Introduction
Lipoprotein (a) [Lp(a)] is a low-density,
cholesterol-containing Lp, whose role in cardiovascular disease
(CVD) is currently being re-evaluated. Lp(a) is different from
other low-density lipoproteins, as its structure comprises
apolipoprotein(a) attached to surface apolipoprotein B100(1). Increasing evidence suggests that
serum Lp(a) levels are genetically determined, remain unaltered for
years on a given individual and are not affected by diet or
exercise (2).
In clinical practice, a significant number of CVD
patients, with the target lipid levels set by the guidelines
achieved, continue to remain at risk. In this setting, Lp(a) role
in CVD prognosis is regaining interest. In a large Japanese study,
Lp(a) levels >30 mg/dl were reported to confer additional risk
for worse prognosis after percutaneous coronary intervention (PCI),
despite the fact that those patients had reached the target lipid
levels according to current guidelines at the time the study was
conducted, i.e., low-density lipoprotein (LDL) <100 mg/dl
(3). It is notable that 90% of
healthy Japanese present Lp(a) values <30 mg/dl (4).
Among acute myocardial infarction (AMI) patients
submitted to primary PCI within 24 h of its onset, those with Lp(a)
levels >40 mg/dl presented higher major adverse cardiac events
(MACE), specifically cardiac death, myocardial infarction and/or
revascularization during the following 5 years. Lp(a) levels were
an independent predictor of revascularization of new coronary
(non-culprit) lesions (5), while
revascularization for new lesions was the main component driving
MACE incidence.
Although Lp(a) is related to the arteriosclerotic
process (6), there is currently no
adequate amount of data for the inclusion of Lp(a) levels as a
primary therapeutic target in the treatment of coronary artery
disease (CAD) patients. In this framework, the current
retrospective study aimed to investigate the association of Lp(a)
levels with the adverse cardiovascular events presented in a 10
year follow-up of CVD patients with dyslipidemia and its
association with the major cardiovascular risk factors.
Materials and methods
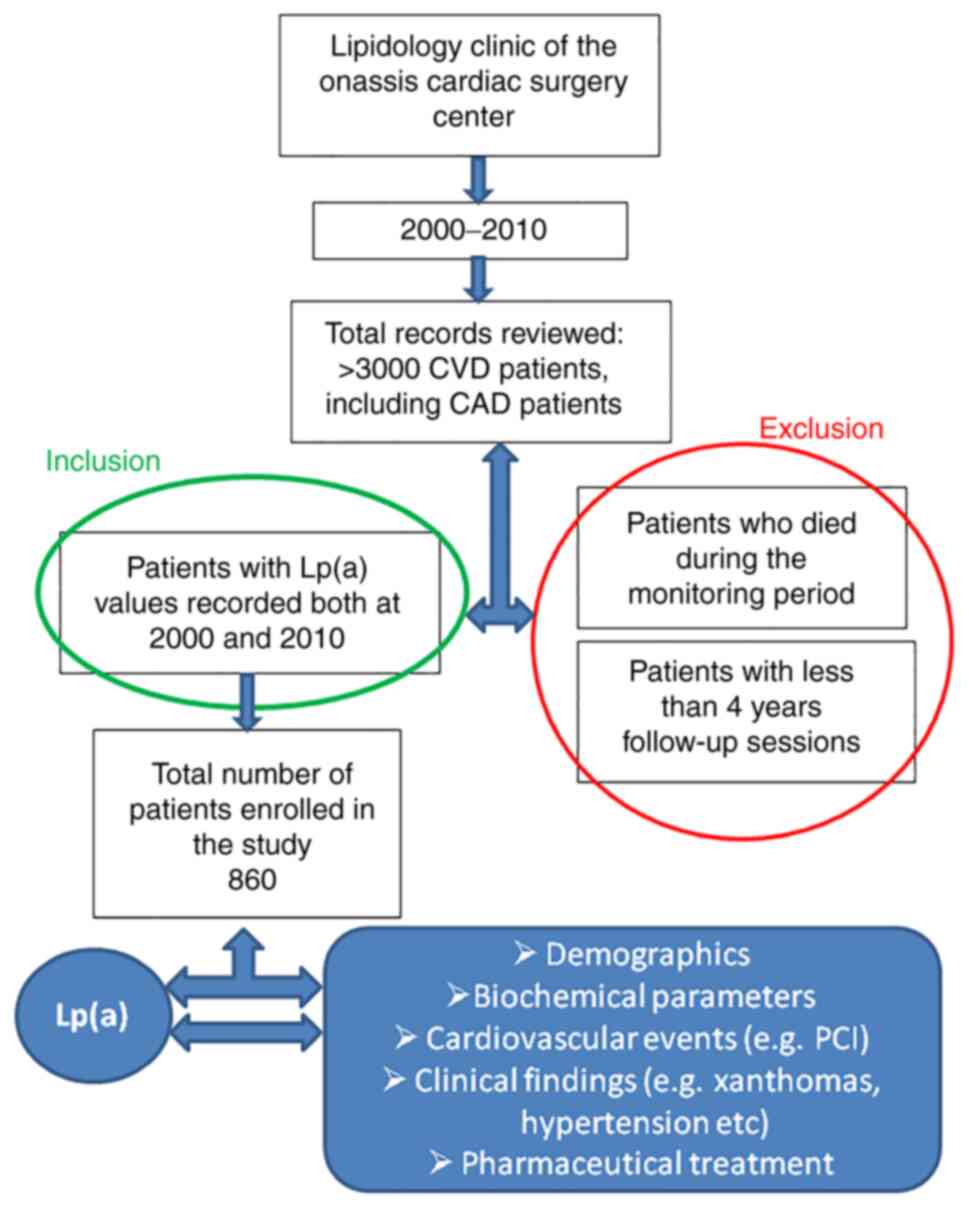
In the present retrospective cohort study,
consecutive CVD patients, including CAD patients, who were
monitored as outpatients at the lipidology clinic of the Onassis
Cardiac Surgery Center for a period of ~10 years (2000-2010), were
included. A total of more than 3,000 files were originally screened
(Table I). Data collection was
recorded at two distinct time intervals: at baseline and at the end
of the follow-up period. Only patients with Lp(a) values recorded
at both time intervals were enrolled in the study. Patients with
fewer than 4 years follow-up sessions, as well as those who died
during the monitoring period, were excluded. Age was not considered
as an exclusion criterion (Fig.
