Introduction
Subarachnoid hemorrhage (SAH), primarily caused by a
ruptured aneurysm, is a life-threatening neurological disease,
accounting for 5% of all stroke types (1). The incidence of SAH is 8-9 per 100,000
individuals (2). Despite
significant improvements in the diagnosis and surgical treatment of
SAH, 25% of patients succumb to the disease, and 50% of survivors
suffer significant disabilities (3,4).
Previous clinical and experimental evidence supports
the hypothesis that inflammatory factors are the basis of vascular
constriction and brain damage associated with SAH (5,6).
Jedrzejowska-Szypulka et al (7) observed that increased levels of
interleukin-1β (IL-1β) can cause nerve toxicity and brain edema by
destroying the blood-brain barrier (BBB). A previous study revealed
that IL-6 and IL-8 levels were increased following SAH (8). Furthermore, inhibition of tumor
necrosis factor-α (TNF-α) was demonstrated to decrease apoptosis in
the hippocampus following SAH (9).
As inflammation is a key factor involved in the
pathology of SAH, the identification of a novel cytokine involved
in inflammation may aid in the detailed characterization of the
mechanisms of SAH.
Mitogen and stress-activated protein kinase 1 (MSK1)
is a nuclear protein kinase (10)
that contains 2 kinase domains: A C-terminal kinase domain
associated with the Ca2+/calmodulin-dependent kinase
family and an N-terminal kinase domain associated with the
cAMP-dependent, cGMP-dependent and protein kinase C kinase family.
It was originally identified through its homology to the N-terminal
ribosomal S6 kinase domain (11).
MSK1 is involved in the cAMP/cAMP dependent protein kinase
(PKA)/cAMP responsive element binding protein 1 (CREB) pathway and
the activation of NF-κB, which are key mediators in the
transcription of genes involved in inflammatory responses (12). Several studies have demonstrated
that MSK1 serves a positive role in the regulation of the activity
of pro-inflammatory transcription factors implicated in asthma
(13), and in vitro data
suggest that MSK1 is involved in negative feedback pathways that
are critical in preventing uncontrolled inflammation in macrophages
(14). In addition, MSK1 may be
implicated in the anti-inflammatory properties of glucocorticoids
through its translocation from the nucleus to the cytoplasm
(15).
Despite evidence of a correlation between MSK1
expression and regulation of inflammation, specific genetic
evidence supporting the role of MSK1 in central nervous system
(CNS) inflammation following SAH has not been reported to date.
Therefore, we hypothesized that MSK1 may be closely
associated with inflammation during the process of SAH, and the
present study aimed to identify the potential role of MSK1 in
inflammation and subsequent brain damage development following
SAH.
Materials and methods
Animals and surgical procedures
In the animal model, a total of 60 Sprague-Dawley
rats (age, 3 months; weight, 280-320 g) were obtained from the
Shandong Experimental Animal Center. All animals were maintained on
a 12-h light/dark cycle with ad libitum access to water and
food and the animal room had a temperature of 20-25˚C and a
relative humidity of 45-50%. The SAH model was established as
described previously (9). In the
study group, the death rate was 0.
The animals were anesthetized with intraperitoneal
(IP) injection of 10% chloral hydrate (400 mg/kg). Then, the
animals were placed in a stereotactic frame with the head tilted
down at ~30˚. A midline scalp incision was made in the neck and the
atlanto-occipital membrane was exposed upon separating the
underlying muscle. Then, the membrane was punctured with a 27-gage
needle, and 0.3 ml autologous femoral arterial blood was injected
into the cisterna magna within 10 min with an infusion pump.
Animals in the sham group were injected with 0.3 ml sterile saline.
Rats had ad libitum access to water and food upon recovery
from anesthesia. The animals with SAH were randomly divided into 7
subgroups and sacrificed by decapitation prior to anesthesia with
10% chloral hydrate (400 mg/kg; IP) at 2 h, 6 h, 12 h, day 1, day
3, day 5 and day 7 post-SAH (n=6). Sham animals (n=6) experienced
the same surgery but without injection of blood into the cisterna
magna. Consequently, the sham animals were sacrificed 24 h
following the sham surgery. No signs of peritonitis such as
decreased food intake and abdominal swelling were observed
following the administration of 10% chloral hydrate. Death was
verified using the following criteria: Confirmation of the absence
of visible breathing and measurable heartbeat, confirmation of
pupil dilation and absence of the tail pinch reflex. The animal use
and care protocols, including all surgical procedures, were
approved by the Animal Care and Use Committee of Taishan Medical
University and conformed to the Guide for the Care and Use of
Laboratory Animals by the National Institute of Health (16).
Hematoxylin and eosin (H&E)
staining
Microscope slides containing cryosections of tissue
were fixed in 100% alcohol for 5 mins and then immersed in
H2O for 30 sec with manual agitation. The slides were
placed in a Coplin jar containing Mayer's hematoxylin and agitated
for 30 sec. Following rinsing of the slides in H2O for 1
min and staining with 1% eosin Y solution for 10-30 sec, the
sections were dehydrated with 2 washes in 95% experimental ethanol
and 2 washes in 100% experimental ethanol for 30 sec each. The
alcohol was extracted with 2 washes in xylene, and 1-2 drops of
mounting medium were then added to the slides, which were covered
with a coverslip. All steps were at room temperature. The stained
slides were examined with a light microscope (Leica Microsystems,
Inc.) at x100 and x400 magnification.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
TRIzol® Reagent (Thermo Fisher
Scientific, Inc.) was used to isolate total RNA from rat tissues,
and the RNA concentration was determined by spectrophotometric
analysis (optical density 260/280). cDNA was synthesized with
Reverse Transcriptase reagent (Takara Biotechnology Co., Ltd.).
RT-qPCR was performed using Stratagene Mx3000P qPCR system (Agilent
Technologies, Inc.) in a reaction that contained 2 µl cDNA, 12.5 µl
SYBR-Green (Takara Biotechnology Co., Ltd.), 1 µl each forward and
reverse primers (10 µM) and RNase-free H2O to a final
volume of 25 µl. The following primers were used: MSK1 forward (FW)
5'-CCTCAAGAC CCCATGCTTCA-3'; MSK1 reverse (REV) 5'-ACTTCTGTC
ATGGGACTGGA-3'; TNF-α FW 5'-TGCCTATGTCTCAGC CTCTTC-3'; TNF-α REV
5'-GAGGCCATTTGGGAACTT CT-3'; IL-1β FW 5'-TGAGCACCTTCTTTTCCTTCA-3';
IL-1β REV 5'-TTGTCTAATGGGAACGTCACAC-3'; GAPDH FW
5'-GAGGCCGGTGCTGAGTATGT-3'; and GAPDH REV
5'-GGTGGCAGTGATGGCATGGA-3'. The thermocycling conditions used to
perform the qPCR were as listed: Initial denaturation at 95˚C for
30 sec, followed by 40 cycles of denaturation at 95˚C for 5 sec and
annealing at 60˚C for 30 sec. The expression levels of target genes
were evaluated by using the 2-ΔΔCq method (17). All samples were analyzed in
triplicate.
