1. Introduction
Endometriosis is a common female disorder, affecting
up to 15% of all women (1). in
which endometrial tissue adheres and proliferates ectopically to
other organs outside the uterus, usually in the abdominal cavity
(2). Classical hypotheses for the
pathogenesis of endometriosis include the retrograde menstruation
theory and the theory of vascular or lymphatic metastasis, while
recent research has revealed the emerging role of polygenic and
epigenetic mechanisms (3-5).
Furthermore, various factors contribute to the pathogenesis of
endometriosis, including inflammation, immunological factors,
angiogenesis, and resistance to apoptosis (2,6).
Protein homeostasis in cells is finely regulated by
a variety of mechanisms, and it affects a wide range of cellular
functions, including regulation of cell proliferation,
differentiation, senescence, death, and cell-cell interactions
(7,8). The unfolded protein response (UPR),
also known as endoplasmic reticulum (ER) stress, is one of the key
mechanisms for maintaining proteostasis by removing proteins that
are abnormally folded during synthesis in the ER (9,10).
Recently, it has been reported that the levels of UPR-related mRNA
are higher than normal in patients with endometriosis (11,12),
with ER stress being associated with the production of inflammatory
cytokines in the endometrium (13)
and the invasive behavior of endometrial stromal cells (14). ER stress in endometrial cells is
also influenced by the menstrual cycle, being suppressed by
estrogen and heightened by progesterone (15). Since a sufficiently excessive UPR
can directly induce apoptosis, researchers are investigating its
potential use as a treatment option for endometriosis (16).
Myrrh, a gum resin derived from Commiphora
myrrha (Nees) English, a small thorny tree belonging to the
genus Commiphora, has a rich historical background of use in
both the Eastern and Western cultures for making perfume, incense,
and medicinal remedies (17-19).
Traditionally, myrrh has been employed to alleviate various
conditions, including wounds, pain, fractures, mouth ulcers,
arthritis, gastrointestinal disorders, and infections (18,19).
Recent studies have reported a range of effects associated with
myrrh and its chemical compounds, including anti-inflammatory,
anti-cancer, analgesic, antioxidant, and anti-microbial properties
(19-22).
Two studies have reported that myrrh-containing nutrient
supplements, composed of alpha-lipoic acid and
palmitoylethanolamide, are effective in alleviating pain in
patients with endometriosis (23,24).
However, whether myrrh inhibits endometriosis foci themselves has
not yet been studied.
In this study, we verified that myrrh inhibits the
growth of endometriosis in an animal model and used RNA sequencing
to identify the underlying molecular mechanisms. The results
revealed that myrrh increased UPR and promoted apoptosis.
Co-treatment with a UPR inhibitor reduced the apoptotic effect of
myrrh. Therefore, we suggest myrrh to be a potent and promising
candidate for the development of novel drugs aimed at combating
endometriosis.
Materials and methods
Preparation of myrrh extract
Water-extracted myrrh was purchased from the Korea
Plant Extract Bank (KPEB), located at the Korea Research Institute
of Bioscience and Biotechnology (Chungju, Korea). The original
plant, C. myrrha, was cultivated in India, and the
corresponding specimen was deposited in the KPEB (lot number:
PBC-124AS). The myrrh extract was dissolved in dimethylsulphoxide
(Sigma-Aldrich; Merck KGaA) and immediately reconstituted with
culture media for in vitro experiments or corn oil for animal
studies.
Network pharmacologic analysis
The Traditional Chinese Medicine Integrative
Database (TCMID, http://bidd.group/TCMID/) (25) and Herbal Ingredients' Targets
Platform 2.0 (HIT 2.0, http://hit2.badd-cao.net/) (26) were used to identify possible target
genes for myrrh. Potential pathways associated with myrrh were
investigated using Cytoscape with the aid of the JEPPETO plugin
(27,28). Visualization was performed by
constructing a scatterplot with the XD-score and q-value as the
axes. The DisGeNet database (https://www.disgenet.org/) (29) was employed to identify potential
target genes for endometriosis. A Venn diagram was generated to
illustrate the 26 genes shared by the prospective targets of myrrh
and endometriosis. These common genes were then analyzed to
determine the protein-protein interaction (PPI) network of the
target genes using Cytoscape (version 3.9.1, https://cytoscape.org/) with the STRING application
(30).
LC-MS analysis
C. myrrha was extracted with 100% methanol at
room temperature to obtain the methanol extract. The extract was
suspended in water and successively partitioned with n-hexane,
methylene chloride (CH2Cl2; MC) and water.
Each solvent fraction was filtered and concentrated under reduced
pressure to yield the n-hexane, MC and water fractions. An aliquot
(5 mg) of each fraction was dissolved in 1 mL of acetonitrile and
filtered through a 0.20 µm hydrophilic PTFE membrane filter
(Advantec) prior to LC-MS analysis. LC-MS analysis was performed
using an Agilent 1290 Infinity II UPLC system (Agilent
Technologies, Inc.) coupled with a SCIEX ZenoTOF 7600 mass
spectrometer (SCIEX). Chromatographic separation was conducted on a
Phenomenex Kinetex XB-C18 column (1.7 µm, 50x2.1 mm; Phenomenex).
The mobile phase consisted of HPLC grade water containing 0.1%
formic acid (solvent A) and HPLC grade acetonitrile containing 0.1%
formic acid (solvent B). The gradient elution program was as
follows: isocratic at 30% B (0-0.5 min), 30-100% B (0.5-10 min),
followed by washing with 100% B (10-18 min) and reconditioning with
30% B for 2 min. The flow rate was set to 0.3 ml/min, and the
injection volume was 1 µl. UV detection was performed at 200 nm. MS
data were collected in negative ionization mode using a TOF/MS
acquisition. The TOF/MS ranges were set to 100-1,000 Da. The ion
source parameters were as follows: curtain gas 35 psi; CAD gas 7;
ion source gas 50 psi; source temperature 500˚C; spray voltage
-4,500 V. The chemical formulas of detected compounds were
predicted using the Formula Finder function in SCIEX OS software.
All solvents were analytical grade and purchased from Daejung
Chemicals. The raw and processed LC-MS datasets generated in this
study are available in the Harvard Dataverse repository at
https://dataverse.harvard.edu/dataset.xhtml?persistentId=doi:10.7910/DVN/UUSTOQ
Animal care
Five-week-old female C57BL/6 mice were procured from
Orient Bio and housed in animal facilities maintained by the
Institute of Experimental Animal Research at the Pusan National
University. The animal facility maintained a temperature of 22±2˚C,
with 50-60% humidity levels and a 12-h light/dark cycle. Anesthesia
and euthanasia were performed via inhalation of isoflurane and
CO2 exposure, respectively. All experimental procedures
were approved by the Institutional Animal Care and Use Committee of
the Pusan National University (PNU-2020-2679, Busan, Korea).
Mouse model of endometriosis
Endometriosis was induced based on previously
published protocol (31). To
eliminate variations associated with the estrous cycle, ovariectomy
was performed on 6-week-old female C57BL/6 mice on day -21,
followed by a recovery period. Two weeks post-ovariectomy (day -7),
the mice received a subcutaneous injection of 100 mg/kg β-estradiol
(Santa Cruz Biotechnology, Inc.) to provide hormonal support. On
day 0, uteri were harvested from syngeneic donor mice, minced using
Gentle Max (Miltenyi Biotec), and intraperitoneally inoculated into
recipient mice at a 1:1 ratio. One day after transplantation, myrrh
was administered orally five days per week, and 100 mg/kg
β-estradiol was injected subcutaneously once per week for three
weeks.