1).
 | Table IDemographics, somatometrics and
clinical history of the study population. |
Table I
Demographics, somatometrics and
clinical history of the study population.
| Parameter | Mean ± standard
deviation | Range |
|---|
| Age | | |
|
Baseline | 57.5±10.3 | 26-80 |
|
Follow-up | 67.9±10.3 | 38-90 |
| Height (cm) | 170.1±8.7 | 150-191 |
| Weight (kg) | 79.6±13.4 | 52-115 |
| Systolic blood
pressure (mmHg) | | |
|
Baseline | 132±14.2 | 100-170 |
|
Follow-up | 132±12.2 | 100-176 |
| Diastolic blood
pressure (mmHg) | | |
|
Baseline | 79.3±8.10 | 60.0-100 |
|
Follow-up | 79.6±7.45 | 60.0-100 |
| BMI
(kg/m2) | 27.3±3.4 | 19.6-38.8 |
| Sex | | |
|
Male (n,
%) | 628, 73.0 | |
|
Female (n,
%) | 232, 27.0 | |
| Parameter | Baseline | Follow-up | P-value |
| Smoking (n, %) | | | |
|
Yes | 260, 30.2 | 108, 12.6 | 0.015 |
|
No | 344, 40.0 | 340, 39.5 | 0.567 |
|
Ex-smoker | 256, 29.8 | 412, 47.9 | 0.009 |
| Xanthomas (n, %) | 32, 3.7 | 40, 4.72 | 0.500a |
|
Lp(a)
(mg/dl) (± standard deviation) | 104±31.5 | 95.5±36.4 | |
| Intermittent
claudication (n, %) | 20, 2.32 | 52, 6.04 |
0.039a |
|
Lp(a)
(mg/dl) (± standard deviation) | 91.3±31.9 | 76.4±66.0 | |
| Arterial
hypertension (n, %) | 400, 47.6 | 492, 57.2 |
0.005a |
|
Lp(a)
(mg/dl) (± standard deviation) | 71.3±44.3 | 63.7±41.3 | |
| Diabetes mellitus
(n, %) | 116, 13.5 | 208, 24.2 |
<0.001a |
|
Lp(a)
(mg/dl) (± standard deviation) | 73.6±44.3 | 59.9±37.1 | |
| Myocardial
infarction (n, %) | 72, 8.4 | 100, 11.6 |
<0.001a |
|
STEMI | 44 | 51 | |
|
Non
STEMI | 28 | 49 | |
|
Lp(a)
(mg/dl) (± standard deviation) | 74±41.8 | 74.2±41.9 | |
The current study was approved by the Ethics and
Bioethics Committee of Onasseio Cardiac Surgery Center, Athens
(reference no. 368/05.09.2008; Management Board approval
14.01.2009).
All study participants were informed in detail and
agreed at the time of their evaluation to the publication of
associated data as appropriate, fully respecting their anonymity
and medical ethics.
Biochemical analysis
Almost all subjects in the study had had a complete
determination of the lipid profile, which included, apart from
Lp(a), levels of total serum cholesterol, high density lipoprotein
(HDL)- and LDL-cholesterol, triglycerides, apolipoprotein A1 and
apolipoprotein B 100 levels. Blood glucose levels, fibrinogen,
homocysteine, and C-reactive protein (CRP) were also measured as
part of the inflammatory profile of individuals with dyslipidemia
and coronary heart disease.
Total cholesterol and triglycerides were determined
by CHOD Abell-Kendall and Lipase/GPO-PAP enzyme chromatographic
methods, respectively, using an automated biochemical analyzer,
while LDL cholesterol, according to the Friedewald formula
(triglycerides needed to be <400 mg/dl). HDL-C was enzymatically
determined from the supernatant after precipitation of the
remaining lipoproteins using phosphotungstic acid and magnesium
chloride. Apolipoprotein A1, apolipoprotein B and CRP were
calculated by an immunoenzymatic assay using nephelometry (mg/dl)
and homocysteine by chemiluminescence.
All measurements were performed on Roche Integra
Biochemical analyzer (Roche Diagnostics) with the commercially
available kits (Roche). The laboratory is subject to external
quality control Randox International Quality Assessment Scheme
(RIQAS) of the company RANDOX with a monthly ‘certificate of
acceptable performance’ and an ISO 9001 certificate.
For Lp(a) determination, all measurements were
typically performed after an overnight fasting, the nephelometric
method (INA) was applied with the help of particles coated with
specific Lp(a) antibody (Dade Behring Marburg GmbH, USA) (7).
Statistical analysis
Continuous variables were expressed with the use of
mean values and standard deviation, while nominal variables were
expressed by frequencies and percentage frequencies. A paired
samples t-test was used to identify differences between values of
the same continuous variable at two different time points.
Differences in mean values of different continuous variables were
estimated with the use of independent samples t-test.
The Kruskal-Wallis non-parametric test followed by
Dunn's test was used to compare differences between >2 groups of
continuous non-normally distributed independent variables.
Spearman's correlation coefficient was used to explore non-linear
correlations, as depicted by Scatter plots, between two variables.
McNemar test was used for the estimation of changes in independent
variables between two measurements of the same variable. Graphical
representation of continuous variables was made with the use of box
plots and scatter plots. Kaplan-Meier survival curves were produced
and the log-rank test was used to study the need (risk) for PCI
over time (event of interest) in relation to Lp(a) levels.
Statistical analysis was performed with the use of IBM SPSS
Statistics 23 statistical package (IBM Corp.). P<0.005 was
considered to indicate a statistically significant difference.
