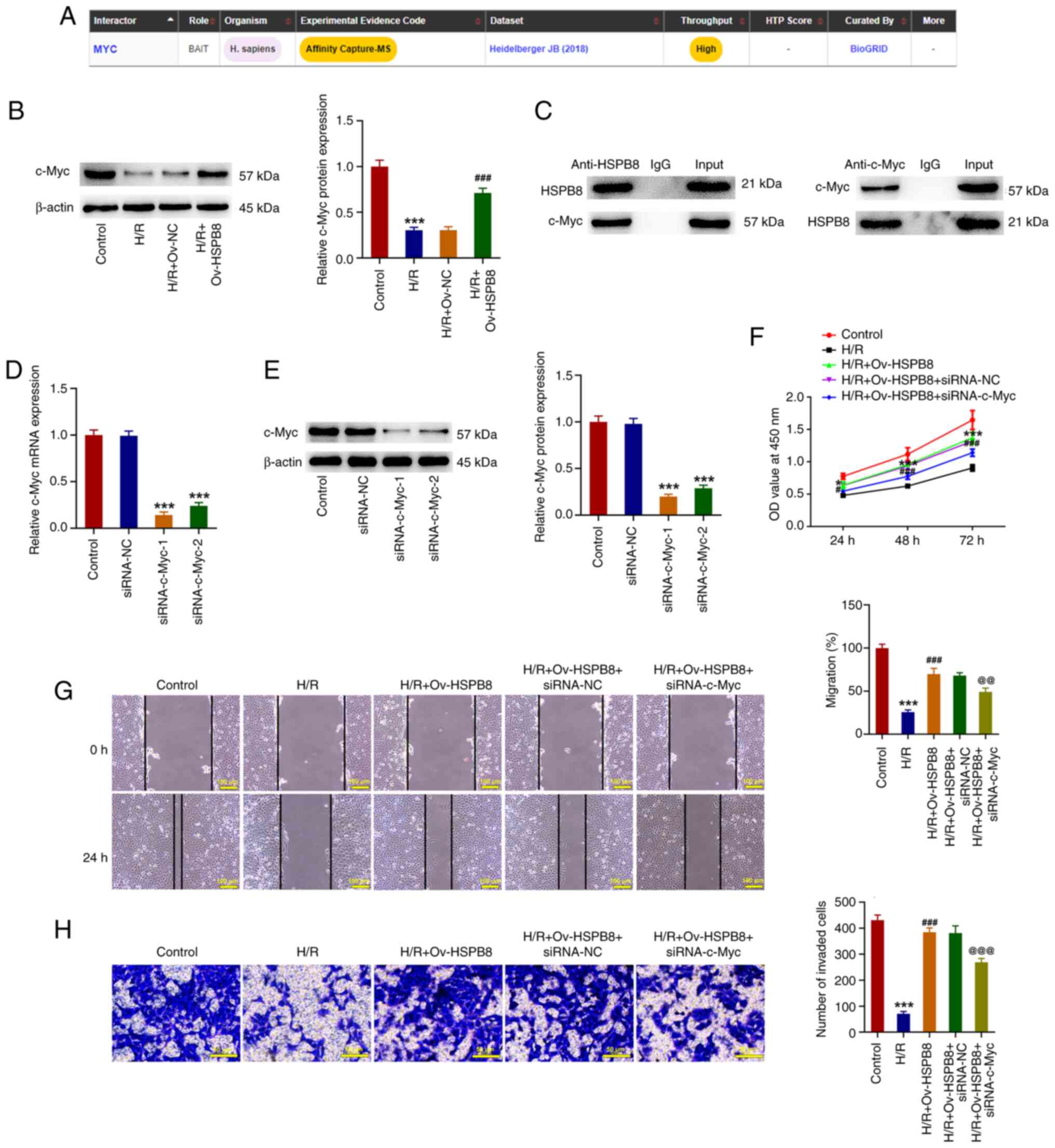
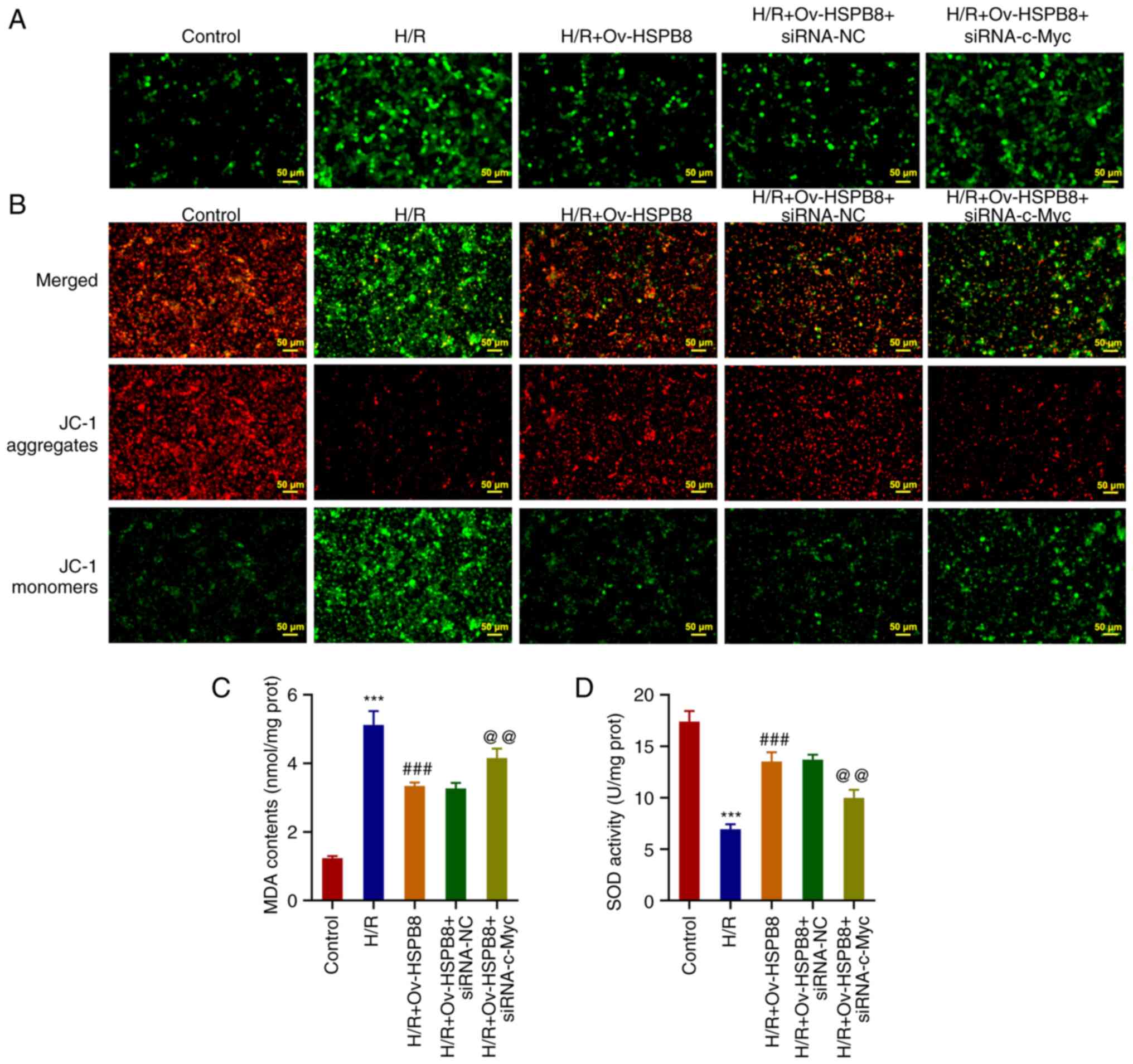
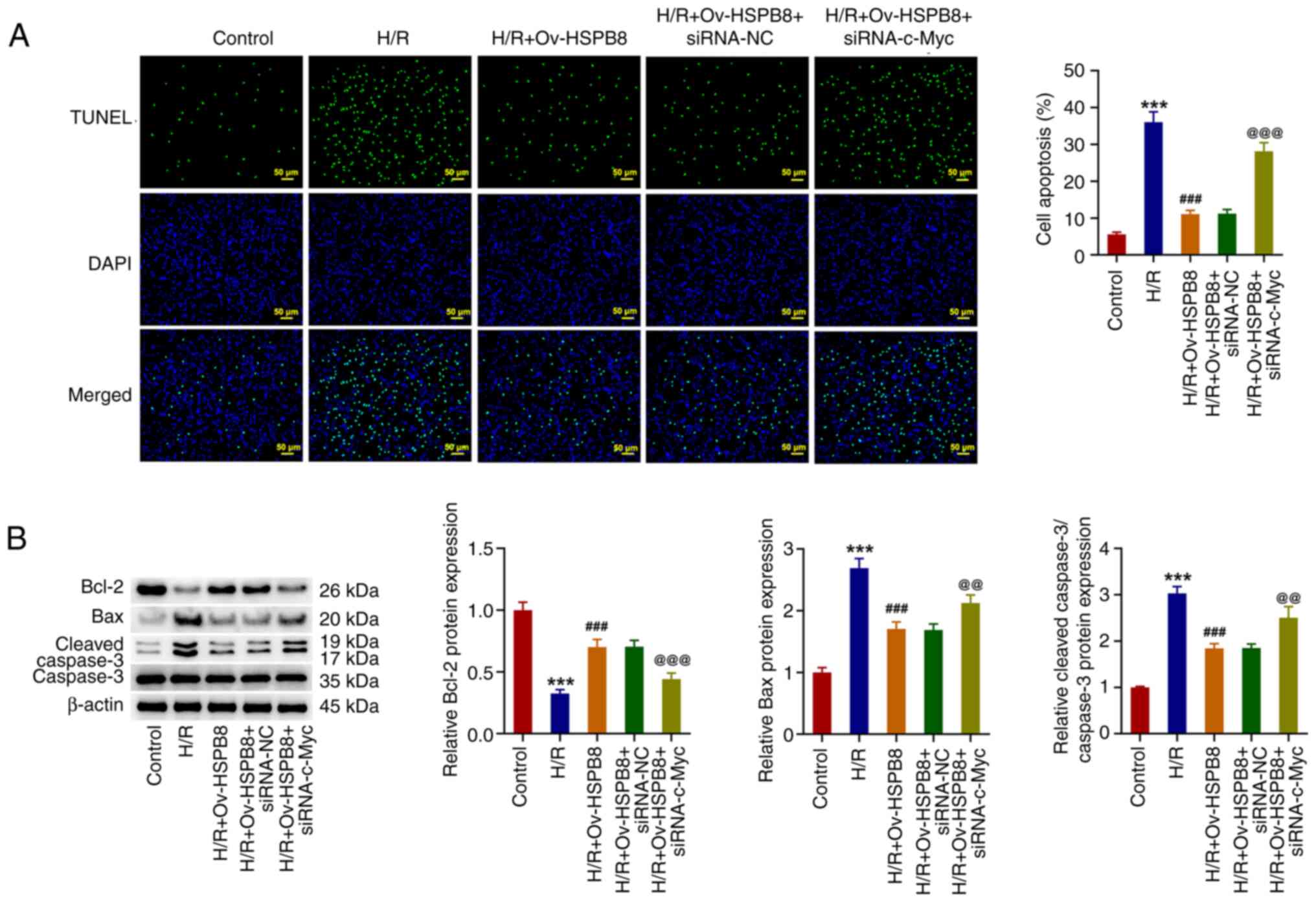
|
1
|
Chang KJ, Seow KM and Chen KH:
Preeclampsia: Recent advances in predicting, preventing, and
managing the maternal and fetal life-threatening condition. Int J
Environ Res Public Health. 20(2994)2023.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Kingwell K: RNA interference: Pre-empting
preeclampsia. Nat Rev Drug Discov. 18(18)2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Serra B, Mendoza M, Scazzocchio E, Meler
E, Nolla M, Sabrià E, Rodríguez I and Carreras E: A new model for
screening for early-onset preeclampsia. Am J Obstet Gynecol.
222:608.e1–608.e18. 2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Phipps EA, Thadhani R, Benzing T and
Karumanchi SA: Pre-eclampsia: Pathogenesis, novel diagnostics and
therapies. Nat Rev Nephrol. 15:275–289. 2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Abbas Y, Turco MY, Burton GJ and Moffett
A: Investigation of human trophoblast invasion in vitro. Hum Reprod
Update. 26:501–513. 2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Sato Y: Endovascular trophoblast and
spiral artery remodeling. Mol Cell Endocrinol.
