Introduction
Neonatal acute kidney injury (AKI) refers to the
rapid decline of neonatal renal function caused by various
etiologies, which is common in neonates admitted to the neonatal
intensive care unit (NICU) and is associated with increased
morbidity and mortality (1). The
incidence of AKI in the NICU is estimated to be ~30% and those with
AKI exhibit >4-fold higher independent odds of death (2). The main causes of neonatal AKI
include perinatal asphyxia, followed by congenital kidney and
urinary tract malformations, congenital heart disease, and sepsis.
The clinical manifestations are oliguria or anuria, electrolyte
disorder, imbalance of acid-base balance, and increased
concentration of metabolites in plasma (3). At present, there are no effective
prevention and treatment measures for neonatal AKI.
Continuous renal replacement therapy (CRRT), also
known as continuous blood purification (CBP), removes harmful
substances continuously, rapidly, and directly. In recent years,
with the development of CBP technology, the application of CBP has
been expanded from AKI in children to neonatal AKI (4). However, there is still a lack of
large sample multicenter research data of CRRT regarding neonatal
AKI treatment. A study conducted by The Children's Hospital of
Ankara University in Turkey retrospectively evaluated NICU and PICU
hospitalized patients who received CRRT from February 2010 to
November 2015, proving that CRRT is a life-saving method, and an
experienced team can be applied to critical children with AKI and
fluid overload of any age and weight (5). In the present study, the clinical
data of 25 AKI neonates treated with CRRT in the NICU were analyzed
in order to explore the potential application of CRRT and its
efficacy in the treatment of neonatal AKI.
Materials and methods
Ethics approval and consent to
participate
All parents of the infants signed the informed
consent for the study. The present study was approved (approval no.
2012C001-F01) by the Ethics Committee of Shanghai Children's
Hospital, School of Medicine, Shanghai Jiao Tong University
(Shanghai, China).
Subjects of the study
From November 2016 to June 2021, 41 critically ill
neonates who received CRRT in the NICU of Shanghai Children's
Hospital, School of Medicine, Shanghai Jiao Tong University were
included in the study. Among them, 25 AKI neonates were treated
with CRRT as the subjects of the present study (cases treated with
CRRT for other reasons, such as metabolic diseases were excluded).
According to the diagnostic criteria of neonatal AKI (Table I), 25 cases were divided into two
groups: AKI stage 0-1 (15 cases) and AKI stage 2-3 (10 cases). The
gestational age (GA) of the 25 AKI neonates ranged from
33+4 to 40+1 weeks, the age of admission
ranged from 2 to 28 days, and the birth weight range was
2,250-4,000 g. A total of 14 males, 11 females, 18 term infants,
and 7 preterm infants were included. The primary diseases included
10 cases of severe asphyxia at birth, 8 cases of neonatal
septicemia, and 7 cases of congenital metabolic diseases.
 | Table INeonatal KDIGO acute kidney injury
definition. |
Table I
Neonatal KDIGO acute kidney injury
definition.
| Stage | SCr | Urine output over 24
h |
|---|
| 0 | No change in SCr or
an increase of <0.3 mg/dl | >1 ml/kg/h |
| 1 | SCr increase of ≥0.3
mg/dl within 48 h or an SCr | |
| | increase of ≥1.5 to
1.9 x reference SCra
within 7 days | >0.5 and ≤1
ml/kg/h |
| 2 | SCr increase of ≥2 to
2.9 x reference SCra | >0.3 and ≤0.5
ml/kg/h |
| 3 | SCr increase of ≥3 x
reference SCra or an
SCr | |
| | increase of ≥2.5
mg/dla or receipt of
dialysis | ≤0.3 ml/kg/h |
Diagnostic criteria for neonatal
AKI
According to the AKI clinical practice guidelines
issued by the Global Committee for the Improvement of the Prognosis
of Kidney Diseases: Improving Global Outcomes (KDIGO) in
2013(6), the current diagnostic
criteria of neonatal AKI mainly depend on the changes in the
concentration of serum creatinine and the volume of urine (Table I). Since the serum creatinine
levels of neonates are influenced by maternal factors, premature
delivery, hyperbilirubinemia, and other factors and the majority of
the neonatal cases with AKI are non-oliguric cases with AKI, the
early diagnosis of neonatal AKI is considerably difficult (7).
CRRT treatment for AKI neonates
The indications for CRRT were as follows: CRRT
treatment was initiated as long as the diagnostic criteria of AKI
were met and irrespective of the factors causing AKI or its stage
(8,9).
The following method of CRRT was used: i) Operating
methods: The equipment used was Plasauto IQ21, which was a blood
purifier instrument manufactured in Japan. Continuous veno-venous
hemodialysis filtration (CVVHDF) mode was selected according to the
molecular weight of solute removal. The CRRT instrument was divided
into the external blood circulation and the filter. The specific
operation process was as follows: External blood from the
circulation was mixed with physiological saline containing heparin
and filtered; 68 ml red cell suspension was used to pre-fill the
external blood circulation sample and the new sample was filtered.
