Introduction
Sepsis is one of the most common causes of acute
kidney injury (AKI) in critically ill patients (1). Compared with AKI caused by other
causes, sepsis-induced AKI increases the length of hospital stay,
medical resource consumption and mortality, and adversely affects
the overall clinical prognosis of patients (2). The mortality rate of toxic patients
after kidney injury is as high as 70% (3), whilst the survival rate of renal
replacement therapy is 13.8% (4).
Previous studies have reported that the pathophysiological
mechanism of septic AKI may be associated with overactivation of
the immune system, uncontrolled inflammation and dysfunction of the
coagulation system and energy metabolism (5,6).
The kidney has two sets of microcirculatory
structures, namely the glomerulus and peritubular microcirculatory
networks located in the renal cortex and renal medulla (7). The dense distribution and complex
structure of the capillary network of the kidney make it more
susceptible to oxidative stress and inflammation, rendering the
kidney highly susceptible to hypoperfusion and hypoxia (7). Adaptive changes occur in an
environment with unbalanced oxygen consumption, including apoptosis
of tubular epithelial cells (TECs) or even necrosis (8). In addition, abnormal activation of
white blood cells and coagulation disorders are also causes of AKI
(9). The aforementioned mechanisms
make the kidney one of the organs most susceptible to sepsis.
A previous study has demonstrated that the severity
of sepsis is associated with platelet activation (10). Platelets are widely involved in the
processes of coagulation, inflammatory factor release and immune
cell activation (9). Platelet
activation dependent granule external membrane protein (PADGEM or
CD62P, also known as P-selectin) protein is expressed on the
surface of the α-granule membrane secreted after platelet
activation and is a marker of platelet activation (11). It serves a key role in the process
of platelet-mediated immune cell activation (12). Ticagrelor is a novel anti-platelet
drug with a strong effect and is primarily used for the prevention
and treatment of acute coronary syndrome in clinical practice by
targeting the P2Y12 receptor (13).
It can inhibit platelet activation and reduce the secretion of
CD62P (14). In the present study,
the aim was to explore the mechanism of platelet activation of
CD62P protein in septic AKI, and the effect of ticagrelor in
decreasing the secretion of CD62 in a septic AKI rat model, in an
attempt to provide theoretical support for the prevention or
treatment of septic AKI.
Materials and methods
Animals and study design
Included in the present study were 50 healthy male
8-week-old SD rats (weighing 260-300 g; Chinese Academy of
Agricultural Sciences of Institute of Veterinary Medicine) after
adaptation to the standard laboratory conditions with free access
to food and water except modeling. The temperature in the vivarium
was 21-22˚C, the humidity was 40-60% and the day/night cycle was
13/11 h. The animals were equally randomized into five groups: i)
Normal group (without laparotomy and intervention); ii) sham group
(laparotomy without intervention); iii) group receiving cecal
ligation and puncture without any treatment (CLP); iv) group
receiving a clinical dose of 8.6 mg/kg body weight ticagrelor
(CCD); and v) group receiving a loading dose of 46.42 mg/kg body
weight of ticagrelor (CLD). Ticagrelor was administered via oral
gavage 12 h before modeling, immediately after modeling and 12 h
after modeling; the different time points were based on the
pharmacokinetics of ticagrelor according to the drug instructions
(15). Ticagrelor was provided by
the Department of Medicinal Chemistry, AstraZeneca Branch, Lanzhou,
China. The present study was approved by the Research Ethics
Committee of the First Hospital of Lanzhou University (Lanzhou,
China).
CLP critical procedure
After routine shaving and disinfection of the
abdominal area, a mid-line incision (~10 mm) was made after
successful induction of anesthesia via intraperitoneal (i.p.)
injection of 1% pentobarbital sodium (40 mg/kg). The cecum in the
lower left area of the abdominal cavity was exposed. Two transmural
injuries were made to the ligated section following cecal ligation
using polyglactin sutures using a 20-gauge needle to allow
peritoneal dissemination of bacteria. The abdominal cavity was then
closed by applying corresponding sutures, followed by fluid
resuscitation. Animals in the sham group underwent the same
procedure without cecal puncture and ligation, and animals in the
normal group did not undergo any procedures.
Preparation and detection of serum and
platelet-rich plasma (PRP)
After 24 h modeling, 1% pentobarbital sodium (40
mg/kg) was injected i.p. into the abdomen. Whole blood drawn from
the heart was centrifuged at 4˚C and 1,700 x g for 15 min to obtain
serum samples. Serum was stored at -20˚C, and serum creatinine
(SCr) and interleukin-1β (IL-1β) levels were measured using ELISAs
(SCr, cat. no. ml059158; https://www.mlbio.cn/goods-59158.html; IL-1β cat. no.
ml037361; https://www.mlbio.cn/goods-37361.html; Shanghai
Enzyme-linked Biotechnology Co., Ltd.). The remaining blood was
injected into the anticoagulant tube and centrifuged at 2,700 x g
for 5 min to isolate PRP at 4˚C, and the level of CD62P was
detected using an ELISA (cat. no. ml028431; Shanghai Enzyme-linked
Biotechnology Co., Ltd.; https://www.mlbio.cn/goods-28431.html). After the
blood was collected, the rats were still under anesthesia and were
euthanized using 25% potassium chloride (75 mg/kg) until the heart
stopped.
