Introduction
Endometrial cancer (EC) is one of the commonest
gynecological malignancies and has the sixth-highest incidence of
cancer in women worldwide (1).
Despite significant advances in the diagnosis and treatment, the
prognosis for patients with advanced and recurring EC remains poor
(2). Exploring the molecular
mechanisms of EC to develop new treatment strategies is critical
for improving the prognosis of patients.
The tumor microenvironment (TME) is linked to the
tumorigenesis and progression of the disease (3). The TME consists primarily of cancer
cells, cancer-associated fibroblasts, immune cells and non-cellular
components, with macrophages being the most abundant immune cells
(4). M2 macrophages, also known as
tumor-associated macrophages (TAMs), are closely linked to the
development and progression of various cancers (5,6).
TAMs secrete a variety of mediators, such as cytokines and
chemokines, to suppress anti-tumor immune responses and promote
cancer cell proliferation, invasion and spreading (7,8).
Studies show that tumor cells direct macrophages toward the M2
phenotype to promote malignant progression (9). Inhibiting M2 macrophage polarization
can slow the progression of cancer (10). The underlying mechanisms by which
cancer cells control macrophage M2 polarization, however, remain to
be elucidated.
Insulin-like growth factor binding protein-related
protein 1 (IGFBP-rP1, also known as IGFBP7) is a secreted protein
of the insulin-like growth factor binding protein superfamily that
has been linked to insulin resistance (11). IGFBP-rP1 appears to be a tumor
suppressor in a variety of cancers, including colorectal cancer and
breast cancer (11,12). A previous study has linked elevated
IGFBP-rP1 levels to a lower risk of EC (13). Hu et al (14) proposed that IGFBP-rP1 plays an
active role in promoting the interaction between cancer cells and
TME, which could explain why cancer cells adhere, invade and
migrate. The potential mechanism of IGFBP-rP1 in EC, however,
remains unknown.
The present study aimed to look at the effect of
IGFBP-rP1 on EC cell proliferation and apoptosis, as well at the
role of IGFBP-rP1 in the formation of M2 TAMs.
Materials and methods
Cell cultures
Human endometrial cancer cell lines (Ishikawa,
HEC-1A, RL95-2, HEC-1B and AN3CA) and human mononuclear cells
(THP-1) were purchased from Procell Life Science & Technology
Co., Ltd. All cells were cultured in Dulbecco's modified Eagle's
medium (Gibco; Thermo Fisher Scientific, Inc.) containing 10% fetal
bovine serum (FBS; Gibco; Thermo Fisher Scientific, Inc.), 100
µg/ml penicillin and 100 µg/ml streptomycin (HyClone; Cytiva) and
cultured at 37˚C in 5% CO2.
Reverse transcription-quantitative
(RT-q) PCR
Total RNA was extracted from EC cells
(1x105 cells/well) according to the manufacturer's
protocols using the TRIzol® kit (Thermo Fisher
Scientific, Inc.). A cDNA synthesis kit (Thermo Fisher Scientific,
Inc.) was used to transcribe total RNA into cDNA in accordance with
the manufacturer's protocols. The SYBR Green One-step RT-PCR Master
Mix (Thermo Fisher Scientific, Inc.) and 7900 real-time PCR system
(Thermo Fisher Scientific, Inc.) was used for RT-qPCR. The
following thermocycling conditions were used for qPCR: Initial
pre-denaturation at 95˚C for 120 sec; 40 cycles of denaturation at
95˚C for 15 sec, annealing at 58˚C for 30 sec and elongation at
72˚C for 30 sec. The primers used were: IGFBP-rP1, Forward 5'-AGC
TGT GAG GTC ATC GGA AT-3', Reverse 5'-CAG CAC CCA GCC AGT TAC
TT-3'; GAPDH, Forward 5'-GGA GCG AGA TCC CTC CAA AAT-3', Reverse
5'-GGC TGT TGT CAT ACT TCT CAT GG-3'. The 2-ΔΔCq method
was used to calculate fold changes in the gene expression
normalized to GAPDH (15). At
least three replicate wells were performed for each group.
Western blot analysis
RIPA lysis buffer (Beyotime Institute of
Biotechnology) was used to extract total protein from EC cells. The
protein concentration of the supernatants was assessed using the
Bradford Protein Assay kit (Beyotime Institute of Biotechnology).
Equal amounts of total protein (30 µg) was then separated on 10%
SDS-PAGE before being transferred to PVDF membranes. TBS-Tween-20
(Beyotime Institute of Biotechnology) containing 5% skimmed milk
powder was used to block the membranes for 1 h at 37˚C, which were
then incubated with primary antibodies overnight at 4˚C. The
primary antibodies were as follows: anti-IGFBP-rP1 (cat. no.
MAB1334; 1:500; R&D Systems), anti-AKT (cat. no. ab108202;
1:500; Abcam), anti-phosphorylated (p-)AKT (cat. no. 4060; 1:2,000;
Cell Signaling Technology, Inc.), anti-Bax (cat. no. ab32503;
1:1,000; Abcam), anti-Caspase 3 (cat no. 19677-1-AP; 1:500;
ProteinTech Group, Inc.), anti-Bcl-2 (cat. no. ab182858; 1:2,000;
Abcam) and anti-GAPDH (cat. no. ab8245; 1:1,000; Abcam). The
membranes were incubated with HRP-conjugated anti-mouse IgG (cat.
no. 7076; 1:5,000; Cell Signaling Technology, Inc.) secondary
antibody for 2 h at 37˚C. The membranes were developed using an
enhanced chemiluminescence system (Beyotime Institute of
Biotechnology). The density of each protein blot was compared with
that of GAPDH using ImageJ software (version 1.46r; National
Institutes of Health) and was shown as a ratio to the endogenous
control.
