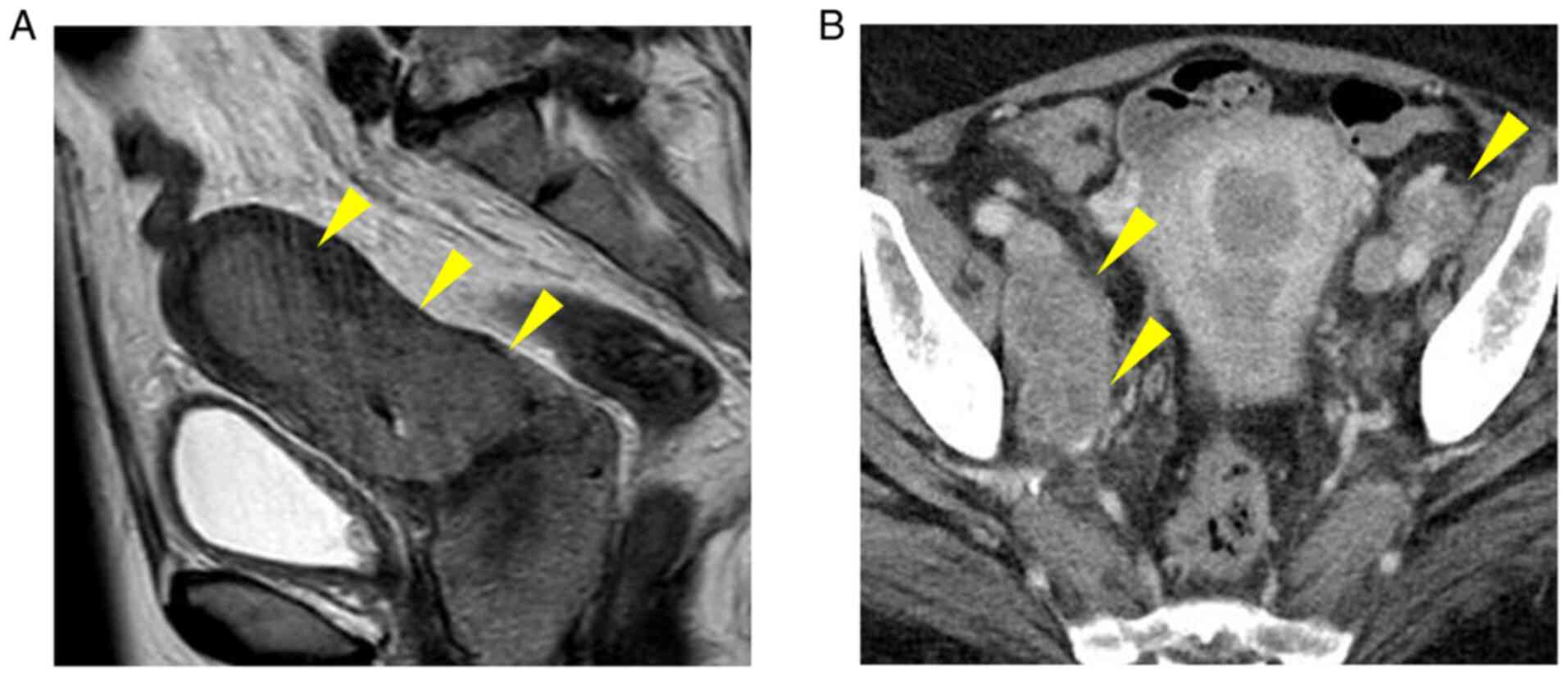
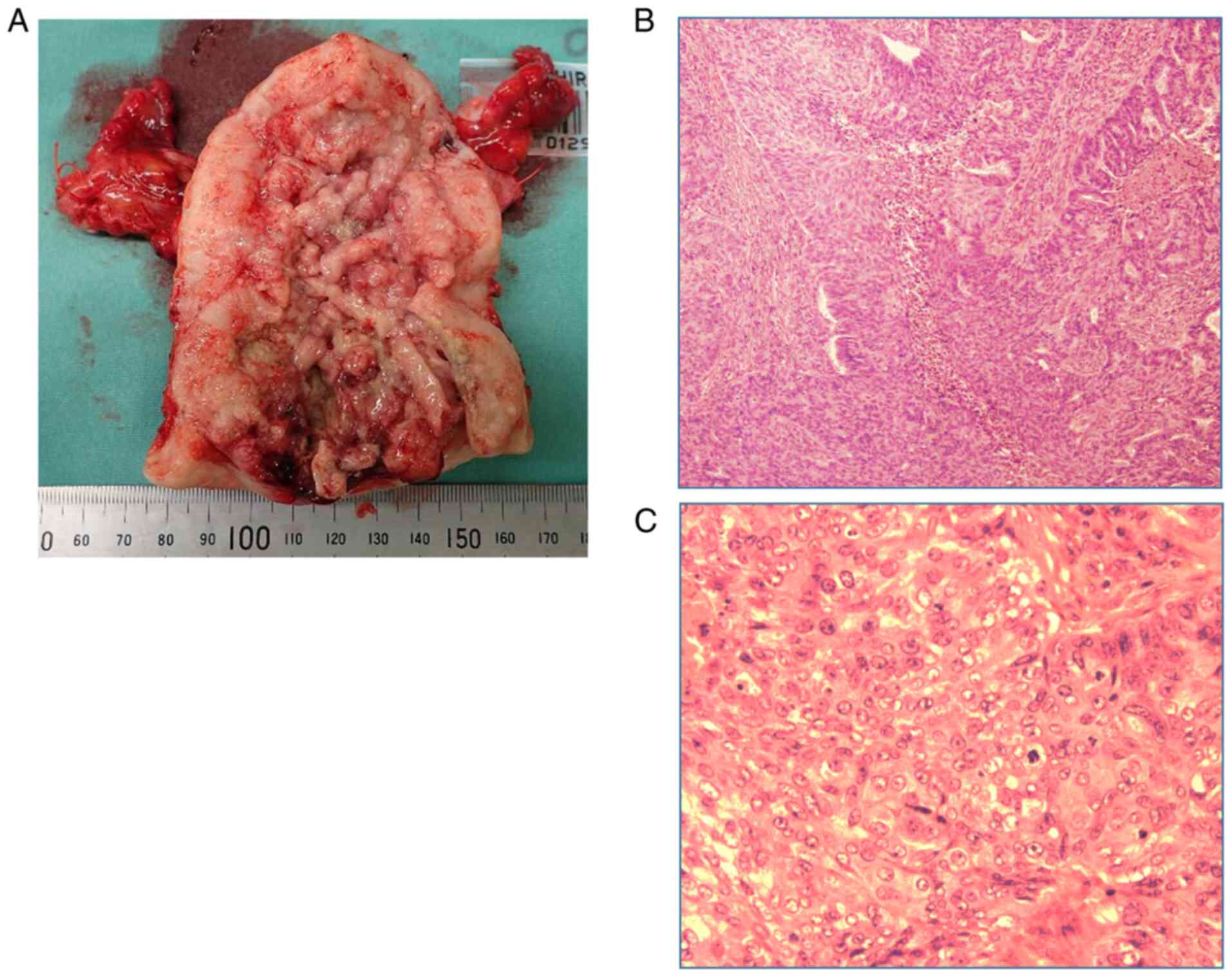
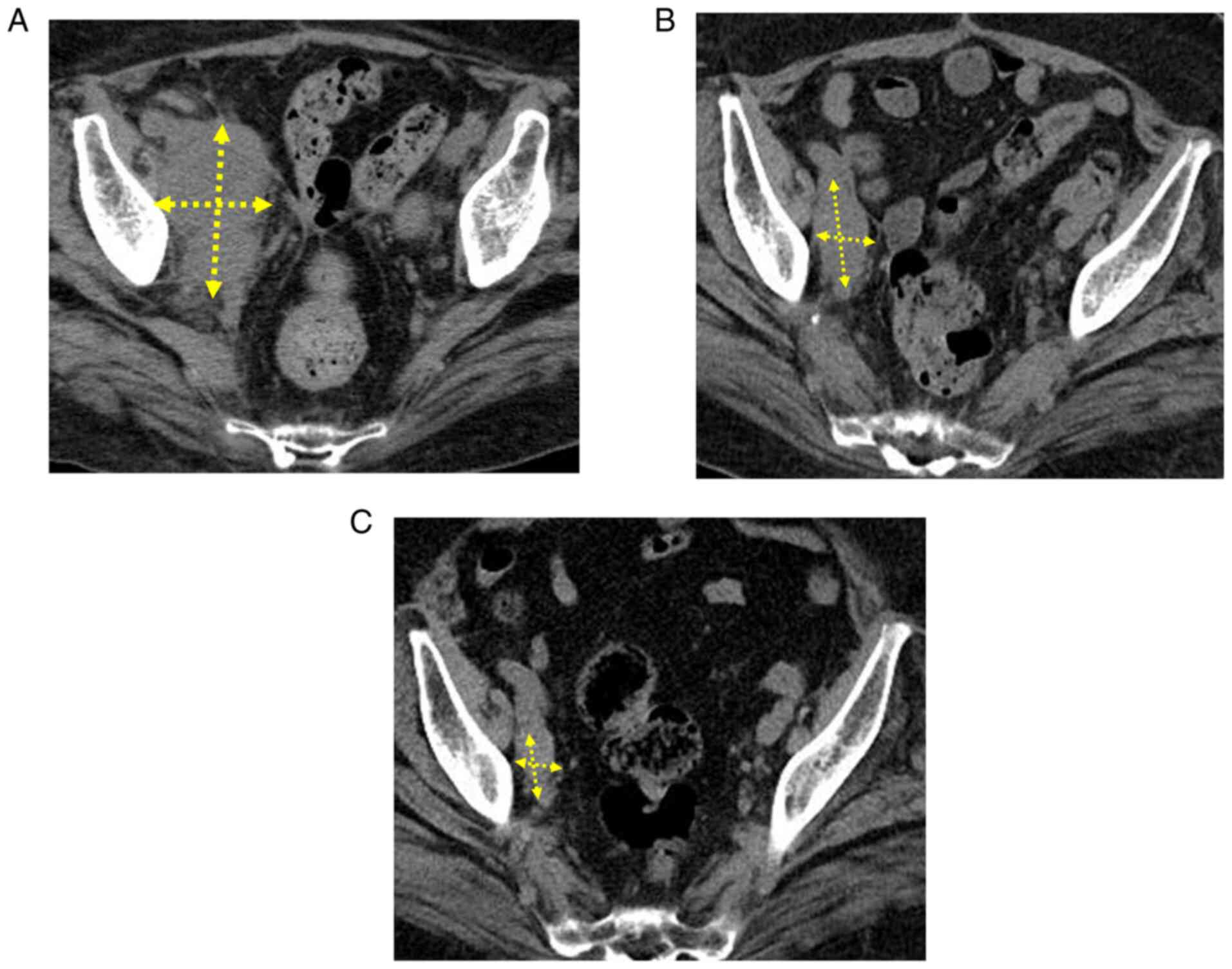
|
1
|
Yamagami W, Mikami M, Nagase S, Tabata T,
Kobayashi Y, Kaneuchi M, Kobayashi H, Yamada H, Hasegawa K,
Fujiwara H, et al: Japan Society of Gynecologic Oncology 2018
guidelines for treatment of uterine body neoplasms. J Gynecol
Oncol. 31(e18)2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Brooks RA, Fleming GF, Lastra RR, Lee NK,
Moroney JW, Son CH, Tatebe K and Veneris JL: Current
recommendations and recent progress in endometrial cancer. CA