Results
Demographics, biochemical markers and
treatment
A total of 860 CVD patients were finally enrolled in
the study, including CAD patients and patients with high lipid
levels. The demographics of the study population at baseline are
summarized in Table I. It is worth
noting that upon the end of the follow-up, smokers were reduced by
12.6%, with ex-smokers raising to 47.9% of all patients.
Biochemical markers levels at baseline and at the
end of the follow-up are presented in Table II.
 | Table IIBiochemical profile of the study
population. |
Table II
Biochemical profile of the study
population.
| | Baseline
values | Follow-up | |
|---|
| Parameter | No. of
patients | Mean | Standard
deviation | Mean | Standard
deviation | P-value |
|---|
| Glucose
(mg/dl) | 856 | 98.4 | 15.9 | 104 | 19.7 |
<0.001 |
| Creatinine
(mg/dl) | 664 | 1 | 0.3 | 1.6 | 3.9 | 0.054 |
| Urea (mg/dl) | 629 | 39 | 15.2 | 43 | 24 | 0.004 |
| Uric acid
(mg/dl) | 524 | 6 | 1.7 | 6.1 | 1.4 | 0.459 |
| Homocysteine
(µmol/l) | 480 | 13.7 | 5.6 | 11.3 | 3.3 |
<0.001 |
| CRP (mg/l) | 584 | 2.0 | 0.2 | 2.0 | 0.5 | 0.592 |
| Total
cholesterola
(mg/dl) | 740 | 186.4 | 32.4 | 169.8 | 26.4 |
<0.001 |
|
Triglycerides1 (mg/dl) | 647 | 119 | 48.4 | 113.1 | 44.8 | 0.074 |
| HDLa (mg/dl) | 668 | 43.5 | 10.8 | 46.4 | 11.4 |
<0.001 |
| LDLa (mg/dl) | 664 | 117.6 | 28 | 101.8 | 23.4 |
<0.001 |
| CPK (U/l) | 513 | 99.1 | 52.7 | 109.8 | 54 | 0.009 |
| Lp(a) (mg/dl) | 860 | 72.8 | 45.2 | 68.3 | 41.8 |
<0.001 |
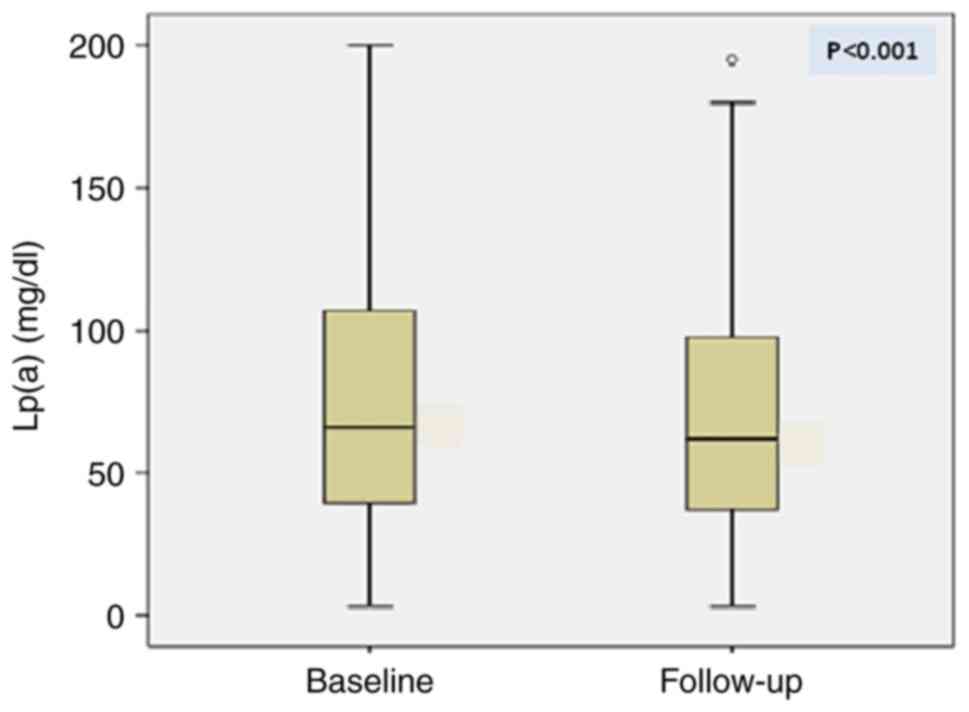
A statistically significant reduction in Lp(a)
levels was observed during the follow up period (72.8±45.6 vs.
68.3±41.8 mg/dl; P<0.001), (Fig.
2).
Lp(a) levels according to
anti-lipidemic treatment
Table III
presents the anti-lipidemic treatment applied in the study
population.
 | Table IIIAnti-lipidemic treatment applied in
the study population. |
Table III
Anti-lipidemic treatment applied in
the study population.
| Variable | Statins | Fibrates | Nicotinic acid
derivatives/n-3 fatty acids | No
treatmenta |
|---|
| Number of patients
treated | 756 | 11 | 9 | 22 |
|
Co-administration | - | 7c | 9c | |
| Lp(a),
mg/dlb | 75.3±45.4 | 52.8±48.6 | 65.0±54.8 | |
| Active
substances | Atrovastatin
(271) | Bezafibrate
(4) | Acipimox (8) | |
| (number of patients
treated) | Simvastatin
(280) | Fenofibrate
(6) | Eicosapentaenoic
acid (1) | |
| | Pravastatin
(133) | Gemfibrozil
(1) | | |
| | Fluvastatin
(64) | | | |
| | Rosuvastatin
(8) | | | |
Lp(a) levels at baseline were higher with the use of
statins (75.3±45.4 mg/dl) compared with the rest of the study
population (both those with other anti-lipidemic treatment and
those with no treatment) (54.1±40.3 mg/dl in the rest of the study
population; P=0.027). Similar results were obtained at the end of
the follow-up period (Lp(a)=70.2±41.4 mg/dl in the statin group vs.
