Introduction
The pandemic created by SARS CoV-2 infection still
represents a pressing medical problem considering the multitude of
risk factors for severe disease and the lack of specific symptoms
(1-3).
Sanitary education of the population and vaccination have served an
essential role in prophylaxis by helping individuals understand the
risks they are exposed to (4-7).
Literature highlights that cutaneous manifestation
of SARS CoV-2 infection presents as lesions with varying morphology
that could be classified in four categories: Acro-papular lesions,
urticarial eruption, vascular (chilblain-like lesions, commonly
known as COVID-19 toes, livedoid and purpuric lesions) and
exanthema (morbilliform and papulo-vesicular rash and
varicella-like eruption) (8-13).
Stevens-Johnson syndrome (SJS) has the potential to
be a lethal skin reaction that has a mortality rate of up to 30%,
which is caused by an immune-complex-mediated hypersensitivity
reaction. The clinical presentation appears as mucosal and
cutaneous tenderness accompanied by erythema, hemorrhagic erosions,
and epidermal detachment that can be described as blisters and
areas of denuded skin, accompanied by systemic symptoms (14,15).
This disease is a dermatological emergency. The recognition in
association with prompt and appropriate management can save the
patient (16). The present study is
a case report of a 77-year-old male with a metabolic, cardiologic
and neurological history diagnosed with SARS CoV-2 infection
associated with SJS. Few cases have been reported concerning this
association, which raises the question of whether, in the case of
our patient, SJS appeared independently from COVID-19 or was the
primary manifestation of the disease (17-23).
Case report
The present article reports the case of a
77-year-old male patient with a history of stroke, stage-2 arterial
hypertension, dyslipidemia, obesity and gout, together with an
underlying treatment: Aspirin, 75 mg; bisoprolol, 2.5 mg bidaily;
atorvastatin, 10 mg/day; vinopectine, 10 mg bidaily; and
allopurinol, 100 mg bidaily. The gout medication was prescribed 14
days before admission to our hospital.
Initially, the patient presented to the Emergency