The blood circulation volume of CRRT was 38 ml (arterial line +
venous line). The CRRT instrument was purchased from Japan Lai Fuen
Co., Ltd. and the filtering capacity was 30 ml. For CRRT, 4Fr, 5Fr,
single-tube, and a double-chamber central venous catheter were
used. The arterial foramen was located at the telecentric end and
the venous foramen at the proximal end, 2 to 3 mm apart. The blood
recirculation was <10%. The most common puncture sites were the
femoral vein, the internal jugular vein, and the umbilical vein,
which could be used for neonates with an age <7 days. The blood
purifier pipeline was connected for bypass. ii) The following CBP
parameters were used: The initial flow rate of the blood pump was 3
ml/(kg/min) which was increased to 5 ml/(kg/min) according to the
blood pressure. The replacement fluid and the dialysate had the
following volume range: 20-30 ml/(kg/h) for the replacement fluid,
and 15-25 ml/(min/m2) for the dialysate. The dehydration
speed was calculated by the following formula: Filter pump -
dialysis pump-rehydration pump, uninterrupted flow. iii) The
following dialysis and replacement fluids were used: Baxter
dialysate (Baxter International, Inc.). The replacement solution
was prepared by NICU nurses in the unit. The Ports scheme was
adopted to improve the formula; specifically, Ringer's solution
(3,000 ml) was mixed with 5% glucose solution (100 ml), 10% calcium
chloride solution (7.5 ml), 50% magnesium sulfate solution (1.6 ml)
and 5% sodium bicarbonate solution (200 ml). The ionic
concentrations of the formula components were the following: 130.0
mmol/l sodium ion, 4.0 mmol/l potassium ion, 28.0 mmol/l
bicarbonate ion, 1.5 mmol/l calcium ion, 3.2 mmol/l magnesium ion
and 109.0 mmol/l chloride ion. The final concentration of glucose
was 0.2 g/l. The ion concentration was adjusted according to
electrolyte monitoring.
The following maintenance of CRRT was used: i)
Anticoagulation was achieved by filtering and pre-filling with
heparin solution. Heparin anticoagulation was used to maintain
prothrombin time at 25-40 sec; the activated partial thromboplastin
time range was 80-120 sec. The dosage range of heparin was
generally 5-40 U/(kg/h). ii) The replacement of the filter
membranes was performed in case blockage was noted during
treatment. The termination indications of the CRRT were as follows:
A neonatal urine volume of AKI >2 ml/(kg/h), the presence of
anhydrous solution, and an electrolyte acid-base balance
disorder.
Plasma exchange (PE) treatment for AKI
neonates
PE is a special support and treatment method, which
is a part of blood purification technology. It refers to extracting
the blood of a patient, separating and filtering out the pathogenic
substances existing in the blood circulation of the body, such as
poisons, pathogens, autoantibodies and metabolites, and then
returning them to the patient and replenishing the same amount of
replacement fluid, so as to achieve the purpose of treatment and
support. Its technologies mainly include total plasma exchange
(TPE) and double filtration plasma exchange (DFPP) (10).
To perform PE, 4.0 or 5.0Fr double lumen central
venous catheters, and internal jugular vein, subclavian vein or
femoral vein catheters were selected. The single exchange dose was:
Plasma volume (L)=0.08 x weight (kg) x (1-hematocrit), and the
general replacement plasma volume was 50-80 ml/kg per newborn. The
replacement solution is usually: i) Plasma products, including
fresh plasma, fresh frozen plasma and purified plasma protein. The
main advantage is that these plasma products contain most
coagulation factors, albumin and immunoglobulins, and are suitable
for patients with deficiency of coagulation factors or other
factors. ii) Human albumin: 5% human albumin is commonly used. Due
to the low concentration of potassium, calcium and magnesium in
human serum albumin, attention should be paid to adjusting the
electrolyte balance during the treatment to avoid causing
hypokalemia and hypocalcemia. iii) Crystal solution: Including
normal saline, glucose normal saline, and Ringer's solution, used
to supplement the loss of various electrolytes in plasma. The
exchange rate, generally, is the amount of plasma exchanged and is
50-80 ml/kg each time, and the blood flow rate is 3-5 ml/kg. The
displacement pump speed was 1/4-1/3 of the blood flow rate. The
general treatment time was 2-3 h. The anticoagulant selected was
unfractionated heparin continuously infused at 10-20 U/kg per hour
during the replacement process, and the addition is expected to
stop 30 min before the end of the process. Heparin dosage should be
adjusted individually according to the coagulation status of
patients. Attention should be paid to monitoring electrolyte, blood
gas and fibrinogen levels while also monitoring vital signs.
Calcium chloride or calcium gluconate can be continuously
supplemented during blood replacement.
Observation indices
The observation indices included the following: i)
The changes in the serum levels of potassium, sodium, urea
nitrogen, and creatinine and the urine volume were assessed prior
to CRRT treatment, at 12, 24 and 48 h and at the end of CRRT
treatment; ii) the changes in the renal function prior to, 24 h
after and at the end of the CRRT treatment in AKI neonates
classified as stage 0-1 and 2-3; iii) the clinical outcome of 25
AKI neonates; iv) the occurrence of CRRT-related complications,
such as pipeline blockage, hypothermia, hemorrhage, thrombosis,
infection and thrombocytopenia.
