Introduction
An ectopic pregnancy (EP) represents the
implantation of the gestational sac outside the endometrium of the
uterine cavity, and is overall considered a public health problem,
judging by the high incidence number reported in recent decades and
by its potentially life-threatening medical emergency due to tubal
rupture. Previous findings revealed that in the UK the incidence of
EP is 11.5 per 1,000 pregnancies and the mortality is 0.4 per 1,000
EP (1): studies from the United
States showed the lowest and highest reported incidence,
respectively, to be 6.4 and 20.7 per 1,000 EP (2,3). The
main risk factors are represented by a previous EP, congenital
genital tract anomalies, tumors or history of tubal surgery that
distort the normal anatomy, inflammatory pelvic disease, tubal
sterilization, combined hormonal contraception or intrauterine
device, increasing age, cigarette smoking, assisted reproductive
techniques and other medical conditions related to the history and
treatment of infertility (4-7).
The most common implantation site of an EP is the fallopian tube
(96%); other anatomic sites are ovary, abdominal cavity,
endocervical canal, hysterotomy scar and the myometrium (8).
As a result of technological progress in
high-resolution transvaginal ultrasonography and in the sensitive
testing of beta subunit of human chorionic gonadotropin hormone
(β-hCG), the EP can be earlier diagnosed, avoiding further
complications and improving morbidity and mortality. An early
diagnosis of EP allows a more conservative resolution of the
pathology. The physician may choose between medical treatment of
the EP using methotrexate (MTX), surgical treatment or even
expectant management, in selected cases.
The clinical features of EP are represented by a
history of amenorrhea, pelvic pain and metrorrhagia (9). An EP may be diagnosed in 90% of cases
by systematic transvaginal examination corroborated with β-hCG
monitoring (10-15).
The ultrasonographic diagnosis of an EP is based on the
visualization outside the uterus of the gestational sac, which
contains a yolk sac or embryo with or without cardiac activity
(11). The most common finding
present in at least 89% of EP cases is represented by a complex
inhomogeneous extraovarian adnexal mass; another suggestive
ultrasonographic finding is described as a tubal ring which
represents an extraovarian adnexal mass incorporating an empty
gestational sac (11). There are
also findings that only suggest the presence of an EP, but they are
not diagnostic. Endometrial thickness can be an indicator of EP;
during normal intrauterine pregnancy the ultrasound shows a mean
endometrial thickness of 17.2 mm versus a mean of 12.7 mm in EP
(15). Douglas and Morrison's pouch
should also be inspected for fluid (15). β-hCG produced by the placental
syncytiotrophoblast cells is essential for the diagnosis of viable
pregnancy (16-18).
Intrauterine viable pregnancy is supported by the tendency of β-hCG
to double every 48-72 h; a suboptimal increase, below 66%, is
highly suggestive for an EP (19).
A β-hCG level between 1,000 and 3,500 mUI/ml, and the absence of a
gestational sac inside the uterine cavity, highly suggests an EP
(16). The differential diagnosis
should include miscarriage, implantation bleeding of an
intrauterine pregnancy, subchorionic hematoma, gynecologic
pathology (ovarian torsion, uterine fibroids, cervical polyps) and
gestational trophoblastic disease (19).
When diagnosing a tubal pregnancy (TP), the
treatment options depend on the clinical and paraclinical
evaluation of the patient, particularly on the hemodynamic status.
The treatment options of TP include an expectative approach with
monitoring of the β-hCG value trend, MTX therapy administered
intramuscularly, intravenously or injected directly into the
gestational sac and surgical treatment consisting of salpingostomy
or salpingectomy. The current approach is to avoid surgical
treatment and to preserve fertility whenever possible. MTX-based
therapy has become increasingly popular in recent years for the
medical treatment of TP, along with the close monitoring of the
β-hCG value dynamics (20,21). MTX therapy is also used in
combination with uterine artery embolization or supra-selective
embolization of the tubal branches of uterine and ovarian arteries
for the treatment of cervical pregnancies or caesarean scar
pregnancies, the depth of implantation of the gestational sac being
dependent on the location of the EP (21-23).
Because of the life-threatening potential, all cases
of diagnosed TP must be evaluated in order to establish the need
for emergency surgery (hemodynamically unstable patient and/or
suspicion of imminent or ongoing tubal rupture). If the case does
not require immediate surgery, the next step is the evaluation of
the β-hCG serum level. If the β-hCG serum value is above 5,000
mIU/ml, surgery is preferred. Furthermore, for patients with lower
β-hCG serum values it is mandatory to evaluate via ultrasonography
the presence or absence of embryonic cardiac activity; if present,
surgery is preferred (21,23). MTX therapy may be suggested if the
embryonic cardiac activity is absent and if there are no
contraindications for such therapy including hypersensitivity to
MTX, heterotopic pregnancy, breastfeeding, medical conditions such
as immunodeficiency, active pulmonary disease or peptic ulcer, or
extensive abnormalities in hematologic, hepatic or renal laboratory
tests (24,25). Concerning patients with renal
insufficiency, MTX having a renal clearance, one dose could cause
severe complications including acute respiratory distress, bone
marrow suppression, or bowel ischemia (26,27).