Room of Sf. Spiridon County Hospital for a non-pruriginous
generalized maculopapular-erythematous eruption with a tendency
towards confluence, accompanied by low back pain, headache and
orbital pain. Considering the epidemiological context, a reverse
transcription PCR for SARS CoV-2 virus and a CT scan were
performed. The result of the molecular test was positive, and the
CT examination demonstrated bilateral centrilobular emphysema and
bilateral apical pachypleuritis. In the inferior two-thirds of the
lungs, bilateral, extensive areas of pulmonary condensation were
observed that were predominantly located subpleurally,
heterogeneous and imprecise. Based on these results, the patient
was directed to Sf. Parascheva Clinical Hospital of Infectious
Diseases, which was a designated first-line COVID-19 hospital.
At admission, the patient had a general fair status
and was conscious. He was experiencing bradylalia, but stable both
hemodynamically and in terms of respiration (blood pressure, 106/67
mmHg; heart rate, 95 beats/min; oxygen saturation, 98% ambient
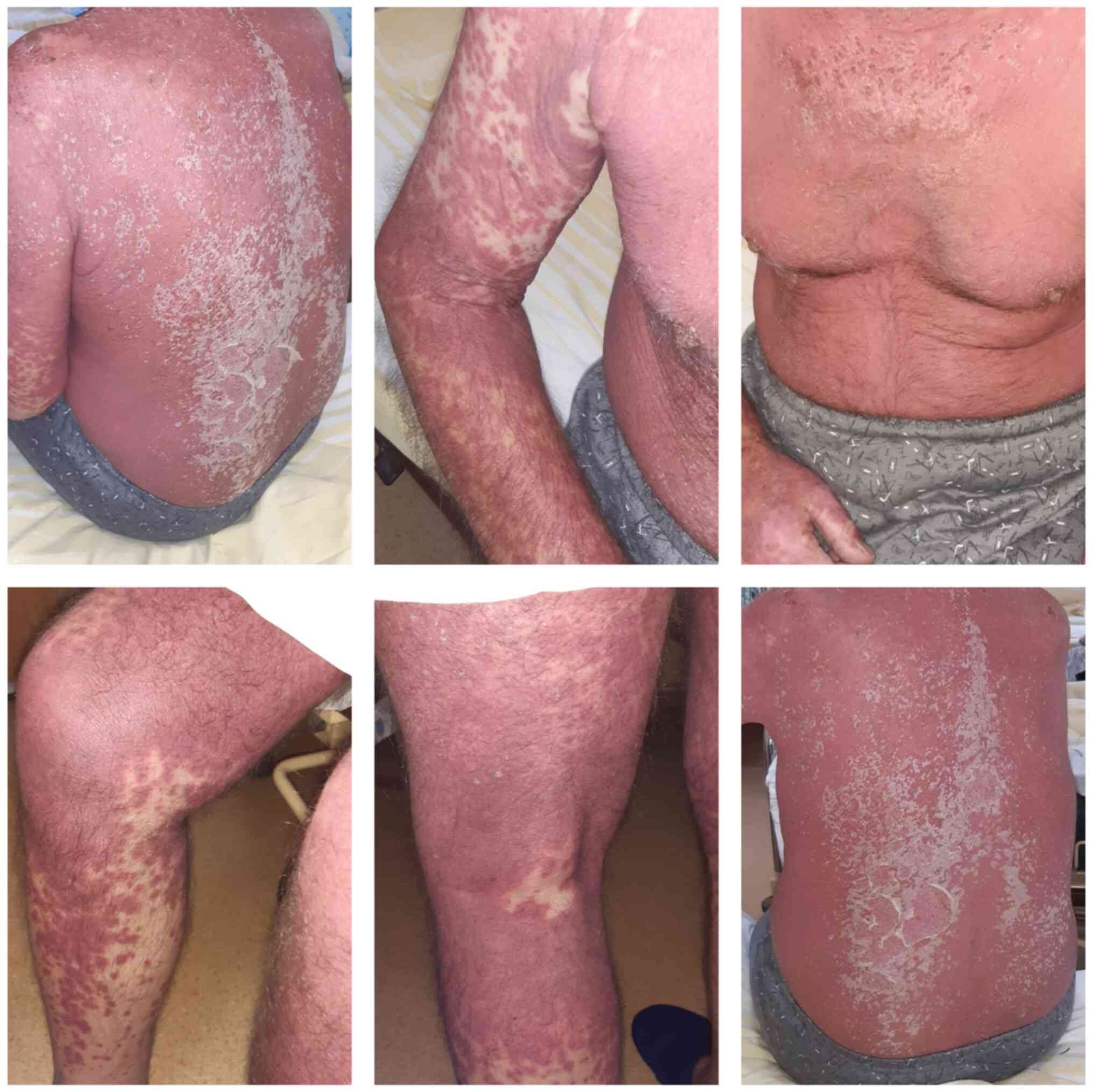
air). This was associated with the aforementioned lesions, as well
as peri-oronasal meliceric crusts and desquamation of the skin on
the third anterosuperior and posterior thorax, scalp and forehead
(Fig. 1).
Considering the clinical and paraclinical evidence
(Table I), the suspicion of SJS was
raised, and a dermatological consultation was requested, which
confirmed the diagnosis. The recommendations were to stop the
administration of allopurinol and administer methylpredinisolone at
250 mg/day, 20 mg bilastine bidaily, vitamin C intravenously at 500
mg bidaily, gluconic calcium at 10 ml/day (94 mg/ml), vitonal and
gentamicin cream (applied bidaily on the lesions located on the
peri-oronasal area) and a cream consisting of 5 g urea, 1 g
hydrocortisone and 100 g Vaseline® (applied bidaily over
all affected areas). In addition, antibiotic (meropenem, 4 g/day;
linezolid, 1.2 g/day), anticoagulant (enoxaparine sodium, 0.6 mg
bidaily), acetaminophen (500 mg) and acetylcysteine (600 mg/daily)
were administered.
 | Table ILaboratory data. |
Table I
Laboratory data.
