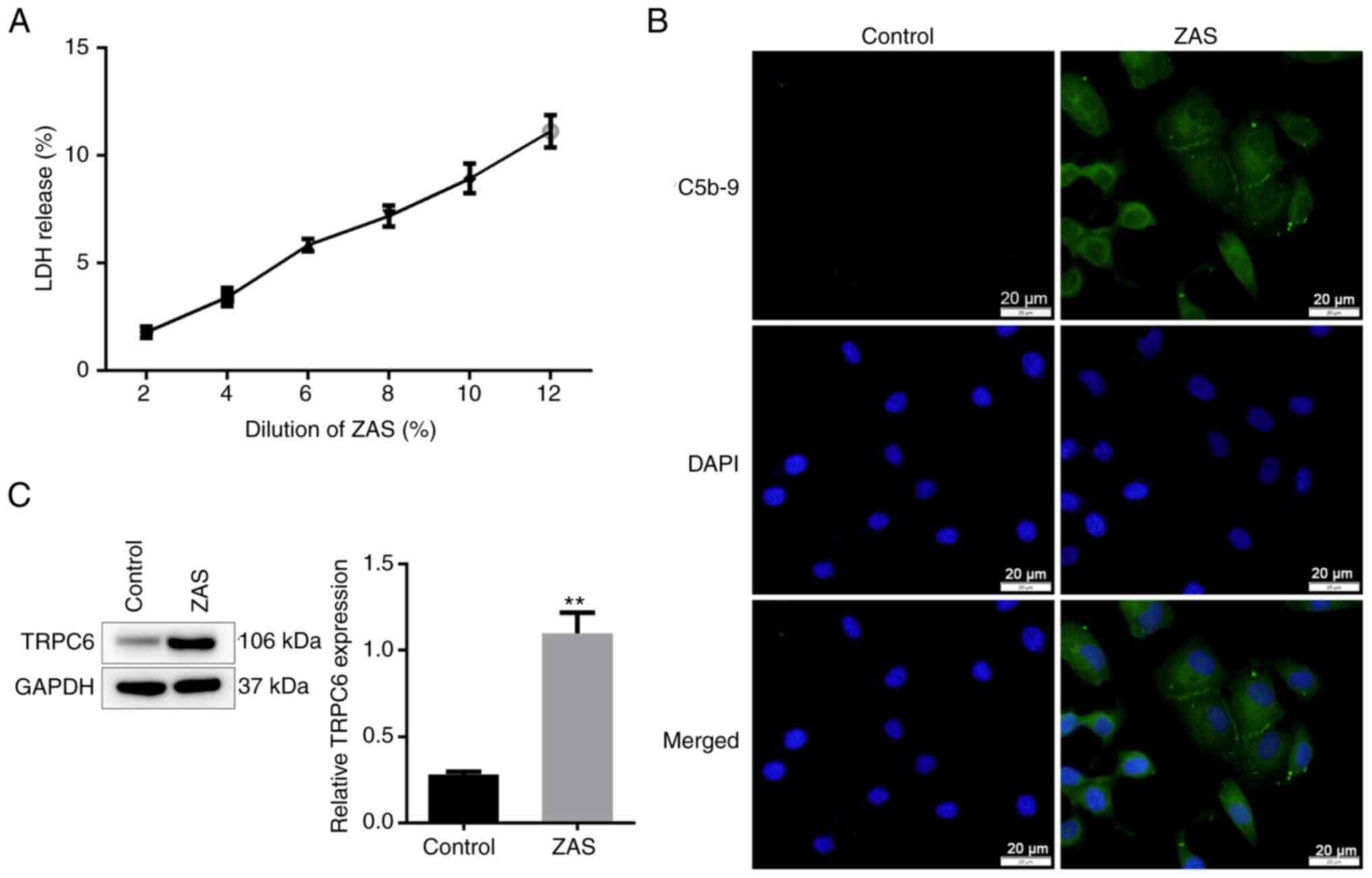
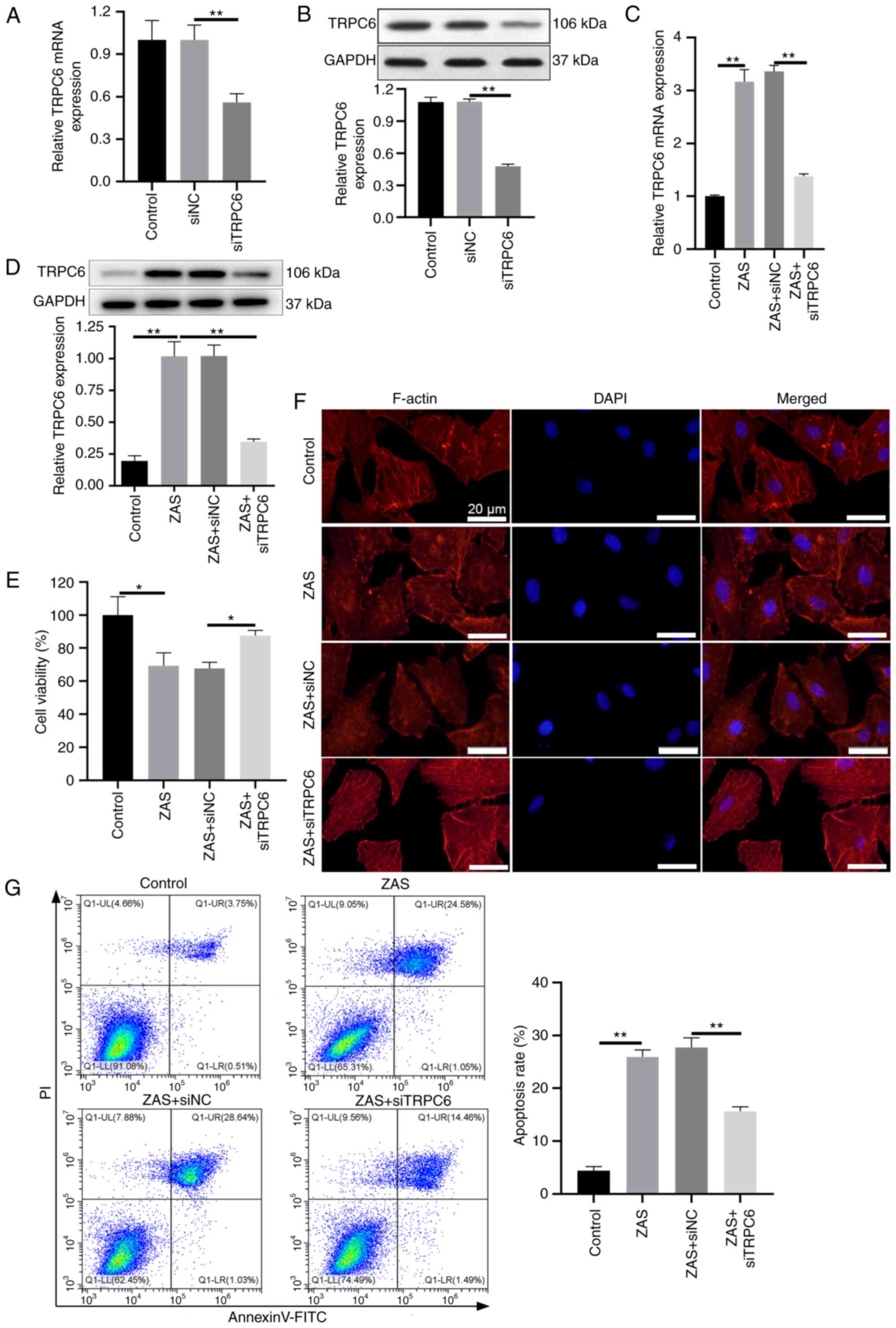
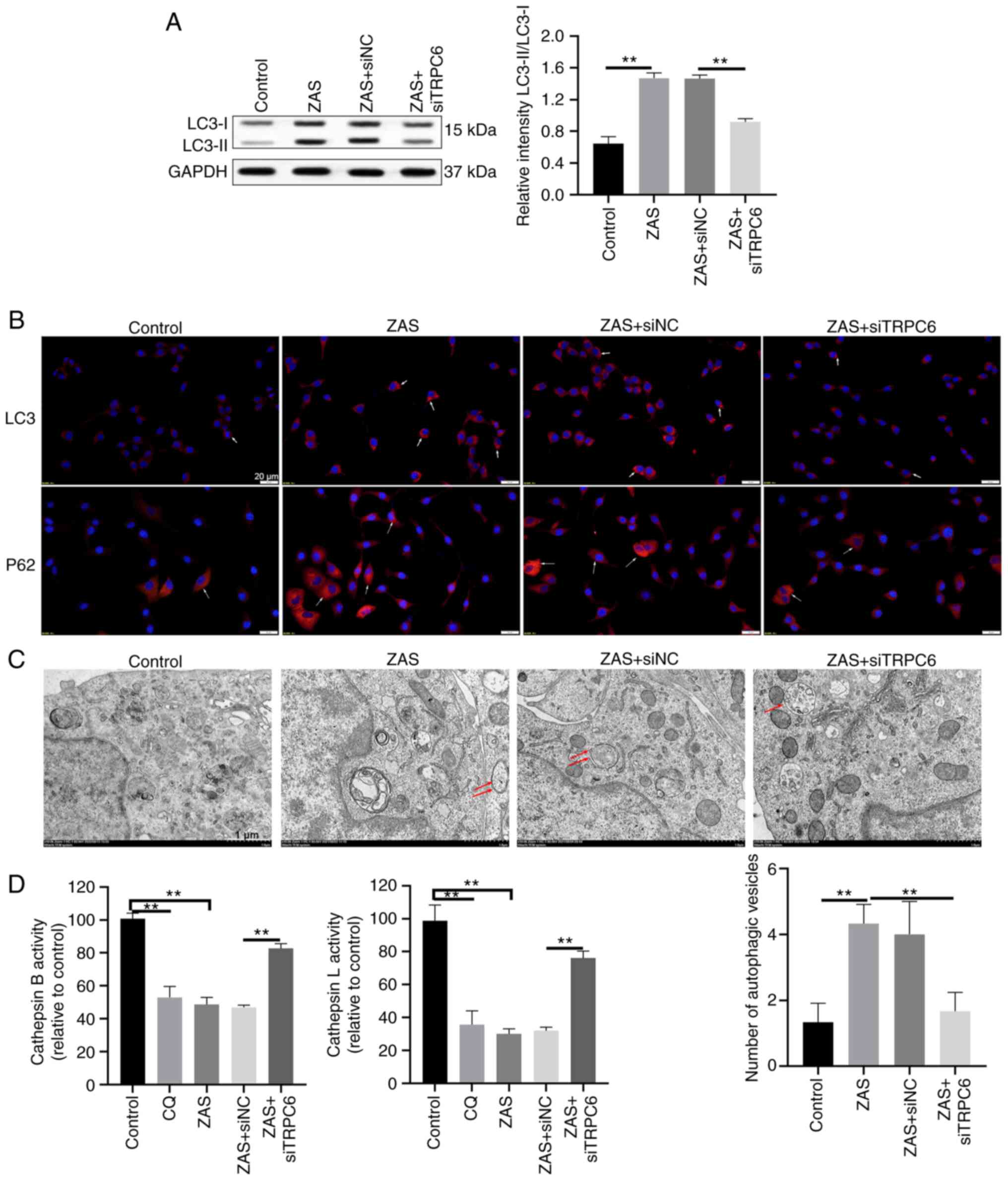
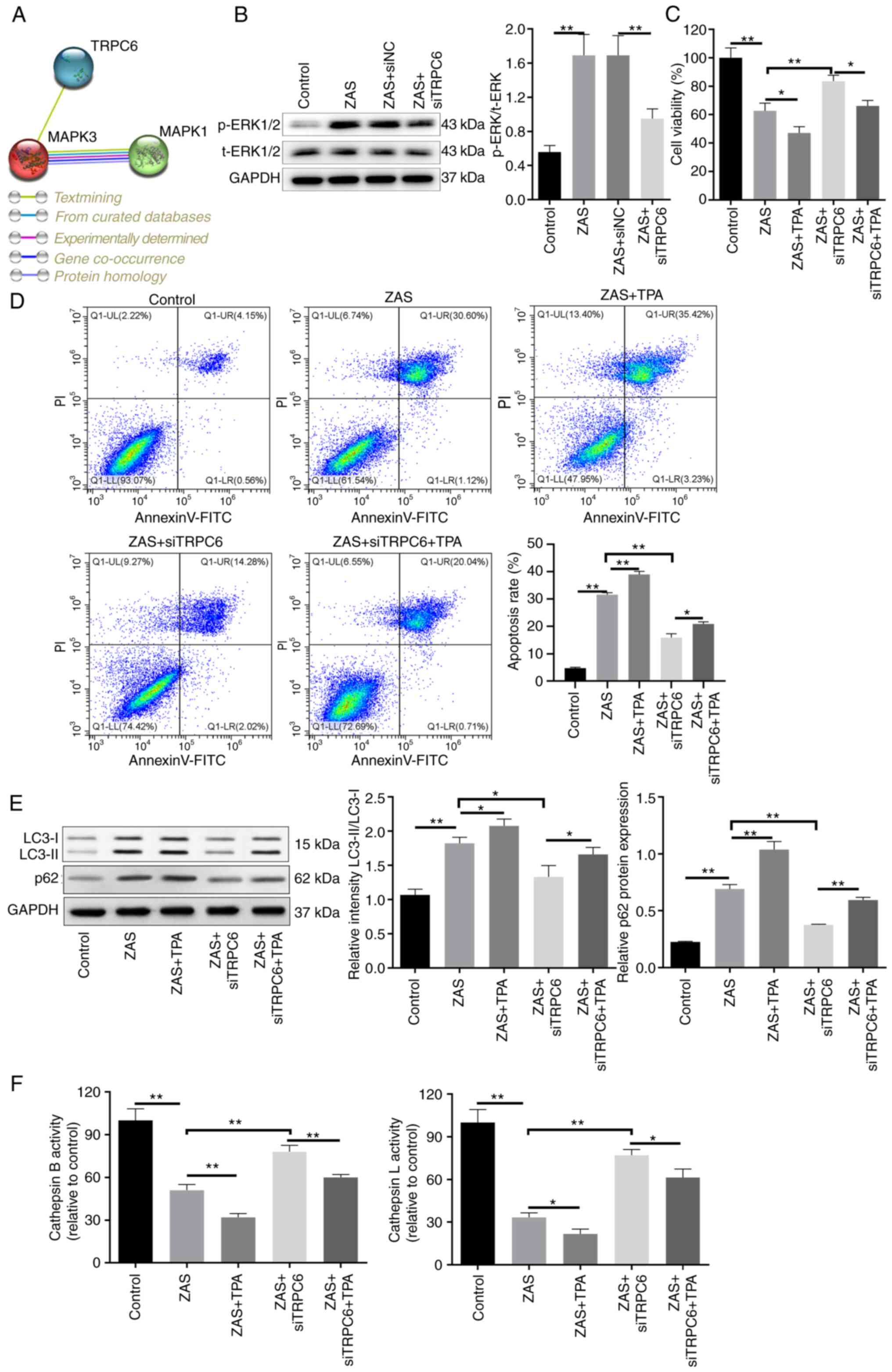
|
1
|
Keri KC, Blumenthal S, Kulkarni V, Beck L
and Chongkrairatanakul T: Primary membranous nephropathy:
Comprehensive review and historical perspective. Postgrad Med J.
95:23–31. 2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Safar-Boueri L, Piya A, Beck LH Jr and
Ayalon R: Membranous nephropathy: Diagnosis, treatment, and
monitoring in the post-PLA2R era. Pediatr Nephrol. 36:19–30.
2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Liu W, Gao C, Liu Z, Dai H, Feng Z, Dong
Z, Zheng Y, Gao Y, Tian X and Liu B: Idiopathic membranous
nephropathy: Glomerular pathological pattern caused by extrarenal
immunity activity. Front Immunol. 11(1846)2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Ma H, Sandor DG and Beck LH Jr: The role
of complement in membranous nephropathy. Semin Nephrol. 33:531–542.
2013.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Nagata M: Podocyte injury and its
consequences. Kidney Int. 89:1221–1230. 2016.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Galindo-Izquierdo M and Pablos Alvarez JL:
Complement as a therapeutic target in systemic autoimmune diseases.