Finally, the personal preference of the patient should be
considered, as the patient should also be perfectly compliant with
close monitoring.
The criteria for expectant management of TP include
asymptomatic patients, low serum quantitative β-hCG values (<200
mIU/ml), pelvic ultrasound with no extra uterine mass and no extra
uterine gestational sac, compliant patient with the proposed
strategy, understanding the risks and clinical significance of an
EP, patients wanting to avoid both surgery or MTX therapy and
patients prepared to reach a medical unit if clinical conditions
worsened (28).
MTX is usually used as a cytostatic, as well as an
antineoplastic, immunosuppressive and anti-inflammatory agent,
which inhibits cell proliferation and protein synthesis by
suppressing the metabolism of purine bases and nucleic acids.
Tetrahydrofolic acid is required in the synthesis of purine bases
and nucleic acids and it is produced after the reaction between
dehydrofolate reductase and folic acid. MTX is a folic acid
analogue, which presents a similar structure and acts as an
antagonist by binding to dehydrofolate reductase, leading to lower
levels of tetrahydrofolic acid. MTX also affects cells with a high
rate of division (29). This
characteristic is used in the treatment of EP, active rheumatoid
arthritis, non-Hodgkin's lymphoma, breast cancer, cutaneous T-cell
lymphoma, gestational trophoblastic disease, desmoid tumors and
other neoplastic conditions (30-33).
The evaluation and treatment protocol, after the
positive diagnosis of a tubal pregnancy, includes several steps:
retreatment laboratory testing, ceasing any administration of folic
acid supplements, avoidance of non-steroidal anti-inflammatory drug
therapy, as well as physical activity and sexual intercourse,
repeated pelvic examinations due to possible tubal rupture, and sun
exposure to reduce the risk of MTX dermatitis (26). The next step involves selecting the
MTX therapy protocol: single dose, two-dose or multiple-dose
protocol. Single-dose protocol suggests a single intramuscular dose
of MTX, calculated as 50 mg/m2 of the body surface area
(34-37).
The β-hCG serum level is measured on day 1, 4 and 7. If the β-hCG
level decreases less than 15% between day 4 and 7, a second dose
should be administered (25,26,38,39);
this occurs in approximately 15-20% of cases. Further monitoring
involves weekly β-hCG serum level dosing, until an undetectable
level is reached. If the decline of β-hCG values is not favorable,
surgery should be performed. Two-dose protocol includes two MTX
doses of 50 mg/m2 offered on day 1 and 4. The β-hCG
serum level is tested on day 1 and 7 and should decrease more than
15%; if not, a third dose is administered, followed by retesting on
day 11. If the decrease is less than 15%, a fourth dose may be
offered, with retesting on day 14. If the decline is again not
reassuring, surgery should be performed (39). Multiple-dose protocol involves MTX
therapy administered on days 1, 3, 5 and 7 (1 mg/kg per day,
intramuscular) combined with 0.1 mg/kg leucovorin on days 2, 4, 6
and 8. In the single-dose protocol, leucovorin is not recommended.
The β-hCG level is monitored on days 1, 3, 5 and 7, and this level
is expected to decrease more than 15% from the previous testing.
After the treatment is finished, the monitoring phase is initiated
by weekly β-hCG testing until undetectable.
Thus, the aim of the present study was to highlight
the necessity for revising current guidelines for EP medical
treatment using MTX, in order to manage this pathology optimally
and to select carefully the proper treatment, whether medical,
surgical or expectant management, so that morbidity is reduced to a
minimum.
Patients and methods
Patients
We conducted a retrospective study, including 61
women aged between 17 and 46 years diagnosed with EP and treated
with MTX at the Department of Obstetrics and Gynecology of the
Bucharest University Emergency Hospital, during the period, January
2014-December 2020. The cases were analyzed by collecting data from
the Medical Statistics Department of the hospital, without
inclusion of personal information. The agreement of the ethics
committee (no. 17622/10.04.2018) was required and obtained from the
University Emergency Hospital of Bucharest without the need of
informed consent for study participation of the patient/legal
representative in the case of minors. In total, 509 patients
diagnosed with EP were analyzed during the mentioned period,
extracting from the database, the medically treated patients, who
were advocated in two groups: successful medically treated group
(42 patients) and unsuccessful medically treated group (19
patients). Conservatory treated patients were analyzed using
clinical and hemodynamic parameters, ultrasound evaluation and a
β-hCG seriated values trend aiming to identify the actual and
practical predictive profile of successful medically treated cases
of EP.
