Introduction
Chronic hepatitis B (CHB) mainly comprises a chronic
immune-mediated inflammatory injury to the liver as a result of
persistent hepatitis B virus (HBV) infection (1). Therefore, effective antiviral therapy
is crucial for viral clearance and the improvement of disease
outcomes. The current goals of treatment are to reduce the
occurrence of liver cancer and disease progression to cirrhosis
(2). Several segregating markers
have been adopted in the clinical setting to measure the treatment
response of hepatitis B e antigen (HBeAg)-positive patients who
have been receiving antiviral therapy. The markers include an
HBV-DNA decline to undetectable levels, transaminase normalization,
HBeAg and hepatitis B surface antigen (HBsAg) level decline
following treatment (2).
IL-21 is a type I cytokine that shares the common
γ-chain receptor subunit with the IL-2, IL-4, IL-7, IL-9 and IL-15
cytokines. It is produced by activated CD4+ T cells,
particularly follicular T helper (Tfh) cells, T helper 17 cells and
activated natural killer (NK) T cells (3-6).
IL-21 is considered a bridge between innate and adaptive immunity
with immune-enhancing and immune-regulatory effects on B-, T- and
NK-cell responses (7,8). IL-21 is derived from CD4+ T
cells and acts as an effective intermediate cytokine for the
optimal generation of specific CD8+ T- and B-cell
responses in the process of HBV infection. The expression of lL-21
is closely associated with HBV clearance (9-11),
clearance of HBV antigens (12-14),
occurrence of HBV-associated liver cirrhosis, liver failure and
liver cancer (15,16). During the follow-up period of
treatment-naïve HBeAg-negative patients with raised alanine
aminotransferase (ALT), the patients undergoing spontaneous HBsAg
seroconversion exhibited higher serum levels of IL-21 compared with
those of the non-convertors (17).
A previous study in HBeAg-positive CHB patients undergoing
telbivudine monotherapy indicated that serum IL-21 levels at
treatment week 12 were significantly higher in patients who
achieved HBeAg seroconversion. This marker was able to
independently predict HBeAg seroconversion in the first year of
treatment (12). In patients with
coexistence of HBeAg-positive CHB and nonalcoholic fatty liver
disease (NAFLD) receiving entecavir monotherapy, the elevation of
IL-21 levels from baseline to week 12 was significantly higher in
the early (24 weeks) virological response (EVR) group and the
elevated levels of IL-21 at treatment week 12 were able to predict
EVR in CHB+ NAFLD patients (18).
The virological response on nucleos(t)ide analogue
(NA) therapy is associated with the dynamic virus-host immune
interaction and the antiviral agents selected. Particularly in
resource-constrained settings, suboptimal virological response
(SOR), defined as detectable HBV DNA after 24-48 weeks of NA
monotherapy without resistance mutation, frequently occurs
(19). Patients with SOR on NA
monotherapy require an optimized strategy as salvage therapy.
However, to the best of our knowledge, the
association between the dynamic changes of IL-21 levels and the
treatment outcomes among patients with SOR to NAs who receive
salvage therapy has not been previously reported. Therefore, the
present study aimed to investigate whether IL-21 level changes at
treatment week 12 in these patients are able to predict HBeAg loss
or decline at week 104 of salvage therapy.
The major aims of the present study were the
following: i) To investigate the dynamic changes of IL-21 levels
during salvage therapy in patients with SOR; and ii) to assess the
association between IL-21 levels and treatment outcomes of salvage
therapy in patients with SOR.
Subjects and methods
Study subjects
A total of 24 patients with CHB who participated in
a multicenter randomized controlled clinical trial (ClinicalTrials.gov identifier: NCT01829685) for the
treatment of SOR to NAs at Beijing Ditan Hospital, Capital Medical
University (Beijing, China) were recruited for the present study
between April 2011 and April 2014. Additional informed consent was
obtained from these patients for the use of their clinical data,
for specimen collection and for the measurement of IL-21 levels.
The inclusion criteria were as follows: CHB with HBeAg-positive
status, HBV-DNA levels >1,000 IU/ml following 24 weeks of
monotherapy with telbivudine (LdT) or entecavir (ETV) at 0.5 mg
daily or 48 weeks of adefovir dipivoxil (ADV) monotherapy and
undetected antiviral resistant mutants. The patients were excluded
from the study if they had any of the following conditions:
Positivity for HCV, HDV or HIV, history of alcohol consumption
(alcohol consumption > 20 g per day for females or >30 g per
day for males), hepatic decompensation, presence of other liver
diseases or severe systemic diseases or administration of
immunomodulator therapy within 6 months.
Among the 24 enrolled subjects, 21 were on ADV, 2 on
LdT and 1 on ETV therapy at 0.5 mg daily. These patients randomly
received one of the three salvage oral regimens: ETV at 1.0 mg
daily, ETV at 1.0 mg plus ADV at 10 mg daily or ETV at 0.5 mg plus
ADV at 10 mg daily for 104 weeks. During the baseline and follow-up
visits at weeks 12, 24, 36, 52, 64, 76, 88 and 104, 2 3 ml of serum
was obtained from each subject and stored at -80˚C for measurement
of cytokine concentrations. In addition, blood samples were
collected for biochemical, serological and HBV-DNA assays.
At week 104 of salvage therapy, SOR patients were
classified into either the complete response (CR) group if they
achieved HBeAg loss and undetectable levels of serum HBV-DNA (the
lower limit of detection was 20 IU/ml) or the no complete response
(NCR) group if they remained HBeAg-positive independent of HBV-DNA
levels. In the NCR group, the patients were further divided into
two subgroups of virological response (VR) compared with no
virological response (NVR) based on their levels of viremia
(undetectable vs. detectable). For secondary assessment, the SOR
patients were classified into either a HBeAg level decline (ED)
group if the decline level of serum HBeAg (log10 S/CO)
from baseline to week 104 was 50% or more or a no HBeAg level
decline (NED) group if the decline level did not reach 50%.
