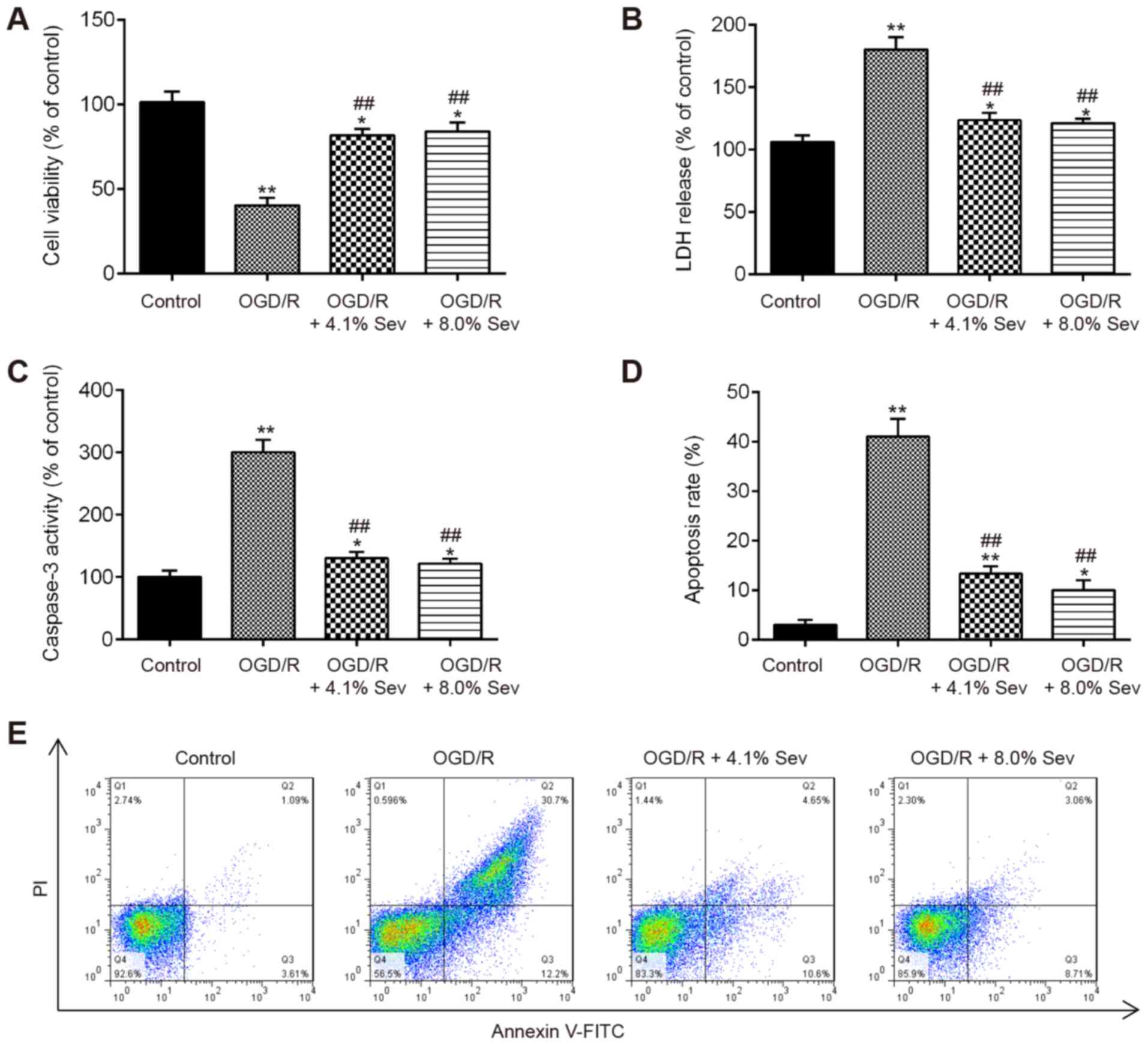
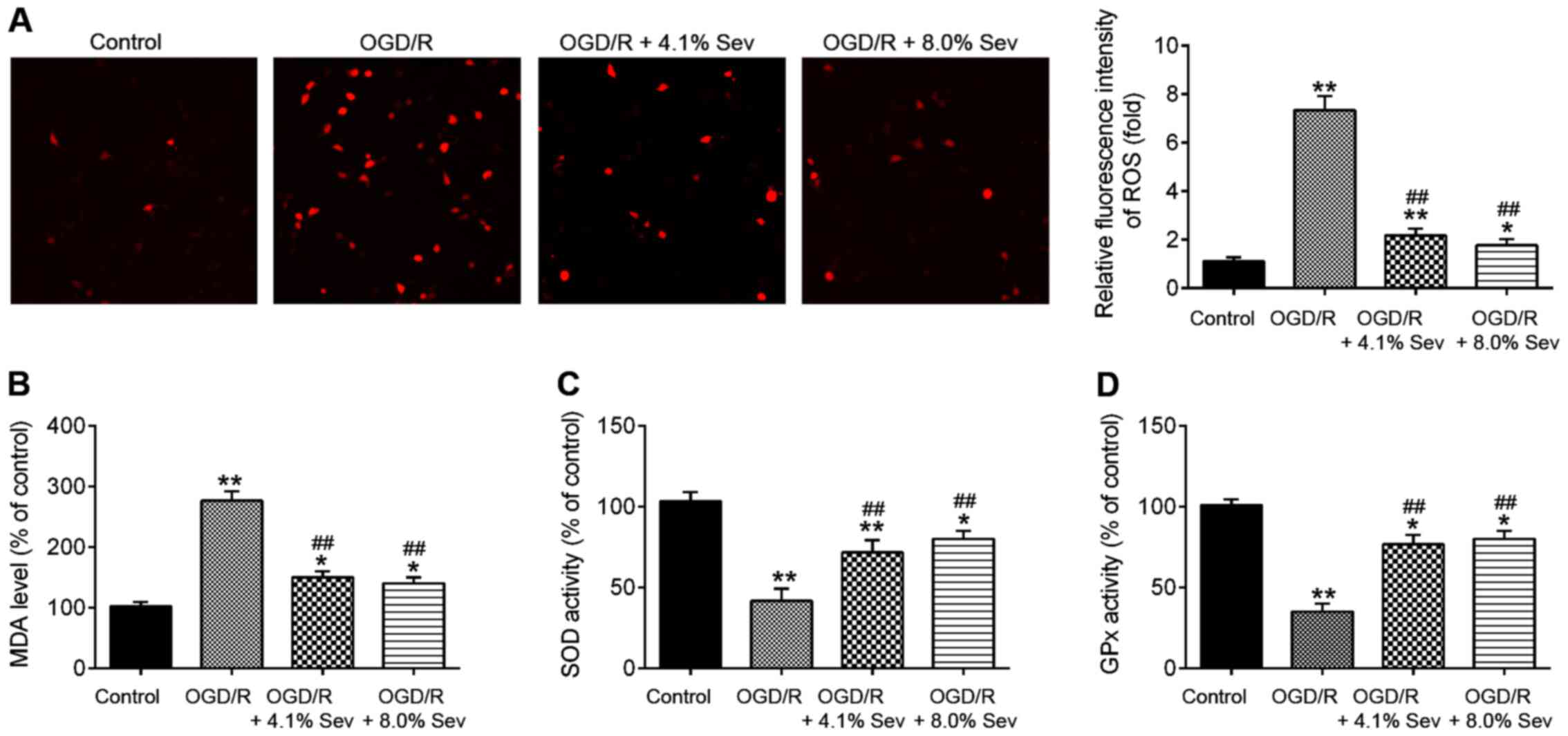
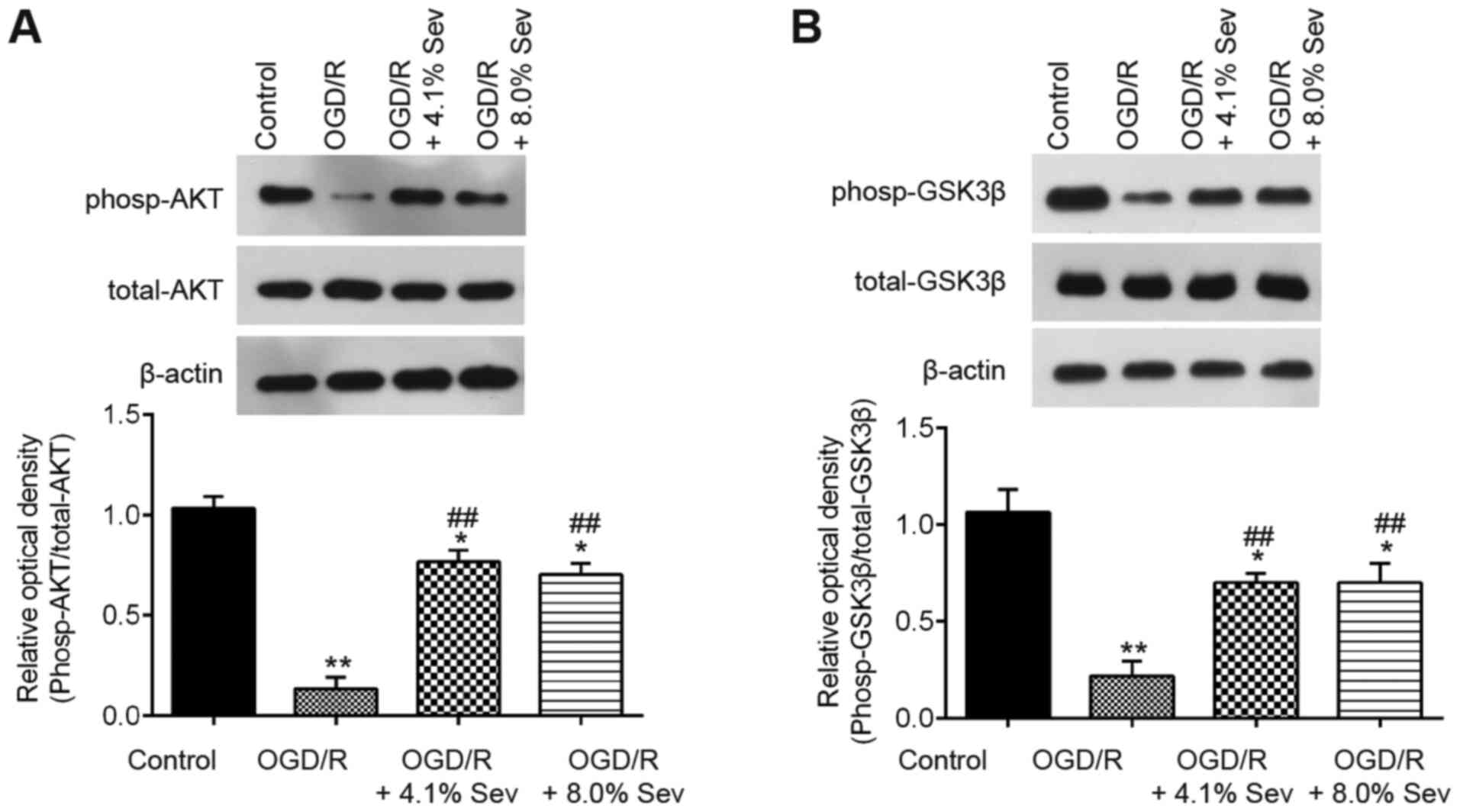
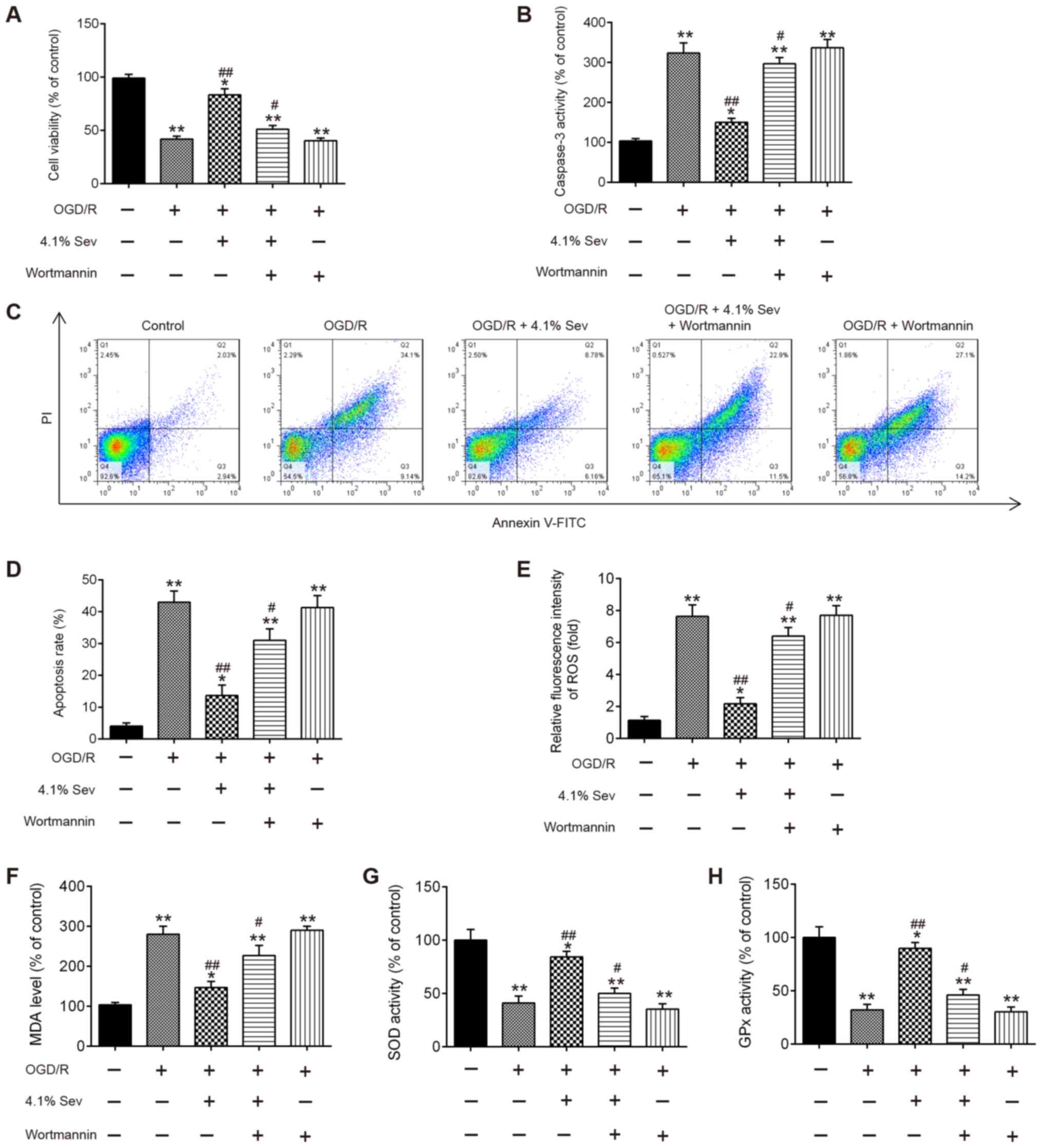
|
1
|
Feigin VL: Stroke epidemiology in the
developing world. Lancet. 365:2160–2161. 2005.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Surinkaew P, Sawaddiruk P, Apaijai N,
Chattipakorn N and Chattipakorn SC: Role of microglia under cardiac
and cerebral ischemia/reperfusion (I/R) injury. Metab Brain Dis.
33:1019–1030. 2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Schrepfer E and Scorrano L: Mitofusins,
from mitochondria to metabolism. Mol Cell. 61:683–694.
2016.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Wu H, Tang C, Tai LW, Yao W, Guo P, Hong