The doses of myrrh used in animal study were
determined based on the in vitro potency in 12Z cells. The GI50
(the concentration causing 50% growth inhibition) at 24 h, which
was 38.22 µg/ml, which we converted to a mouse dose using the
following formula (32).
Dose (µg/kg)=Concentration (µg/ml) x
Dosing volume (ml/kg)
with a standard oral gavage volume of 10 ml/kg (200
µl/20 g). This GI50-converted dose was 0.382 mg/kg (38.22 µg/ml X
10 ml/kg). To span a conservative range around the GI50-converted
dose, we used 0.7 mg/kg (~2X) and 3.5 mg/kg (~10X) once daily.
Mice were euthanized under CO2 inhalation
at a volume displacement rate of 50% one day after the final
β-estradiol injection, and the number and weight of endometriotic
lesions were assessed. Each experimental group comprised five
mice.
Terminal deoxynucleotidyl transferase
dUTP nick end labeling (TUNEL) assay
Endometriotic lesions were fixed with 4%
paraformaldehyde at 4˚C for 30 min. Labeling was performed using
the TUNEL Assay Kit-BrdU-Red (ab66110; Abcam). Labeled apoptotic
cells were imaged at Ex/Em 488/576 nm by fluorescence microscopy
(Microscope stand Axio Observer 7, Zeiss).
Cell culture
Immortalized human endometrial stromal T-HESCs were
obtained from the American Type Culture Collection (#CRL-4003), and
immortalized human endometriotic 12Z cells were purchased from
Applied Biological Materials (#T0764). T-HESCs were cultured in
phenol red-free DMEM/F12 (Thermo Fisher Scientific, Inc.)
supplemented with 10% charcoal-filtered (Sigma-Aldrich; Merck KGaA)
fetal bovine serum (FBS; Thermo Fisher Scientific, Inc.), 1% ITS
premix (BD Biosciences), and 1% penicillin/streptomycin (Thermo
Fisher Scientific, Inc.). 12Z cells were maintained in DMEM/F12
supplemented with 10% charcoal-filtered FBS and 1%
penicillin/streptomycin. Both cell types were cultured at 37˚C in a
humidified atmosphere containing 5% CO2.
Cell death and apoptosis
Cell viability was assessed using
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT;
Sigma-Aldrich; Merck KGaA). Myrrh was added at indicated
concentrations to 12Z cells seeded in 96-well plates. Cell
viability was determined by measuring absorbance at 450 nm using a
SPARK spectrophotometer (Tecan). The 50% growth inhibition dose
(GI50) was calculated using GraphPad Prism software
(GraphPad). Apoptosis was assessed using a propidium iodide
(PI)/Annexin V assay kit (BD Biosciences). 12Z cells were plated in
six-well plates for PI/Annexin V detection. Fluorescence-activated
cell sorting (FACS) analysis was performed to assess the percentage
of PI/Annexin V-positive cells at excitation (Ex) 494/emission (Em)
525 nm for Annexin V, using Attune X (Thermo Fisher Scientific,
Inc.), and at Ex 535/Em 617 nm for PI. The FlowJo application (BD
Biosciences) was used to analyze the FACS data.
Immunoblot analysis
Immunoblot analysis was used to investigate
apoptotic signaling proteins in the cells. RIPA buffer (Thermo
Fisher Scientific, Inc.) containing a protease inhibitor cocktail
(Roche) was used to extract total proteins, and the protein
concentration was measured using the Bradford assay. The proteins
were separated using sodium dodecyl sulfate-polyacrylamide gel
electrophoresis and transferred onto polyvinylidene fluoride
membranes (Amersham Bioscience). Subsequently. the membranes were
subjected to overnight incubated at 4˚C with primary antibodies,
including anti-human poly (ADP-ribose) polymerase (PARP, #9542s;
Cell Signaling Technology, Inc.), caspases-3 (#9665s; Cell
Signaling Technology, Inc.), caspase-9 (#9508s; Cell Signaling
Technology, Inc.), Bax (NB100-56095; Novus Biologicals), Bcl-2
(NB100-56098; Novus Biologicals), p53 (sc-6243; Santa Cruz
Biotechnology, Inc.) and glyceraldehyde 3-phosphate dehydrogenase
(GAPDH, sc-32233; Santa Cruz Biotechnology, Inc.). Following
thorough washing with Tris-buffered saline, horseradish
peroxidase-conjugated secondary antibodies, including anti-rabbit
IgG or anti-mouse IgG (Invitrogen; Thermo Fisher Scientific, Inc.),
were applied to the membranes at room temperature for 1 h. Specific
bands were developed using the ImageQuant LAS 4000
chemiluminescence imaging equipment (GE Healthcare) and an
Immunoblot detection kit from Bio-Rad.
In situ crosslinking
Cells were washed twice with PBS and incubated with
freshly prepared 3.6% formaldehyde in PBS at room temperature for
5-10 min. Crosslinking was quenched by addition of 1.25 M glycine
to a final concentration of 0.125 M and incubated 5 min at room
temperature, followed by three washes with PBS. Cells were lysed in
NP-40 lysis buffer (50 mM Tris-HCl pH 7.5, 150 mM NaCl, 1% NP-40,
protease inhibitor cocktail) on ice for 15-30 min and cleared by
centrifugation (12,000 x g, 10 min, 4˚C). Protein concentration was
measured (BCA), and equal amounts of protein were mixed with
non-reducing Laemmli sample buffer. Proteins were separated on
SDS-PAGE, transferred to PVDF membranes, and immunoblotted with
primary antibodies as indicated.
RNA sequencing
12Z cells were cultured at 5x105/well in
six-well plates and treated with either vehicle (DMSO) or Myrrh
(100 µg/ml) for 12 h. Cells cultured in three different wells for
each treatment group were used for RNA sequencing, conducted by DNA
Link (Seoul, Korea) services. The total RNA quality was assessed
using a 2100 Expert Bioanalyzer with an RNA 6000 Nano Kit (Agilent
Technologies, Inc.), ensuring that the RNA integrity level of all
samples exceeded 7.0.
The raw RNA sequencing data have been deposited in
the NCBI Gene Expression Omnibus (GEO) under accession number
GSE246172 (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE246172).
Gene set enrichment analysis
(GSEA)
GSEA was performed using the Broad Institute GSEA
software (version 4.3.0, available at http://www.gsea-msigdb.org/gsea/downloads.jsp) as
described previously (33,34). For Hallmark and KEGG results,
statistical analysis was performed using |NES|>1 and FDR q-value
<0.25 as cutoff values. The biological processes of the
Differentially expressed genes (DEGs) were analyzed using Hallmark,
the Kyoto Encyclopedia of Genes and Genomes (KEGG), and Gene
Ontology (GO). Results of network analysis of GO genes with
P<0.001 and FDR Q<0.5 cutoff values were displayed using
Cytoscape.
Differentially expressed gene and
heatmap analysis
Transcriptome analysis was performed as previously
described using the R program DESeq2 (version 4.1.1, https://www.r-project.org/) (34,35).
Genes with P-values and Log2|FoldChange (FC)| values less than 0.05
and greater than 1, were identified as DEGs. Heat maps for the 156
DEGs (134 upregulated and 22 downregulated) identified in the GSEA
related to endometriosis pathways were generated using Morpheus, a
versatile matrix visualization and analysis program from the Broad
Institute (https://software.broadinstitute.org/morpheus/).