| | Date of
measurement |
|---|
| Parameter | 20.11 | 23.11 | 24.11 | 26.11 | 30.11 | 3.12 |
|---|
| Leukocytes (per
mm3) | 27,840 | NA | 12,820 | 12,320 | 9,690 | 10,210 |
| Neutrophil (%) | 70.70 | NA | 79.2 | 82.3 | 87.3 | 83.40 |
| Lymphocytes (%) | 11.20 | NA | 14.3 | 11 | 9.2 | 13.50 |
| Platelets (per
mm3) | 349,000 | NA | 274,000 | 233,000 | 79,000 | 117,000 |
| C-reactive protein
(mg/l) | 27.3 | NA | 31.57 | NA | 58.66 | 53.46 |
| ESR (mm/h) | 18 | NA | 20 | NA | 40 | 85 |
| INR | 1.33 | NA | NA | NA | | 2.42 |
| Fibrinogen
(g/l) | 1.8 | NA | NA | NA | 3.29 | 3.29 |
| IL-6 (pg/ml) | | 27.19 | NA | NA | NA | NA |
| D-dimer | 1235 | NA | NA | NA | NA | NA |
| Urea (mg/dl) | 172 | NA | 86 | 85 | 102 | 118 |
| Creatinine
(mg/dl) | 1.75 | NA | 1.2 | 0.95 | 0.96 | 1.11 |
| Glucose
(mg/dl) | 140 | NA | 111 | NA | 103 | 103 |
| Na (mmol/l) | 141 | NA | 146.1 | 146.6 | 146.7 | 146.7 |
| K (mmol/l) | 4.37 | NA | 3.99 | 4.05 | 4.80 | 4.58 |
| Cl (mmol/l) | 97.7 | NA | 102.6 | 103.1 | 105.2 | 105.4 |
| HCO3
(mmol/l) | NA | 21.4 | NA | 13.6 | NA | NA |
| ALT(U/l) | 37 | NA | 62 | NA | 63 | 58 |
| AST(U/l) | 39 | NA | 80 | NA | 88 | 88 |
| Bilirubin
(mg/dl) | 1.25 | NA | 1.31 | 1.60 | 1.15 | 2.64 |
| Ionic calcium
(mg/dl) | NA | NA | NA | 4.72 | NA | 4.40 |
| HIV serology | NA | Negative | NA | NA | NA | NA |
| LDH (U/l) | NA | NA | NA | NA | 609 | NA |
| Total protein
(g/l) | 60.78 | NA | NA | NA | 75.21 | NA |
| Ferritin
(ng/ml) | NA | 511 | NA | NA | NA | NA |
The algorithm of drug causality for epidermal
necrolysis (ALDEN) and the severity-of-illness score for toxic
epidermal necrolysis (SCORTEN) were calculated. The ALDEN score for
the patient was 5, corresponding to a ‘probable’ causal link,
suggesting that the implicated drug in our case could be
allopurinol (Table II). In
addition, the SCORTEN was 3 for this patient, indicating a
mortality rate of 35.3% (Table
III).
 | Table IIALDEN results for allopurinol. |
Table II
ALDEN results for allopurinol.
| Score | Value |
|---|
| Delay from initial
drug intake to index day | +3 |
| Drug present in the
body (on index day) | 0 |
|
Pre-challenge/Re-challenge | 0 |
| De-challenge | 0 |
| Type of drug
(notoriety) | +3 |
| Other cause | -1 |
| Total ALDEN
score | 5a |
 | Table IIISCORTEN score. |
Table III
SCORTEN score.
