Introduction
It is common to obtain metaphase II (MII), MI or
germinal vesicle (GV) oocytes in in vitro fertilization
(IVF). Cases of spontaneous oocyte activation (parthenogenesis)
in vivo have previously been reported (1-4).
However, the mechanisms of the oocyte activation in natural or
stimulated cycles have remained to be clarified. In the present
study, a case of recurrent parthenogenesis was reported and it was
attempted to find common characteristics through reviewing the
literature.
Case report
Patient history
A 33-year-old female patient and her 32-year-old
husband presented at The Center for Reproductive Medicine and
Infertility, The Fourth Hospital of Shijiazhuang, Hebei Medical
University in March 2021 due to infertility for a year. The patient
had a regular menstrual cycle and remarried a year ago. The patient
had delivered two full-term babies with her ex-husband in 2012 and
2014. In 2016, a laparoscopic bilateral salpingectomy had been
performed for an ectopic pregnancy. Chromosomal analysis indicated
a normal female chromosome complement (46,XX). The patient's
husband's semen parameters were in the normal range and the
chromosome karyotype was normal (46,XY).
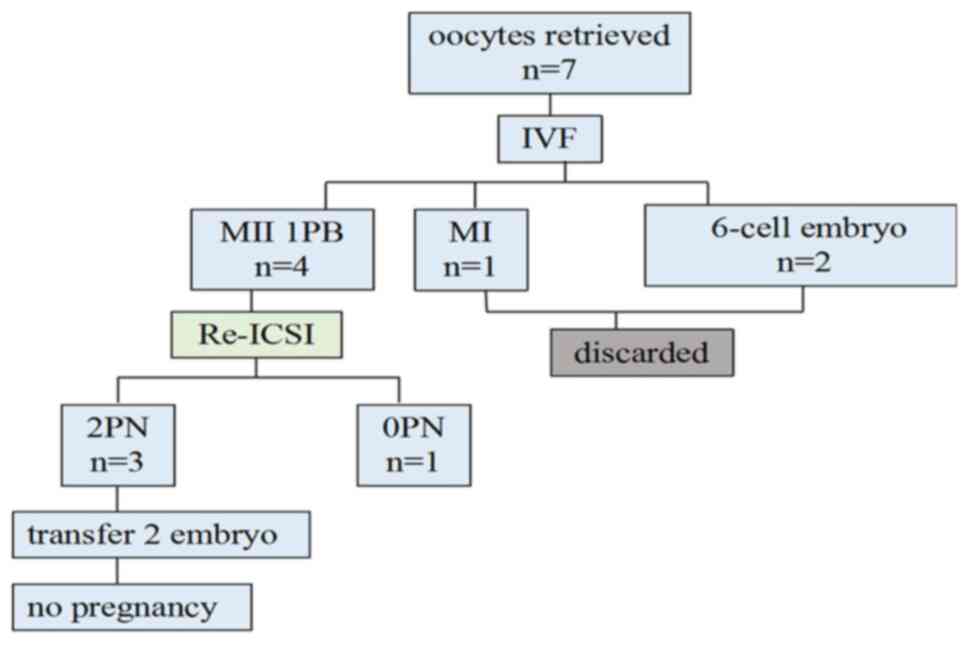
Cycle 1 (IVF+rescue ICSI)
In June 2021, a long program of fertility treatment
due to infertility following salpingectomy was adopted using
triptorelin, in association with follicle-stimulating hormone (FSH)
and human menopausal gonadotropin (5). On the day of the trigger, the patient
produced four follicles of 18 mm, two of 17 mm and one of 16 mm
under ultrasound. Ovum pick up was scheduled after 36 h. This time,
seven oocytes were obtained and short-time fertilization was
performed. At four hours after fertilization, cumulus cells were
removed. A total of four MII oocytes with only one polar body, one
MI oocyte and two 6-cell embryos were retrieved. Consequently,
rescue ICSI was performed and three out of four injected oocytes
were fertilized normally, while one remained unfertilized. Embryo
transfer was performed 72 h after oocyte retrieval, resulting in no
pregnancy (Fig. 1).