Association of outcome measures with
IL-21 levels
The primary endpoint was the difference in IL-21
levels between the CR and NCR groups at week 12. The serum IL-21
levels and other baseline markers were assessed for predicting
complete response. Subsequently, within the NCR group, the IL-21
levels in the two subgroups of VR vs. NVR were compared. Secondary
endpoints included the comparison of IL-21 levels between the ED
and NED groups and the dynamic changes of IL-21 levels from
baseline to week 104.
Laboratory tests and assays
The liver function of the patients was measured
using the Hitachi 7600-020 full-automatic biochemical analyzer by
rate essay. HBV serological markers were detected using the
ARCHITECT i2000SR full-automatic immunoassay analyzer (Abbott
Laboratories) by chemiluminescence microparticle immunoassay. HBV
viral load was quantified using the Roche
Lightcycler®480 system by real-time quantitative PCR,
which had a detection limit of 20 IU/ml. The concentrations of
serum IL-21 were measured in duplicate using a commercial human
IL-21 ELISA kit (Mabtech) by the double antibody sandwich method.
The units of the IL-21 concentration were pg/ml.
Statistical analysis
Data analysis was conducted using SPSS 24.0 (IBM
Corp.). Continuous data were expressed as either the median
(10-90th percentile) or the mean ± standard deviation according to
whether the data satisfied the assumptions of normal distribution.
The independent-samples t-test (unpaired), the Mann-Whitney U-test
and the Chi-square test were used for comparisons between two
groups. One-way or two-way repeated-measures ANOVA was used for
comparisons of repeated sample data. One-way ANOVA was used for
univariate analysis. When the spherical data distribution
assumption was not satisfied, a multivariate test or
Greenhouse-Geisser correction were adopted. Bonferroni's post-hoc
test was used for multiple comparisons. Multi-way ANOVA and
logistic regression analysis were used to assess the predictive
value of each variable regarding the treatment response. Receiver
operating characteristic curves were constructed to identify the
optimal cut-off values for predicting treatment outcomes and for
calculating the sensitivity and specificity of the predictions
using these values. Spearman's rank order correlation coefficient
was determined to assess the correlation. All statistical analyses
were based on two-tailed hypothesis tests and P<0.05 was
considered to indicate statistical significance.
Results
Baseline characteristics
Following approval by the Institutional Review Board
(IRB) of Beijing Ditan Hospital, Capital Medical University
(Beijing, China; IRB no. NFEC-201011-K1), 24 subjects consented and
were enrolled in the study between April 2011 and April 2014. The
subjects of the SOR group included 21 patients on ADV, 2 on LdT and
1 on ETV at 0.5 mg daily. The subjects included 15 males and 9
females with a median age of 30.5 (range, 23.5-51.0) years. The
median levels of HBeAg and HBV DNA were 2.7 (0.2-3.1)
log10 S/CO and 5.2 (3.5-7.5) log10 IU/ml,
respectively. The baseline characteristics of the patients are
provided in Table I.
 | Table IBaseline clinical characteristics of
the study subjects assigned to different groups. |
Table I
Baseline clinical characteristics of
the study subjects assigned to different groups.
