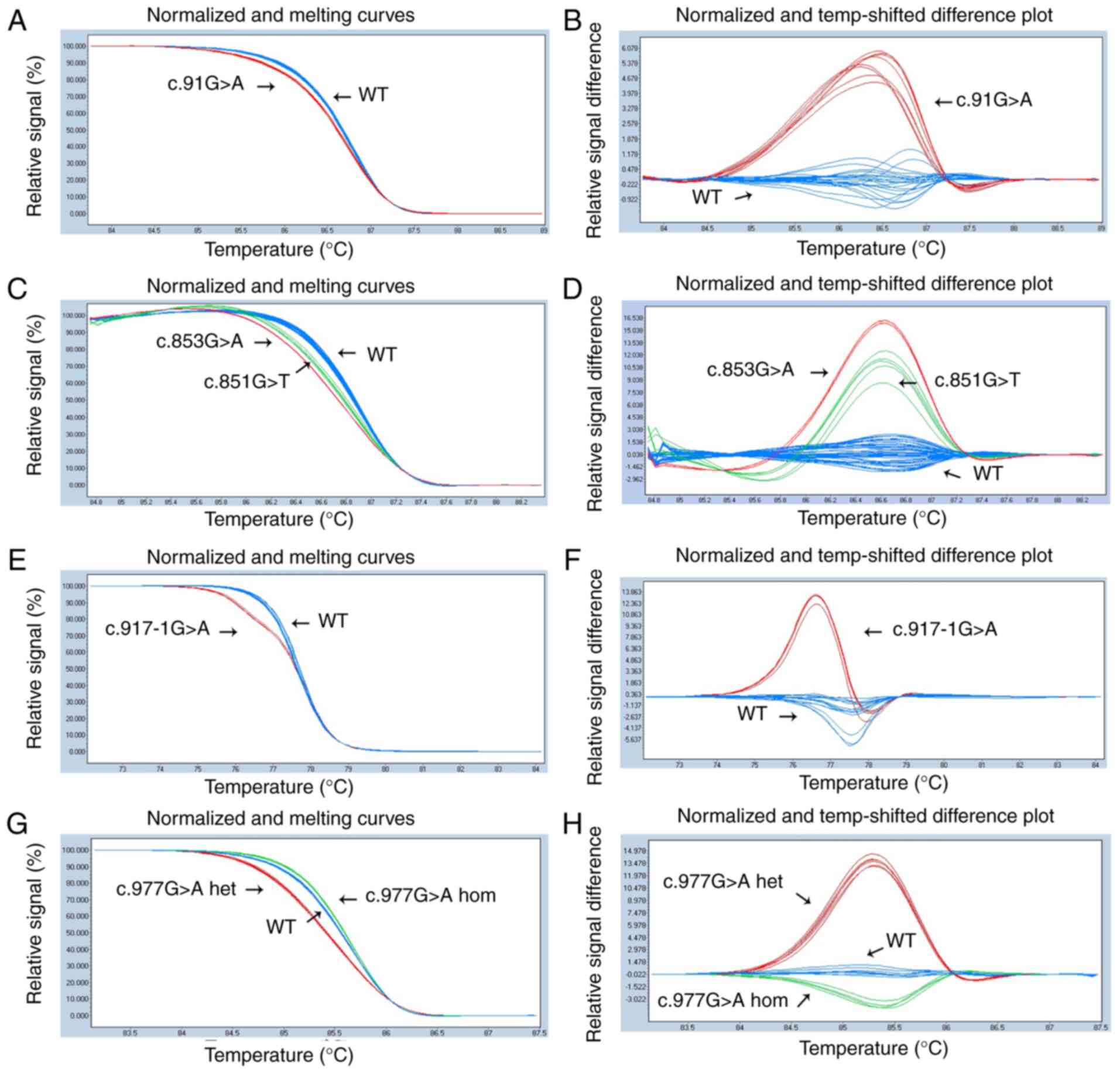
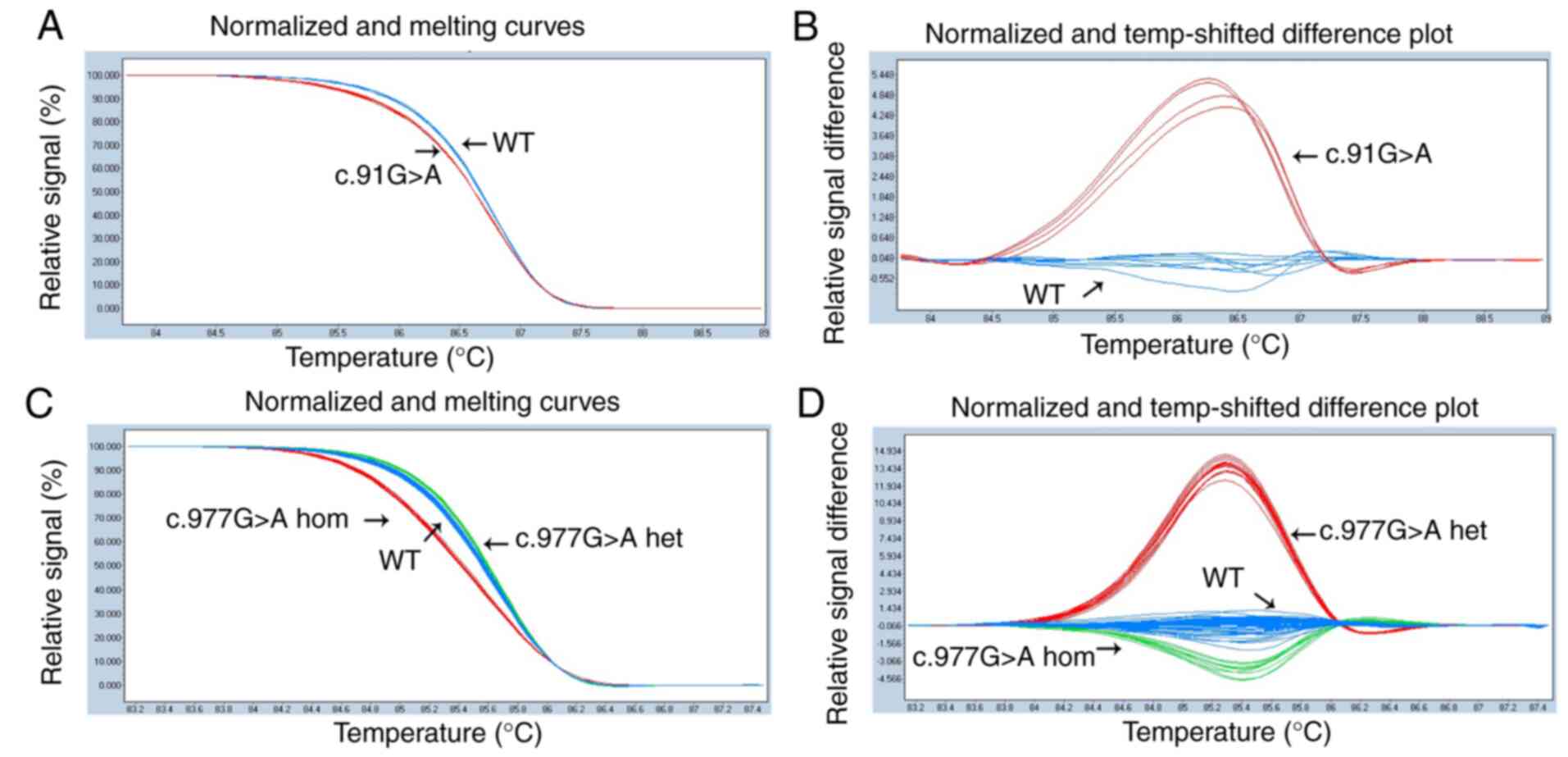
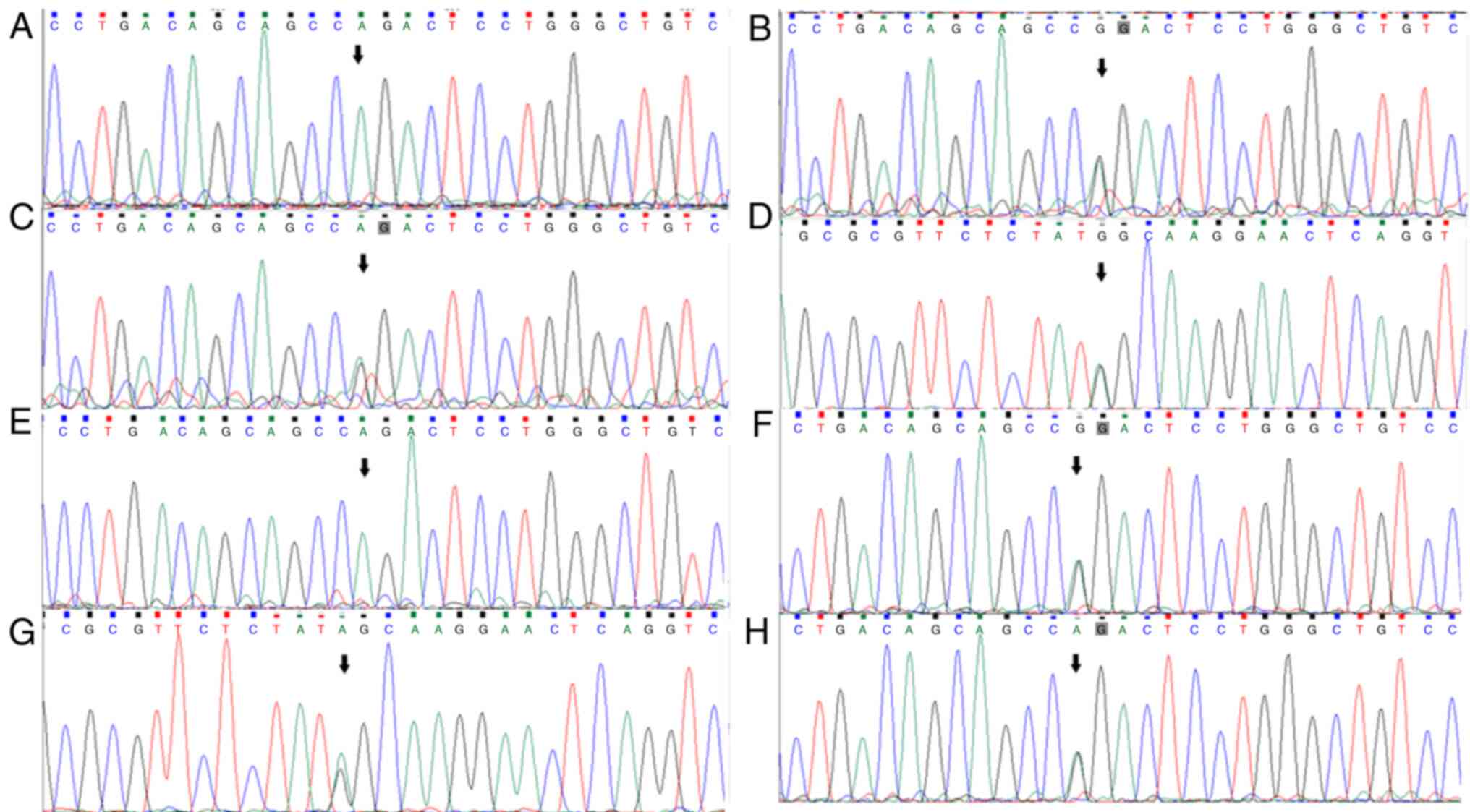
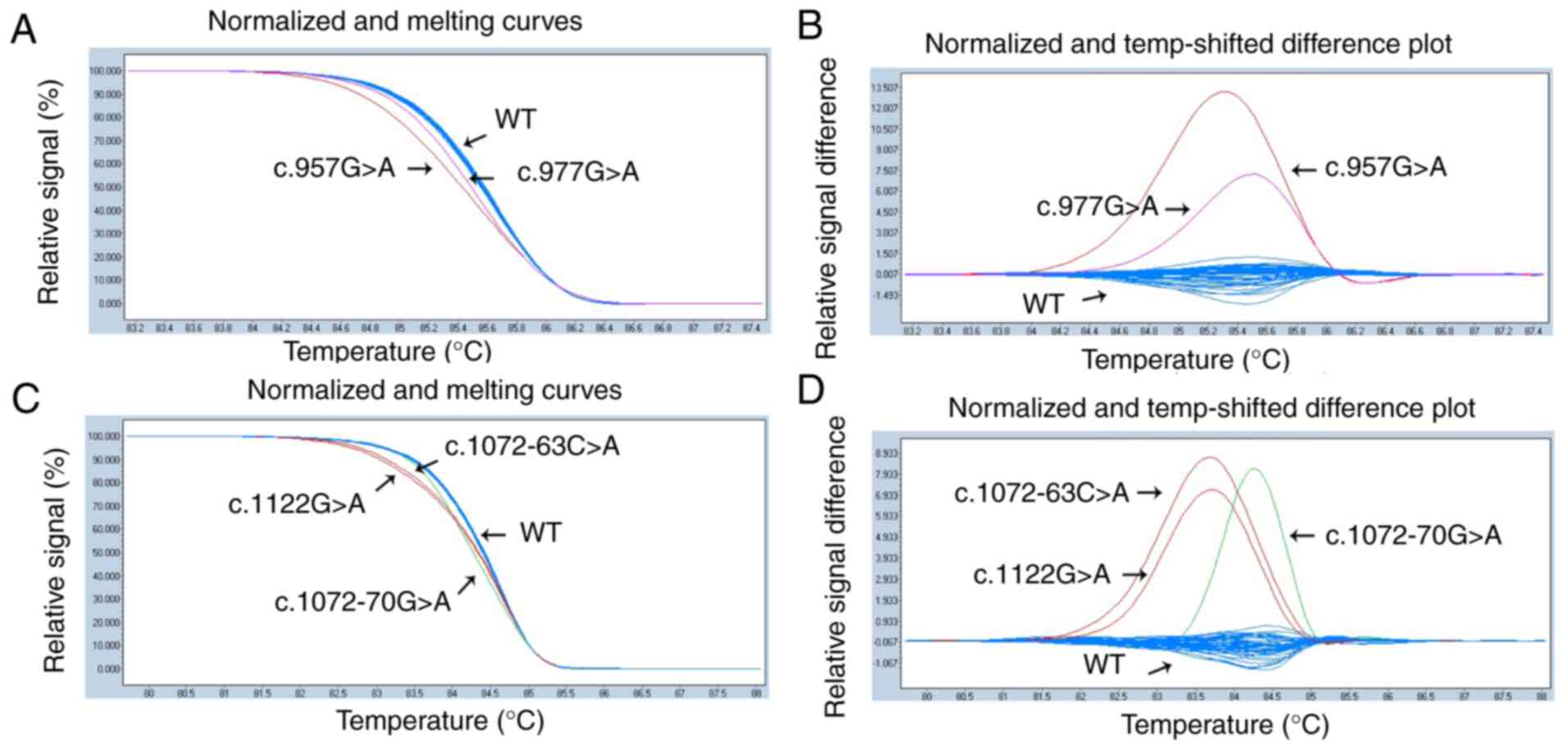
|
1
|
van Kuilenburg AB, Dobritzsch D, Meijer J,
Krumpel M, Selim LA, Rashed MS, Assmann B, Meinsma R, Lohkamp B,
Ito T, et al: β-ureidopropionase deficiency: Phenotype, genotype
and protein structural consequences in 16 patients. Biochim Biophys