53.6±42.0 mg/dl; P=0.031). Fibrates and nicotinic acid derivatives
lower Lp(a) levels at the end of the follow-up by ~10% with no
statistical significance.
Lp(a) levels and clinical history of
patients
The clinical history of the patients and the
respective Lp(a) levels at the two monitored time intervals of the
present study are also summarized in Table I. Patients that presented xanthomas
increased non-statistically significantly by eight patients (0.9%
of the patients) at the end of the follow-up period (McNemar test;
P=0.5). Intermittent claudication diagnosis, on the other hand, was
not verified in four (0.5%) of these patients in the second
evaluation, but appears at another 32 patients (3.7%; McNemar test;
P=0.039). New presentation of arterial hypertension at the end of
the follow-up was reported for 92 patients (10.7%) (McNemar test;
P=0.005), while another 92 patients were diagnosed with increased
blood glucose levels at the second evaluation (diabetes mellitus,
24.2%) (McNemar test; P<0.001). The increase in the frequency of
high blood pressure and diabetes mellitus at the end of the
follow-up is statistically significantly associated with the Lp(a)
levels at that time interval (bivariate correlation, P=0.02 and
P=0.046, respectively), while there was no statistically
significant correlation between the incidences of hypertension and
diabetes and Lp(a) values or the change in Lp(a) values at baseline
and at the end of the follow-up period.
Lp(a) correlations
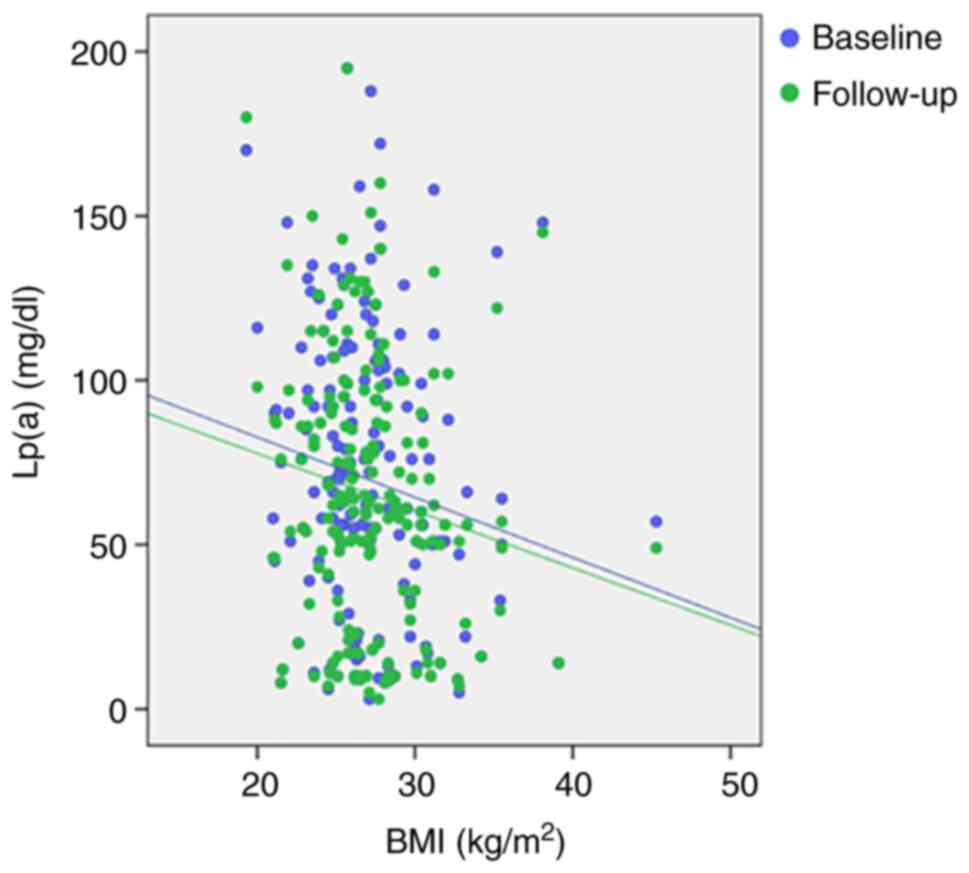
Lp(a) levels both at baseline and at the end of the
follow-up were weakly and negatively correlated with body mass
index (BMI) (Spearman's correlation r=-0.171, P=0.022; r=-0.153,
P=0.040, respectively; Fig.
3).
Lp(a) values correlation to biochemical and
hematological measurements are presented in Table IV. Fasting glucose levels and
hematocrit were significantly but weakly inversely correlated to
Lp(a) values both at baseline and at the end of the follow up
period. Total cholesterol and LDL levels were correlated
significantly but not strongly to Lp(a) values both at baseline and
at study end. No sex-related differences regarding Lp(a) levels
were found in our study population.
 | Table IVCorrelation of Lp(a) levels with
clinical parameters monitored during the study. |
Table IV
Correlation of Lp(a) levels with
clinical parameters monitored during the study.
| | Βaseline Lp(α)
values | Follow-up Lp (α)
values |
|---|
| Parameter |
Rsa | P-value |
Rsa | P-value |
|---|
| SBP (mmHg) | -0.004 | 0.954 | -0.140 | 0.072 |
| DBP (mmHg) | -0.047 | 0.503 | -0.098 | 0.210 |
| Fasting glucose
(mg/dl) | -0.151 | 0.027 | -0.202 | 0.003 |
| Creatinine
(mg/dl) | 0.104 | 0.152 | 0.042 | 0.576 |
| Urea (mg/dl) | -0.069 | 0.351 | -0.004 | 0.957 |
| Uric acid
(mg/dl) | -0.081 | 0.334 | 0.063 | 0.457 |
| Hematocrit (%) | -0.202 | 0.003 | -0.143 | 0.037 |
| Blood Platelets
(per µl) | 0.014 | 0.833 | 0.132 | 0.054 |
| Homocysteine
(µmol/l) | 0.112 | 0.192 | 0.044 | 0.624 |
| CRP (mg/l) | -0.021 | 0.790 | 0.007 | 0.931 |
| Total
cholesterolb
(mg/dl) | 0.200 | 0.006 | 0.269 |
<0.001 |
|
Triglyceridesb (mg/dl) | 0.028 | 0.706 | -0.015 | 0.837 |
| HDLb (mg/dl) | -0.061 | 0.415 | -0.134 | 0.068 |
| LDLb (mg/dl) | 0.218 | 0.003 | 0.287 |
<0.001 |
| Apolipoprotein
Ab (mg/dl) | 0.061 | 0.562 | 0.014 | 0.968 |
Lp(a) levels and AMI, PCI
The vast majority of patients who suffered a new AMI
during the follow up period had Lp(a) levels >30 mg/dl (24/28
patients, mean ± standard deviation Lp(a), 83.1±36.6, P=0.001).