| Item | SOR (n=24) | CR (n=4) | NCR (n=20) | VR (n=11) | NVR (n=9) |
|---|
| Gender (M/F) | 15/9 | 2/2 | 13/7 | 5/6 | 8/1 |
| Age (years) | 30.5
(23.5-51.0) | 34.0
(30.0-40.5) | 29.0
(23.1-55.8) | 28.0
(20.6-58.6) | 32.0
(25.0-42.6) |
| Initial antiviral
therapy (ADV/LdT/ETV) | 21/2/1 | 3/1/0 | 18/1/1 | 11/0/0 | 7/1/1 |
| Salvage regimen
(A/B/C)a | 4/9/11 | 0/2/2 | 4/7/9 | 2/4/5 | 2/3/4 |
| HBV-DNA
(log10 IU/ml) | 5.2 (3.5-7.5) | 3.8 (3.6-4.4) | 5.3 (3.5-7.6) | 5.3 (3.6-7.8) | 6.0 (3.4-7.5) |
| ALT (ULN) | 0.9 (0.5-3.1) | 1.5 (0.6-2.4) | 0.8 (0.4-3.1) | 0.7 (0.3-3.4) | 0.8 (0.6-2.7) |
| TBil (µmol/l)
HBsAg | 11.8
(6.9-20.5) | 13.1
(8.6-14.7) | 11.2
(6.3-22.8) | 12.2
(6.6-30.2) | 10.2
(5.3-18.8) |
| (log10
IU/ml) HBeAg | 4.3 (3.1-4.8) | 3.4 (1.9-4.2) | 4.3 (3.3-4.8) | 3.8 (3.3-4.5) | 4.6 (4.4-4.9) |
| (log10
S/CO) | 2.7 (0.2-3.1) | 0.5 (0.04-2.4) | 2.7 (1.1-3.1) | 2.3 (0.3-2.9) | 2.9 (2.8-3.1) |
Changes in virological markers and
IL-21 concentration during treatment
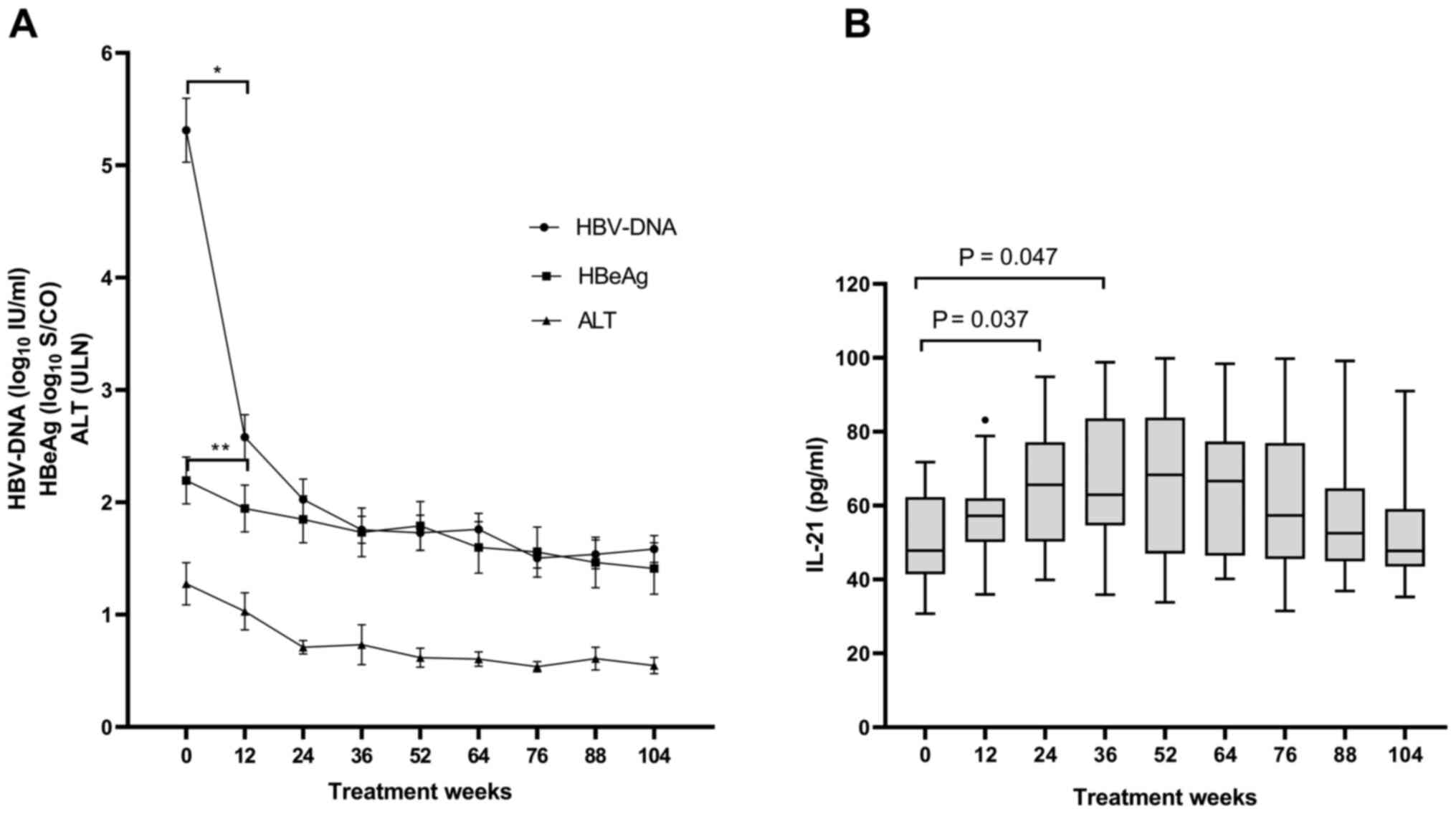
The changes in the serum HBV-DNA, HBeAg and ALT
levels in the patients who received salvage treatment for 104 weeks
are presented in Fig. 1A. Serum
HBV-DNA levels declined as the salvage treatment continued
(P<0.05) and the decline of the HBV-DNA levels during the
initial 12 weeks of treatment was the most significant (baseline,
5.3±1.4 log10 IU/ml vs. week 12, 2.6±1.0
log10 IU/ml; P<0.001). At week 104, 15 patients had
undetectable levels of serum HBV-DNA. In addition to the decline in
serum HBV-DNA levels, serum HBeAg levels declined accordingly
(P=0.037), particularly from baseline [2.7 (0.2-3.1)
log10 S/CO] to week 12 [2.3 (0.0-3.0) log10
S/CO] (P=0.01) and HBeAg loss occurred in 4 patients at week 104.
Mean ALT levels returned to normal by week 24.
Repeated-measures analysis of the IL-21 levels
indicated that the serum IL-21 levels were elevated from baseline
to week 36 (P=0.007) and subsequently, they were gradually reduced
to reach the baseline level again by week 104 (Fig. 1B). The mean IL-21 concentration at
week 24 (65.4±16.4 pg/ml) and week 36 (67.1±18.5 pg/ml) was
significantly higher than that at the baseline (50.4±12.2 pg/ml;
P=0.037 and 0.047, respectively). The serum IL-21 concentration
exhibited the most obvious increase during the initial 12-week
period of salvage treatment, which was consistent with the period
when serum HBV-DNA levels exhibited the greatest reduction.
Spearman's correlation analysis indicated that serum
IL-21 concentrations may exhibit a moderate negative correlation
with HBeAg levels (log10 S/CO) at baseline
(rs=-0.386; P=0.062) and at week 12
(rs=-0.392; P=0.058) (Fig.
S1). However, no significant correlation was noted between
serum IL-21 and serum ALT levels, or between serum IL-21 and serum
HBV DNA levels at any of the time-points assessed.
Association between IL-21
concentration and treatment response
Based on the treatment outcomes at week 104,
patients with SOR under salvage treatment were divided into the two
following groups: CR (n=4) and NCR (n=20). The latter included two
subgroups: VR (n=11) and NVR (n=9). No significant difference in
gender, age, initial antiviral therapy and salvage regimens was
identified between the CR and the NCR groups, as well as between
the VR and the NVR groups. The baseline characteristics of these
groups are summarized in Table I.
