Introduction
Leiomyomas are benign tumors originating from smooth
muscle cells (1). The prevalence
of uterine leiomyomas in women varies between 4.5-68.6%, depending
on racial and ethnic characteristics. However, because diagnosis is
mainly sought in symptomatic patients, this condition is arguably
more prevalent than clinical records and published data indicate.
In fact, uterine leiomyomas are the most commonly occurring benign
tumors in women of reproductive age. They tend to exert major
negative impact on morbidity and quality of life and are the most
frequent indication for hysterectomy (2).
Extrauterine leiomyomas tend to be extremely rare
and challenging to diagnose. Cases of ovarian, bladder, or urethral
leiomyomas have been reported in the literature. Other rare cases
feature unusual growth patterns, including benign metastasizing
leiomyoma, diffuse peritoneal leiomyomatosis, parasitic leiomyomas,
retroperitoneal leiomyoma, cotyledonoid dissecting leiomyoma and
intravenous leiomyomatosis (1).
Of these, intravenous leiomyomatosis is known to
behave in a particularly noteworthy way. Although benign, tumors of
this type have the potential to become clinically aggressive, with
intraluminal development in the intrauterine veins, spreading also
to the systemic veins. So far, ~300 cases have been described in
the scientific literature available in English, and most were
diagnosed by cardiologists even if cardiac involvement is extremely
rare.
Such cases illustrate the clinical importance of
improving the diagnosis of uterine myomas, especially since
successful treatment requires complete excision and, also, the rate
of recurrence seems high. The fact that these tumors tend to be
hormone sensitive offers novel treatment options (3,4). In
0.25-0.41% of women with uterine leiomyomas, the condition can
affect the venous system and advance into the inferior vena cava.
Occasionally, the spread can reach the right chambers of the heart,
causing heart failure or cardiogenic shock (5,6).
The patient's consent for publication and Cuza-Voda
Clinical Hospital of Obstetrics and Gynecology ethical committee
approval (no. 14183/25.10.2022) was obtained.