Kidney biopsy process and histological
lesions
After 24 h modeling and whole blood removal, rats
were euthanized using 25% potassium chloride (75 mg/kg) until the
heart stopped before the bilateral kidneys were removed. The right
kidney was immediately stored in a -80˚C refrigerator, and the left
kidney was fixed using 10% formaldehyde in room temperature
(20-25˚C) and >8 h, embedded in paraffin, sliced into 4-µm-thick
sections and stained using hematoxylin and eosin (H&E) 5 min in
room temperature. All the tissue sections were independently
evaluated in a blinded manner under an optical microscope by the
investigators (magnification, x400; cat. no. BX51-32H01; Olympus
Corporation). The scores were determined as follows: i) 0, not
detected, such that the glomeruli and renal tubules were unchanged;
ii) 1, where 1-10% tubules involved, part of the renal tubules were
occluded, the brush border of the renal tubular cells was absent;
iii) 2, 10-25% tubules involved; iv) 3, 25-50% tubules involved;
and v) 4, >50% of tubules involved (16).
Determination of renal tissue
homogenate
The right kidney tissue was prepared into a 10%
tissue homogenate with pre-cooled saline and renal cortex, and
centrifuged at 4˚C and 1,300 x g for 20 min to obtain the
supernatant. To determine the neutrophil content in the renal
tissue, an ELISA (cat. no. ml003250; Shanghai Enzyme-linked
Biotechnology Co., Ltd.; https://www.mlbio.cn/goods-3250.html) was used to
detect myeloperoxidase (MPO) activity, as the MPO level represents
an indicator of global tissue neutrophil recruitment (17).
Assessment of renal cell
apoptosis
This procedure was performed as previously described
(18). Fresh kidney tissue
fragments were added to a sufficient volume of type II collagenase
digestive solution (cat. no. 17101-015; Thermo Fisher Scientific,
Inc.) and digested in a thermostatic shaker at 37˚C for 5 min. The
protected sample was incubated for 15 min at 37˚C away from light
and gently agitated every 5 min. All supernatants were collected
and centrifuged at 4˚C and 70 x g for 5 min. The bottom sediment
was removed by filtration through a 200-mesh stainless steel mesh
to remove the residual tissue and then centrifuged at 30 x g for 2
min at 4˚C. The supernatant was discarded, and the isolated kidney
cells were collected and counted under a fluorescence microscope
(magnification, x400). The cells were stained and protected from
light using an FITC-Annexin V Apoptosis Detection kit at room
temperature (20-25˚C, stain for 15 min) stored at 4˚C (cat. no.
C1062S; Beyotime Institute of Biotechnology) and renal cell
apoptosis was detected using the BD FACSVerse™ flow cytometer (BD
Biosciences) analyzed by Flowjo (V10.5.2; BD Biosciences) and
inverted fluorescence microscopy (magnification, x400; cat. no.
IX53-DP73; Olympus Corporation). There was not enough renal tissue
to repeat the staining protocol whilst including a stain such as
DAPI in the fluorescence experiments, which represented a
limitation of the present study.
Statistical analysis
Statistical analysis was performed using the SPSS
16.0 statistical software package (SPSS, Inc.). The measurement
data are expressed as the mean ± SD. One-way ANOVA and Tukey's post
hoc test were used for comparison between groups.
Non-parametrically distributed data was analyzed using
Kruskal-Wallis with Dunn's post hoc test. P<0.05 was considered
to indicate a statistically significant difference.
Results
General condition of rats
No significant change was noticed in the rats of the
normal group. The rats in the sham group were all in a good mental
condition after surgery, with normal water feeding, stable
breathing and sensitive to stimuli. Meanwhile, septicemia symptoms,
such as refusal to eat and drink, drowsiness, hair erection and
diarrhea were observed in CLP group, and hematuria appeared in some
rats. Compared with the CLP group, the aforementioned conditions
were milder in CCD and CLD groups, but more severe than in the sham
group.