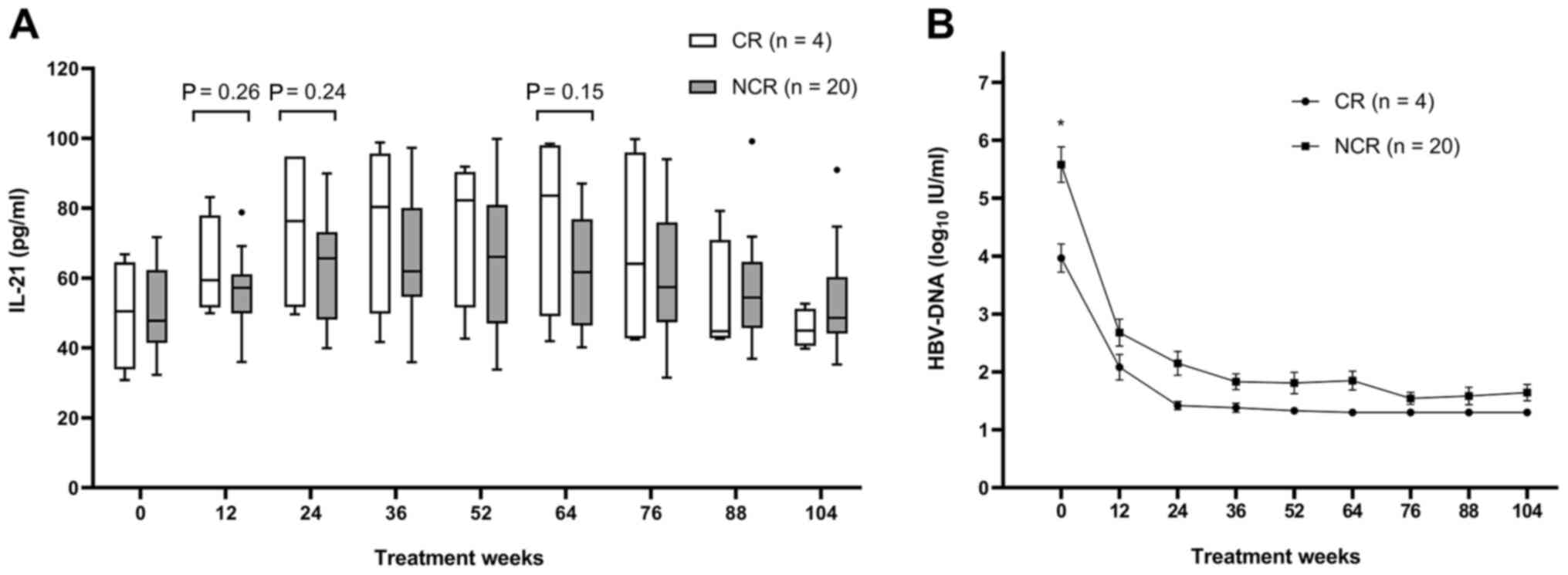
The changes in serum IL-21 and HBV-DNA levels of CR and NCR groups
over 104 weeks of treatment are presented in Fig. 2A and B. Although IL-21 levels appeared to be
higher in the CR group compared with those in the NCR group from
weeks 12 to 76, the differences exhibited no statistical
significance at any of the time-points of investigation. HBV-DNA
levels were significantly lower in the CR group compared with those
in the NCR group at baseline (P=0.032), but there were no
significant differences at other time-points.
Using univariate analysis, the influence of certain
parameters, including age, gender, initial antiviral therapy,
salvage regimens, serum HBV-DNA levels, serum ALT levels, serum
HBeAg levels and serum IL-21 concentrations on the achievement of
complete response was determined (Table II). No significant effect on the
treatment response was noted with regard to the use of the
different salvage regimens. Furthermore, the serum IL-21 levels at
week 12 did not differ between the CR and the NCR groups (63.0±14.4
vs. 55.9±10.5 pg/ml; P=0.26). All variables with a P<0.15 were
included in the multivariate analysis. The variables with the
strongest predictive value regarding CR included the HBeAg levels
at baseline (R2=0.25) and the HBV-DNA levels at baseline
(R2=0.16). Logistic regression analysis demonstrated
that the influence of baseline HBV-DNA on CR was not significant
(P=0.075). Following adjustment for the confounding factor
(baseline HBV-DNA), the independent effect of baseline HBeAg levels
became insignificant (P=0.110; Table
SI).
 | Table IIUnivariate analysis of factors
possibly associated with HBeAg loss. |
Table II
Univariate analysis of factors
possibly associated with HBeAg loss.
| Variable/treatment
week | CR (n=4) | NCR (n=20) | P-value |
|---|
| Age
(years)a | 34.0
(30.0-40.5) | 29.0
(23.1-55.8) | 0.347 |
| Male sex (n,
%)b | 2(50) | 13(65) | 0.615 |
| Initial antiviral
therapy (ADV/LdT/ETV)b | 3/1/0 | 18/1/1 | 0.437 |
| Salvage regimen
(A/B/C)b,c | 0/2/2 | 4/7/9 | 1.000 |
| HBV-DNA
(log10 IU/ml)d | | | |
|
0 | 3.8 (3.6-4.4) | 5.3 (3.5-7.6) | 0.032 |
|
12 | 2.1 (1.5-2.5) | 2.9 (1.3-4.1) | 0.276 |
|
24 | 1.4 (1.3-1.5) | 2.0 (1.3-3.4) | 0.137 |
| ALT
(ULN)a | | | |
|
0 | 1.5 (0.6-2.4) | 0.8 (0.4-3.1) | 0.477 |
|
12 | 1.3 (0.5-2.8) | 0.8 (0.4-1.9) | 0.157 |
|
24 | 0.8 (0.5-1.4) | 0.6 (0.4-1.1) | 0.273 |
| Baseline HBeAg
(log10 S/CO)a | 0.5 (0.04-2.4) | 2.7 (1.1-3.1) | 0.135 |
| IL-21
(pg/ml)d | | | |
|
0 | 49.6±15.9 | 50.5±11.9 | 0.900 |
|
12 | 63.0±14.4 | 55.9±10.5 | 0.262 |
|
24 | 74.3±23.9 | 63.6±14.7 | 0.243 |
|
12-0e | 13.3±9.8 | 5.4±13.5 | 0.282 |
Serum IL-21 levels appeared to be higher in the VR
group compared with those in the NVR group from baseline to week
36, but the differences were not significant. Serum IL-21 levels at
week 12 did not differ between the VR and NVR groups (58.4±12.2 vs.
52.9±7.6 pg/ml; P=0.26). However, the HBV-DNA levels were
significantly lower in the VR than those in the NVR group at week
12 (1.9±0.6 vs. 3.6±0.6 log10 IU/ml; P<0.001).