Transfections
The pcDNA3.1-IGFBP-rP1 (p-IGFBP7) containing
full-length IGFBP-rP1 coding sequence (GeneBank accession,
BC017201.2) and empty pcDNA3.1 (p-NC1), pcDNA3.1-AKT (p-AKT)
containing full-length AKT coding sequence (GeneBank accession,
MG516906.1) and empty pcDNA3.1 (p-NC2) were obtained from Sangon
Biotech Co., Ltd. HEC-1B and AN3CA cells were seeded into six-well
plates and cultured for 24 h until 80% confluence. Then, HEC-1B and
AN3CA cells were transfected with p-IGFBP7 (2 µg) or p-AKT (2 µg)
using Lipofectamine® 3000 Reagent (Invitrogen; Thermo
Fisher Scientific, Inc.) in accordance with the manufacturer's
instructions for 6 h at 37˚C. In rescue experiments, cells were
co-transfected with p-IGFBP7 (1 µg) and p-AKT (1 µg) at the same
time. At 48 h post-transfection, cells were collected and used for
subsequent experiments.
MTT assay
The cell proliferation was measured using the MTT
assay (MilliporeSigma). Briefly, HEC-1B and AN3CA cells were
inoculated in 96-well plates containing 200 µl of DMEM medium at
5x103 cells/well. In order to determine the adherence of
cells to the floor of plate wells, the plates were incubated for 24
h at 37˚C and 5% carbon dioxide. After the indicated treatment,
cells were incubated with 5 mg/ml MTT solution (20 µl for each
well) and the plates were incubated at 37˚C for 4 h. The wells were
then emptied and 150 µl of dimethyl sulfoxide (DMSO) was added to
each well. A Multiska FC microplate reader (Thermo Fisher
Scientific, Inc.) was then used to calculate the optical density at
490 nm.
Flow cytometry
Apoptosis was evaluated using Annexin V-fluorescein
isothiocyanate (FITC) and propidium iodide (PI) (Beyotime Institute
of Biotechnology). Briefly, after the indicated treatment, the
cells were collected and washed with PBS. Annexin-binding buffer
was used to re-suspend HEC-1B and AN3CA cells that were then
incubated with Annexin V-FITC and PI for 10 min at room
temperature. Apoptosis was assessed by flow cytometer (Beckman
Coulter, Inc.) with Cell Quest software v6.0 (BD Biosciences).
FITC/PI denoted living cells, FITC+/PI indicated early apoptotic
cells, FITC+/PI+ represented late apoptotic cells and FITC/PI+
depicted necrotic cells.
Glutathione S-transferase (GST)
pull-down assay
GST or GST-fusion proteins were expressed and
purified in accordance with the manufacturer's protocol (Cytiva).
His-tagged Protein Purification kit was purchased from GenScript.
HEC-1B and AN3CA cells were transformed with the plasmid expressing
GST fusion proteins (2 µg) or GST control (2 µg). In
vitro-translated His-IGFBP-rP1 (2 µg) was incubated with
GST-AKT fusion proteins (2 µg) or GST alone (2 µg) for 3 h at 4˚C.
GST or GST-AKT were bound to glutathione-Sepharose beads (Cytiva).
The mixture was washed eight times with PBS to thoroughly remove
unbound proteins. A volume of 50 µl of SDS-PAGE loading buffer (1x)
was added, boiled for 5 min and centrifuged at 12,000 x g for 1 min
at 4˚C. The absorbed proteins were analyzed using western
blotting.
Co-immunoprecipitation (co-IP)
assay
Whole-cell lysate (400 µg) were treated with
anti-AKT antibodies (1:100; cat. no. ab183556; Abcam) or IgG
control antibody (1:100; cat. no. ab172730; Abcam) in TBS buffer
(40 mM Tris-HCl pH 7.5, 130 mM NaCl) for 1 h at room temperature,
and then was centrifuged at 14,000 x g for 15 min at 4˚C. Next,
protein A agarose beads (2 µg/ml) were added to the supernatant;
then, it was shaken at 4˚C for 10 min on horizontal ice to remove
non-specific foreign proteins and reduce the background. Then,
protein A beads (2 µg/ml; cat. no. #sc-2003; Santa Cruz
Biotechnology, Inc.) were removed after centrifuging at 14,000 x g
for 15 min at 4˚C. The mixture of antibody and tissue lysate was
slowly shaken at 4˚C overnight. Subsequently, 100 µl of protein A
agarose beads were added to capture the antibody and its bound
proteins, and the antigen-antibody mixture was slowly shaken at 4˚C
overnight. The mixture was centrifuged at 14,000 x g for 5 sec at
4˚C, the agarose bead antibody complex was collected, the
supernatant was removed and washed with cooled PBS buffer for three
times. Then, the released proteins were analyzed by western blot
analysis using anti-IGFBP-rP1 (1:500) or anti-AKT antibodies
(1:500).
Production and differentiation of
macrophages
THP-1 cells were treated with 100 ng/ml Phorbol
12-myristate 13-acetate (PMA) (cat. no. P1585; MilliporeSigma) for
24 h at room temperature to generate THP-1 macrophages (M0
macrophages) (16). To simulate
the formation of TAMs, M0 macrophages were co-cultured with HEC-1B
and AN3CA cells transfected with IGFBP-rP1 overexpression vector
(p-IGFBP7) and/or AKT overexpression vector (p-AKT) in a 6-well
Transwell co-culture system for 48 h at room temperature.
ELISA
The concentration of inducible nitric oxide synthase
(iNOS; cat. no. ab253217; Abcam), TNF-α (cat. no. ab181421; Abcam),
CDl63 (cat. no. ab274394; Abcam), arginase-1 (Arg-1; cat. no.
