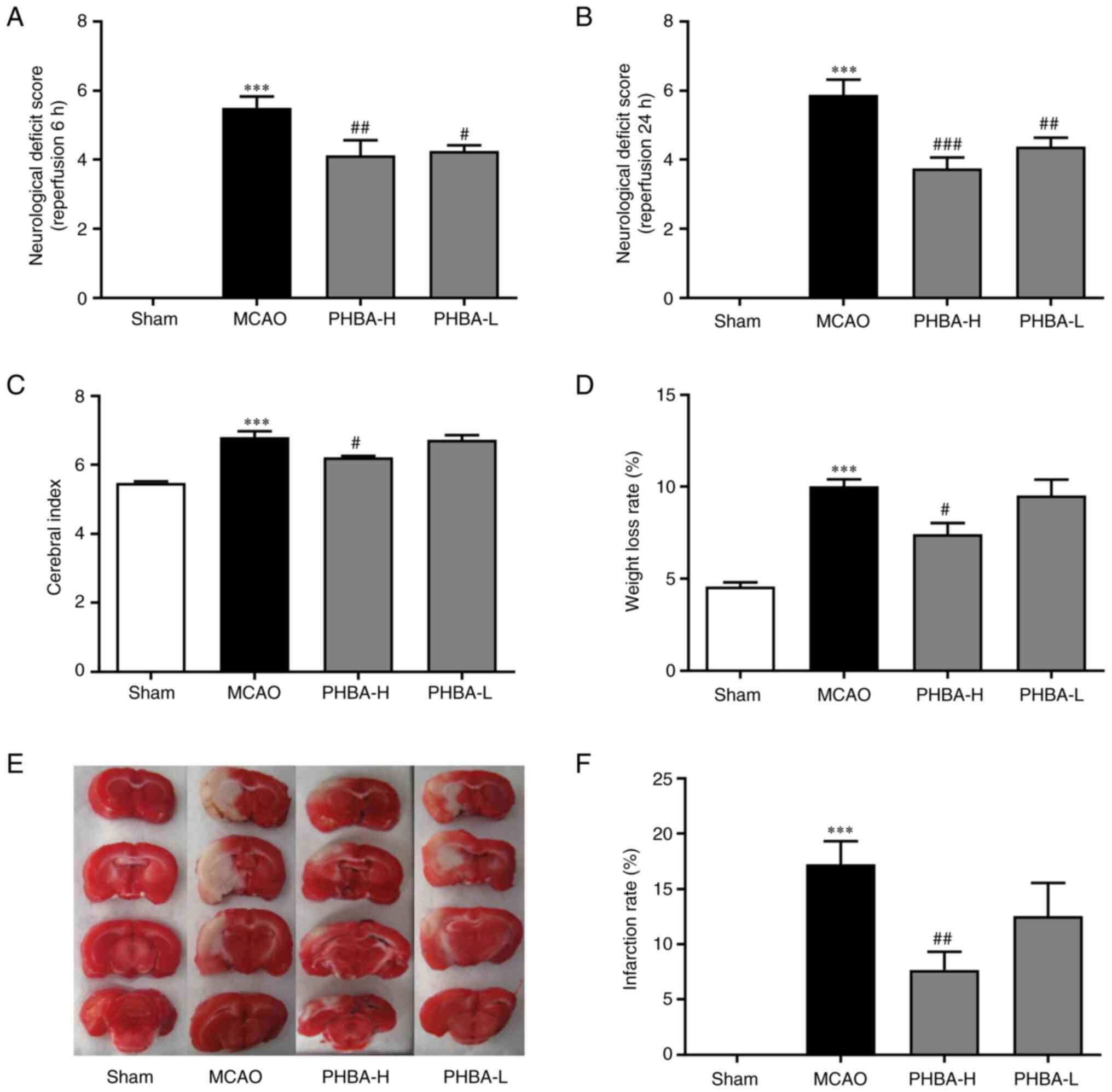
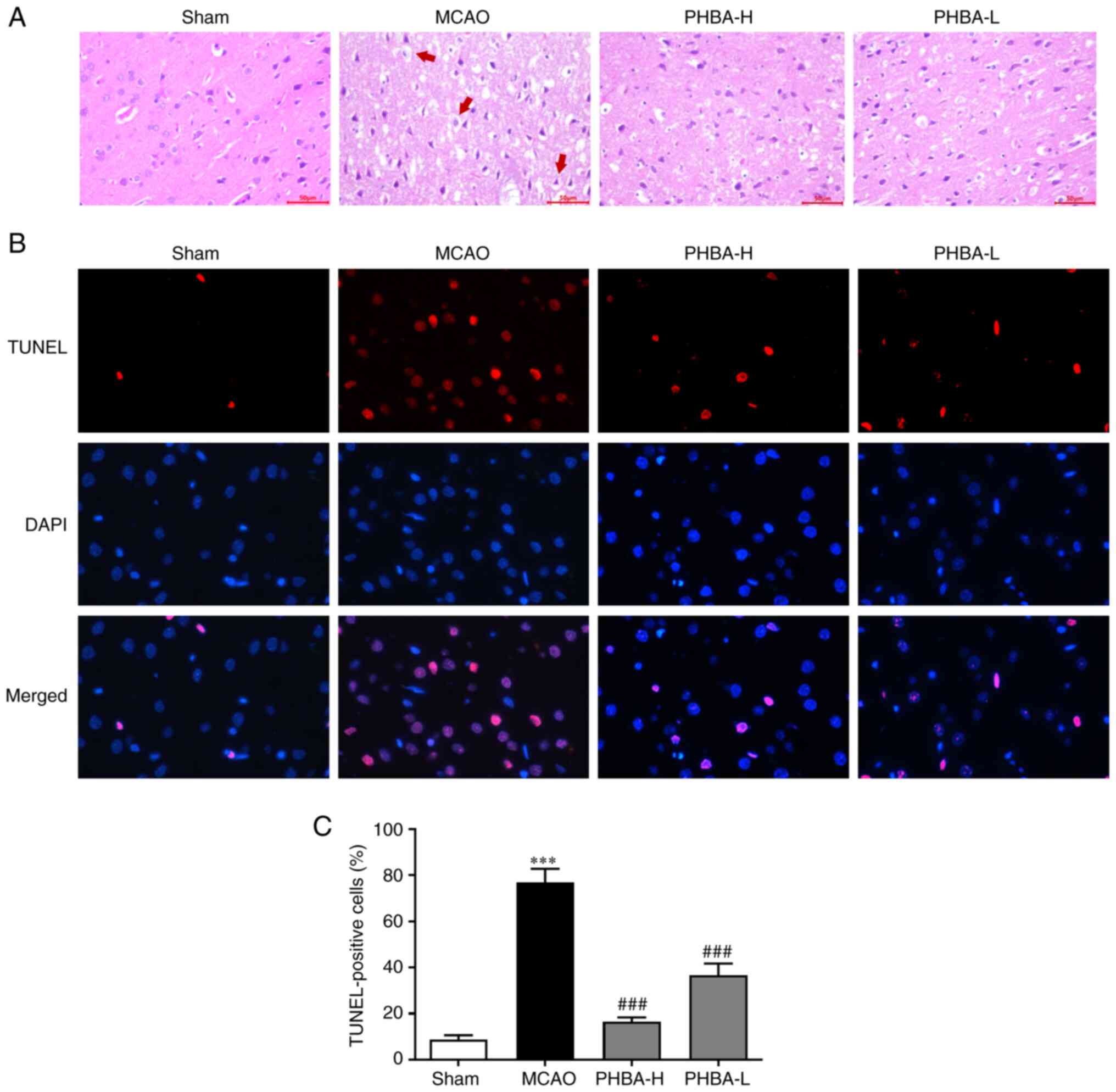
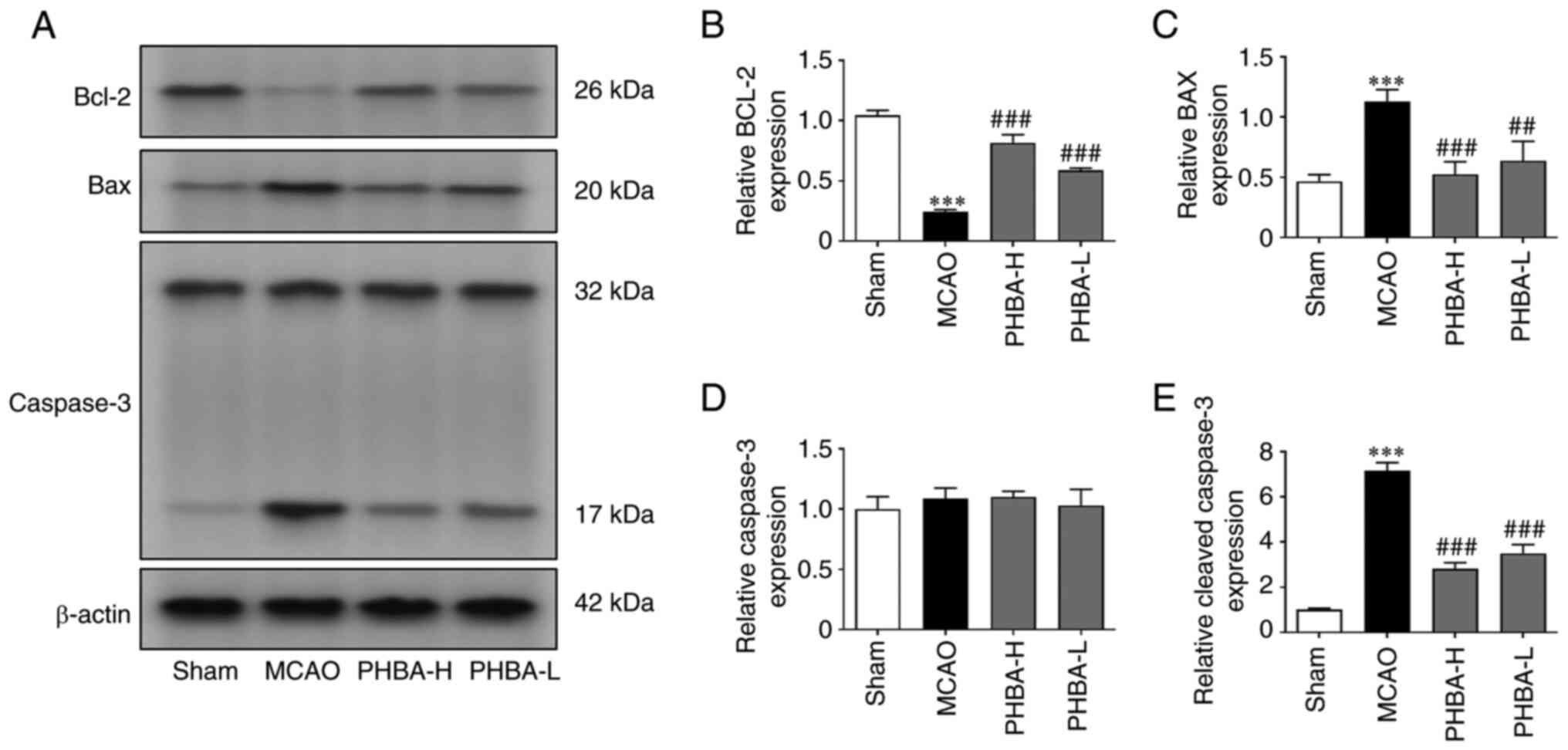
