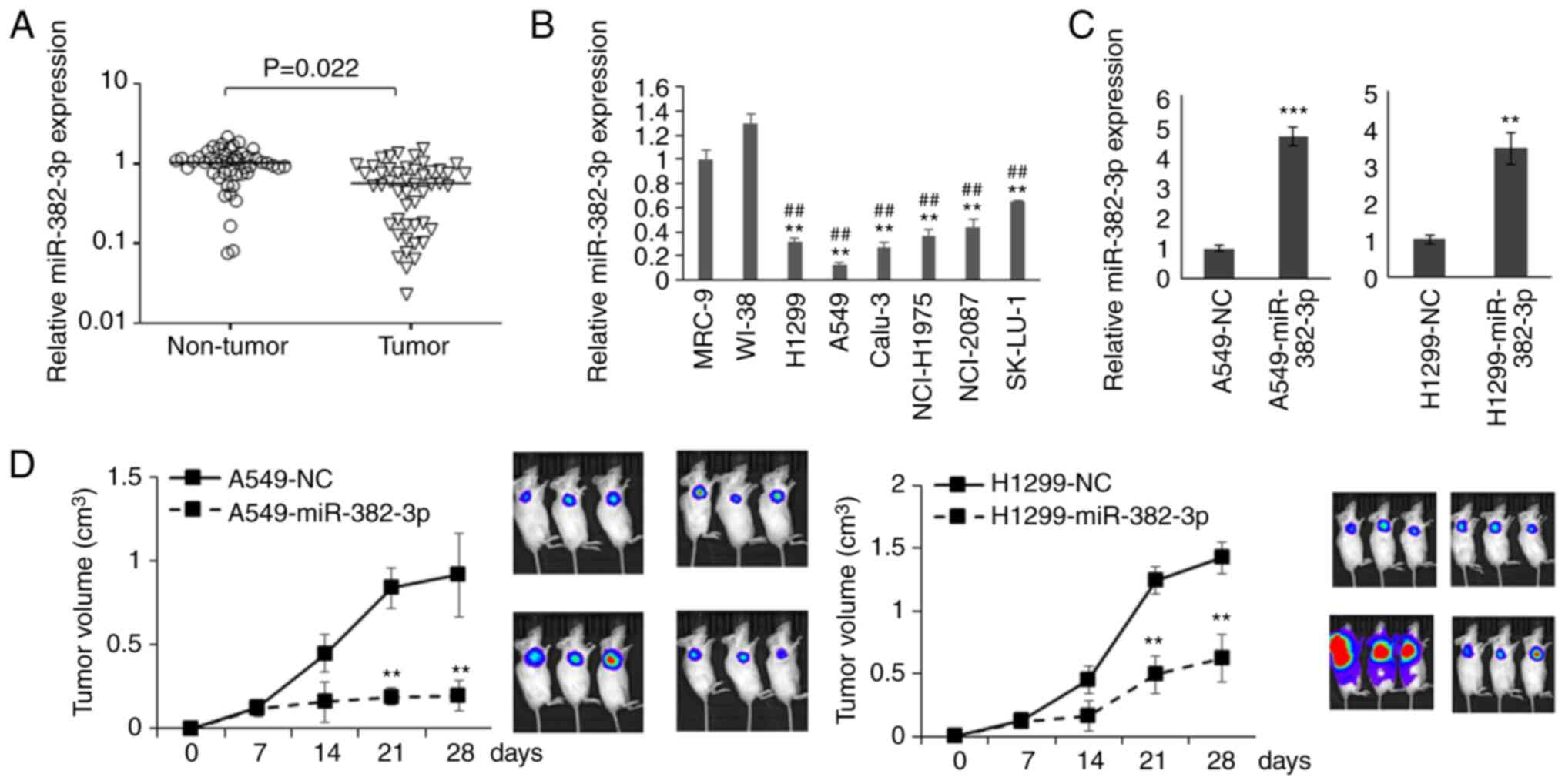
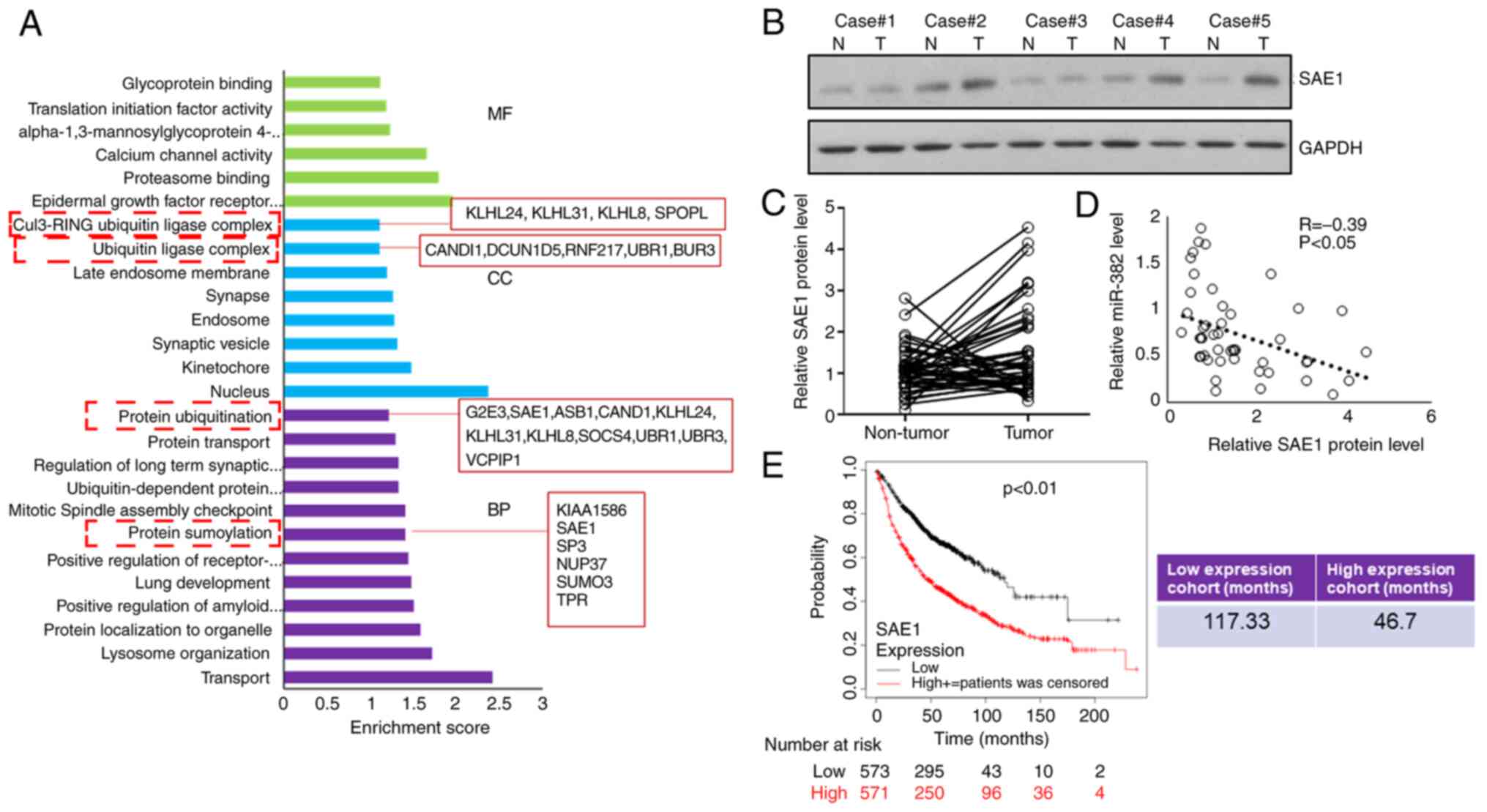
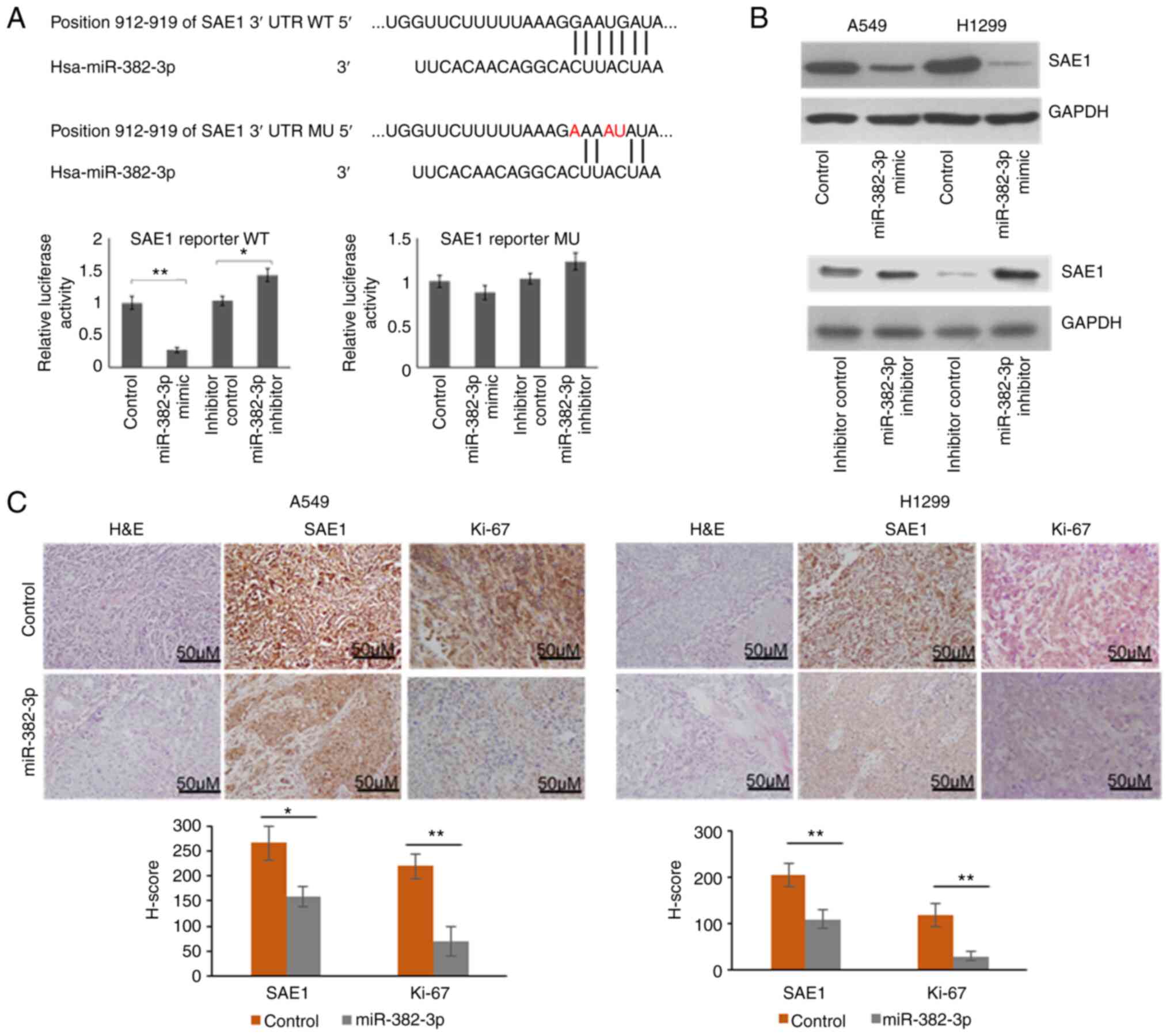
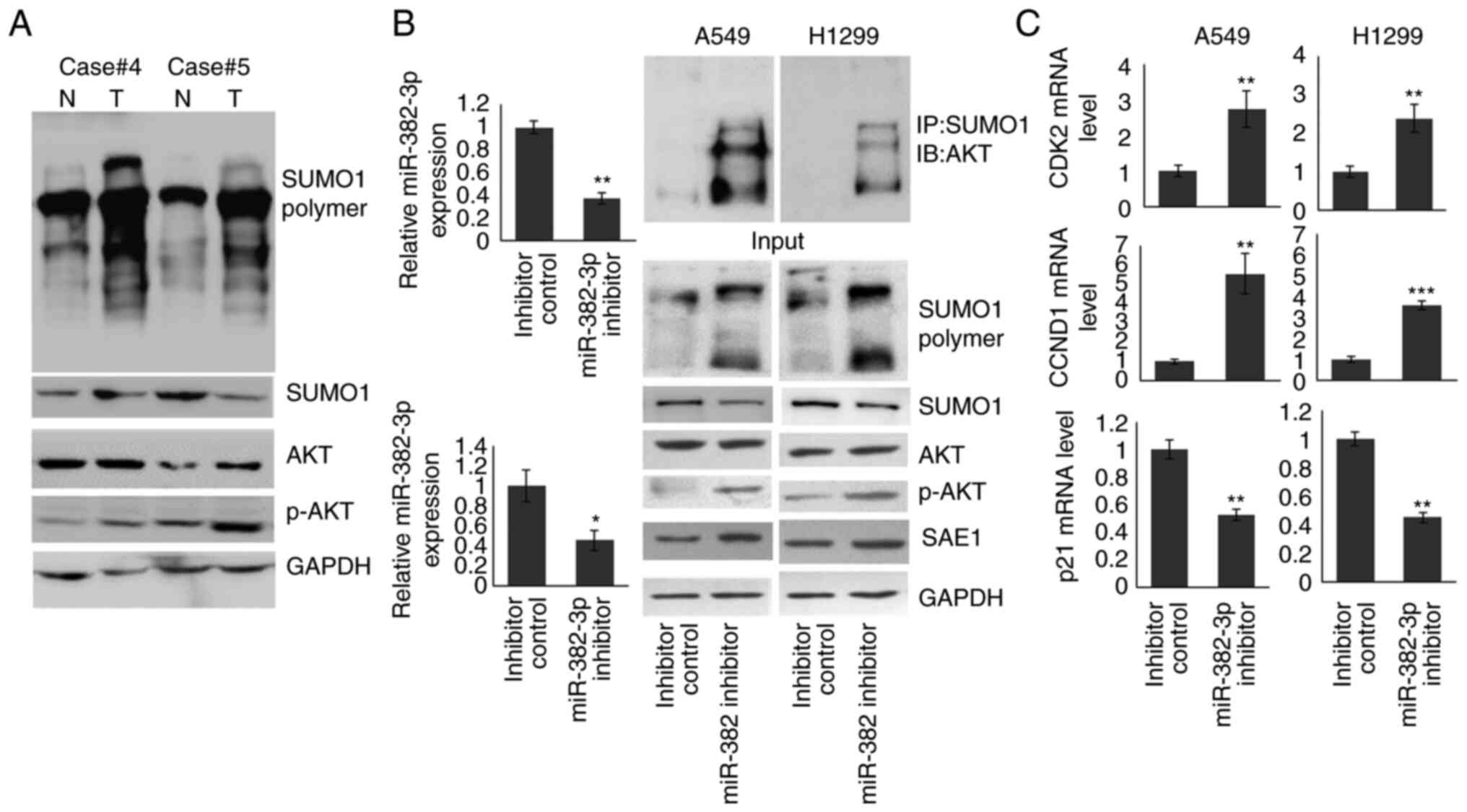
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2015. CA Cancer J Clin. 65:5–29. 2015.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Chen Z, Fillmore CM, Hammerman PS, Kim CF
and Wong KK: Non-small-cell lung cancers: A heterogeneous set of
diseases. Nat Rev Cancer. 14:535–546. 2014.PubMed/NCBI View
Article : Google Scholar
|
|
3
|
Li Q, Liu M, Ma F, Luo Y, Cai R, Wang L,
Xu N and Xu B: Circulating miR-19a and miR-205 in serum may predict
the sensitivity of luminal A subtype of breast cancer patients to
neoadjuvant chemotherapy with epirubicin plus paclitaxel. PLoS One.