The inclusion criteria consisted of pregnant
patients without evidence of an intrauterine pregnancy on
transvaginal ultrasound, associated with visualization of an
extrauterine gestational sac with embryo or yolk sac visible on
TVUS, plus MTX administration after informed consent was obtained.
The exclusion criteria comprised ruptured ectopic pregnancies
exclusively surgically treated, renal, pulmonary and liver disease,
immunodeficiency, and concomitant intra- and extra-uterine
pregnancies.
Methods
Unit protocol for admitted hemodynamic stable
patients suspected of EP based on pelvic pain and/or scant brown
metrorrhagia, a routine pregnancy test and a transvaginal
ultrasound examination, included: clinical observation and caution
for pelvic-abdominal pain, metrorrhagia or faintness, blood
pressure and pulse monitoring, β-hCG trend, repeated transvaginal
ultrasound, complete blood count, liver and renal tests, blood type
and Rh, Human Immunodeficiency and hepatitis B and C virus
screening. The patients fulfilling the criteria for medical
treatment and requiring MTX administration underwent the single- or
multiple-dose protocols with further monitoring of the clinical
status, ultrasound evolution and β-hCG seriated values during
treatment as well as β-hCG monitoring after the protocol ended
until the β-hCG serum level became undetectable. All the patients
receiving MTX were informed of the risks involved and signed an
informed consent form, in order to receive the medication. Patients
with Rh incompatibility received an intramuscular dose of 300 mcg
of specific anti-D immunoglobulin. Patients with hemoperitoneum or
with decreasing β-hCG serum levels were excluded from the study.
Absolute contraindications of MTX treatment were considered:
hemodynamic instability, hemoperitoneum, intrauterine pregnancy,
hypersensitivity to MTX, severe medullary depression, active
pulmonary pathology, peptic ulcer, severe renal pathology, severe
liver pathology, or the impossibility of consistent monitoring of
β-hCG. Patients with contraindications for MTX therapy and those
who left the hospital contrary to medical opinion were excluded
from the study. The patients with relative contraindications