503(110699)2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Kingdom JC, Audette MC, Hobson SR, Windrim
RC and Morgen E: A placenta clinic approach to the diagnosis and
management of fetal growth restriction. Am J Obstet Gynecol. 218
(2S):S803–S817. 2018.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Collins SL, Birks JS, Stevenson GN,
Papageorghiou AT, Noble JA and Impey L: Measurement of spiral
artery jets: General principles and differences observed in
small-for-gestational-age pregnancies. Ultrasound Obstet Gynecol.
40:171–178. 2012.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Myatt L and Roberts JM: Preeclampsia:
Syndrome or disease? Curr Hypertens Rep. 17(83)2015.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Pennington KA, Schlitt JM, Jackson DL,
Schulz LC and Schust DJ: Preeclampsia: Multiple approaches for a
multifactorial disease. Dis Model Mech. 5:9–18. 2012.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Reister F, Heyl W, Kaufmann P and Rath W:
Trophoblast invasion in pre-eclampsia. Zentralbl Gynakol.
121:587–590. 1999.PubMed/NCBI(In German).
|
|
12
|
Rana S, Lemoine E, Granger JP and
Karumanchi SA: Preeclampsia: Pathophysiology, challenges, and
perspectives. Circ Res. 124:1094–1112. 2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Zsengellér ZK, Rajakumar A, Hunter JT,
Salahuddin S, Rana S, Stillman IE and Ananth Karumanchi S:
Trophoblast mitochondrial function is impaired in preeclampsia and
correlates negatively with the expression of soluble fms-like
tyrosine kinase 1. Pregnancy Hypertens. 6:313–319. 2016.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Chiarello DI, Abad C, Rojas D, Toledo F,
Vázquez CM, Mate A, Sobrevia L and Marín R: Oxidative stress:
Normal pregnancy versus preeclampsia. Biochim Biophys Acta Mol
Basis Dis. 1866(165354)2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Dai H and Lu X: MGST1 alleviates the
oxidative stress of trophoblast cells induced by
hypoxia/reoxygenation and promotes cell proliferation, migration,
and invasion by activating the PI3K/AKT/mTOR pathway. Open Med
(Wars). 17:2062–2071. 2022.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Haram K, Mortensen JH, Myking O, Magann EF
and Morrison JC: The role of oxidative stress, adhesion molecules
and antioxidants in preeclampsia. Curr Hypertens Rev. 15:105–112.
2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Xu Z, Jin X, Cai W, Zhou M, Shao P, Yang
Z, Fu R, Cao J, Liu Y, Yu F, et al: Proteomics analysis reveals
abnormal electron transport and excessive oxidative stress cause
mitochondrial dysfunction in placental tissues of early-onset
preeclampsia. Proteomics Clin Appl. 12(e1700165)2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Yu L, Liang Q, Zhang W, Liao M, Wen M,
Zhan B, Bao H and Cheng X: HSP22 suppresses diabetes-induced
endothelial injury by inhibiting mitochondrial reactive oxygen
species formation. Redox Biol. 21(101095)2019.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Li X, Sun C, Chen J, Ma JF and Pan YH:
ERK-CREB pathway is involved in HSPB8-mediated glioma cell growth
and metastatic properties. Exp Mol Pathol. (104653)2021.PubMed/NCBI View Article : Google Scholar : (Epub ahead of
print).
|
|
20
|
Boczek EE, Fürsch J, Niedermeier ML,
Jawerth L, Jahnel M, Ruer-Gruß M, Kammer KM, Heid P, Mediani L,
Wang J, et al: HspB8 prevents aberrant phase transitions of FUS by
chaperoning its folded RNA-binding domain. Elife.
10(e69377)2021.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Matsushima-Nishiwaki R, Toyoda H,
Takamatsu R, Yasuda E, Okuda S, Maeda A, Kaneoka Y, Yoshimi N,
Kumada T and Kozawa O: Heat shock protein 22 (HSPB8) reduces the
migration of hepatocellular carcinoma cells through the suppression
of the phosphoinositide 3-kinase (PI3K)/AKT pathway. Biochim
Biophys Acta Mol Basis Dis. 1863:1629–1639. 2017.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Piccolella M, Crippa V, Cristofani R,
Rusmini P, Galbiati M, Cicardi ME, Meroni M, Ferri N, Morelli FF,
Carra S, et al: The small heat shock protein B8 (HSPB8) modulates
proliferation and migration of breast cancer cells. Oncotarget.