Double immunofluorescent staining
Immunofluorescent staining was performed according
to our previous study (18). Brain
tissue was fixed in 4% paraformaldehyde for 3 h at 4˚C and then
incubated in 20% saccharose for 2 days at 4˚C, followed by
incubation in 30% sucrose for an additional 2 days at 4˚C to remove
the H2O content. Sections measuring 8 µm in thickness
were prepared and blocked with 5% normal fetal bovine serum in PBS
(Sigma-Aldrich; Merck KGaA) containing 0.1% Triton X-100 for 2 h at
room temperature. The slices were then incubated overnight at 4˚C
with primary antibodies against MSK1 (cat. no. ab81294; 1:200;
Abcam), TNF-α (cat. no. ab13597; 1:200; Abcam), IL-1β (cat. no.
ab9722; 1:100; Abcam) and glial fibrillary acidic protein (GFAP;
cat. no. sc7114; 1:100; Santa Cruz Biotechnology, Inc.). On the
following day, the corresponding secondary antibodies (cat. nos.
sc-2030 and sc-2354; 1:100; Santa Cruz Biotechnology, Inc.) were
added and incubated for 2 h at room temperature in the dark.
Following 3 washes in PBS, the slides were covered with a
microscopic glass coverslip and observed under a Leica fluorescence
microscope at x100 and x400 magnification (Leica Microsystems
Inc.). Negative controls were prepared by omitting the primary
antibodies.
Cell culture and treatment
Primary astrocytes were obtained from the cerebral
cortex of 10 neonatal (2 day-old) male Sprague-Dawley rat pups
obtained from Shandong Experimental Animal Center. The pups were
decapitated and their brains were rapidly removed before the
cerebral cortices were trypsinized and dissociated by trituration.
The dissociation mixture was plated at a density of
5x107 cells per 75 cm2 flask in Dulbecco's
modified Eagle's medium (DMEM) with F12 nutrient (1:1; Gibco;
Thermo Fisher Scientific, Inc.). Cells were maintained in complete
culture medium for 7-8 days. Prior to experimental treatments,
cultures of astrocytes were passaged twice. The culture medium was
exchanged for serum-free DMEM/F12 and experiments were initiated 24
h later. To assess the effect of lipopolysaccharide (LPS), cells
were allowed to reach 80% confluence. At the time of treatment, 1
µg/ml LPS was added to the culture medium. Cells were stimulated
with LPS for 24 h. The reagents were administered directly to the
growth medium for 48 h. Non-treated cells were included as controls
in all experiments.
Transfection
Astrocytes were seeded at a density of
2x105 cells/ml in a 6-well plate prior to transfection.
After 24 h, 10 µl Lipofectamine 2000 reagent in 450 µl
Opti-MEM® (Invitrogen; Thermo Fisher Scientific, Inc.)
and 500 µl transfection mixture containing 4 mg enhanced green
fluorescent protein (EGFP)-N3-MSK1 plasmid (or EGFP-N3 mock plasmid
as control) were added to each well. The cells were incubated for
48 h after transfection and then harvested for experiments. The
experiments were repeated ≥3 times. Plasmids were synthesized and
donated by the Nerve Regeneration Laboratory of Nantong University.
The EGFP-N3-MSK-1 contained the full sequence of MSK-1, while the
control plasmid contained the insert
5'-TACAAGTAAAGCGGCCGCGACT-3'.
Western blot analysis
For western blot analysis, the cells were washed
twice in ice-cold PBS and then homogenized in lysis buffer (1%
sodium deoxycholate, 50 mmol/l Tris, 1% NP-40 Lysis Buffer), 1%
Triton X-100, 5 mmol/l EDTA, 1% SDS, 1 mmol/l phenylmethylsulfonyl
fluoride 10 IU/ml aprotinin and 1 IU/ml leupeptin). The mixture was
collected with a cell scraper. After centrifugation at 14,000 x g
for 15 min at 4˚C, the supernatant was collected. Then, a BCA kit
(Thermo Fisher Scientific, Inc.) was used to determine the protein
concentration. The samples were boiled for 5 min at 100˚C upon
adding SDS sample buffer. Samples (80 µg/lane) were subjected to
electrophoresis in 10% SDS-PAGE gels for 40 min at 70 V followed by
90 min at 120 V, and then transferred onto polyvinylidene
difluoride membranes for 2.5 h at 180 mA. The membranes were
blocked with 5% non-fat milk for 2 h at room temperature and then
incubated with the appropriate primary antibodies MSK1 (cat. no.
ab81294; 1:200; Abcam), TNF-α (cat. no. ab13597;1:200; Abcam),
IL-1β (cat. no. ab9722; 1:100; Abcam), β-actin (cat. no. sc-81178;
1:100; Santa Cruz Biotechnology, Inc.) at 4˚C overnight. The
membrane was then washed with PBS + 1% Tween-20 3 times and
incubated with the appropriate horseradish peroxidase-conjugated
secondary antibody (cat. no. A-16078; 1:2,000; Pierce; Thermo
Fisher Scientific, Inc.) for 2 h at room temperature. The blotted
protein bands were developed using ECL Chemiluminescent Substrate
Reagent kit (Thermo Fisher Scientific, Inc.) and exposed to X-ray
film. The films were estimated using a Molecular Dynamics
densitometer (Scion Corporation). The relative protein expression
levels were normalized to β-actin and quantified by Quantity One
software (Version 4.6.6; Bio-Rad Laboratories, Inc.) through
measuring the relative band density.
Statistical analysis
All values are expressed as means ± standard error
of the mean and each experiment was repeated ≥3 times. SPSS v.21.0
(IBM Corp.) was used for statistical analysis of the data.
Statistical evaluation was processed by two-tailed Student's t-test
between two groups. Statistical evaluation of multiple groups was
performed using a one-way analysis of variance followed by
Dunnett's post hoc test. P<0.05 was considered to indicate a
statistically significant difference. The correlations between
MSK1, IL-1β and TNF-α were determined by Spearman's analysis.