consisting in β-hCG serum level of over 5,000 mIU/ml, uterine tube
diameter ≥3.5 cm, present fetal cardiac activity or an embryo sac
larger than 4 cm were accepted for MTX therapy if they refused
laparoscopic management as a first line of treatment.
Statistical analysis
The data were collected using Office-Excel version
14.7.7. SPSS 12.0 (IBM Corp.) Pearson's correlation was used for
statistical analysis, and the statistical significance threshold
was set at P<0.05. The characteristics of each group were
calculated using ‘Descriptive statistics’.
Results
During the selected period, 509 patients were
diagnosed with extrauterine pregnancy, including 13 patients who
were discharged on request or left the hospital contrary to medical
advice from the first day of hospitalization, after a single
sampling of β-hCG. Medically treated patients were 61 (11.98%).
Patient supervision was performed using repeated dosing of β-hCG
serum levels every 48 h and also by using transvaginal ultrasound.
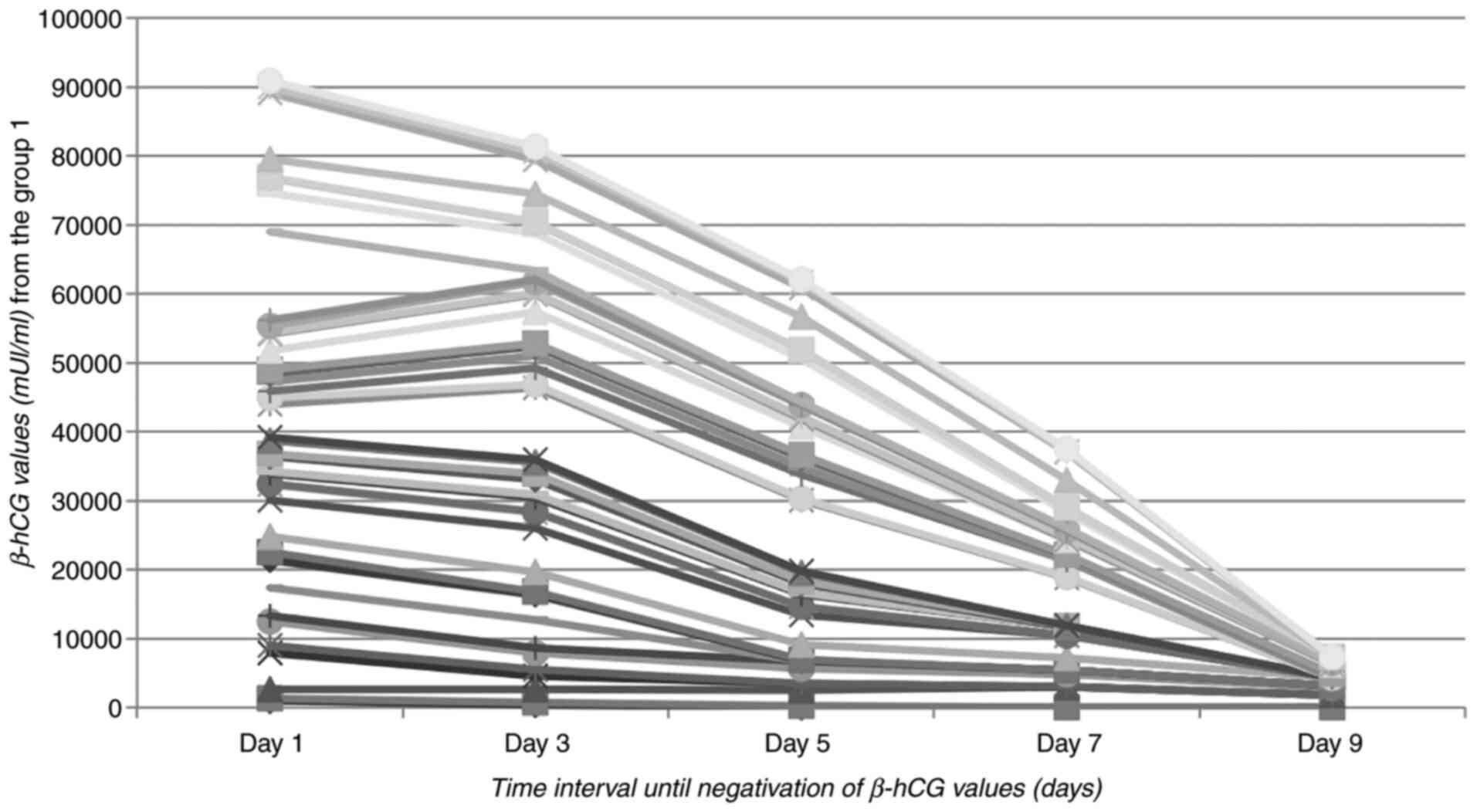
The MTX therapy was successful for 42 patients (68.85% of cases)
representing group 1 of our study, β-hCG serum levels decreasing
during hospitalization (Fig. 1). In
8 cases, a slight increase in the β-hCG value was observed, mainly
consisting of several tens of units in the first 48 h after the
administration of MTX, which led to its subsequent decrease
thereafter. The average period of hospitalization was 9 days, with
a minimum of 3 and a maximum of 22 days.
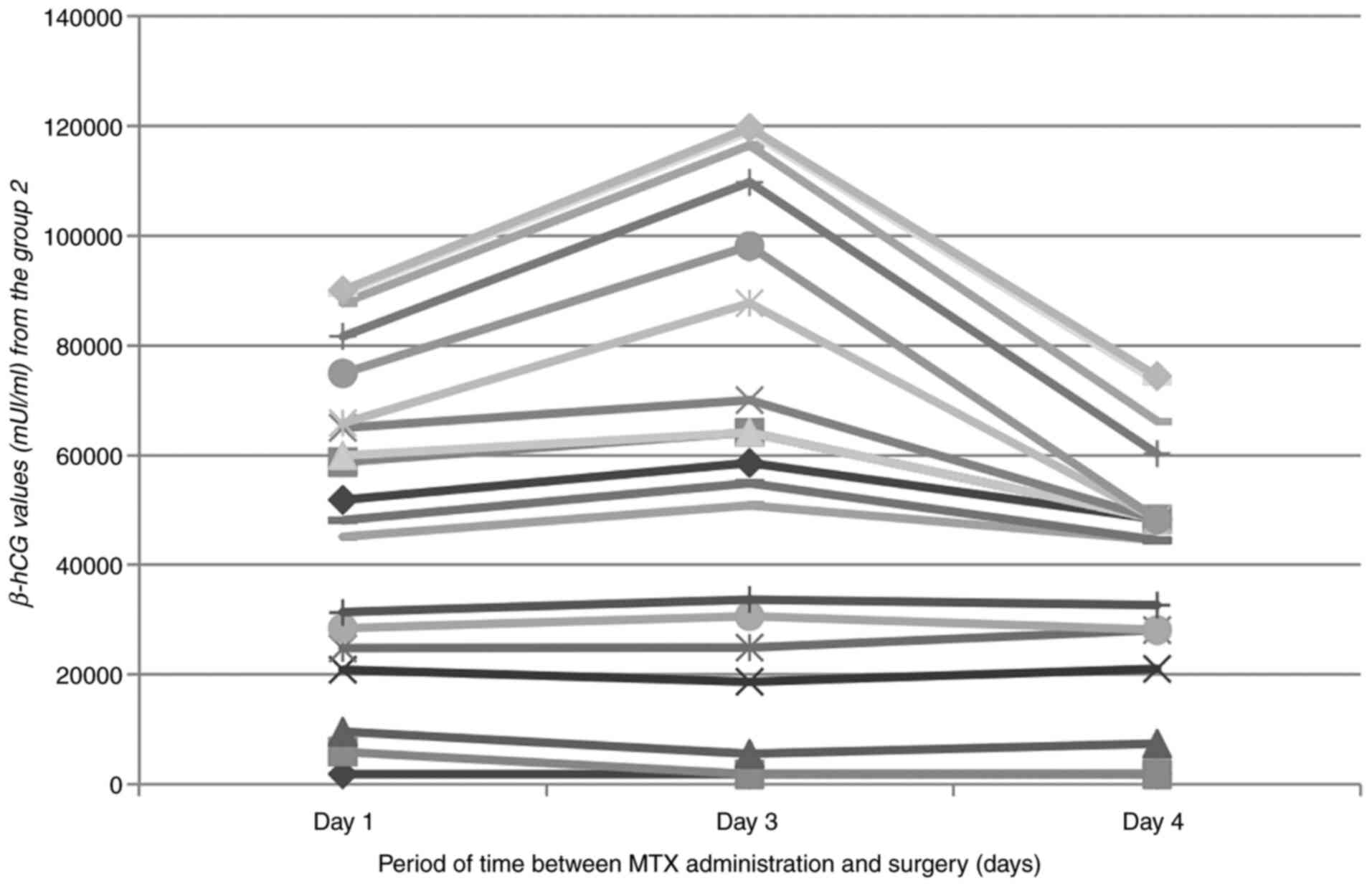
In the second group, including 19 cases of 61
medically treated (31.15%), MTX therapy failed and surgery was
necessary, as the β-hCG serum level increased or the patients'
symptomatology during hospitalization worsened, in most cases
complaining of acute pelvic pain. In 3 cases the patients who
received MTX were discharged, but returned 10-15 days later to the
emergency ward with hemoperitoneum (Fig. 2): one patient was discharged on
demand 48 h after MTX administration, with an insignificant β-hCG
decrease, from 1,908 to 1,846 mIU/ml (3.24% decrease), one patient
was also discharged on demand after a 48 h increase in β-hCG from