Cells. 10(148)2021.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Yang P, Skiba NP, Tewkesbury GM, Treboschi
VM, Baciu P and Jaffe GJ: Complement-mediated regulation of
apolipoprotein E in cultured human RPE cells. Invest Ophthalmol Vis
Sci. 58:3073–3085. 2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Fishelson Z and Kirschfink M: Complement
C5b-9 and Cancer: Mechanisms of cell damage, cancer counteractions,
and approaches for intervention. Front Immunol.
10(752)2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Takano T, Elimam H and Cybulsky AV:
Complement-mediated cellular injury. Semin Nephrol. 33:586–601.
2013.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Bateman RM, Sharpe MD, Jagger JE, Ellis
CG, Solé-Violán J, López-Rodríguez M, Herrera-Ramos E,
Ruíz-Hernández J, Borderías L, Horcajada J, et al: 36th
International symposium on intensive care and emergency medicine:
Brussels, Belgium. 15-18 March 2016. Crit Care. 20 (Suppl
2)(S94)2016.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Tan Y and Zhao MH: Complement in
glomerular diseases. Nephrology (Carlton). 23 (Suppl 4):S11–S15.
2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Xie H, Yang L, Yang Y, Jiang W, Wang X,
Huang M, Zhang J and Zhu Q: C5b-9 membrane attack complex activated
NLRP3 inflammasome mediates renal tubular immune injury in
trichloroethylene sensitized mice. Ecotoxicol Environ Saf.
208(111439)2021.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wang X, Liu J, Tian R, Zheng B, Li C,
Huang L, Lu Z, Zhang J, Mao W, Liu B, et al: Sanqi oral solution
mitigates proteinuria in rat passive heymann nephritis and blocks
podocyte apoptosis via Nrf2/HO-1 Pathway. Front Pharmacol.
12(727874)2021.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Zhou XJ, Klionsky DJ and Zhang H:
Podocytes and autophagy: A potential therapeutic target in lupus
nephritis. Autophagy. 15:908–912. 2019.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Yoshibayashi M, Kume S, Yasuda-Yamahara M,
Yamahara K, Takeda N, Osawa N, Chin-Kanasaki M, Nakae Y, Yokoi H,
Mukoyama M, et al: Protective role of podocyte autophagy against
glomerular endothelial dysfunction in diabetes. Biochem Biophys Res
Commun. 525:319–325. 2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Liu WJ, Li ZH, Chen XC, Zhao XL, Zhong Z,
Yang C, Wu HL, An N, Li WY and Liu HF: Blockage of the
lysosome-dependent autophagic pathway contributes to complement
membrane attack complex-induced podocyte injury in idiopathic
membranous nephropathy. Sci Rep. 7(8643)2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Liu X, Yao X and Tsang SY:
Post-Translational modification and natural mutation of TRPC
Channels. Cells. 9(135)2020.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Hou X, Huang M, Zeng X, Zhang Y, Sun A, Wu
Q, Zhu L, Zhao H and Liao Y: The Role of TRPC6 in renal
ischemia/reperfusion and cellular hypoxia/reoxygenation injuries.
Front Mol Biosci. 8(698975)2021.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Dryer SE, Roshanravan H and Kim EY: TRPC
channels: Regulation, dysregulation and contributions to chronic
kidney disease. Biochim Biophys Acta Mol Basis Dis. 1865:1041–1066.
2019.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Moller CC, Wei C, Altintas MM, Li J, Greka
A, Ohse T, Pippin JW, Rastaldi MP, Wawersik S, Schiavi S, et al:
Induction of TRPC6 channel in acquired forms of proteinuric kidney
disease. J Am Soc Nephrol. 18:29–36. 2007.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Yu S and Yu L: Dexamethasone resisted
podocyte injury via stabilizing TRPC6 expression and distribution.
Evid Based Complement Alternat Med. 2012(652059)2012.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Shengyou Y and Li Y: The effects of
siRNA-silenced TRPC6 on podocyte autophagy and apoptosis induced by
AngII. J Renin Angiotensin Aldosterone Syst. 16:1266–1273.
2015.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Zhang MH, Fan JM, Xie XS, Deng YY, Chen
YP, Zhen R, Li J, Cheng Y and Wen J: Ginsenoside-Rg1 protects
podocytes from complement mediated injury. J Ethnopharmacol.