BMS2216; Thermo Fisher Scientific, Inc.) and mannose receptor (MR;
cat. no. ab277420; Abcam) in the culture medium was measured by
ELISA-kits according to the manufacturer's instructions,
respectively.
Statistical analysis
All statistical analyses were performed using SPSS
19.0 statistical software (IBM Corp.). Data were analyzed by
unpaired Student's t-test or one-way ANOVA followed by Tukey's post
hoc test and were presented as the mean ± standard deviation of
three independent experiments. P<0.05 was considered to indicate
a statistically significant difference.
Results
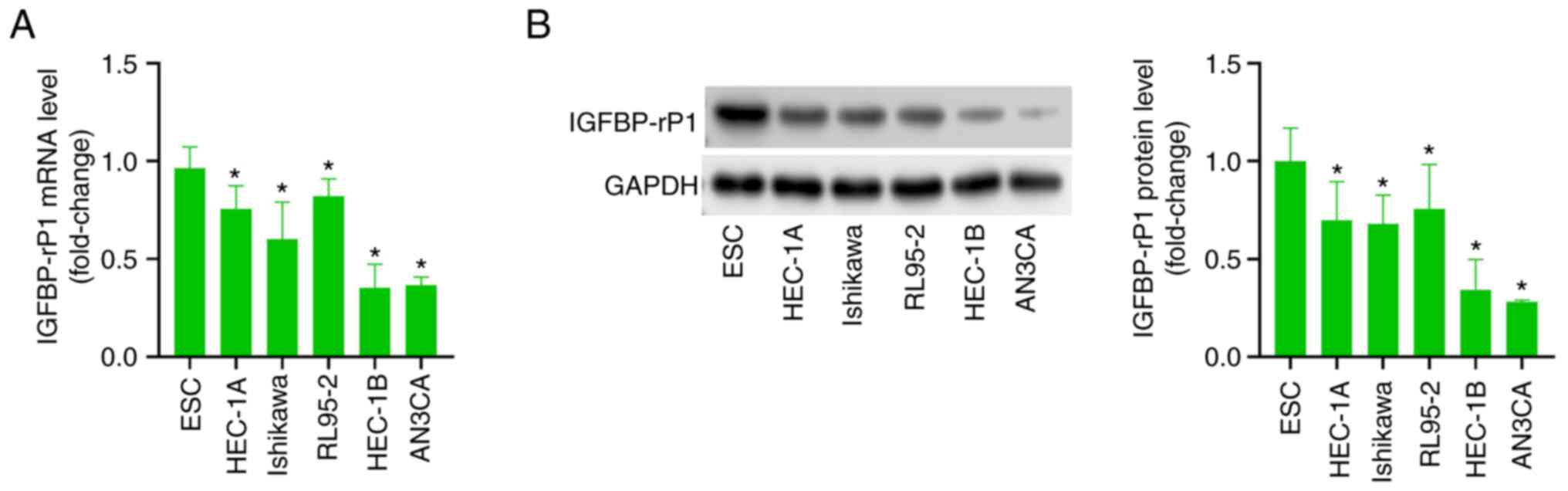
The expression of IGFBP-rP1 in EC
cells is downregulated
RT-qPCR showed that compared with normal endometrial
epithelial (ESC) cells, IGFBP-rP1 mRNA level in Ishikawa, HEC-1A
and RL95-2 was markedly diminished, especially in HEC-1B and AN3CA
cells (Fig. 1A). Similarly, the
expression of IGFBP-rP1 protein in EC cell lines was significantly
lower than that of normal ESC cells, especially in HEC-1B and AN3CA
cells (Fig. 1B). Therefore, HEC-1B
and AN3CA cells were chosen for follow-up experiments.
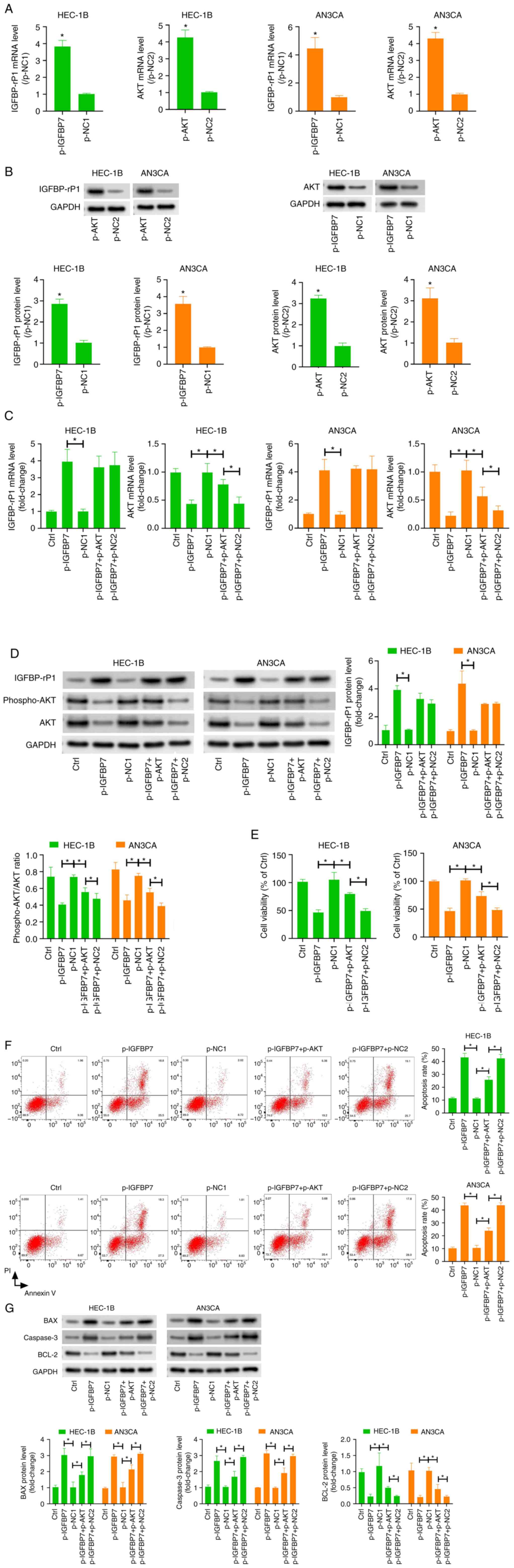
Overexpression of IGFBP-rP1 affects
the proliferation and apoptosis of EC cells by regulating the
PI3K/AKT pathway
To overexpress IGFBP-rP1, the pcDNA3.1-IGFBP-rP1
(p-IGFBP7) vector was transfected into HEC-1B and AN3CA cells.