3,682 to 3,738 mIU/ml and another patient was discharged after a
decrease in β-hCG from 13,760 to 4,085 mIU/ml (total decrease of
70.3%). From the 19 cases needing surgery, 9 hospitalized patients
including the three patients that were discharged and returned with
acute pelvic pain and signs of peritoneal irritation, required
emergency surgery as the second line of therapy. In patients where
MTX therapy failed and hemoperitoneum was detected, marked
increasing of the β-hCG serum level was observed during repeated
determinations, except for the 2 cases described above (Fig. 2). At the time of hemoperitoneum
formation in the other patients, the β-hCG level was at an average
of 4,238 mIU/ml. In 10 cases, elective surgery was performed, due
to the maintenance or increase of the β-hCG value: 8 patients
underwent laparoscopic intervention and 2 patients had an
exploratory laparotomy performed. In the group of hemodynamically
stable patients, an average β-hCG value of 7,083 mIU/ml was
detected. Patients with β-hCG values under 2,000 mIU/ml had a
success rate of 82.7%, statistically significant correlation
between the initial β-hCG level (mIU/ml) and the group in which the
patient was included (P=0.003) (Fig.
1). The failures of medical therapy required emergency surgery
(14.75%) and elective surgery (16.39%).
Discussion
There are several studies revealing the success rate
of a single dose administration of MTX up to 94%, the
administration of a single intramuscular dose being better
tolerated by the patient and better assimilated rather than the
sequence of administration of smaller, serial doses (34,35,37-40).
The success rate of MTX is lower when embryonic
heartbeats are detected, correlating with a higher β-hCG serum
level (41). The most important
factor regarding the prediction of the MTX therapy success rate
represents the initial value of the β-hCG level (42). In our study, patients with values
<2,000 mIU/ml showed the best response, with higher values
positively correlated with being in-group 2, rather than in-group
1. There was no statistically significant correlation between the
age of the patients and the success of the conservative
therapy.
