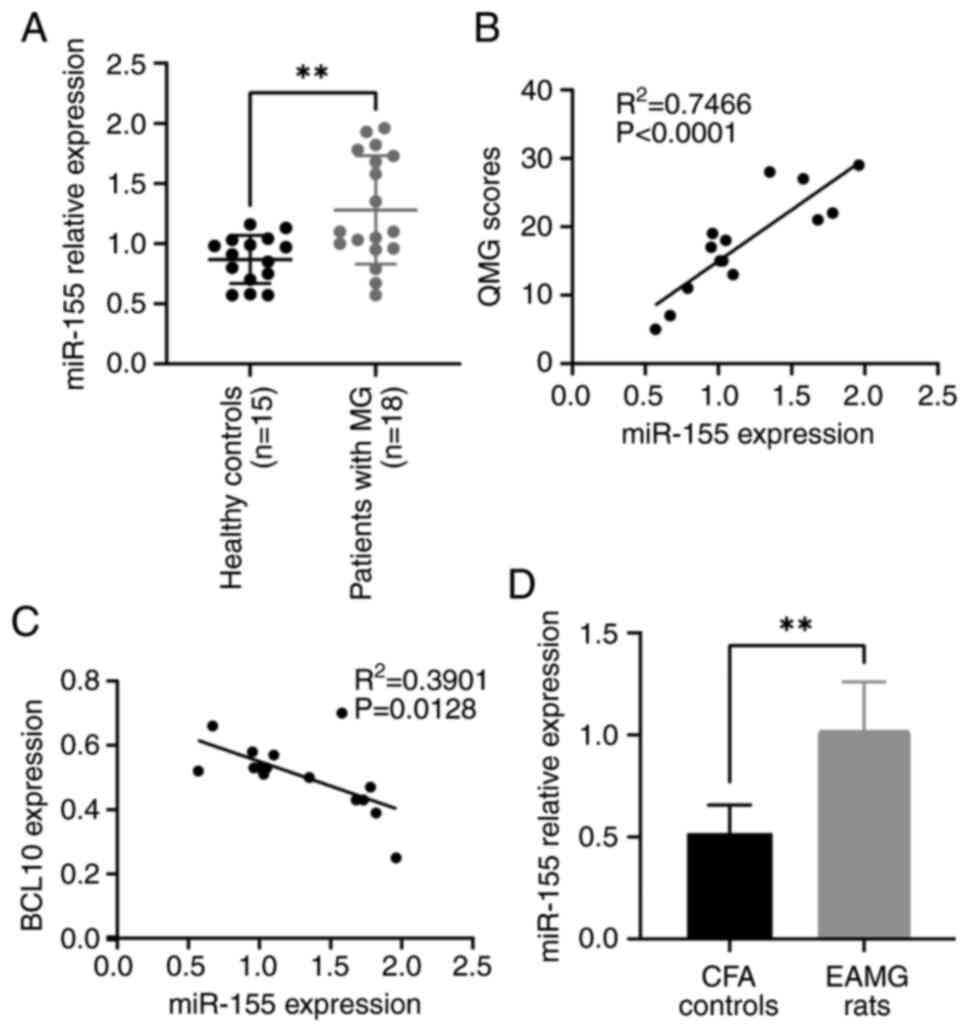
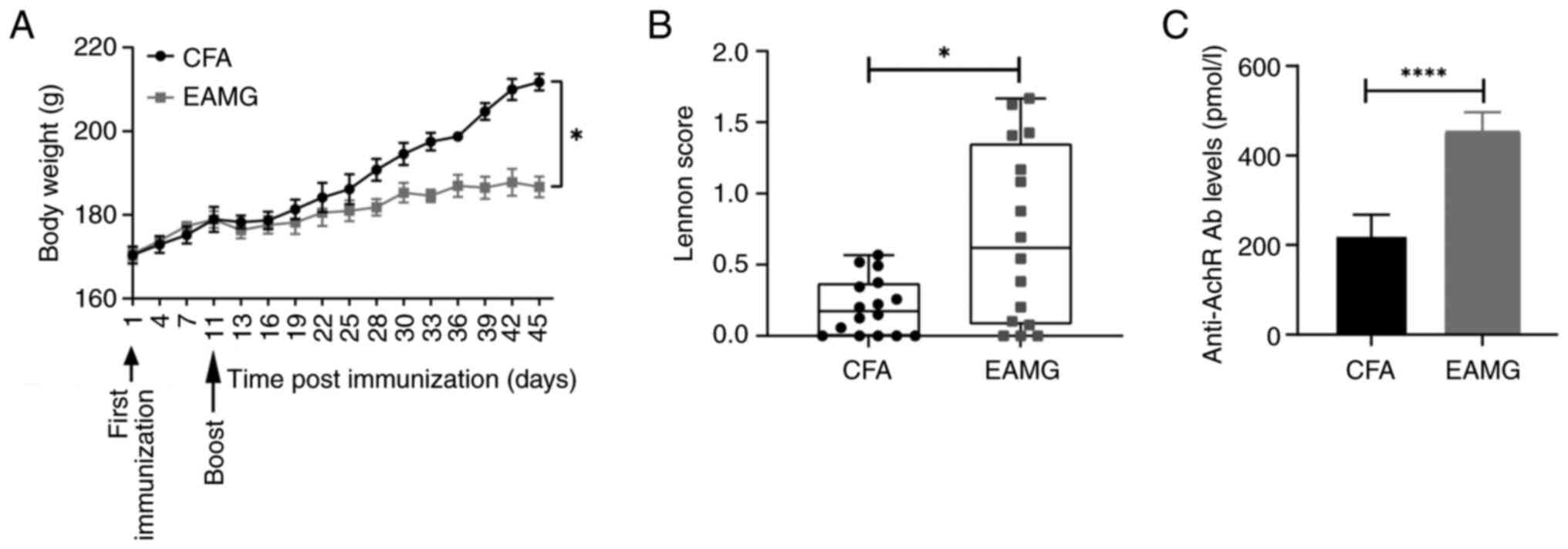
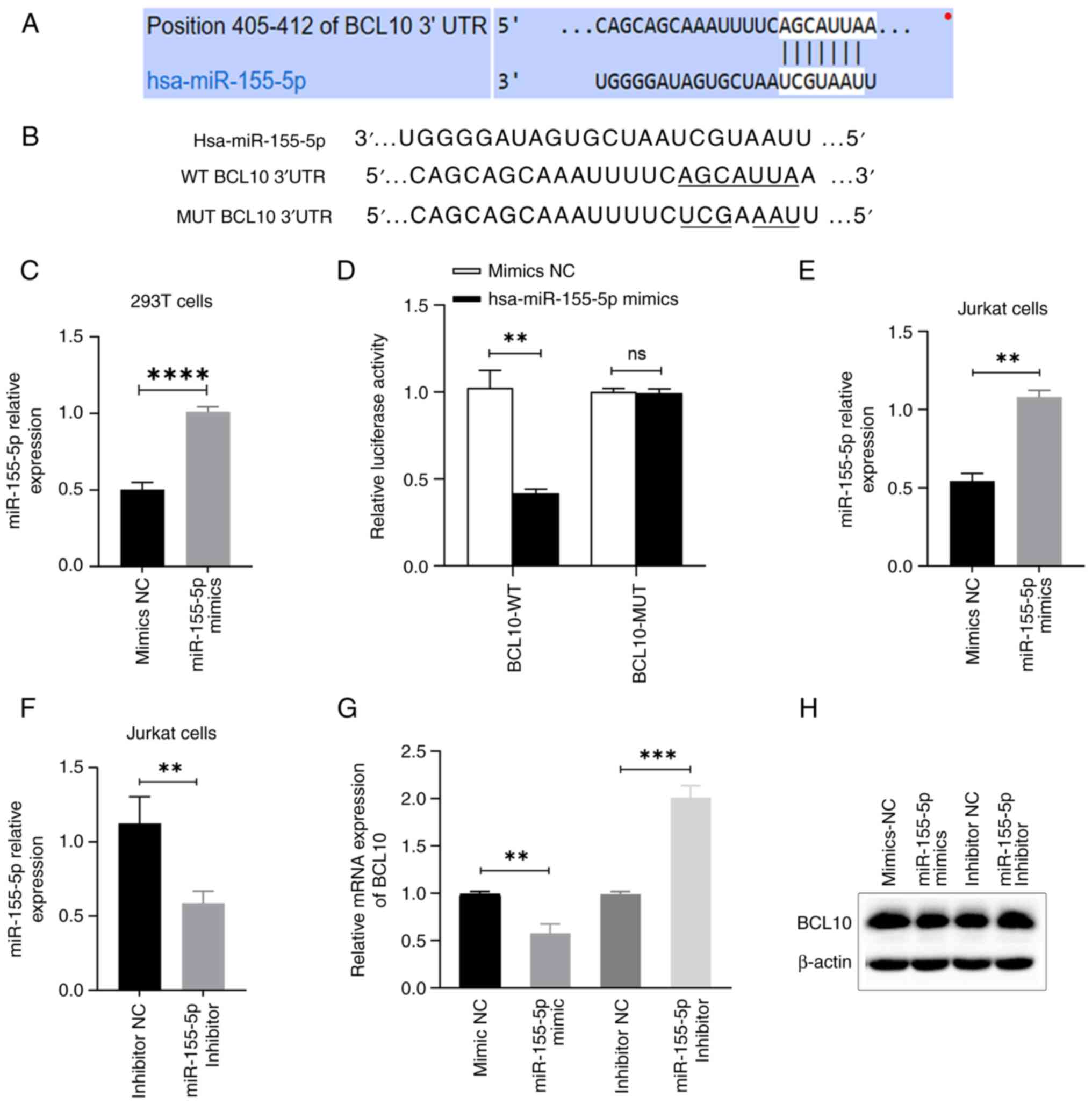
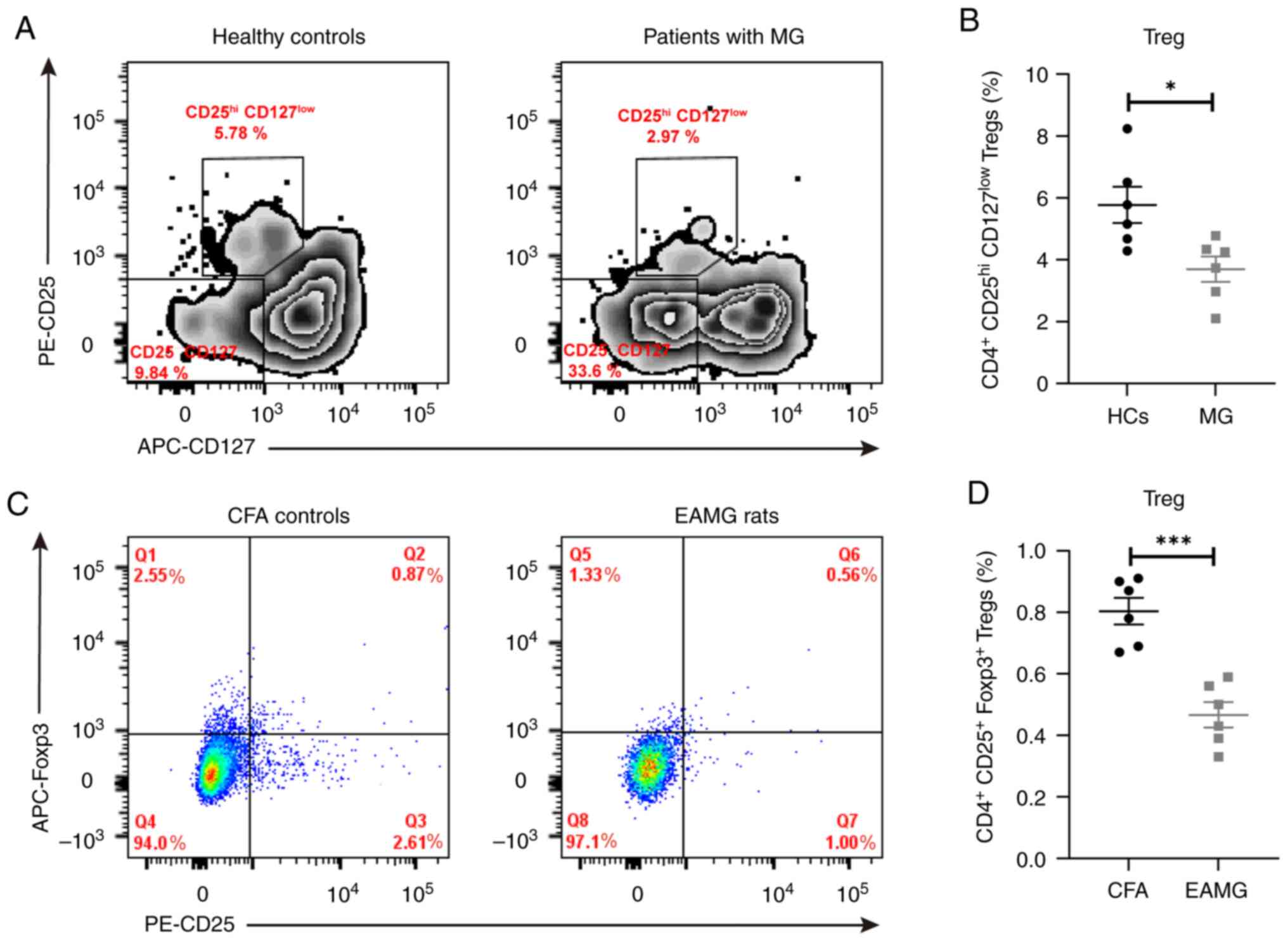
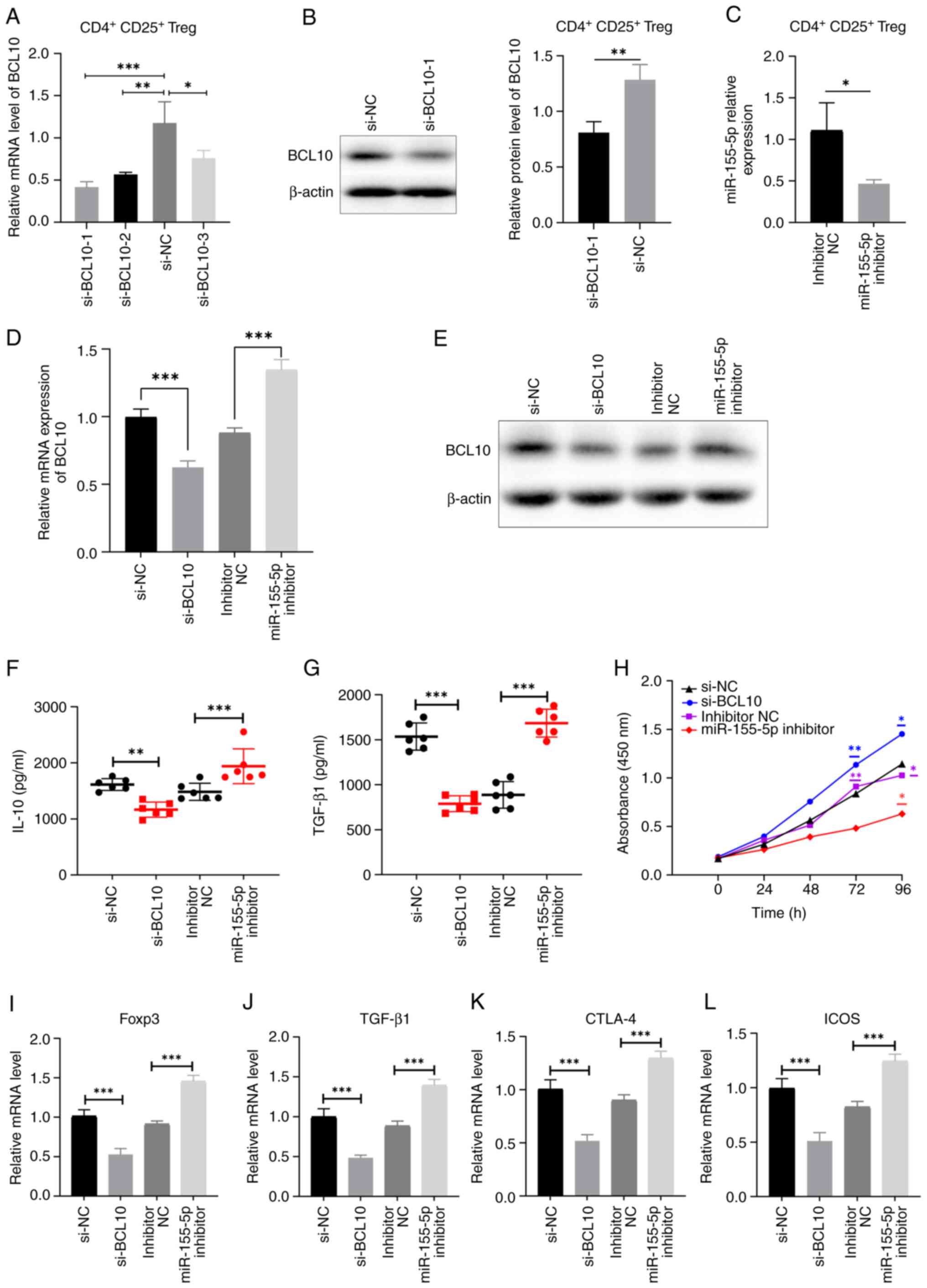
|
1
|
Gilhus NE, Tzartos S, Evoli A, Palace J,
Burns TM and Verschuuren JJGM: Myasthenia gravis. Nat Rev Dis
Primers. 5(30)2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Dresser L, Wlodarski R, Rezania K and