Case presentation
A 44-year-old woman was admitted to the gynecology
department of the Cuza-Voda Clinical Hospital of Obstetrics and
Gynecology complaining of aggravation of her symptoms over the
previous six months. She had initially presented with menorrhagia
two years before, when clinical and paraclinical evaluation had
revealed a 5-cm intramural uterine leiomyoma. Due to the Covid-19
pandemic, the patient was too frightened to seek timely medical
assistance. When she did present, she refused the recommended
hospital admission and surgical intervention. She later returned
for re-evaluation after completing her Covid-19 vaccination scheme.
By then, she had already been experiencing menometrorrhagia for six
months, during which time her treatment consisting of progestins
failed to produce significant results in terms of controlling the
bleeding. The patient had no relevant medical history besides two
uneventful cesarean section deliveries and there was no family
history of uterine fibroids.
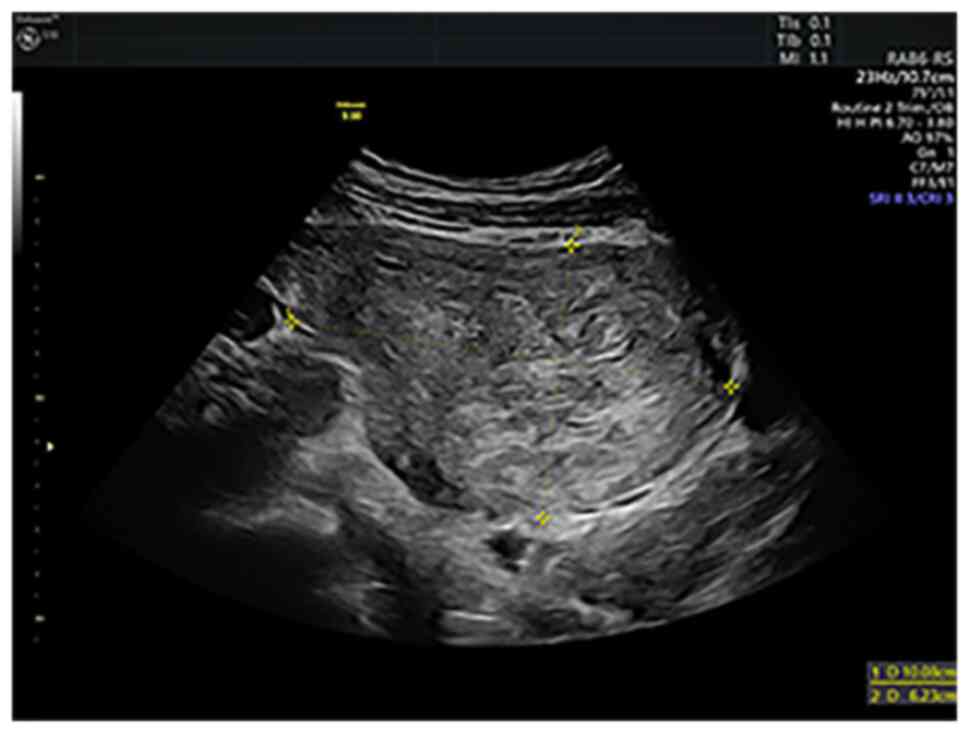
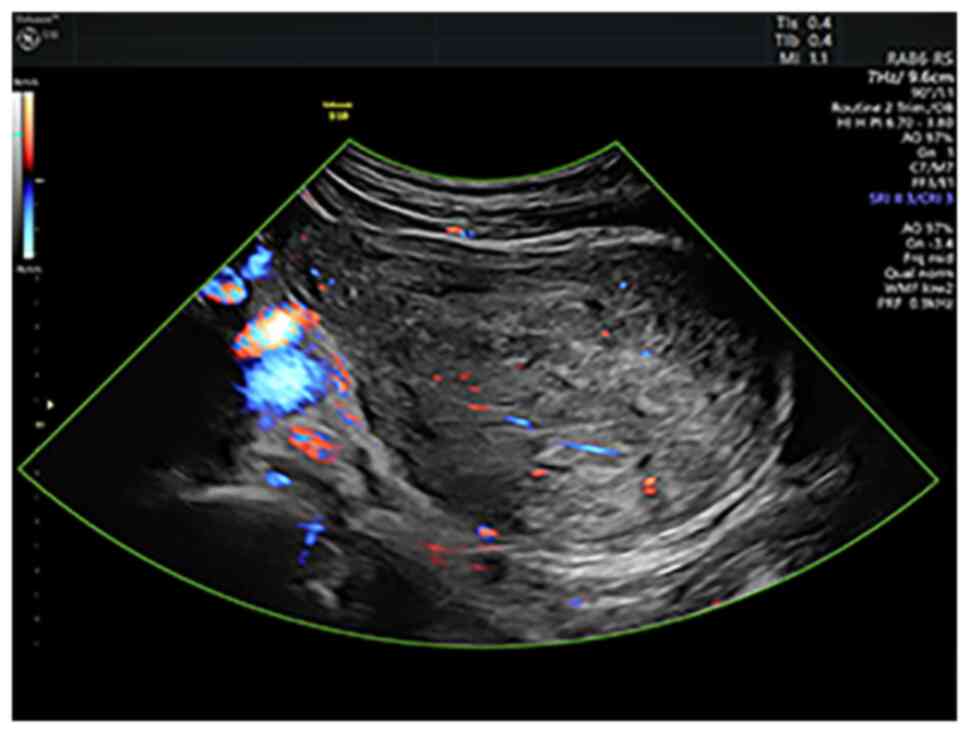
