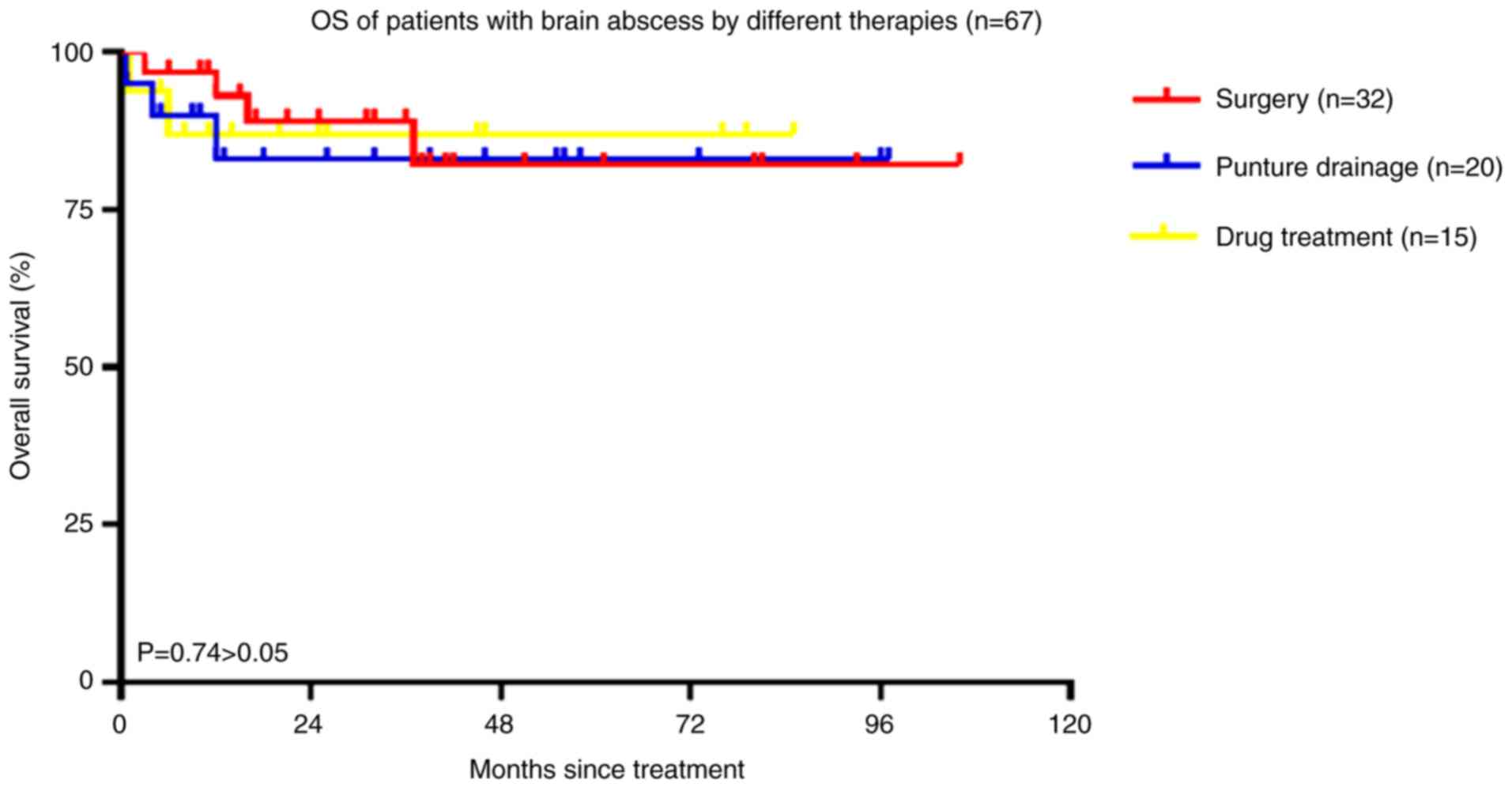
(P=0.74; Fig. 6).