Quantitative RT-PCR
Total RNA was extracted from 12Z cells using the
GeneJET RNA Purification Kit (Thermo Fisher Scientific). An equal
quantity of total RNA (1 µg) from each sample were subjected to
reverse transcription using oligo-dT primers and M-MLV reverse
transcriptase (Thermo Fisher Scientific). Quantitative real-time
PCR was conducted through the StepOnePlus Real-Time PCR system
(Applied Biosystems) using the RealHelix qPCR kit (NanoHelix). The
relative mRNA levels were normalized to the 18S rRNA levels, and
the primer details used in this study are listed in Table I.
 | Table ISequence of primers used for reverse
transcription-quantitative PCR. |
Table I
Sequence of primers used for reverse
transcription-quantitative PCR.
| Gene | Forward
(5'-3') | Reverse
(5'-3') |
|---|
|
PPP1R15A |
TCCTCTGGCAATCCCCCATA |
TGGTTTTCAGCCCCAGTGTT |
| DDIT3 |
CTGGAAAGCAGCGCATGAAG |
CGCTCGATTTCCTGCTTGAG |
| XBP1 |
CTGCCAGAGATCGAAAGAAGGC |
CTCCTGGTTCTCAACTACAAGGC |
| EIF2AK3 |
GTCCCAAGGCTTTGGAATCTGTC |
CCTACCAAGACAGGAGTTCTGG |
| ATF6 |
AGCAGGAACTCAGGGAGTGA |
GGTAGCTGGTAACAGCAGGT |
| ERN1 |
GGACAGTGAATCTGGGGACG |
GGTCTCCACAGCGACATTGA |
| TRAF6 |
ATCCCACGGAACCCAAAAGG |
CTCCGAAGGCTACCCATGTC |
| ATF4 |
TCAGTCCCTCCAACAACAGC |
TCTGGCATGGTTTCCAGGTC |
| 18S
rRNA |
GTAACCCGTTGAACCCCATT |
CCATCCAATCGGTAGTAGCG |
Hematoxylin and eosin (H&E)
staining
Kidneys and livers were removed from euthanised mice
at the end of the experiment. Tissues were fixed in formaldehyde,
embedded in paraffin blocks and sectioned by microtome. Sections
were deparaffinised, dehydrated and embedded in H&E solution
for histopathological examination.
Statistics
The mean and standard error of the mean were used to
express the values of the numerical data. The two-tailed Student's
t-test was used for comparisons between two different groups, while
one-way analysis of variance and Tukey's post hoc tests were used
for comparisons between multiple groups. These analyses were
performed using the GraphPad Prism software package.
Results
Network pharmacological prediction of
the anti-endometriosis effects of myrrh
The list of potential myrrh target genes obtained
from the TCMID and HIT databases (Table SI) was used for network analysis
using the JEPPTO plug-in of Cytoscape software. The results
revealed significant alterations in apoptosis, mitochondrial
damage, proteolytic regulation, stress response, endometrial
cancer, and reproductive tract development, which could be
associated with endometriosis (Fig.
S1A, B, and C; Table
SII, Table SIII, Table SIV, Table SV and Table SVI) (36-38).
Therefore, we postulated that myrrh might be a potential drug for
treating endometriosis, and this hypothesis was confirmed by
comparing myrrh target genes with endometriosis-related genes in
the DisGeNet database (Table
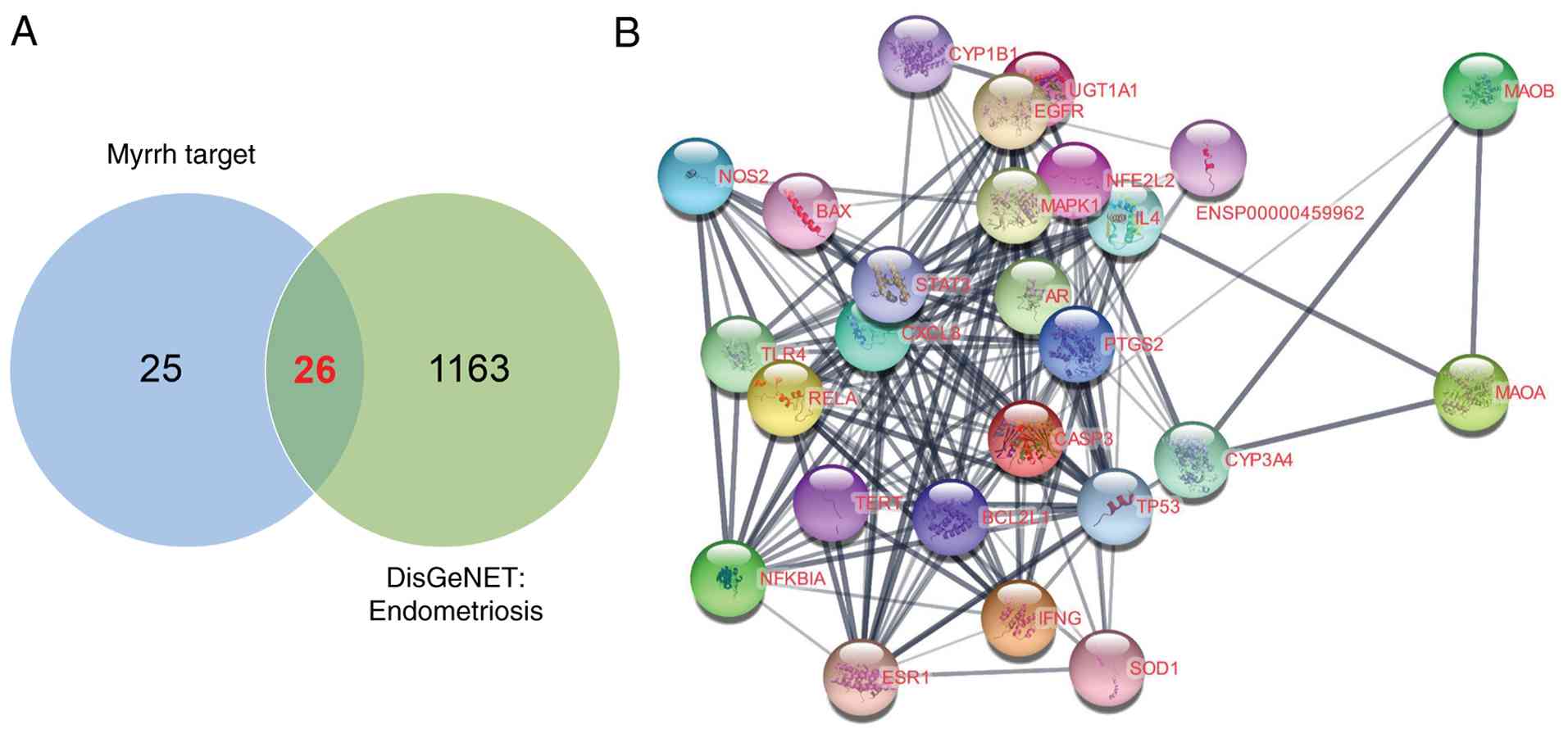
SVII). Twenty-six genes were identified as common targets of
endometriosis and myrrh, representing over half of the potential
myrrh targets (Fig. 1A). These 26
genes exhibited strong PPIs, including AR, BAX, BCL2L1, CASP3,
CXCL8, CYP1B1, CYP3A4, EGFR, ESR1, IFNG, IL4, MAPK1, NFE2L2,
NFKBIA, NOS2, PTGS2, RELA, SOD1, STAT3, TERT, TLR4, TP53,
TRPV1, and UGT1A1, with more than four interactions
(Fig. 1B). These nodes are
involved in key biological processes, including apoptosis and cell
survival (BAX, BCL2L1, CASP3, TP53), inflammation and immune
response (CXCL8, IFNG, IL4, NOS2, RELA, STAT3, TLR4, NFKBIA),
hormone signaling (AR, ESR1, EGFR), drug metabolism and
detoxification (CYP1B1, CYP3A4, UGT1A1, NFE2L2, SOD1), telomere
maintenance (TERT), pain signaling (TRPV1), and signaling pathways
(MAPK1).