Upon admission, the ultrasound examination revealed
a 10-cm anterior intramural and subserosal uterine leiomyoma
without unusual features, but there were other ultrasound findings
suggestive of adenomyosis in the posterior uterine wall (Figs. 1 and 2). After counseling, the patient opted
for a total abdominal hysterectomy with bilateral salpingectomy and
conservation of both ovaries to prevent the effects of estrogenic
privation. Due to the localization of the myoma towards the broad
ligament, its dimensions, and the suspected adenomyosis, it was
decided to proceed with open surgery. The intervention itself was
undertaken without difficulties and complications. The patient made
a favorable postsurgical recovery and was discharged after 4 days
of hospitalization.
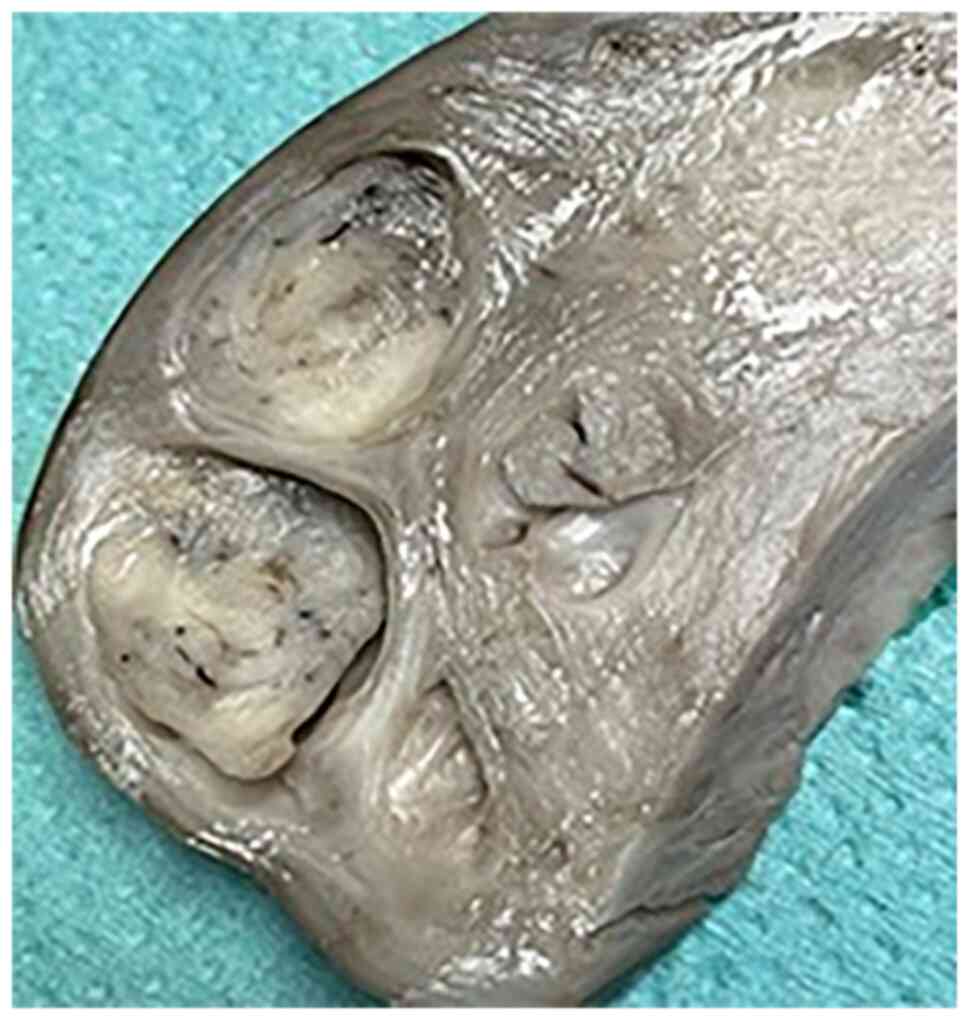
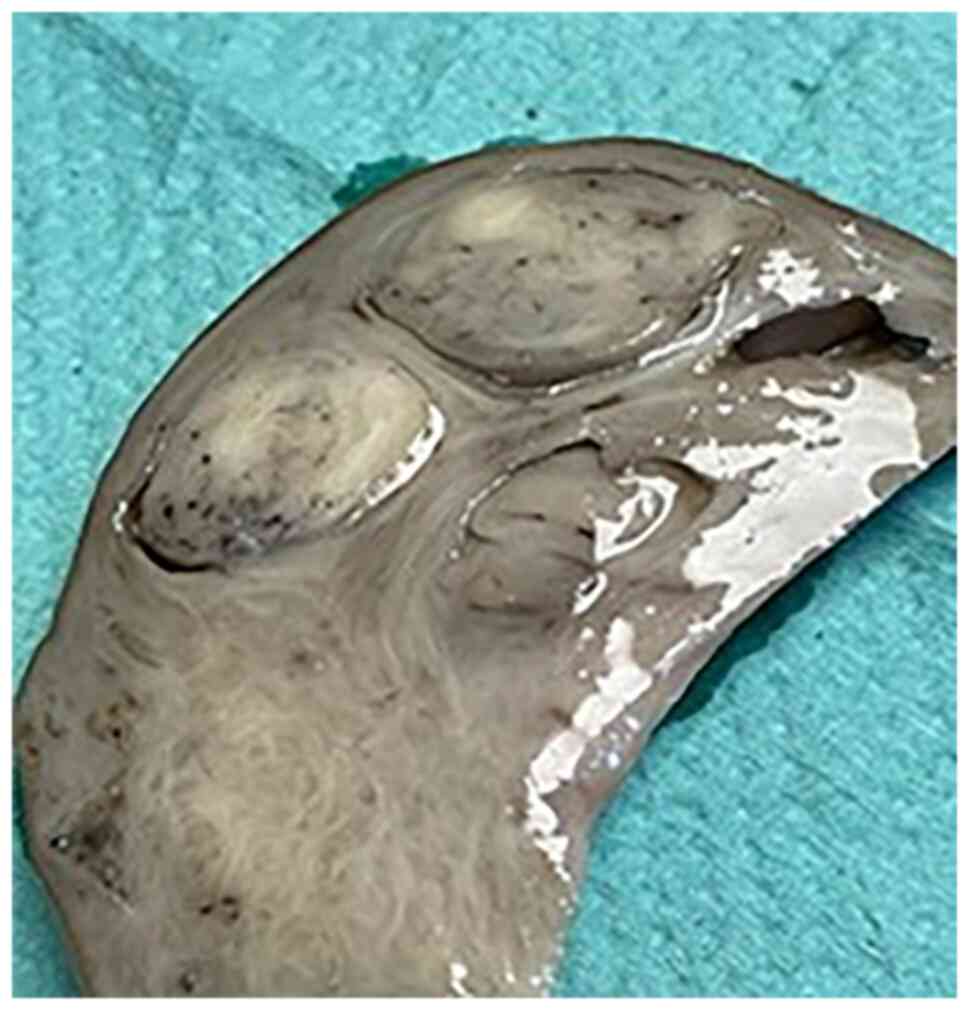
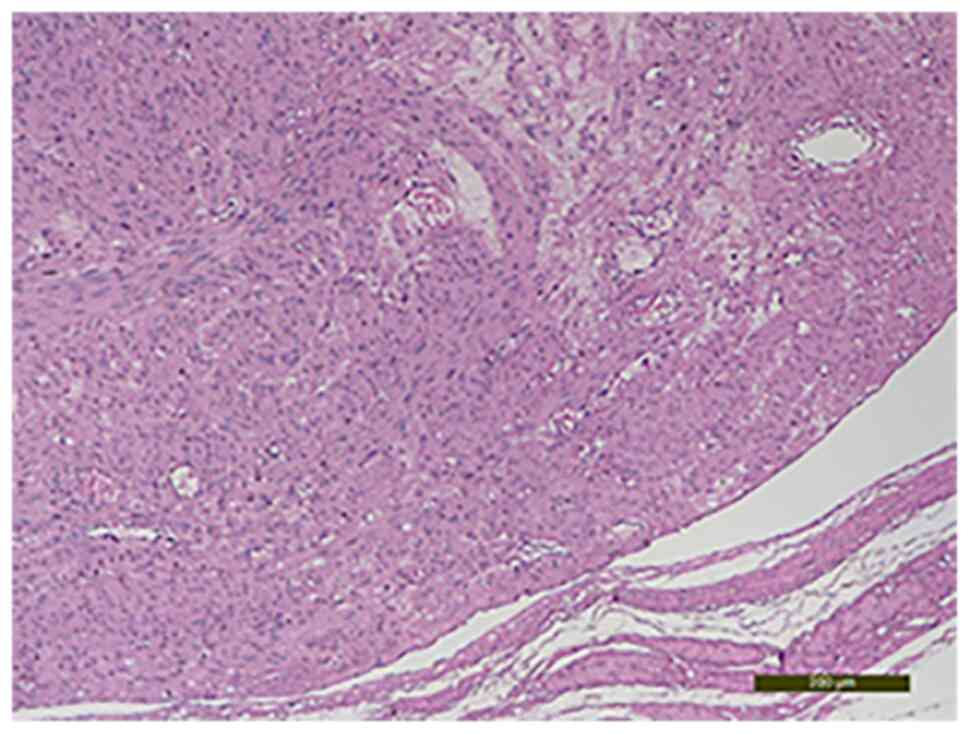
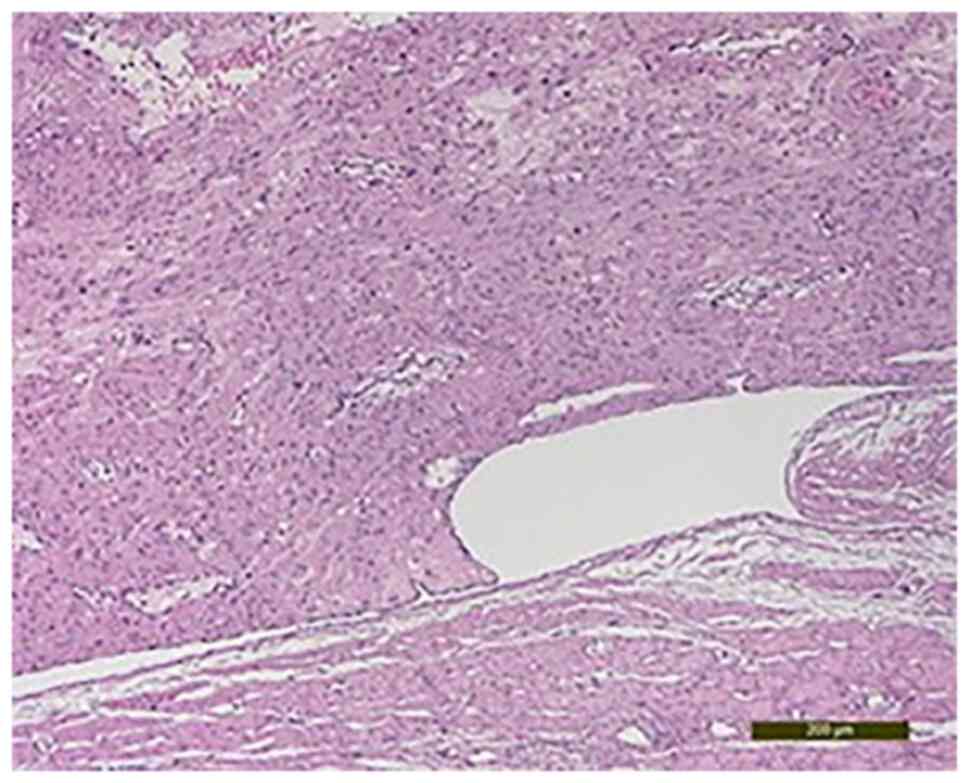
The gross pathological examination revealed a 10-cm
uterine mass with intramural proliferation and fascicular
architecture (Figs. 3 and 4).