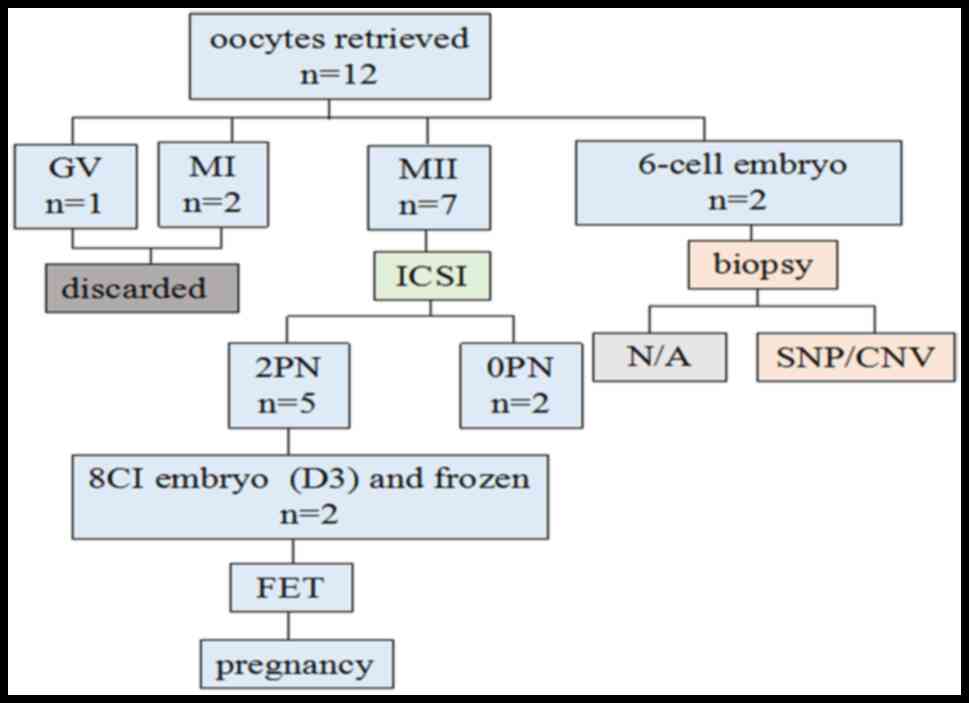
Cycle 2 (ICSI)
The patient underwent a second cycle after an
interval of one month. An antagonist protocol commenced after the
baseline scan and then hormonal treatment on day two (6). The patient was stimulated with
urinary (u)FSH 225 IU. The antagonist was given from day seven. The
trigger was administered on day 17 of stimulation with recombinant
human chorionic gonadotropin, 0.25 µg subcutaneously. The patient
had eight follicles of 18 mm and four follicles of 15-17 mm in size
on the day of the trigger under ultrasound. Egg retrieval was
performed 36 h after the trigger. A total of twelve oocytes were
recovered by ultrasound-guided transvaginal aspiration. After
removing the cumulus cells, seven MII, two MI and one GV oocyte, as
well as two 6-cell embryos were retrieved. A total of seven MII
oocytes were injected and five 2PN were observed (D1); two
good-quality 8CI (cell) embryos were formed and frozen (D3).
Subsequently, two frozen embryos were thawed and transferred to the
patient, resulting in a normal single pregnancy (Fig. 2).
Genetic analysis
Due to IVF, the two 6-cell embryos in the first