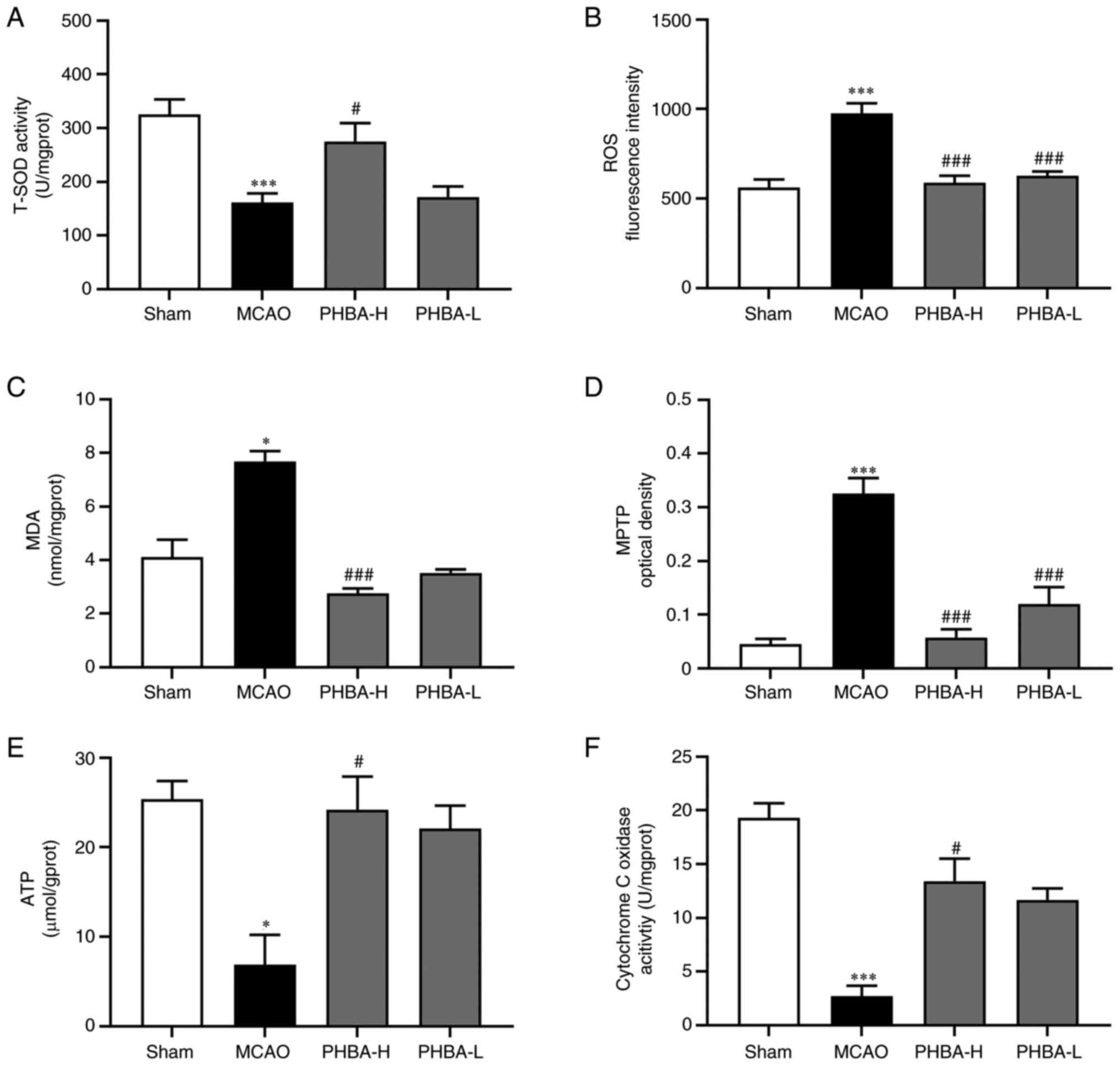
|
1
|
Wisen WP, Evans WR, Sure VN, Sperling JA,
Sakamuri SS, Mostany R and Katakam PV: Nitric oxide synthase
inhibitor is an effective therapy for ischemia-reperfusion injury
in mice. FASEB J. 36 (Suppl 1)(R5443)2022.