Acta. 1822:1096–1108. 2012.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Kuhara T, Ohse M, Inoue Y and Shinka T:
Five cases of beta-ureidopropionase deficiency detected by GC/MS
analysis of urine metabolome. J Mass Spectrom. 44:214–221.
2009.PubMed/NCBI View
Article : Google Scholar
|
|
3
|
Nakajima Y, Meijer J, Dobritzsch D, Ito T,
Meinsma R, Abeling NG, Roelofsen J, Zoetekouw L, Watanabe Y,
Tashiro K, et al: Clinical, biochemical and molecular analysis of
13 Japanese patients with β-ureidopropionase deficiency
demonstrates high prevalence of the c.977G>A (p.R326Q) mutation
[corrected]. J Inherit Metab Dis. 37:801–812. 2014.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Wittwer CT: High-resolution DNA melting
analysis: Advancements and limitations. Hum Mutat. 30:857–859.
2009.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Wittwer CT, Reed GH, Gundry CN,
Vandersteen JG and Pryor RJ: High-resolution genotyping by amplicon
melting analysis using LCGreen. Clin Chem. 49:853–860.
2003.PubMed/NCBI View
Article : Google Scholar
|
|
6
|
Gundry CN, Vandersteen JG, Reed GH, Pryor
RJ, Chen J and Wittwer CT: Amplicon melting analysis with labeled
primers: A closed-tube method for differentiating homozygotes and
heterozygotes. Clin Chem. 49:396–406. 2003.PubMed/NCBI View
Article : Google Scholar
|
|
7
|
Lyon E and Wittwer CT: LightCycler
technology in molecular diagnostics. J Mol Diagn. 11:93–101.