Profiling of C. myrrha extract by
LC-MS analysis
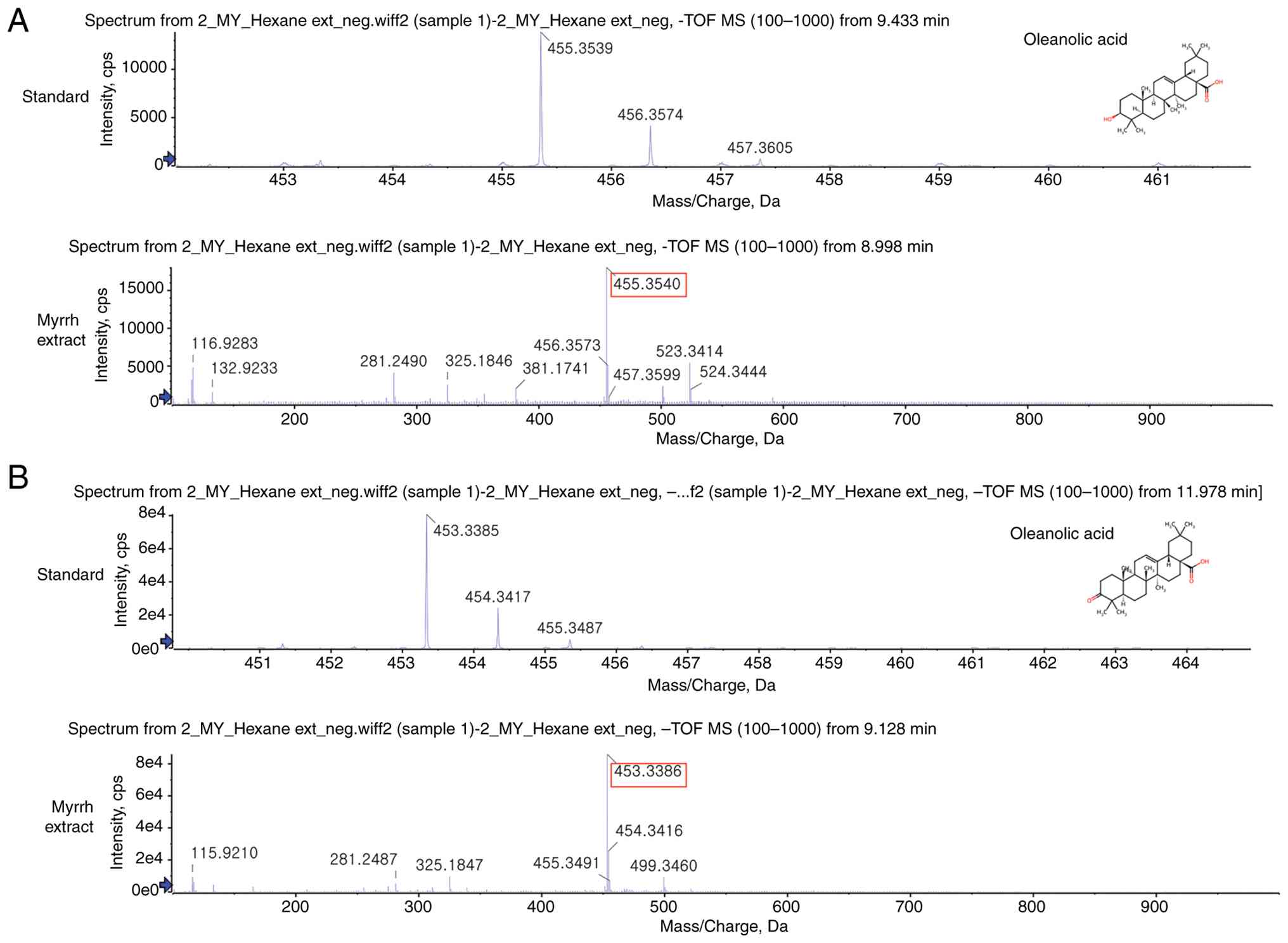
LC-MS analysis performed to confirm the chemical
composition of C. myrrha extract detected oleanolic acid and
oleanonic acid, triterpenoid compounds with anti-inflammatory
activity (Fig. 2). Oleanolic acid
and oleanonic acid have been previously reported as major bioactive
triterpenoids present in C. myrrha resin (19,39).
The detection of these specific acids highlights the potential
bioactivity of the extract, supports its traditional and
pharmacological relevance, contributes to the chemical profiling of
C. myrrha, and provides valuable data for further research
on therapeutic applications and quality control standards in
natural product studies.
Effects of myrrh on murine models of
endometriosis
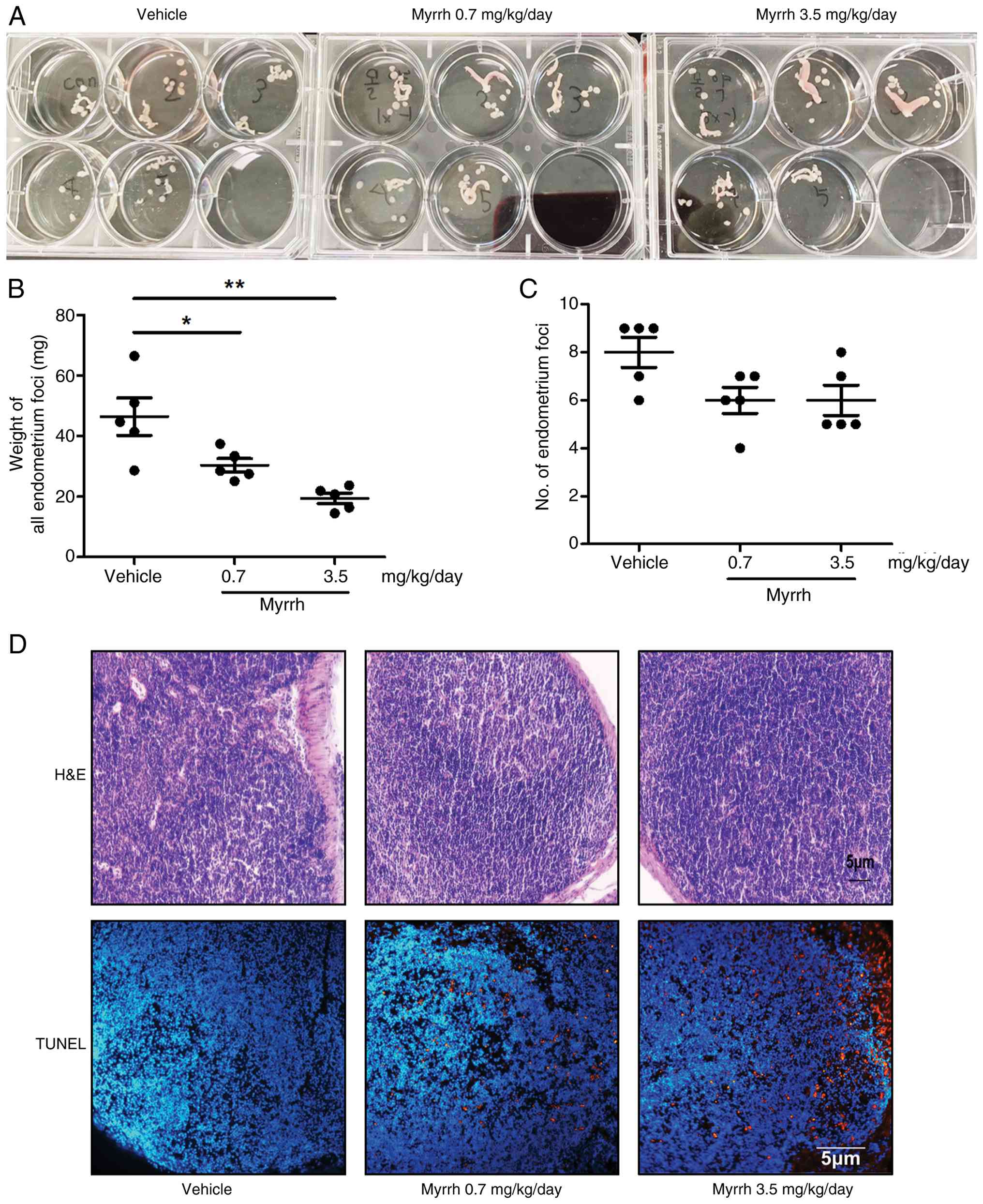
To determine the efficacy of myrrh against
endometriosis, we established an endometriosis model using
allogeneic uterine transplantation in genetically identical
immunocompetent mice. Starting the day after inducing experimental
endometriosis, myrrh was administered orally five times a week at
two concentrations (0.7 or 3.5 mg/kg/day). Three weeks after myrrh
administration, we examined the number and weight of endometriotic
foci, which are cyst-like ectopic lesions attached to the
intestinal or intraperitoneal tissues. Compared to the vehicle,
myrrh significantly reduced the weight of ectopic endometriosis
foci at both high and low concentrations (Fig. 3A and B). However, no significant reduction was
observed in the number of endometriotic foci (Fig. 3C). The Terminal deoxynucleotidyl
transferase dUTP nick end labeling (TUNEL) assay was used to
confirm apoptosis in ectopic lesions. The drug was found to cause
apoptosis at both low and high concentrations (Fig. 3D). As a brief confirmation of
safety at effective concentrations, microscopic examination of
hematoxylin and eosin-stained sections of kidney and liver tissue
confirmed that the dose of myrrh used in the in vivo experiments
did not exhibit significant toxicity (Fig. S2A and B). These results suggested that oral
administration of myrrh might inhibit the growth of ectopic
endometriotic tissues, as hypothesized by network pharmacology.