| Prognostic
factor | Score |
|---|
| Age >40
years | 1 |
| Associated
cancer | 0 |
| Heart rate >120
bpm | 0 |
| Serum blood urea
>28 mg/dl | 1 |
| Detached or
compromised body surface >10% | 1 |
| Serum bicarbonate
<20 mmol/l | 0 |
| Serum glucose
>250 mg/dl | 0 |
| Total SCORTEN | 3 |
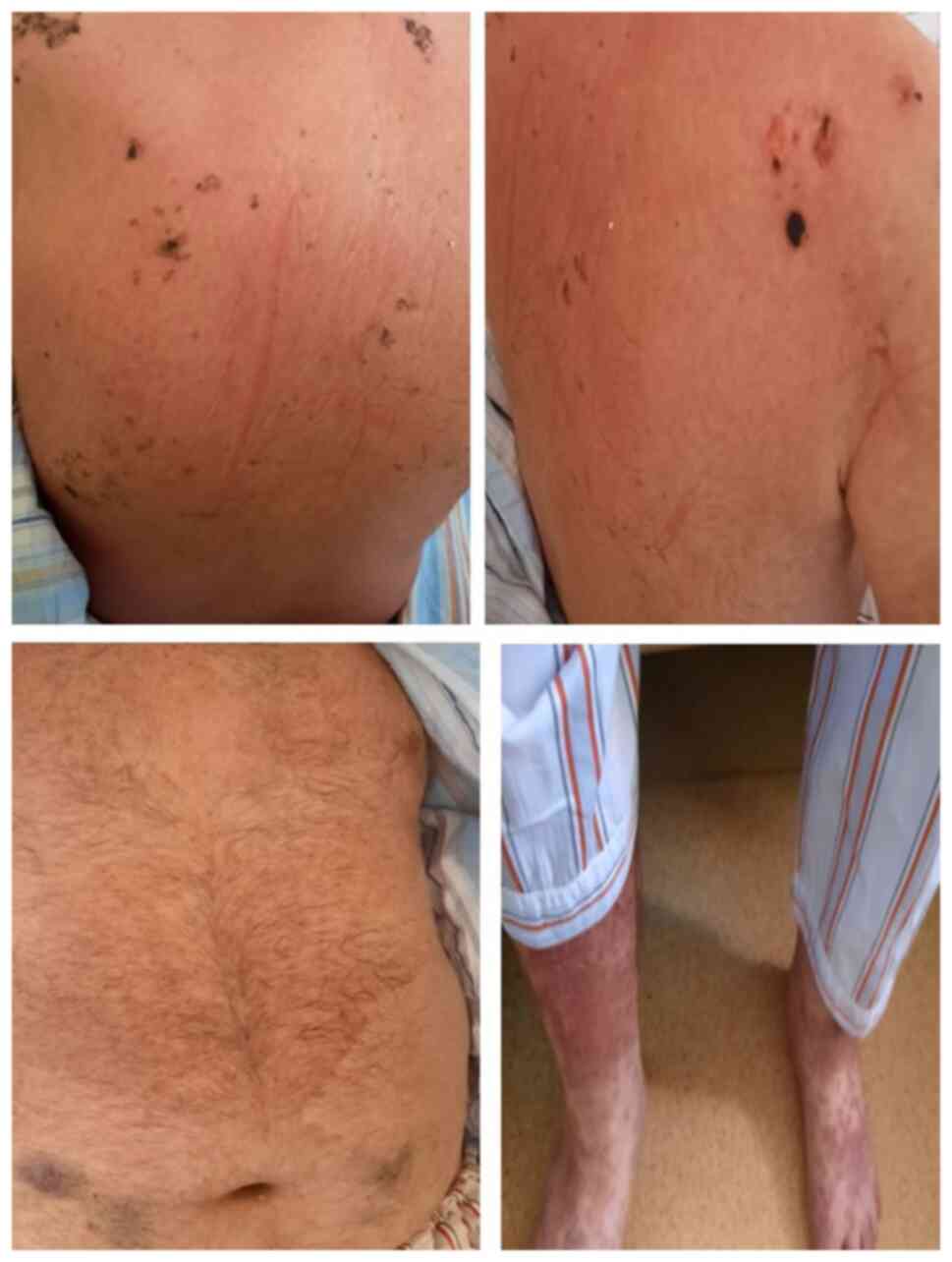
After five days of treatment, the dermatological
aspects had a favorable evolution, with healing of most of the
lesions but persistence of those located on the inferior limbs
(Fig. 2). However, the general
condition of the patient started to deteriorate. On the 7th day of
admission, the patient desaturated to 76% ambient air, requiring an
oxygen supplement that corrected saturation to 93% with 15 l of
additional oxygen. Therefore, an IL-1 inhibitor was added to his
treatment (200 mg on day 1, then 100 mg/day for four days).
Considering his status, an intensive care unit consultation was
requested, arterial gases were measured, which suggested that the
patient was in metabolic acidosis (low PaCO2 and
HCO3), and recommendations for intensive care therapy
were given.
After 20 days of hospitalization, the patient had a
fatal outcome.