cycle were not further analyzed. The two 6-cell embryos in the ICSI
cycle were observed for three days and exhibited no changes from
their previous state. The embryos had a normal morphology and
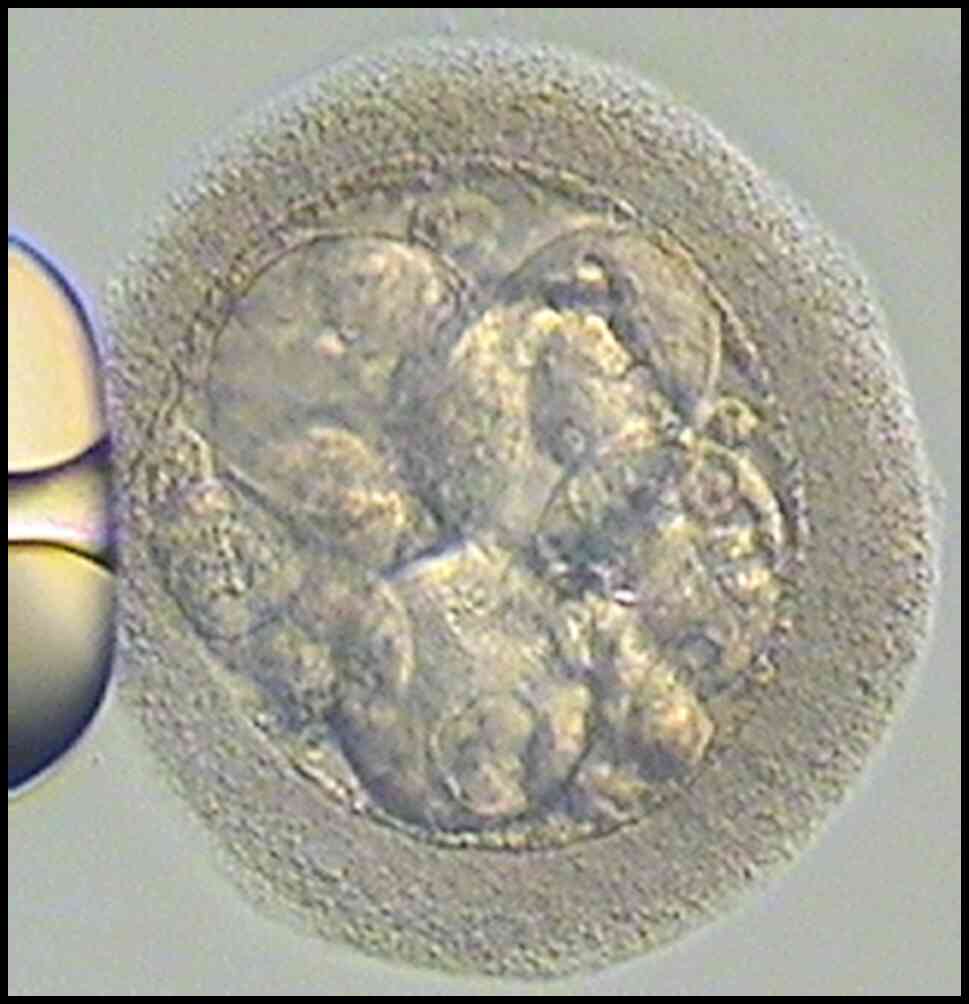
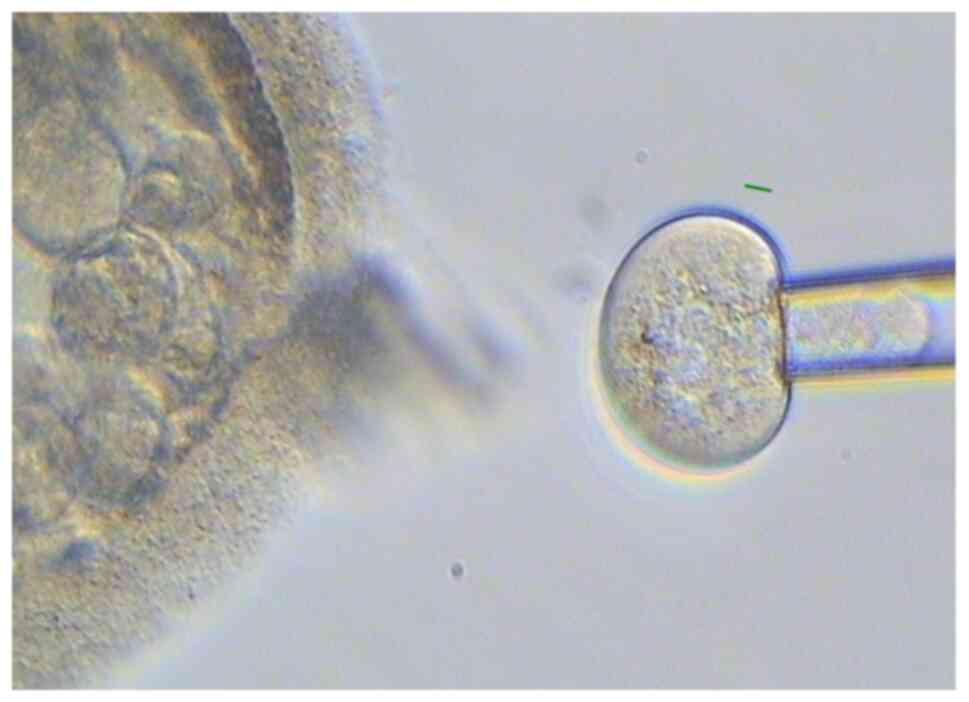
certain blastomeres had distinct nuclei. A blastomere biopsy was
then performed on them using a laser (Hamilton Thorne) (Figs. 3 and 4). The blastomere was aspirated from the
embryo and released into the medium for amplification (YK001B;
Yikon). The amplification results were detected by copy number
variation (CNV) analysis and next-generation sequencing (NGS)
(Illumina, Inc.) (7). The NGS data
that were used for SNP chip (Illumina, Inc.) analysis indicated
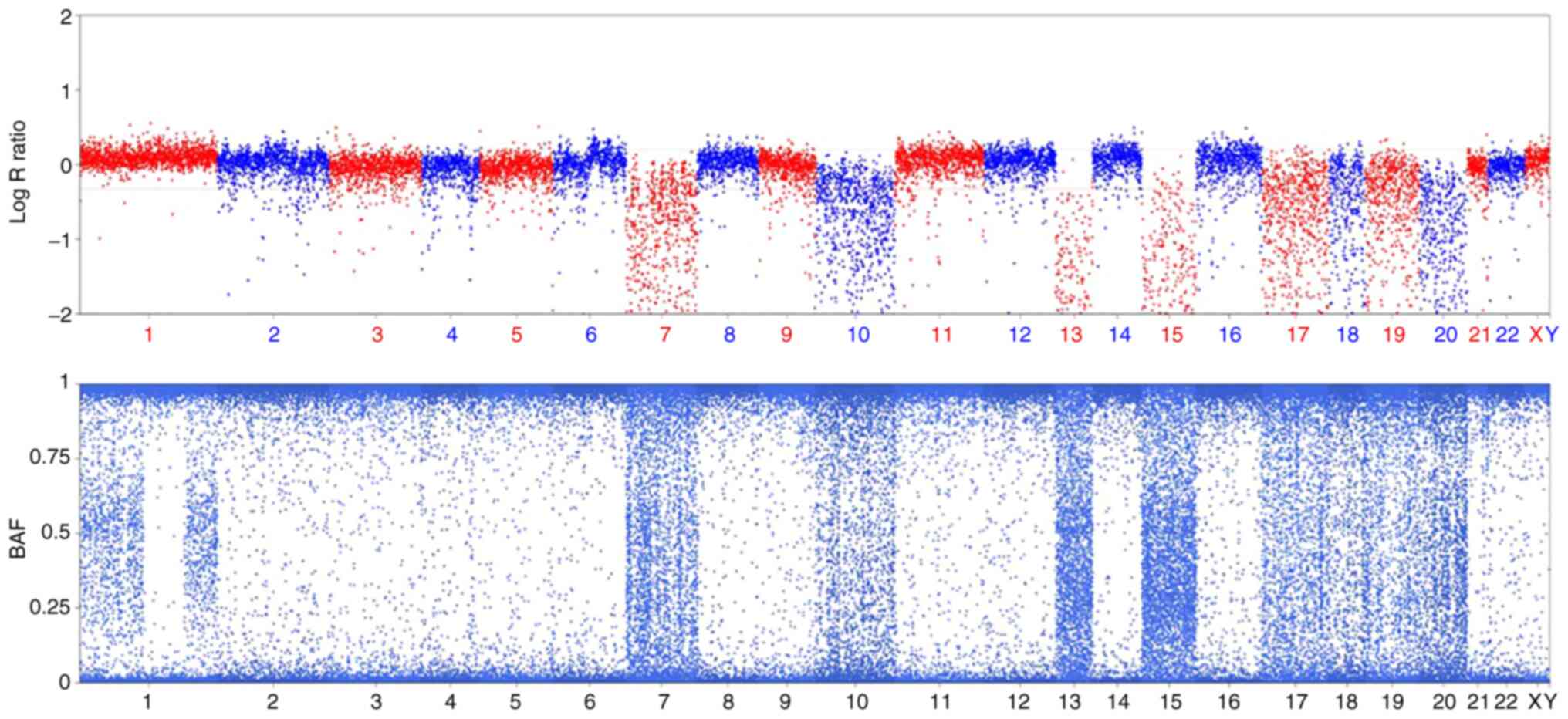
that one 6-cell embryo was not able to be obtained using the
amplification concentrate (Fig.
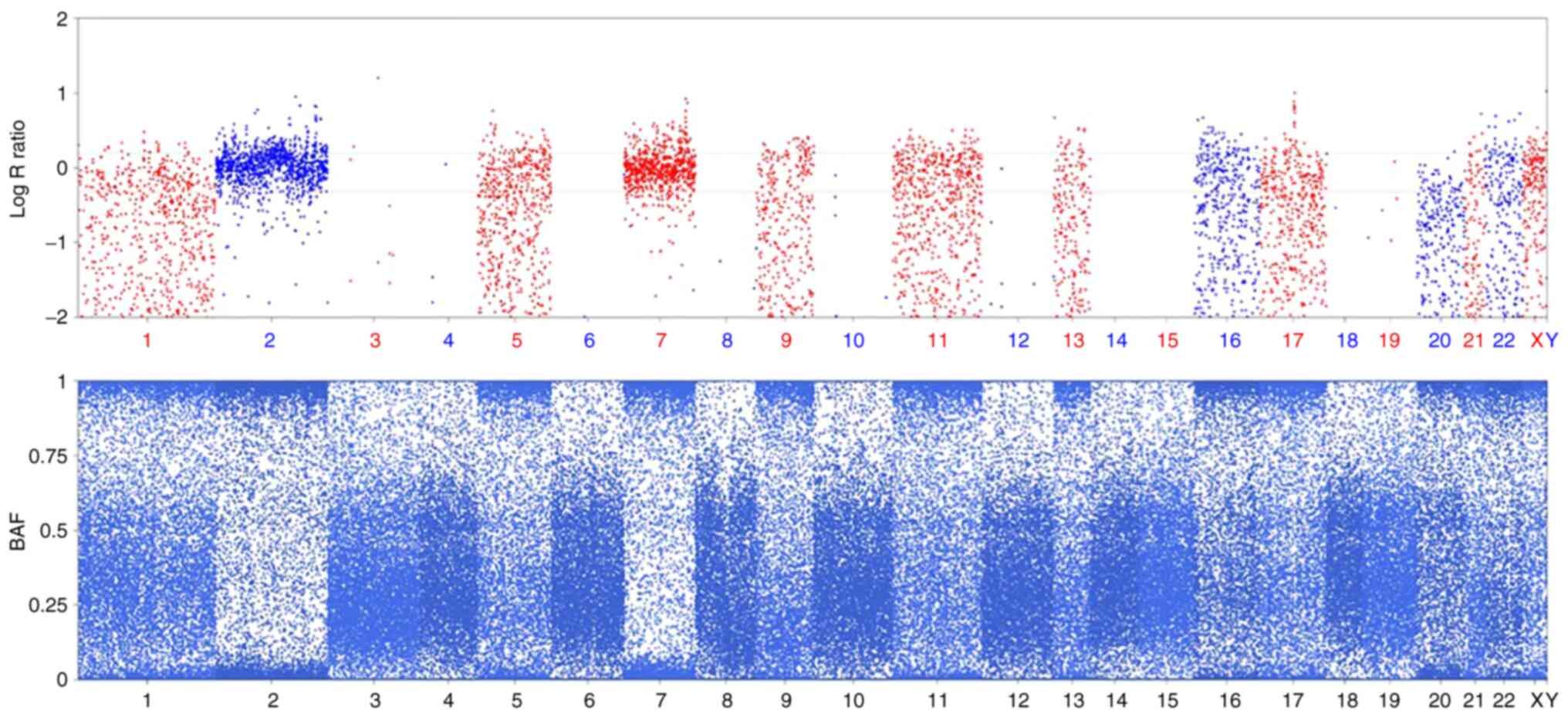
5). The other 6-cell embryo SNPs reported those chromosomes
with one copy had a signal pattern similar to that obtained for
uniparental disomy (UPD) based on the B-allele frequency value
combined with the LogR Ratio, which may be ascertained by the
