|
1
|
Zhang F, Wei K, Slowikowski K, Fonseka CY,
Rao DA, Kelly S, Goodman SM, Tabechian D, Hughes LB, Salomon-Escoto
K, et al: Defining inflammatory cell states in rheumatoid arthritis
joint synovial tissues by integrating single-cell transcriptomics
and mass cytometry. Nat Immunol. 20:928–942. 2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Aletaha D, Neogi T, Silman AJ, Funovits J,
Felson DT, Bingham CO III, Birnbaum NS, Burmester GR, Bykerk VP,
Cohen MD, et al: 2010 Rheumatoid arthritis classification criteria:
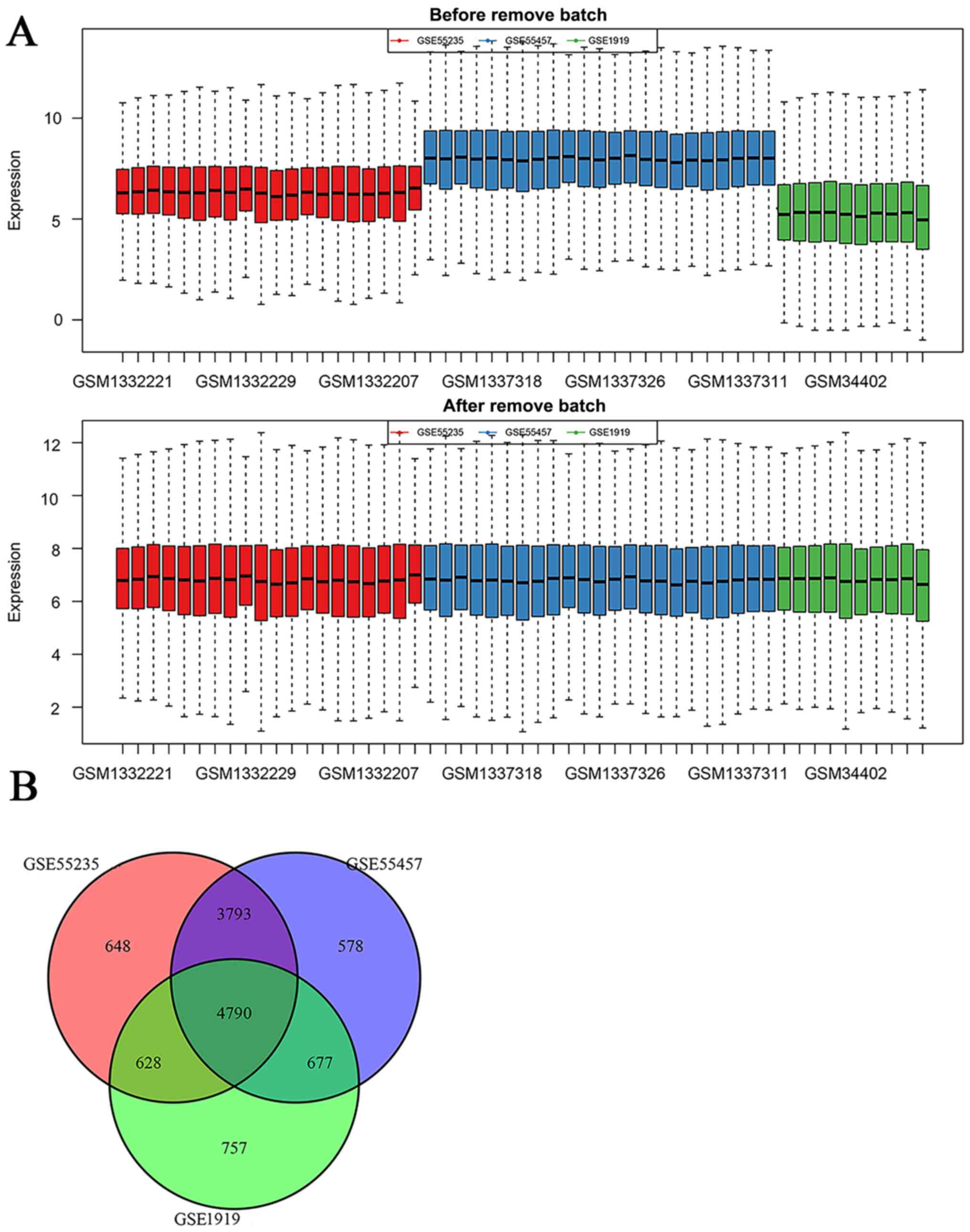
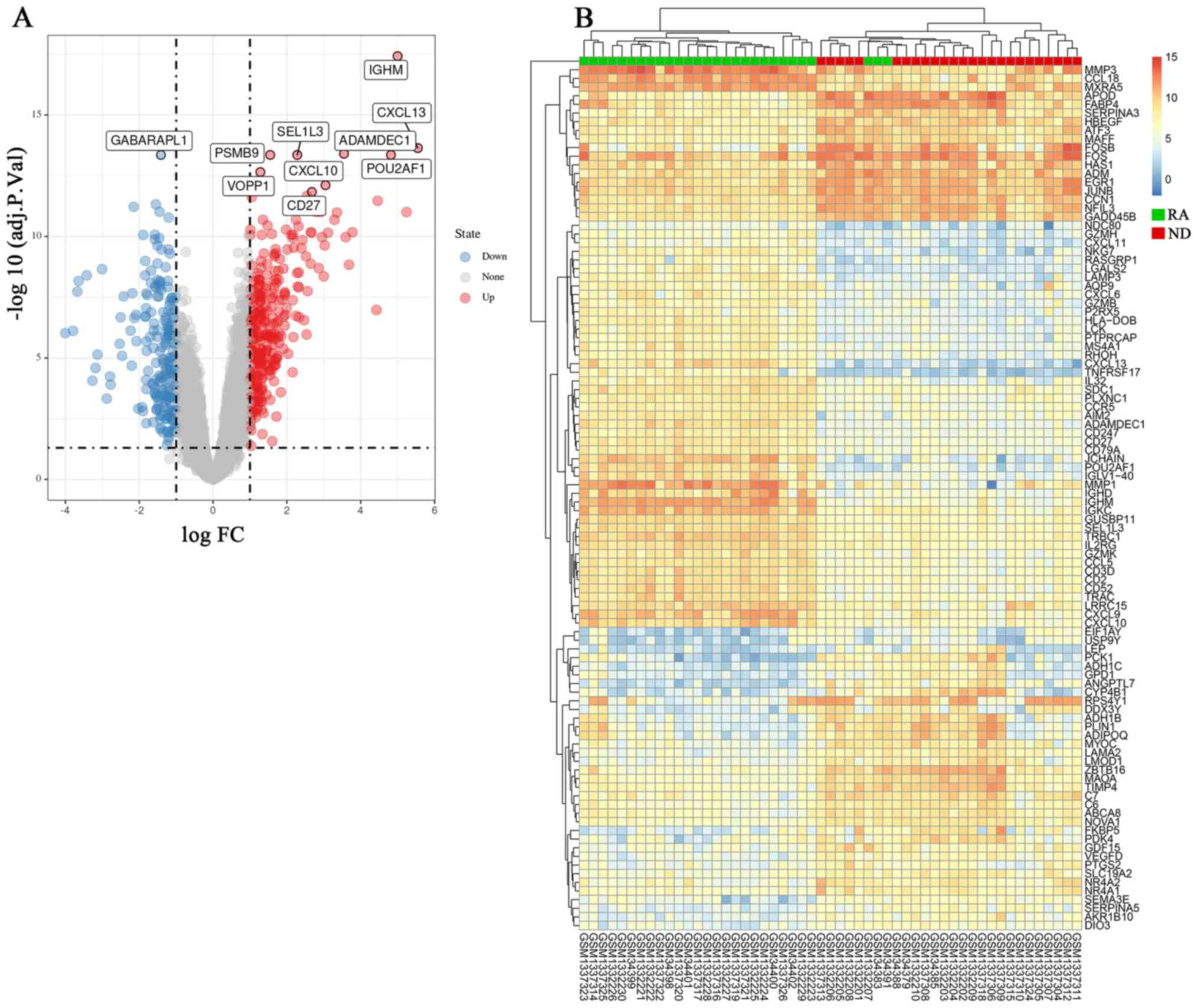
An American college of rheumatology/European league against
rheumatism collaborative initiative. Ann Rheum Dis. 69:1580–1588.
2010.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Jacob L, Rockel T and Kostev K: Depression
risk in patients with rheumatoid arthritis in the United Kingdom.