Discussion
SJS/toxic epidermal necrolysis (TEN) represents a
significant dermatological emergency, being one of the most severe
cutaneous adverse reactions and associated with a high risk of
mortality. SJS/TEN, due to an immune-complex-mediated
hypersensitivity reaction, involves the mucous membranes and skin
(24,25). Initial symptoms can be unspecific
and include fever, cough, sore throat or eye discomfort, which are
followed by the cutaneous manifestations (26).
SJS/TEN is drug-induced in 70-80% of cases. Graft
versus-host disease is another well-established but rare cause,
independent of drugs (27). A few
cases are related to infections (such as with Mycoplasma
pneumoniae), while others remain unexplained (idiopathic forms)
(28).
This pathology represents a delayed reaction that
usually occurs 4-28 days from the moment of exposure to a drug
(29); thus, it is of utmost
importance to conduct an in-depth anamnesis and a thorough
retrospective pharmacological investigation for an extended period
of time preceding the onset of skin manifestations.
The drugs that are associated with SJS/TEN include
anticonvulsants, allopurinol, sulfonamides, antibiotics (such as
penicillin, cephalosporins, quinolones and minocycline),
acetaminophen and nonsteroidal anti-inflammatory drugs (30-33).
The ALDEN score is one of the most valuable tools in
the assessment of SJS/TEN, which helps identify the possible drug
associated with the severe cutaneous adverse reaction, as well as
the drugs that can still be administered to the patient (34). The algorithm gives the suspected
causal drug taken by the patient a score that sums between -12 and
10, which corresponds to the probability of having caused the
reaction. The total score corresponds to ‘causal links’ that range
from ‘very unlikely’ to ‘very probable’ (35). SJS can occur as a rare side effect
of allopurinol, which, in this case, could have been favored by the
immune stimulation induced by the SARS CoV-2 virus. This idea is
supported by the fact that SJS/TEN has been associated with viral
replication (human immunodeficiency virus and cytomegalovirus)
(36-39),
suggesting this could also be possible in SARS CoV-2 infection in
our case. Therefore, it may be hypothesized that allopurinol was
the causative agent of SJS/TEN, although the fact that SJS/TEN
could be a cutaneous manifestation of SARS CoV-2 infection and or
represent a consequence in this type of viral infection should not
be discounted.
The severity of SJS/TEN can be assessed using
SCORTEN, which is a severity-of-illness scale that was defined in
2000 and is a specific predictor of mortality. The score includes
the following variables: Age >40, the presence of neoplasia,
heart rate >120 bpm, serum blood urea >28 mg/dl, serum
glucose >250 mg/dl, serum HCO3 <20 mmol/l and
>10% detached body surface. Each constant receives one point,
and the final score ranges from 0 to 7. (40). In the presented case, the patient
had an initial score of 3, which indicated mortality of 35.3% As
the status of the patient started to deteriorate, and the
HCO3 level decreased to 13.6 mmol/l, the prognosis of
mortality grew to 58.3%.
In our case, although correct dermatological
treatment led to a favorable evolution of the skin lesions
(41,42), the patient's condition was
ultimately influenced by the complication of SARS CoV-2 infection,
which progressed to respiratory failure associated with major
hydro-electrolytic and acid-base imbalance. Together with the
negative prognostic factors that the patient presented
(hypertension, obesity and dyslipidemia) (43-45),
this led to a fatal outcome.
The SCORTEN in our patient led to an estimated
mortality rate of 35.3% that later grew to 58.3% as a result of
bicarbonate levels being <20 mmol/l (Table III). Therefore, one might ask
whether SJS/TEN influenced the unfavorable evolution of the patient
and if SJS/TEN can appear in (or perhaps be a predictor of) severe
forms of SARS CoV-2 infection.
In conclusion, although SJS/TEN is a rare pathology,
it represents a major dermatological emergency. The combination
between SJS/TEN and COVID-19 can have a fatal outcome if not
recognized and promptly treated. To the best of our knowledge, this
is the first case of SARS CoV-2 and SJS/TEN association in Romania.
This association raises multiple questions regarding the
possibility of SJS/TEN being a cutaneous manifestation of COVID-19.