|
|
2
|
Chen G, Shan X, Li L, Dong L, Huang G and
Tao H: circHIPK3 regulates apoptosis and mitochondrial dysfunction
induced by ischemic stroke in mice by sponging miR-148b-3p via
CDK5R1/SIRT1. Exp Neurol. 355(114115)2022.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Huang Y, Ye Z, Ma T, Li H, Zhao Y, Chen W,
Wang Y, Yan X, Gao Y and Li Z: Carbon monoxide (CO) modulates
hydrogen peroxide (H2O2)-mediated cellular
dysfunction by targeting mitochondria in rabbit lens epithelial
cells. Exp Eye Res. 169:68–78. 2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Guan R, Zou W, Dai X, Yu X, Liu H, Chen Q
and Teng W: Mitophagy, a potential therapeutic target for stroke. J
Biomed Sci. 25(87)2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Duan C, Kuang L, Hong C, Xiang X, Liu J,
Li Q, Peng X, Zhou Y, Wang H, Liu L and Li T: Mitochondrial Drp1
recognizes and induces excessive mPTP opening after hypoxia through
BAX-PiC and LRRK2-HK2. Cell Death Dis. 12(1050)2021.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Carinci M, Vezzani B, Patergnani S,
Ludewig P, Lessmann K, Magnus T, Casetta I, Pugliatti M, Pinton P
and Giorgi C: Different roles of mitochondria in cell death and
inflammation: Focusing on mitochondrial quality control in ischemic
stroke and reperfusion. Biomedicines. 9(169)2021.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Lee HK, Jang JY, Yoo HS and Seong YH:
Neuroprotective effect of 1,3-dipalmitoyl-2-oleoylglycerol derived
from rice bran oil against cerebral ischemia-reperfusion injury in
rats. Nutrients. 14(1380)2022.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Pasala PK, Abbas Shaik R, Rudrapal M, Khan
J, Alaidarous MA, Jagdish Khairnar S, Bendale AR, Naphade VD, Kumar
Sahoo R, Zothantluanga JH and Walode SG: Cerebroprotective effect
of Aloe Emodin: In silico and in vivo studies. Saudi J Biol Sci.
29:998–1005. 2022.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Uzdensky AB: Apoptosis regulation in the
penumbra after ischemic stroke: Expression of pro- and
antiapoptotic proteins. Apoptosis. 24:687–702. 2019.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Yan H, Huang W, Rao J and Yuan J: miR-21
regulates ischemic neuronal injury via the p53/Bcl-2/Bax signaling
pathway. Aging (Albany NY). 13:22242–22255. 2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Yang M, He Y, Deng S, Xiao L, Tian M, Xin
Y, Lu C, Zhao F and Gong Y: Mitochondrial quality control: A
pathophysiological mechanism and therapeutic target for stroke.
Front Mol Neurosci. 14(786099)2022.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Hu Q, Zuo T, Deng L, Chen S, Yu W, Liu S,
Liu J, Wang X, Fan X and Dong Z: β-Caryophyllene suppresses
ferroptosis induced by cerebral ischemia reperfusion via activation
of the NRF2/HO-1 signaling pathway in MCAO/R rats. Phytomedicine.
102(154112)2022.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Zhan HD, Zhou HY, Sui YP, Du XL, Wang WH,
Dai L, Sui F, Huo HR and Jiang TL: The rhizome of Gastrodia
elata blume-an ethnopharmacological review. J Ethnopharmacol.
189:361–385. 2016.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Wang T, Chen H, Xia S, Chen X, Sun H and
Xu Z: Ameliorative effect of parishin C against cerebral
ischemia-induced brain tissue injury by reducing oxidative stress
and inflammatory responses in rat model. Neuropsychiatr Dis Treat.
17:1811–1823. 2021.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Zhang ZL, Gao YG, Zang P, Gu PP, Zhao Y,
He ZM and Zhu HY: Research progress on mechanism of gastrodin and
p-hydroxybenzyl alcohol on central nervous system. Zhongguo Zhong
Yao Za Zhi. 45:312–320. 2020.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
16
|
Yang F, Li G, Lin B and Zhang K: Gastrodin
suppresses pyroptosis and exerts neuroprotective effect in
traumatic brain injury model by inhibiting NLRP3 inflammasome
signaling pathway. J Integr Neurosci. 21(72)2022.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Liu Y, Lu YY, Huang L, Shi L, Zheng ZY,
Chen JN, Qu Y, Xiao HT, Luo HR and Wu GS: Para-hydroxybenzyl
alcohol delays the progression of neurodegenerative diseases in
models of caenorhabditis elegans through activating multiple
cellular protective pathways. Oxid Med Cell Longev.
2022(8986287)2022.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Gong X, Cheng J, Zhang K, Wang Y, Li S and
Luo Y: Transcriptome sequencing reveals Gastrodia elata
Blume could increase the cell viability of eNPCs under hypoxic
condition by improving DNA damage repair ability. J Ethnopharmacol.