The pro-apoptotic effects of myrrh on
endometriosis cells
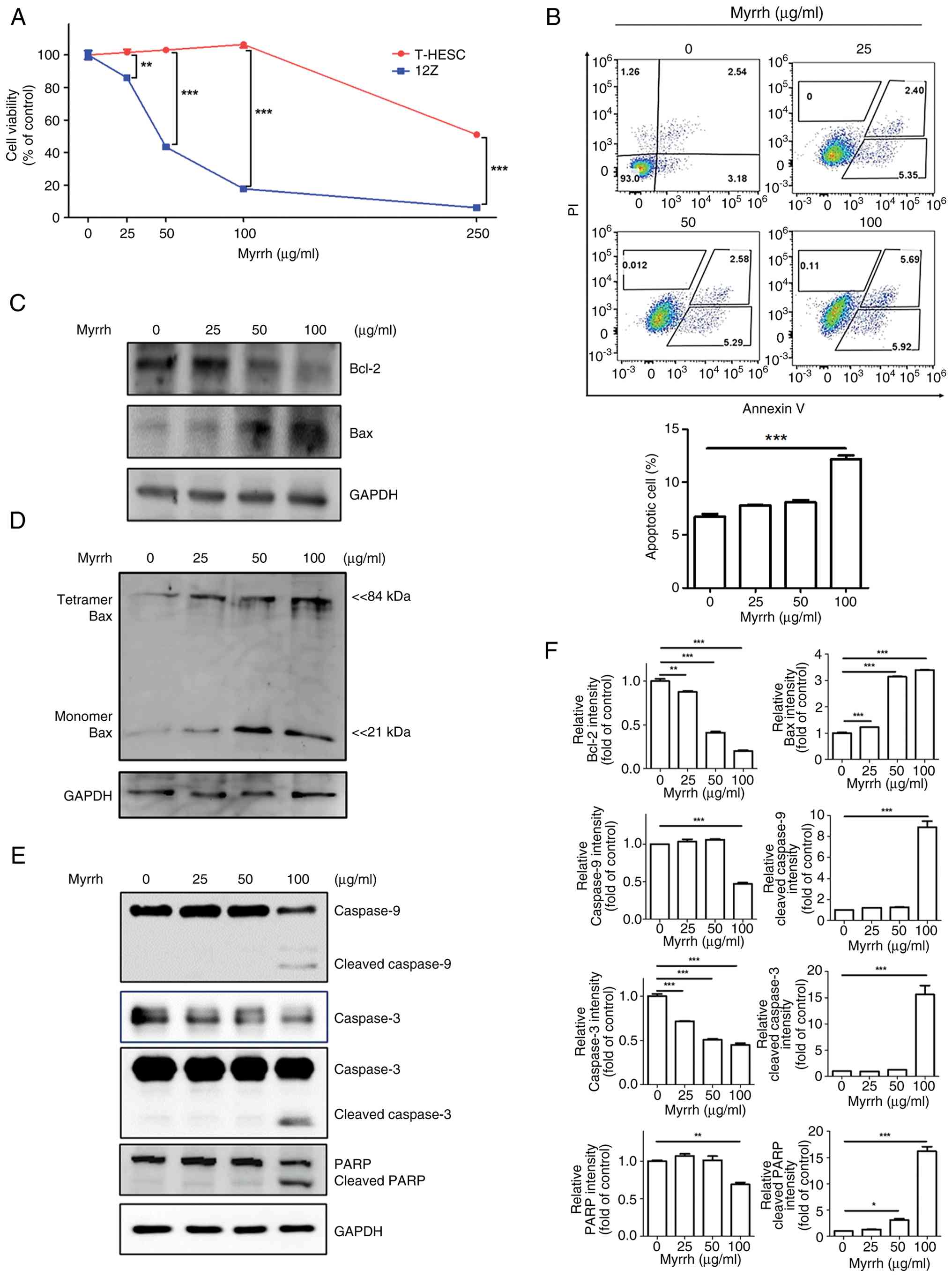
We investigated the inhibitory effects of myrrh on
the growth of normal endometrial T-HESCs and ectopic endometriotic
12Z cells. Both cell lines exhibited a dose-dependent reduction in
growth after myrrh treatment. However, cytotoxicity was observed at
lower concentrations in 12Z cells than in T-HESCs (Fig. 4A). The half-maximal inhibitory
concentrations of myrrh on the growth of T-HESCs and 12Z cells were
185.8 and 38.22 µg/ml, respectively, indicating that endometriotic
12Z cells were more sensitive to myrrh-induced cytotoxicity than
normal endometrial T-HESCs.
To determine the mechanism underlying the decreased
viability of myrrh-treated 12Z cells, we examined whether apoptosis
was induced. The results of the PI/Annexin V apoptosis assay
demonstrated that myrrh promoted apoptosis of 12Z cells in a
dose-dependent manner, similar to the results of the cell viability
assay. A slight increase in apoptotic cells (Annexin V-positive)
was observed at a concentration of 25 µg/ml, and a marked increase
in apoptotic cells was seen from 100 µg/ml (Fig. 4B). The levels of proteins involved
in intracellular apoptotic signaling in myrrh-treated 12Z cells
demonstrated that myrrh treatment decreased Bcl-2 protein
expression and increased Bax protein expression (Fig. 4C). This was accompanied by
increased Bax tetramer formation (Fig.
4D), indicating that myrrh promotes Bax activation and
mitochondrial apoptotic signaling. Myrrh also promoted the
conversion of caspase-3, -9 and PARP from their pro- to
active-cleavage forms (Fig. 4E).
The Immunoblot results are quantitatively supported by
densitometric analysis presented (Fig.
