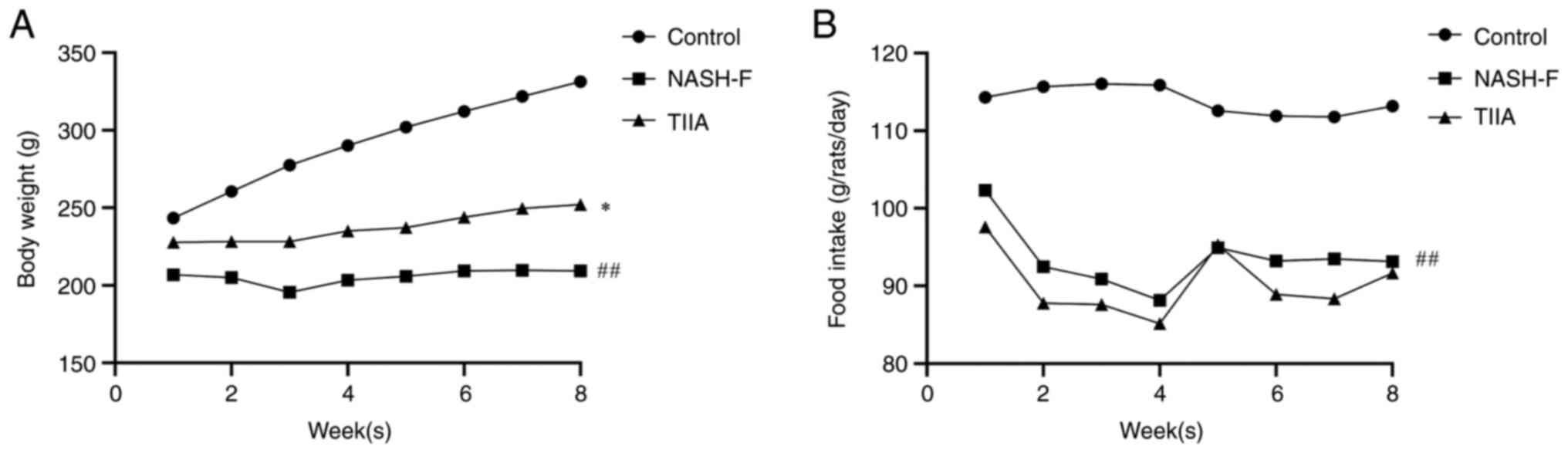
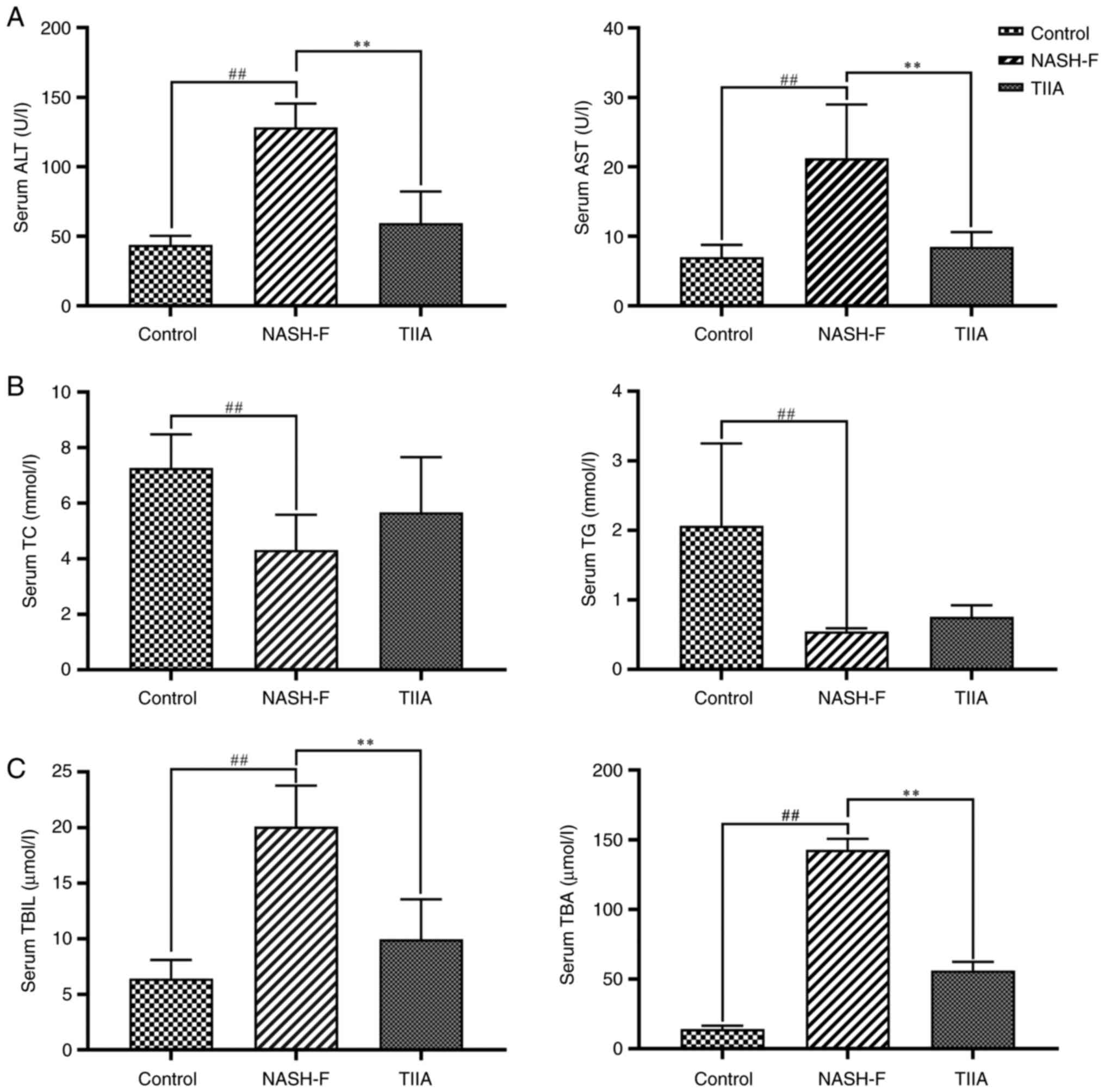
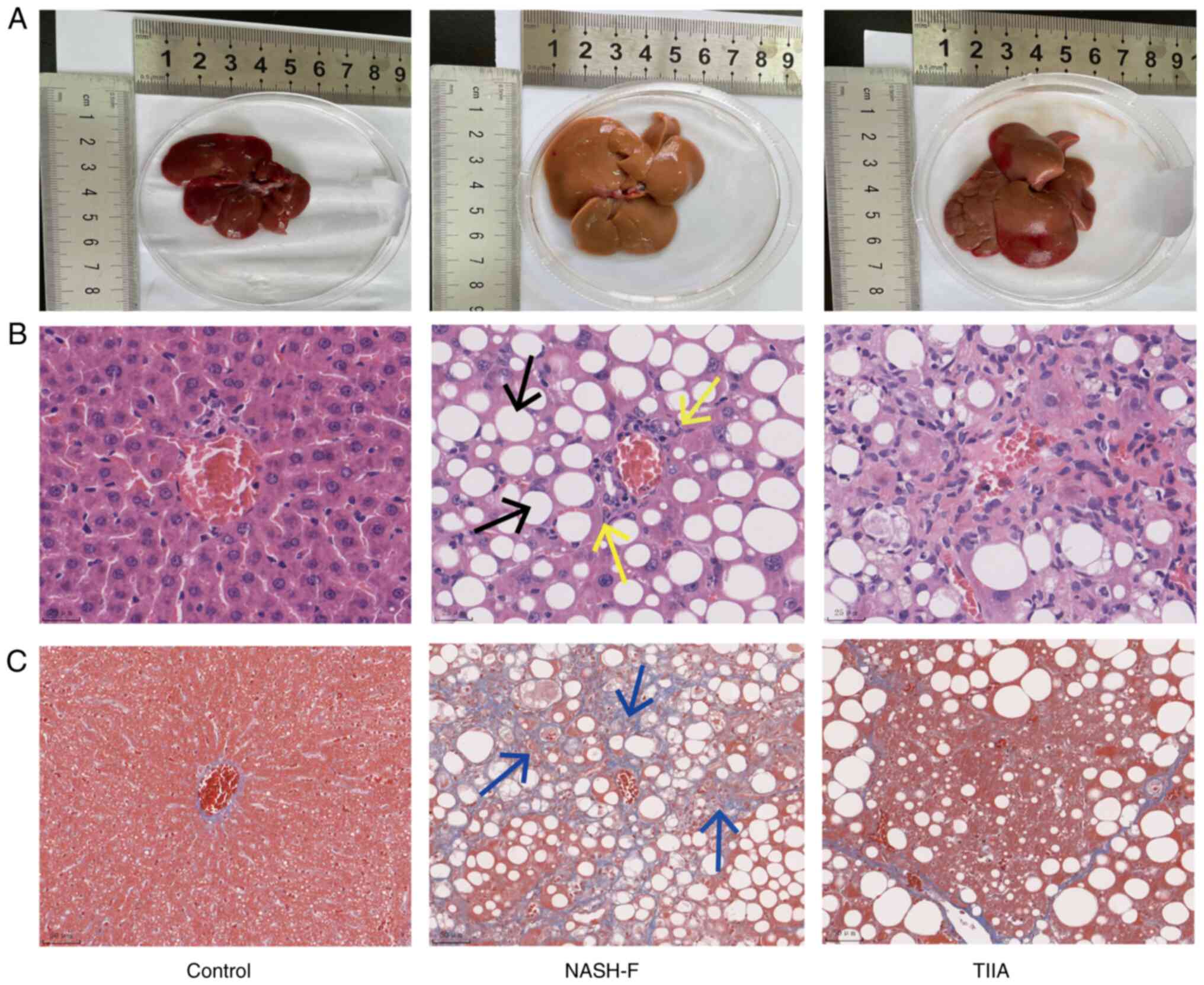
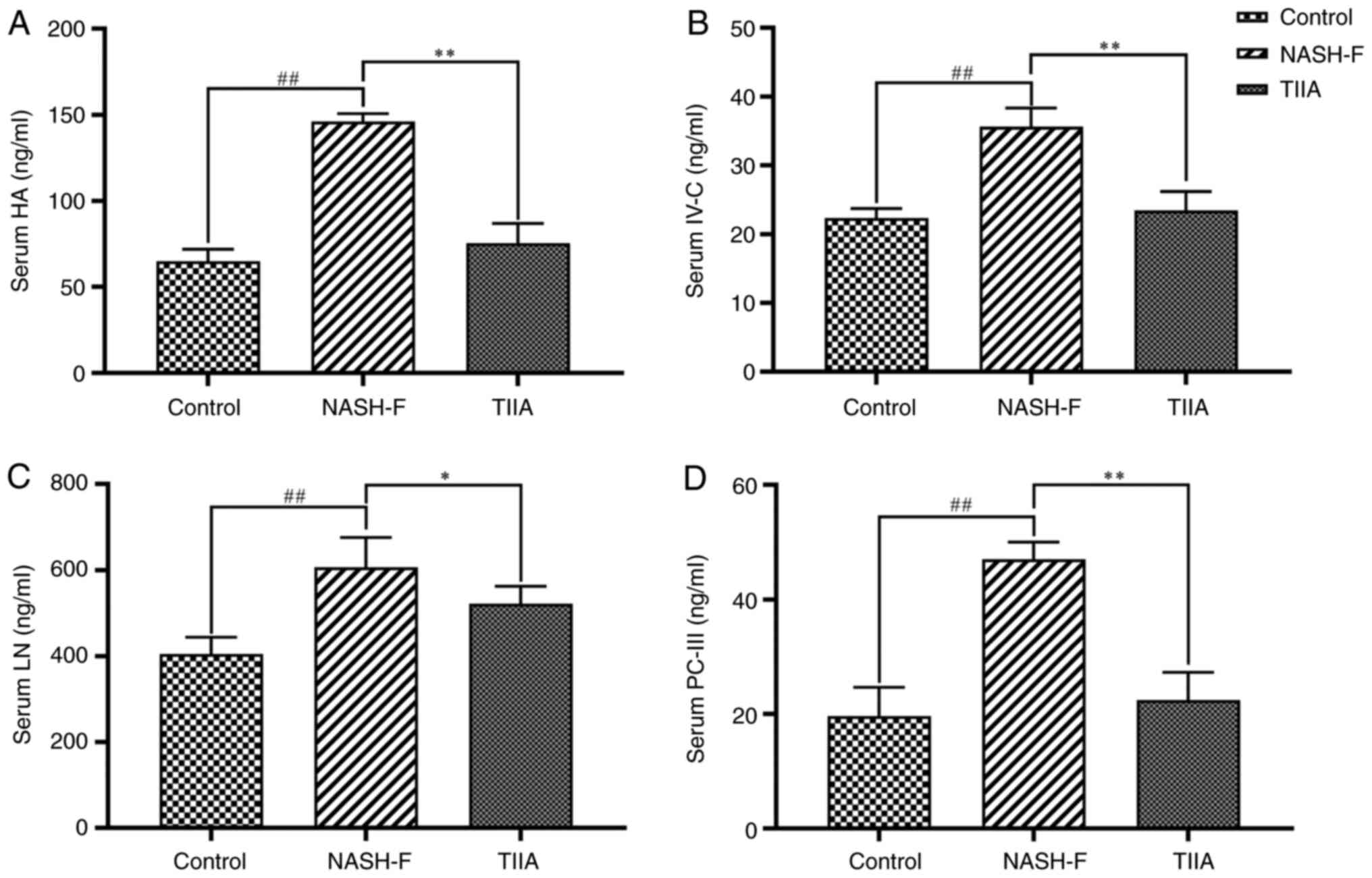