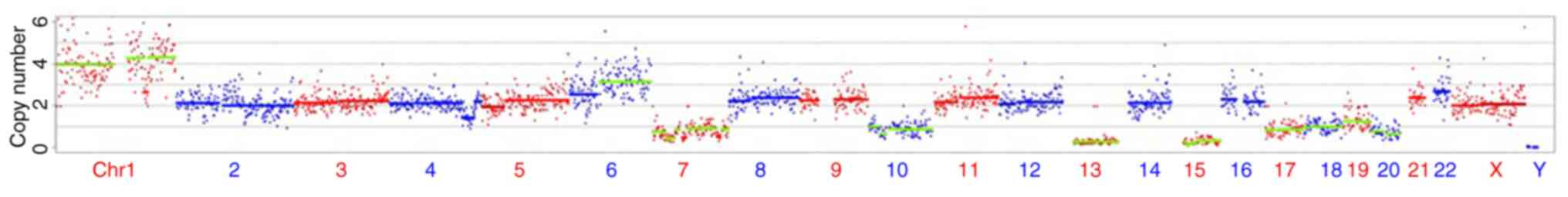
absence of heterozygous sites on certain chromosomes (Fig. 6). The CNV results indicated a
genotype of
39,XX,+1,+1,+1,+1,+6q,+6q,+6q,-7p(x1),-10(x1),-13(x0),-15(x0),-17(x1),-18(x1),-19(x1),-20(x1)
(Fig. 7). The participant provided
written informed consent and the ethics committee of The Fourth
Hospital of Shijiazhuang (Shijiazhuang, China) approved the present
study.
Literature review (search terms:
Parthenogenesis in human case IVF in PubMed)
Parthenogenesis and spontaneous oocyte activation in
unfertilized oocytes has been reported previously in humans and
cases are presented in Table I
(1-4).
 | Table IReview of cases of parthenogenesis
reported in the previous literature and the present case
report. |
Table I
Review of cases of parthenogenesis
reported in the previous literature and the present case
report.
| Author (year) | Country | Patient age,
years | Cycle | History of
infertility | Protocol | 1PN/embryo | Test | Result | (Refs.) |
|---|
| Ye (2019) | China | 38 | 1st | Bilateral tubal
obstruction | GnRH-antagonist | One 1PN 30 h 8-cell
embryos | CNV/SNP | No pregnancy | 1 |
| Combelles (2010) | USA | 32 | 1st/2nd | Recurrent