J, Yang X, Li X, Jin Z, Ke J and Wang Y: Flurbiprofen axetil
attenuates cerebral ischemia/reperfusion injury by reducing
inflammation in a rat model of transient global cerebral
ischemia/reperfusion. Biosci Rep. 38(BSR20171562)2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Zhang Y, Li D, Luo J, Chen S, Dou X, Han M
and Zhang H: Pharmacological postconditioning with sevoflurane
activates PI3K/AKT signaling and attenuates cardiopulmonary
bypass-induced lung injury in dog. Life Sci. 173:68–72.
2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Lu CC, Tsai CS, Ho ST, Chen WY, Wong CS,
Wang JJ, Hu OYP and Lin CY: Pharmacokinetics of sevoflurane uptake
into the brain and body. Anaesthesia. 58:951–956. 2003.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Xiong XQ, Lin LN, Wang LR and Jin LD:
Sevoflurane attenuates pulmonary inflammation and
ventilator-induced lung injury by upregulation of HO-1 mRNA
expression in mice. Int J Nanomedicine. 6:1075–1081.
2013.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Li XQ, Cao XZ, Wang J, Fang B, Tan WF and
Ma H: Sevoflurane preconditioning ameliorates neuronal deficits by
inhibiting microglial MMP-9 expression after spinal cord
ischemia/reperfusion in rats. Mol Brain. 7(69)2014.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Park HP, Jeong EJ, Kim MH, Hwang JW, Lim
YJ, Min SW, Kim CS and Jeon YT: Effects of sevoflurane on neuronal
cell damage after severe cerebral ischemia in rats. Korean J
Anesthesiol. 61:327–331. 2011.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Canas PT, Velly LJ, Labrande CN, Guillet
BA, Sautou-Miranda V, Masmejean FM, Nieoullon AL, Gouin FM, Bruder
NJ and Pisano PS: Sevoflurane protects rat mixed cerebrocortical
neuronal-glial cell cultures against transient oxygen-glucose
deprivation: Involvement of glutamate uptake and reactive oxygen
species. Anesthesiology. 105:990–998. 2006.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Wang Z, Ye Z, Huang G, Wang N, Wang E and
Guo Q: Sevoflurane post-conditioning enhanced hippocampal neuron
resistance to global cerebral ischemia induced by cardiac arrest in
rats through PI3K/Akt survival pathway. Front Cell Neurosci.