4F). These results suggested that myrrh activates apoptotic
signaling via the mitochondrial pathway.
Mechanism of action of myrrh revealed
by RNA sequencing
To investigate how myrrh regulates cell death at the
gene level, we conducted RNA sequencing. GSEA analysis utilizing
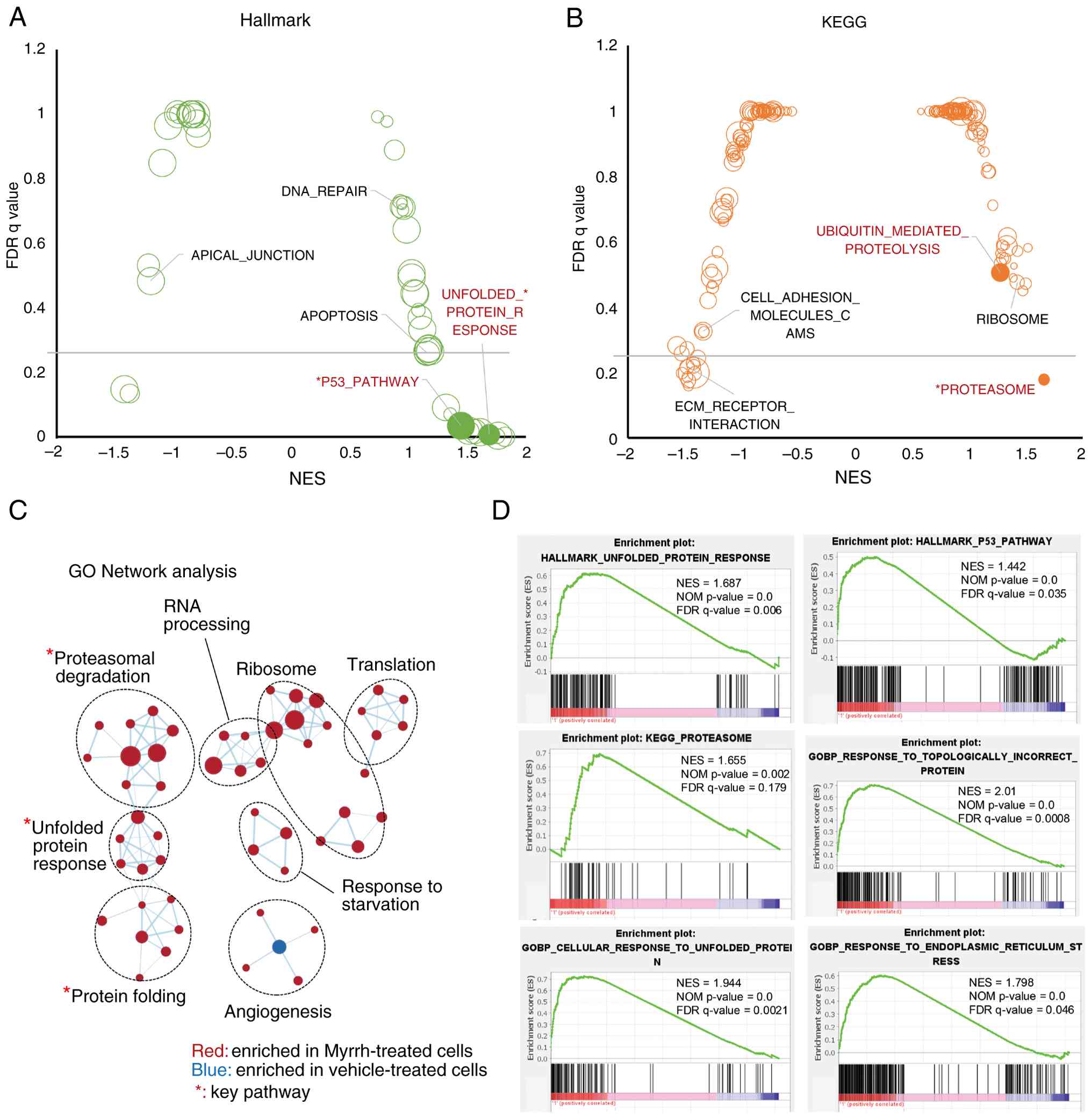
Hallmark (Fig. 5A), KEGG (Fig. 5B), and GO networks (Fig. 5C), revealed the upregulation of
pathways including p53, proteasome, ER stress, protein folding,
protein degradation, and apoptosis, whereas cell adhesion pathways,
such as ECM receptor interactions, were downregulated (Fig. 5D). Pathways such as ribosome and
translation and response to starvation were also upregulated in the
GO network analysis. DEG analysis revealed that 1499 genes were
significantly upregulated, whereas 1497 genes were significantly
downregulated (Fig. 6A). For the
156 (134 upregulated and 22 downregulated) genes included in the
pathway shown in Figure 5D, the
relative expression levels were summarized by heat map analysis
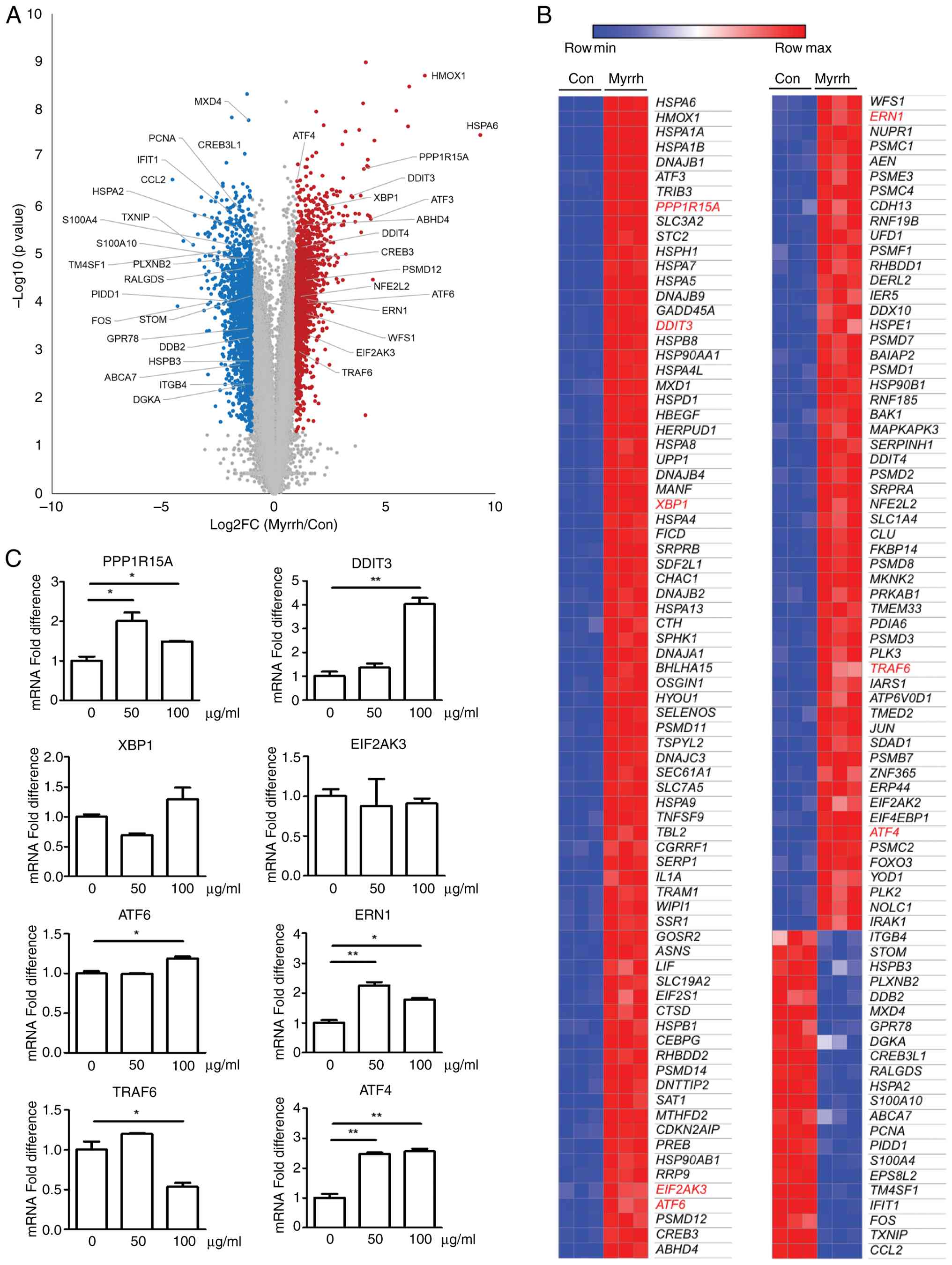
(Fig. 6B). Among them, the
expression of nine genes (PPP1R15A, DDIT3, XBP1, EIF2AK3, ATF6,
ERN1, TRAF6, and ATF4) known to be critical for the UPR
response was validated by qRT-PCR (Fig. 6C). The mRNA levels of PPP1R15A,
DDIT3, ERN1, ATF6, and ATF4 were increased by
myrrh treatment (50 or 100 µg/ml). In contrast, XBP1,
EIF2AK3, and TRAF6, either decreased or remained
unchanged. Moreover, while no significant change was observed in
TP53 gene expression, p53 protein levels decreased following myrrh
treatment, suggesting that this pathway was not significantly
involved in the mechanism of action of myrrh (Fig. S3). These results suggested the
possible involvement of the UPR in the myrrh-induced apoptosis.