9(e104870)2014.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Maes OC, Chertkow HM, Wang E and Schipper
HM: MicroRNA: Implications for Alzheimer disease and other human
CNS disorders. Curr Genomics. 10:154–168. 2009.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Xu J, Li Y, Wang F, Wang X, Cheng B, Ye F,
Xie X, Zhou C and Lu W: Suppressed miR-424 expression via
upregulation of target gene Chk1 contributes to the progression of
cervical cancer. Oncogene. 32:976–987. 2013.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Farazi TA, Hoell JI, Morozov P and Tuschl
T: MicroRNAs in human cancer. Adv Exp Med Biol. 774:1–20.
2013.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Fang H, Liu Y, He Y, Jiang Y, Wei Y, Liu
H, Gong Y and An G: Extracellular vesicle-delivered miR-505-5p, as
a diagnostic biomarker of early lung adenocarcinoma, inhibits cell
apoptosis by targeting TP53AIP1. Int J Oncol. 54:1821–1832.
2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Vivanco I and Sawyers CL: The
phosphatidylinositol 3-Kinase AKT pathway in human cancer. Nat Rev
Cancer. 2:489–501. 2002.PubMed/NCBI View
Article : Google Scholar
|
|
9
|
Tsurutani J, Fukuoka J, Tsurutani H, Shih
JH, Hewitt SM, Travis WD, Jen J and Dennis PA: Evaluation of two
phosphorylation sites improves the prognostic significance of Akt
activation in non-small-cell lung cancer tumors. J Clin Oncol.
24:306–314. 2006.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Yoshizawa A, Fukuoka J, Shimizu S, Shilo
K, Franks TJ, Hewitt SM, Fujii T, Cordon-Cardo C, Jen J and Travis
WD: Overexpression of phospho-eIF4E is associated with survival
through AKT pathway in non-small cell lung cancer. Clin Cancer Res.
16:240–248. 2010.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Li R, Wei J, Jiang C, Liu D, Deng L, Zhang
K and Wang P: Akt SUMOylation regulates cell proliferation and
tumorigenesis. Cancer Res. 73:5742–5753. 2013.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Kessler BM, Bursomanno S, McGouran JF,
Hickson ID and Liu Y: Biochemical and mass spectrometry-based
approaches to profile SUMOylation in human cells. Methods Mol Biol.
1491:131–144. 2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Seeler JS and Dejean A: SUMO and the
robustness of cancer. Nat Rev Cancer. 17:184–197. 2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Inamura K, Shimoji T, Ninomiya H,
Hiramatsu M, Okui M, Satoh Y, Okumura S, Nakagawa K, Noda T,
Fukayama M and Ishikawa Y: A metastatic signature in entire lung
adenocarcinomas irrespective of morphological heterogeneity. Hum
Pathol. 38:702–709. 2007.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Orlans FB: Regulation of animal
experimentation: United States of America. Acta Physiol Scand
Suppl. 554:138–152. 1986.PubMed/NCBI
|
|
16
|
Guide for the Care and Use of Laboratory
Animals. Washington, DC, 1996.
|
|
17
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Zhang S, Ge W, Zou G, Yu L, Zhu Y, Li Q,
Zhang Y, Wang Z and Xu T: MiR-382 targets GOLM1 to inhibit
metastasis of hepatocellular carcinoma and its down-regulation
predicts a poor survival. Am J Cancer Res. 8:120–131.
2018.PubMed/NCBI
|
|
19
|
Tan H, He Q, Gong G, Wang Y, Li J, Wang J,
Zhu D and Wu X: miR-382 inhibits migration and invasion by
targeting ROR1 through regulating EMT in ovarian cancer. Int J
Oncol. 48:181–190. 2016.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Xu M, Jin H, Xu CX, Sun B, Song ZG, Bi WZ
and Wang Y: miR-382 inhibits osteosarcoma metastasis and relapse by
targeting Y box-binding protein 1. Mol Ther. 23:89–98.
2015.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Kim VN, Han J and Siomi MC: Biogenesis of
small RNAs in animals. Nat Rev Mol Cell Biol. 10:126–139.
2009.PubMed/NCBI View
Article : Google Scholar
|
|
22
|
Okamura K: Diversity of animal small RNA
pathways and their biological utility. Wiley Interdiscip Rev RNA.
3:351–368. 2012.PubMed/NCBI View
Article : Google Scholar
|