Results
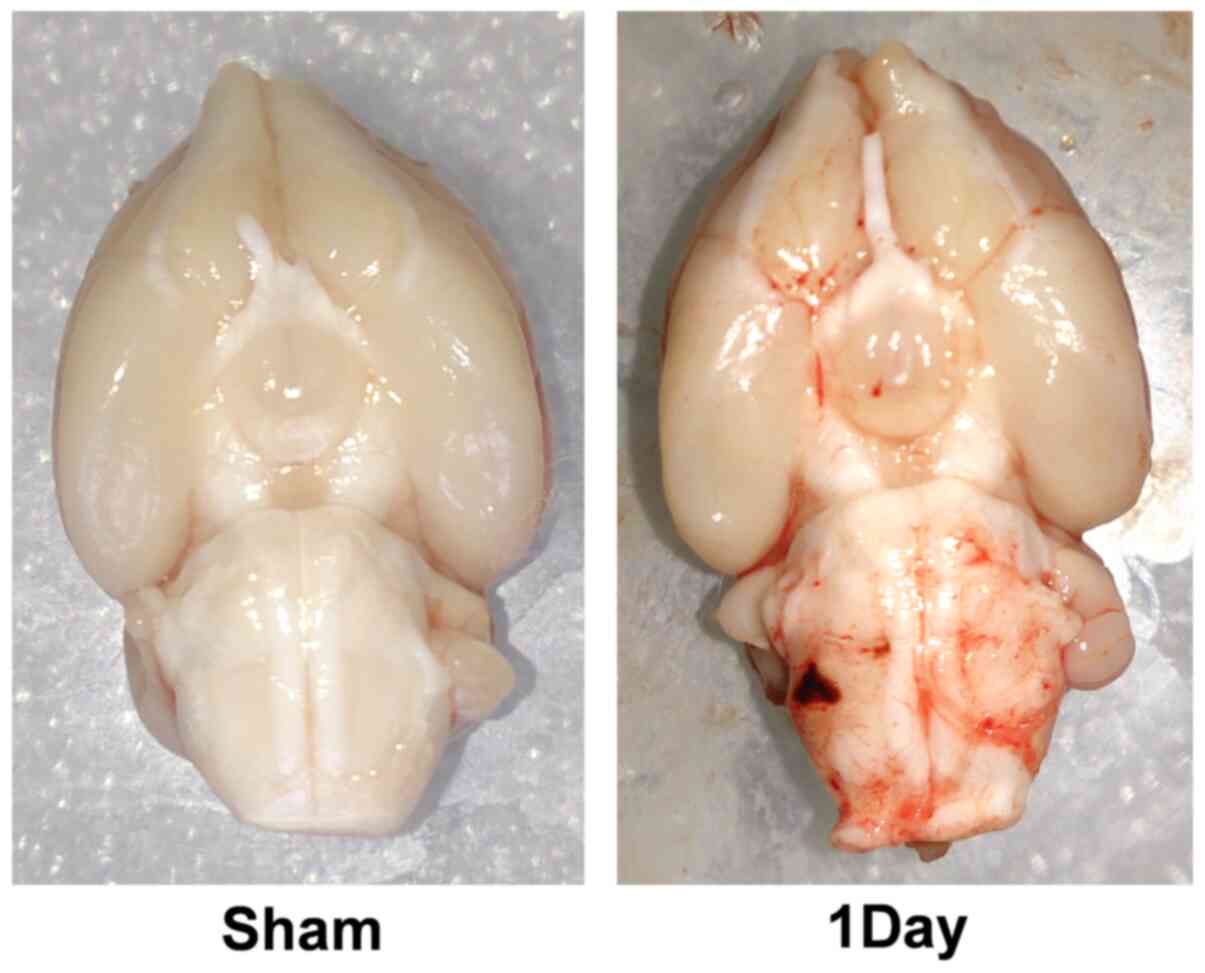
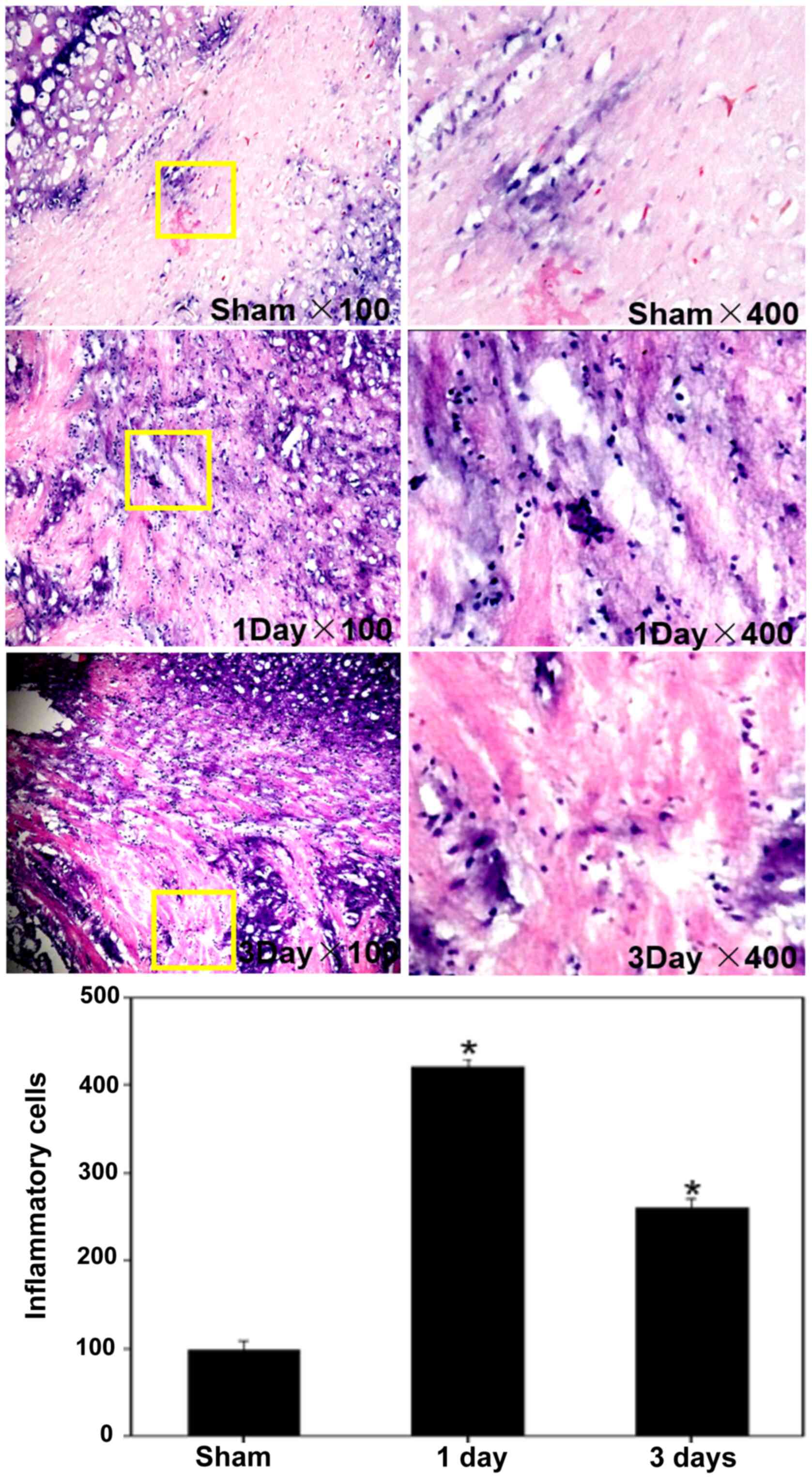
Infiltration of inflammatory cells
following SAH
Immunohistochemical staining was performed on
cross-sections of adult rat brains. The number of inflammatory
cells infiltrated in the day-1 group (412.6±61.9
cells/mm2; n=6) was markedly elevated compared with the
sham group (113.0±47.5 cells/mm2; n=6). There was a
statistically significant difference among the sham, the day-1 and
the day-3 groups (Figs. 1 and
2). The results revealed a high
density of inflammatory cells (predominantly monocytic and
neutrophilic granulocytes) in the cortex, which indicated that the
inflammatory reaction appeared at an early stage following SAH.
Expression of MSK1, TNF-α and IL-1β
during inflammatory responses following SAH
Although MSK1 is known to be expressed at relatively
high levels in the nervous system, its function is well understood.
In order to clarify whether MSK1 participates in the
pro-inflammatory reaction during the process of SAH, the present
study firstly examined the mRNA expression levels of MSK1, IL-1β
and TNF-α in the brain cortex at various survival times following
SAH. The quantitative analysis demonstrated high expression of MSK1
mRNA in the control and sham groups. Furthermore, the level of MSK1
in the cortex decreased gradually following SAH, and then peaked at
day 3 post-SAH and increased during the following days. Conversely,
quantitative analysis of the levels of IL-1β and TNF-α mRNA
revealed that they were increased in the cortex as early as 2 h
after SAH, reaching peak levels at day 1 after SAH and gradually
decreasing during the subsequent days. MSK1 levels peaked 2 days
later compared with those of IL-1β and TNF-α. There was a marked
positive correlation between IL-1β and TNF-α mRNA levels (r=0.741;
P<0.05). In addition, negative correlations were observed
between MSK1 and IL-1β and TNF-α mRNA levels (r=-0.679 and -0.709,
respectively; both P<0.05; Fig.