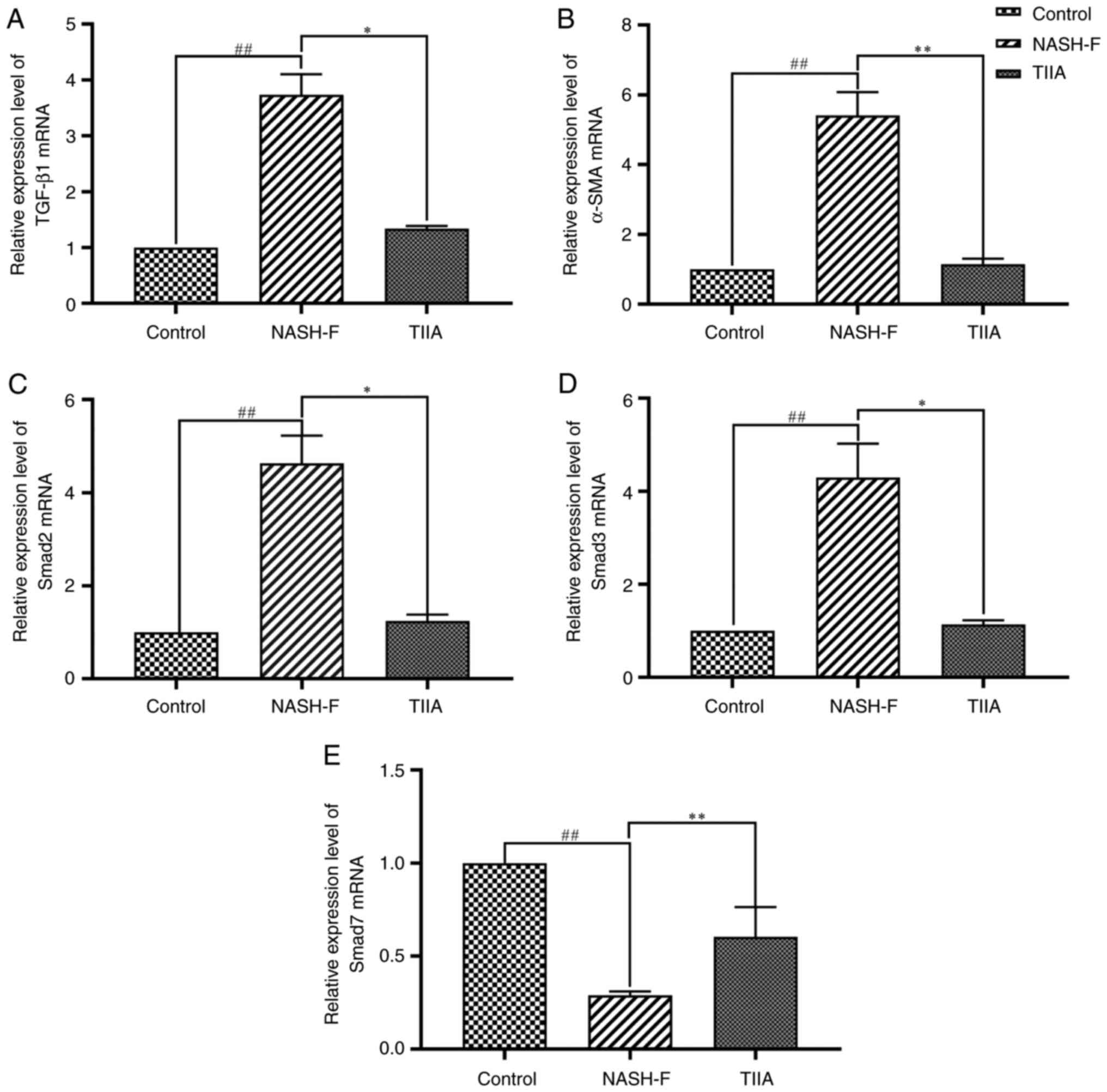
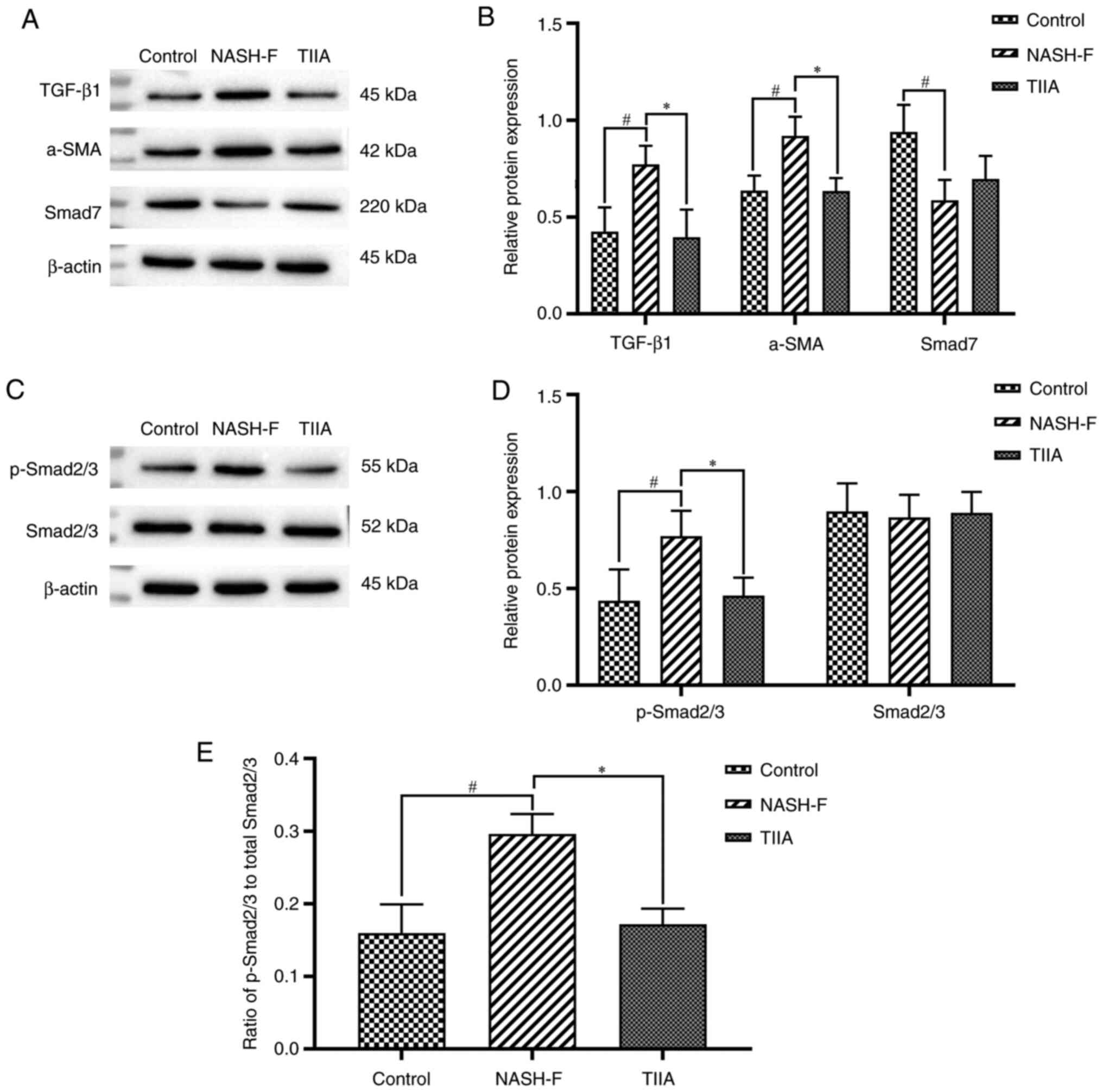
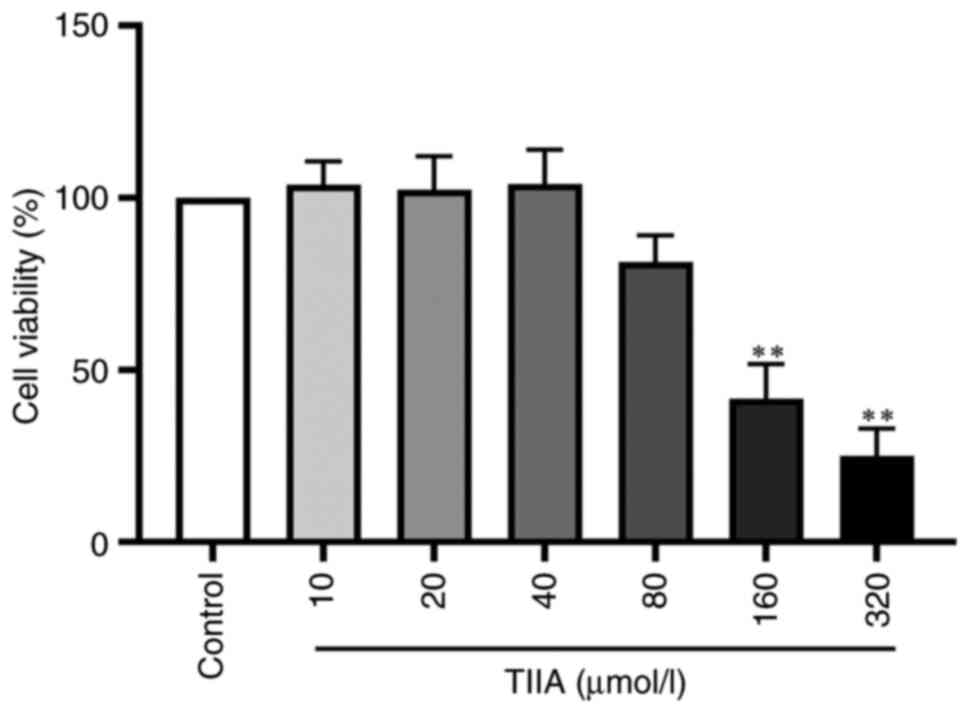
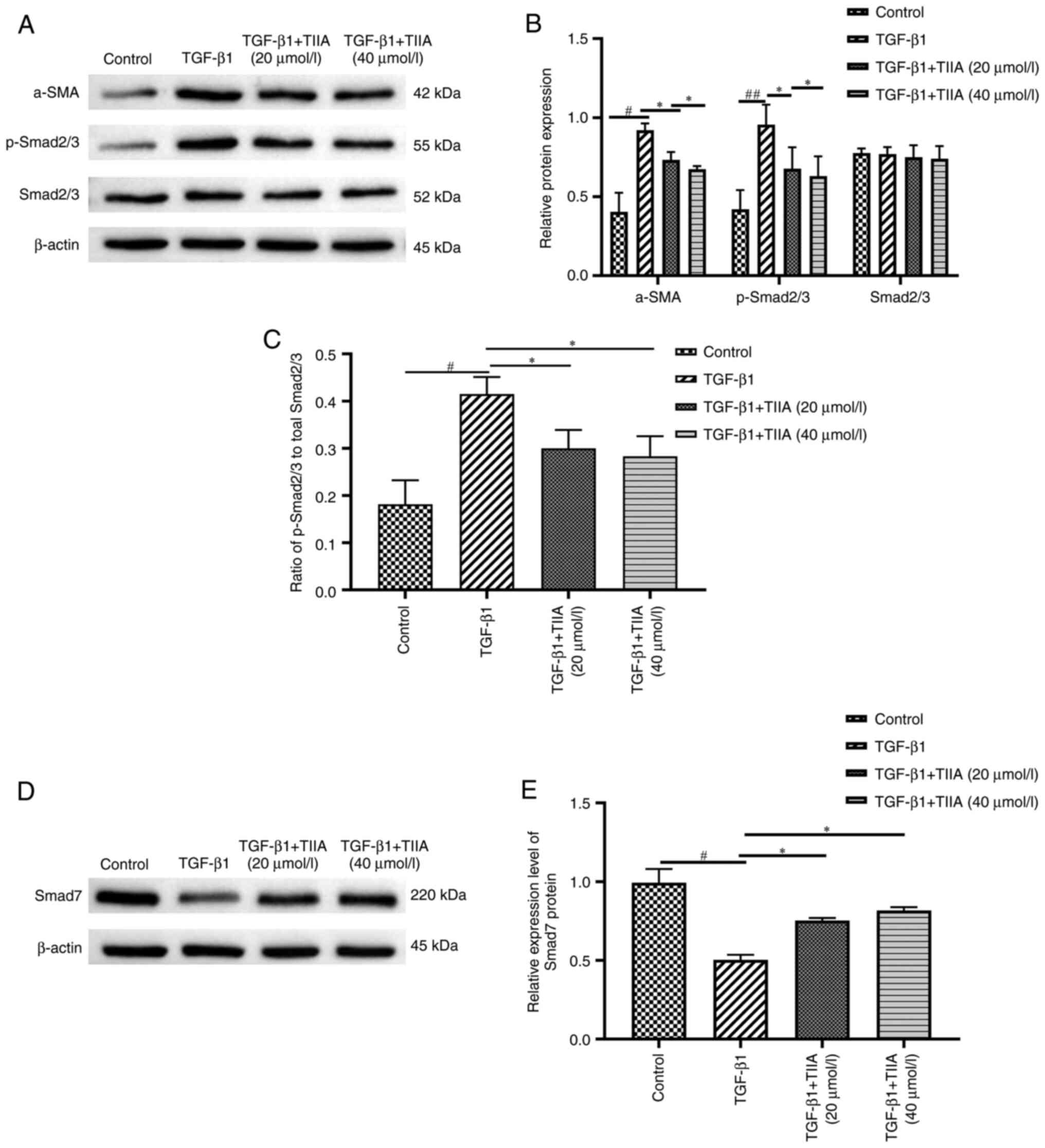
|
1
|
Eslam M, Newsome PN, Sarin SK, Anstee QM,
Targher G, Romero-Gomez M, Zelber-Sagi S, Wai-Sun Wong V, Dufour
JF, Schattenberg JM, et al: A new definition for metabolic
dysfunction-associated fatty liver disease: An international expert
consensus statement. J Hepatol. 73:202–209. 2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Arab JP, Arrese M and Trauner M: Recent
insights into the pathogenesis of nonalcoholic fatty liver disease.
Annu Rev Pathol. 13:321–350. 2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Powell EE, Wong VW and Rinella M:
Non-alcoholic fatty liver disease. Lancet. 397:2212–2224.