Evaluation of efficacy
The curative effect was evaluated as follows: The
changes in the renal function, urine volume, blood electrolyte
levels, and acid-base balance prior to and following CRRT treatment
were analyzed. In case the aforementioned indicators were
apparently improved and/or no improvement was noted, the treatment
was considered to be ineffective.
Evaluation of safety
In the present study, the failure rate of
intravenous catheterization in neonates and the incidence rate of
CRRT-related complications were used to evaluate the safety of CRRT
in the application of AKI in neonates, both of which were expressed
by the percentage of failure of catheterization and CRRT related
complications.
Statistical analysis
SPSS 22.0 (IBM Corp.) was used to analyze the data.
The measurement data in the present study followed a normal
distribution and were expressed by the mean ± standard deviation
(x±s). One way repeated measures ANOVA was used to test the effect
of CRRT on serum sodium, potassium, blood urea nitrogen, blood
creatinine and urine output of neonates with AKI at various time
points of treatment, and Bonferroni's post hoc test was used for
pairwise comparison between groups. Two-way repeated measures ANOVA
was used to test the effect of CRRT on urine output and serum
creatinine of neonates with AKI stage 0-1 and AKI stage 2-3.
P<0.05 was considered to indicate a statistically significant
difference.
Results
General information
The GA of 25 AKI newborns ranged from
33+4 to 40+1 weeks; the age range of
admission was 2-28 days; the birth weight range was 2,250~4,000 g,
with an average weight of 3,090±550 g. A total of 15 cases with AKI
were identified as stage 0-1 and 10 cases with AKI as stage 2-3.
The primary diseases included 10 cases of severe asphyxia at birth
(including 1 case of traumatic asphyxia), 8 cases of neonatal
septicemia, 7 cases of congenital metabolic diseases, including 3
cases of urea circulation disorder, 1 case of
pseudohypoaldosteronism, 1 case of midchain acyl coenzyme A
dehydrogenase deficiency, 1 case of methylmalonate acidemia, and 1
case of liver failure. CVVHDF was used in 23 cases and PE was used
in 2 cases combined with CRRT. The mean time of CRRT treatment was
90.1±32.6 h.
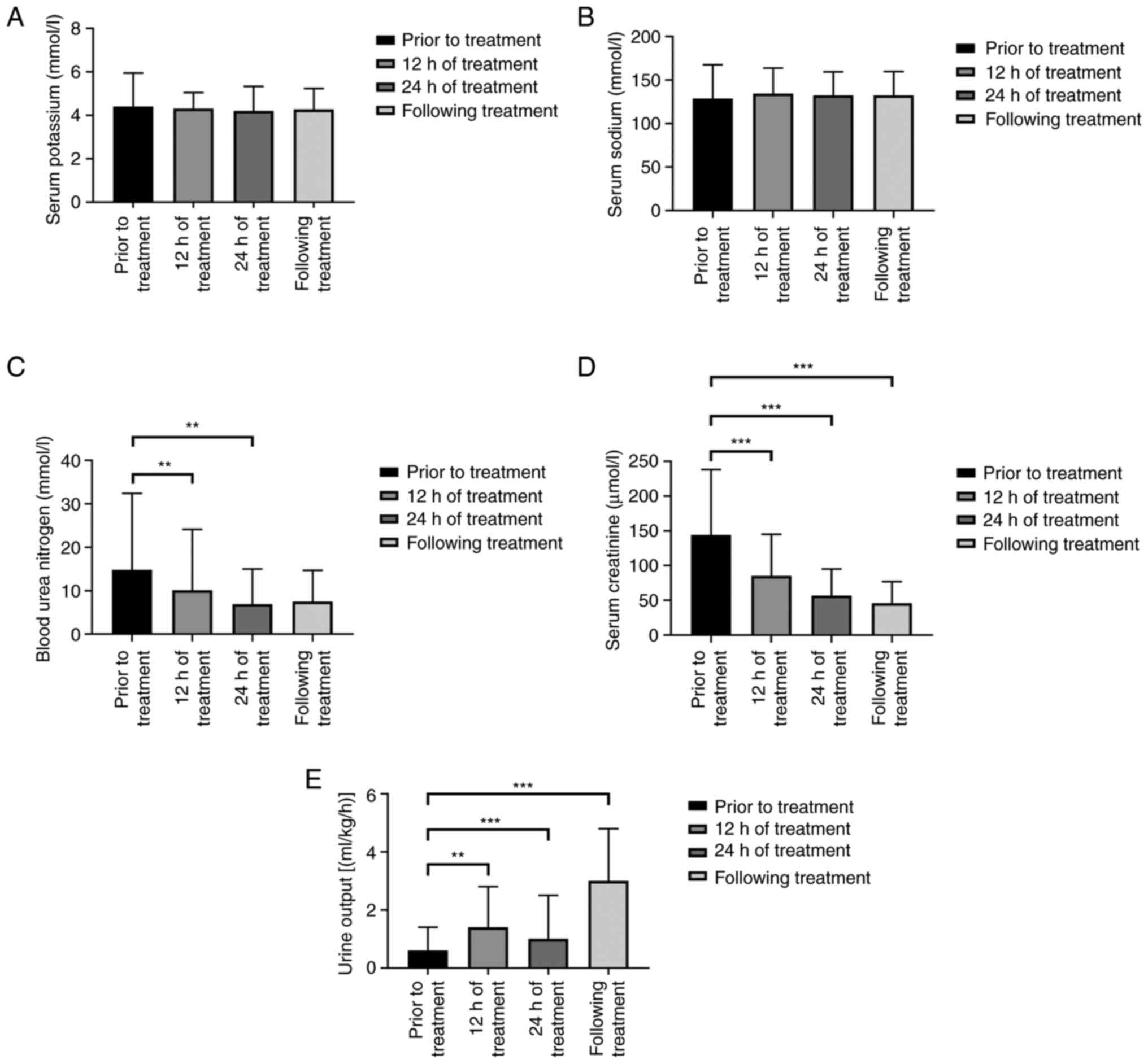
Changes in renal function and
electrolyte indicator levels prior and following CRRT
treatment
The results of one-way repeated measurement ANOVA
showed that there were significant differences in creatinine
levels, the blood urea nitrogen levels and urine output at each
time-point of CRRT treatment (Table
II and Fig. 1).