Discussion
In the present study, it was demonstrated that brain
abscesses were different between the deep and superficial regions
in terms of thickness of the abscess wall and infiltration by
macrophages/microglia. Furthermore, the factors affecting the wall
of a brain abscess were analyzed. The formation of a capsule in a
brain abscess has a significant effect on limiting pathogen
diffusion. Despite previous studies illustrating the mechanisms of
fibrotic processes (25) in the
CNS, these mechanisms remain to be fully elucidated and previous
studies have rarely focused on the relationship between the
location and the fibrosis of brain abscesses.
In the cohort of the present study, patients with
abscess in deep regions more frequently had focal neurological
deficits (72.73%) and impaired consciousness (22.73%) compared with
patients with abscess in superficial brain regions (41.10 and
9.89%, respectively). Furthermore, the complications of deep-brain
abscess included dyskinesia following treatment, which caused most
patients with deep-brain abscess to suffer with movement disorders
during the follow-up (26).
By measuring the thickness of the wall of brain
abscesses in images, it was determined that the wall of deep-brain
abscesses was thicker than that of superficial-brain abscesses. The
process of brain-abscess encapsulation involves the formation of a
fibrotic wall. A key feature of fibrotic wall formation is
extensive proliferation and activation of fibrotic tissue (20,27,28);
fibroblasts secrete α-smooth muscle actin, which is a constituent
of the fibrotic abscess capsule (29). Previous studies demonstrated that
alternatively activated macrophages enhanced fibrogenesis of
fibroblasts by providing profibrogenic factors, such as
transforming growth factor β1 and platelet-derived growth factors
(19). Fibrotic wall formation was
associated with increased numbers of alternatively activated
microglia and macrophages (19) and
relative markers for these cells were also indicated to increase,
in accordance with fibrotic wall formation (20).
Microglia/macrophage activation is an important
feature of a brain abscess (30).
Studies have indicated that activated glial cells and infiltrated
peripheral immune cells continuously release pro-inflammatory
mediators. These mediators enhance the subsequent recruitment and
activation of newly recruited inflammatory cells and glial cells;
this effectively maintains the antimicrobial inflammatory response
and ultimately achieves the goal of eliminating pathogenic
microorganisms (31,32). Of note, the present study indicated
that the number of infiltrating macrophages in deep-brain abscesses
was greater than that in superficial-brain abscess walls. Studies
in the rat brain have indicated that macrophages are mainly
distributed in the gray matter of the brain, particularly the basal
ganglia, hippocampus, olfactory telencephalon and substantia nigra
(33). In addition, the basal
ganglia are rich in blood vessels, so the content of macrophages in
brain abscesses of the basal ganglia may be higher.
There is a large time range in the symptom/sign
duration, as was particularly evident in the case of a
five-year-old female patient with epilepsy for ~730 days, who, due
to poor economic conditions and personal factors, was not diagnosed
accurately and treated effectively until they received surgical
treatment at our hospital. At the final follow-up, it was
determined that the seizure frequency had gradually decreased after
the brain abscess was cured. The thickness of the wall of the brain
abscess may change during a long evolutionary period. Comparing the
CT appearances and the neuropathologic findings in dogs, Britt
et al (13) and Enzmann
et al (34) determined that
ring-shaped contrast enhancement appeared on CT imaging in the
early cerebritis stage and well-encapsulated brain abscesses
occurred at the age of 14 days and beyond through the accumulation
of fibroblasts and neovascularization, with the thickness and
diameter of ring-like abscesses decreasing as cerebritis later
receded. Pathological abscess formation processes may slow down in
the chronically immunosuppressed host (35). However, the results of the present
study indicated that time was not significantly correlated with the
thickness of a brain abscess. There was no significant difference
between the deep and superficial brain abscesses over time. This
may be due to a number of reasons, including the fact that most
patients present at the stage of capsule formation or later; the
thickness of the wall of a brain abscess may undergo no significant
changes in the absence of any therapeutic intervention. Similarly,
pathophysiological analysis of brain abscesses suggests that
activation of microglia/macrophages is also evident during early
encephalitis (days 1-3) and continues throughout abscess
development. The stage of advanced encephalitis (days 4-9) is
associated with macrophage and lymphocyte infiltration; the final
capsule phase occurs after day 10 and is associated with the
formation of well-vascularized abscess walls (32). Combined with the results of the
above study, the present study observed that the subsequent immune
response to pathogenic bacteria became gradually stronger. The
immune response to pathogens may be able to destroy surrounding
normal brain tissue (31,36,37).