Whether this association is a simple coincidence or complication of
the physiopathological events of the infection with the new
coronavirus remains to be determined.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
CM, GAL and FDP designed the study. CS and AV
contributed to data extraction and quality assessment. CM, GAL and
AV were responsible for the analysis and discussion of data. CM,
GAL and FDP drafted the manuscript. AV, CMA and CS revised the
manuscript critically and made substantial intellectual
contributions. All authors read and approved the final manuscript.
CM, GAL, CMA and CS confirm the authenticity of all the raw
data.
Ethics approval and consent to
participate
Written informed consent was obtained from the
patient prior to admission.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Lacatusu GA, Vasilescu C, Mihai IF,
Filip-Ciubotaru F, Vata A and Manciuc C: COVID-19 and air
conditioning-is there an environmental link? Environ Eng Manag J.
19:1255–1260. 2020.
|
|
2
|
Manciuc C, Nemescu D, Vata A and Lacatusu
GA: SARS-CoV-2 infection and diabetes mellitus: A North Eastern
Romanian experience. Exp Ther Med. 21(279)2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Docea AO, Tsatsakis A, Albulescu D,
Cristea O, Zlatian O, Vinceti M, Moschos SA, Tsoukalas D, Goumenou
M, Drakoulis N, et al: A new threat from an old enemy: Re-emergence
of coronavirus (Review). Int J Mol Med. 45:1631–1643.
2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Tanasa IA, Manciuc C, Carauleanu A,
Navolan DB, Bohiltea RE and Nemescu D: Anosmia and ageusia
associated with coronavirus infection (COVID-19)-what is known? Exp
Ther Med. 20:2344–2347. 2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Calina D, Docea AO, Petrakis D, Egorov AM,
Ishmukhametov AA, Gabibov AG, Shtilman MI, Kostoff R, Carvalho F,
Vinceti M, et al: Towards effective COVID-19 vaccines: Updates,
perspectives and challenges (Review). Int J Mol Med. 46:3–16.
2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Manciuc DC, Iordan IF, Adavidoaiei AM and
Largu MA: Risks of leptospirosis linked to living and working
environments. Environ Eng Manag J. 17:749–753. 2018.
|
|
7
|
Manciuc C, Dorobăţ C, Hurmuzache M and
Nicu M: Leptospirosis: Clinical and environmental aspects of the
Iaşi County. Environ Eng Manag J. 6:133–136. 2007.
|
|
8
|
Gisondi P, PIaserico S, Bordin C, Alaibac
M, Girolomoni G and Naldi L: Cutaneous manifestations of SARS-CoV-2
infection: A clinical update. J Eur Acad Dermatol Venereol.
34:2499–2504. 2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Marzano AV, Genovese G, Fabbrocini G,
Pigatto P, Monfrecola G, Piraccini BM, Veraldi S, Rubegni P, Cusini
MP, Caputo V, et al: Varicella-like exanthem as a specific
COVID-19-associated skin manifestation: Multicenter case series of
22 patients. J Am Acad Dermatol. 83:280–285. 2020.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Casas CG, Català A, Hernández GC,
Rodríguez-Jiménez P, Fernández-Nieto D, Lario ARV, Fernández IN,
Ruiz-Villaverde R, Falkenhain-López D, Velasco ML, et al:
Classification of the cutaneous manifestations of COVID-19: A rapid
prospective nationwide consensus study in Spain with 375 cases. Br
J Dermatol. 183:71–77. 2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Dominguez-Santas M, Diaz-Guimaraens B,
Abellas PG, Real CMG, Burgos-Blasco P and Suarez-Valle A: Cutaneous
small-vessel vasculitis associated with novel 2019 coronavirus
SARS-CoV-2 infection (COVID-19). J Eur Acad Dermatol Venereol.
34:e536–e537. 2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Manalo IF, Smith MK, Cheeley J and Jacobs
R: A dermatologic manifestation of COVID-19: Transient livedo
reticularis. J Am Acad Dermatol. 83(700)2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Estébanez A, Pérez-Santiago L, Silva E,
Guillen-Climent S, García-Vázquez A and Ramón MD: Cutaneous
manifestations in COVID-19: A new contribution. J Eur Acad Dermatol
Venereol. 34:e250–e251. 2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Oakley AM and Krishnamurthy K: Stevens
Johnson Syndrome. In: StatPearls. StatPearls Publishing, Treasure
Island, FL, 2021.
|
|
15
|
Dutt J, Sapra A, Sheth-Dutt P, Bhandari P
and Gupta S: Stevens-Johnson syndrome: A perplexing diagnosis.