3).
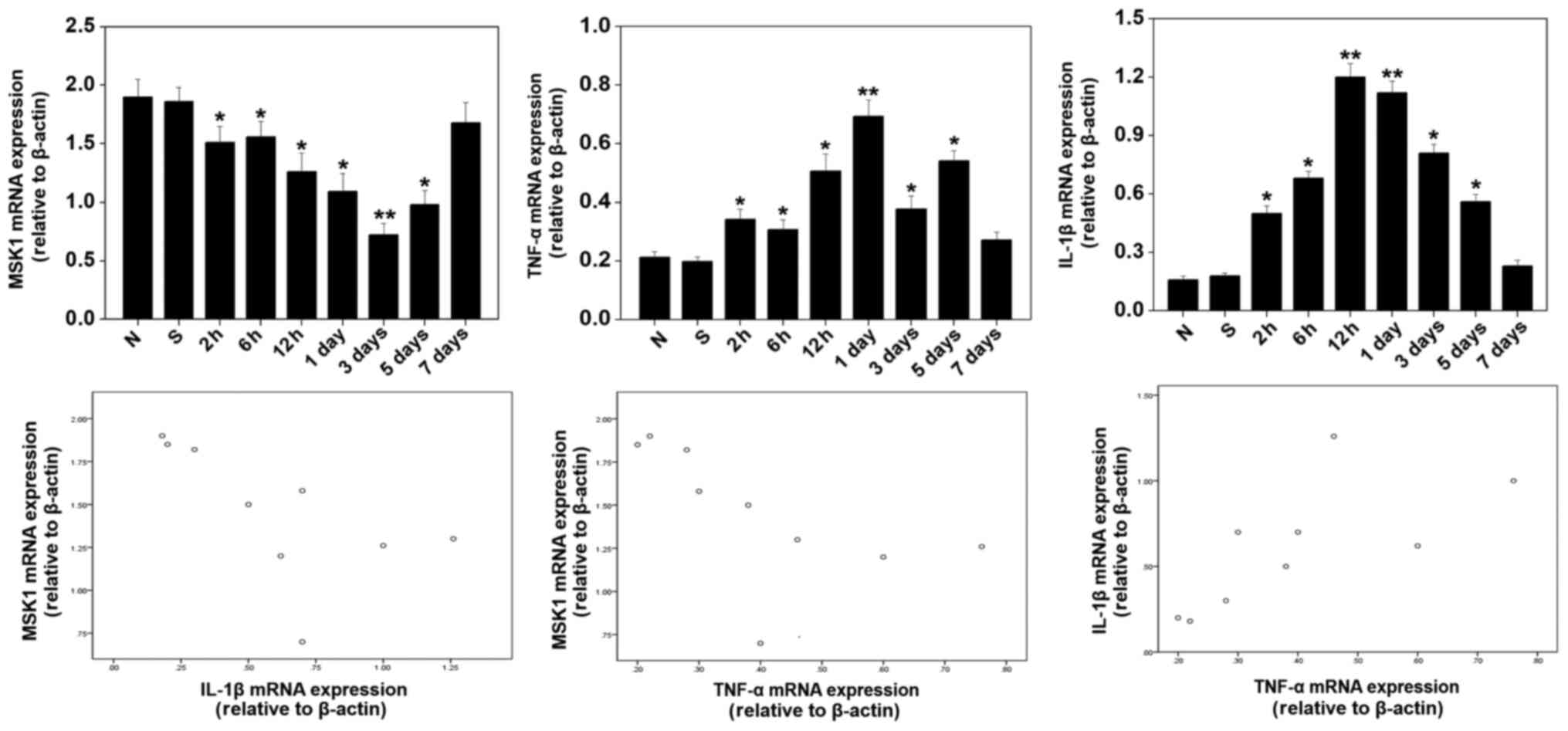 | Figure 3mRNA expression of MSK1, IL-1β, TNF-α
in the brain cortex at various survival time intervals after SAH.
The quantitative analysis demonstrated the high expression of MSK1
mRNA in the control and sham groups. The level of MSK1 decreased
gradually after SAH, then peaked at 3 days, and was increased
during the following days in the cortex. Conversely, analysis of
the levels of IL-1β and TNF-α mRNA indicated that these mRNA
gradually increased following SAH, reaching peak levels at 1 day,
and then gradually decreased during the following days in the
cortex. The correlation was determined by Spearman's analysis. Bars
represent the mean ± SEM (n=6 in each group). Statistical analysis
was performed using one-way analysis of variance followed by
Dunnett's post-hoc test. *P<0.05 and
**P<0.01 vs. sham and normal groups. MSK1, mitogen
and stress-activated protein kinase 1; IL-1β, interleukin-1β;
TNF-α, tumor necrosis factor-α; SAH, subarachnoid hemorrhage. |
Co-localization of MSK1 with TNF-α,
IL-1β and GFAP
Double immunofluorescence staining for MSK1, TNF-α,
IL-1β and GFAP in the cortex was performed at day 1 after SAH to
further explore the association between MAK1 and pro-inflammatory
cytokines. Following injury, sections of adult rat brains were
labeled with MSK1 and GFAP (an astrocyte marker), and
co-localization of MSK1 with GFAP was observed in the brain tissue.
Furthermore, immunofluorescent staining was performed, and the
merged images revealed that MSK1 was located in the same plane of
view in the brain cortex as TNF-α and IL-1β (Fig. 4).