Compared with the p-NC group, IGFBP-rP1 mRNA and protein levels in
the p-IGFBP7 group were increased significantly, confirming the
successful transfection (Fig. 2A
and B). Successful transfection of
pcDNA3.1-AKT vector significantly increased the expression of AKT
mRNA and protein in HEC-1B and AN3CA cells (Fig. 2A and B). Compared with the p-NC group, the
expression of AKT and p-AKT in HEC-1B and AN3CA cells in the
p-IGFBP7 group was significantly reduced (Fig. 2C and D), which suggested that overexpression of
IGFBP7 inhibited the activation of PI3K/AKT pathway in HEC-1B and
AN3CA cells. To verify this conclusion, we co-transfected HEC-1B
and AN3CA cells with the p-IGFBP7 and p-AKT. Compared to the
p-IGFBP7 group, the expression of AKT and p-AKT proteins were
markedly increased in the p-IGFBP7 + p-AKT group (Fig. 2C and D).
To further evaluate the effect of IGFBP-rP1 on the
biological characteristics of EC cells, MTT assay was used to
evaluate cell proliferation. The results showed that compared to
the p-NC group, the proliferation of HEC-1B and AN3CA cells were
markedly decreased in p-IGFBP7 group (Fig. 2E). Additionally, the proliferation
of EC cells was significantly increased in p-IGFBP7 + p-AKT group
compared with the p-IGFBP7 + p-NC2 group (Fig. 2E). Flow cytometry also showed that
the apoptosis of HEC-1B and AN3CA cells was evidently increased in
p-IGFBP7 group compared with the p-NC1 group (Fig. 2F). Consistently, the expression of
BAX and Caspase-3, while the expression of BCL-2 was markedly
decreased in p-IGFBP7 group compared with the p-NC1 group (Fig. 2G), which further confirmed the
induction of EC cell apoptosis by IGFBP-rP1. Compared to the
p-IGFBP7 group, the apoptosis of HEC-1B and AN3CA cells was
markedly decreased in p-IGFBP7 + p-AKT group (Fig. 2F and G). Taken together, our results indicate
that IGFBP-rP1 inhibited the malignant phenotype of EC cells by
inhibiting the activated PI3K/AKT pathway.
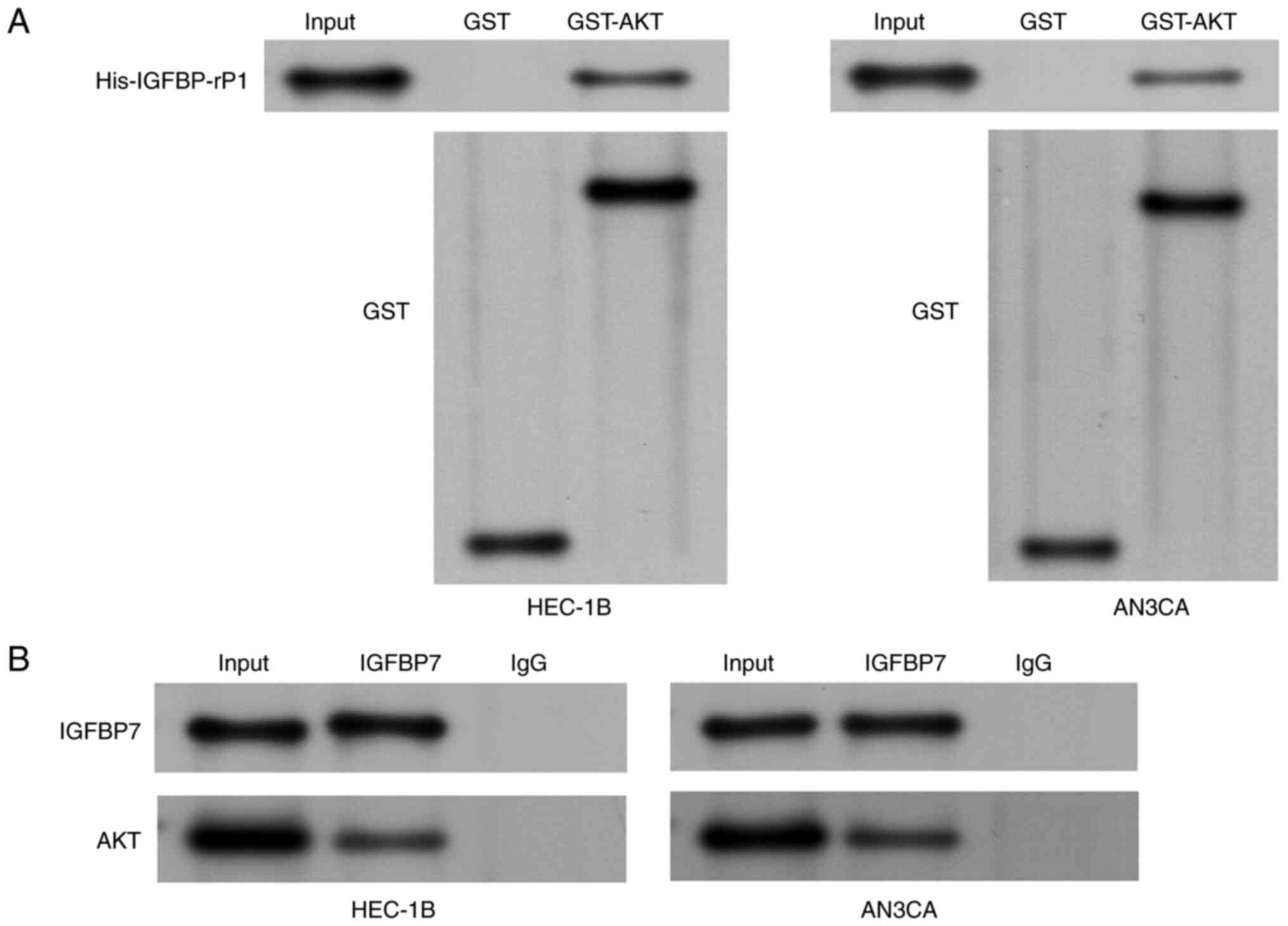
The interaction of IGFBP-rP1 and AKT
in EC cells
The association between IGFBP-rP1 and AKT was
evaluated. A GST pull-down assay was performed to assess the
binding ability of IGFBP-rP1 to AKT (Fig. 3A). The results confirmed the direct
interaction between IGFBP-rP1 and AKT. The direct interaction of
IGFBP-rP1 and AKT was also confirmed by co-immunoprecipitation
assay in HEC-1B and AN3CA cells (Fig.
3B).
Overexpression of IGFBP-rP1 affects
the polarization of EC-related macrophages by regulating the
PI3K/AKT pathway
TAMs are a type of M2 macrophages with
tumor-promoting effects that play a key role in mediating the