Cancer J Clin. 69:258–279. 2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Le DT, Durham JN, Smith KN, Wang H,
Bartlett BR, Aulakh LK, Lu S, Kemberling H, Wilt C, Luber BS, et
al: Mismatch repair deficiency predicts response of solid tumors to
PD-1 blockade. Science. 357:409–413. 2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Akagi K, Oki E, Taniguchi H, Nakatani K,
Aoki D, Kuwata T and Yoshino T: Real-world data on microsatellite
instability status in various unresectable or metastatic solid
tumors. Cancer Sci. 112:1105–1113. 2021.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Marabelle A, Le DT, Ascierto PA, Di
Giacomo AM, De Jesus-Acosta A, Delord JP, Geva R, Gottfried M,
Penel N, Hansen AR, et al: Efficacy of pembrolizumab in patients
with noncolorectal high microsatellite instability/mismatch
repair-deficient cancer: Results from the phase II KEYNOTE-158
study. J Clin Oncol. 38:1–10. 2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Robert C, Ribas A, Hamid O, Daud A,
Wolchok JD, Joshua AM, Hwu WJ, Weber JS, Gangadhar TC, Joseph RW,
et al: Durable complete response after discontinuation of
pembrolizumab in patients with metastatic melanoma. J Clin Oncol.