Soliven B: Myasthenia gravis: Epidemiology, pathophysiology and
clinical manifestations. J Clin Med. 10(2235)2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Danikowski KM, Jayaraman S and Prabhakar
BS: Regulatory T cells in multiple sclerosis and myasthenia gravis.
J Neuroinflammation. 14(117)2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Cron MA, Guillochon É, Kusner L and Le
Panse R: Role of miRNAs in normal and myasthenia gravis thymus.
Front Immunol. 11(1074)2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Villegas JA, Van Wassenhove J, Le Panse R,
Berrih-Aknin S and Dragin N: An imbalance between regulatory T
cells and T helper 17 cells in acetylcholine receptor-positive
myasthenia gravis patients. Ann N Y Acad Sci. 1413:154–162.
2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Balandina A, Lécart S, Dartevelle P,
Saoudi A and Berrih-Aknin S: Functional defect of regulatory
CD4(+)CD25+ T cells in the thymus of patients with autoimmune
myasthenia gravis. Blood. 105:735–741. 2005.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Thiruppathi M, Rowin J, Ganesh B, Sheng
JR, Prabhakar BS and Meriggioli MN: Impaired regulatory function in
circulating CD4(+)CD25(high)CD127(low/-) T cells in patients with
myasthenia gravis. Clin Immunol. 145:209–223. 2012.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Battaglia A, Di Schino C, Fattorossi A,
Scambia G and Evoli A: Circulating CD4+CD25+ T regulatory and
natural killer T cells in patients with myasthenia gravis: A flow
cytometry study. J Biol Regul Homeost Agents. 19:54–62.