8:10400–10415. 2017.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Shu B, Zhou Y, Liang Q, He C and Li F:
HSPB8 promoted intrahepatic cholangiocarcinoma progression by
enhancing epithelial-mesenchymal transition and autophagy. Exp Mol
Pathol. 123(104691)2021.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Li F, Yang B, Li T, Gong X, Zhou F and Hu
Z: HSPB8 over-expression prevents disruption of blood-brain barrier
by promoting autophagic flux after cerebral ischemia/reperfusion
injury. J Neurochem. 148:97–113. 2019.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Azar C, Valentine MC, Trausch-Azar J, Rois
L, Mahjoub M, Nelson DM and Schwartz AL: RNA-Seq identifies genes
whose proteins are upregulated during syncytia development in
murine C2C12 myoblasts and human BeWo trophoblasts. Physiol Rep.
9(e14671)2021.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Dhanasekaran R, Deutzmann A,
Mahauad-Fernandez WD, Hansen AS, Gouw AM and Felsher DW: The MYC
oncogene-the grand orchestrator of cancer growth and immune
evasion. Nat Rev Clin Oncol. 19:23–36. 2022.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Sun M, Chen H, Liu J, Tong C and Meng T:
MicroRNA-34a inhibits human trophoblast cell invasion by targeting
MYC. BMC Cell Biol. 16(21)2015.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Hu X, Ao J, Li X, Zhang H, Wu J and Cheng
W: Competing endogenous RNA expression profiling in pre-eclampsia
identifies hsa_circ_0036877 as a potential novel blood biomarker
for early pre-eclampsia. Clin Epigenetics. 10(48)2018.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Hu X, Li X, Tian GG, Zhang H, Cheng W and
Wu J: Expression profiling dataset of competing endogenous RNA in
pre-eclampsia. Data Brief. 27(104795)2019.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Medina-Bastidas D, Guzmán-Huerta M,
Borboa-Olivares H, Ruiz-Cruz C, Parra-Hernández S, Flores-Pliego A,
Salido-Guadarrama I, Camargo-Marín L, Arambula-Meraz E and
Estrada-Gutierrez G: Placental microarray profiling reveals common
mRNA and lncRNA expression patterns in preeclampsia and
intrauterine growth restriction. Int J Mol Sci.
21(3597)2020.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Yang Z, Bai B, Luo X, Xiao X, Liu X, Ding
Y, Zhang H, Gao L, Li J and Qi H: Downregulated Krüppel-like factor
8 is involved in decreased trophoblast invasion under
hypoxia-reoxygenation conditions. Reprod Sci. 21:72–81.
2014.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Thompson LP, Pence L, Pinkas G, Song H and
Telugu BP: Placental hypoxia during early pregnancy causes maternal
hypertension and placental insufficiency in the hypoxic guinea pig
model. Biol Reprod. 95(128)2016.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Masturzo B, Di Martino D, Prefumo F,
Cavoretto P, Germano C, Gennarelli G, Roletti E, Bottazzoli E, Fusè
F, Ferrazzi E, et al: Higher rate of early-onset preeclampsia in
pregnancies following oocyte donation according to increasing
maternal age. Arch Gynecol Obstet. 300:861–867. 2019.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Fox R, Kitt J, Leeson P, Aye CYL and
Lewandowski AJ: Preeclampsia: Risk factors, diagnosis, management,
and the cardiovascular impact on the offspring. J Clin Med.
8(1625)2019.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Merviel P, Carbillon L, Challier JC,
Rabreau M, Beaufils M and Uzan S: Pathophysiology of preeclampsia:
Links with implantation disorders. Eur J Obstet Gynecol Reprod
Biol. 115:134–147. 2004.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Huppertz B: The critical role of abnormal
trophoblast development in the etiology of preeclampsia. Curr Pharm
Biotechnol. 19:771–780. 2018.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Hemberger M: Health during pregnancy and
beyond: Fetal trophoblast cells as chief co-ordinators of
intrauterine growth and reproductive success. Ann Med. 44:325–337.
2012.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Sánchez-Aranguren LC, Prada CE,
Riaño-Medina CE and Lopez M: Endothelial dysfunction and
preeclampsia: role of oxidative stress. Front Physiol.