Cureus. 12(e7374)2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Dodiuk-Gad RP, Chung WH, Valeyrie-Allanore
L and Shear NH: Stevens-Johnson syndrome and toxic epidermal
necrolysis: An update. Am J Clin Dermatol. 16:475–493.
2015.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Abdelgabar A and Elsayed M: Case of
erythema multiforme/Stevens-Johnson syndrome: An unusual
presentation of COVID-19. J R Coll Physicians Edinb. 51:160–161.
2021.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Pudukadan D and John B: Toxic epidermal
necrolysis and coronavirus disease 2019: A rare association. J Skin
Sex Transm Dis. 3:184–187. 2021.
|
|
19
|
Shahraki T, Hassanpour K, Arabi A, Ansari
I and Sadoughi MM: Corona virus disease 2019-associated
Stevens-Johnson syndrome: A case report. BMC Ophthalmol.
21(274)2021.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Rossi CM, Beretta FN, Traverso G,
Mancarella S and Zenoni D: A case report of toxic epidermal
necrolysis (TEN) in a patient with COVID-19 treated with
hydroxychloroquine: Are these two partners in crime? Clin Mol
Allergy. 18(19)2020.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Narang I, Panthagani AP, Lewis M, Chohan
B, Ferguson A and Nambi R: COVID-19-induced toxic epidermal
necrolysis. Clin Exp Dermatol. 46:927–929. 2021.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Tanaka A, Isei M, Kikuzawa C, Hinogami H,
Nishida K, Gohma I and Ogawa Y: Development of toxic epidermal
necrolysis in a coronavirus disease 2019 patient with recurrence of
positive SARS-CoV-2 viral RNA. J Dermatol. 48:e144–e145.
2021.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Besari AM, Lim JA, Vellaichamy PT, Hussain
FA, Kamaludin Z and Nor M: Stevens-Johnson syndrome as a primary
skin manifestation of COVID-19. Postgrad Med J.
20(140778)2021.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Nassif A, Bensussan A, Boumsell L, Deniaud
A, Moslehi H, Wolkenstein P, Bagot M and Roujeau JC: Toxic
epidermal necrolysis: Effector cells are drug-specific cytotoxic T
cells. J Allergy Clin Immunol. 114:1209–1215. 2004.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Chung WH, Hung SI, Hong HS, Hsih MS, Yang
LC, Ho HC, Wu JY and Chen YT: Medical genetics: A marker for
Stevens-Johnson syndrome. Nature. 428(486)2004.PubMed/NCBI View
Article : Google Scholar
|
|
26
|
Harr T and French LE: Toxic epidermal
necrolysis and Stevens-Johnson syndrome. Orphanet J Rare Dis.
5(39)2010.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Hazin R, Ibrahimi OA, Hazin MI and
Kimyai-Asadi A: Stevens-Johnson syndrome: Pathogenesis, diagnosis,
and management. Ann Med. 40:129–138. 2008.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Levy M and Shear NH: Mycoplasma pneumoniae
infections and Stevens-Johnson syndrome. Report of eight cases and
review of the literature. Clin Pediatr (Phila). 30:42–49.
1991.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Jawaro T, Kumar A, Pistun O and Dixit D:
Stevens-Johnson syndrome associated with chlordiazepoxide. J Pharm
Technol. 34:82–85. 2018.PubMed/NCBI View Article : Google Scholar
|
|
30
|
De Luca F, Losappio LM, Mirone C,
Schroeder JW, Citterio A, Aversano MG, Scibilia J and Pastorello
EA: Tolerated drugs in subjects with severe cutaneous adverse
reactions (SCARs) induced by anticonvulsants and review of the
literature. Clin Mol Allergy. 15(16)2017.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Techasatian L, Panombualert S, Uppala R
and Jetsrisuparb C: Drug-induced Stevens-Johnson syndrome and toxic
epidermal necrolysis in children: 20 years study in a tertiary care
hospital. World J Pediatr. 13:255–260. 2017.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Frey N, Bodmer M, Bircher A, Jick SS,
Meier CR and Spoendlin J: Stevens-Johnson syndrome and toxic
epidermal necrolysis in association with commonly prescribed drugs
in outpatient care other than anti-epileptic drugs and antibiotics:
A population-based case-control study. Drug Saf. 42:55–66.