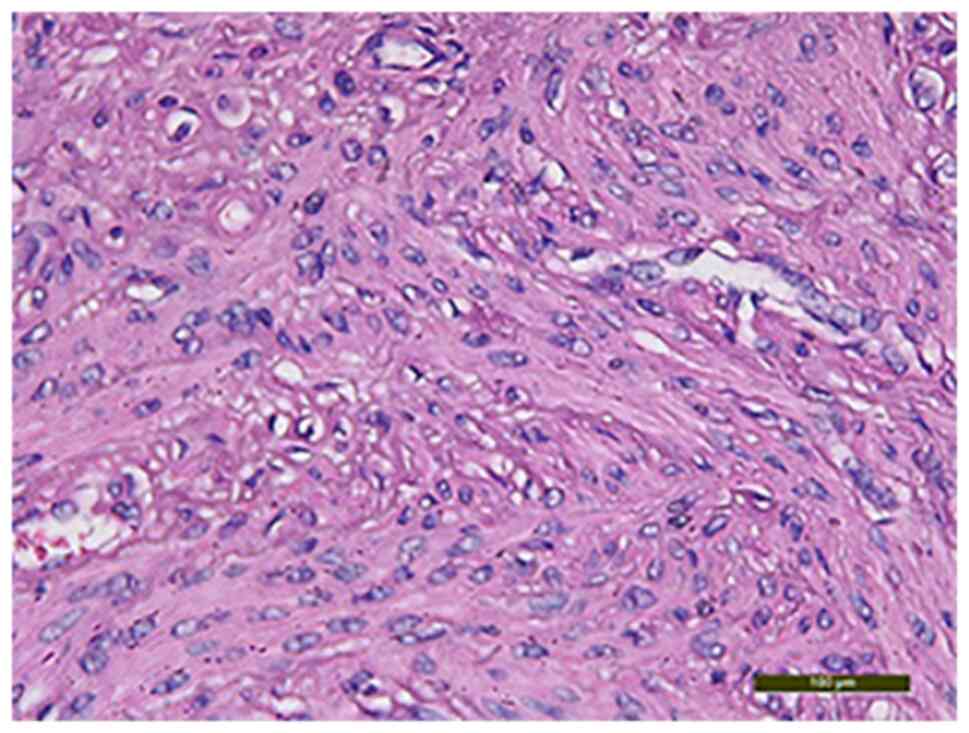
The histological examination with hematoxylin-eosin
staining showed fusiform cells that had moderate nuclear atypia,
rare mitosis, areas of sclerosis and hyalinosis, interstitial edema
and degenerative modifications. The specimen was processed
according to standard histological technique and the slides were
stained with the conventional hematoxylin and eosin staining (HE)
method (7). Briefly, the uterine
mass tissues was fixed in formaldehyde (neutral buffered formalin
10%) for 24 h at room temperature (24˚C) and then the specimen was
dehydrated in alcohol, cleared in xylene and embedded in paraffin.
Clarification was achieved by passing the fragments through three
xylene baths to remove alcohol from the tissues, for 4-6 h. The
impregnation with paraffin was achieved by placing the tissue
fragments in three paraffin baths for a minimum of 24 h at a
temperature of 56˚C. The slides obtained were placed in a
thermostat at 58˚C for 24 h and later stained using HE staining.
The paraffin blocks were sectioned at 4 µm. The tumor with
intravascular growth featured uniform spindle-shaped smooth muscle
cells (Fig. 5, Fig. 6 and Fig. 7). Also, the presence of adenomyosis
was confirmed. This diagnosis was not suspected prior to the