2005.PubMed/NCBI
|
|
9
|
Fuchs S, Aricha R, Reuveni D and Souroujon
MC: Experimental autoimmune myasthenia gravis (EAMG): From
immunochemical characterization to therapeutic approaches. J
Autoimmun. 54:51–59. 2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Gradolatto A, Nazzal D, Truffault F,
Bismuth J, Fadel E, Foti M and Berrih-Aknin S: Both treg cells and
tconv cells are defective in the myasthenia gravis thymus: Roles of
IL-17 and TNF-α. J Autoimmun. 52:53–63. 2014.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Rodríguez-Perea AL, Arcia ED, Rueda CM and
Velilla PA: Phenotypical characterization of regulatory T cells in
humans and rodents. Clin Exp Immunol. 185:281–291. 2016.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Gavin MA, Rasmussen JP, Fontenot JD, Vasta
V, Manganiello VC, Beavo JA and Rudensky AY: Foxp3-dependent
programme of regulatory T-cell differentiation. Nature.
445:771–775. 2007.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Vrolix K, Fraussen J, Losen M, Stevens J,
Lazaridis K, Molenaar PC, Somers V, Bracho MA, Le Panse R,
Stinissen P, et al: Clonal heterogeneity of thymic B cells from
early-onset myasthenia gravis patients with antibodies against the
acetylcholine receptor. J Autoimmun. 52:101–112. 2014.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Serr I, Fürst RW, Ott VB, Scherm MG,
Nikolaev A, Gökmen F, Kälin S, Zillmer S, Bunk M, Weigmann B, et
al: miRNA92a targets KLF2 and the phosphatase PTEN signaling to
promote human T follicular helper precursors in T1D islet
autoimmunity. Proc Natl Acad Sci USA. 113:E6659–E6668.
2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Serr I, Scherm MG, Zahm AM, Schug J, Flynn
VK, Hippich M, Kälin S, Becker M, Achenbach P, Nikolaev A, et al: A
miRNA181a/NFAT5 axis links impaired T cell tolerance induction with
autoimmune type 1 diabetes. Sci Transl Med.
10(eaag1782)2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Zhou X, Jeker LT, Fife BT, Zhu S, Anderson
MS, McManus MT and Bluestone JA: Selective miRNA disruption in T
reg cells leads to uncontrolled autoimmunity. J Exp Med.
205:1983–1991. 2008.PubMed/NCBI View Article : Google Scholar
|
|
17
|
O'Connell RM, Kahn D, Gibson WS, Round JL,
Scholz RL, Chaudhuri AA, Kahn ME, Rao DS and Baltimore D:
MicroRNA-155 promotes autoimmune inflammation by enhancing
inflammatory T cell development. Immunity. 33:607–619.
2010.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Seddiki N, Brezar V, Ruffin N, Lévy Y and
Swaminathan S: Role of miR-155 in the regulation of lymphocyte
immune function and disease. Immunology. 142:32–38. 2014.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Ghafouri-Fard S, Azimi T, Hussen BM,
Taheri M and Jalili Khoshnoud R: A review on the role of non-coding
RNAs in the pathogenesis of myasthenia gravis. Int J Mol Sci.
22(12964)2021.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Wang YZ, Tian FF, Yan M, Zhang JM, Liu Q,
Lu JY, Zhou WB, Yang H and Li J: Delivery of an miR155 inhibitor by
anti-CD20 single-chain antibody into B cells reduces the
acetylcholine receptor-specific autoantibodies and ameliorates
experimental autoimmune myasthenia gravis. Clin Exp Immunol.