miscarriages | LDLL | i) IVF 1PN/embryo
ii)14 1PN | i) None ii)
FISH/PCR/SNP | No pregnancy | 2 |
| Oliveira (2004) | Brazil | 29 | 1st | Teratoma left
oophorectomy | Long protocol | One 4-cell develop
7-cell | FISH | Pregnancy | 3 |
| Socolov (2015) | African
ethnicity | 38 | 1st | Salpingectomy | Antagonist | An extruded oocyte
with one nucleus | None | No pregnancy | 4 |
| (2021) | China | 33 | 1st/2nd | Ectopic pregnancy;
salpingectomy | i) Long protocol ii)
Antagonist | i) Two 6-cell embryos
ii) Two 6-cell embryos | i) None ii)
CNV/SNP | i) No pregnancy ii)
Pregnancy | Present study |
For a 38-year-old female patient with a history of
bilateral tubal obstruction, one pronucleus was retrieved prior to
ICSI and it developed into an embryo 30 h post-retrieval. The
genome electrophoresis assay of the individual blastomere indicated
that only one of eight blastomeres contained the genome. The CNV
results indicated a genotype of 48, XX,+17,+17. The NGS data were
used for MultiSNPs analysis and the results indicated that the SNPs
of the biopsied embryo were exclusively consistent with those of
the maternal side (1) (Table I).