In the present study, the failure rate of MTX
treatment was 31.15%, with almost equal percentages of patients
requiring emergency surgery and elective surgery due to increased
β-hCG levels, even when two doses of MTX were administered
(difference not statistically significant). The high failure rate
compared to other studies was correlated with the β-hCG value
measured before starting the treatment, which depended on the time
frame of when the patient presented to the hospital. Patients
usually presented to the hospital when pelvic pain occurred and
required immediate care. MTX therapy was also attempted in patients
presenting higher β-hCG values, the success rate being of 50% in
patients with β-hCG values above 4,000 mIU/ml at the moment of the
healthcare unit admission.
Worldwide, the inpatient hospital treatment of EP
has decreased while multiple health care visits for a single EP
have increased. Taking into account that after MTX administration,
rigorous patient supervision is fundamental, with β-hCG monitoring,
clinical and hemodynamic parameters monitoring, and any complaints
of abdominal-pelvic pain, Kehr's sign (acute pain in the tip of the
shoulder), abnormal uterine bleeding, faintness, nausea,
tachycardia and blood pressure decrease should raise the suspicion
of intraperitoneal bleeding, we strongly recommend that each
country to adapt the management of surveillance of medically
treated EP cases upon the real promptitude and performance of its
own healthcare system. Due to the lack of specific guidance, low
level of medical education of some patients and the deficiencies in
informational system of the medical care, inpatient hospital
treatment of ectopic pregnancy until the β-hCG value has decreased
to under the life-threatening level is optimal.
MTX consistently targets rapidly dividing cells,
such as gastrointestinal tract cells, epidermal and hematopoietic
cells, which are most commonly affected. The side effects of MTX
administration consist of nausea, intestinal transit disorders,
fatigue, abdominal pain, pancytopenia, increased risk of infections
and dermatitis (43). High elevated
serum liver enzymes, chronic liver injury, cirrhosis and portal
hypertension may occur when given in high doses. As a result of
cellular destruction, an elevated uric acid concentration in
patients receiving MTX is expected. Breastfeeding is
contraindicated because MTX is distributed in breast milk (33). Due to the hepatotoxic effects, it is
imperative to screen for chronic viral hepatitis B and C and to
recommend reducing alcohol consumption to a minimum in the
immediate period following MTX administration. The patient should
be counseled to avoid excessive exposure to sunlight and UV lamps
for a period of 4 weeks, as there have been recorded cases of
dermatitis. Due to the risk of pancytopenia and the increased risk
of infection, we recommend the use of external menstrual pads and
the avoidance of intravaginal tampons in case of spotting or
vaginal bleeding.
In conclusion, the advantage of using MTX in
extrauterine pregnancies consists in surgery avoidance, with the
possibility of maintaining fertility and the prevention of
long-term complications of surgery such as adhesions that may cause
chronic pain and modify the quality of life. Medical management of
EP should be considered whenever a patient meets the criteria. The
success rate of MTX is correlated with lower initial β-hCG serum
levels, especially below 2,000 mIU/ml; MTX therapy efficiency
decreases with higher β-hCG serum values. At present, the suggested
superior cut-off β-hCG value proposed is 5,000 mIU/ml for the
medical treatment of EP in the majority of international
guidelines. Given our 50% success rate for ectopic pregnancies with
β-hCG values over 4,000 mIU/ml, we recommend revising the actual
criteria, thereby decreasing the superior cut-off value for optimal
future results. With the introduction of MTX therapy, the need for
surgery has been significantly reduced, sparing the unnecessary use
of resources, avoiding possible surgical complications, providing
comfort to the patients and reducing morbidity caused by the
surgery. In addition, medical therapy is less invasive, has a
diminished cost and presents a similar intrauterine pregnancy rate
for future pregnancies.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Any additional information concerning the study can
be requested from the corresponding author on reasonable
request.
Authors' contributions
CG, REB, and BMM conceived the article and wrote the
draft of manuscript. CG, DIM and TAG collected, analyzed and
interpreted the patient data regarding the methotrexate
administration for the treatment of ectopic pregnancy. REB, TAG and
BMM performed the literature search and conducted the follow-up of
the patients. CAZ and FF performed statistical analysis and study
description, making a substantial contribution to the conception of
the work and interpretation of data. CG, REB and DIM revised the
manuscript critically for important intellectual content. The
authenticity of all the raw data was assessed by CG, FF and DIM.
All authors have read and approved the final manuscript.
Ethics approval and consent to
participate
The present study was conducted in accordance with
the World Medical Association Declaration of Helsinki. The data
collected retrospectively did not contain personal information and
only the Ethics Committee agreement no. 17622/10.04.2018 of the
University Emergency Hospital of Bucharest was required and
obtained without the need of informed consent or the consent of the
patient/legal representative in the case of minors.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Tay JI, Moore J and Walker JJ: Ectopic
pregnancy. West J Med. 173:131–134. 2000.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Van Den Eeden SK, Shan J, Bruce C and
Glasser M: Ectopic pregnancy rate and treatment utilization in a
large managed care organization. Obstet Gynecol. 105:1052–1057.