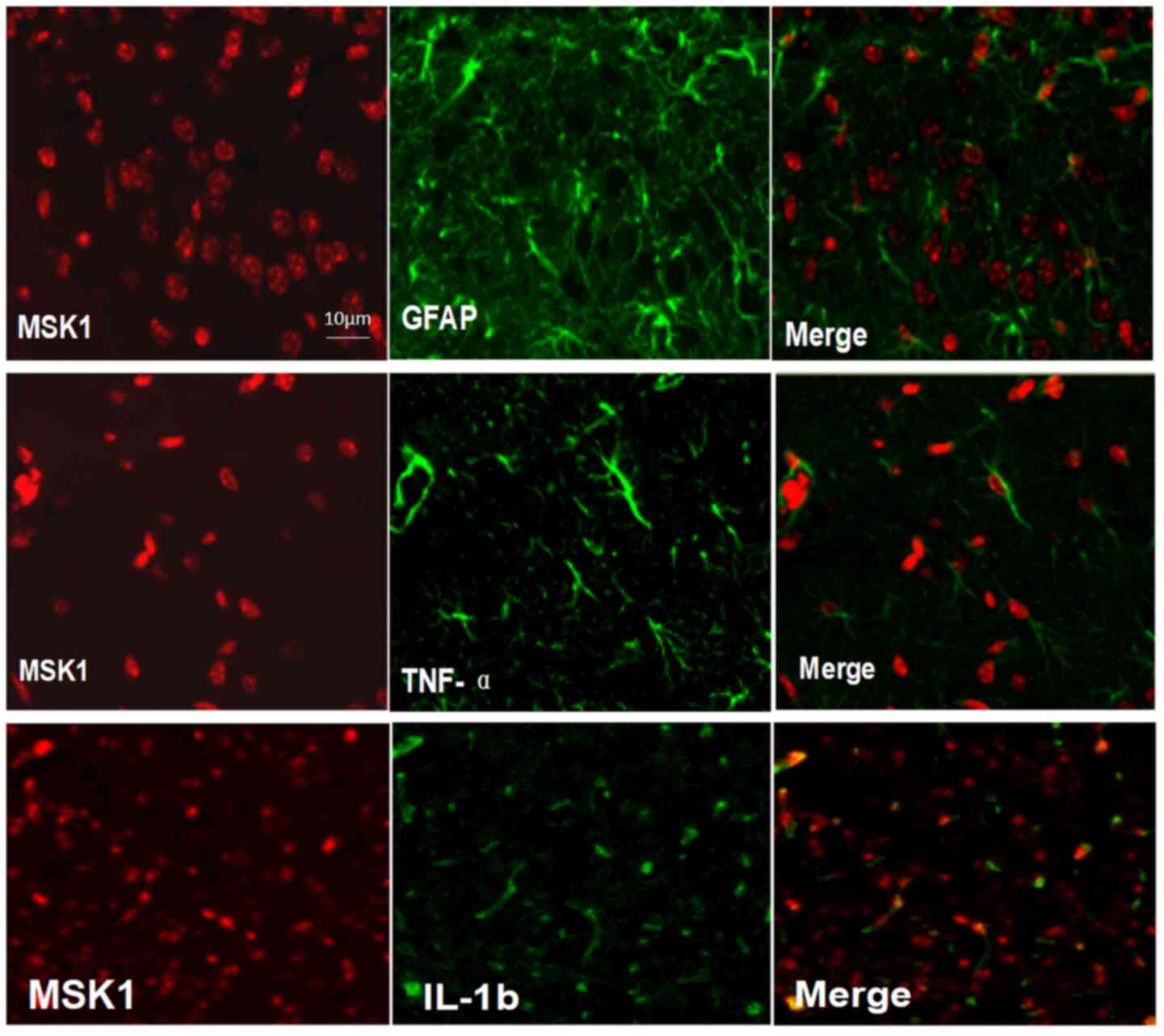 | Figure 4Double immunofluorescence staining
for MSK1, TNF-α, IL-1β and GFAP in brain cortex tissues at 1 day
after SAH. In adult rat brain tissues after injury, sections were
labeled with MSK1 and GFAP, an astrocytes marker, and the
co-localization of MSK1 with GFAP was demonstrated in the brain
tissues. In addition, immunofluorescence staining for MSK1, TNF-α
and IL-1β was also performed, and the merged images indicated that
MSK1 was closely positioned to TNF-α and IL-1β, in the same view in
each section. Scale bar=10 µm. MSK1, mitogen and stress-activated
protein kinase 1; IL-1β, interleukin-1β; TNF-α, tumor necrosis
factor-α; GFAP, glial fibrillary acidic protein. |
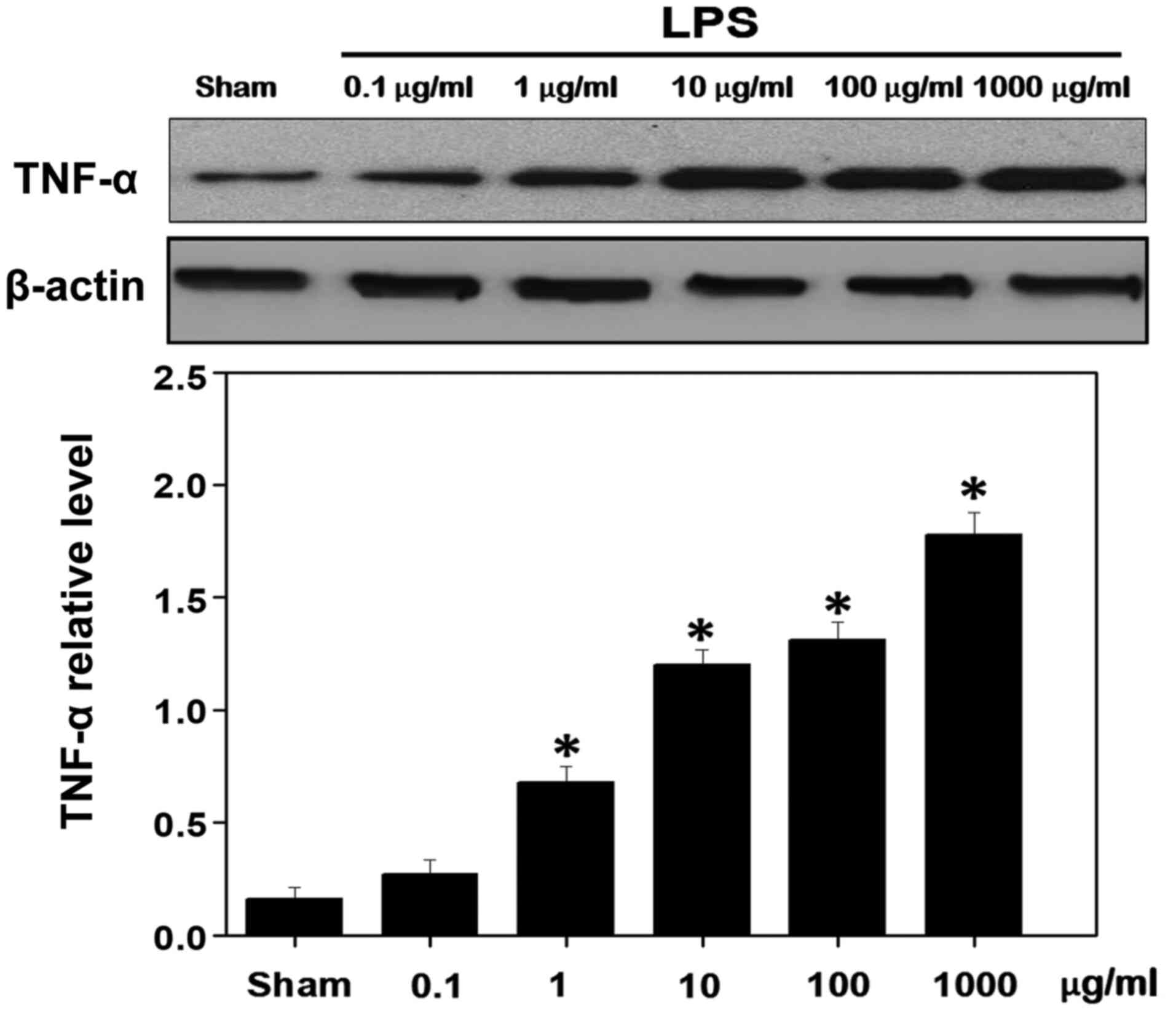
MSK1 inhibits LPS-induced inflammation
in cultured primary astrocytes
To additionally verify the aforementioned results,
LPS was used to induce an inflammatory reaction in cultured primary
astrocytes. It was observed that the TNF-α levels were
significantly increased in the presence of minimum concentrations
of LPS (1 µg/ml) (Fig. 5). To
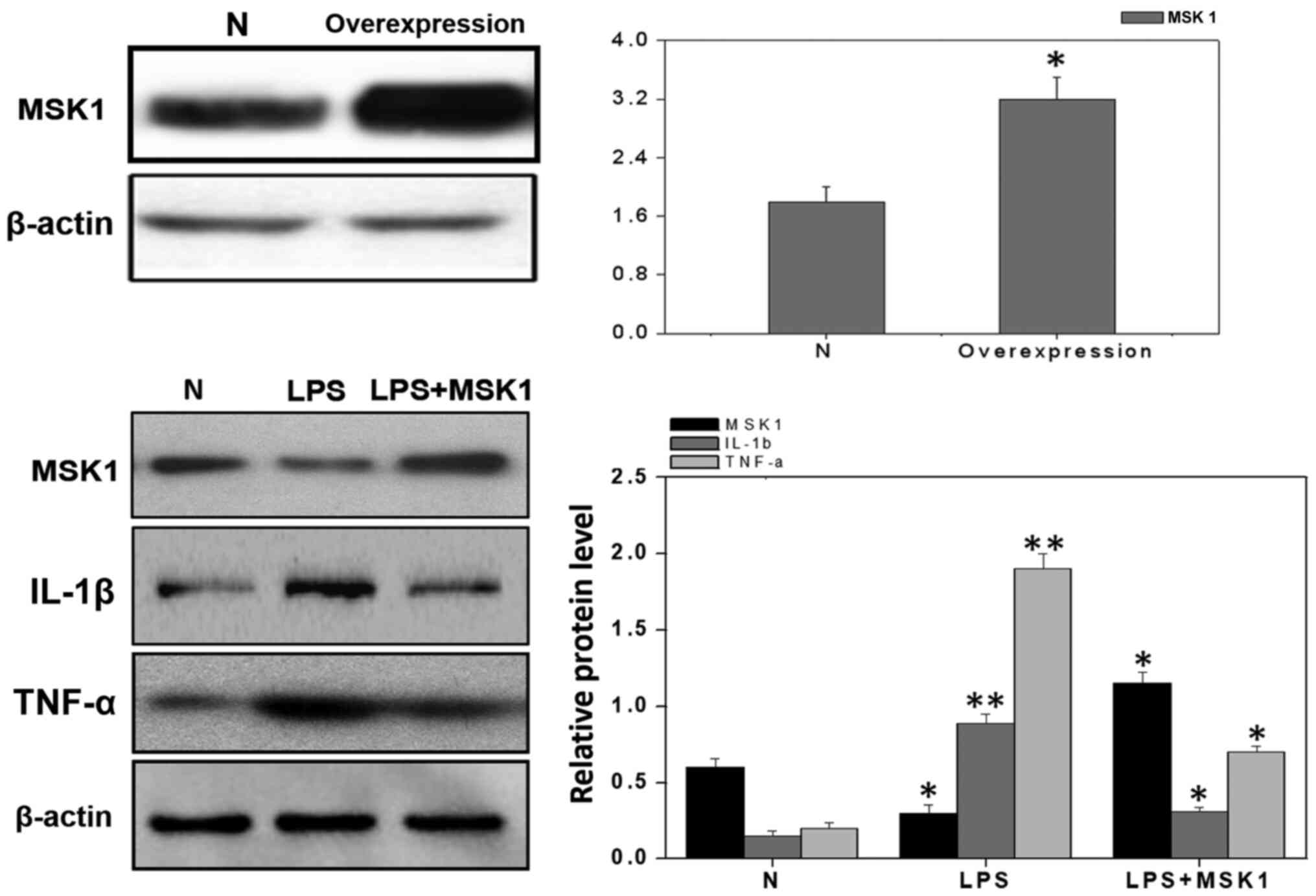
investigate whether MSK1 inhibits the expression of inflammatory
factors in these cells, the cultured primary astrocytes were
transfected with plasmids encoding MSK1. Western blot analysis was
used to examine the levels of TNF-α in MSK1-transfected astrocytes
in the presence of 1 µg/ml LPS. The results demonstrated that the
overexpression of MSK1 decreased the expression of TNF-α (Fig. 6).
Discussion
SAH remains a devastating disease with high
morbidity and mortality rates, and 50% of survivors suffer
neurological dysfunctions that range from mild cognitive deficits
to disabling cerebral infarctions. The majority of previous studies
have focused on cerebral vasospasm (CVS), which is considered the
major cause of cerebral ischemia and poor outcomes following SAH
(19-21).
However, clinical evidence suggests that certain patients
deteriorate neurologically without accompanying vasospasm following
SAH. In addition, preventing vasospasm does not always improves
outcomes (22). The results of the
CONSCIOUS-1 trial (trial no. 00111085), which investigated the role
of clazosentan in preventing the occurrence of CVS following an
aneurysmal SAH, also demonstrated that the pathophysiology
underlying SAH is multifactorial and other pathological mechanisms
independent of vasospasm may be responsible for poor clinical
outcomes (23-25).
Both animal models and human studies have
demonstrated that inflammation is an important factor following
SAH, inducing both direct brain injury and vasospasm, which in turn