282(114646)2022.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Duan X, Wang W, Liu X, Yan H, Dai R and
Lin Q: Neuroprotective effect of ethyl acetate extract from
Gastrodia elata against transient focal cerebral ischemia in
rats induced by middle cerebral artery occlusion. J Tradit Chin
Med. 35:671–678. 2015.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Jiang G, Hu Y, Liu L, Cai J, Peng C and Li
Q: Gastrodin protects against MPP(+)-induced oxidative stress by up
regulates heme oxygenase-1 expression through p38 MAPK/Nrf2 pathway
in human dopaminergic cells. Neurochem Int. 75:79–88.
2014.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Luo L, Kim SW, Lee HK, Kim ID, Lee H and
Lee JK: Anti-Zn2+-toxicity of 4-hydroxybenzyl alcohol in
astrocytes and neurons contribute to a robust neuroprotective
effects in the postischemic brain. Cell Mol Neurobiol. 38:615–626.
2018.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Li X, Xiang B, Shen T, Xiao C, Dai R, He F
and Lin Q: Anti-neuroinflammatory effect of
3,4-dihydroxybenzaldehyde in ischemic stroke. Int Immunopharmacol.
82(106353)2020.PubMed/NCBI View Article : Google Scholar : (Epub ahead of
print).
|
|
23
|
Kim HJ, Hwang IK and Won MH: Vanillin,
4-hydroxybenzyl aldehyde and 4-hydroxybenzyl alcohol prevent
hippocampal CA1 cell death following global ischemia. Brain Res.
1181:130–141. 2007.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Wu J, Wu B, Tang C and Zhao J: Analytical
techniques and pharmacokinetics of Gastrodia elata blume and
its constituents. Molecules. 22(1137)2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Liu CS, Liang X, Wei XH, Chen FL, Tang QF
and Tan XM: Comparative pharmacokinetics of major bioactive
components from Puerariae radix-gastrodiae rhizome extracts and
their intestinal absorption in rats. J Chromatogr B Analyt Technol
Biomed Life Sci. 1105:38–46. 2019.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Loh YC, Oo CW, Tew WY, Wen X, Wei X and
Yam MF: The predominance of endothelium-derived relaxing factors
and beta-adrenergic receptor pathways in strong vasorelaxation
induced by 4-hydroxybenzaldehyde in the rat aorta. Biomed
Pharmacother. 150(112905)2022.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Lee JY, Jang YW, Kang HS, Moon H, Sim SS
and Kim CJ: Anti-inflammatory action of phenolic compounds from
Gastrodia elata root. Arch Pharm Res. 29:849–858.
2006.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Ha JH, Lee DU, Lee JT, Kim JS, Yong CS,
Kim JA, Ha JS and Huh K: 4-Hydroxybenzaldehyde from Gastrodia
elata B1. is active in the antioxidation and GABAergic
neuromodulation of the rat brain. J Ethnopharmacol. 73:329–333.
2000.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Feng J, Sun H, Xu Y, Yang Q and He F:
Gastrointestinal absorption characteristics of
p-hydroxybenzaldehyde, a constituent of Gastrodia elata.
Pharmacology and Clinics of Chinese Materia Medica. 37:23–28.
2021.
|
|
30
|
Zhu YP, Li X, Du Y, Zhang L, Ran L and
Zhou NN: Protective effect and mechanism of p-hydroxybenzaldehyde
on blood-brain barrier. Zhongguo Zhong Yao Za Zhi. 43:1021–1027.
2018.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
31
|
Ha JH, Shin SM, Lee SK, Kim JS, Shin US,
Huh K, Kim JA, Yong CS, Lee NJ and Lee DU: In vitro effects of
hydroxybenzaldehydes from Gastrodia elata and their
analogues on GABAergic neurotransmission, and a structure-activity
correlation. Planta Med. 67:877–880. 2001.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Gorsky A, Monsour M, Nguyen H, Castelli V,
Lee JY and Borlongan CV: Metabolic switching of cultured
mesenchymal stem cells creates super mitochondria in rescuing
ischemic neurons. Neuromolecular Med: Jul 20, 2022 (Epub ahead of
print).
|
|
33
|
Brenza TM, Ghaisas S, Ramirez JEV,
Harischandra D, Anantharam V, Kalyanaraman B, Kanthasamy AG and
Narasimhan B: Neuronal protection against oxidative insult by
polyanhydride nanoparticle-based mitochondria-targeted antioxidant
therapy. Nanomedicine. 13:809–820. 2017.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Buchke S, Sharma M, Bora A, Relekar M,
Bhanu P and Kumar J: Mitochondria-targeted, nanoparticle-based
drug-delivery systems: Therapeutics for mitochondrial disorders.
Life (Basel). 12(657)2022.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Li N, Qin S, Xie L, Qin T, Yang Y, Fang W
and Chen MH: Elevated serum potassium concentration alleviates
cerebral ischemia-reperfusion injury via mitochondrial
preservation. Cell Physiol Biochem. 48:1664–1674. 2018.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Zhang M, Yang JK and Ma J: Regulation of
the long noncoding RNA XIST on the inflammatory polarization of
microglia in cerebral infarction. Exp Ther Med.