Thus, it is not surprising that mechanisms of reducing defensive
responses and promoting wound repair and healing are part of
immunity. Previous studies have demonstrated the mechanisms by
which the innate immune response may be reduced, including the
following: Reducing pathogen load, decomposing pro-inflammatory
mediators, managing the negative feedback of activating immune
cells and controlling the activation pathway (38). Van Rossum et al (39) indicated that peripheral macrophages
and dead or dying immune cells spread throughout the vascular
system and are eventually removed from circulation, although the
movement of cells and solutes from the blood vessels to the brain
parenchyma remains to be characterized (38). Due to the long duration of
symptoms/signs (41.88±90.15 days) and differences in individual
immunity and fibrosis, the imaging results of brain abscesses and
the low bacterial culture rate of patients in this group indicate
that most patients may be in the period of declining immune defense
response. Macrophages/microglia were in retreat and the number of
infiltrating macrophages in brain abscess was not linearly related
to the thickness of the abscess wall. In addition, the time of
symptoms/signs may not accurately reflect the pathophysiological
stage of the brain abscess. It is possible that there is no linear
correlation between the time of symptoms/signs.
The brain is well protected from invading pathogens
by the two major barriers [the blood-brain barrier (BBB) and the
blood-cerebrospinal fluid barrier]. However, bacteria have
developed a wide variety of different strategies to cross these
barriers and reach the CNS (40,41).
They use a variety of different virulence factors to invade the
host cell, including factors that mediate intracellular survival,
induction of host-cell signaling and inflammatory response; these
factors enable them to attach to and traverse the two barriers
(40). Certain mechanisms may vary
with different pathogens (40). For
instance, Streptococcus pneumoniae may translocate across
the BBB by altering expression of the Neisseria adhesin A for
attachment and interacting with the BBB (42-44).
However, Escherichia coli relies on type 1 fimbriae, outer
membrane protein A, IbeA; a virulence factor of Eschericia
coli, and cytotoxic necrotizing factor 1 to attach and invade
the BBB (45-48).
Due to the small number of positive cases determined by bacterial
culture in the present study, a detailed classification was not
easy to perform using statistical methods and the following groups
were created: The Gram staining positive group, Gram staining
negative group, a fungal group and a mixed bacterial group.
Different pathogenic bacteria of brain abscesses may have different
effects on macrophage infiltration, but no studies have been
performed to investigate such differences. In the present study,
the extent of macrophage infiltration in fungal brain abscesses was
the lowest, which may be related to their ability to evade an
immune response by associating with Toll-like receptor (TLR) action
and anti-inflammatory response (49); an example is C. albicans,
which may induce immune suppression by activating TLR-2, leading to
the release of IL-10, an anti-inflammatory cytokine that activates
CD4+CD25+ T-regulatory cells (50).
In the present study, the extent of macrophage
infiltration caused by gram-negative bacteria in brain abscesses
was the highest. However, the number of cases with a positive
culture of gram-negative bacteria was lower in deep-brain abscesses
and it was not possible to perform a statistical analysis.
Therefore, a statistical analysis of relevant factors in brain
abscesses caused by Staphylococcus, which is a common
pathogenic bacteria in brain abscesses, was performed (3,51). Of
note, in the brain abscesses caused by Staphylococci, the
thickness of the deep-brain abscess wall was thicker than that of
the superficial-brain abscess wall. The amount of macrophage
infiltration in the deep brain abscess was higher than that in the
superficial brain abscess and the thickness of the wall of brain
abscesses was positively correlated with the number of macrophages.