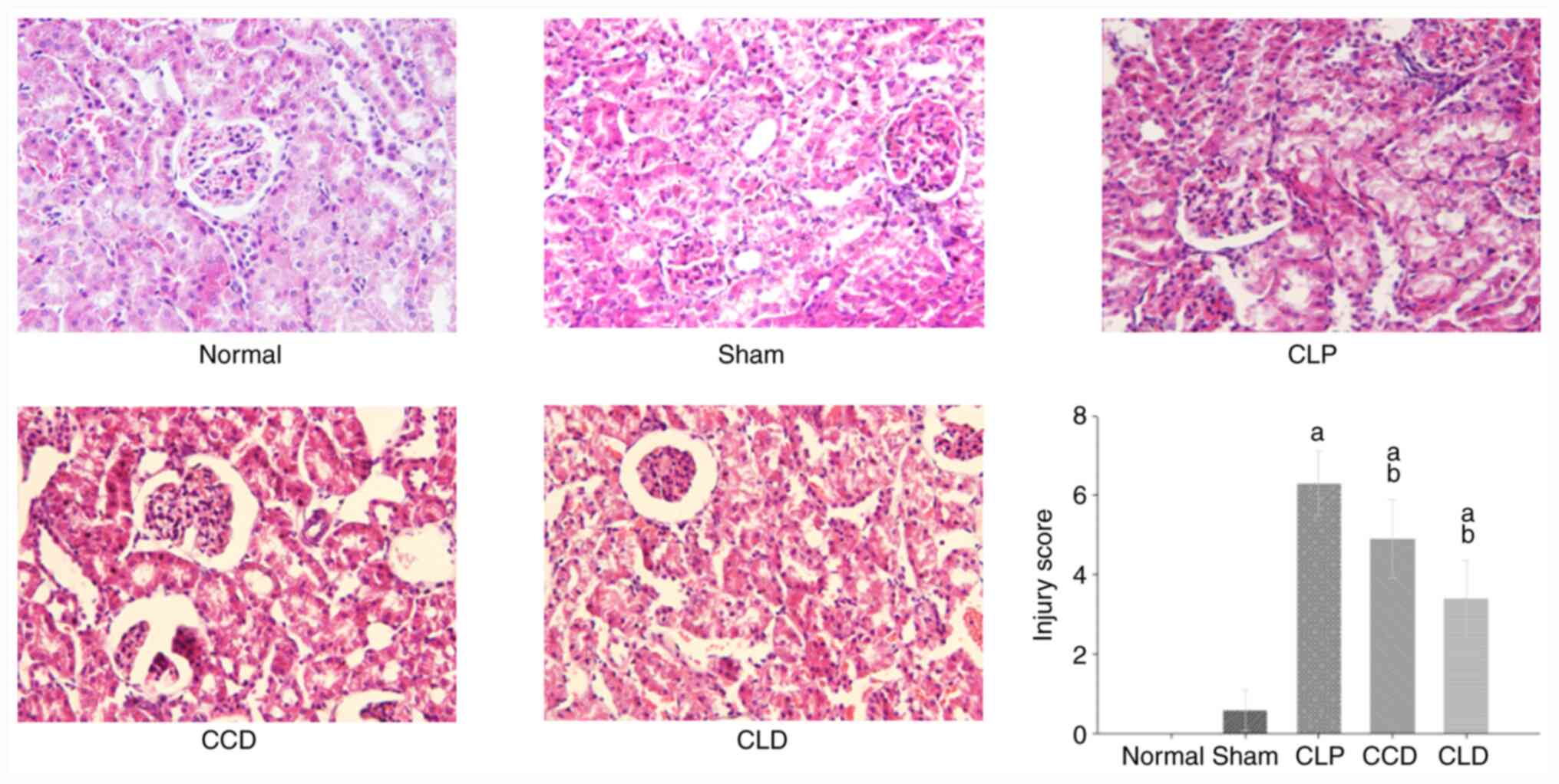
Histological findings
The effect of ticagrelor on sepsis-induced renal
histological alterations was evaluated using H&E staining
(16). The results indicated that
the glomeruli and renal tubules were unchanged in the normal group,
where cells were arranged regularly, the structure was intact, the
nucleus was central, the nucleolus was clear and the plasma was
evenly stained. The findings in sham group were similar to those in
the normal group, except that the tubular cells were slightly
edematous. In the CLP group, interstitial congestion, edema and
inflammatory cell infiltration were observed in the renal tissue,
and part of the renal tubules were occluded, the brush border of
the renal tubular cells was absent, and the top surface of the cell
membrane was vacuolated and protruded into the lumen. Parts of the
renal tubules were compensated for dilatation, the epithelium of
the tube was flat, the nucleus disappeared, the basement membrane
was exposed and the Tamm-Horsfall protein cast was observable. In
the CCD group, the structure of the renal tubules was less damaged,
with no luminal occlusion, less inflammatory cell infiltration,
slight damage to the renal tubular epithelial cells and edema of
vacuolated changes. In the CLD group, only tubular cell edema was
seen, and no inflammatory cell infiltration was observed. Compared
with the sham group, the injury score were increased in the CLP,
CCD and CLD groups (P<0.05). Moreover, compared with the CLP
group, the injury score were decreased in the CCD and CLD groups
(P<0.05; Fig. 1).