176:207–221. 2014.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Rosenbaum M, Gewies A, Pechloff K, Heuser
C, Engleitner T, Gehring T, Hartjes L, Krebs S, Krappmann D,
Kriegsmann M, et al: Bcl10-controlled Malt1 paracaspase activity is
key for the immune suppressive function of regulatory T cells. Nat
Commun. 10(2352)2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Yang D, Zhao X and Lin X: Bcl10 is
required for the development and suppressive function of
Foxp3+ regulatory T cells. Cell Mol Immunol. 18:206–218.
2021.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Wei TT, Cheng Z, Hu ZD, Zhou L and Zhong
RQ: Upregulated miR-155 inhibits inflammatory response induced by
C. albicans in human monocytes derived dendritic cells via
targeting p65 and BCL-10. Ann Transl Med. 7(758)2019.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Berrih-Aknin S, Frenkian-Cuvelier M and
Eymard B: Diagnostic and clinical classification of autoimmune
myasthenia gravis. J Autoimmun. 48–49, 143-148. 2014.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Jaretzki A III, Barohn RJ, Ernstoff RM,
Kaminski HJ, Keesey JC, Penn AS and Sanders DB: Myasthenia gravis:
Recommendations for clinical research standards. Task force of the
medical scientific advisory board of the myasthenia gravis
foundation of America. Neurology. 55:16–23. 2000.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Bedlack RS, Simel DL, Bosworth H, Samsa G,
Tucker-Lipscomb B and Sanders DB: Quantitative myasthenia gravis
score: Assessment of responsiveness and longitudinal validity.
Neurology. 64:1968–1970. 2005.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Baggi F, Annoni A, Ubiali F, Milani M,
Longhi R, Scaioli W, Cornelio F, Mantegazza R and Antozzi C:
Breakdown of tolerance to a self-peptide of acetylcholine receptor
alpha-subunit induces experimental myasthenia gravis in rats. J
Immunol. 172:2697–2703. 2004.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Laferriere CA and Pang DS: Review of
intraperitoneal injection of sodium pentobarbital as a method of
euthanasia in laboratory rodents. J Am Assoc Lab Anim Sci.
59:254–263. 2020.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Losen M, Martinez-Martinez P, Molenaar PC,
Lazaridis K, Tzartos S, Brenner T, Duan RS, Luo J, Lindstrom J and
Kusner L: Standardization of the experimental autoimmune myasthenia
gravis (EAMG) model by immunization of rats with Torpedo
californica acetylcholine receptors-recommendations for methods and
experimental designs. Exp Neurol. 270:18–28. 2015.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Cossarizza A, Chang HD, Radbruch A, Acs A,
Adam D, Adam-Klages S, Agace WW, Aghaeepour N, Akdis M, Allez M, et
al: Guidelines for the use of flow cytometry and cell sorting in
immunological studies (second edition). Eur J Immunol.
49:1457–1973. 2019.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Robertson B, Dalby AB, Karpilow J,
Khvorova A, Leake D and Vermeulen A: Specificity and functionality
of microRNA inhibitors. Silence. 1(10)2010.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Ha M and Kim VN: Regulation of microRNA
biogenesis. Nat Rev Mol Cell Biol. 15:509–524. 2014.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Landgraf P, Rusu M, Sheridan R, Sewer A,
Iovino N, Aravin A, Pfeffer S, Rice A, Kamphorst AO, Landthaler M,
et al: A mammalian microRNA expression atlas based on small RNA
library sequencing. Cell. 129:1401–1414. 2007.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Muljo SA, Ansel KM, Kanellopoulou C,
Livingston DM, Rao A and Rajewsky K: Aberrant T cell
differentiation in the absence of Dicer. J Exp Med. 202:261–269.