Rheumatol Ther. 4:195–200. 2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Kharlamova N, Jiang X, Sherina N, Potempa
B, Israelsson L, Quirke AM, Eriksson K, Yucel-Lindberg T, Venables
PJ, Potempa J, et al: Antibodies to porphyromonas gingivalis
indicate interaction between oral infection, smoking, and risk
genes in rheumatoid arthritis etiology. Arthritis Rheumatol.
68:604–613. 2016.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Kurowska M, Rudnicka W, Kontny E, Janicka
I, Chorazy M, Kowalczewski J, Ziółkowska M, Ferrari-Lacraz S, Strom
TB and Maśliński W: Fibroblast-like synoviocytes from rheumatoid
arthritis patients express functional IL-15 receptor complex:
Endogenous IL-15 in autocrine fashion enhances cell proliferation
and expression of Bcl-x(L) and Bcl-2. J Immunol. 169:1760–1767.
2002.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Chen S, Yang Y, Feng H, Wang H, Zhao R and
Liu H: Baicalein inhibits interleukin-1β-induced proliferation of
human rheumatoid arthritis fibroblast-like synoviocytes.
Inflammation. 37:163–169. 2014.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Lu YC, Chuang CH, Chuang KH, Chen IJ,
Huang BC, Lee WH, Wang HE, Li JJ, Cheng YA, Cheng KW, et al:
Specific activation of pro-Infliximab enhances selectivity and
safety of rheumatoid arthritis therapy. PLoS Biol.
17(e3000286)2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Bao J, Yue T, Li T, He DY and Bao YX: Good