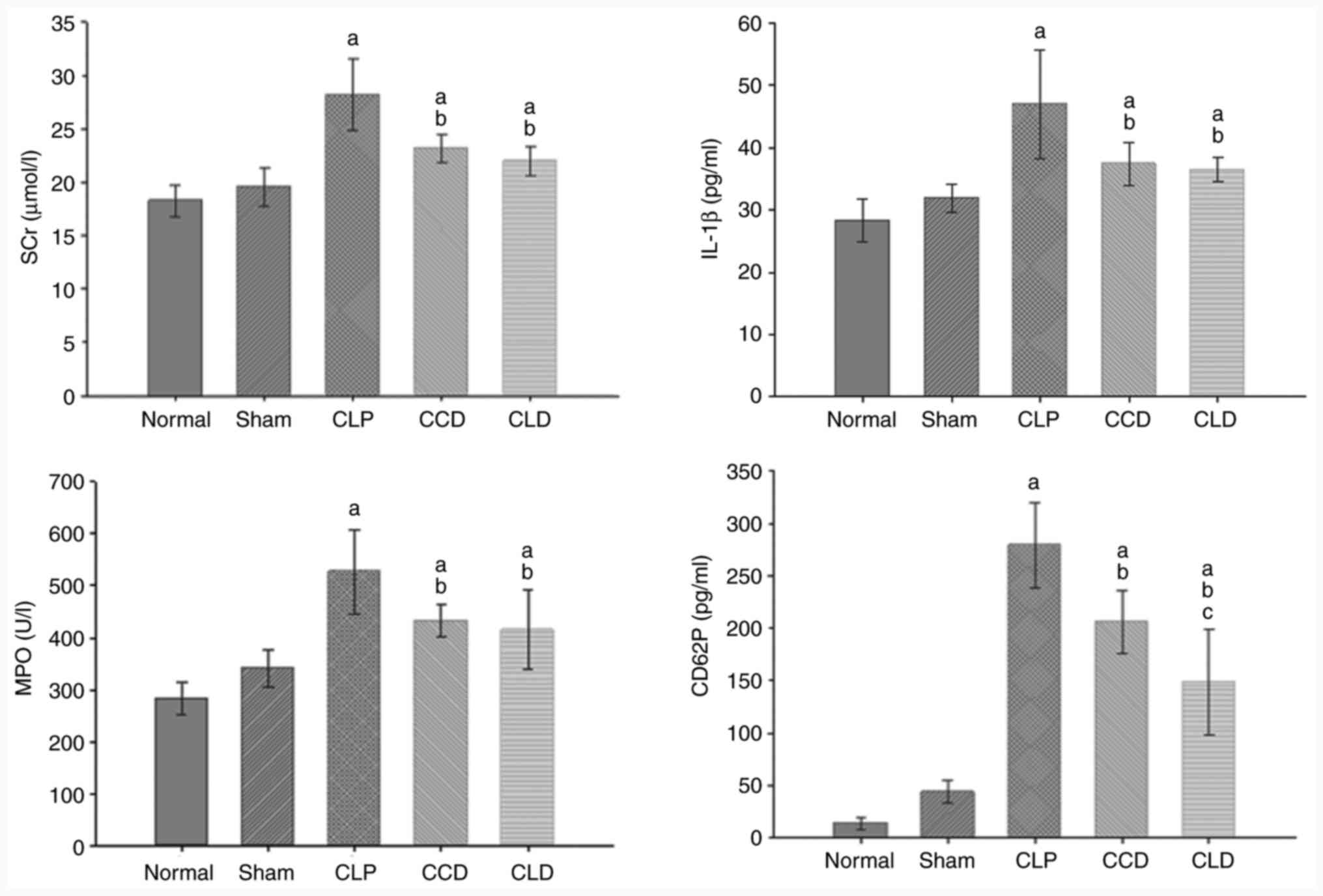
Changes in SCr, CD62P, IL-1β and renal
tissue MPO levels
Renal function is usually represented by SCr levels,
platelet activity by CD62P levels, the degree of neutrophil
accumulation in the kidney by MPO activity, and inflammatory factor
levels by IL-1β levels (19-22).
Compared with the normal and sham groups, SCr, CD62P, IL-1β and
renal tissue MPO were increased in the CLP group (P<0.05).
Moreover, compared with the CLP group, SCr, CD62P, IL-1β and renal
tissue MPO were decreased in the CCD and CLD groups (P<0.05).
Finally, compared with the CCD group, CD62P in the PRP of the CLD
group was decreased (P<0.05; Fig.
2).
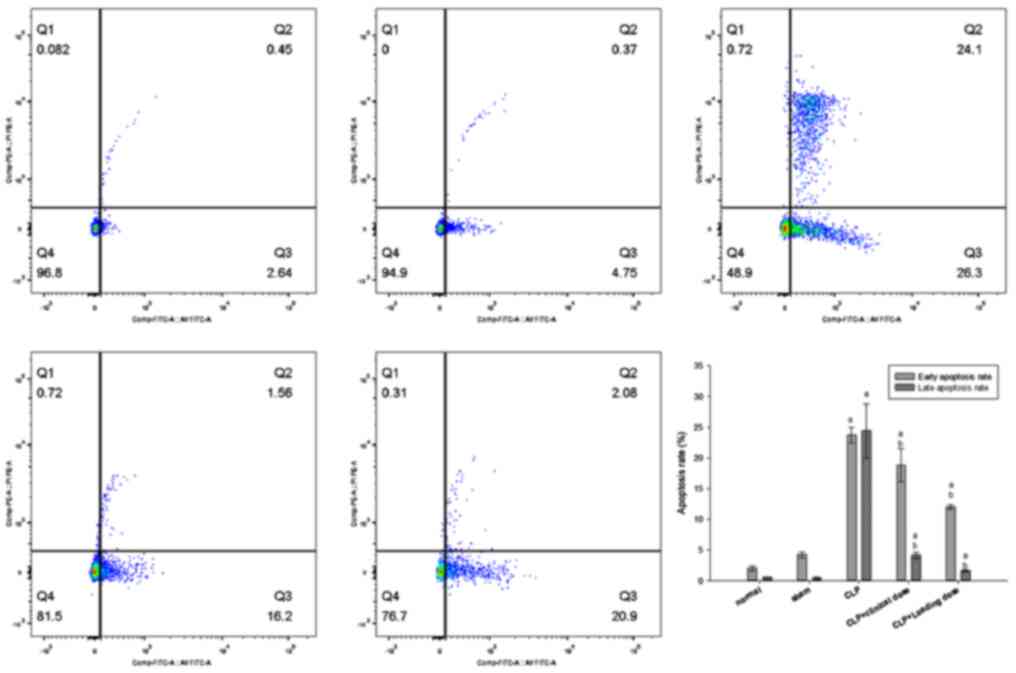
Cell apoptosis in the kidney Flow
cytometric analysis of the renal cell apoptosis rate
In the normal and sham groups, some early apoptotic
cells of Annexin V+/propidium iodide (PI)-
were observed, but very few Annexin V+/PI+
intermediate and advanced apoptotic cells or secondary necrotic
cells were observed. Compared with the normal and sham groups, a
large number of Annexin V+/PI- early
apoptotic cells and Annexin V+/PI+
intermediate-to-advanced apoptotic cells or secondary necrotic
cells were observed in the CLP group, with significant differences
between the groups (P<0.05). Compared with the CLP group, the
number of early, intermediate and advanced apoptotic cells was
significantly decreased in the CCD and CLD groups (P<0.05). The
change in renal cell apoptosis was consistent with that of SCr in
all groups, suggesting that renal cell apoptosis was associated
with septic AKI (Fig. 3).