Women appeared to develop AMI later than men (58.8±8.02 vs.
50.6±9.08 years of age).
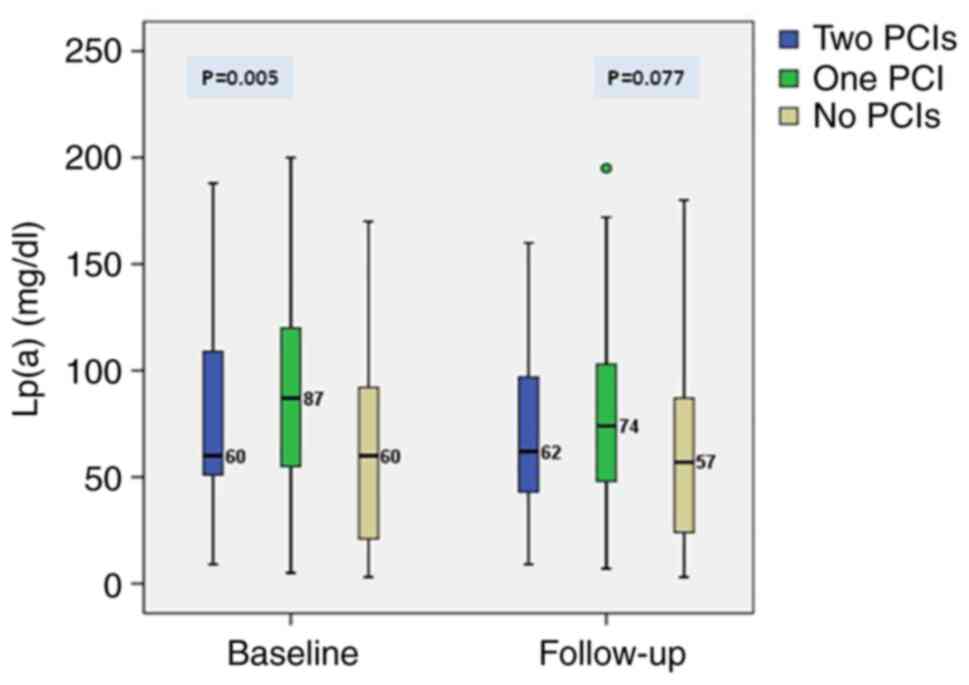
At baseline, patients already submitted to PCI
(n=272) had significantly higher Lp(a) compared with the rest of
study patients (83.4±45.1 vs. 67.7±44.5 mg/dl, P=0.016). Similar
findings were observed at the end of the follow-up (PCI patients
n=424, mean ± standard deviation, 76.7±41.8 vs. 64.1±41.3 mg/dl,
P=0.038). At baseline Lp(a) levels were significant different
between patient with a clinical history of one, two or no PCI in
their clinical history. At study end, the said differences in Lp(a)
seemed to be attenuated (Fig.
4).
Kaplan-Meier survival analysis
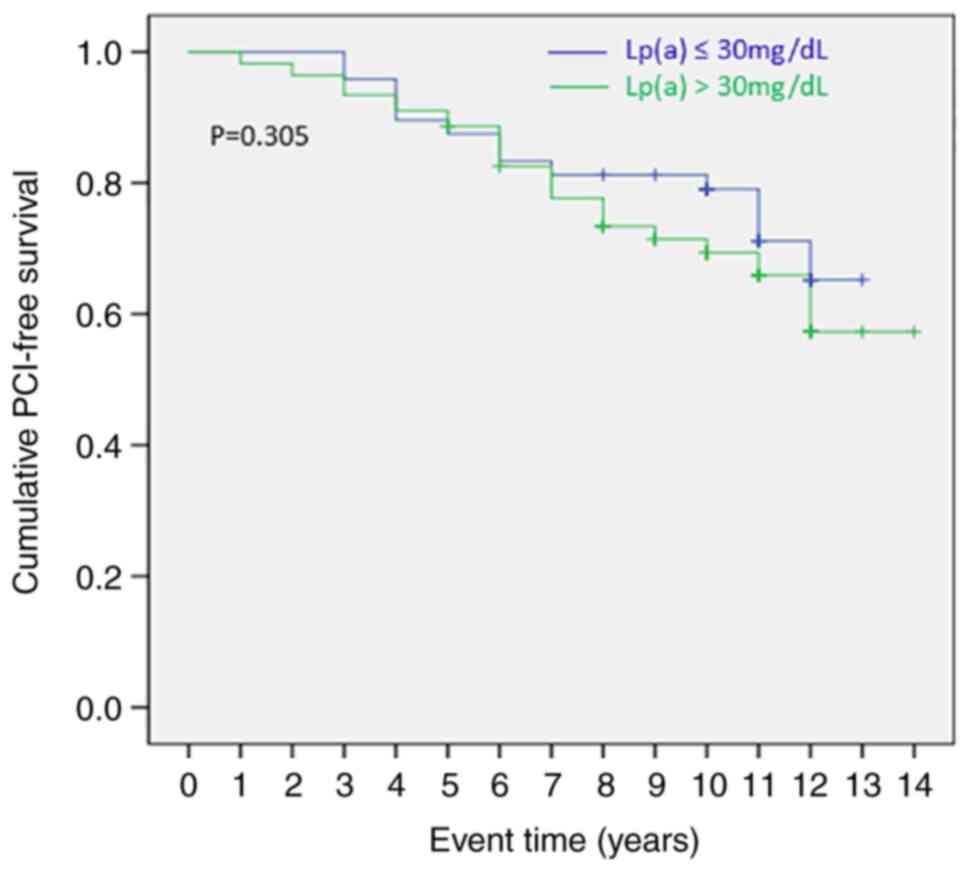
Kaplan-Meier survival analysis did not find
statistically significant differences in the event time (occurrence
of a PCI until the second evaluation) between patients with low
Lp(a) levels (≤30 mg/dl compared with patients with higher Lp(a)
levels (>30 mg/dl; log-rank P=0.305; Fig. 5). In addition, no significant
differences were found by Kaplan-Meier survival analysis when the
threshold for Lp(a) was set to 50 mg/dl (log-rank P=0.866). Even a
higher Lp(a) cut-off value of 80 mg/dl did not lead to
significantly different results in the time a PCI should occur
(log-rank P=0.145) (data not shown).