Certain models have demonstrated that Staphylococcus aureus
and its cell wall product peptidoglycan had a strong stimulatory
effect on pro-inflammatory mediators produced by primary microglial
cells (36), inducing a rapid
elevation and sustained high levels of pro-inflammatory cytokines
and chemokines, including IL-1β, TNF, IL-12 p40, C-X-C motif
chemokine ligand 2, C-C motif chemokine ligand (CCL)2, CCL3 and
CCL4 (52-54).
The release of pro-inflammatory cytokines led to the destruction of
the BBB and vascular cell adhesion molecules also promoted the
involvement of macrophages in the evolution of brain abscesses
(32). These changes are associated
with the persistence of microglia/macrophages in brain abscesses.
In brain abscesses caused by the same pathogen, the enhanced immune
response and the secretion of inflammatory factors led to an
increased number of macrophage infiltration and thickening of the
wall of the deep-brain abscess. Due to the small number of positive
cases on bacterial culture, only the corresponding results of brain
abscess mainly caused by Staphylococcus are available in the
present study. Whether the wall thickness of brain abscesses caused
by any other pathogenic microorganisms is positively correlated
with the extent of macrophage infiltration requires to be further
verified in larger studies.
However, in the present study, no difference in the
thickness of the brain-abscess wall was obtained among the
different types of pathogens. This result may be influenced by
immune differences mainly caused by the various pathogenic
microorganisms.
In the present study, 68 patients were successfully
followed up, including 16 patients with deep-brain abscess. A total
of 10 patients (including 3 patients with abscesses in the deep
brain) died after the surgery; all of these had a certain type of
predisposing condition, such as history of operations, congenital
heart disease, malignant tumors, cerebral hemorrhage or diabetes.
Among the 10 patients, two patients died from malignant tumors and
the cause of death in the remainder of the patients was associated
with illnesses prior to treatment. During follow-up, there was no
evidence of recurrence of brain abscess after treatment in either
the living patients nor the deceased ones. Therefore, no
statistical calculation or discussion of progression-free survival
was performed. There was no statistically significant difference in
the OS of patients with deep-brain abscess compared to those with
superficial-brain abscess, which may be related to the less
patients with superficial-brain abscess. Furthermore, there was no
significant difference in OS among the different treatment groups.
Therefore, in the present study, brain abscess had a low mortality
rate (12.00%), in comparison to the proportion of 40% in 1960 and
15% in the past decade (10). Of
note, the mortality of the patients with deep-brain abscess (19%)
was higher than that of the patients with abscess in superficial
brain regions (10%), however, no significant difference was
observed. However, the missing follow-up of certain patients may
have influenced the accuracy of the outcomes reported in the
present study.
In conclusion, the present study reported on the
increased thickness of abscesses and the increased number of
macrophages/microglia in the wall of deep-brain abscesses in
comparison to shallow-brain abscesses. Brain abscesses at different
sites caused by the same pathogen exhibited differences in wall
thicknesses and extent of macrophage infiltration, suggesting that
the thickness of the wall of brain abscesses in the deep brain may
be associated with the extent of macrophage infiltration. Further
studies are required to illustrate the formation mechanisms of
abscesses in the deep and superficial brain. In addition, due to
the high mortality and disability rate of deep brain abscesses and
the poor effect of antibiotic treatment alone based on the previous
treatment experiences, surgical treatment of abscesses combined
with drug treatment is encouraged. Choosing the correct treatment
method according to the pathological stage of an abscess and its
location may improve patient outcomes.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
SH designed the study. ZY, YY, NL and PW acquired
important data and revised the manuscript for important
intellectual content. YY, NL, ZY and XQ performed the histological
examination of the samples. PW, ZY, LZ and MH analyzed the data. ZY
and SH wrote the article. ZY and SH confirm the authenticity of all
the raw data. All authors have read and approved the final
manuscript.