response to infliximab in rheumatoid arthritis following failure of
interleukin-1 receptor antagonist. Int J Rheum Dis. 19:370–376.
2016.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Zhang R, Yang X, Wang J, Han L, Yang A,
Zhang J, Zhang D, Li B, Li Z and Xiong Y: Identification of
potential biomarkers for differential diagnosis between rheumatoid
arthritis and osteoarthritis via integrative genome-wide gene
expression profiling analysis. Mol Med Rep. 19:30–40.
2019.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Jiang P and Liu XS: Big data mining yields
novel insights on cancer. Nat Genet. 47:103–104. 2015.PubMed/NCBI View
Article : Google Scholar
|
|
11
|
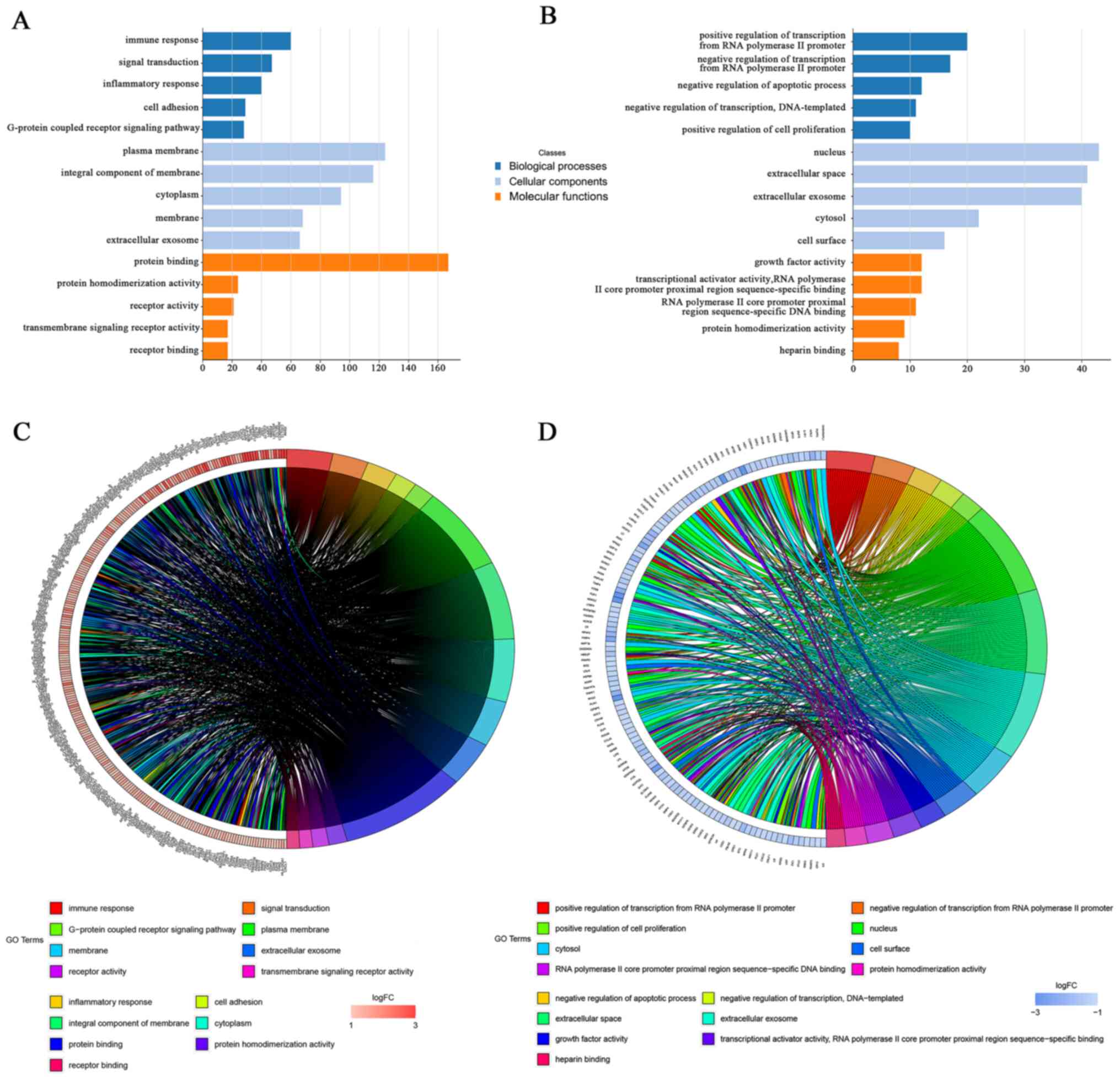
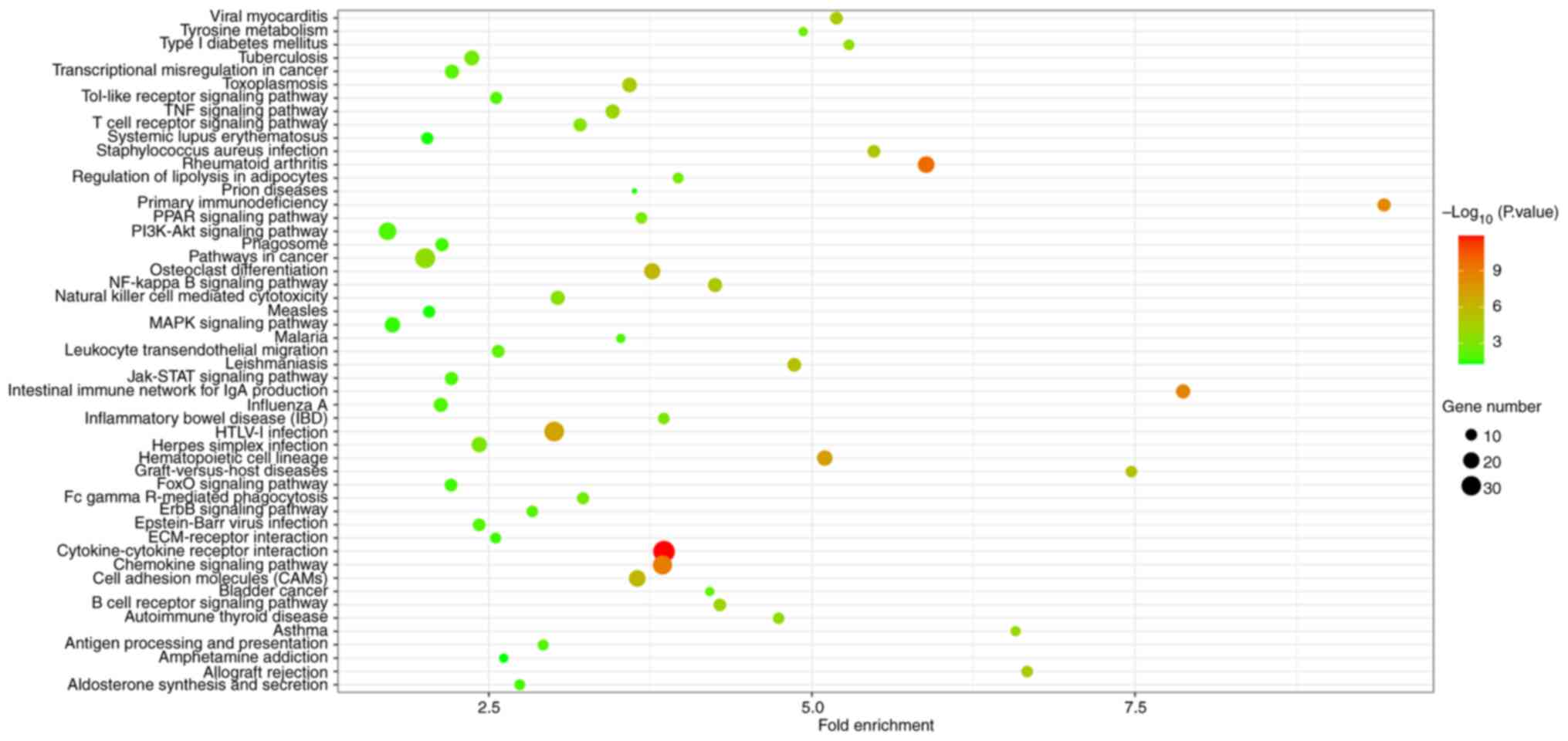
Huang da W, Sherman BT and Lempicki RA:
Systematic and integrative analysis of large gene lists using DAVID
bioinformatics resources. Nat Protoc. 4:44–57. 2009.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Chen B, Zhao Y, Li S, Yang L, Wang H, Wang
T, Shi B, Gai Z, Heng X, Zhang C, et al: Variations in oral
microbiome profiles in rheumatoid arthritis and osteoarthritis with
potential biomarkers for arthritis screening. Sci Rep.
8(17126)2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Woetzel D, Huber R, Kupfer P, Pohlers D,
Pfaff M, Driesch D, Häupl T, Koczan D, Stiehl P, Guthke R and Kinne
RW: Identification of rheumatoid arthritis and osteoarthritis
patients by transcriptome-based rule set generation. Arthritis Res