22(924)2021.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Bederson JB, Pitts LH, Tsuji M, Nishimura
MC, Davis RL and Bartkowski H: Rat middle cerebral artery
occlusion: Evaluation of the model and development of a neurologic
examination. Stroke. 17:472–476. 1986.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Tian J, Yao H, Liu Y, Wang X, Wu J, Wang
J, Yu D, Xie Y, Gao J, Zhu Y and Yang C: Extracellular vesicles
from bone marrow stromal cells reduce the impact of stroke on glial
cell activation and blood brain-barrier permeability via a putative
miR-124/PRX1 signalling pathway. Eur J Neurosci. 56:3786–3805.
2022.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Lee J, Kang CG, Park CR, Hong IK and Kim
DY: The neuroprotective effects of pregabalin after cerebral
ischemia by occlusion of the middle cerebral artery in rats. Exp
Ther Med. 21(165)2021.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Yu SS, Zhao J, Zheng WP and Zhao Y:
Neuroprotective effect of 4-hydroxybenzyl alcohol against transient
focal cerebral ischemia via anti-apoptosis in rats. Brain Res.
1308:167–175. 2010.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Cui L, Zhang X, Yang R, Liu L, Wang L, Li
M and Du W: Baicalein is neuroprotective in rat MCAO model: Role of
12/15-lipoxygenase, mitogen-activated protein kinase and cytosolic
phospholipase A2. Pharmacol Biochem Behav. 96:469–475.
2010.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Jang JH, Son Y, Kang SS, Bae CS, Kim JC,
Kim SH, Shin T and Moon C: Neuropharmacological potential of
Gastrodia elata blume and its components. Evid Based
Complement Alternat Med. 2015(309261)2015.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Liu Y, Gao J, Peng M, Meng H, Ma H, Cai P,
Xu Y, Zhao Q and Si G: A review on central nervous system effects
of gastrodin. Front Pharmacol. 9(24)2018.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Xiang B, Xiao C, Shen T and Li X:
Anti-inflammatory effects of anisalcohol on
lipopolysaccharide-stimulated BV2 microglia via selective
modulation of microglia polarization and down-regulation of NF-κB
p65 and JNK activation. Mol Immunol. 95:39–46. 2018.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Shi A, Xiang J, He F, Zhu Y, Zhu G, Lin Y
and Zhou N: The phenolic components of Gastrodia elata
improve prognosis in rats after cerebral ischemia/reperfusion by
enhancing the endogenous antioxidant mechanisms. Oxid Med Cell
Longev. 2018(7642158)2018.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Li L, Wan G, Han B and Zhang Z:
Echinacoside alleviated LPS-induced cell apoptosis and inflammation
in rat intestine epithelial cells by inhibiting the mTOR/STAT3
pathway. Biomed Pharmacother. 104:622–628. 2018.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Liu B, Li F, Shi J, Yang D, Deng Y and
Gong Q: Gastrodin ameliorates subacute phase cerebral
ischemia-reperfusion injury by inhibiting inflammation and
apoptosis in rats. Mol Med Rep. 14:4144–4152. 2016.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Mi Y, Mao Y, Cheng H, Ke G, Liu M, Fang C
and Wang Q: Studies of blood-brain barrier permeability of
gastrodigenin in vitro and in vivo. Fitoterapia.
140(104447)2020.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Wang S, Nan Y, Zhu W, Yang T, Tong Y and
Fan Y: Gastrodin improves the neurological score in MCAO rats by
inhibiting inflammation and apoptosis, promoting revascularization.
Int J Clin Exp Pathol. 11:5343–5350. 2018.PubMed/NCBI
|
|
50
|
Ning L, Sun H, Feng J, Yang Q, Zhou Y and
He F: Tissue distribution of p-hydroxybenzaldehyde metabolite 4-HBA
in rats. J Shanghai Univ Tradit Chin Med. 35:65–70. 2021.
|
|
51
|
Kim HS, Choi JA, Kim BY, Ferrer L, Choi J,
Wendisch VF and Lee JH: Engineered corynebacterium glutamicum as
the platform for the production of aromatic aldehydes. Front Bioeng
Biotechnol. 10(880277)2022.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Duan XH, Li ZL, Yang DS, Zhang FL, Lin Q
and Dai R: Study on the chemical constituents of Gastrodia
elata. Zhong Yao Cai. 36:1608–1611. 2013.PubMed/NCBI(In Chinese).
|
|
53
|
Lemmerman LR, Harris HN, Balch MHH,
Rincon-Benavides MA, Higuita-Castro N, Arnold DW and Gallego-Perez
D: Transient middle cerebral artery occlusion with an intraluminal
suture enables reproducible induction of ischemic stroke in mice.
Bio Protoc. 12(e4305)2022.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Yuan Y, Xia F, Gao R, Chen Y, Zhang Y,
Cheng Z, Zhao H and Xu L: Kaempferol mediated AMPK/mTOR signal
pathway has a protective effect on cerebral ischemic-reperfusion
injury in rats by inducing autophagy. Neurochem Res. 47:2187–2197.