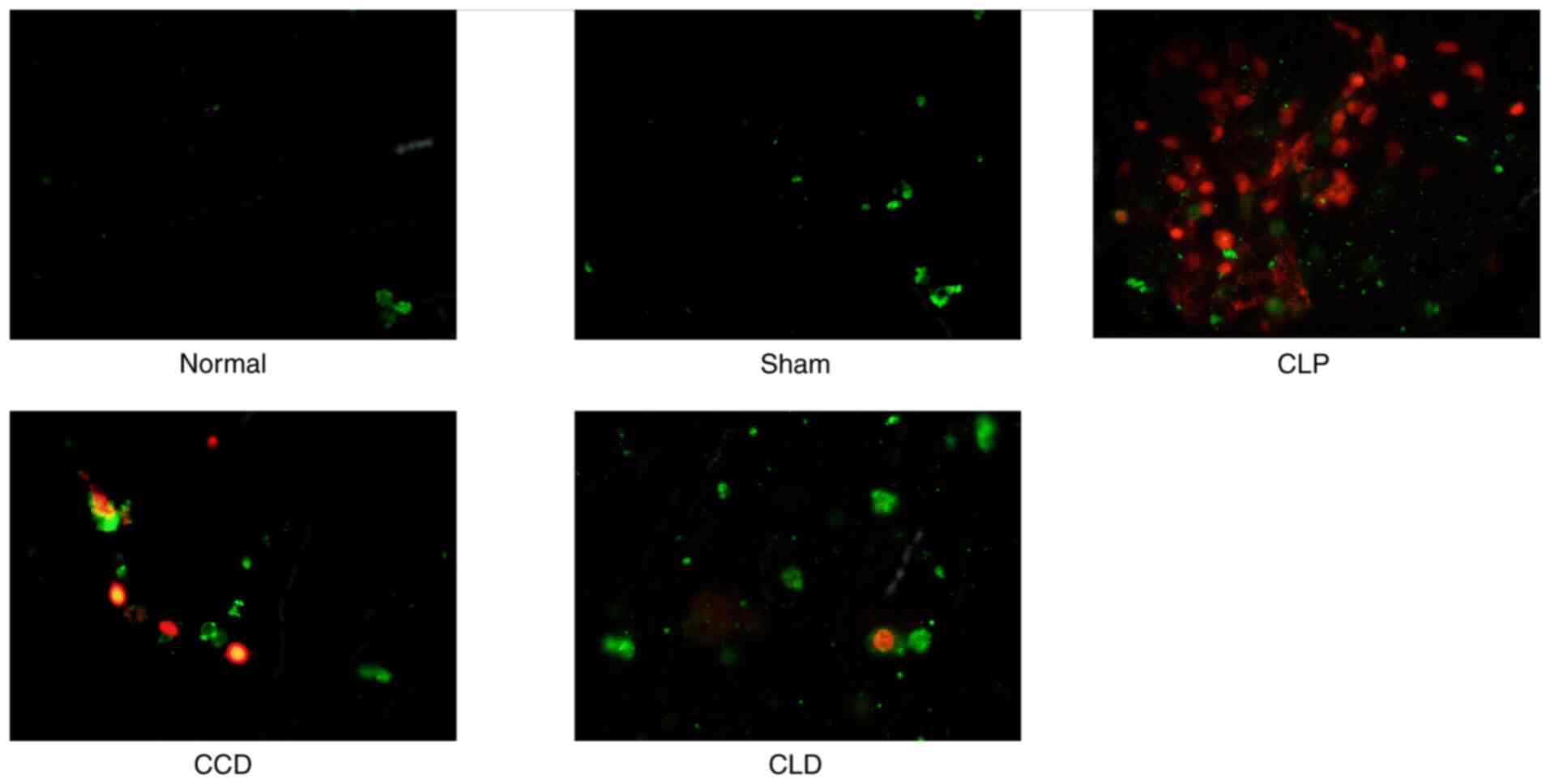
Inverted fluorescence microscopy of
renal cell apoptosis
Inverted fluorescence microscopy for double-stained
cells revealed that: i) The early apoptotic cell membrane showed
green fluorescence (FITC fluorescence); ii) the intermediate and
advanced stage apoptotic cell nucleus exhibited red fluorescence
(PI fluorescence) and the cell membrane displayed green
fluorescence; and iii) only red fluorescent nuclei were seen in
late apoptotic and dead cells, and the cell membranes were
disintegrated. Few green fluorescent cell membranes were observed
in the normal and sham groups. Large numbers of cells were observed
with red-fluorescent nuclei and green-fluorescent membranes or with
red-fluorescent nuclei only in the CLP group, while in the CCD and
CLD groups there were primarily green fluorescence-stained cells,
and some nuclei were stained with red fluorescence. In addition,
there were more cells that stained red in the CCD group compared
with CLD groups (Fig. 4).