2019.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Diphoorn J, Cazzaniga S, Gamba C,
Schroeder J, Citterio A, Rivolta AL, Vighi GD and Naldi L:
REACT-Lombardia study group. REACT-lombardia study group:
Incidence, causative factors and mortality rates of Stevens-Johnson
syndrome (SJS) and toxic epidermal necrolysis (TEN) in northern
Italy: Data from the REACT registry. Pharmacoepidemiol Drug Saf.
25:196–203. 2016.PubMed/NCBI View
Article : Google Scholar
|
|
34
|
Lerch M, Mainetti C, Beretta-Piccoli BT
and Harr T: Current perspectives on Stevens-Johnson syndrome and
toxic epidermal necrolysis. Clin Rev Allergy Immunol. 54:147–176.
2018.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Honma M, Tobisawa S, Iinuma S, Shibuya T,
Komatsu S, Takahashi I, Ishida-Yamamoto A and Iizuka H: Toxic
epidermal necrolysis with prominent facial pustules: A case with
reactivation of human herpesvirus 7. Dermatology. 221:306–308.
2010.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Peter J, Choshi P and Lehloenya RJ: Drug
hypersensitivity in HIV infection. Curr Opin Allergy Clin Immunol.
19:272–282. 2019.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Yang CW, Cho YT, Hsieh YC, Hsu SH, Chen KL
and Chu CY: The interferon-γ-induced protein 10/CXCR3 axis is
associated with human herpesvirus-6 reactivation and the
development of sequelae in drug reaction with eosinophilia and
systemic symptoms. Br J Dermatol. 183:909–919. 2020.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Tagajdid MR, Doblali T, Elannaz H, Hammi
S, Belfequih B and Mrani S: Reactivation of cytomegalovirus in a
patient with Stevens-Johnson syndrome-toxic epidermal necrolysis.
Iran J Med Sci. 38 (2 Suppl):S195–S197. 2013.PubMed/NCBI
|
|
39
|
Richard EB, Hamer D, Musso MW, Short T and
O'Neal HR Jr: Variability in management of patients with SJS/TEN: A
survey of burn unit directors. J Burn Care Res. 39:585–592.
2018.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Bastuji-Garin S, Fouchard N, Bertocchi M,
Roujeau JC, Revuz J and Wolkenstein P: SCORTEN: A
severity-of-illness score for toxic epidermal necrolysis. J Invest
Dermatol. 115:149–153. 2000.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Papp A, Sikora S, Evans M, Song D,
Kirchhof M, Miliszewski M and Dutz J: Treatment of toxic epidermal
necrolysis by a multidisciplinary team. A review of literature and
treatment results. Burns. 44:807–815. 2018.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Miliszewski MA, Kirchhof MG, Sikora S,
Papp A and Dutz JP: Stevens-Johnson syndrome and toxic epidermal
necrolysis: An analysis of triggers and implications for improving
prevention. Am J Med. 129:1221–1225. 2016.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Di Stadio A, Ricci G, Greco A, de
Vincentis M and Ralli M: Mortality rate and gender differences in
COVID-19 patients dying in Italy a comparison with other countries.
Eur Rev Med Pharmacol Sci. 24:4066–4067. 2020.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Onder G, Rezza G and Brusaferro S:
Case-fatality rate and characteristics of patients dying in
relation to COVID-19 in Italy. JAMA. 323:1775–1776. 2020.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He
JX, Liu L, Shan H, Lei CL, Hui DSC, et al: China medical treatment
expert group for Covid-19: Clinical characteristics of coronavirus
disease 2019 in China. N Engl J Med. 382:1708–1720. 2020.PubMed/NCBI View Article : Google Scholar
|