2022.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Zhu A, Lin Y, Hu X, Lin Z, Lin Y, Xie Q,
Ni S, Cheng H, Lu Q, Lai S, et al: Treadmill exercise decreases
cerebral edema in rats with local cerebral infarction by modulating
AQP4 polar expression through the caveolin-1/TRPV4 signaling
pathway. Brain Res Bull. 188:155–168. 2022.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Jin M, Kim KM, Lim C, Cho S and Kim YK:
Neuroprotective effects of Korean White ginseng and Red ginseng in
an ischemic stroke mouse model. J Ginseng Res. 46:275–282.
2022.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Qi S, Zhang X, Fu Z, Pi A, Shi F, Fan Y,
Zhang J, Xiao T, Shang D, Lin M, et al:
(±)-5-bromo-2-(5-fluoro-1-hydroxyamyl) benzoate protects against
oxidative stress injury in PC12 cells exposed to
H2O2 through activation of Nrf2 pathway.
Front Pharmacol. 13(943111)2022.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Song ZL, Bai F, Zhang B and Fang J:
Synthesis of Dithiolethiones and Identification of Potential
Neuroprotective Agents via Activation of Nrf2-Driven Antioxidant
Enzymes. J Agric Food Chem. 68:2214–2231. 2020.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Huang SS, Su HH, Chien SY, Chung HY, Luo
ST, Chu YT, Wang YH, MacDonald IJ, Lee HH and Chen YH: Activation
of peripheral TRPM8 mitigates ischemic stroke by topically applied
menthol. J Neuroinflammation. 19(192)2022.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Wu J, Jin Z, Yang X and Yan LJ:
Post-ischemic administration of 5-methoxyindole-2-carboxylic acid
at the onset of reperfusion affords neuroprotection against stroke
injury by preserving mitochondrial function and attenuating
oxidative stress. Biochem Biophys Res Commun. 497:444–450.
2018.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Narne P, Pandey V and Phanithi PB:
Interplay between mitochondrial metabolism and oxidative stress in
ischemic stroke: An epigenetic connection. Mol Cell Neurosci.
82:176–194. 2017.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Li Y, Zhang X, Ma A and Kang Y: Rational
application of β-hydroxybutyrate attenuates ischemic stroke by
suppressing oxidative stress and mitochondrial-dependent apoptosis
via activation of the Erk/CREB/eNOS pathway. ACS Chem Neurosci.
12:1219–1227. 2021.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Kaushik P, Ali M, Salman M, Tabassum H and
Parvez S: Harnessing the mitochondrial integrity for
neuroprotection: Therapeutic role of piperine against experimental
ischemic stroke. Neurochem Int. 149(105138)2021.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Morkuniene R, Rekuviene E and
Kopustinskiene DM: Rotenone decreases ischemia-induced injury by
inhibiting mitochondrial permeability transition: A study in brain.
Methods Mol Biol. 2497:63–72. 2022.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Huang P, Wu SP, Wang N, Seto S and Chang
D: Hydroxysafflor yellow A alleviates cerebral ischemia reperfusion
injury by suppressing apoptosis via mitochondrial permeability
transition pore. Phytomedicine. 85(153532)2021.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Su F, Yang H, Guo A, Qu Z, Wu J and Wang
Q: Mitochondrial BKCa mediates the protective effect of low-dose
ethanol preconditioning on oxygen-glucose deprivation and
reperfusion-induced neuronal apoptosis. Front Physiol.
12(719753)2021.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Malko P and Jiang LH: TRPM2
channel-mediated cell death: An important mechanism linking
oxidative stress-inducing pathological factors to associated
pathological conditions. Redox Biol. 37(101755)2020.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Xu M, Ma Q, Fan C, Chen X, Zhang H and
Tang M: Ginsenosides Rb1 and Rg1 protect primary cultured
astrocytes against oxygen-glucose deprivation/reoxygenation-induced
injury via improving mitochondrial function. Int J Mol Sci.
20(6086)2019.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Pozdnyakov DI, Chernikov MV, Sarkisyan KK
and Rybalko IE: Neuroprotective potential of pyrimidine-4-H1-OHa
derivatives in experimental cerebral ischemia. Zh Nevrol Psikhiatr
Im S S Korsakova. 121:63–68. 2021.PubMed/NCBI View Article : Google Scholar : (In Russian).
|
|
70
|
Morse PT, Goebel DJ, Wan J, Tuck S, Hakim
L, Hüttemann CL, Malek MH, Lee I, Sanderson TH and Hüttemann M:
Cytochrome c oxidase-modulatory near-infrared light penetration
into the human brain: Implications for the noninvasive treatment of
ischemia/reperfusion injury. IUBMB Life. 73:554–567.
2021.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Ma Y, Zhao P, Zhu J, Yan C, Li L, Zhang H,
Zhang M, Gao X and Fan X: Naoxintong protects primary neurons from
oxygen-glucose deprivation/reoxygenation induced injury through
PI3K-Akt signaling pathway. Evid Based Complement Alternat Med.