Ethics approval and consent to
participate
This study was approved by the Ethics Committee of
Sanbo Brain Hospital (Beijing, China). Written informed consent for
publication was obtained from all parents, legal guardians and
participants.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Carpenter J, Stapleton S and Holliman R:
Retrospective analysis of 49 cases of brain abscess and review of
the literature. Eur J Clin Microbiol Infect Dis. 26:1–11.
2007.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Reddy JS, Mishra AM, Behari S, Husain M,
Gupta V, Rastogi M and Gupta RK: The role of diffusion-weighted
imaging in the differential diagnosis of intracranial cystic mass
lesions: A report of 147 lesions. Surg Neurol. 66:246–250-251.
2006.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Helweg-Larsen J, Astradsson A, Richhall H,
Erdal J, Laursen A and Brennum J: Pyogenic brain abscess, a 15 year
survey. BMC Infect Dis. 12(332)2012.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Nicolosi A, Hauser WA, Musicco M and
Kurland LT: Incidence and prognosis of brain abscess in a defined
population: Olmsted County, Minnesota, 1935-1981.
Neuroepidemiology. 10:122–131. 1991.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Felsenstein S, Williams B, Shingadia D,
Coxon L, Riordan A, Demetriades AK, Chandler CL, Bassi S,
Koutoumanou E, Stapleton S, et al: Clinical and microbiologic
features guiding treatment recommendations for brain abscesses in
children. Pediatr Infect Dis J. 32:129–135. 2013.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Nathoo N, Nadvi SS, Narotam PK and van
Dellen JR: Brain abscess: Management and outcome analysis of a
computed tomography era experience with 973 patients. World
Neurosurg. 75:716–726; discussion 612-617. 2011.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Xiao F, Tseng MY, Teng LJ, Tseng HM and
Tsai JC: Brain abscess: Clinical experience and analysis of
prognosticfactors. Surg Neurol. 63:442–450. 2005.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Seydoux C and Francioli P: Bacterial brain
abscesses: Factors influencing mortality and sequelae. Clin Infect
Dis. 15:394–401. 1992.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Kao PT, Tseng HK, Liu CP, Su SC and Lee
CM: Brain abscess: Clinical analysis of 53 cases. J Microbiol
Immunol Infect. 36:129–136. 2003.PubMed/NCBI
|
|
10
|
Brouwer MC, Coutinho JM and van de Beek D:
Clinical characteristics and outcome of brain abscess: Systematic
review and meta-analysis. Neurology. 82:806–813. 2014.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Brouwer MC, Tunkel AR, McKhann GM II and
van de Beek D: Brain abscess. N Engl J Med. 371:447–456.
2014.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Tunkel AR: Brain abscess. In: Bennett JE,
Dolin R, Blaser M, (eds). Principles and practice of infectious
diseases. 8th edition. Philadelphia: Elsevier; pp1265-1272,
2015.
|
|
13
|
Britt RH, Enzmann DR and Yeager AS:
Neuropathological and computerized tomographic findings in
experimental brain abscess. J Neurosurg. 55:590–603.
1981.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Mathisen GE and Johnson JP: Brain abscess.
Clin Infect Dis. 25:763–781. 1997.PubMed/NCBI View
Article : Google Scholar
|
|
15
|
Sauermann R, Karch R, Langenberger H,
Kettenbach J, Mayer-Helm B, Petsch M, Wagner C, Sautner T,
Gattringer R, Karanikas G and Joukhadar C: Antibiotic abscess
penetration: Fosfomycin levels measured in pus and simulated
concentration-time profiles. Antimicrob Agents Chemother.
49:4448–4454. 2005.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Gerding DN, Kozak AJ, Peterson LR and Hall
WH: Failure of single doses of cefazolin and cefamandole to
penetrate experimental chronic Escherichia coli abdominal
abscesses. Antimicrob Agents Chemother. 17:1023–1029.