Discussion
Ticagrelor is widely used for the prevention and
treatment of acute coronary syndrome in clinical practice. It is
the first reversible P2Y12 receptor antagonist that can effectively
decrease platelet activation and aggregation (8). Ticagrelor inhibits further release of
the platelet agonist adiponectin from dense particles by
antagonizing the P2Y12 receptor, thereby achieving the effect of
inhibiting platelet activation and aggregation (23). Compared with clopidogrel, ticagrelor
has a more potent antiplatelet effect (14). Moreover, pharmacokinetics reveal
that the inhibitory effect of ticagrelor on platelets is
concentration-dependent, there is a longer time window between
antithrombotic and bleeding accidents, and its clinical safety is
higher (23). A previous study have
demonstrated that ticagrelor can also mimic the anti-inflammatory
effect of adenosine (24). However,
there are few studies reporting the use of ticagrelor for the
treatment of septic AKI. The aim of the present study was to
explore the association between CD62P and septic AKI, and the
effect of ticagrelor on septic AKI.
Sepsis is a serious medical condition with complex
pathogenesis, resulting in high morbidity and mortality (25). The exact molecular mechanisms
underlying this disease are yet to be elucidated and effective
therapeutic targets need to be developed. In the present study, a
rat model of sepsis-induced AKI was established via CLP.
Postoperatively, the rats were generally presented in a poor
condition. Pus was observed in the abdominal cavity of the
sacrificed animals and the ligated cecal segments became black and
necrotic. SCr level was increased and the pathological sections
revealed that the kidney tissue was severely damaged. All these
findings indicated that the rat septic AKI model was constructed
successfully.
A clinical study has previously demonstrated that
the severity of sepsis was associated with platelet activation
(16). During the activation of
platelets after sepsis, CD62P is converted from the internal
storage protein of the cell to a platelet membrane surface protein,
eventually being expressed on the surface of platelet cell
membranes as cell adhesion receptors; thus, it represents a marker
for detecting platelet activation (26). The present study demonstrated that
the CD62P concentration in septic AKI animals of the CLP group was
higher than that in normal and sham groups, indicating that CD62P
was to some extent associated with septic AKI.
Lerolle et al (27) found extensive infiltration of
neutrophils in the glomeruli, renal interstitia and capillaries
upon autopsy of patients with sepsis AKI. The authors therefore
speculated that accumulation of neutrophils in the kidney was
involved in the pathogenesis of septic AKI. In the present study,
in the CLP group, H&E staining of the renal tissue revealed
that inflammatory cells accumulated in the kidney, renal tubular
epithelial cells were damaged and Tamm-Horsfall protein casts were
observed, indicating that the occurrence of septic AKI was partly
associated with the accumulation of renal inflammatory cells.
To determine the association of neutrophils and
their secreted factors with septic AKI, compared with those in the
Normal group and the Sham group, serum IL-1β levels and MPO
activity were detected in the renal tissue, and it was revealed
that both MPO activity and IL-1β levels were increased in the CLP
group, indicating that neutrophil infiltration in the renal tissue
and imbalance of the inflammatory response mediated septic AKI, and
that this mechanism was associated with the increase of CD62P
concentration. As a cell adhesion receptor, CD62P mediates
P-selectin glycoprotein ligand-1 and glycoprotein Ib interactions
between platelets and neutrophils, triggering a series of cellular
responses within neutrophils (28).
Notably, other studies reported that CD62P protein directly induced
the expression of IL-1β and other cytokines, while IL-1β is an
important pro-inflammatory factor that could promote the bone
marrow to release neutrophils, chemotactic monocytes and
multinuclear cells, and infiltrate the local inflammation (29,30).
To summarize, CD62P mediates the accumulation of
neutrophils in the kidney, which is the initiating factor of AKI in
sepsis. The activation of platelet mass initiates disseminated
intravascular coagulation, which causes microcirculation
disturbance of the renal tissue, and aggravates local circulatory
disorders and inflammation unbalanced response in renal tissue
(31). The aforementioned factors
work synergistically to induce the development of septic AKI.
It was demonstrated in the present study that SCr,
CD62P, IL-1β and renal MPO were decreased after ticagrelor
intervention in the CCD and CLD groups, and H&E staining of
renal tubular epithelial cells and renal tubules was mild with no
tubular deposition observed. In addition, the decrease in CD62P via
ticagrelor concentration was associated with similar reductions in
the renal injury marker SCr, the proinflammatory factor IL-1β and
MPO, the aggregation marker of central granulocytes in the kidney,
suggesting that ticagrelor attenuated the severity of septic AKI by
inhibiting the secretion of CD62P and the inflammatory response
mediated by CD62P, such as reducing the release of proinflammatory
factor IL-1β, inflammatory response and chemotaxis of neutrophils,
thereby reducing neutrophil accumulation in the kidney. At the same
time, the expression of CD62P in the CCD group was significantly
higher than the CLD group. It is speculated that there is a
dose-dependent association between the inhibition of CD62P
expression and ticagrelor. Certain scholars hypothesize that
ticagrelor may become one of the novel targeted drugs for the
treatment of septic AKI (32).