connection between cells in the tumor microenvironment (5). In order to confirm the effect of
IGFBP-rP1 on macrophages at the cellular level, human THP-1
monocytes were treated with PMA for 24 h to induce M0 macrophages
(15). Subsequently, M0 cells were
co-cultured with EC cells overexpressing IGFBP-rP1 and/or
overexpressing AKT for 48 h to produce TAMs. Compared with M0, TAMs
showed lower levels of M1 markers iNOS (Fig. 4A) and TNF-α (Fig. 4B) and higher levels of M2 markers
CD163 (Fig. 4C), Arg-1 (Fig. 4D) and MR (Fig. 4E), suggesting that M0 macrophages
were induced by cancer cells to differentiate into M2 macrophages.
Compared with M0 + p-NC1 group, the expression of iNOS and TNF-α
was increased (Fig. 4A and
B), while the expression of CD163,
Arg-1 and MR was decreased in M0 + p-IGFBP7 group (Fig. 4C-E), suggesting that overexpression
of IGFBP-rP1 inhibited M2 polarization. However, compared to the M0
+ p-IGFBP7 + p-NC2 group, the expression of iNOS and TNF-α was
decreased, while the expression of CD163, Arg-1 and MR was
increased in M0 + p-IGFBP7 + p-AKT group, suggesting that the
overexpression of AKT in EC cells abolished the inhibitory effect
of IGFBP-rP1 on M2 polarization (Fig.
4A-D).
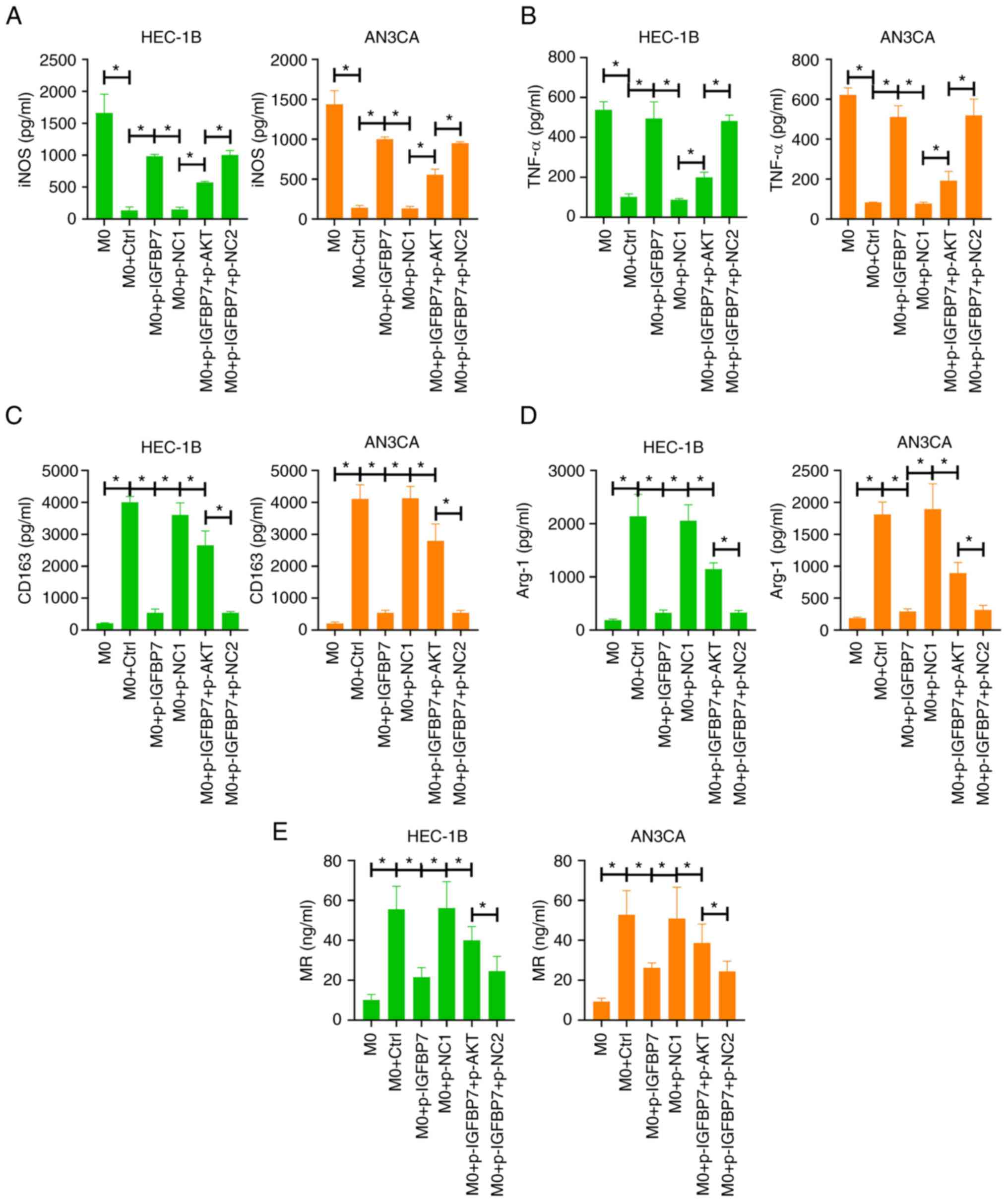 | Figure 4Overexpression of IGFBP-rP1 affects
the polarization of EC-related macrophages by regulating the
PI3K/AKT pathway. Human THP-1 monocytes were treated with PMA for
24 h to induce M0 macrophages. Then, M0 cells were co-cultured with
EC cells overexpressing IGFBP-rP1 and/or overexpressing AKT for 48
h to produce TAMs. The level of M1 markers, (A) iNOS and (B) TNF-α
and M2 markers, (C) CD163, (D) Arg-1 and (E) MR were analyzed using
ELISA. *P<0.05. IGFBP-rP1, insulin-like growth factor
binding protein-related protein 1; EC, endometrial carcinoma; TAMs,
tumor-associated macrophages; iNOS, inducible nitric oxide
synthase; NC, negative control; Arg-1, arginase-1; MR, mannose
receptor. |
Discussion
IGFBP-rP1 is a potential tumor suppressor gene in a
variety of cancers, including EC (13). Nonetheless, its mechanism in EC
remains to be elucidated. The present study showed that IGFBP-rP1
has low expression levels in EC cells. It was found in in
vitro experiments that overexpressed IGFBP-rP1 inhibited the
proliferation and induced apoptosis of EC cells. These findings
suggested that IGFBP-rP1 might play a tumor suppressor role in
EC.
The PI3K/AKT pathway is a classic pathway that
regulates cell proliferation, apoptosis and metastasis (16,17).
A previous study reported that the activation of the PI3K/AKT
pathway is related to the continuous growth of various solid
tumors, including EC (18). AKT