Ther. 16(R84)2014.PubMed/NCBI View
Article : Google Scholar
|
|
14
|
Ungethuem U, Haeupl T, Witt H, Koczan D,
Krenn V, Huber H, von Helversen TM, Drungowski M, Seyfert C, Zacher
J, et al: Molecular signatures and new candidates to target the
pathogenesis of rheumatoid arthritis. Physiol Genomics.
42A:267–282. 2010.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Leek JT and Storey JD: Capturing
heterogeneity in gene expression studies by surrogate variable
analysis. PLoS Genet. 3:1724–1735. 2007.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Phipson B, Lee S, Majewski IJ, Alexander
WS and Smyth GK: Robust hyperparmeter estimation protects against
hypervariable genes and improves power to detect differential
expression. Ann Appl Stat. 10:946–963. 2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Leek JT, Johnson WE, Parker HS, Jaffe AE
and Storey JD: The sva package for removing batch effects and other
unwanted variation in high-throughput experiments. Bioinformatics.
28:882–883. 2012.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Huang da W, Sherman BT and Lempicki RA:
Bioinformatics enrichment tools: Paths toward the comprehensive
functional analysis of large gene lists. Nucleic Acids Res.
37:1–13. 2009.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Dennis G Jr, Sherman BT, Hosack DA, Yang
J, Gao W, Lane HC and Lempicki RA: DAVID: Database or annotation,
visualization, and integrated discovery. Genome Biol.