Validation of myrrh-induced
cytotoxicity and ER stress
To confirm the involvement of ER stress in
myrrh-induced cytotoxicity, cell viability was determined after
simultaneous treatment with tauroursodeoxycholic acid (TUDCA), a
wide-range ER stress inhibitor. The results indicated that the
myrrh-induced decrease in 12Z cell viability was significantly
reversed by co-treatment with TUDCA (Fig. 7A). These results suggested that the
differential cytotoxicity of myrrh against endometriotic 12Z cells
may be, at least in part, attributed to the induction of ER stress
(Fig. 7B).
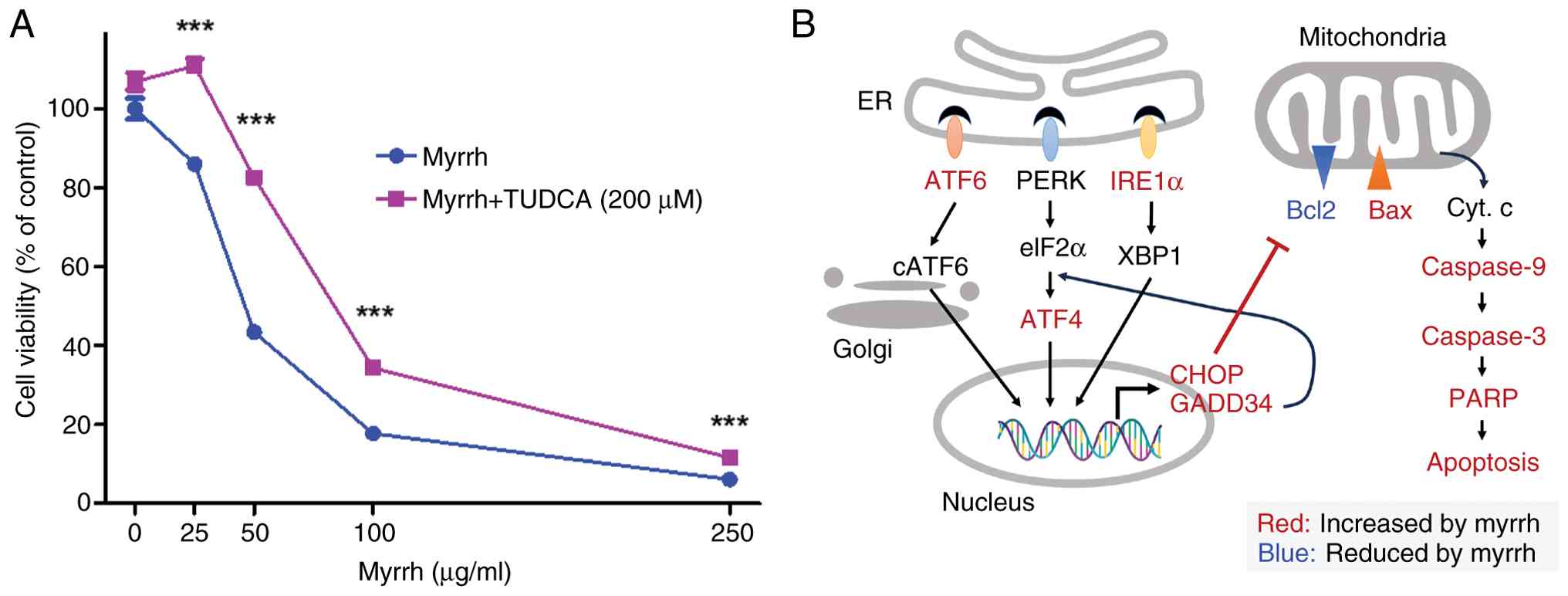 | Figure 7Mechanism of action of myrrh in
endometriosis. (A) 12Z were treated with the indicated
concentrations of myrrh and TUDCA (200 µM). Cell viability was
analyzed using MTT after 24 h, measured at a wavelength of 450 nm.
(B) Schematic representation summarizing the experimental results
and the potential mechanism by which myrrh exerts palliative
effects on endometriosis. ***P<0.001. TUDCA,
tauroursodeoxycholic acid; ATF6, activating transcription factor 6;
IRE1α, inositol-requiring enzyme 1 alpha; ATF4, activating
transcription factor 4; CHOP, C/EBP homologous protein; GADD34,
growth arrest and DNA damage-inducible protein 34; Bax,
Bcl-2-associated X protein; Bcl2, B-cell lymphoma 2; Caspase 3,
cysteine-aspartic acid protease 3; Caspase 9, cysteine-aspartic
acid protease 9; PARP, poly(ADP-ribose) polymerase. |
Discussion
The most common symptoms of endometriosis are
dysmenorrhea and pelvic pain; endometriosis is the second leading
cause of chronic pelvic pain (1,6).
Traditional medicine has approached the discovery of treatments for
endometriosis within the category of dysmenorrhea or pelvic pain
(40). The combination of myrrh
and frankincense has been reported to alleviate chronic pelvic pain
by modulating inflammation and TRPV1 signaling pathway (41-43).
Our previous research revealed that frankincense can alleviate
endometriosis by promoting apoptosis and inhibiting intercellular
adhesion at the animal and cellular levels (28). In addition, previous studies on
Prunella vulgaris used dienogest as a positive control
(44). Combining the data from the
previous study and the current results, it can be found that 3.5
mg/kg of myrrh is similar to 1 mg/kg of dienogest in terms of the
effect on weight of endometriosis foci. Myrrh has been used in
traditional medicine for a variety of painful conditions,
particularly for chronic pelvic pain, such as the herbal formula
Shaofuzhuyu Decoction, which is a standard prescription used to
treat blood stasis syndrome in gynecological conditions (45). Clinical studies have also suggested
the potential benefits of nutritional supplements containing myrrh
for endometriosis (23,24).