Predictors of serum HBeAg level
decline on salvage therapy
The decline of HBeAg levels at week 104 was present
in 7 patients of the ED group, and absent in 17 patients of the NED
group. In the ED group, the mean HBeAg level declined remarkably
from 1.63±1.34 to 0.06±0.49 log10 S/CO over 104 weeks of
salvage therapy (P=0.013). Furthermore, four of these patients
achieved HBeAg loss over 104 weeks. In addition, the changes in the
median HBeAg levels in the NED group ranged from 2.73 (0.87-3.10)
to 1.98 (0.70-2.97) log10 S/CO and none of the cases
exhibited any HBeAg loss (Fig. 3A).
At week 104, the proportion of HBeAg loss in the ED group was
significantly higher than that in the NED group (57.1% vs. 0;
P=0.003).
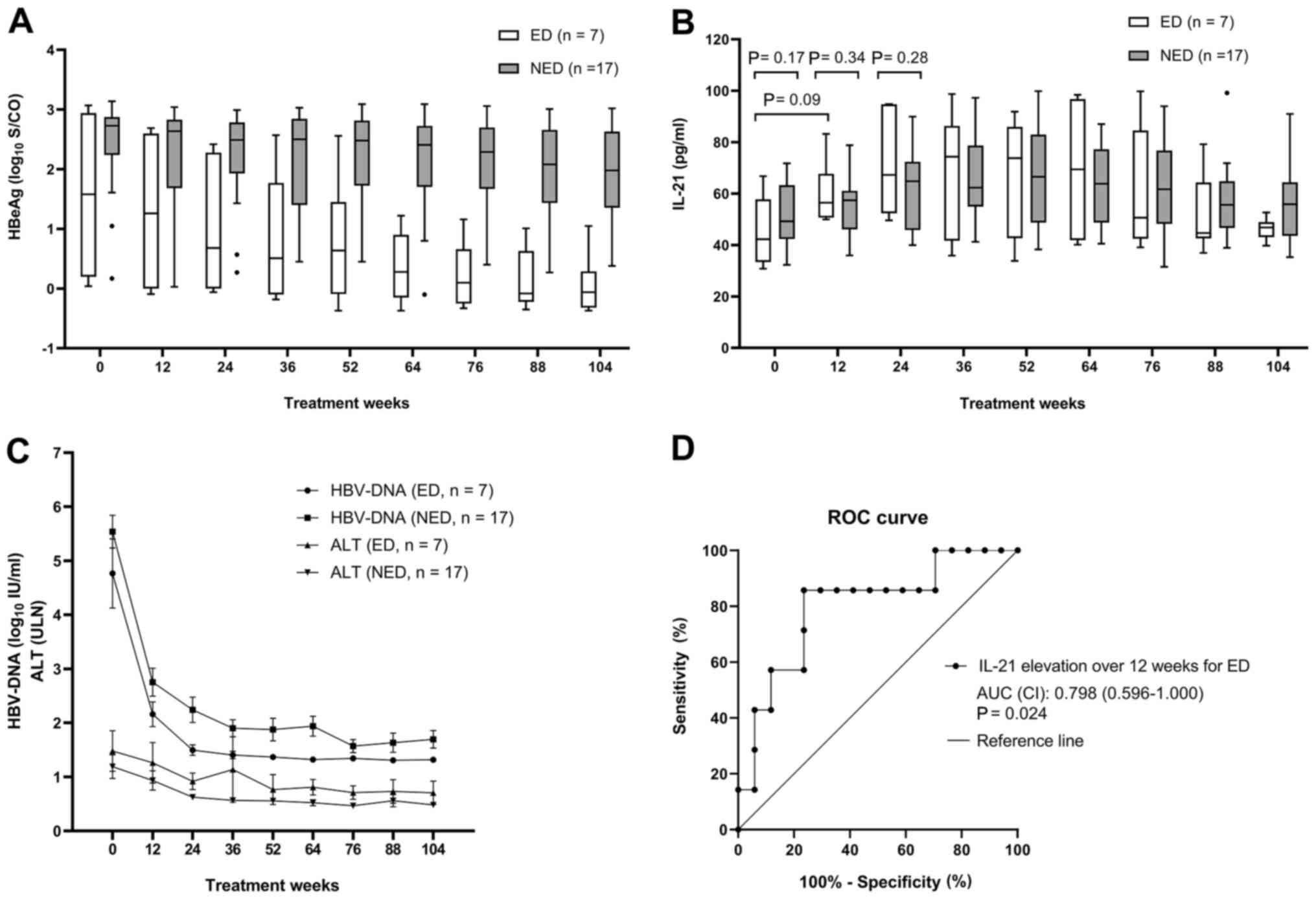 | Figure 3Prediction of serum HBeAg level
decline. (A) Comparison of the serum HBeAg levels (median, 25-75th
percentile, range) during 104 weeks of salvage therapy in the ED
and NED groups. (B) Comparison of the serum IL-21 concentrations
(median, 25-75th percentile, range) during 104 weeks of salvage
therapy in the ED and NED groups. (C) Comparison of the serum
HBV-DNA and ALT levels during 104 weeks of salvage therapy in the
ED and NED groups. (D) ROC curve indicating the influence of the
elevated levels of IL-21 over the initial 12 weeks of treatment on
the probability of HBeAg decline during 104 weeks of salvage
therapy. An AUC of 1.0 is indicative of a maximum diagnostic value,
whereas an AUC<0.5 is insufficient. Groups: ED, patients with a
decline in the serum level of HBeAg (log10S/CO) from
baseline to week 104 by ≥50%; NED, patients with a decline in the
serum level of HBeAg (log10S/CO) from baseline to week
104 by <50%. NED, no HBeAg level decline. The dots indicate
outlier; ROC, receiver operating characteristic; AUC, area under
the curve; HBV, hepatitis B virus; ALT, alanine aminotransferase;
ULN, upper limit of normal; HBeAg, hepatitis B e antigen. |
Repeated-measures analysis indicated that serum
IL-21 levels were elevated from baseline to week 36 and
subsequently, they decreased gradually until week 104 in both the
ED and the NED groups (Fig. 3B).