5(372)2014.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Murata M, Fukushima K, Takao T, Seki H,
Takeda S and Wake N: Oxidative stress produced by xanthine oxidase
induces apoptosis in human extravillous trophoblast cells. J Reprod
Dev. 59:7–13. 2013.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Staff AC: The two-stage placental model of
preeclampsia: An update. J Reprod Immunol. 134-135:1–10.
2019.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Yang HY: MiR-133b regulates oxidative
stress injury of trophoblasts in preeclampsia by mediating the
JAK2/STAT3 signaling pathway. J Mol Histol. 52:1177–1188.
2021.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Xu X, Ye X, Zhu M, Zhang Q, Li X and Yan
J: FtMt reduces oxidative stress-induced trophoblast cell
dysfunction via the HIF-1α/VEGF signaling pathway. BMC Pregnancy
Childbirth. 23(131)2023.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Zhang K, Yin W, Ma L, Liu Z and Li Q:
HSPB8 facilitates prostate cancer progression via activating the
JAK/STAT3 signaling pathway. Biochem Cell Biol. 101:1–11.
2023.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Shen J, Li M and Min L: HSPB8 promotes
cancer cell growth by activating the ERK-CREB pathway and is
indicative of a poor prognosis in gastric cancer patients. Oncol
Rep. 39:2978–2986. 2018.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Yu LL, Wang Y, Xiao ZK and Chen SS: Heat
shock protein B8 promotes proliferation and migration in lung
adenocarcinoma A549 cells by maintaining mitochondrial function.
Mol Cell Biochem. 476:187–197. 2021.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Cheng J, Ji M, Jing H and Lin H: DUSP12
ameliorates myocardial ischemia-reperfusion injury through
HSPB8-induced mitophagy. J Biochem Mol Toxicol.
37(e23310)2023.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Li F, Tan J, Zhou F, Hu Z and Yang B: Heat
shock protein B8 (HSPB8) reduces oxygen-glucose
deprivation/reperfusion injury via the induction of mitophagy. Cell
Physiol Biochem. 48:1492–1504. 2018.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Duffy MJ, O'Grady S, Tang M and Crown J:
MYC as a target for cancer treatment. Cancer Treat Rev.
94(102154)2021.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Fatma H, Maurya SK and Siddique HR:
Epigenetic modifications of c-MYC: Role in cancer cell
reprogramming, progression and chemoresistance. Semin Cancer Biol.
83:166–176. 2022.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Dai M, Song J, Wang L, Zhou K and Shu L:
HOXC13 promotes cervical cancer proliferation, invasion and Warburg
effect through β-catenin/c-Myc signaling pathway. J Bioenerg
Biomembr. 53:597–608. 2021.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Shan N, Ni LY, Zhu YS, Shen Y, Gao ZJ and
Cai ZZ: Girdin influences pancreatic cancer progression by
regulating c-MYC expression. Neoplasma. 69:193–202. 2022.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Ali A, Ali A, Devi HS, Daddam JR, Sarwar R
and Badrealam FK: The HDAC2/YY1/c-Myc signaling axis regulates lung
cancer cell migration and proliferation. Environ Toxicol.
38:1989–2001. 2023.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Wu Z, Cheng S, Wang S, Li W and Liu J:
C-MYC ameliorates ventricular remodeling of myocardial infarction
rats via binding to the promoter of microRNA-29a-3p to facilitate
TET2 expression. Int J Cardiol. 357:105–112. 2022.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Prieto J, García-Cañaveras JC, León M,
Sendra R, Ponsoda X, Izpisúa Belmonte JC, Lahoz A and Torres J:
c-MYC triggers lipid remodelling during early somatic cell
reprogramming to pluripotency. Stem Cell Rev Rep. 17:2245–2261.
2021.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Kfoury A, Armaro M, Collodet C,
Sordet-Dessimoz J, Giner MP, Christen S, Moco S, Leleu M, de Leval
L, Koch U, et al: AMPK promotes survival of c-Myc-positive melanoma
cells by suppressing oxidative stress. EMBO J.
37(e97673)2018.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Pu HH, Duan J, Wang Y, Fan DX, Li DJ and
Jin LP: Thymic stromal lymphopoietin promotes the proliferation of
human trophoblasts via phosphorylated STAT3-mediated c-Myc
upregulation. Placenta. 33:387–391. 2012.PubMed/NCBI View Article : Google Scholar
|