 | Table IIChanges of renal function and
electrolyte indexes of 25 neonates with AKI before and after CRRT
therapy (x±s). |
Table II
Changes of renal function and
electrolyte indexes of 25 neonates with AKI before and after CRRT
therapy (x±s).
| Time-points | Serum potassium
(mmol/l) | Serum Sodium
(mmol/l) | Blood urea nitrogen
(mmol/l) | Serum creatinine
(µmol/l) | Urine output
(ml/kg/h) |
|---|
| Prior to
treatment | 4.41±1.54 | 128.74±38.98 | 14.8±17.6 | 144±94 | 0.6±0.8 |
| 12 h of
treatment | 4.31±0.74 | 134.45±29.50 |
10.1±14.0a | 85±60b | 1.4±1.4a |
| 24 h of
treatment | 4.20±1.14 | 132.34±27.25 | 6.9±8.1a | 57±38a | 1.9±1.5b |
| Following
treatment | 4.27±0.97 | 132.60±27.27 | 7.5±7.2 | 46±31b |
3.0±1.8b |
| F-value | 0.198 | 0.490 | 7.201 | 28.648 | 22.581 |
| P-value | 0.897 | 0.508 | 0.008 | <0.001 | <0.001 |
The results of Bonferroni multiple mean comparison
showed that the level of serum creatinine and blood urea nitrogen
prior to CRRT was significantly higher than that at 12 and 24 h as
well as following treatment. In addition, the urine output was
significantly increased at 12 and 24 h and following CRRT treatment
than that prior to treatment (Table
II and Fig. 1).
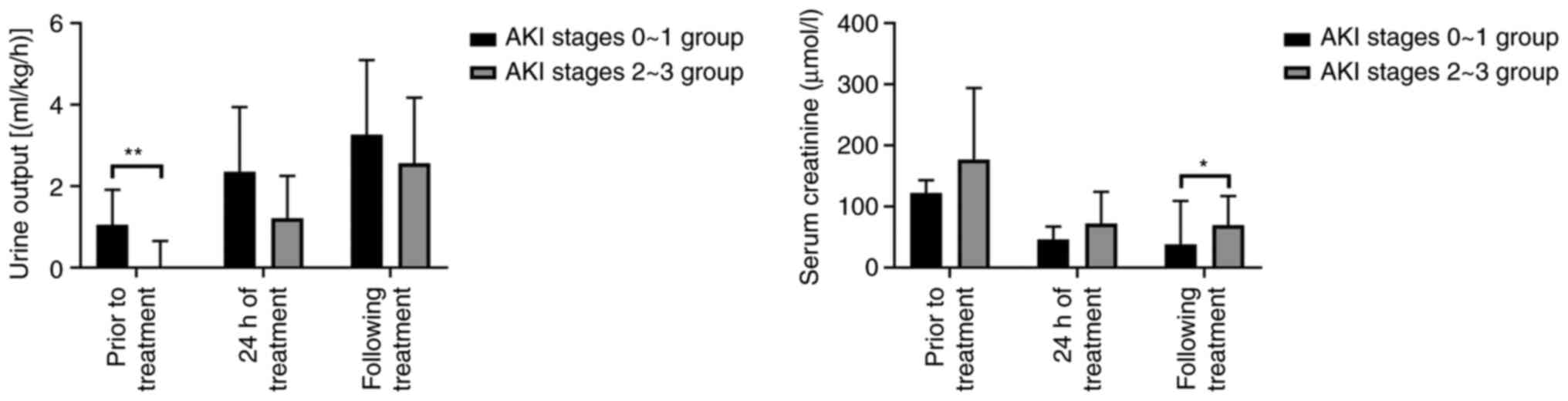
Comparison of urine output and serum
creatinine levels prior to and following CRRT treatment for the AKI
neonates of the two groups
The serum creatinine level of neonates with AKI
stage 0-1 was significantly lower than that with AKI stage 2-3
following CRRT (F=5.005; P=0.035). At each time-point following
CRRT treatment, the urine output level of neonates with AKI at any
stage was significantly improved (Table III and Fig. 2).