2005.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Hoover KW, Tao G and Kent CK: Trends in
the diagnosis and treatment of ectopic pregnancy in the United
States. Obstet Gynecol. 115:495–502. 2010.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Bouyer J, Coste J, Shojaei T, Pouly JL,
Fernandez H, Gerbaud L and Job-Spira N: Risk factors for ectopic
pregnancy: A comprehensive analysis based on a large case-control,
population-based study in France. Am J Epidemiol. 157:185–194.
2003.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Malacova E, Kemp A, Hart R, Jama-Alol K
and Preen DB: Long-term risk of ectopic pregnancy varies by method
of tubal sterilization: A whole-population study. Fertil Steril.
101:728–734. 2014.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Nybo Andersen AM, Wohlfahrt J, Christens
P, Olsen J and Melbye M: Maternal age and fetal loss: Population
based register linkage study. BMJ. 320:1708–1712. 2000.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Bohîlțea RE, Zugravu CA, Neacsu A, Navolan
D, Berceanu C, Nemescu D, Bodean O, Turcan N, Baros Al and Cîrstoiu
MM: The prevalence of Vitamin D deficiency and its obstetrical
effects. A prospective study on Romanian patients. Rev Chim.
70:1228–1233. 2019.
|
|
8
|
Bouyer J, Coste J, Fernandez H, Pouly JL
and Job-Spira N: Sites of ectopic pregnancy: A 10 year
population-based study of 1800 cases. Hum Reprod. 17:3224–3230.
2002.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Belics Z, Gérecz B and Csákány MG: Early
diagnosis of ectopic pregnancy. Orv Hetil. 155:1158–1166.
2014.PubMed/NCBI View Article : Google Scholar : (In Hungarian).
|
|
10
|
Van Mello NM, Mol F, Ankum WM, Mol BW, Van
der Veen F and Hajenius PJ: Ectopic pregnancy: How the diagnostic
and therapeutic management has changed. Fertil Steril.
98:1066–1073. 2012.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Webster K, Eadon H, Fishburn S and Kumar
G: Guideline Committee. Ectopic pregnancy and miscarriage:
Diagnosis and initial management: Summary of updated NICE guidance.
BMJ. 367(I6283)2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Atri M, Leduc C, Gillett P, Bret PM,
Reinhold C, Kintzen G, Aldis AE and Thibodeau M: Role of
endovaginal sonography in the diagnosis and management of ectopic
pregnancy. Radigraphics. 16:755–774; discussion 775.
1996.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Dialani V and Levine D: Ectopic pregnancy:
A review. Ultrasound Q. 20:105–117. 2004.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Winder S, Reid S and Condous G: Ultrasound
diagnosis of ectopic pregnancy. Australas J Ultrasound Med.
14:29–33. 2011.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Moschos E and Twickler DM: Endometrial
thickness predicts intrauterine pregnancy in patients with
pregnancy of unknown location. Ultrasound Obstet Gynecol.
32:929–934. 2008.PubMed/NCBI View
Article : Google Scholar
|
|
16
|
Surampudi K and Gundabattula SR: The role
of serum beta hCG in early diagnosis and management strategy of
ectopic pregnancy. J Clin Diagn Res. 10:QC08–QC10. 2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Cole LA: Individual deviations in human
chorionic gonadotropin concentrations during pregnancy. Am J Obstet
Gynecol. 204(349. e1-7)2011.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Seeber BE: What serial hCG can tell you,
and cannot tell you, about an early pregnancy. Fertil Steril.
98:1074–1077. 2012.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Dogra V, Paspulati RM and Bhatt S: First
trimester bleeding evaluation. Ultrasound Q. 21:69–85; quiz 149-50,
153-4. 2005.PubMed/NCBI
|
|
20
|
Van Mello NM, Mol F, Adriaanse AH, Boss
EA, Dijkman AB, Doornbos JP, Emanuel MH, Friederich J,
Leeuw-Harmsen Lv, Lips JP, et al: The METEX study: Methotrexate
versus expectant management in women with ectopic pregnancy: A
randomised controlled trial. BMC Women's Health.