2016(5815946)2016.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Zeng JW, Chen BY, Lv XF, Sun L, Zeng XL,
Zheng HQ, Du YH, Wang GL, Ma MM and Guan YY: Transmembrane member
16A participates in hydrogen peroxide-induced apoptosis by
facilitating mitochondria-dependent pathway in vascular smooth
muscle cells. Br J Pharmacol. 175:3669–3684. 2018.PubMed/NCBI View Article : Google Scholar
|
|
73
|
He F, Wu Q, Xu B, Wang X, Wu J, Huang L
and Cheng J: Suppression of Stim1 reduced intracellular calcium
concentration and attenuated hypoxia/reoxygenation induced
apoptosis in H9C2 cells. Biosci Rep. 37(BSR20171249)2017.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Momenabadi S, Vafaei AA, Zahedi Khorasani
M and Vakili A: Pre-ischemic oxytocin treatment alleviated neuronal
injury via suppressing NF-κB, MMP-9, and apoptosis regulator
proteins in a mice model of stroke. Cell J. 24:337–345.
2022.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Huang LY, Song JX, Cai H, Wang PP, Yin QL,
Zhang YD, Chen J, Li M, Song JJ, Wang YL, et al: Healthy
serum-derived exosomes improve neurological outcomes and protect
blood-brain barrier by inhibiting endothelial cell apoptosis and
reversing autophagy-mediated tight junction protein reduction in
rat stroke model. Front Cell Neurosci. 16(841544)2022.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Cory S and Adams JM: The Bcl2 family:
Regulators of the cellular life-or-death switch. Nat Rev Cancer.
2:647–656. 2002.PubMed/NCBI View
Article : Google Scholar
|
|
77
|
Zhai Y, Liu B, Wu L, Zou M, Mei X and Mo
X: Pachymic acid prevents neuronal cell damage induced by
hypoxia/reoxygenation via miR-155/NRF2/HO-1 axis. Acta Neurobiol
Exp (Wars). 82:197–206. 2022.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Yan Y, Liu Y, Yang Y, Ding Y and Sun X:
Carnosol suppresses microglia cell inflammation and apoptosis
through PI3K/AKT/mTOR signaling pathway. Immunopharmacol
Immunotoxicol. 1–11. 2022.PubMed/NCBI View Article : Google Scholar : (Epub ahead of
print).
|
|
79
|
Liu X, Zhu X, Chen M, Ge Q, Shen Y and Pan
S: Resveratrol protects PC12 cells against OGD/R-induced apoptosis
via the mitochondrial-mediated signaling pathway. Acta Biochim
Biophys Sin (Shanghai). 48:342–353. 2016.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Wang PC, Wang SX, Yan XL, He YY, Wang MC,
Zheng HZ, Shi XG, Tan YH and Wang LS: Combination of paeoniflorin
and calycosin-7-glucoside alleviates ischaemic stroke injury via
the PI3K/AKT signalling pathway. Pharm Biol. 60:1469–1477.
2022.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Khaksar S, Bigdeli M, Samiee A and
Shirazi-Zand Z: Antioxidant and anti-apoptotic effects of
cannabidiol in model of ischemic stroke in rats. Brain Res Bull.
180:118–130. 2022.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Li Z, Xiao G, Wang H, He S and Zhu Y: A
preparation of Ginkgo biloba L. leaves extract inhibits the
apoptosis of hippocampal neurons in post-stroke mice via regulating
the expression of Bax/Bcl-2 and caspase-3. J Ethnopharmacol.
280(114481)2021.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Yang L, Tao Y, Luo L, Zhang Y, Wang X and
Meng X: Dengzhan Xixin injection derived from a traditional Chinese
herb Erigeron breviscapus ameliorates cerebral ischemia/reperfusion
injury in rats via modulation of mitophagy and mitochondrial
apoptosis. J Ethnopharmacol. 288(114988)2022.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Nhu NT, Li Q, Liu Y, Xu J, Xiao SY and Lee
SD: Effects of mdivi-1 on neural mitochondrial dysfunction and
mitochondria-mediated apoptosis in ischemia-reperfusion injury
after stroke: A systematic review of preclinical studies. Front Mol
Neurosci. 14(778569)2021.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Gan L, Liao S, Tong Y, Li W, Peng W and
Deng S: Long noncoding RNA H19 mediates neural stem/progenitor
cells proliferation, differentiation and apoptosis through the p53
signaling pathway after ischemic stroke. Biochem Biophys Res
Commun. 597:8–15. 2022.PubMed/NCBI View Article : Google Scholar : (Epub ahead of
print).
|
|
86
|
MacDougall G, Anderton RS, Mastaglia FL,
Knuckey NW and Meloni BP: Mitochondria and neuroprotection in
stroke: Cationic arginine-rich peptides (CARPs) as a novel class of
mitochondria-targeted neuroprotective therapeutics. Neurobiol Dis.
121:17–33. 2019.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Shefa U, Jeong NY, Song IO, Chung HJ, Kim
D, Jung J and Huh Y: Mitophagy links oxidative stress conditions
and neurodegenerative diseases. Neural Regen Res. 14:749–756.
2019.PubMed/NCBI View Article : Google Scholar
|
|
88
|
Awooda HA, Lutfi MF, Sharara GG and Saeed
AM: Oxidative/nitrosative stress in rats subjected to focal
cerebral ischemia/reperfusion. Int J Health Sci (Qassim). 9:17–24.
2015.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Chen Y: Disturbed cerebral circulation and
metabolism matters: A preface to the special issue ‘stroke and
energy metabolism’: A preface to the special issue ‘stroke and
energy metabolism’. J Neurochem. 160:10–12. 2022.PubMed/NCBI View Article : Google Scholar
|