 | Table IIIComparison of urine output and serum
creatinine levels prior to and following CRRT treatment of AKI
neonates in 2 groups (x±s). |
Table III
Comparison of urine output and serum
creatinine levels prior to and following CRRT treatment of AKI
neonates in 2 groups (x±s).
| A, Urine output
(ml/kg/h). |
|---|
| Group | n | Prior to CRRT
treatment | CRRT treatment at
24 h | Following CRRT
treatment |
|---|
| AKI stage 0-1
group | 15 | 1.05±0.86 |
2.35±1.59b |
3.26±1.84c |
| AKI stage 2-3
group | 10 | 0.03±0.62 |
1.21±1.04a |
2.56±1.61c |
| F-value | | 13.786 | 4.008 | 0.965 |
| P-value | | 0.001 | 0.057 | 0.336 |
| B, Serum creatinine
(µmol/l) |
| Group | n | Prior to CRRT
treatment | CRRT treatment at
24 h | Following CRRT
treatment |
| AKI stage 0-1
group | 15 | 122±71 | 46±21b | 38±21b |
| AKI stage 2-3
group | 10 | 177±117 | 72±52c | 69±48b |
| F-value | | 2.215 | 3.013 | 5.005 |
| P-value | | 0.150 | 0.096 | 0.035 |
Efficacy evaluation of CRRT treatment
for AKI neonates
All 25 cases of AKI neonates were effectively
treated, and the indicators, such as renal function, urine volume,
blood electrolyte, acid-base balance, and hemodynamics were
significantly improved following CRRT treatment (Table II).
Safety evaluation of CRRT treatment
for AKI neonates
Intravenous catheterization in 25 neonates with AKI
was successfully completed. Thrombocytopenia was noted in 4 cases,
obstruction in 2 cases and hypotension in 2 cases. No hypothermia,
bleeding, thrombosis or infection occurred (data not shown).
Clinical outcomes of 25 neonates with
AKI
Among the 25 AKI newborns, 16 cases (14 cases of AKI
stage 0-1 and 2 cases of AKI stage 2-3) were cured and discharged.
The remaining 9 AKI neonates (3 cases of AKI stage 0-1 and 6 cases
of AKI stage 2-3) exhibited functional dysfunction in ≥4 organs,
deep coma, and multiple organ dysfunction syndrome (MODS) prior to
CRRT treatment (data not shown). These neonates survived the AKI
oliguria stage following CRRT treatment; however, they could not
recover due to neurological injury. Therefore, their families
consented to treatment discontinuation and these neonates succumbed
to AKI.
Discussion
AKI is a common and life-threatening complication in
critically ill neonates. Neonatal AKI is a clinical syndrome
characterized by an acute decrease of glomerular filtration rate in
a short time period caused by various factors, leading to the
disturbance of acid-base balance, disrupted electrolyte levels, and
accumulation of metabolic waste (11). Neonatal AKI can be caused by
various pathogenic factors prior to, during, and following delivery
(12). According to the nature and
location of kidney injury, the etiology of AKI can be divided into
three major categories including pre-renal AKI, renal AKI, and
post-renal AKI (13,14). The risk factors for neonatal AKI
include neonatal asphyxia, low Apgar score at birth, very low to
extremely low birth weight of neonates, cardiac arrest,
endotracheal intubation, maternal and prenatal medications, sepsis,
septic shock, nephrotoxic agents, extracorporeal membrane pulmonary
oxygenation, and cardiac surgery (15,16).
CRRT, also known as continuous blood purification
(CBP), is a method of continuous and slow removal of water and
solute (17). In recent years,
with the rapid development of the CRRT methodology, this treatment
has become an important type of critical care medicine, playing a
key role in the treatment of neonatal AKI and critically ill
neonates; this type of treatment can significantly improve the
efficacy and prognosis of neonatal AKI (18). In 1995, at the first international
academic conference of CRRT held in San Diego (USA), CRRT was
formally defined as ‘all blood purification technologies that can
continuously remove solutes and support the functions of organs’
(19).
The main principles of CRRT are dispersion,
convection, adhesion and adsorption. Dispersion can remove small
molecules, such as water, electrolytes, urea nitrogen, and
creatinine. Convection can clear the molecular materials found in
the serum, such as cytokines and inflammatory mediators (20). The successful application of blood
purification techniques in adult patients with AKI was first
reported in 1977. Following several years of investigation of the
application of CRRT in the treatment of AKI in adults and children,
Ronco et al (21) reported
the application of continuous arteriovenous hemofiltration (CAVH)
in 1985. CAVH is one of the models of CRRT. CAVH mainly uses the
difference in arterial venous pressure of the human body as the
driving force of extracorporeal circulation. Although it exhibits
the characteristics of self-limited ultrafiltration, it can
effectively reduce the risk of excessive ultrafiltration,
overspeed, and result in hypotension. Following these studies, a
new starting point for the treatment of neonatal AKI with CRRT was
highlighted.