Combelles et al (2) reported a case with recurrent
miscarriages and prolonged infertility in which spontaneous
activation of oocytes with parthenogenetic evolution was the likely
cause. Following the unexpected presence of cleaved embryos at the
fertilization check at the first IVF attempt, oocytes and embryos
were subsequently analyzed in an ICSI/preimplantation genetic
diagnosis (PGD) case. Fluorescence in situ hybridization
(FISH) revealed aneuploidy in all seven blastomeres analyzed, with
all but two lacking Y chromosomes. Microarray SNP analysis
indicated an exclusively maternal origin of all blastomeres
analyzed, which was further confirmed by PCR (Table I).
A 29-year-old female had undergone left oophorectomy
due to an ovarian teratoma. The patient had 13 MII oocytes and one
4-cell embryo retrieved prior to injection in an ICSI cycle. The
4-cell (parthenote) embryo developed into a 7-cell embryo by the
next day. FISH returned two signals for the X chromosome in each
blastomere that was analyzed. Other oocytes were fertilized
normally and three were transferred, resulting in a normal
singleton pregnancy and the birth of a healthy baby (Table I) (3).
In another study, the patient was a G1P0 38-year-old
female of African ethnicity who had experienced a previous ectopic
pregnancy, which had been treated by salpingectomy in search of an
IVF with donor sperm. One oocyte was retrieved, donated sperms were
used for IVF and an extruded oocyte with one nucleus separate from
the granulosa cell wall was described. At 40 h, the aspect was of
parthenogenic oocytes in a three-cell cluster, one cell with one
nucleus, the others with high granulation and no visible
pro-nucleus (4) (Table I).
Through the literature review, it was discovered
that parthenogenesis and spontaneous oocyte activation may occur in
humans of different ethnicities/regions, such as Chinese, American,
Brazilian and African ethnicity patients. All of these patients had
a history of either recurrent miscarriages or bilateral tubal
obstruction with or without ovarian/fallopian tube surgery. In
certain cases, embryos appear as 1PN and cleave later and in
others, they appear as four-to six-cell embryos directly.
Discussion
In the studies retrieved in the literature review,
oocyte activation, parthenogenesis and UPD were mentioned. This
raises the question of whether there are any differences and
correlations between oocyte activation, parthenogenesis and
UPD.
Oocyte activation is a series of events that
converts an MII-arrested oocyte into a fertilized egg ready to
begin embryogenesis. In most mammals, oocyte activation is a
spatial-temporal regulated process triggered by sperm entry
(8).
By activating mouse eggs with experimentally
controlled and precisely defined calcium transients, Ducibella
et al (9) demonstrated that
each of the early events of mammalian oocyte activation is
initiated by a different number of calcium transients, while their
completion requires a greater number of calcium transients than for
their initiation (8). A variety of
artificial activating methods is used in human assisted
reproduction treatment, including physical, mechanical or chemical
stimuli, which provoke one or more calcium rises in the oocyte
cytoplasm (8). However,
spontaneous oocyte activation may occur under experimental
conditions, providing genetic material for PGD diagnostics, as
reported by Paffoni et al (10).
Parthenogenesis, a unique form of reproduction, is
normally inhibited in mammals and a human embryo with
parthenogenetic origin is not considered capable of producing
offspring (1). It was reported
that parthenogenetic activation took place around the time of
fertilization of a sperm missing a sex chromosome, resulting in the
generation of the upid(AC)mat 46,XX cell lineage by endoreplication
of one blastomere containing a female pronucleus and the 45,X cell
lineage by union of male and female pronuclei (9,11).
Artificial activation may also be used to obtain
parthenogenetic mammalian embryos that may serve as a model to
study biochemical and morphological events. Fresh and aged human
oocytes may be activated parthenogenetically using a calcium
ionophore. Ethanol was proven to be a poor activating agent. Human
parthenotes may complete division to the eight cell stage (11,12).
Parthenogenesis may also be induced by exposure of unfertilized
oocytes to strontium-containing medium (13). Certain early human pregnancy losses
may involve oocytes that have been parthenogenetically activated
spontaneously (2,14).
UPD refers to the situation in which both homologues
of a chromosomal region/segment have originated from only one
parent. UPD and mosaic aneuploidy arise from mitotic or meiotic
events (15). As a consequence of
UPD, or deficiency of part of a chromosome, there are two types of
developmental risk: Aberrant dosage of genes regulated by genomic
imprinting and homozygosity of a recessive mutation (16). This may involve the entire
chromosome or only a small segment. Uniparental disomy in the human
blastocyst is exceedingly rare (17).