Although the difference in mean IL-21 levels between the ED and the
NED groups at week 12 was not statistically significant, the
increase of serum IL-21 levels from baseline to week 12 (IL-21
level at week 12 minus IL-21 level at baseline) was higher in the
ED group than that in the NED group (15.6±8.3 vs. 3.1±13.2 pg/ml;
P=0.03). When compared to the NED group, the HBV-DNA levels
appeared to be lower in the ED group and the ALT levels appeared to
be higher; however, none of these differences were significant
(Fig. 3C).
Univariate analysis was used to examine the
influence of age, gender (male vs. female), initial antiviral
therapy, salvage regimen, serum HBV-DNA levels, serum ALT levels,
serum HBeAg levels and serum IL-21 concentration on the probability
of HBeAg-decline (Table III). The
different initial antiviral therapies and salvage regimens had no
influence on treatment outcomes. The variable that was significant
in the univariate analysis was elevated levels of serum IL-21 from
baseline to week 12 (P=0.03). All variables with a P<0.10 were
included in the multivariate analysis. The variables with the
strongest predictive value on HBeAg decline included the elevated
levels of IL-21 over the initial 12 weeks of treatment
(R2=0.20) and the ALT levels at week 24
(R2=0.21). Following adjustment for confounding factors,
logistic regression analysis demonstrated that the elevation of
IL-21 has an independent predictive value for the decline of HBeAg
levels (odds ratio=1.137; R2=0.23; P=0.047; Table SII). A receiver operating
characteristic curve was generated to assess the use of IL-21
elevation over the initial 12 weeks to predict the HBeAg decline at
week 104 (Fig. 3D). The area under
ROC curve was 0.798 (CI, 0.596-1.000; P=0.024). The optimal cut-off
value for IL-21 elevation over 12 weeks was 12.6 pg/ml, at which
the sensitivity and specificity for predicting an HBeAg decline
were 85.7 and 76.5%, respectively. Elevated levels of IL-21 at 12
weeks by <12.6 pg/ml had a negative predictive value for HBeAg
decline of 92.9%.
 | Table IIIUnivariate analysis of factors
possibly associated with HBeAg-decline. |
Table III
Univariate analysis of factors
possibly associated with HBeAg-decline.
| Variable/treatment
week | ED (n=7) | NED (n=17) | P-value |
|---|
| Age
(years)a | 31.0
(28.4-48.8) | 28.0
(23.6-43.2) | 0.354 |
| Male sex (n,
%)b | 3 (42.9) | 12 (70.6) | 0.356 |
| Initial antiviral
therapy (ADV/LdT/ETV)b | 7/1/0 | 15/1/1 | 1.000 |
| Salvage regimen
(A/B/C)b,c | 1/2/4 | 3/7/7 | 0.840 |
| HBV-DNA
(log10 IU/ml)d | | | |
|
0 | 3.9 (3.6-6.8) | 5.3 (4.1-7.1) | 0.147 |
|
12 | 2.3 (1.5-2.8) | 2.9 (1.3-4.0) | 0.209 |
|
24 | 1.5 (1.3-1.8) | 2.1 (1.3-3.3) | 0.187 |
| ALT
(ULN)a | | | |
|
0 | 1.4 (0.6-2.7) | 0.8 (0.5-2.6) | 0.619 |
|
12 | 1.1 (0.5-2.2) | 0.8 (0.4-1.5) | 0.349 |
|
24 | 0.9 (0.5-1.4) | 0.6 (0.4-0.8) | 0.087 |
| Baseline HBeAg
(log10 S/CO)a | 1.6 (0.04-3.0) | 2.7 (0.9-3.1) | 0.418 |
| IL-21
(pg/ml)d | | | |
|
0 | 44.9±13.0 | 52.6±11.6 | 0.169 |
|
12 | 60.6±11.8 | 55.7±11.0 | 0.340 |
|
24 | 71.2±19.3 | 63.0±15.1 | 0.275 |
|
12-0e | 15.6±8.3 | 3.1±13.2 | 0.030 |
Discussion
The present study was the first prospective
longitudinal observation on serum IL-21 levels and virological
markers in patients with SOR receiving ETV with or without ADV as
the salvage therapy. The results indicated that the elevation of
the levels of IL-21 from baseline to week 12 was significantly
higher in those patients who achieved a decline of HBeAg at
treatment week 104 and that the elevation of IL-21 levels over the
initial 12 weeks of treatment had an independent effect on/was able
to predict HBeAg decline at week 104. In the present study, the
patients with SOR who presented with an apparent HBeAg decline over
104 weeks of salvage therapy were more likely to achieve HBeAg loss
(the proportion of HBeAg loss in the ED group and NED group was
57.1% vs. 0; P=0.003) and HBeAg seroconversion. Therefore, the
elevation of IL-21 levels at week 12 may be associated with immune
response and favorable treatment outcomes of salvage therapy in
patients with SOR. These results are consistent with those reported
in previous studies. Ma et al (12) observed that high serum levels of
IL-21 following 12 weeks of antiviral therapy predicted HBeAg
seroconversion in patients with CHB. Li et al (14) demonstrated that the HBV-specific
circulating chemokine C-X-C motif receptor 5 (CXCR5)+
CD4+ T cells (Tfh cells) were able to promote anti-HBe
production by autologous B cells via IL-21 in order to benefit
HBeAg seroconversion. All of these results implied that the
upregulation of serum IL-21 levels during the early stage of
antiviral treatment probably had a positive correlation with the
immune response required for HBeAg seroconversion and favorable
clinical outcomes. The study by Wu et al (20) further highlighted that IL-10 and
IL-12 were predictors of early spontaneous HBeAg seroconversion.
These studies suggested the utility of serum IL levels for
predicting disease outcomes in the clinic.
However, the results of the present study indicated
no association between IL-21 levels at treatment week 12 and the
clinical outcomes of HBeAg loss at treatment week 104, which
differed from the study of Ma et al (12). Although this discrepancy may be
attributed to the small sample size of the present study, it is
more likely due to the unique clinical features of the patients
with SOR to antiviral treatment. That these patients may have
exhibited differences in their immune status, since they had a
treatment history. For instance, the patients with SOR in the
present study exhibited relatively lower levels of HBV-DNA, ALT and
HBeAg compared with those of the treatment-naïve patients in the
study of Ma et al (12).