leads to brain ischemia (5,26). An increased level of
pro-inflammatory factors in the cerebrospinal fluid of patients
with SAH is associated with poor outcomes (27,28).
In the present study, H&E staining demonstrated
a high density of inflammatory cells in brain cortex 1 day after
SAH. To evaluate the underlying role of inflammation, IL-1β and
TNF-α were detected as inflammation-associated factors. IL-1β, an
important initiator of inflammation, signals through the IL-1
receptor to trigger multiple cellular responses. IL-1β is released
from activated microglia upon brain inflammatory reaction (7,9). TNF-α
is another pro-inflammatory cytokine that can enhance vascular
permeability, and serves a role in the recruitment of inflammatory
cells and endothelial injury (29).
The data from the present study demonstrated that the mRNA
expression levels of TNF-α and IL-1β in injured brain tissues were
significantly increased as early as 1 h after SAH compared with the
sham group. These results suggested that upregulation of IL-1β and
TNF-α may contribute to the initial brain inflammatory response,
which then triggers the subsequent pathophysiological process,
including endothelium dysfunction, BBB disruption, brain edema and
neuron cell apoptosis.
However, the mechanisms of inflammation behind the
pathology of SAH remain unclear. NF-κB, which can be activated by
IL-1β and Toll-like receptors, has been suggested to be the most
important regulator during the inflammatory process subsequent to
SAH (30,31). NF-κB is also the transcription
factor target of MSK1, which can be activated in vivo
downstream of mitogen-activated protein kinase (MAPK)2/ERK2 by
phosphorylation of Thr-581(32).
The MAPK and NF-κB signaling pathways are crucial in
generating inflammatory responses (33). MAPKs can regulate cell
proliferation, apoptosis, differentiation and inflammatory
responses through phosphorylation of their respective substrates
(34,35). Among the MAPK targets, MSK1 is
activated downstream of p38 and ERK1/2(11), and can mediate the phosphorylation
of the NF-kB p65 subunit at Ser-276 upon IL-1β or TNF-α treatment
(13). TNF-α induces the
phosphorylation of MSK1 at Ser-360, Ser-376 and Thr-581 in airway
smooth muscle cells. Dimethyl fumarate, a non-specific NF-kB
inhibitor, attenuated TNF-α-triggered MSK1 Ser-376
autophosphorylation and IL-1β-triggered MSK1/2 phosphorylation in
human keratinocytes (36). There
have been multiple previous studies describing the activation of
CREB, proto-oncogene c-Fos and NF-kb in inflammatory responses
following subarachnoid hemorrhage (37-40).