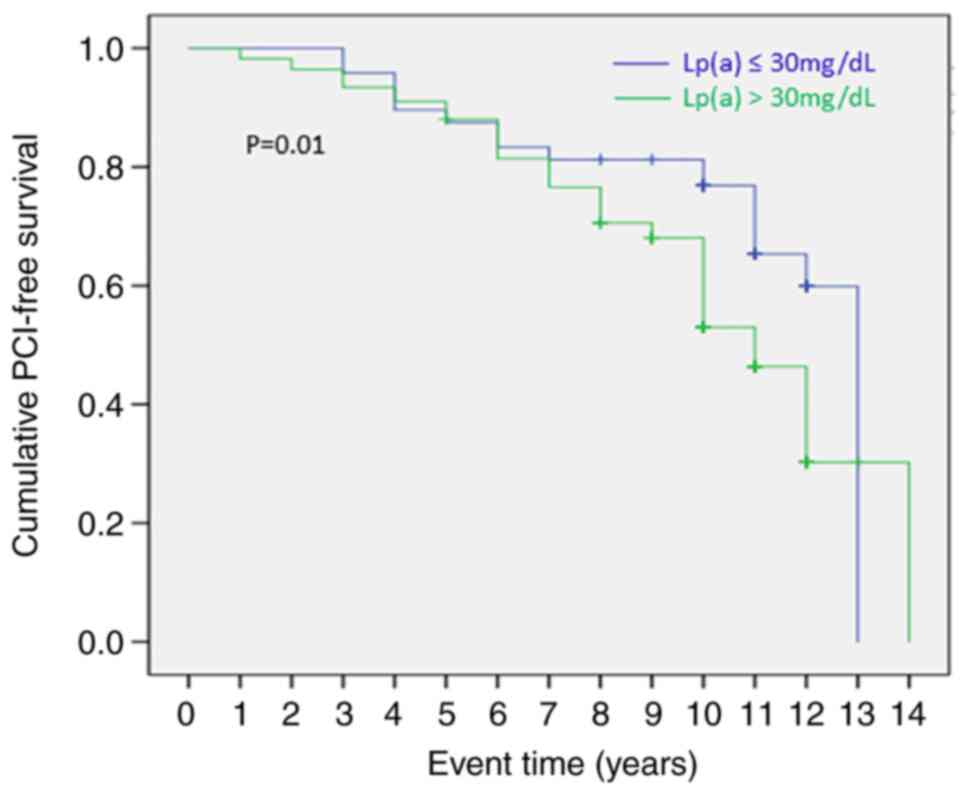
On the other hand, Kaplan-Meier analysis for
patients with at least one PCI in the monitoring period including
as events in the analysis (second or third PCIs in the monitoring
period), the mean event time for a PCI during the study period was
significantly shorter (10.2 years vs. 11 years, P=0.01) for
patients with Lp(a) levels >30 mg/dl (Fig. 6), suggesting that when taking into
account the most vulnerable coronary patients with more than one
PCI, Lp(a) values >30 mg/dl are associated with shorter need for
revascularization.
Discussion
Mendelian randomization studies offer new evidence
for Lp(a) role in promoting CVD (6,8).
Genetic polymorphisms leading to increased Lp(a) levels are in fact
associated with CVD and its adverse events, such as MI (9). The effects of anthropometric
parameters and everyday activities on Lp(a) levels have also been
studied. Reports of a non-fully explained rise of Lp(a) with diet
and exercise exist in literature (10). At the same time, it is found that
an interaction with additive effect exists between BMI and Lp(a) in
the risk for a first MI (11). In
the current study, Lp(a) levels decreased significantly during the
follow-up period, while a weak inverse relationship was observed
between Lp(a) levels and BMI.
Lp(a) is causally linked to atherosclerotic disease
progression. Pathophysiology describes the ability of Lp(a) to
enter the vessel wall and accumulate in the macrophages along with
cholesterol, thus leading to the formation of foam cells, fatty
streaks and atherosclerotic plaques. At the same time, Lp(a)
inactivates transforming growth factor and augments smooth muscle
cell proliferation in atherosclerotic lesions (5,12).
Lp(a) ability to promote atherosclerosis is mediated
also by oxidized phospholipids (OxPL) of which Lp(a) is the
preferential carrier (13). The
role of OxPL is thought to be crucial in plaque destabilization.
OxPl are immunogenic and are found in atherosclerotic lesions. OxPL
modify Lp(a) primarily by covalent binding to its unique apo(a)
component and promote inflammation, endothelial dysfunction and
calcification. Plaque vulnerability, evident by thin cap
fibroatheroma, a state with augmented potential for plaque rupture,
is linked to OxPL and Lp(a) presence. In advanced ‘vulnerable’
human carotid artery thin-cap fibroatheroma the 2 molecules were
found to co-exist (14). At the
same time Apo(a) possesses unique properties leading to arterial
wall inflammation and atherosclerosis progression (14).