36:1668–1674. 2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Garon EB, Hellmann MD, Rizvi NA, Carcereny
E, Leighl NB, Ahn MJ, Eder JP, Balmanoukian AS, Aggarwal C, Horn L,
et al: Five-year overall survival for patients with advanced
non-small-cell lung cancer treated with pembrolizumab: Results from
the phase I KEYNOTE-001 study. J Clin Oncol. 37:2518–2527.
2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Nagai T, Mogami T, Takeda T, Tomiyama N
and Yasui T: A case of secondary adrenocortical insufficiency due
to isolated adrenocorticotropic hormone deficiency with empty sella
syndrome after pembrolizumab treatment in a patient with metastatic
renal pelvic cancer. Urol Case Rep. 39(101766)2021.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Tsang ES, Walker EJ, Carnevale J, Fisher
GA and Ko AH: Durable response after immune checkpoint
inhibitor-related diabetes in mismatch repair-deficient pancreatic
cancer. Immunotherapy. 13:1249–1254. 2021.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Eguchi S, Shinkawa H, Sato Y, Nakai K,
Takemura S, Tanaka S, Amano R, Kimura K, Ohira G, Nishio K, et al:
Durable response after discontinuation of pembrolizumab therapy for
intrahepatic cholangiocarcinoma: A case report. Clin J
Gastroenterol. 14:858–865. 2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Viale G, Trapani D and Curigliano G:
Mismatch repair deficiency as a predictive biomarker for
immunotherapy efficacy. Biomed Res Int.
2017(4719194)2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Maleki Vareki S, Garrigós C and Duran I:
Biomarkers of response to PD-1/PD-L1 inhibition. Crit Rev Oncol
Hematol. 116:116–124. 2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Chalmers ZR, Connelly CF, Fabrizio D, Gay
L, Ali SM, Ennis R, Schrock A, Campbell B, Shlien A, Chmielecki J,
et al: Analysis of 100,000 human cancer genomes reveals the
landscape of tumor mutational burden. Genome Med.
9(34)2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Duffy MJ and Crown J: Biomarkers for
predicting response to immunotherapy with immune checkpoint
inhibitors in cancer patients. Clin Chem. 65:1228–1238.
2019.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Mulder EE, de Joode K, Litière S, Ten Tije
AJ, Suijkerbuijk KP, Boers-Sonderen MJ, Hospers GAP, de Groot JW,
van den Eertwegh AJ, Aarts MJ, et al: Early discontinuation of PD-1
blockade upon achieving a complete or partial response in patients
with advanced melanoma: The multicentre prospective Safe Stop
trial. BMC Cancer. 21(323)2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Makker V, Colombo N, Casado Herráez A,
Santin AD, Colomba E, Miller DS, Fujiwara K, Pignata S, Baron-Hay
S, Ray-Coquard I, et al: Lenvatinib plus pembrolizumab for advanced
endometrial cancer. N Engl J Med. 386:437–448. 2022.PubMed/NCBI View Article : Google Scholar
|