4(P3)2003.PubMed/NCBI
|
|
20
|
Shannon P, Markiel A, Ozier O, Baliga NS,
Wang JT, Ramage D, Amin N, Schwikowski B and Ideker T: Cytoscape: A
software environment for integrated models of biomolecular
interaction networks. Genome Res. 13:2498–2504. 2003.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Franceschini A, Szklarczyk D, Frankild S,
Kuhn M, Simonovic M, Roth A, Lin J, Minguez P, Bork P, von Mering C
and Jensen LJ: STRING v9.1: Protein-protein interaction networks,
with increased coverage and integration. Nucleic Acids Res. 41
(Database Issue):D808–D815. 2013.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Bader GD and Hogue CW: An automated method
for finding molecular complexes in large protein interaction
networks. BMC Bioinformatics. 4(2)2003.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Song E, Song W, Ren M, Xing L, Ni W, Li Y,
Gong M, Zhao M, Ma X, Zhang X and An R: Identification of potential
crucial genes associated with carcinogenesis of clear cell renal
cell carcinoma. J Cell Biochem. 119:5163–5174. 2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Yamanaka H: TNF as a target of
inflammation in rheumatoid arthritis. Endocr Metab Immune Disord
Drug Targets. 15:129–134. 2015.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Huang CC, Chiou CH, Liu SC, Hu SL, Su CM,
Tsai CH and Tang CH: Melatonin attenuates TNF-α and IL-1β
expression in synovial fibroblasts and diminishes cartilage
degradation: Implications for the treatment of rheumatoid
arthritis. J Pineal Res. 66(e12560)2019.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Shen HH, Yang YX, Meng X, Luo XY, Li XM,
Shuai ZW, Ye DQ and Pan HF: NLRP3: A promising therapeutic target
for autoimmune diseases. Autoimmun Rev. 17:694–702. 2018.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Smolen JS, Aletaha D and McInnes IB:
Rheumatoid arthritis. Lancet. 388:2023–2038. 2016.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Smatti MK, Cyprian FS, Nasrallah GK, Al
Thani AA, Almishal RO and Yassine HM: Viruses and autoimmunity: A
review on the potential interaction and molecular mechanisms.
Viruses. 11(762)2019.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Smolen JS and Aletaha D: Rheumatoid
arthritis therapy reappraisal: Strategies, opportunities and
challenges. Nat Rev Rheumatol. 11:276–289. 2015.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Mackay CR: Chemokines: Immunology's high
impact factors. Nat Immunol. 2:95–101. 2001.PubMed/NCBI View
Article : Google Scholar
|
|
32
|
Combe B, Pope RM, Fischbach M, Darnell B,
Baron S and Talal N: Interleukin-2 in rheumatoid arthritis:
Production of and response to interleukin-2 in rheumatoid synovial
fluid, synovial tissue and peripheral blood. Clin Exp Immunol.
59:520–528. 1985.PubMed/NCBI
|
|
33
|
Scheen AJ, Esser N and Paquot N:
Antidiabetic agents: Potential anti-inflammatory activity beyond
glucose control. Diabetes Metab. 41:183–194. 2015.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Ma MHY, Defranoux N, Li W, Sasso EH,
Ibrahim F, Scott DL and Cope AP: A multi-biomarker disease activity
score can predict sustained remission in rheumatoid arthritis.
Arthritis Res Ther. 22(158)2020.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Lee SS, Joo YS, Kim WU, Min DJ, Min JK,
Park SH, Cho CS and Kim HY: Vascular endothelial growth factor
levels in the serum and synovial fluid of patients with rheumatoid
arthritis. Clin Exp Rheumatol. 19:321–324. 2001.PubMed/NCBI
|
|
36
|
Zhang Y, Qiu H, Zhang H, Wang L, Zhuang C
and Liu R: Vascular endothelial growth factor A (VEGFA)
polymorphisms in Chinese patients with rheumatoid arthritis. Scand
J Rheumatol. 42:344–348. 2013.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Piao CH, Bui TT, Fan YJ, Nguyen TV, Shin
DU, Song CH, Lee SY, Shin HS, Kim HT and Chai OH: In vivo
and in vitro anti-allergic and anti-inflammatory effects of
Dryopteris crassirhizoma through the modulation of the NF-ĸB
signaling pathway in an ovalbumin-induced allergic asthma mouse
model. Mol Med Rep. 22:3597–3606. 2020.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Lu W and Li G: Identification of key genes
and pathways in rheumatoid arthritis gene expression profile by
bioinformatics. Acta Reumatol Port. 43:109–131. 2018.PubMed/NCBI
|
|
39
|
Tang JR, Michaelis KA, Nozik-Grayck E,
Seedorf GJ, Hartman-Filson M, Abman SH and Wright CJ: The NF-κB
inhibitory proteins IκBα and IκBβ mediate disparate responses to
inflammation in fetal pulmonary endothelial cells. J Immunol.
190:2913–2923. 2013.PubMed/NCBI View Article : Google Scholar
|