In the current study, it was demonstrated that the
apoptosis rate of renal cells in CLP group was increased compared
with the normal and sham groups, suggesting that renal cell
apoptosis is one of the pathophysiological mechanisms underlying
septic AKI. Mariano et al (33) used the plasma from patients with
severe sepsis or septic shock to culture renal TECs and found that
TECs still underwent functional changes and apoptosis even without
hypoperfusion or hypoxia; therefore, ischemia and hypoxia may not
be the primary factors of renal cell apoptosis. In the present
study, it was revealed that the apoptosis rate was decreased in the
CCD and CLD groups compared with the CLP group, suggesting that the
inhibition of CD62P secretion using ticagrelor reduced the
apoptosis rate of renal cells. The lack of caspase-3 level
measurements and TUNEL staining of tissue slices are potential
limitations of the current study.
In summary, the present study demonstrated that
sepsis results in excessive platelet activation, causing an
increase in the blood CD62P protein concentration and the renal
cell apoptosis rate, which may be one of the key pathophysiological
mechanisms underlying septic AKI. In sepsis, the abnormal increase
in CD62P protein induces the release of the pro-inflammatory factor
IL-1β, which adheres to neutrophils, indicating that CD62P-mediated
inflammation and renal cell apoptosis are the key mechanisms
underlying the development of septic AKI (34). Ticagrelor exerts certain renal
protective effects by inhibiting the inflammatory response mediated
by CD62P, alleviating systemic and renal local inflammation and
reducing renal cell apoptosis. These findings may provide novel
evidence-based insight and experimental information to aid in the
prevention and treatment of septic AKI. In the current study, only
animal experiments were conducted; in future, the mechanism
underlying ticagrelor regulation of CD62P expression (potentially
via the NF-κB pathway) should be examined at the cellular level and
alleviating the sepsis-induced AKI.
Acknowledgements
The authors would like to thank Dr Yan-Long Pan
(Department of Research and Test Center, Gansu University of
Traditional Chinese Medicine, Lanzhou, China) for his help in the
guidance of the experimental methods and Professor Shun Xing Zhang
(Foreign Language Teaching and Research Section, Second Military
Medical University, Shanghai, China), who helped revise the English
in the paper.
Funding
The present work was supported by the First Hospital of Lanzhou
University (grant no. ldyyyn2018-49) from the fund program named
Fund of First Hospital of Lanzhou University.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
CY made substantial contributions to conception and
design, performed the experiments and participated in drafting the
manuscript. CMG has confirmed the authenticity of all the raw data,
and revising the manuscript critically for important intellectual
content. CMG has been involved in analyzing the data and drafting
the manuscript. YQM made substantial contributions to design the
experiment and critically revised the manuscript for important
intellectual content. NX and XQW contributed to acquisition and
analysis of data, YQM critically revised the manuscript for
important intellectual content. All authors have read and approved
the final version of this manuscript.
Ethics approval and consent to
participate
The present study was approved by the Research
Ethics Committee of the First Hospital of Lanzhou University
(Lanzhou, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Thakar CV, Christianson A, Freyberg R,
Almenoff P and Render ML: Incidence and outcomes of acute kidney
injury in intensive care units: A Veterans administration study.
Crit Care Med. 37:2552–2558. 2009.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Kim WY, Huh JW, Lim CM, Koh Y and Hong SB:
A comparison of acute kidney injury classifications in patients
with severe sepsis and septic shock. Am J Med Sci. 344:350–356.
2012.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Peng Q and Zhang L, Ai Y and Zhang L:
Epidemiology of acute kidney injury in intensive care septic
patients based on the KDIGO guidelines. Chin Med J (Eng).
127:1820–1826. 2014.PubMed/NCBI
|
|
4
|
Chen YJ, Gong CL and Tan F: Effects of
dexmedetomidine pretreatment on inflammatory factors and oxidative
stress in rats with sepsis-induced renal injury. J Southern Med
Univ. 35:1472–1475. 2015.
|
|
5
|
Engelmann B and Massberg S: Thrombosis as
an intravascular effector of innate immunity. Nat Rev Immunol.
13:34–45. 2013.PubMed/NCBI View
Article : Google Scholar
|
|
6
|
Schrier RW and Wang W: Acute renal failure
and sepsis. N Engl J Med. 35:159–169. 2004.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Ergin B, Kapucu A, Demirci-Tansel C and
Ince C: The renal microcirculation in sepsis. Nephrol Dial
Transplantat. 30:169–177. 2015.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Evans RG, Ince C, Joles JA, Smith DW, May
CN, O'Connor PM and Gardiner BS: Haemodynamic influences on kidney
oxygenation: Clinical implications of integrative physiology. Clin
Exp Pharmacol Physiol. 40:106–122. 2013.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Levy MM, Fink MP, Marshall JC, Abraham E,
Angus D, Cook D, Cohen J, Opal SM, Vincent JL and Ramsay G:
SCCM/ESICM/ACCP/ATS/SIS. 2001 SCCM/ESICM/ACCP/ATS/SIS international
sepsis definitions conference. Crit Care Med. 31:250–1256.
2003.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Yu LP: Mesh Meta-analysis of the
differences in the effects of different ADP receptor antagonists on
platelet function in patients with coronary heart disease.
Practical Drugs Clini, 2016.
|
|
11
|
Thomas MR and Storey RF: The role of
platelets in inflammation. Thromb Haemost. 114:449–458.
2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Woollard KJ and Chin-Dusting J: P-selectin
antagonism in inflammatory disease. Curr Pharm Des. 16:4113–4118.