The present study focused on the involvement of MSK1 downregulation
in the inflammatory responses following SAH in rats. We plan to
investigate the role of activated MSK1 kinase and the effect of a
kinase-deficient mutant of MSK1 in subsequent studies, and to
examine the roles of other molecules such as CREB, c-Fos and NF- kB
in vivo and in vitro. However, the exact mechanism of
action of MSK1 in response to TNF-α and IL-1β during the
inflammatory response following SAH remains unknown, and to the
best of our knowledge, direct genetic evidence supporting the role
of MSK1 in CNS inflammation following SAH has not been reported to
date.
In the present study, the expression of TNF-α and
IL-1β gradually increased 1 h after SAH, peaked at day 1 post-SAH
and decreased during the following days. Notably, the expression of
MSK1 exhibited a negative correlation with TNF-α and IL-1β
expression. It gradually decreased 1 h after SAH, peaked at day 3
post-SAH and increased during the following days. Double
immunofluorescence staining revealed that TNF-α and GFAP were
labeled and co-localized in adult rat brain at day 1 after SAH. In
addition, microscopy analysis revealed that MSK1 was closely
positioned to TNF-α and IL-1β in the same view in the brain cortex.
These data suggested that MSK1 may be associated with the
inflammatory response subsequent to SAH. MSK1 may mediate
inflammatory responses via a negative feedback loop to decrease the
expression of pro-inflammation factors. The observed
co-localization of MSK1 and TNF-α supported this hypothesis, but
the detailed mechanisms require further investigation.
It is well-known that MSK1 has a high level of
expression in astrocytes (41),
which is associated with numerous inflammatory responses of the CNS
(42). Therefore, the present study
established an LPS-induced astrocytic inflammation model to confirm
whether MSK1 serves a role in pro-inflammatory cytokine expression.
The effect of MSK1 overexpression on astrocytes was analyzed by
western blot analysis. The results suggested that MSK1 inhibited
astrocytic inflammation by attenuating the expression of IL-1β and
TNF-α. During the inflammatory response, NF-kB potentially connects
MSK1 with pro-inflammatory factors, although future studies
investigating the effect of kinase-deficient mutant of MSK1 using
small interference RNA or specific inhibitors are required to
confirm this hypothesis.
Despite it is known that inflammatory reactions
occur upon SAH, a link must be identified between inflammation and
poor outcomes following SAH in order to consider inflammation as an
important therapeutic target. Our in vivo and in
vitro experiments implicated that M SK1 may be a key
inflammatory regulator. Regulation of MSK1 expression in activated
astrocytes may provide a novel strategy to protect against
inflammation-induced injury.
Acknowledgements
Not applicable
Funding
Funding: The present study was supported by the National Natural
Science Foundation of China (grant no. 81671129) and the Natural
Science Foundation of Fujian (grant no. 13185026).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
BN, ZL, FL and BZ designed the experiments. BN, ZL,
JW, XC and PJ performed the in vitro and in vivo
experiments. LN analyzed the data and images. BN and ZL wrote the
manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
The animal use and care protocols, including all
surgical procedures, were approved by The Animal Care and Use
Committee of Taishan Medical University (approval no. 2014-002) and
conformed to the Guide for the Care and Use of Laboratory Animals
by the National Institute of Health
Patient approval for publication
Not applicable
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
van Gijn J, Kerr RS and Rinkel GJ:
Subarachnoid haemorrhage. Lancet. 369:306–318. 2007.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Alg VS, Sofat R, Houlden H and Werring DJ:
Genetic risk factors for intracranial aneurysms: a meta-analysis in
more than 116,000 individuals. Neurology. 80:2154–2165.
2013.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Connolly ES Jr, Rabinstein AA, Carhuapoma
JR, Derdeyn CP, Dion J, Higashida RT, Hoh BL, Kirkness CJ, Naidech
AM, Ogilvy CS, et al: American Heart Association Stroke Council;
Council on Cardiovascular Radiology and Intervention; Council on
Cardiovascular Nursing; Council on Cardiovascular Surgery and
Anesthesia; Council on Clinical Cardiology: Guidelines for the
management of aneurysmal subarachnoid hemorrhage: A guideline for
healthcare professionals from the American Heart
Association/american Stroke Association. Stroke. 43:1711–1737.
2012.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Rinkel GJ and Algra A: Long-term outcomes
of patients with aneurysmal subarachnoid haemorrhage. Lancet
Neurol. 10:349–356. 2011.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Sehba FA, Pluta RM and Zhang JH:
Metamorphosis of subarachnoid hemorrhage research: From delayed
vasospasm to early brain injury. Mol Neurobiol. 43:27–40.
2011.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Provencio JJ: Inflammation in subarachnoid
hemorrhage and delayed deterioration associated with vasospasm: A
review. Acta Neurochir Suppl (Wien). 115:233–238. 2013.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Jedrzejowska-Szypulka H, Straszak G and
Laryszbrysz M: Interleukin-1beta plays a role in the activation of
peripheral leukocytes after blood-brain barrier rupture in the
course of subarachnoid hemorrhage. Curr Neurovasc Res. 7:39–48.
2010.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Xie X, Wu X, Cui J, Li H and Yan X:
Increase ICAM-1 and LFA-1 expression by cerebrospinal fluid of
subarachnoid hemorrhage patients: Involvement of TNF-α. Brain Res.
1512:89–96. 2013.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Jiang Y, Liu DW, Han XY, Dong YN, Gao J,
Du B, Meng L and Shi JG: Neuroprotective effects of anti-tumor
necrosis factor-alpha antibody on apoptosis following subarachnoid
hemorrhage in a rat model. J Clin Neurosci. 19:866–872.
2012.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Drobic B, Espino PS and Davie JR: Mitogen-
and stress-activated protein kinase 1 activity and histone h3
phosphorylation in oncogene-transformed mouse fibroblasts. Cancer
Res. 64:9076–9079. 2004.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Deak M, Clifton AD, Lucocq LM and Alessi
DR: Mitogen- and stress-activated protein kinase-1 (MSK1) is
directly activated by MAPK and SAPK2/p38, and may mediate
activation of CREB. EMBO J. 17:4426–4441. 1998.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Vermeulen L, De Wilde G, Van Damme P,
Vanden Berghe W and Haegeman G: Transcriptional activation of the
NF-kappaB p65 subunit by mitogen- and stress-activated protein
kinase-1 (MSK1). EMBO J. 22:1313–1324. 2003.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Reber L, Vermeulen L, Haegeman G and
Frossard N: Ser276 phosphorylation of NF-kB p65 by MSK1 controls
SCF expression in inflammation. PLoS One. 4(e4393)2009.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Ananieva O, Darragh J, Johansen C, Carr
JM, McIlrath J, Park JM, Wingate A, Monk CE, Toth R, Santos SG, et
al: The kinases MSK1 and MSK2 act as negative regulators of
Toll-like receptor signaling. Nat Immunol. 9:1028–1036.