1980.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Mutsaers SE, Bishop JE, McGrouther G and
Laurent GJ: Mechanisms of tissue repair: From wound healing to
fibrosis. Int J Biochem Cell Biol. 29:5–17. 1997.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Chambers RC, Laurent GJ and
Westergren-Thorsson G: Cadmium inhibits proteoglycan and
procollagen production by cultured human lung fibroblasts. Am J
Respir Cell Mol Biol. 19:498–506. 1998.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Song E, Ouyang N, Hörbelt M, Antus B, Wang
M and Exton MS: Influence of alternatively and classically
activated macrophages on fibrogenic activities of human
fibroblasts. Cell Immunol. 204:19–28. 2000.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Aldrich A and Kielian T: Central nervous
system fibrosis is associated with fibrocyte-like infiltrates. Am J
Pathol. 179:2952–2962. 2011.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Villa C, Miquel C, Mosses D, Bernier M and
Di Stefano AL: The 2016 World Health Organization classification of
tumours of the central nervous system. Presse Med. 47:e187–e200.
2018.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Han S, Wang PF, Xing YX, Song HW, Yao K
and Lin ZX: Human Cytomegalovirus (HCMV) infection was not
correlated with overall survival in glioblastomas. Neoplasma.
65:431–435. 2018.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Gould PV and Saikali S: A comparison of
digitized frozen section and smear preparations for intraoperative
neurotelepathology. Anal Cell Pathol (Amst). 35:85–91.
2012.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Zheng PP, van der Weiden M and Kros JM:
Fast tracking of co-localization of multiple markers by using the
nanozoomer slide scanner and NDPViewer. J Cell Physiol.
229:967–973. 2014.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Meneghin A and Hogaboam CM: Infectious
disease, the innate immune response, and fibrosis. J Clin Invest.
117:530–538. 2007.PubMed/NCBI View
Article : Google Scholar
|
|
26
|
Bhatia R, Tandon P and Misra NK:
Inflammatory lesions of the basal ganglia and thalamus: Review of
twenty-one cases. Neurosurgery. 19:983–988. 1986.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Burt AD: Pathobiology of hepatic stellate
cells. J Gastroenterol. 34:299–304. 1999.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Phan SH: Role of the myofibroblast in
pulmonary fibrosis. Kidney Int Suppl. 54 (Suppl):S46–S48.
1996.PubMed/NCBI
|
|
29
|
Clouthier DE, Comerford SA and Hammer RE:
Hepatic fibrosis, glomerulosclerosis, and a lipodystrophy-like
syndrome in PEPCK-TGF-beta1 transgenic mice. J Clin Invest.
100:2697–713. 1997.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Kielian T and Hickey WF: Proinflammatory
cytokine, chemokine, and cellular adhesion molecule expression
during the acute phase of experimental brain abscess development.
Am J Pathol. 157:647–658. 2000.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Baldwin AC and Kielian T: Persistent
immune activation associated with a mouse model of
Staphylococcus aureus-induced experimental brain abscess. J
Neuroimmunol. 151:24–32. 2004.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Kielian T: Immunopathogenesis of brain
abscess. J Neuroinflammation. 1(16)2004.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Lawson LJ, Perry VH, Dri P and Gordon S:
Heterogeneity in the distribution and morphology of microglia in
the normal adult mouse brain. Neuroscience. 39:151–710.
1990.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Enzmann DR, Britt RH and Yeager AS:
Experimental brain abscess evolution: Computed tomographic and
neuropathologic correlation. Radiology. 133:113–122.
1979.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Obana WG, Britt RH, Placone RC, Stuart JS
and Enzmann DR: Experimental brain abscess development in the
chronically immunosuppressed host. Computerized tomographic and
neuropathological correlations. J Neurosurg. 65:382–391.
1986.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Kielian T, Barry B and Hickey WF: CXC
chemokine receptor-2 ligands are required for neutrophil-mediated
host defense in experimental brain abscesses. J Immunol.
166:4634–4643. 2001.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Kielian T, Cheung A and Hickey WF:
Diminished virulence of an alpha-toxin mutant of Staphylococcus
aureus in experimental brain abscesses. Infect Immun.