At present, the application of CRRT in children's
diseases has been developed, and includes uremia, refractory
hyperkalemia, volume overload, severe or metabolic acidosis with
volume overload, some dialysis poisoning, some serious electrolyte
disorders and anuria AKI (22).
Studies have shown that there is an independent association between
the degree of fluid overload at the beginning of CRRT and adverse
outcomes, including increased mortality and duration of ECMO
support. Intervention before significant fluid overload may be a
clinical treatment target (23).
CRRT can accurately control neonatal blood volume
via continuous, slow, and gradual control, which maintains fluid
balance and hemodynamic stability (24). Neonates, notably premature infants,
exhibit reduced blood volume and difficulty in vascular
catheterization (25). Therefore,
the developers of the blood purification equipment had limited
power to develop specific CRRT filters for neonates, and the CRRT
method was restricted from newborns for a long time period
(26). With the development of
catheterization technology and the research and development of an
instrument suitable for neonatal CRRT, CRRT is more and more widely
used in neonatal diseases. A study has shown that CRRT is safe and
effective in the treatment of neonatal hereditary metabolic
diseases and hyperammonemia (27).
Successful treatment of neonatal AKI with CRRT has been reported,
and CRRT has been gradually applied to premature infants and
infants with low birth weight (28). In the present study, it was found
that the serum urea nitrogen and creatinine levels were
significantly decreased following 24 h of CRRT treatment in
neonates with AKI stage 0-1. At each time-point in the study (prior
to CBP treatment, 24 h following CBP treatment and following CRRT
treatment), serum creatinine levels of stage 2-3 newborns with AKI
were still significantly higher than those with stage 0-1 AKI, but
urine output was lower than those with stages 0-1 AKI. Therefore,
it was considered that the treatment time required to significantly
reduce serum creatinine levels and improved urine output in AKI
stage 2-3 neonates was significantly higher compared with that of
the AKI stage 0-1 group. If CRRT treatment is provided promptly in
cases with AKI stage 0-1, it can improve the kidney function
avoiding more persistent damage to the kidney. In the present
study, the efficacy of CRRT treatment in cases with AKI stage 0-1
was improved compared with that noted in the cases of AKI stage
2-3. Therefore, the present study demonstrated that the optimal
selection for the application of the CRRT treatment is to treat
neonates with AKI stage 0-1.
Lee and Cho (29)
retrospectively analyzed the neonatal cases treated by CRRT at the
Samsung Medical Center (Seoul, South Korea) from 2007 to 2014. The
medical center treated 34 AKI neonates with CRRT, with a birth
weight of 800-4,100 g. A total of 15 premature infants (GA 25-36
+6 weeks) and 19 full-term infants were included in that
study. The results indicated that CRRT was effective in treating
AKI caused by fluid overload. Following treatment, 20 patients
(58.8%) survived and 14 (41.2%) succumbed to AKI. The total number
of patient was 14 and included 12 premature infants and 2 full-term
infants. Therefore, neonatal or low birth weight should not be
contraindicated for CRRT. CRRT is feasible and effective for
neonates or infants with low birth weight; however, there are still
apparent risks for premature infants.
In 2016, a ‘precise CRRT consensus meta-analysis’
study clearly proposed that CRRT therapy should be initiated as
soon as possible when the metabolic and fluid management
requirements of the body exceed the capacity of the kidney
(30). This indicates that as long
as the body's metabolism and the fluid management exceed the
capacity of the kidneys, CRRT should be treated quickly even in
case of lack of renal injury. According to the neonatal KDIGO AKI
definition, AKI stage 0-1 includes the early-stage cases with AKI
and reversible renal injury, while AKI stage 2-3 corresponds to the
state of renal failure. The complications of CRRT are important
indices for safety evaluation. Certain complications in the
treatment of AKI neonates have been identified following CRRT
treatment, such as difficulty or failure of catheterization,
hypothermia, hypotension, pipe blockage, thrombosis, bleeding,
thrombocytopenia and bloodstream infection (31). In the present study, CRRT was used
to treat 4 patients complicated with thrombocytopenia, 2 patients
with pipe obstruction, and 2 patients with hypotension. No
hypothermia, bleeding, thrombosis, or bloodstream infection
occurred. The reason for hypotension may be volume-related factors,
such as extremely rapid dehydration. The causes of pipeline
blockage may be the slow blood flow rate and unsatisfactory
anticoagulation control. The causes of thrombocytopenia may include
sepsis, anticoagulants, disseminated intravascular coagulation,
bleeding, and the effects of the filter membranes. Adequate
puncture technique, timely and close monitoring, and strict aseptic
operation are the key factors required to reduce or prevent CRRT
complications (4). During the
treatment of CRRT, the coagulation function, blood gas analysis and
trace blood glucose were strictly monitored every 2-4 h. In
addition, liver and kidney function and blood electrolyte levels
were detected every 6-12 h. Therefore, it is suggested that during
treatment, children should be monitored, their vital signs closely
observed, and various indicators timely detected, such as the
balance of access volume, coagulation function, hemodynamics, blood
electrolytes, and blood glucose. The monitoring of these indices,
aims to avoid or reduce the complications of CRRT.