While the conclusions of the present studies are not in line with
those of the above study, the data provide important clinical
information for the management of patients with SOR. The present
study suggested that unlike those in the treatment-naïve
population, serum IL-21 levels in patients with SOR may not be used
as a predictor of HBeAg loss. The present results expanded the
current knowledge regarding patients with SOR receiving salvage
antiviral therapy. Giarda et al (21) demonstrated no association between
serum IL-21 levels and HBeAg seroconversion among patients with HBV
and HIV coinfection following antiviral treatment for both
diseases. IL-21 levels were considerably low during HBV-active
antiretroviral therapy and it was speculated that the immune
response associated with IL-21 was not the driving factor for HBeAg
seroconversion.
IL-21 is a cytokine that is mainly secreted by
activated CD4+ T cells and natural killer T cells.
Previous studies have indicated that IL-21 is able to promote the
proliferation of HBcAg-specific IFN-γ+ CD8+ T
cells, which is involved in the control of HBV replication
(10,11,22).
In addition, IL-21 is able to directly or, via CXCR5+
CD4+ T cells (Tfh cells), indirectly contribute to the
production of anti-HBe by B cells, which is associated with HBeAg
seroconversion (14,23,24).
Earlier studies have suggested discrepancies in the correlation
between serum IL-21 levels and HBV DNA, HBeAg and ALT levels
(11,12,15,22,25-27).
The present study indicated that the serum IL-21 levels at baseline
exhibited no correlation with serum HBV DNA or serum ALT levels.
However, IL-21 levels may exhibit a negative correlation with serum
HBeAg levels (rs=-0.386, P=0.062). These data support
the notion that IL-21 may have no direct involvement in the control
of HBV replication and liver inflammation, but they indicate the
possibility that IL-21 is associated with the immune response to
HBV. Such a response may contribute to an HBeAg level decline and
HBeAg seroconversion.
Serum IL-21 levels were upregulated from baseline to
week 36 of the salvage treatment, notably in the first 12 weeks of
treatment. During this period, serum HBV-DNA levels declined
significantly and serum ALT levels normalized in the majority of
the study subjects. The upregulation of IL-21 levels may suggest
that the defective HBV-specific immune response in patients with
SOR is partially recovered. Following week 36, the immune response
to HBV was gradually reduced, as the antiviral drugs further
suppressed HBV replication, resulting in a gradual decrease of the
IL-21 concentration to the baseline levels.
Although the present study was the first to
investigate the association between serum IL-21 levels and
treatment outcomes in a prospective cohort of patients with SOR,
certain limitations should be highlighted. IL-21 levels were only
measured in serum extracted from the peripheral blood, which may
not completely reflect IL-21 levels and immune reactions in the
liver. Future studies investigating IL-21 levels in liver tissues
are required. In addition, the sample size was small and additional
studies with a larger number of subjects are required to confirm
the present results. Finally, only a limited number of parameters
were assessed and it was not possible to measure T-cell specific
immune responses to HBV. As the immune reaction to HBV involves
complex interactions between varieties of immune cells and a broad
range of cytokines, further studies should involve more cytokines
associated with immune response. Furthermore, the studies should be
conducted at the immune cellular level. The mechanism of the role
of IL-21 in the immune response to HBV requires further
research.
In conclusion, serum IL-21 levels at treatment week
12 were associated with a decline in HBeAg levels at treatment week
104 in patients with SOR receiving antiviral treatment of ETV with
or without ADV. Elevated serum levels of IL-21 at treatment week 12
were an independent predictor for HBeAg decline at treatment week
104.
Supplementary Material
Correlation between the serum IL-21
concentrations and serum HBeAg levels (log10 S/CO) at
baseline and week 12.
Logistic regression analysis model for
variables associated with HBeAg loss.
Logistic regression analysis model for
variables associated with HBeAg decline.
Acknowledgements
Not applicable.
Funding
The present study was supported in part by the
National Science and Technology Major Project of China (grant no.
2018ZX10302206-003-006), Beijing Hospitals Authority Clinical
Medicine Development of Special Funding Support (grant no.
XMLX201837) and the Digestive Medical Coordinated Development
Center of Beijing Hospitals Authority (grant no. XXT26).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
HX and SL made substantial contributions to the
conception and design. YL, SJ, GY, JC, CQP and SL contributed to
the acquisition, analysis and interpretation of data. YL, CQP, HX
and SL were involved in drafting the manuscript and revising it
critically for important intellectual content. All authors read and
approved the final version of the manuscript to be published and
agreed to be accountable for all aspects of the work.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of the Beijing Ditan Hospital, Capital Medical University
(Beijing, China; no. NFEC-201011-K1) and written informed consent
was obtained from each patient.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Chang JJ and Lewin SR: Immunopathogenesis
of hepatitis B virus infection. Immunol Cell Biol. 85:16–23.
2007.PubMed/NCBI View Article : Google Scholar
|
|
2
|
European Association for the Study of the
Liver. Electronic address: simpleeasloffice@easloffice.eu;
European Association for the Study of the Liver. EASL 2017 Clinical
Practice Guidelines on the management of hepatitis B virus
infection. J Hepatol. 67:370–398. 2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Parrish-Novak J, Dillon SR, Nelson A,
Hammond A, Sprecher C, Gross JA, Johnston J, Madden K, Xu W, West
J, et al: Interleukin 21 and its receptor are involved in NK cell
expansion and regulation of lymphocyte function. Nature. 408:57–63.
2000.PubMed/NCBI View
Article : Google Scholar
|
|
4
|
Bryant VL, Ma CS, Avery DT, Li Y, Good KL,
Corcoran LM, de Waal Malefyt R and Tangye SG: Cytokine-mediated
regulation of human B cell differentiation into Ig-secreting cells:
Predominant role of IL-21 produced by CXCR5+ T follicular helper
cells. J Immunol. 179:8180–8190. 2007.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Wei L, Laurence A, Elias KM and O'Shea JJ:
IL-21 is produced by Th17 cells and drives IL-17 production in a
STAT3-dependent manner. J Biol Chem. 282:34605–34610.