UPD almost always arises in connection with a
numerical or structural chromosomal aberration. UPD cases in which
the causative cytogenetic event is still present, even if only in
the mosaic state, provide deep insight into the abilities of
gametes or embryonic cells to repair chromosomal imbalances and/or
rearrangements (18).
In conclusion, there are certain correlations
between oocyte activation, parthenogenesis and UPD, but the
following are the differences, which are well established: The
incidence of UPD of any chromosome is estimated to account for
~1:3,500 live births. For certain chromosomes, UPD does not exert
any adverse effect on any individual. However, for other
chromosomes, it may result in abnormalities due to aberrant genomic
imprinting (16). In humans,
parthenogenesis always leads to teratoma and never to a viable
individual (1).
In the present study, the first cycle used was IVF
short-time fertilization. After the removal of the cumulus cells,
two early cleavage embryos were discovered. ICSI was performed in
the second cycle and prior to injection, two 6-cell embryos were
discovered and tested. Although recurrent spontaneous
parthenogenesis was observed, this did not influence the other MII
oocyte fertilization and the achievement of pregnancy. It was
hypothesized that the bilateral salpingectomy resulted in recurrent
spontaneous oocyte activation. This is a similar case to that
reported by Combelles et al (2), where the patient had two cycles of
spontaneous oocyte activation and underwent left oophorectomy due
to an ovarian teratoma.
In humans, parthenogenesis never results in a viable
individual. In the present study, the parthenogenetic embryos were
observed for three days and exhibited no changes. Although there
were two parthenogenetic embryos, this did not influence the other
oocyte fertilization and the resulting pregnancy. In the present
case, the healthy baby that was born originated from the other
normal oocyte.
The present results indicated that the first 6-cell
embryo was not able to be observed in the amplification
concentrate. The reason was probably that not every blastomere
contains genomes. As with the previously reported case, only one of
eight blastomeres contained genomes, indicating that the oocyte
underwent cleavage without genome replication, or another
probability was that what was assumed to be the 7 blastomeres were
actually embryo fragments rather than blastomeres (1).
In the present study, the SNP and CNV results for
the second 6-cell embryo revealed a genotype of
39,XX,+1,+1,+1,+1,+6q,+6q,+6q,-7p(x1),-10(x1),-13(x0),-15(x0),-17(x1),-18(x1),-19(x1),-20(x1).
Only those chromosomes with the presence of one copy exhibited a
signal pattern similar to that of a UPD and other chromosomes
rather appeared to have a trisomy or tetrasomy copy. The reason may
be due to amplification concentration, or other unknown aspects.
The mechanisms of the formation of the UPD included trisomy rescue,
with and without concomitant trisomy, monosomy rescue and mitotic
formation of a mosaic segmental UPD (15). UPD was also identified with one
cell line having complete maternal UPD consistent with a
parthenogenetic origin (14).
Chromosomal segregation was analyzed, revealing a highly probable
UPD as the reason for parent/child allele mismatches (19). This indicates that massively
imbalanced embryos were identified, as new single-cell genomic
methodologies have further pinpointed the existence of mixoploidy
in cleavage-stage embryos (20).
Although repeated spontaneous parthenogenesis
appeared in the patient of the present study, other oocytes were
able to be fertilized normally and the patient became pregnant. The
clinical value of the case report was that it demonstrated that
pregnancy outcomes were not influenced by the fact that
parthenogenesis was observed in part of the oocytes. As with the
case reported by Oliveira et al (3), although there was a parthenogenetic
origin of the ovarian teratoma, there were eight normally
fertilized embryos, three of which were transferred, resulting in a
normal single pregnancy and the birth of a healthy baby.
In conclusion, in the present study, a massively
imbalanced parthenogenesis embryo was detected, which may occur in
patients from various regions, who always have a history of either
recurrent miscarriages or bilateral tubal obstruction with or
without ovarian/fallopian tube surgery.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The sequencing data are available from a curated
repository (https://submit.ncbi.nlm.nih.gov/sra/metadata_file/SUB11494870/processed-ok;
Homo sapiens genome sequencing, SRR19261840, PRJNA838268).
All other data are available from the corresponding author upon
reasonable request.
Authors' contributions
YJ conceived the study and wrote the manuscript. GS,
JY, XZ and XW performed experiments and analyzed data. YC, ZH and
WH supervised the study. GS and XW confirm the authenticity of all
raw data. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
The Fourth Hospital of Shijiazhuang ethics committee
approved this study (approval no. 20210081; July 06, 2021). The
procedures used in this study adhere to the tenets of the
Declaration of Helsinki.
Patient consent for publication
The patient gave written informed consented to the
publication of data and images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ye Y, Li N, Yan X, Wu R, Zhou W, Cheng L
and Li Y: Genetic analysis of embryo in a human case of spontaneous
oocyte activation: A case report. Gynecol Endocrinol. 36:294–296.