2021.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Thibaut R, Gage MC, Pineda-Torra I,
Chabrier G, Venteclef N and Alzaid F: Liver macrophages and
inflammation in physiology and physiopathology of non-alcoholic
fatty liver disease. FEBS J: Apr 15, 2021 (Epub ahead of
print).
|
|
5
|
Zhang CY, Yuan WG, He P, Lei JH and Wang
CX: Liver fibrosis and hepatic stellate cells: Etiology,
pathological hallmarks and therapeutic targets. World J
Gastroenterol. 22:10512–10522. 2016.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Zhang Y, Miao H, Yan H, Sheng Y and Ji L:
Hepatoprotective effect of forsythiae fructus water extract against
carbon tetrachloride-induced liver fibrosis in mice. J
Ethnopharmacol. 218:27–34. 2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Liu J, Kong D, Qiu J, Xie Y, Lu Z, Zhou C,
Liu X, Zhang R and Wang Y: Praziquantel ameliorates
CCl4-induced liver fibrosis in mice by inhibiting
TGF-β/Smad signalling via up-regulating Smad7 in hepatic stellate
cells. Br J Pharmacol. 176:4666–4680. 2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Walton KL, Johnson KE and Harrison CA:
Targeting TGF-β mediated SMAD signaling for the prevention of
fibrosis. Front Pharmacol. 8(461)2017.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Karin D, Koyama Y, Brenner D and Kisseleva
T: The characteristics of activated portal
fibroblasts/myofibroblasts in liver fibrosis. Differentiation.
92:84–92. 2016.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Massagué J: TGF-β signalling in context.
Nat Rev Mol Cell Biol. 13:616–130. 2012.PubMed/NCBI View
Article : Google Scholar
|
|
11
|
Fabregat I, Moreno-Càceres J, Sánchez A,
Dooley S, Dewidar B, Giannelli G and Ten Dijke P: IT-LIVER
Consortium. TGF-β signalling and liver disease. FEBS J.
283:2219–2232. 2016.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Giannelli G, Mikulits W, Dooley S,
Fabregat I, Moustakas A, Ten Dijke P, Portincasa P, Winter P,
Janssen R, Leporatti S, et al: The rationale for targeting TGF-β in
chronic liver diseases. Eur J Clin Invest. 46:349–361.
2016.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Yan C, Wang L, Li B, Zhang BB, Zhang B,
Wang YH, Li XY, Chen JX, Tang RX and Zheng KY: The expression
dynamics of transforming growth factor-β/Smad signaling in the
liver fibrosis experimentally caused by clonorchis sinensis.
Parasit Vectors. 8(70)2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Li B, Yan C, Wu J, Stephane K, Dong X,
Zhang YZ, Zhang Y, Yu Q and Zheng KY: Clonorchis sinensis ESPs
enhance the activation of hepatic stellate cells by a cross-talk of
TLR4 and TGF-β/Smads signaling pathway. Acta Trop.
205(105307)2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
El Nashar EM, Alghamdi MA, Alasmari WA,
Hussein MMA, Hamza E, Taha RI, Ahmed MM, Al-Khater KM and
Abdelfattah-Hassan A: Autophagy promotes the survival of adipose
mesenchymal stem/stromal cells and enhances their therapeutic
effects in cisplatin-induced liver injury via modulating
TGF-β1/Smad and PI3K/AKT signaling pathways. Cells.
10(2475)2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Dooley S, Hamzavi J, Breitkopf K,
Wiercinska E, Said H, Lorenzen J, Ten Dijke P and Gressner AM:
Smad7 prevents activation of hepatic stellate cells and liver
fibrosis in rats. Gastroenterology. 125:178–191. 2003.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Choi JH, Jin SW, Choi CY, Kim HG, Lee GH,
Kim YA, Chung YC and Jeong HG: Capsaicin inhibits
dimethylnitrosamine-induced hepatic fibrosis by inhibiting the
TGF-β1/Smad pathway via peroxisome proliferator-activated receptor
gamma activation. J Agric Food Chem. 65:317–326. 2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Bai G, Yan G, Wang G, Wan P and Zhang R:
Anti-hepatic fibrosis effects of a novel turtle shell decoction by
inhibiting hepatic stellate cell proliferation and blocking
TGF-β1/Smad signaling pathway in rats. Oncol Rep. 36:2902–2910.
2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Peng Y, Yang T, Huang K, Shen L, Tao Y and
Liu C: Salvia miltiorrhiza ameliorates liver fibrosis by
activating hepatic natural killer cells in vivo and in vitro. Front
Pharmaco. 9(762)2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Yan Q, Mao Z, Hong J, Gao K, Niimi M,
Mitsui T and Yao J: Tanshinone IIA stimulates cystathionine γ-lyase
expression and protects endothelial cells from oxidative injury.