Rapid angiographic progression of CAD could be
attributed in part to Lp(a) as a study with repetitive coronary
angiography on average 60 days from each other described (15). The partial structural homology of
Lp(a) with plasminogen was pinpointed but also intracoronary plaque
rupture and thrombosis through the plasminogen-like apo(a) moiety
could be the missing link. Lp(a) preferentially enters and deposits
to the vessel wall as a result of apo(a) binding to extracellular
matrix proteins (16). An
apolipoprotein(a) antisense oligonucleotide ended phase 2 trials
showing promising results in safe reduction of Lp(a) (17). However today no drugs specifically
aimed at lowering Lp(a) are currently clinically available
(18). With the approval of the
antisense oligonucleotide for clinical use, individuals with
established CVD will probably be selected as the first recipients
in an effort to minimize recurrent CVD events to them (19).
It is estimated that Lp(a) levels >25-30 mg/dl
are present in ~30% of Caucasians and 60-70% of African Americans
(i.e., 100,000,000 Americans) (14).
Regarding secondary vascular events in CAD patients,
Lp(a) cut-off values as low as 19 mg/dl are important in
discriminating patients with a higher probability of MACE for a
follow-up period of 3 years post MI (20). Konishi et al (3) report that in patients on statin
therapy undergoing PCI for the first time, lp(a) levels >21.5
mg/dl were linked to higher rates of MACE, namely cardiac mortality
and acute coronary syndromes. In the present study, on the other
hand, Lp(a) levels >30 mg/dl were significantly linked to an
earlier need of revascularization, only when repeated
revascularizations in the study monitoring period were included in
the Kaplan Meier survival analysis. It is interesting to note that
LDL levels in the study population of the present study were close
enough to current guidelines at the time the study was
conducted.
It is known that niacin and estrogens lower Lp(a) up
to 30%, but that statins either have no effect or increase Lp(a)
levels, occasionally significantly (21). In the current study, statin
treatment led to higher Lp(a) levels both at baseline and at the
end of the follow-up.
In the primary care setting, when the findings of
two former large studies, the European Prospective Investigation of
Cancer (EPIC)-Norfolk prospective population study and the
Copenhagen City Heart Study (CCHS) prospective population study,
were re-analyzed, at LDL-C levels below <96 mg/dl, the risk
associated with elevated Lp(a) was attenuated (22). No significant interaction existed
between corrected LDL and Lp(a) levels on CVD risk, thus meaning
that both high Lp(a) and LDL levels contribute to CVD occurrence
independently. It is interesting to note that in the said studies
the cut-off Lp(a) of 50 mg/dl represents different percentile
values: the 87th percentile in EPIC-Norfolk and the 80th percentile
in CCHS cohort. In the current study with mean LDL values slightly
over 100 mg/dl, different Lp(a) cut-off values were weakly able to
discriminate progressive CAD evident by the need for
revascularization.
New evidence from meta-analysis of large community
trials suggests that a 10 mg/dl lower plasma Lp(a) was associated
with a multifactorial adjusted hazard ratio for MACE of 0.96 (95%
CI, 0.94-0.98) during 5 years of follow-up in patients with CVD
(19). In addition, in the most
recent Plaque at RISK study, patients with symptomatic carotid
artery stenosis were characterized by increased Lp(a) which was
associated with degree of stenosis and other vulnerable plaque
characteristics (23).
In conclusion, the current study reported that
patients with high Lp(a) values are more prone to the occurrence of
5 MI, while the Lp(a) cut-off value of 30 mg/dl is linked in CVD
patients to an earlier need for PCI, especially in the most
vulnerable group of patients with more than one (recurrent)
revascularizations.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
AH and KT organized and performed the research,
collected relevant information, wrote the manuscript and performed
overall project management. AH and GK confirm the authenticity of
all the raw data. CT performed the statistical analysis, data
assessment and manuscript preparation. GK, AV, IH and DAS performed
the statistical analysis and the evaluation of the results, and
were involved in the preparation and writing of the research
article. HM, ME and ST reviewed the manuscript and comprehensively
assessed the study design and the data analysis, prepared and wrote
the manuscript, organized the references and reviewed the current
study. All authors have read and approved the final version of this
manuscript.
Ethics approval and consent to
participate
The current study was approved by the Ethics and
Bioethics Committee of Onasseio Cardiac Surgery Center, Athens
(reference no. 368/05.09.2008; Management Board approval
14.01.2009). All procedures were in accordance with the ethical
standards of the patient's evaluation reports (institutional and
national) and in agreement with the Helsinki Declaration of 1964
and later versions. All the data were original, used in the current
study with anonymity and confidentiality. Informed consent was
obtained from all study participants at the time of their
evaluation. All study participants were informed in detail and
agreed at the time of their evaluation to the publication of
associated data as appropriate, fully respecting their anonymity
and medical ethics.
Patient consent for publication
Not applicable.
Competing interests
DAS is the Editor-in-Chief for the journal, but had
no personal involvement in the reviewing process, or any influence
in terms of adjudicating on the final decision, for this article.
The other authors declare that they have no competing
interests.
References
|
1
|
Shah NP, Pajidipati NJ, McGarrah RW, Navar
AM, Vemulapalli S, Blazing MA, Shah SH, Hernandez AF and Patel MR:
Lipoprotein (a): An update on a marker of residual risk and
associated clinical manifestations. Am J Cardiol. 126:94–102.
2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Seed M, Ayres KL, Humphries SE and Miller
GJ: Lipoprotein (a) as a predictor of myocardial infarction in
middle-aged men. Am J Med. 110:22–27. 2001.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Konishi H, Miyauchi K, Kasai T, Tsuboi S,
Ogita M, Naito R, Sai E, Fukushima Y, Katoh Y, Okai I, et al:
Impact of lipoprotein(a) as residual risk on long-term outcomes in
patients after percutaneous coronary intervention. Am J Cardiol.