2007.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Coquet JM, Kyparissoudis K, Pellicci DG,
Besra G, Berzins SP, Smyth MJ and Godfrey DI: IL-21 is produced by
NKT cells and modulates NKT cell activation and cytokine
production. J Immunol. 178:2827–2834. 2007.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Spolski R and Leonard WJ: Interleukin-21:
Basic biology and implications for cancer and autoimmunity. Annu
Rev Immunol. 26:57–79. 2008.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Mantellone G, Pallone F and Macdonald TT:
Interleukin-21 as a new therapeutic target for immune-mediated
diseases. Trends Pharmacol Sci. 30:441–447. 2009.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Li HJ, Kang FB, Li BS, Yang XY, Zhang YG
and Sun DX: Interleukin-21 inhibits HBV replication in vitro.
Antivir Ther. 20:583–590. 2015.PubMed/NCBI View
Article : Google Scholar
|
|
10
|
Zhong H, Xibing G, Yaping D, Zheng W,
Decai F, Xiaoye G, Hangyuan W, Dong W and Zhonghua L: Interleukin-7
in patients with chronic hepatitis B may have effect on T
follicular helper cells and specific cellular immunity. Hepat Mon.
16(e36068)2016.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Li L, Liu M, Cheng LW, Gao XY, Fu JJ, Kong
G, Feng X and Pan XC: HBcAg-specific IL-21-producing CD4+ T cells
are associated with relative viral control in patients with chronic
hepatitis B. Scand J Immunol. 78:439–446. 2013.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Ma SW, Huang X, Li YY, Tang LB, Sun XF,
Jiang XT, Zhang YX, Sun J, Liu ZH, Abbott WG, et al: High serum
IL-21 levels after 12 weeks of antiviral therapy predict HBeAg
seroconversion in chronic hepatitis B. J Hepatol. 56:775–781.
2012.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Publicover J, Goodsell A, Nishimura S,
Vilarinho S, Wang ZE, Avanesyan L, Spolski R, Leonard WJ, Cooper S
and Baron JL: IL-21 is pivotal in determining age-dependent
effectiveness of immune responses in a mouse model of human
hepatitis B. J Clin Invest. 121:1154–1162. 2011.PubMed/NCBI View
Article : Google Scholar
|
|
14
|
Li Y, Ma S, Tang L, Li Y, Wang W, Huang X,
Lai Q, Zhang M, Sun J, Li CK, et al: Circulating chemokine (C-X-C
Motif) receptor 5(+) CD4(+) T cells benefit hepatitis B e antigen
seroconversion through IL-21 in patients with chronic hepatitis B
virus infection. Hepatology. 58:1277–286. 2013.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Wu X, Su Z, Cai B, Yan L, Li Y, Feng W and
Wang L: Increased circulating follicular regulatory T-like cells
may play a critical role in chronic hepatitis B virus infection and
disease progression. Viral Immunol. 31:379–388. 2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Chen HM, Liu HL, Yang YC, Cheng XL, Wang
YF, Xing FF and Zhao YR: Serum IL-21 levels associated with chronic
hepatitis B and hepatitis B-related liver failure. Exp Ther Med.
7:1013–1019. 2014.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Vyas AK, Sharma BC, Sarin SK and
Trehanpati N: Immune correlates of hepatitis B surface antigen
spontaneous seroconversion in hepatitis B e antigen negative
chronic hepatitis B patients. Liver Int. 38:38–49. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Liu X, Shen Z, Zhang H, Liang J and Lin H:
Interleukin-21 is associated with early antiviral response in
patients with hepatitis B e antigen-positive chronic hepatitis B
and nonalcoholic fatty liver disease. J Interferon Cytokine Res.
36:367–373. 2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Lai CL, Gane E, Liaw YF, Hsu CW,
Thongsawat S, Wang Y, Chen Y, Heathcote EJ, Rasenack J, Bzowej N,
et al: Telbivudine versus lamivudine in patients with chronic
hepatitis B. N Engl J Med. 357:2576–2588. 2007.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Wu JF, Wu TC, Chen CH, Ni YH, Chen HL, Hsu
HY and Chang MH: Serum levels of interleukin-10 and interleukin-12
predict early, spontaneous hepatitis B virus e antigen
seroconversion. Gastroenterology. 138:165–172.e1-e3.
2010.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Giarda P, Avihingsanon A, Sasadeusz J,
Audsley J, Marks P, Matthews G, Ruxrungtham K, Lewin SR and Crane
M: CXCL-10, interleukin-12 and interleukin-21 are not immunological
predictors of HBeAg seroconversion in HIV-1-HBV coinfection
following HBV-active antiretroviral therapy. Antivir Ther.
19:429–433. 2014.PubMed/NCBI View
Article : Google Scholar
|
|
22
|
Li J, Ren W, Ma W, Zhang J, Shi J and Qin
C: Interleukin-21 responses in patients with chronic hepatitis B. J
Interferon Cytokine Res. 35:134–142. 2015.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Crotty S: Follicular helper CD4 T cells
(Tfh). Annu Rev Immunol. 29:621–663. 2011.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Ozaki K, Spolski R, Feng CG, Qi CF, Cheng
J, Sher A, Morse HC III, Liu C, Schwartzberg PL and Leonard WJ: A
critical role for IL-21 in regulating immunoglobulin production.
Science. 298:1630–1634. 2002.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Hu TT, Song XF, Lei Y, Hu HD, Ren H and Hu
P: Expansion of circulating TFH cells and their associated
molecules: Involvement in the immune landscape in patients with
chronic HBV infection. Virol J. 11(54)2014.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Pan Q, Yu Y, Tang Z, Xi M, Jiang H, Xun Y,
Liu X, Liu H, Hu J and Zang G: Increased levels of IL-21 responses
are associated with the severity of liver injury in patients with
chronic active hepatitis B. J Viral Hepat. 21:e78–e88.
2014.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Xing T, Xu H and Yu W: Role of T
follicular helper cells and their associated molecules in the
pathogenesis of chronic hepatitis B virus infection. Exp Ther Med.
5:885–889. 2013.PubMed/NCBI View Article : Google Scholar
|