plays a major role in this signal pathway. p-AKT is related to the
disorder of apoptosis, proliferation and cell motility because of
its role in inducing signals that interfere with the normal
regulatory mechanisms that activate the mTOR (19). In the current study, it was
observed that overexpression of IGFBP-rP1 reduced levels of p-AKT
in EC cells, indicating that the activation of the PI3K/AKT
signaling pathway was repressed. Additionally, the overexpression
of AKT effectively reversed the reduced proliferation and increased
apoptosis caused by IGFBP-rP1. These findings suggested that
IGFBP-rP1 inhibited the PI3K/AKT signaling pathway to exert a tumor
suppressor effect in EC. Importantly, through co-IP and GST
pull-down assays, it was also confirmed that AKT is a key protein
interacting with IGFBP-rP1. Based on these data, it was
hypothesized that IGFBP-rP1 directly binds to AKT to block the
phosphorylation of AKT, thereby inhibiting the PI3K/AKT
pathway.
Previous studies provide novel understanding into
the appearance of TAMs in the tumor microenvironment (20,21).
Studies indicate that the existence of TAMs at tumor sites is
closely related to tumor progression (22,23).
Cassetta et al (24)
isolated TAMs from breast cancer and EC tissues and found that the
TAMs population positively correlated with poorer clinical
prognosis. TAMs, therefore, may be crucial to the occurrence and
development of EC. Gu et al (25) proposed that blocking M2 macrophages
in TME may be a promising target for EC tumor immunotherapy. The
present study used macrophages as a study subject to observe the
effect of IGFBP-Rp1 on macrophage polarization by co-culturing
macrophages with EC cells to mimic the inflammatory
microenvironment of EC in vitro. As expected, M2 macrophage
markers were significantly upregulated after incubating M0
macrophages with the EC cells. In addition, by co-culturing M0
macrophages with the EC cells transfected with IGFBP-rP1
overexpression vector, the changes in cytokine markers in the
macrophages were blocked. These data suggested that the high
expression of IGFBP-rP1 inhibited the M2 differentiation induced by
the EC cells. The role of PI3K/AKT pathway in TAMs has also been
studied. The uncontrolled activation of the PI3K/AKT pathway
induces immune tolerance TME and regulates the transition between
immune stimulation and immunosuppression of TAMs (26). Inhibition of PI3Kγ has been
proposed as a macrophage-based cancer treatment strategy (27). Additionally, changes in AKT
isoforms or AKT activity levels in macrophages determine the
viability of monocytes/macrophages (28,29).