surgery and it was an unusual histopathological finding.
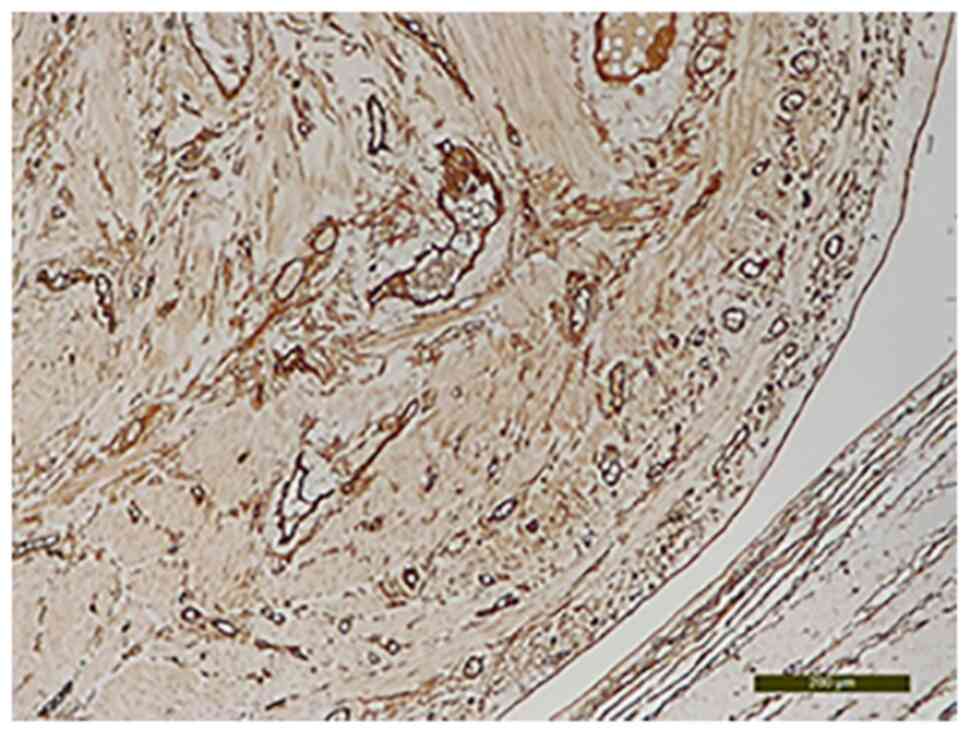
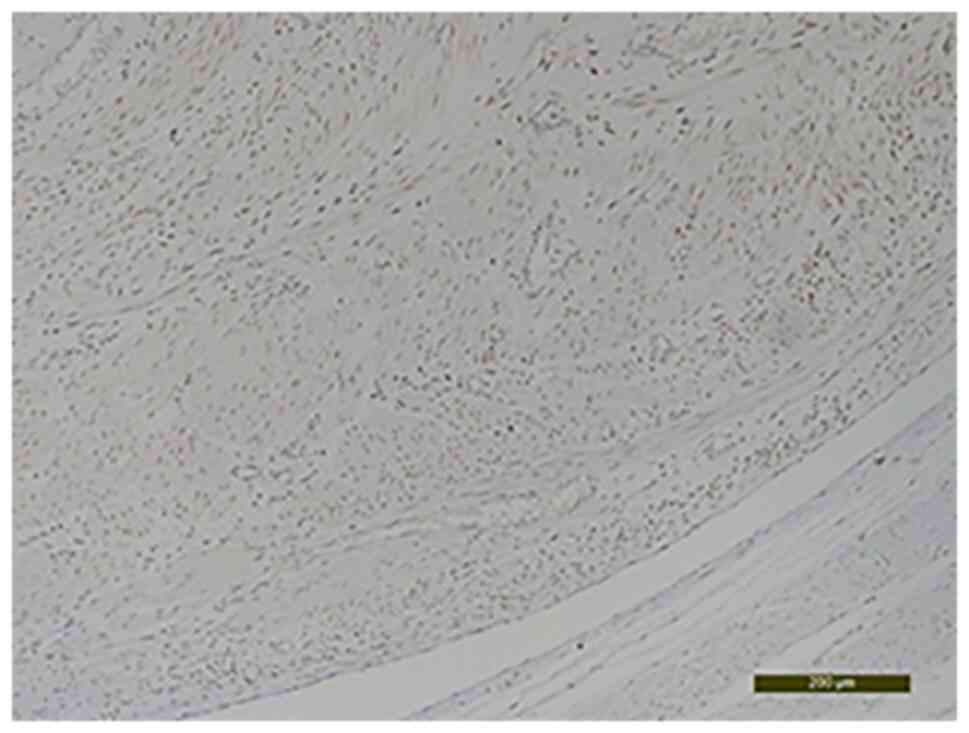
The CD34 analysis established its intravascular
origin (Fig. 8). The
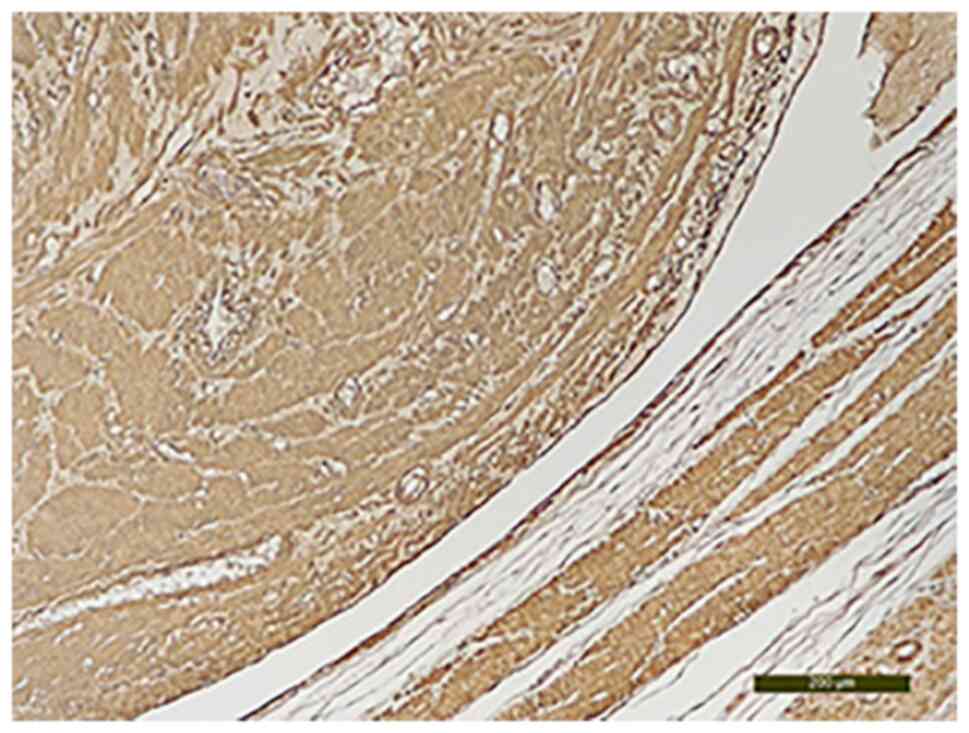
immunohistochemical evaluation revealed that the area of the tumor
that had intravascular development was positive for estrogen
receptors (Fig. 9) and for the
smooth muscle actin marker (Fig.
10). Intravascular origin was evaluated using a CD34 marker for
endothelial cells. Incubation was with the primary antibody
(anti-CD34) by applying the optimal dilution in a humid chamber at
4˚C, overnight. The anatomopathological diagnosis was confirmed by
two experienced pathologists.
After this diagnosis was established, the patient
underwent an abdominal-pelvic magnetic resonance imaging (MRI) and
a pulmonary computed tomography (CT) scan to exclude the presence
of other intravascular tumors. She had no other intravascular
leiomyomatosis lesions, so she did not need to continue with any
other treatment. However, due to the high risk of recurrence,
follow-up evaluations were scheduled at 3-month intervals and
pelvic MRIs at 6 months. At the date of the article submission no
ultrasound or MRI signs of relapse were detected and the patient
had no complaints.