In the present study, 10 cases of severe asphyxia, 8
cases of neonatal septicemia and 7 cases of metabolic crisis of
genetic metabolic disease were found in 25 cases of AKI neonates
(15 of cases AKI stage 0-1 and 10 of cases AKI stage 2-3). A total
of 9 neonates (3 neonates of AKI stage 0-1 and 6 neonates of AKI
stages 2-3) had been in a deep coma prior to CRRT treatment.
Although they had passed the oliguria stage following CRRT
treatment, they could not recover due to neurological injury. Their
families consented in treatment discontinuation and consequently,
these neonates succumbed to AKI. Symons et al (32) retrospectively studied the CRRT data
of 85 children with a body weight <10 kg in 5 hospitals in the
United States. Their study included 13 cases with MODS and 12 cases
of sepsis. Among 16 cases, the lowest weight was 1.5 kg. The
results of their study indicated that the CRRT treatment effect and
prognosis of children with a weight of 3.0-10.0 kg were similar to
those of older children. Therefore, it was suggested, that the
initiation of CRRT treatment at the early stage of AKI may
significantly improve the clinical outcome and prognosis of AKI
infants with low body weight.
The present study contains certain limitations. A
relatively small sample number was included with only 25 neonatal
cases with AKI treated with CRRT. Moreover, the present study was a
single-center retrospective clinical study and provided
insufficient information in assessing the types of CRRT treatment
for their application in neonatal diseases and individualized CRRT
treatment. In the future, a large sample and multi-center clinical
study of CRRT treatment should be carried out to further explore
the application of individualized and precise CRRT treatment for
critically ill neonates.
In conclusion, CRRT is considered to be safe and
effective in the treatment of neonatal AKI. This method should be
an effective measure for the treatment of neonatal AKI. The optimal
stage of initiation of CRRT to treat newborns with AKI should be in
cases with AKI stage 0-1.
Acknowledgements
Not applicable.
Funding
Funding: This study was supported by the Special Medical
Innovation Research Project of Shanghai ‘Scientific and
Technological Innovation Action Plan’ in 2020 (grant no.
20Y11907000).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XZ, XG and CC made substantial contributions to the
conception and design of the study, acquisition of data, and
analysis and interpretation of data. WH and NL participated in data
collection and analysis. CC and XG were involved in drafting the
manuscript and revising it critically for important intellectual
content. WH, NL, XG and CC revised the manuscript and provided
final approval of the version to be published. XG and CC confirm
the authenticity of all the raw data. All authors read and approved
the final manuscript and agree to be accountable for all aspects of
the work in ensuring that questions related to the accuracy or
integrity of any part of the work are appropriately investigated
and resolved.
Ethics approval and consent to
participate
The study protocol and amendments were reviewed and
approved by the Institutional Review Board, in accordance with the
ethical principles of the Declaration of Helsinki. This study was
approved (approval no. 2012C001-F01) by the Ethics Committee of
Shanghai Children's Hospital, School of Medicine, Shanghai Jiao
Tong University. All parents of AKI neonates provided written
informed consent before enrollment.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Kavanaugh KJ, Jetton JG and Kent AL:
Neonatal acute kidney injury: Understanding of the impact on the
smallest patients. Crit Care Clin. 37:349–363. 2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Jetton JG, Boohaker LJ, Sethi SK, Wazir S,
Rohatgi S, Soranno DE, Chishti AS, Woroniecki R, Mammen C, Swanson
JR, et al: Incidence and outcomes of neonatal acute kidney injury
(AWAKEN): A multicentre, multinational, observational cohort study.
Lancet Child Adolesc Health. 1:184–194. 2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Gallo D, de Bijl-Marcus KA, Alderliesten
T, Lilien M and Groenendaal F: Early acute kidney injury in preterm
and term neonates: Incidence, outcome, and associated clinical
features. Neonatology. 118:174–179. 2021.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Diane Mok TY, Tseng MH, Chiang MC, Lin JL,
Chu SM, Hsu JF and Lien R: Renal replacement therapy in the
neonatal intensive care unit. Pediatr Neonatol. 59:474–480.
2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Erkol Tuncer GH, Ekim M, Okulu E, Atasay B
and Kendirli T: Continuous renal replacement therapy in critically
ill children: single-center experience. Turk J Med Sci. 51:188–194.
2021.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Khwaja A: KDIGO clinical practice
guidelines for acute kidney injury. Nephron Clin Pract.
120:c179–c184. 2012.PubMed/NCBI View Article : Google Scholar
|
|
7
|
SooHoo MM, Patel SS, Jaggers J, Faubel S
and Gist KM: Acute kidney injury defined by fluid corrected
creatinine in neonates after the norwood procedure. World J Pediatr
Congenit Heart Surg. 9:513–521. 2018.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Yoon BR, Leem AY, Park MS, Kim YS and
Chung KS: Optimal timing of initiating continuous renal replacement
therapy in septic shock patients with acute kidney injury. Sci Rep.
9(11981)2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Suzuki J, Ohnuma T, Sanayama H, Ito K,
Fujiwara T, Yamada H, Lefor AK and Sanui M: The optimal timing of
continuous renal replacement therapy according to the modified
RIFLE classification in critically ill patients with acute kidney
injury: A retrospective observational study. Renal Replacement
Therapy. 3(30)2017.
|
|
10
|
Roman-Filip C, Catană MG, Bereanu A,
Lăzăroae A, Gligor F and Sava M: Therapeutic plasma exchange and
double filtration plasmapheresis in severe neuroimmune disorders.