8(10)2008.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Kirk E, Condous G, Haider Z, Syed A, Ojha
K and Bourne T: The conservative management of cervical ectopic
pregnancies. Ultrasound Obstet Gynecol. 27:430–437. 2006.PubMed/NCBI View
Article : Google Scholar
|
|
22
|
Mesogitis S, Pilalis A, Daskalakis G,
Papantoniou N and Antsaklis A: Management of early viable cervical
pregnancy. BJOG. 112:409–411. 2005.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Timor-Tritsch IE, Monteagudo A, Santos R,
Tsymbal T, Pineda G and Arslan AA: The diagnosis, treatment, and
follow-up of cesarean scar pregnancy. Am J Obstet Gynecol. 207:44.
e1–13. 2012.PubMed/NCBI View Article : Google Scholar
|
|
24
|
American College of Obstetricians and
Gynecologists. ACOG Practice Bulletin No. 94: Medical management of
ectopic pregnancy. Obstet Gynecol. 111:1479–1485. 2008.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Practice Committee of American Society for
Reproductive Medicine. Medical treatment of ectopic pregnancy: A
committee opinion. Fertil Steril. 100:638–644. 2013.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Kelly H, Harvey D and Moll S: A cautionary
tale: Fatal outcome of methotrexate therapy given for management of
ectopic pregnancy. Obstet Gynecol. 107:439–441. 2006.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Teal SB: A cautionary tale: Fatal outcome
of methotrexate therapy given for management of ectopic pregnancy.
Obstet Gynecol. 107:1420–1; author reply 1421. 2006.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Korhonen J, Stenman UH and Ylöstalo P:
Low-dose oral methotrexate with expectant management of ectopic
pregnancy. Obstet Gynecol. 88:775–778. 1996.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Bleyer WA: The clinical pharmacology of
methotrexate: New applications of an old drug. Cancer. 41:36–51.
1978.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Kremer JM, Lawrence DA, Hamilton R and
McInnes IB: Long-term study of the impact of methotrexate on serum
cytokines and lymphocyte subsets in patients with active rheumatoid
arthritis: Correlation with pharmacokinetic measures. RMD Open.
2(e000287)2016.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Moura CS, Schieir O, Valois MF, Thorne C,
Bartlett SJ, Pope JE, Hitchon CA, Boire G, Haraoui B, Hazlewood GS,
et al: Treatment strategies in early rheumatoid arthritis
methotrexate management: Results from a prospective cohort.
Arthritis Care Res (Hoboken). 72:1104–1111. 2020.PubMed/NCBI View Article : Google Scholar
|
|
32
|
National Comprehensive Cancer Network
(NCCN). NCCN clinical practice guidelines in oncology. https://www.nccn.org/professionals/physician_gls
(Accessed on August 19, 2021).
|
|
33
|
National Center for Biotechnology
Information. PubChem Database. Methotrexate, 2021 https://pubchem.ncbi.nlm.nih.gov/compound/Methotrexate
(Accessed on 14 March 2021).
|
|
34
|
Lipscomb GH, Bran D, McCord ML, Portera JC
and Ling FW: Analysis of three hundred fifteen ectopic pregnancies
treated with single-dose methotrexate. Am J Obstet Gynecol.
178:1354–1358. 1998.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Barnhart KT, Gosman G, Ashby R and Sammel
M: The medical management of ectopic pregnancy: A meta-analysis
comparing ‘single dose’ and ‘multidose’ regimens. Obstet Gynecol.
101:778–784. 2003.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Morlock RJ, Lafata JE and Eisenstein D:
Cost-effectiveness of single-dose methotrexate compared with
laparoscopic treatment of ectopic pregnancy. Obstet Gynecol.
95:407–412. 2000.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Lipscomb GH: Medical therapy for ectopic
pregnancy. Semin Reprod Med. 25:93–98. 2007.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Stovall TG, Ling FW, Gray LA, Carson SA
and Buster JE: Methotrexate treatment of unruptured ectopic
pregnancy: A report of 100 cases. Obstet Gynecol. 77:749–753.
1991.PubMed/NCBI
|
|
39
|
Kirk E, Condous G, Van Calster B, Haider
Z, Van Huffel S, Timmerman D and Bourne T: A validation of the most
commonly used protocol to predict the success of single-dose
methotrexate in the treatment of ectopic pregnancy. Hum Reprod.
22:858–863. 2007.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Lipscomb GH, Stovall TG and Ling FW:
Nonsurgical treatment of ectopic pregnancy. N Engl J Med.
343:1325–1329. 2000.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Erdem M, Erdem A, Arslan M, Oç A,
Biberoglu K and Gursoy R: Single-dose methotrexate for the
treatment of unruptured ectopic pregnancy. Arch Gynecol Obstet.
270:201–204. 2004.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Liposcomb GH, McCord ML, Stovall TG, Huff
G, Portera SG and Ling TW: Predictors of success of methotrexate
treatment in women with tubal ectopic pregnancies. N Engl J Med.
341:1974–1978. 1999.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Alur-Gupta S, Cooney LG, Senapati S,
Sammel MD and Banhart KT: Two dose versus single dose of
methotrexate for treatment of ectopic pregnancy: A meta-analysis.
Am J Obstet Gynecol. 221:95–108.e2. 2019.PubMed/NCBI View Article : Google Scholar
|