10(271)2016.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Hu X, Zhang Y, Li W, Liu J and Li Y:
Preconditioning with sevoflurane ameliorates spatial learning and
memory deficit after focal cerebral ischemia-reperfusion in rats.
Int J Dev Neurosci. 31:328–333. 2013.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wang G, Wang T, Zhang Y, Li F, Yu B and
Kou J: Schizandrin protects against OGD/R-induced neuronal injury
by suppressing autophagy: Involvement of the AMPK/mTOR pathway.
Molecules. 24(3624)2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Liang H, Gu M, Yang C, Wang H, Wen X and
Zhou Q: Sevoflurane inhibits invasion and migration of lung cancer
cells by inactivating the p38 MAPK signaling pathway. J Anesth.
26:381–392. 2012.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Pyo H, Yang MS, Jou I and Joe EH:
Wortmannin enhances lipopolysaccharide-induced inducible nitric
oxide synthase expression in microglia in the presence of
astrocytes in rats. Neurosci Lett. 346:141–144. 2003.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Rössler OG, Giehl KM and Thiel G:
Neuroprotection of immortalized hippocampal neurones by
brain-derived neurotrophic factor and Raf-1 protein kinase: Role of
extracellular signal-regulated protein kinase and
phosphatidylinositol 3-kinase. J Neurochem. 88:1240–1252.
2004.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Tang Y, Vater C, Jacobi A, Liebers C, Zou
X and Stiehler M: Salidroside exerts angiogenic and cytoprotective
effects on human bone marrow-derived endothelial progenitor cells
via Akt/mTOR/p70S6K and MAPK signalling pathways. Br J Pharmacol.
171:2440–2456. 2014.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Landucci E, Lattanzi R, Gerace E,
Scartabelli T, Balboni G, Negri L and Pellegrini-Giampietro DE:
Prokineticins are neuroprotective in models of cerebral ischemia
and ischemic tolerance in vitro. Neuropharmacology. 108:39–48.
2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Ye Z, Xia P, Cheng ZG and Guo Q:
Neuroprotection induced by sevoflurane-delayed post-conditioning is
attributable to increased phosphorylation of mitochondrial GSK-3β
through the PI3K/Akt survival pathway. J Neurol Sci. 348:216–225.
2015.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Chen H, Yoshioka H, Kim GS, Jung JE, Okami
N, Sakata H, Maier CM, Narasimhan P, Goeders CE and Chan PH:
Oxidative stress in ischemic brain damage: Mechanisms of cell death
and potential molecular targets for neuroprotection. Antioxid Redox
Signal. 14:1505–1517. 2011.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Xu HX, Pan W, Qian JF, Liu F, Dong HQ and
Liu QJ: MicroRNA-21 contributes to the puerarin-induced
cardioprotection via suppression of apoptosis and oxidative stress
in a cell model of ischemia/reperfusion injury. Mol Med Rep.
20:719–727. 2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Siti HN, Kamisah Y and Kamsiah J: The role
of oxidative stress, antioxidants and vascular inflammation in
cardiovascular disease (Review). Vascul Pharmacol. 71:40–56.
2015.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Lai Z, Zhang L, Su J, Cai D and Xu Q:
Sevoflurane postconditioning improves long-term learning and memory
of neonatal hypoxia-ischemia brain damage rats via the
PI3K/Akt-mPTP pathway. Brain Res. 1630:25–37. 2016.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Li P, Zhang Y and Liu H: The role of
Wnt/β-catenin pathway in the protection process by dexmedetomidine
against cerebral ischemia/reperfusion injury in rats. Life Sci.
236(116921)2019.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Patel SS and Goa KL: Sevoflurane. A review
of its pharmacodynamic and pharmacokinetic properties and its
clinical use in general anaesthesia. Drugs. 51:658–700.
1996.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Wise-Faberowski L, Raizada MK and Sumners
C: Desflurane and sevoflurane attenuate oxygen and glucose
deprivation-induced neuronal cell death. J Neurosurg Anesthesiol.