2008.PubMed/NCBI View
Article : Google Scholar
|
|
15
|
Beck IM, Vanden Berghe W, Vermeulen L,
Bougarne N, Vander Cruyssen B, Haegeman G and De Bosscher K:
Altered subcellular distribution of MSK1 induced by glucocorticoids
contributes to NF-kappaB inhibition. EMBO J. 27:1682–1693.
2008.PubMed/NCBI View Article : Google Scholar
|
|
16
|
National Research Council (US) Institute
for Laboratory Animal Research: Guide for the Care and Use of
Laboratory Animals. National Academies Press (US), Washington, DC,
1996.
|
|
17
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2-ΔΔCT method. Methods. 25:402–408. 2001.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Ning B, Guo G, Liu H, Ning L, Sun BL, Li
Z, Wang S, Lv ZW and Fan CD: MSK1 downregulation is associated with
neuronal and astrocytic apoptosis following subarachnoid hemorrhage
in rats. Oncol Lett. 14:2940–2946. 2017.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Dorsch NW: Cerebral arterial spasm - a
clinical review. Br J Neurosurg. 9:403–412. 1995.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Dziurdzik P, Krawczyk L, Jalowiecki P,
Kondera-Anasz Z and Menon L: Serum interleukin-10 in ICU patients
with severe acute central nervous system injuries. Inflamm Res.
53:338–343. 2004.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Ostrowski RP, Colohan AR and Zhang JH:
Molecular mechanisms of early brain injury after subarachnoid
hemorrhage. Neurol Res. 28:399–414. 2006.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Macdonald RL, Kakarieka A, Mayer SA,
Pasqualin A, Rufenacht DA, Schmiedek P and Kassell NF: Prevention
of cerebral vasospasm after aneurysmal subarachnoid hemorrhage with
clazosentan, an endothelin receptor antagonist. Neurosurgery.
59(453)2006.
|
|
23
|
Cahill J, Calvert JW and Zhang JH:
Mechanisms of early brain injury after subarachnoid hemorrhage. J
Cereb Blood Flow Metab. 26:1341–1353. 2006.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Macdonald RL, Pluta RM and Zhang JH:
Cerebral vasospasm after subarachnoid hemorrhage: The emerging
revolution. Nat Clin Pract Neurol. 3:256–263. 2007.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Macdonald RL, Kassell NF, Mayer S,
Ruefenacht D, Schmiedek P, Weidauer S, Frey A, Roux S and Pasqualin
A: CONSCIOUS-1 Investigators. Clazosentan to overcome neurological
ischemia and infarction occurring after subarachnoid hemorrhage
(CONSCIOUS-1): Randomized, double-blind, placebo-controlled phase 2
dose-finding trial. Stroke. 39:3015–3021. 2008.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Hanafy KA, Grobelny B, Fernandez L, Kurtz
P, Connolly ES, Mayer SA, Schindler C and Badjatia N: Brain
interstitial fluid TNF-alpha after subarachnoid hemorrhage. J
Neurol Sci. 291:69–73. 2010.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Kaynar MY, Tanriverdi T, Kafadar AM,
Kacira T, Uzun H, Aydin S, Gumustas K, Dirican A and Kuday C:
Detection of soluble intercellular adhesion molecule-1 and vascular
cell adhesion molecule-1 in both cerebrospinal fluid and serum of
patients after aneurysmal subarachnoid hemorrhage. J Neurosurg.
101:1030–1036. 2004.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Kikuchi T, Okuda Y, Kaito N and Abe T:
Cytokine production in cerebrospinal fluid after subarachnoid
haemorrhage. Neurol Res. 17:106–108. 1995.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Aoki T and Nishimura M: Targeting chronic
inflammation in cerebral aneurysms: Focusing on NF-kappaB as a
putative target of medical therapy. Expert Opin Ther Targets.
14:265–273. 2010.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Greenhalgh AD, Brough D, Robinson EM,
Girard S, Rothwell NJ and Allan SM: Interleukin-1 receptor
antagonist is beneficial after subarachnoid haemorrhage in rat by
blocking haem-driven inflammatory pathology. Dis Model Mech.
5:823–833. 2012.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Ma CX, Yin WN and Cai BW: Activation of
TLR4/ NF-κB signaling pathway in early brain injury after
subarachnoid haemorrhage. Neurol Res. 122:1575–1581.
2009.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Arthur JS: MSK activation and
physiological roles. Front Biosci. 13:5866–5879. 2008.PubMed/NCBI View
Article : Google Scholar
|
|
33
|
Arthur JSC and Ley SC: Mitogen-activated
protein kinases in innate immunity. Nat Rev Immunol. 13:679–692.
2013.PubMed/NCBI View
Article : Google Scholar
|
|
34
|
Kim JH, Choi JS and Lee BH: PI3K/Akt and
MAPK pathways evoke activation of FoxO transcription factor to
undergo neuronal apoptosis in brain of the silkworm Bombyx mori
(Lepidoptera: Bombycidae). Cell Mol Biol. 58:OL1780–OL1785.
2012.PubMed/NCBI
|
|
35
|
Pan WW, Li JD, Huang S, Papadimos TJ, Pan
ZK and Chen LY: Synergistic activation of NF-{κ}B by bacterial
chemoattractant and TNF{α} is mediated by p38 MAPK-dependent RelA
acetylation. J Biol Chem. 285:34348–34354. 2010.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Gesser B, Johansen C, Rasmussen MK,
Funding AT, Otkjaer K, Kjellerup RB, Kragballe K and Iversen L:
Dimethylfumarate specifically inhibits the mitogen and
stress-activated kinases 1 and 2 (MSK1/2): Possible role for its
anti-psoriatic effect. J Invest Dermatol. 127:2129–2137.
2007.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Peng Y, Jin J, Fan L, Xu H, He P, Li J,
Chen T, Ruan W and Chen G: Rolipram Attenuates Early Brain Injury
Following Experimental Subarachnoid Hemorrhage in Rats: Possibly
via Regulating the SIRT1/NF-κB Pathway. Neurochem Res. 43:785–795.
2018.PubMed/NCBI View Article : Google Scholar
|
|
38
|
You W, Zuo G, Shen H, Tian X, Li H, Zhu H,
Yin J, Zhang T and Wang Z: Potential dual role of nuclear
factor-kappa B in experimental subarachnoid hemorrhage-induced
early brain injury in rabbits. Inflamm Res. 65:975–984.
2016.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Mo J, Enkhjargal B, Travis ZD, Zhou K, Wu
P, Zhang G, Zhu Q, Zhang T, Peng J, Xu W, et al: AVE 0991
attenuates oxidative stress and neuronal apoptosis via
Mas/PKA/CREB/UCP-2 pathway after subarachnoid hemorrhage in rats.
Redox Biol. 20:75–86. 2019.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Lv O, Zhou F, Zheng Y, Li Q, Wang J and
Zhu Y: Mild hypothermia protects against early brain injury in rats
following subarachnoid hemorrhage via the TrkB/ERK/CREB signaling
pathway. Mol Med Rep. 14:3901–3907. 2016.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Ning B, Li Z, Zhu N, Hou G and Pang Q:
Traumatic brain injury induces a downregulation of MSK1 in rat
brain cortex. J Mol Neurosci. 49:380–386. 2013.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Myer DJ, Gurkoff GG, Lee SM, Hovda DA and
Sofroniew MV: Essential protective roles of reactive astrocytes in
traumatic brain injury. Brain. 129:2761–2772. 2006.PubMed/NCBI View Article : Google Scholar
|