2010.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wang Y and Chai YF: Relationship of sepsis
severity and platelet change. Pract Clin Med (Jiangxi). 19:16–18.
2018.
|
|
14
|
Gu X, Fu X, Wang Y, Zhang W, Fan W, Jiang
Y, Hao G, Miao Q, Li Y and Zhi W: Comparison of ticagrelor and
high-dose clopidogrel on the platelet functions in patients with
inadequate response to clopidogrel. Am J Cardiovasc Dis. 7:1–8.
2017.PubMed/NCBI
|
|
15
|
Zhao W and Sun GZ: Conversion of dosage
between different kinds of experimental animals. Animal Husbandry
Veterinary Sci Techn Infor. 5:52–53. 2012.
|
|
16
|
Jin H, Zhang Y, Ding Q, Wang SS, Rastogi
P, Dai DF, Lu D, Purvis M, Cao C, Wang A, et al: Epithelial innate
immunity mediates tubular cell senescence after kidney injury. JCI
Insight. 4(e125490)2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Singbartl K and Ley K: Leukocyte
recruitment and acute renal failure. J Mol Med (Berl). 82:91–101.
2004.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Gu G, Gao F, Zhi Y and Yin ZX: Comparison
of methods of isolation, preparation and culture about rat kidney
cells. Wei Sheng Yan Jiu. 34:574–576. 2005.PubMed/NCBI(In Chinese).
|
|
19
|
Wasung ME, Chawla LS and Madero M:
Biomarkers of renal function, which and when? Clin Chim Acta.
438:350–357. 2015.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Yip HK, Chen SS, Liu JS, Chang HW, Kao YF,
Lan MY, Chang YY, Lai SL, Chen WH and Chen MC: Serial changes in
platelet activation in patients after ischemic stroke: Role of
pharmacodynamic modulation. Stroke. 35:1683–1687. 2004.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Mullane KM, Kraemer R and Smith B:
Myeloperoxidase activity as a quantitative assessment of neutrophil
infiltration into ischemie myocardium. J Pharmacol Methods.
14:157–167. 1985.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Meduri GU, Headley S, Kohler G, Stentz F,
Tolley E, Umberger R and Leeper K: Persistent elevation of
inflammatory cytokines predicts a poor outcome in ARDS: Plasma
IL-1beta and IL-6 levels are consistent and efficient predictors of
outcome over time. Chest. 107:1062–1073. 1995.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Husted S and Van Giezen JJ: Ticagrelor:
The first reversibly binding oral P2Y12 receptor antagonist.
Cardiovas Ther. 27:259–274. 2009.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Cattaneo M, Schulz R and Nylander S:
Adenosine-mediated effects of ticagrelor: Evidence and potential
clinical relevance. J Am Coll Cardiol. 63:2503–2509.
2014.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Fani F, Regolisti G, Delsante M,
Cantaluppi V, Castellano G, Gesualdo L, Villa G and Fiaccadori E:
Recent advances in the pathogenetic mechanisms of sepsis-associated
acute kidney injury. J Nephrol. 31:351–359. 2018.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Furie B, Furie BC and Flaumenhaft R: A
journey with platelet P-selectin: The molecular basis of granule
secretion, signalling and cell adhesion. Thromb Haemost.
86:214–221. 2001.PubMed/NCBI
|
|
27
|
Lerolle N, Nochy D, Guérot E, Bruneval P,
Fagon JY, Diehl JL and Hill G: Histopathology of septic shock
induced acute kidney injury: Apoptosis and leukocytic infiltration.
Intensive Care Med. 36:471–478. 2010.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Chappell D, Heindl B, Jacob M, Annecke T,
Chen C, Rehm M, Conzen P and Becker BF: Sevoflurane reduces
leukocyte and platelet adhesion after ischemia-reperfusion by
protecting the endothelial glycocalyx. Anesthesiologists.
115:483–491. 2011.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Jansen MP, Florquin S and Roelofs JJ: The
role of platelets in acute kidney injury. Nat Rev Nephrol.
14:457–471. 2018.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Lukens JR, Gurung P, Vogel P, Johnson GR,
Carter RA, McGoldrick DJ, Bandi SR, Calabrese CR, Vande Walle L,
Lamkanfi M and Kanneganti TD: Dietary modulation of the microbiome
affects autoinflammatory disease. Nature. 516:246–249.
2014.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Wang TY and Zhang ZX: Progress in
treatment of diffuse intravascular coagulation. Chin Pediatric
Emergency Med. 15:405–407. 2008.
|
|
32
|
Sexton TR, Zhang G, Macaulay TE, Callahan
LA, Charnigo R, Vsevolozhskaya OA, Li Z and Smyth S: Ticagrelor
reduces thromboinflammatory markers in patients with pneumonia.
JACC Basic Transl Sci. 3:435–449. 2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Mariano F, Cantaluppi V, Stella M,
Romanazzi GM, Assenzio B, Cairo M, Biancone L, Triolo G, Ranieri VM
and Camussi G: Circulating plasma factors induce tubular and
glomerular alterations in septic burns patients. Crit Care.
12(R42)2008.PubMed/NCBI View
Article : Google Scholar
|
|
34
|
Li X, Li Y, Shen K, Li H and Bai J: The
protective effect of ticagrelor on renal function in a mouse model
of sepsis-induced acute kidney injury. Platelets. 30:199–205.
2019.PubMed/NCBI View Article : Google Scholar
|