15:193–199. 2003.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Wen XR, Fu YY, Liu HZ, Wu J, Shao XP,
Zhang XB, Tang M, Shi Y, Ma K, Zhang F, et al: Neuroprotection of
sevoflurane against ischemia/reperfusion-induced brain injury
through inhibiting JNK3/caspase-3 by enhancing akt signaling
pathway. Mol Neurobiol. 53:1661–1671. 2016.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Coyle JT and Puttfarcken P: Oxidative
stress, glutamate, and neurodegenerative disorders. Science.
262:689–695. 1993.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Shen MH, Zhang CB, Zhang JH and Li PF:
Electroacupuncture attenuates cerebral ischemia and reperfusion
injury in middle cerebral artery occlusion of rat via modulation of
apoptosis, inflammation, oxidative stress, and excitotoxicity. Evid
Based Complement Alternat Med. 2016(9438650)2016.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Sosunov SA, Ameer X, Niatsetskaya ZV,
Utkina-Sosunova I, Ratner VI and Ten VS: Isoflurane anesthesia
initiated at the onset of reperfusion attenuates oxidative and
hypoxic-ischemic brain injury. PLoS One.
10(e0120456)2015.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Chen L, Wei X, Hou Y, Liu X, Li S, Sun B,
Liu X and Liu H: Tetramethylpyrazine analogue CXC195 protects
against cerebral ischemia/reperfusion-induced apoptosis through
PI3K/Akt/GSK3β pathway in rats. Neurochem Int. 66:27–32.
2014.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Liu H, Liu X, Wei X, Chen L, Xiang Y, Yi F
and Zhang X: Losartan, an angiotensin II type 1 receptor blocker,
ameliorates cerebral ischemia-reperfusion injury via
PI3K/Akt-mediated eNOS phosphorylation. Brain Res Bull. 89:65–70.
2012.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Zhang J, Deng Z, Liao J, Song C, Liang C,
Xue H, Wang L, Zhang K and Yan G: Leptin attenuates cerebral
ischemia injury through the promotion of energy metabolism via the
PI3K/Akt pathway. J Cereb Blood Flow Metab. 33:567–574.
2013.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Martin M, Rehani K, Jope RS and Michalek
SM: Toll-like receptor-mediated cytokine production is
differentially regulated by glycogen synthase kinase 3. Nat
Immunol. 6:777–784. 2005.PubMed/NCBI View
Article : Google Scholar
|
|
35
|
Jope RS and Johnson GVW: The glamour and
gloom of glycogen synthase kinase-3. Trends Biochem Sci. 29:95–102.
2004.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Arslan F, Lai RC, Smeets MB, Akeroyd L,
Choo A, Aguor ENE, Timmers L, van Rijen HV, Doevendans PA,
Pasterkamp G, et al: Mesenchymal stem cell-derived exosomes
increase ATP levels, decrease oxidative stress and activate
PI3K/Akt pathway to enhance myocardial viability and prevent
adverse remodeling after myocardial ischemia/reperfusion injury.
Stem Cell Res. 10:301–312. 2013.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Li Y, Zhu W, Tao J, Xin P, Liu M, Li J and
Wei M: Fasudil protects the heart against ischemia-reperfusion
injury by attenuating endoplasmic reticulum stress and modulating
SERCA activity: The differential role for PI3K/Akt and JAK2/STAT3
signaling pathways. PLoS One. 7(e48115)2012.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Zhang JF, Zhang L, Shi LL, Zhao ZH, Xu H,
Liang F, Li HB, Zhao Y, Xu X, Yang K and Tian YF: Parthenolide
attenuates cerebral ischemia/reperfusion injury via Akt/GSK-3β
pathway in PC12 cells. Biomed Pharmacother. 89:1159–1165.
2017.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Gerace E, Scartabelli T,
Pellegrini-Giampietro DE and Landucci E: Tolerance induced by
(S)-3,5-dihydroxyphenylglycine postconditioning is mediated by the
PI3K/Akt/GSK3β signalling pathway in an in vitro model of cerebral
ischemia. Neuroscience. 433:221–229. 2020.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Lee DH, Lee YJ and Kwon KH:
Neuroprotective effects of astaxanthin in oxygen-glucose
deprivation in SH-SY5Y cells and global cerebral ischemia in rat. J
Clin Biochem Nutr. 47:121–129. 2010.PubMed/NCBI View Article : Google Scholar
|