Antioxidants (Basel). 10(1007)2021.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Chen Z, Gao X, Jiao Y, Qiu Y, Wang A, Yu
M, Che F, Li S, Liu J, Li J, et al: Tanshinone IIA exerts
anti-inflammatory and immune-regulating effects on vulnerable
atherosclerotic plaque partially via the TLR4/MyD88/NF-κB signal
pathway. Front Pharmacol. 10(850)2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Subedi L and Gaire BP: Tanshinone IIA: A
phytochemical as a promising drug candidate for neurodegenerative
diseases. Pharmacol Res. 169(105661)2021.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Bi Z, Wang Y and Zhang W: A comprehensive
review of tanshinone IIA and its derivatives in fibrosis treatment.
Biomed Pharmacother. 137(111404)2021.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Shi MJ, Yan XL, Dong BS, Yang WN, Su SB
and Zhang H: A network pharmacology approach to investigating the
mechanism of Tanshinone IIA for the treatment of liver fibrosis. J
Ethnopharmacol. 253(112689)2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Sun X, Tan Y, Lyu J, Liu HL, Zhao ZM and
Liu CH: Active components formulation developed from fuzheng huayu
recipe for anti-liver fibrosis. Chin J Integr Med: Sep 28, 2021
(Epub ahead of print).
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Chalasani N, Younossi Z, Lavine JE,
Charlton M, Cusi K, Rinella M, Harrison SA, Brunt EM and Sanyal AJ:
The diagnosis and management of nonalcoholic fatty liver disease:
Practice guidance from the American association for the study of
liver diseases. Hepatology. 67:328–357. 2018.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Wong VW, Wong GL, Choi PC, Chan AW, Li MK,
Chan HY, Chim AM, Yu J, Sung JJ and Chan HL: Disease progression of
non-alcoholic fatty liver disease: A prospective study with paired
liver biopsies at 3 years. Gut. 59:969–974. 2010.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Taylor RS, Taylor RJ, Bayliss S, Hagström
H, Nasr P, Schattenberg JM, Ishigami M, Toyoda H, Wai-Sun Wong V,
Peleg N, et al: Association between fibrosis stage and outcomes of
patients with nonalcoholic fatty liver disease: A systematic review
and meta-analysis. Gastroenterology. 158:1611–1625.e12.
2020.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Li R, Li J, Huang Y, Li J, Yan S, Lin J,
Chen Y, Wu L, Liu B, Wang G and Lan T: Polydatin attenuates
diet-induced nonalcoholic steatohepatitis and fibrosis in mice. Int
J Biol Sci. 14:1411–1425. 2018.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Li X, Wang TX, Huang X, Li Y, Sun T, Zang
S, Guan KL, Xiong Y, Liu J and Yuan HX: Targeting ferroptosis
alleviates methionine-choline deficient (MCD)-diet induced NASH by
suppressing liver lipotoxicity. Liver Int. 40:1378–1394.
2020.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Zisser A, Ipsen DH and Tveden-Nyborg P:
Hepatic stellate cell activation and inactivation in
NASH-fibrosis-roles as putative treatment targets? Biomedicines.
9(365)2021.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Czaja AJ: Hepatic inflammation and
progressive liver fibrosis in chronic liver disease. World J
Gastroenterol. 20:2515–2532. 2014.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Eom YW, Shim K and Baik SK: Mesenchymal
stem cell therapy for liver fibrosis. Korean J Intern Med.
30:580–589. 2015.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Seki E and Brenner DA: Recent advancement
of molecular mechanisms of liver fibrosis. J Hepatobiliary Pancreat
Sci. 22:512–518. 2015.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Xu S, Mao Y, Wu J, Feng J, Li J, Wu L, Yu
Q, Zhou Y, Zhang J and Chen J: TGF-β/Smad and JAK/STAT pathways are
involved in the anti-fibrotic effects of propylene glycol alginate
sodium sulphate on hepatic fibrosis. J Cell Mol Med. 24:5224–5237.
2020.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Friedman SL: Hepatic stellate cells:
Protean, multifunctional, and enigmatic cells of the liver. Physiol
Rev. 88:125–172. 2008.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Li Z, Wang Z, Dong F, Shi W, Dai W, Zhao
J, Li Q, Fang ZE, Ren L, Liu T, et al: Germacrone attenuates
hepatic stellate cells activation and liver fibrosis via regulating
multiple signaling pathways. Front Pharmacol.
12(745561)2021.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Yu K, Li Q, Shi G and Li N: Involvement of
epithelial-mesenchymal transition in liver fibrosis. Saudi J
Gastroenterol. 24:5–11. 2018.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Zhou QY, Yang HM, Liu JX, Xu N, Li J, Shen
LP, Zhang YZ, Koda S, Zhang BB, Yu Q, et al: MicroRNA-497 induced
by clonorchis sinensis enhances the TGF-β/Smad signaling pathway to
promote hepatic fibrosis by targeting Smad7. Parasit Vectors.
14(472)2021.PubMed/NCBI View Article : Google Scholar
|