115:157–160. 2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Abe A and Noma A: Studies on
apolipoprotein(a) phenotypes. Part 1. Phenotype frequencies in a
healthy Japanese population. Atherosclerosis. 96:1–8.
1992.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Ikenaga H, Ishihara M, Inoue I, Kawagoe T,
Shimatani Y, Miura F, Nakama Y, Dai K, Otani T, Ejiri K, et al:
Usefulness of lipoprotein (a) for predicting progression of
non-culprit coronary lesions after acute myocardial infarction.
Circ J. 75:2847–2852. 2011.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Nordestgaard BG and Langsted A:
Lipoprotein (a) as a cause of cardiovascular disease: Insights from
epidemiology, genetics, and biology. J Lipid Res. 57:1953–1975.
2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Tsarouhas K, Tsitsimpikou C, Haliassos A,
Georgoulias P, Koutsioras I, Kouretas D, Kogias J, Liosis I,
Rentoukas E and Kyriakides Z: Study of insulin resistance, TNF-α,
total antioxidant capacity and lipid profile in patients with
chronic heart failure under exercise. In Vivo. 25:1031–1037.
2011.PubMed/NCBI
|
|
8
|
Clarke R, Peden JF, Hopewell JC, Kyriakou
T, Goel A, Heath SC, Parish S, Barlera S, Franzosi MG, Rust S, et
al: Genetic variants associated with Lp(a) lipoprotein level and
coronary disease. N Engl J Med. 361:2518–2528. 2009.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Kamstrup PR, Tybjaerg-Hansen A, Steffensen
R and Nordestgaard BG: Genetically elevated lipoprotein(a) and
increased risk of myocardial infarction. JAMA. 301:2331–2339.
2009.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Randall OS, Feseha HB, Illoh K, Xu S,
Ketete M, Kwagyan J, Tilghman C and Wrenn M: Response of
lipoprotein(a) levels to therapeutic life-style change in obese
African-Americans. Atherosclerosis. 172:155–160. 2004.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Teng RL, Wang H, Sun BC, Cai DP and He YM:
Interaction between lipoprotein (a) levels and body mass index in
first incident acute myocardial infarction. BMC Cardiovasc Disord.
20(350)2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Grainger DJ, Kirschenlohr HL, Metcalfe JC,
Weissberg PL, Wade DP and Lawn RM: Proliferation of human smooth
muscle cells promoted by lipoprotein(a). Science. 260:1655–1658.
1993.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wilson DP, Jacobson TA, Jones PH,
Koschinsky ML, McNeal CJ, Nordestgaard BG and Orringer CE: Use of
lipoprotein(a) in clinical practice: A biomarker whose time has
come. A scientific statement from the national lipid association. J
Clin Lipidol. 13:374–392. 2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Tsimikas S and Hall JL: Lipoprotein(a) as
a potential causal genetic risk factor of cardiovascular disease: A
rationale for increased efforts to understand its pathophysiology
and develop targeted therapies. J Am Coll Cardiol. 60:716–721.
2012.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Terres W, Tatsis E, Pfalzer B, Beil FU,
Beisiegel U and Hamm CW: Rapid angiographic progression of coronary
artery disease in patients with elevated lipoprotein(a).
Circulation. 91:948–950. 1995.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Reyes-Soffer G, Ginsberg HN, Berglund L,
Duell PB, Heffron SP, Kamstrup PR, Lloyd-Jones DM, Marcovina SM,
Yeang C, Koschinsky ML, et al: Lipoprotein(a): A genetically
determined, causal, and prevalent risk factor for atherosclerotic
cardiovascular disease: A scientific statement from the American
heart association. Arterioscler Thromb Vasc Biol. 42:e48–e60.
2022.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Schreml J and Gouni-Berthold I:
Apolipoprotein(a) antisense oligonucleotides: A new treatment
option for lowering elevated lipoprotein(a)? Curr Pharm Des.
23:1562–1570. 2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Viney NJ, van Capelleveen JC, Geary RS,
Xia S, Tami JA, Yu RZ, Marcovina SM, Hughes SG, Graham MJ, Crooke
RM, et al: Antisense oligonucleotides targeting apolipoprotein(a)
in people with raised lipoprotein(a): Two randomised, double-blind,
placebo-controlled, dose-ranging trials. Lancet. 388:2239–2253.
2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Madsen CM, Kamstrup PR, Langsted A, Varbo
A and Nordestgaard BG: Lipoprotein(a)-lowering by 50 mg/dl (105
nmol/l) may be needed to reduce cardiovascular disease 20% in
secondary prevention: A population-based study. Arterioscler Thromb
Vasc Biol. 40:255–266. 2020.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Mitsuda T, Uemura Y, Ishii H, Takemoto K,
Uchikawa T, Koyasu M, Ishikawa S, Miura A, Imai R, Iwamiya S, et
al: Lipoprotein(a) levels predict adverse vascular events after
acute myocardial infarction. Heart Vessels. 31:1923–1929.
2016.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Taleb A, Witztum JL and Tsimikas S:
Oxidized phospholipids on apoB-100-containing lipoproteins: A
biomarker predicting cardiovascular disease and cardiovascular
events. Biomark Med. 5:673–694. 2011.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Verbeek R, Hoogeveen RM, Langsted A,
Stiekema LCA, Verweij SL, Hovingh GK, Wareham NJ, Khaw KT,
Boekholdt SM, Nordestgaard BG and Stroes ESG: Cardiovascular
disease risk associated with elevated lipoprotein(a) attenuates at
low low-density lipoprotein cholesterol levels in a primary
prevention setting. Eur Heart J. 39:2589–2596. 2018.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Dilba K, van Dijk AC, Crombag GAJC, van
der Steen AFW, Daemen MJ, Koudstaal PJ, Nederkoorn PJ, Hendrikse J,
Kooi ME, van der Lugt A and Wentzel JJ: Association between
intraplaque hemorrhage and vascular remodeling in carotid arteries:
The plaque at RISK (PARISK) study. Cerebrovasc Dis. 50:94–99.
2021.PubMed/NCBI View Article : Google Scholar
|