137:99–107. 2011.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Liu Y, Zhang J, Wang Y and Zeng X: Apelin
involved in progression of diabetic nephropathy by inhibiting
autophagy in podocytes. Cell Death Dis. 8(e3006)2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Ishikawa S, Tsukada H and Bhattacharya J:
Soluble complex of complement increases hydraulic conductivity in
single microvessels of rat lung. J Clin Invest. 91:103–109.
1993.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Schmittgen TD and Livak KJ: Analyzing
Real-time PCR data by the comparative C(T) method. Nat Protoc.
3:1101–1108. 2008.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Pasquier B: Autophagy inhibitors. Cell Mol
Life Sci. 73:985–1001. 2016.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Jefferson JA, Pippin JW and Shankland SJ:
Experimental models of membranous nephropathy. Drug Discov Today
Dis Models. 7:27–33. 2010.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Papagianni AA, Alexopoulos E, Leontsini M
and Papadimitriou M: C5b-9 and adhesion molecules in human
idiopathic membranous nephropathy. Nephrol Dial Transplant.
17:57–63. 2002.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Zheng R, Deng Y, Chen Y, Fan J, Zhang M,
Zhong Y, Zhu R and Wang L: Astragaloside IV attenuates complement
membranous attack complex induced podocyte injury through the MAPK
pathway. Phytother Res. 26:892–898. 2012.PubMed/NCBI View
Article : Google Scholar
|
|
31
|
Wang L, Liang H, Sun B, Mi J, Tong X, Wang
Y, Chen M, Yu L, Pan J, Liu S, et al: Role of TRPC6 in periodontal
tissue reconstruction mediated by appropriate stress. Stem Cell Res
Ther. 13(401)2022.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Zhou LF, Chen QZ, Yang CT, Fu ZD, Zhao ST,
Chen Y, Li SN, Liao L, Zhou YB, Huang JR and Li JH: TRPC6
contributes to LPS-induced inflammation through ERK1/2 and p38
pathways in bronchial epithelial cells. Am J Physiol Cell Physiol.
314:C278–C288. 2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Putta P, Smith AH, Chaudhuri P,
Guardia-Wolff R, Rosenbaum MA and Graham LM: Activation of the
cytosolic calcium-independent phospholipase A2 β isoform
contributes to TRPC6 externalization via release of arachidonic
acid. J Biol Chem. 297(101180)2021.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Hartleben B, Godel M, Meyer-Schwesinger C,
Liu S, Ulrich T, Kobler S, Wiech T, Grahammer F, Arnold SJ,
Lindenmeyer MT, et al: Autophagy influences glomerular disease
susceptibility and maintains podocyte homeostasis in aging mice. J
Clin Invest. 120:1084–1096. 2010.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Fang L, Zhou Y, Cao H, Wen P, Jiang L, He
W, Dai C and Yang J: Autophagy attenuates diabetic glomerular
damage through protection of hyperglycemia-induced podocyte injury.
PLoS One. 8(e60546)2013.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Qi YY, Zhou XJ, Cheng FJ, Hou P, Ren YL,
Wang SX, Zhao MH, Yang L, Martinez J and Zhang H: Increased
autophagy is cytoprotective against podocyte injury induced by
antibody and interferon-α in lupus nephritis. Ann Rheum Dis.
77:1799–1809. 2018.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Lenoir O, Jasiek M, Henique C, Guyonnet L,
Hartleben B, Bork T, Chipont A, Flosseau K, Bensaada I, Schmitt A,
et al: Endothelial cell and podocyte autophagy synergistically
protect from diabetes-induced glomerulosclerosis. Autophagy.
11:1130–1145. 2015.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Hou X, Xiao H, Zhang Y, Zeng X, Huang M,
Chen X, Birnbaumer L and Liao Y: Transient receptor potential
channel 6 knockdown prevents apoptosis of renal tubular epithelial
cells upon oxidative stress via autophagy activation. Cell Death
Dis. 9(1015)2018.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Farmer LK, Rollason R, Whitcomb DJ, Ni L,
Goodliff A, Lay AC, Birnbaumer L, Heesom KJ, Xu SZ, Saleem MA and
Welsh GI: TRPC6 binds to and activates calpain, independent of its
channel activity, and regulates podocyte cytoskeleton, cell
adhesion, and motility. J Am Soc Nephrol. 30:1910–1924.
2019.PubMed/NCBI View Article : Google Scholar
|