2009.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Nguyen-Dumont T, Calvez-Kelm FL, Forey N,
McKay-Chopin S, Garritano S, Gioia-Patricola L, De Silva D, Weigel
R, Sangrajrang S, Lesueur F, et al: Description and validation of
high-throughput simultaneous genotyping and mutation scanning by
high-resolution melting curve analysis. Hum Mutat. 30:884–890.
2009.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Montgomery JL, Sanford LN and Wittwer CT:
High-resolution DNA melting analysis in clinical research and
diagnostics. Expert Rev Mol Diagn. 10:219–240. 2010.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Zhou L, Palais RA, Paxton CN, Geiersbach
KB and Wittwer CT: Copy number assessment by competitive PCR with
limiting deoxynucleotide triphosphates and high-resolution melting.
Clin Chem. 61:724–733. 2015.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Chambliss AB, Resnick M, Petrides AK,
Clarke WA and Marzinke MA: Rapid screening for targeted genetic
variants via high-resolution melting curve analysis. Clin Chem Lab
Med. 55:507–516. 2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Zahorakova D, Lelkova P, Gregor V, Magner
M, Zeman J and Martasek P: MECP2 mutations in Czech patients with
Rett syndrome and Rett-like phenotypes: Novel mutations,
genotype-phenotype correlations and validation of high-resolution
melting analysis for mutation scanning. J Hum Genet. 61:617–625.
2016.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Azuara D, Aussó S, Rodriguez-Moranta F,
Guardiola J, Sanjuan X, Lobaton T, Boadas J, Piqueras M, Monfort D,
Guinó E, et al: New methylation biomarker panel for early diagnosis
of dysplasia or cancer in high-risk inflammatory bowel disease
patients. Inflamm Bowel Dis. 24:2555–2564. 2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Müller KE, Zampieri RA, Aoki JI, Muxel SM,
Nerland AH and Floeter-Winter LM: Amino acid permease 3 (aap3)
coding sequence as a target for Leishmania identification and
diagnosis of leishmaniases using high resolution melting analysis.
Parasit Vectors. 11(421)2018.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Rezaei F, Haeili M, Fooladi AI and
Feizabadi MM: High resolution melting Curve analysis for rapid
detection of streptomycin and ethambutol resistance in
Mycobacterium tuberculosis. Maedica (Bucur). 12:246–257.
2017.PubMed/NCBI
|
|
16
|
Fang Y, Cai C, Wang C, Sun B, Zhang X, Fan
W, Hu W, Meng Y, Lin S, Zhang C, et al: Clinical and genetic
analysis of 7 Chinese patients with beta-ureidopropionase
deficiency. Medicine (Baltimore). 98(e14021)2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
van Kuilenburg AB, Meinsma R, Assman B,
Hoffman GF, Voit T, Ribes A, Lorente I, Busch R, Mayatepek E,
Abeling NG, et al: Genetic analysis of the first 4 patients with
beta-ureidopropionase deficiency. Nucleosides Nucleotides Nucleic
Acids. 25:1093–1098. 2006.PubMed/NCBI View Article : Google Scholar
|
|
18
|
van Kuilenburg AB, Meinsma R, Beke E,
Assmann B, Ribes A, Lorente I, Busch R, Mayatepek E, Abeling NG,
van Cruchten A, et al: beta-Ureidopropionase deficiency: An inborn
error of pyrimidine degradation associated with neurological
abnormalities. Hum Mol Genet. 13:2793–2801. 2004.PubMed/NCBI View Article : Google Scholar
|
|
19
|
van Kuilenburg AB, van Lenthe H, Ratmann
GG, Assmann B, Hoffmann GF, Brautigam C and van Gennip AH:
Confirmation of the enzyme defect in the first case of
beta-ureidopropionase deficiency. Beta-alanine deficiency. Adv Exp
Med Biol. 486:243–246. 2000.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Shu J, Lv X, Jiang S, Zhang Y, Zhang C,
Meng Y, Situ A, Xu H and Song L: Genetic analysis of the UPB1 gene
in two new Chinese families with β-ureidopropionase deficiency and
the carrier frequency of the mutation c.977G>A in Northern
China. Childs Nerv Syst. 30:2109–2114. 2014.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Nagai Y, Iwade Y, Hayakawa E, Nakano M,
Sakai T, Mitarai S, Katayama M, Nosaka T and Yamaguchi T: High
resolution melting curve assay for rapid detection of
drug-resistant Mycobacterium tuberculosis. J Infect Chemother.