The present study showed that overexpression of AKT in EC cells
abolished the effect of overexpression of IGFBP-rP1 on M2
polarization, indicating that this regulation was dependent on the
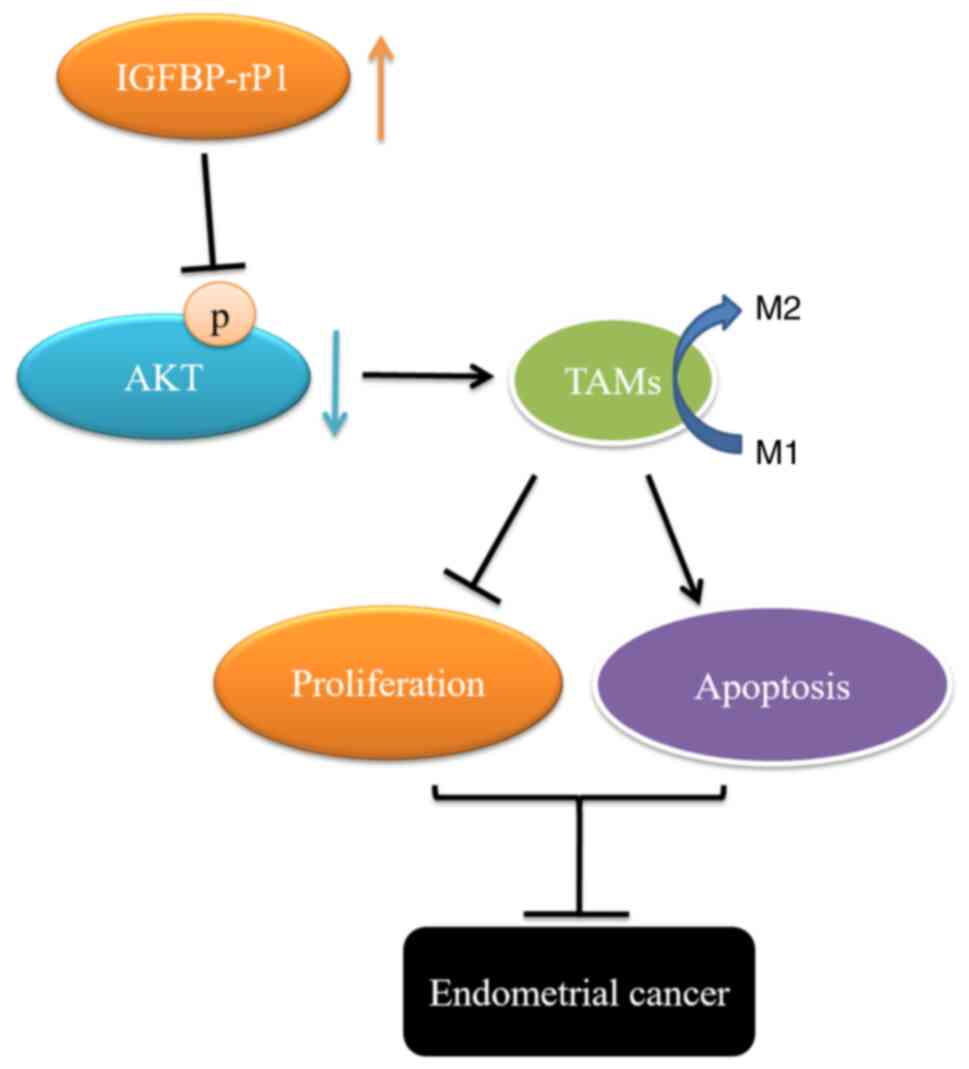
PI3K/AKT pathway. IGFBP-rP1, therefore, acted as an inhibitor of
the M2 polarization of TAMs through the PI3K/AKT signaling pathway
(Fig. 5).
In conclusion, the present study confirmed that
IGFBP-rP1 promotes the proliferation and induces apoptosis of EC
cells. Additionally, IGFBP-rP1 directly binds to AKT to block the
phosphorylation of AKT, thereby inhibiting the PI3K/AKT pathway.
The present study also proved that IGFBP-rP1 recruited M2 TAMs
through PI3K/AKT signals. These results indicated that IGFBP-rP1
can be a capability marker for the treatment of EC in the future.
The lack of clinical and animal studies is a limitation of the
present study and will be fully researched in future studies.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets generated and/or analyzed during the
current study are available from the corresponding author on
reasonable request.
Authors' contributions
JG was the main contributor for designing the study
and writing the manuscript. SS, JL and CW contributed to conducting
the experiments. RD, YH and CZ analyzed the data and revised the
manuscript. All authors read and approved the final manuscript. RD
and CZ confirm the authenticity of all the raw data.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Xiao L, He Y, Peng F, Yang J and Yuan C:
Endometrial cancer cells promote M2-Like macrophage polarization by
delivering exosomal miRNA-21 under hypoxia condition. J Immunol
Res. 2020(9731049)2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Braun MM, Overbeek-Wager EA and Grumbo RJ:
Diagnosis and management of endometrial cancer. Am Fam Physician.
93:468–474. 2016.PubMed/NCBI
|
|
3
|
Hinshaw DC and Shevde LA: The tumor
microenvironment innately modulates cancer progression. Cancer Res.