69:6902–6911. 2001.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Colton CA: Heterogeneity of microglial
activation in the innate immune response in the brain. J
Neuroimmune Pharmacol. 4:399–418. 2009.PubMed/NCBI View Article : Google Scholar
|
|
39
|
van Rossum D, Hilbert S, Strassenburg S,
Hanisch UK and Brück W: Myelin-phagocytosing macrophages in
isolated sciatic and optic nerves reveal a unique reactive
phenotype. Glia. 56:271–283. 2008.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Herold R, Schroten H and Schwerk C:
Virulence factors of meningitis-causing bacteria: Enabling brain
entry across the blood-brain barrier. Int J Mol Sci.
20(5393)2019.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Dando SJ, Mackay-Sim A, Norton R, Currie
BJ, St John JA, Ekberg JA, Batzloff M, Ulett GC and Beacham IR:
Pathogens penetrating the central nervous system: Infection
pathways and the cellular and molecular mechanisms of invasion.
Clin Microbiol Rev. 27:691–726. 2014.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Iovino F, Orihuela CJ, Moorlag HE, Molema
G and Bijlsma JJ: Interactions between blood-borne Streptococcus
pneumoniae and the blood-brain barrier preceding meningitis. PLoS
One. 8(e68408)2013.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Shainheit MG, Mulé M and Camilli A: The
core promoter of the capsule operon of Streptococcus pneumoniae is
necessary for colonization and invasive disease. Infect Immun.
82:694–705. 2014.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Gratz N, Loh LN, Mann B, Gao G, Carter R,
Rosch J and Tuomanen EI: Pneumococcal neuraminidase activates TGF-β
signalling. Microbiology. 163:1198–1207. 2017.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Kim KS: Human meningitis-associated
Escherichia coli. EcoSal Plus: May 1, 2016 (Epub ahead of
print). doi: 10.1128/ecosalplus.ESP-0015-2015.
|
|
46
|
Khan NA, Shin S, Chung JW, Kim KJ, Elliott
S, Wang Y and Kim KS: Outer membrane protein A and cytotoxic
necrotizing factor-1 use diverse signaling mechanisms for
Escherichia coli K1 invasion of human brain microvascular
endothelial cells. Microb Pathog. 35:35–42. 2003.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Huang SH, Wan ZS, Chen YH, Jong AY and Kim
KS: Further characterization of Escherichia coli brain
microvascular endothelial cell invasion gene ibeA by deletion,
complementation, and protein expression. J Infect Dis.
183:1071–1078. 2001.PubMed/NCBI View
Article : Google Scholar
|
|
48
|
Wang MH and Kim KS: Cytotoxic necrotizing
factor 1 contributes to Escherichia coli meningitis. Toxins
(Basel). 5:2270–2280. 2013.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Koutsouras GW, Ramos RL and Martinez LR:
Role of microglia in fungal infections of the central nervous
system. Virulence. 8:705–718. 2017.PubMed/NCBI View Article : Google Scholar
|
|
50
|
van de Veerdonk FL, Kullberg BJ, van der
Meer JW, Gow NA and Netea MG: Host-microbe interactions: Innate
pattern recognition of fungal pathogens. Curr Opin Microbiol.
11:305–312. 2008.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Yang KY, Chang WN, Ho JT, Wang HC and Lu
CH: Postneurosurgical nosocomial bacterial brain abscess in adults.
Infection. 34:247–251. 2006.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Kielian T, Phulwani NK, Esen N, Syed MM,
Haney AC, McCastlain K and Johnson J: MyD88-dependent signals are
essential for the host immune response in experimental brain
abscess. J Immunol. 178:4528–4537. 2007.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Kielian T, Mayes P and Kielian M:
Characterization of microglial responses to Staphylococcus
aureus: Effects on cytokine, costimulatory molecule, and
Toll-like receptor expression. J Neuroimmunol. 130:86–99.
2002.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Kielian T, Esen N and Bearden ED:
Toll-like receptor 2 (TLR2) is pivotal for recognition of S.
aureus peptidoglycan but not intact bacteria by microglia.
Glia. 49:567–756. 2005.PubMed/NCBI View Article : Google Scholar
|