An noteworthy aspect of this case is the
concomitance of a uterine leiomyoma with areas of intravascular
leiomyomatosis and adenomyosis.
Discussion
This is the fourth such case ever reported in
Romania; the first two were described by teams from cardiology
departments, one in 2010(8),
another in 2018(9) and the third
was published in 2019 by peers from a gynecology department
(10). According to other studies
in literature, the age of the patients with this pathology ranges
from 21-80, and the mean age at onset is ~47, so the 44-year-old
patient in the present study fits this general age profile
(11).
‘Cuza-Voda’ Clinical Hospital of Obstetrics and
Gynecology from Iasi s a tertiary referral center where numerous
surgical interventions address uterine leiomyomatosis. Between 2013
and 2019, for instance, 2,196 patients were treated by means of
open hysterectomy and 243 with open myomectomy, while another 188
patients underwent laparoscopic myomectomy and 159 had
hysteroscopic myomectomy. The last 3 years were excluded from this
analysis due to the impact of the Covid-19 pandemic on the typical
schedule of gynecological surgical interventions. The case hereby
described is the first uterine leiomyoma with intravascular
leiomyomatosis that we encountered from 2013 until 2021(12).
Unusual leiomyoma localizations and growth patterns
pose serious diagnostic difficulties and require different
management approaches. Early stage disease has no particularities
in which concerns the clinical manifestations or the ultrasound
features. Occasionally, these tumors may resemble a malignancy but
they are almost always benign, so their clinical and paraclinical
features ought to be carefully investigated for accurate diagnosis
and optimal management (13).
The etiopathogenesis of these less common leiomyomas
remains unknown. There are two hypotheses that attempt to explain
this interesting process. For one, intravascular tumors are thought
to develop secondary to the penetration inside the vascular lumen
of smooth muscle cells from the venous wall. Alternatively, it may
be that the vascular lumen is penetrated by smooth muscle cells
derived from uterine leiomyoma (14). The case described seems to favor
the second hypothesis, as the 10-cm uterine fibroid presented
features of intravascular invasion with intravascular
leiomyomatosis.
Du et al (15) discovered that intravenous
leiomyomatosis appears to be associated with uterine leiomyoma in
~40% of cases, with adenomyosis in 11% of cases and with both types
of lesions in almost 28% of cases. The frequent association with
adenomyosis lesions suggests that the second theory about
intravenous leiomyomatosis origin is more likely. They appear to
share some common etiopathogenic mechanisms.
By evaluating the progression of the tumors prior to
surgery, Ma et al (16)
proposed a classification of the disease into four stages. In the
first stage the tumors invade the vascular lumen, but they are
limited to the pelvic vessels. In the second stage of evolution the
tumors reach the vessels form the abdominal cavity without
affecting the renal vein. In the third stage of disease progression
the tumors reach the renal vein, the inferior vena cava and the
right atrium but without any lesions being detected in the
pulmonary artery. A stage four patient has intravascular tumors in
the pulmonary arteries and/or lungs with intravascular
leiomyomatosis lesions.
Despite the benign nature of intravascular
leiomyomatosis, its evolution may be aggressive and lead to serious
complications. In particular, there are several cases with
intracardiac development reported in the literature which posed
substantial surgical difficulties, as well as cases in which the
tumors determined cardiac insufficiency, cardiac arrest, and even
secondary pulmonary complications, but the data are scarce about
early-stage disease (5,6).
The present case report reflected the negative
impact of the Covid-19 outbreak on patients with gynecological
surgical pathologies in Romania. Not only did the official Covid-19
preventive measures limit access to health care to a certain
degree, but also fear of infection compelled patients to postpone
necessary surgical interventions. Given the potentially aggressive
behavior of this type of smooth muscle tumor, the outcome for our
patient could have been seriously affected by her decision to delay
having the surgical intervention for as long as two years. In this
time, the symptoms worsened, the uterine leiomyoma doubled in size
and it possibly permitted the appearance of areas of intravascular
leiomyomatosis.
The incidence of this pathology seems to be rising.
In literature, this ascendant trend is justified either by more
accurate means to detected it, due to diagnosis techniques
evolution and improvement, or by the blooming of minimally invasive
surgery with morcellation for uterine fibroids, that is assumed to
be one of the potential causes for this rare and intriguing
pathology (17).
The gold standard treatment for this pathology is
considered by many to be hysterectomy with bilateral
salpingoophorectomy (3,4). In literature, the recurrence rate of
these tumors is estimated at ~30%, or even higher in cases with
incomplete surgical resection of the tumors (1). This suggests that patients diagnosed
with this disease need further postoperative follow-up using either
MRI or CT imaging to detect recurrences. The optimal schedule for
the postoperative follow-up has not yet been standardized mostly
because cases are rare and there are a number of unique aspects to
consider. For instance, it was concluded that our patient would
need close follow-up due to her decision to preserve both ovaries.
As such, a follow up plan was established including gynecological
check-ups at 3-month intervals and additional abdominal-pelvic MRI
investigations at every other 6 months check-up.