79:4557–4566. 2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Vitale I, Manic G, Coussens LM, Kroemer G
and Galluzzi L: Macrophages and metabolism in the tumor
microenvironment. Cell Metab. 30:36–50. 2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Kim J and Bae JS: Tumor-Associated
macrophages and neutrophils in tumor microenvironment. Mediators
Inflamm. 2016(6058147)2016.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Genin M, Clement F, Fattaccioli A, Raes M
and Michiels C: M1 and M2 macrophages derived from THP-1 cells
differentially modulate the response of cancer cells to etoposide.
BMC Cancer. 15(577)2015.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Cassetta L and Pollard JW:
Tumor-associated macrophages. Curr Biol. 30:R246–R248.
2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Shapouri-Moghaddam A, Mohammadian S,
Vazini H, Taghadosi M, Esmaeili SA, Mardani F, Seifi B, Mohammadi
A, Afshari JT and Sahebkar A: Macrophage plasticity, polarization,
and function in health and disease. J Cell Physiol. 233:6425–6440.
2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Cersosimo F, Lonardi S, Bernardini G,
Telfer B, Mandelli GE, Santucci A, Vermi W and Giurisato E:
Tumor-Associated macrophages in osteosarcoma: From mechanisms to
therapy. Int J Mol Sci. 21(E5207)2020.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Wang X, Gao S, Song L, Liu M, Sun Z and
Liu J: Astragaloside IV antagonizes M2 phenotype macrophage
polarization-evoked ovarian cancer cell malignant progression by
suppressing the HMGB1-TLR4 axis. Mol Immunol. 130:113–121.
2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Mohd Nafi SN, Siti Azrin AH, Mat Zin AA,
Othman NH and Che Jalil NA: Expression of IGFBP-rP1 in ovarian and
breast cancers in association with diabetes mellitus status. Malays
J Pathol. 41:33–39. 2019.PubMed/NCBI
|
|
12
|
Ruan W, Zhu S, Wang H, Xu F, Deng H, Ma Y
and Lai M: IGFBP-rP1, a potential molecule associated with colon
cancer differentiation. Mol Cancer. 9(281)2010.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Ma Y, Jiang J, Zhang Y, Ding Y, Xu T and
Lu B: IGFBP-rP1 acts as a potential tumor suppressor via the
suppression of ERK signaling pathway in endometrial cancer cells.
Mol Med Rep. 16:1445–1450. 2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Hu S, Chen R, Man X, Feng X, Cen J, Gu W,
He H, Li J, Chai Y and Chen Z: Function and expression of
insulin-like growth factor-binding protein 7 (IGFBP7) gene in
childhood acute myeloid leukemia. Pediatr Hematol Oncol.
28:279–287. 2011.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) Method. Methods. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Ersahin T, Tuncbag N and Cetin-Atalay R:
The PI3K/AKT/mTOR interactive pathway. Mol Biosyst. 11:1946–1954.
2015.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Porta C, Paglino C and Mosca A: Targeting
PI3K/Akt/mTOR signaling in cancer. Front Oncol.
4(64)2014.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Roncolato F, Lindemann K, Willson ML,
Martyn J and Mileshkin L: PI3K/AKT/mTOR inhibitors for advanced or
recurrent endometrial cancer. Cochrane Database Syst Rev.
10(CD012160)2019.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Aoki M and Fujishita T: Oncogenic Roles of
the PI3K/AKT/mTOR Axis. Curr Top Microbiol Immunol. 407:153–189.
2017.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Komohara Y and Takeya M: CAFs and TAMs:
Maestros of the tumour microenvironment. J Pathol. 241:313–315.
2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Cortés M, Sanchez-Moral L, de Barrios O,
Fernández-Aceñero MJ, Martínez-Campanario MC, Esteve-Codina A,
Darling DS, Győrffy B, Lawrence T, Dean DC and Postigo A:
Tumor-associated macrophages (TAMs) depend on ZEB1 for their
cancer-promoting roles. EMBO J. 36:3336–3355. 2017.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Pan Y, Yu Y, Wang X and Zhang T:
Tumor-Associated macrophages in tumor immunity. Front Immunol.
11(583084)2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Ge Z and Ding S: The crosstalk between
tumor-associated macrophages (TAMs) and tumor cells and the
corresponding targeted therapy. Front Oncol.
10(590941)2020.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Cassetta L, Fragkogianni S, Sims AH,
Swierczak A, Forrester LM, Zhang H, Soong DYH, Cotechini T, Anur P,
Lin EY, et al: Human Tumor-Associated macrophage and monocyte
transcriptional landscapes reveal cancer-specific reprogramming,
biomarkers, and therapeutic targets. Cancer Cell. 35:588–602.e10.
2019.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Gu S, Ni T, Wang J, Liu Y, Fan Q and Wang
Y, Huang T, Chu Y, Sun X and Wang Y: CD47 blockade inhibits tumor
progression through promoting phagocytosis of tumor cells by M2
polarized macrophages in endometrial cancer. J Immunol Res.
2018(6156757)2018.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Giannone G, Ghisoni E, Genta S, Scotto G,
Tuninetti V, Turinetto M and Valabrega G: Immuno-Metabolism and
microenvironment in cancer: Key players for immunotherapy. Int J
Mol Sci. 21(E4414)2020.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Kowal J, Kornete M and Joyce JA:
Re-education of macrophages as a therapeutic strategy in cancer.
Immunotherapy. 11:677–689. 2019.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Damien C, Cisse F, Ligot N, Toure ML,
Konaté M, Barry SD, Saw M and Naeije G: Insights in the
pathophysiology of haemorrhagic strokes in a sub-Sahara African
country, an epidemiological and MRI study. Trop Med Int Health.
26:166–172. 2021.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Weichhart T, Hengstschläger M and Linke M:
Regulation of innate immune cell function by mTOR. Nat Rev Immunol.
15(599614)2015.PubMed/NCBI View
Article : Google Scholar
|