2005.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Cobb BS, Nesterova TB, Thompson E,
Hertweck A, O'Connor E, Godwin J, Wilson CB, Brockdorff N, Fisher
AG, Smale ST and Merkenschlager M: T cell lineage choice and
differentiation in the absence of the RNase III enzyme Dicer. J Exp
Med. 201:1367–1373. 2005.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Scherm MG, Serr I, Zahm AM, Schug J,
Bellusci S, Manfredini R, Salb VK, Gerlach K, Weigmann B, Ziegler
AG, et al: miRNA142-3p targets Tet2 and impairs Treg
differentiation and stability in models of type 1 diabetes. Nat
Commun. 10(5697)2019.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Alharris E, Alghetaa H, Seth R, Chatterjee
S, Singh NP, Nagarkatti M and Nagarkatti P: Resveratrol attenuates
allergic asthma and associated inflammation in the lungs through
regulation of miRNA-34a that targets FoxP3 in mice. Front Immunol.
9(2992)2018.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Kim D, Nguyen QT, Lee J, Lee SH, Janocha
A, Kim S, Le HT, Dvorina N, Weiss K, Cameron MJ, et al:
Anti-inflammatory roles of glucocorticoids are mediated by
Foxp3+ regulatory T cells via a miR-342-dependent
mechanism. Immunity. 53:581–596.e5. 2020.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Perl A: Activation of mTOR (mechanistic
target of rapamycin) in rheumatic diseases. Nat Rev Rheumatol.
12:169–182. 2016.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Lu LF, Thai TH, Calado DP, Chaudhry A,
Kubo M, Tanaka K, Loeb GB, Lee H, Yoshimura A, Rajewsky K and
Rudensky AY: Foxp3-dependent microRNA155 confers competitive
fitness to regulatory T cells by targeting SOCS1 protein. Immunity.
30:80–91. 2009.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Zheng Y, Josefowicz SZ, Kas A, Chu TT,
Gavin MA and Rudensky AY: Genome-wide analysis of Foxp3 target
genes in developing and mature regulatory T cells. Nature.
445:936–940. 2007.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Liu X, Luo M, Meng H, Zeng Q, Xu L, Hu B,
Luo Y, Liu C, Luo Z and Yang H: MiR-181a regulates CD4+
T cell activation and differentiation by targeting IL-2 in the
pathogenesis of myasthenia gravis. Eur J Immunol: Jul 26, 2019
(Epub ahead of print).
|
|
44
|
Deretic V, Saitoh T and Akira S: Autophagy
in infection, inflammation and immunity. Nat Rev Immunol.
13:722–737. 2013.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Scheinecker C, Göschl L and Bonelli M:
Treg cells in health and autoimmune diseases: New insights from
single cell analysis. J Autoimmun. 110(102376)2020.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Rao J, Li S, Wang Q, Cheng Q, Ji Y, Fu W,
Huang H, Shi L and Wu X: Comparison of peripheral blood regulatory
T cells and functional subsets between ocular and generalized
myasthenia gravis. Front Med (Lausanne). 9(851808)2022.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Kohler S, Keil TOP, Hoffmann S, Swierzy M,
Ismail M, Rückert JC, Alexander T and Meisel A: CD4+
FoxP3+ T regulatory cell subsets in myasthenia gravis
patients. Clin Immunol. 179:40–46. 2017.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Gertel-Lapter S, Mizrachi K, Berrih-Aknin
S, Fuchs S and Souroujon MC: Impairment of regulatory T cells in
myasthenia gravis: Studies in an experimental model. Autoimmun Rev.
12:894–903. 2013.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Song J, Xi JY, Yu WB, Yan C, Luo SS, Zhou
L, Zhu WH, Lu JH, Dong Q, Xiao BG and Zhao CB: Inhibition of ROCK
activity regulates the balance of Th1, Th17 and Treg cells in
myasthenia gravis. Clin Immunol. 203:142–153. 2019.PubMed/NCBI View Article : Google Scholar
|