2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Combelles CM, Kearns WG, Fox JH and
Racowsky C: Cellular and genetic analysis of oocytes and embryos in
a human case of spontaneous oocyte activation. Hum Reprod.
26:545–552. 2011.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Oliveira FG, Dozortsev D, Diamond MP,
Fracasso A, Abdelmassih S, Abdelmassih V, Gonçalves SP, Abdelmassih
R and Nagy ZP: Evidence of parthenogenetic origin of ovarian
teratoma: Case report. Hum Reprod. 19:1867–1870. 2004.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Socolov R, Ebner T, Gorduza V, Martiniuc
V, Angioni S and Socolov D: Self-oocyte activation and
parthenogenesis: An unusual outcome of a misconducted IVF cycle.
Gynecol Endocrinol. 31:529–530. 2015.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Jiang Y, Cao Q, Zhao X, Li L, Li S and Gao
F: Percutaneous epididymal sperm aspiration and short time
insemination in the treatment of men with obstructive azoospermia.
J Assist Reprod Genet. 30:1175–1179. 2013.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Jiang Y, Song G, Yuan J and Zhang X: ICSI
with all oocytes recurrent Metaphase I characterized by absence
perivitelline space. Open J Obstet Gynecol. 11:1112–1116. 2021.
|
|
7
|
McGowan-Jordan J, Ros JH and Sarah M
(eds): ISCN 2020: An international system for human cytogenomic
nomenclature. 1st edition. S. Karger AG, Basel, pp341-503,
2020.
|
|
8
|
Bonte D, Ferrer-Buitrago M, Dhaenens L,
Popovic M, Thys V, De Croo I, De Gheselle S, Steyaert N, Boel A,
Vanden Meerschaut F, et al: Assisted oocyte activation
significantly increases fertilization and pregnancy outcome in
patients with low and total failed fertilization after
intracytoplasmic sperm injection: A 17-year retrospective study.
Fertil Steril. 112:266–274. 2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Ducibella T, Huneau D, Angelichio E, Xu Z,
Schultz RM, Kopf GS, Fissore R, Madoux S and Ozil JP: Egg-to-embryo
transition is driven by differential responses to Ca(2+)
oscillation number. Dev Biol. 250:280–291. 2002.PubMed/NCBI
|
|
10
|
Paffoni A, Paracchini V, Ferrari S,
Scarduelli C, Seia M, Coviello DA and Ragni G: Use of
parthenogenetic activation of human oocytes as an experimental
model for evaluation of polar body based PGD assay performance. J
Assist Reprod Genet. 28:461–470. 2011.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Kaufman MH: Parthenogenetic activation of
Oocytes. Cold Spring Harb Protoc. 2018:20–23. 2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Yamazawa K, Nakabayashi K, Kagami M, Sato
T, Saitoh S, Horikawa R, Hizuka N and Ogata T: Parthenogenetic
chimaerism/mosaicism with a Silver-Russell syndrome-like phenotype.
J Med Genet. 47:782–785. 2010.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Kaneda M, Takahashi M, Yamanaka KI, Saito
K, Taniguchi M, Akagi S, Watanabe S and Nagai T: Epigenetic
analysis of bovine parthenogenetic embryonic fibroblasts. J Reprod
Dev. 63:365–375. 2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Winston N, Johnson M, Pickering S and
Braude P: Parthenogenetic activation and development of fresh and
aged human oocytes. Fertil Steril. 56:904–912. 1991.PubMed/NCBI
|
|
15
|
Conlin LK, Thiel BD, Bonnemann CG, Medne
L, Ernst LM, Zackai EH, Deardorff MA, Krantz ID, Hakonarson H and
Spinner NB: Mechanisms of mosaicism, chimerism and uniparental
disomy identified by single nucleotide polymorphism array analysis.
Hum Mol Genet. 19:1263–1275. 2010.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Yamazawa K, Ogata T and Ferguson-Smith AC:
Uniparental disomy and human disease: An overview. Am J Med Genet C
Semin Med Genet. 154C:329–334. 2010.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Gueye NA, Devkota B, Taylor D, Pfundt R,
Scott RT Jr and Treff NR: Uniparental disomy in the human
blastocyst is exceedingly rare. Fertil Steril. 101:232–236.
2014.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Liehr T: Formation of UPD. In: Uniparental
Disomy (UPD) in Clinical Genetics. Springer, Berlin, Heidelberg,
2014.
|
|
19
|
Nikitina TV and Nazarenko SA: Mutation in
microsatellite repeats of DNA and embryonal death in humans.
Genetika. 36:965–971. 2000.PubMed/NCBI
|
|
20
|
Masset H, Tšuiko O and Vermeesch JR:
Genome-wide abnormalities in embryos: Origins and clinical
consequences. Prenat Diagn. 41:554–563. 2021.PubMed/NCBI View
Article : Google Scholar
|