Acta Clin Croat. 58:621–626. 2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Uber AM and Sutherland SM: Acute kidney
injury in hospitalized children: Consequences and outcomes. Pediatr
Nephrol. 35:213–220. 2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Mian AN, Askenazi DJ and Mhanna MJ:
Therapeutic options for neonatal acute kidney injury (AKI). Curr
Treat Opt Pediatrics. 2:1–13. 2016.
|
|
13
|
Selewski DT, Charlton JR, Jetton JG,
Guillet R, Mhanna MJ, Askenazi DJ and Kent AL: Neonatal Acute
kidney injury. Pediatrics. 136:e463–e473. 2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Perico N, Askenazi D, Cortinovis M and
Remuzzi G: Maternal and environmental risk factors for neonatal AKI
and its long-term consequences. Nat Rev Nephrol. 14:688–703.
2018.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Safina AI, Daminova MA and Abdullina GA:
Acute kidney injury in neonatal intensive care: Medicines involved.
Int J Risk Saf Med. 27 (Suppl 1):S9–S10. 2015.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Askenazi DJ: AWAKEN-Ing a new frontier in
neonatal nephrology. Front Pediatr. 8(21)2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Goldstein SL: Pediatric continuous renal
replacement therapy for ‘40 years of continuous renal replacement
therapy’. Contrib Nephrol. 194:146–154. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Cho AY, Yoon HJ, Lee KY and Sun IO:
Clinical characteristics of sepsis-induced acute kidney injury in
patients undergoing continuous renal replacement therapy. Ren Fail.
40:403–409. 2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Continuous renal replacement therapies,
international conference. San Diego, California, November 8-10,
1995. Abstracts. Blood Purif. 13:385–402. 1995.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Ricci Z and Goldstein SL: Pediatric
continuous renal replacement therapy. Contrib Nephrol. 187:121–30.
2016.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Ronco C, Ricci Z and Goldstein SL:
(R)evolution in the management of acute kidney injury in newborns.
Am J Kidney Dis. 66:206–211. 2015.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Kes P and Basić Jukić N: Acute kidney
injury in the intensive care unit. Bosn J Basic Med Sci 10 Suppl. 1
(Suppl 1):S8–S12. 2010.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Gorga SM, Sahay RD, Askenazi DJ, Bridges
BC, Cooper DS, Paden ML, Zappitelli M, Gist KM, Gien J, Basu RK, et
al: Fluid overload and fluid removal in pediatric patients on
extracorporeal membrane oxygenation requiring continuous renal
replacement therapy: A multicenter retrospective cohort study.
Pediatr Nephrol. 35:871–882. 2020.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Ronco C, Garzotto F, Brendolan A, Zanella
M, Bellettato M, Vedovato S, Chiarenza F, Ricci Z and Goldstein SL:
Continuous renal replacement therapy in neonates and small infants:
Development and first-in-human use of a miniaturised machine
(CARPEDIEM). Lancet. 383:1807–1813. 2014.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Noh ES, Kim HH, Kim HS, Han YS, Yang M,
Ahn SY, Sung SI, Chang YS and Park WS: Continuous renal replacement
therapy in preterm infants. Yonsei Med J. 60:984–991.
2019.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Garzotto F, Vidal E, Ricci Z, Paglialonga
F, Giordano M, Laforgia N, Peruzzi L, Bellettato M, Murer L and
Ronco C: Continuous kidney replacement therapy in critically ill
neonates and infants: A retrospective analysis of clinical results
with a dedicated device. Pediatr Nephrol. 35:1699–1705.
2020.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Hu Y, Peng XM and Xiao ZH: Application of
continuous renal replacement therapy in the treatment of neonates
with inherited metabolic diseases. Zhongguo Dang Dai Er Ke Za Zhi.
23:488–493. 2021.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
28
|
Pedersen O, Jepsen SB and Toft P:
Continuous renal replacement therapy for critically ill infants and
children. Dan Med J. 59(A4385)2012.PubMed/NCBI
|
|
29
|
Lee ST and Cho H: Fluid overload and
outcomes in neonates receiving continuous renal replacement
therapy. Pediatr Nephrol. 31:2145–2152. 2016.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Kellum JA and Ronco C: The 17th acute
disease quality initiative international consensus conference:
Introducing precision renal replacement therapy. Blood Purif.
42:221–223. 2016.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Riley AA, Watson M, Smith C, Guffey D,
Minard CG, Currier H and Akcan Arikan A: Pediatric continuous renal
replacement therapy: Have practice changes changed outcomes? A
large single-center ten-year retrospective evaluation. BMC Nephrol.
19(268)2018.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Symons JM, Brophy PD, Gregory MJ, McAfee
N, Somers MJ, Bunchman TE and Goldstein SL: Continuous renal
replacement therapy in children up to 10 kg. Am J Kidney Dis.
41:984–989. 2003.PubMed/NCBI View Article : Google Scholar
|