19:1116–1125. 2013.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Sirous M, Khosravi AD, Tabandeh MR,
Salmanzadeh S, Ahmadkhosravi N and Amini S: Molecular detection of
rifampin, isoniazid, and ofloxacin resistance in Iranian isolates
of Mycobacterium tuberculosis by high-resolution melting analysis.
Infect Drug Resist. 11:1819–1829. 2018.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Kramer D, Thunnissen FB, Gallegos-Ruiz MI,
Smit EF, Postmus PE, Meijer CJ, Snijders PJ and Heideman DA: A
fast, sensitive and accurate high resolution melting (HRM)
technology-based assay to screen for common K-ras mutations. Cell
Oncol. 31:161–167. 2009.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Chou LS, Lyon E and Wittwer CT: A
comparison of high-resolution melting analysis with denaturing
high-performance liquid chromatography for mutation scanning:
Cystic fibrosis transmembrane conductance regulator gene as a
model. Am J Clin Pathol. 124:330–338. 2005.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Hondow HL, Fox SB, Mitchell G, Scott RJ,
Beshay V, Wong SQ, kConFab Investigators and Dobrovic A: A
high-throughput protocol for mutation scanning of the BRCA1 and
BRCA2 genes. BMC Cancer. 11(265)2011.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Sirdah MM: Superparamagnetic-bead based
method: An effective DNA extraction from dried blood spots (DBS)
for diagnostic PCR. J Clin Diagn Res. 8:FC01–FC04. 2014.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Natoli ME, Rohrman BA, De Santiago C, van
Zyl GU and Richards-Kortum RR: Paper-based detection of HIV-1 drug
resistance using isothermal amplification and an oligonucleotide
ligation assay. Anal Biochem. 544:64–71. 2018.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Gupta V, Arora R, Ranjan A, Bairwa NK,
Malhotra DK, Udhayasuriyan PT, Saha A and Bamezai R: Gel-based
nonradioactive single-strand conformational polymorphism and
mutation detection: Limitations and solutions. Methods Mol Biol.
291:247–261. 2005.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Ohtomo R, Oka N and Morimoto S:
PCR-denaturing gradient gel electrophoresis as a simple
identification tool of arbuscular mycorrhizal fungal isolates.
Microbes Environ. 34:356–362. 2019.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Er TK and Chang JG: High-resolution
melting: Applications in genetic disorders. Clin Chim Acta.
414:197–201. 2012.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Tong SY and Giffard PM: Microbiological
applications of high-resolution melting analysis. J Clin Microbiol.
50:3418–3421. 2012.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Reed GH and Wittwer CT: Sensitivity and
specificity of single-nucleotide polymorphism scanning by
high-resolution melting analysis. Clin Chem. 50:1748–1754.
2004.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Chen C, Li S, Lu X, Tan B, Huang C and Qin
L: High resolution melting method to detect single nucleotide
polymorphism of VKORC1 and CYP2C9. Int J Clin Exp Pathol.
7:2558–2564. 2014.PubMed/NCBI
|
|
34
|
Słomka M, Sobalska-Kwapis M, Wachulec M,
Bartosz G and Strapagiel D: High resolution melting (HRM) for
high-throughput genotyping-limitations and caveats in practical
case studies. Int J Mol Sci. 18(2316)2017.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Fidlerova J, Kleiblova P, Kormunda S,
Novotny J and Kleibl Z: Contribution of the β-ureidopropionase
(UPB1) gene alterations to the development of
fluoropyrimidine-related toxicity. Pharmacol Rep. 64:1234–1242.
2012.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Johnson MR and Diasio RB: Importance of
dihydropyrimidine dehydrogenase (DPD) deficiency in patients
exhibiting toxicity following treatment with 5-fluorouracil. Adv
Enzyme Regul. 41:151–157. 2001.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Thomas HR, Ezzeldin HH, Guarcello V,
Mattison LK, Fridley BL and Diasio RB: Genetic regulation of
beta-ureidopropionase and its possible implication in altered
uracil catabolism. Pharmacogenet Genom. 18:25–35. 2008.PubMed/NCBI View Article : Google Scholar
|