In conclusion, clinicians must know about these
unusual types of fibroids because their diagnosis is easy to miss
and patients require extensive imagistic exploration to accurately
stage the disease and organize effective management. The high
recurrence rates justify the need for continued imagistic
monitoring of these patients after surgery. Also, the
malignant-like behavior of such tumors further complicates their
management. Last but not least, the incidence of this pathology
appears to be rising, which may be due to more accurate diagnosis
and investigative means, but also to increase risk factors for the
occurrence of hormone-dependent tumors.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
CS, AU, DM were involved in the conception of the
study and revised the manuscript for important intellectual
content. LL performed the anatomopathological and
immunohistochemical examination. BS, SS, IB and MT performed the
data collection and wrote the manuscript. BS, SS, IB and MT confirm
the authenticity of all the raw data. The surgery was performed by
CS with the help of BS and SS. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
Ethics approval was obtained from the Cuza-Voda
Clinical Hospital of Obstetrics and Gynecology ethical committee
approval (no. 14183/25.10.2022).
Patient consent for publication
Patient consent was obtained.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Fasih N, Prasad Shanbhogue AK, Macdonald
DB, Fraser-Hill MA, Papadatos D, Kielar AZ, Doherty GP, Walsh C,
McInnes M and Atri M: Leiomyomas beyond the uterus: Unusual
locations, rare manifestations. Radiographics. 28:1931–1948.
2008.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Giuliani E, As-Sanie S and Marsh EE:
Epidemiology and management of uterine fibroids. Int J Gynecol
Obstet. 149:3–9. 2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Kocica MJ, Vranes MR, Kostic D,
Kovacevic-Kostic N, Lackovic V, Bozic-Mihajlovic V, Velinovic MM,
Mikic Adj and Dimitrijevic-Kalezic N: Intravenous leiomyomatosis
with extension to the heart: Rare or underestimated? J Thorac
Cardiovasc Surg. 130:1724–1726. 2005.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Low HY, Zhao Y, Huang KS, Shen HP, Wu PJ
and Tseng CJ: Intravenous leiomyomatosis of the uterus: A
clinicopathological analysis of nine cases and literature review.
Taiwan J Obstet Gynecol. 56:362–365. 2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Nakai G, Maeda K, Yamamoto K, Yamada T,
Hirose Y, Terai Y, Ohmichi M, Katsumata T and Narumi Y: Uterine
intravenous leiomyomatosis with cardiac extension: Radiologic
assessment with surgical and pathologic correlation. Case Rep
Obstet Gynecol. 2015(576743)2015.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Xu J, Wei M, Miao Q, Zhu B, Yu C and Huang
Y: Perioperative management of intracardiac leiomyomatosis: An
observational cohort study. Medicine (Baltimore).
96(e7522)2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Feldman AT and Wolfe D: Tissue processing
and hematoxylin and eosin staining. Methods Mol Biol. 1180:31–43.
2014.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Galajda Z, Copotoiu C, Suciu H, Tint D,
Glasz T and Deac R: The diagnosis, morphological particularities,
and surgical technique in a case of intravascular leiomyoma
extended to the right heart chambers. J Vasc Surg. 51:1000–1002.
2010.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Brunei-Rusu A: Atypical uterine
leiomyomatosis-case report. Ginecologia. 23(46)2019.
|
|
10
|
Dregoesc IM, Bãlãnescu ŞM, Marc MC and
Iancu AC: Intravascular leiomyoma with intracardiac extension
associated with hepatorenal polycystic disease. Anatol J Cardiol.
20:246–248. 2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Carr RJ, Hui P and Buza N: Intravenous
leiomyomatosis revisited: An experience of 14 cases at a single
medical center. Int J Gynecol Pathol. 34:169–176. 2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Matasariu DR, Ursache A, Himiniuc L, Toma
B, Boiculese VL, Grigore M and Dumitrascu I: Research on myoma in
Northeastern Romania and socio-medical outcomes. Exp Ther Med.
23(30)2022.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Cho JH and Kim SS: Peritoneal
carcinomatosis and its mimics: Review of CT findings for
differential diagnosis. J Belg Soc Radiol. 104(8)2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Li B, Chen X, Chu YD, Li RY, Li WD and Ni
YM: Intracardiac leiomyomatosis: A comprehensive analysis of 194
cases. Interact Cardiovasc Thorac Surg. 17:132–138. 2013.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Du J, Zhao X, Guo D, Li H and Sun B:
Intravenous leiomyomatosis of the uterus: A clinicopathologic study
of 18 cases, with emphasis on early diagnosis and appropriate
treatment strategies. Hum Pathol. 42:1240–1246. 2011.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Ma G, Miao Q, Liu X, Zhang C, Liu J, Zheng
Y, Shao J, Cheng N, Du S, Hu Z, et al: Different surgical
strategies of patients with intravenous leiomyomatosis. Medicine
(Baltimore). 95(e4902)2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Liu X, Hu Y, Chen L and Zhou Q:
Disseminated peritoneal leiomyomatosis: A case report and review of
the literature. J Int Med Res. 49(3000605211033194)2021.PubMed/NCBI View Article : Google Scholar
|