Although these clinical applications of myrrh
suggest its potential as a remedy for endometriosis, direct
evidence had been lacking. Therefore, in this study, employing a
network pharmacology approach, we explored the potential of myrrh
to serve as an inhibitor of pain and inflammation and inducer of
apoptosis, and determined its association with uterine and female
reproductive system disorders. Notably, over half of the myrrh's
known target genes are associated with endometriosis, and these
genes exhibit closely relationships with each other in terms of
PPIs. The prediction that myrrh affects endometriosis was confirmed
at both the animal and cellular levels. In contrast to frankincense
(28), myrrh had a significant
effect only on the size of endometriosis by inducing apoptosis but
not on the adhesion between endometrial and peritoneal cells.
To elucidate the mechanism through which myrrh
inhibits endometriosis, we focused on ER stress-related apoptosis.
ER stress is a stress response that is caused by abnormal folding
of proteins and regulates protein homeostasis. It is initiated by
proteins in the ER membrane, such as IRE1, PERK, and ATF6, and
determines various cellular functions, particularly cell survival
and death (46). Under
physiological conditions, an adaptive UPR reduces intracellular
protein stress by promoting the transcription of chaperone
proteins, refolding misfolded proteins, and degrading misfolded
proteins through ER-associated degradation. However, sustained
elevation of UPR results in a pro-apoptotic UPR that promotes
apoptosis by activating ATF4-CHOP (47,48).
Consistent with the findings of previous studies, our findings
revealed that myrrh increased the expression of key pro-apoptotic
UPR genes, including DDIT4 (CHOP), PPP1R15A (GADD34), ATF6,
ATF4, and ERN1 (IRE1α). We also observed that myrrh
induced a Bcl-2/Bax imbalance, resulting in mitochondria-dependent
apoptosis and myrrh-induced apoptosis was abolished by the ER
stress inhibitor, TUDCA. To confirm the involvement of ER stress in
the cytotoxic effects of myrrh, it would be ideal to perform
knockdown experiments targeting UPR-related genes up-regulated by
myrrh. However, there are many genes increased by myrrh, which
means that knockdown of a single gene may have limited efficacy.
Additionally, simultaneous knockdown of multiple genes can cause
competitive effects or DNA toxicity (49,50),
making it experimentally impractical. Therefore, in this study, we
confirmed that cell survival rates recovered when TUDCA, a
broad-spectrum ER stress inhibitor, was co-administered with myrrh.
It should be noted that myrrh did not uniformly activate all UPR
branches. Our data indicated selective upregulation of
PERK-ATF4-CHOP and ATF6 signaling, while the IRE1-XBP1 branch was
minimally affected. Such branch-specific activation of the UPR is
consistent with the concept that distinct stressors preferentially
engage different UPR arms. Furthermore, although GSEA identified
enrichment of the p53 pathway, this enrichment is thought to
reflect the transcriptional program of p53-regulated genes rather
than TP53 expression itself. This is because neither p53 mRNA
levels observed in RNA sequencing nor p53 protein expression
confirmed by Immunoblot after myrrh-treatment were actually
decreased. This apparent discrepancy suggests that p53 is unlikely
to serve as a key player in myrrh-induced cell death, instead
emphasizing that ER stress-mediated cell death is the primary
mechanism.
Despite the promising results, several limitations
of this study should be acknowledged. First, the murine
endometriosis model and immortalized endometrial cell lines used
may not fully capture the complexity and heterogeneity of human
endometriosis, which could affect the generalizability of the
findings. Second, although RNA sequencing and qRT-PCR analyses
identified key UPR genes upregulated by myrrh, some genes such as
XBP1 and EIF2AK3 did not show consistent changes, and p53 protein
levels decreased despite pathway enrichment, indicating that
additional studies-including targeted knockdown experiments-are
needed to clarify the precise molecular mechanisms. Third, while
the involvement of ER stress was supported using the broad-spectrum
inhibitor TUDCA, single-gene interventions were not performed due
to experimental limitations. Finally, although no significant
toxicity was observed in mice at the effective concentrations used,
further preclinical studies are required to evaluate the safety,
pharmacokinetics, and efficacy of myrrh in humans. These
limitations should be considered when interpreting the
translational potential of our findings.
The role of UPR and ER stress in endometriosis has
been well reviewed by Al-Hetty et al (16). Zhou et al suggested a
possible mechanism by which increased UPR in the hypoxic
environment of the peritoneal cavity may be involved in the
pathogenesis of endometriosis (51). In cancer, the induction of
apoptosis by targeting ER stress has been investigated as a
therapeutic approach (52,53). However, in the context of
endometriosis, only a few studies have identified ER stress as a
potential therapeutic target. Dienogest and tunicamycin have been
shown to suppress endometriosis by increasing ER stress (54,55).
Several plant flavonoids, such as apigenin, chrysin, quercetin, and
kaempferol, may inhibit endometriosis by promoting ER stress
(56-58).
We previously suggested that frankincense, commonly combined with
myrrh for pain relief, reduces the size and number of endometriotic
lesions by activating ER stress (28). The above studies and the results of
this paper confirm the potential of the UPR as a good therapeutic
target to inhibit endometriosis. furthermore previous studies have
shown that myrrh can enhance the activity of cisplatin in human
cervical cancer cells, inducing cell death and autophagy (59). This suggests that myrrh may
potentiate the therapeutic effects of other treatments. Future
research could explore the synergistic effects of myrrh with
treatments targeting ER stress or inflammatory pathways. Such
studies could significantly expand the therapeutic potential of
myrrh and enhance its effectiveness in treating endometriosis and
other related diseases.
Myrrh is a resin extracted from C. myrrha
that contains a wide variety of natural constituents, including
terpenoids and steroids (18,19).
The volatile oils in myrrh, containing monoterpenoids and
sesquiterpenoids, are often volatilized during hydrothermal
extraction (19), resulting in
their detection in very small amounts after extraction. Therefore,
hydrothermally extracted myrrh may contain high-weight molecules,
including diterpenoids, triterpenoids, and steroid-like compounds,
as the major constituents. Previous studies have suggested that
several steroids, steroid-like flavonoids, and triterpenoids can
promote UPR (56-58,60,61).
In contrast to Myrrh, which induces ER stress and promotes
UPR-mediated apoptosis in endometriosis, Z-Guggulsterone primarily
exerts its biological effects through the inhibition of the NF-κB
pathway (62). NF-κB is known to
play a role in modulating ER stress (63), and its inhibition by
Z-Guggulsterone could potentially mitigate ER stress responses.
Similarly, oleanolic acid and oleanonic acid have been reported to
inhibit NF-κB or reduce ER stress (64-66).
This difference in mechanisms suggests that while Myrrh exacerbates
ER stress in endometriosis, several components of myrrh may offer a
protective role against ER stress through its regulation of NF-κB
activity. However, there is currently a lack of studies directly
investigating the relationship between myrrh compounds and ER
stress. Therefore, further research is needed to determine how the
major constituents of myrrh influence ER stress responses. Such
investigations could provide valuable insights into the therapeutic
potential of myrrh in modulating ER stress-related conditions.
Myrrh has a long history of use in flavors,
perfumes, nutraceuticals and as a medicine, and its safety is well
documented. In general, no unusual toxic effects have been reported
in rodents at doses of 500 mg/kg/day for 12 weeks, and no
genotoxicity or carcinogenicity studies have been conducted
(19). Several studies have
reported myrrh essential oils to be irritating to the skin,
respiratory and digestive systems and toxic to goats at
concentrations of 1-5 g/kg/day (18). However, this study used
hydrothermally extracted myrrh to volatilize the essential oils and
used concentrations significantly lower than those toxic to goats.
Additionally, histological examination of the liver and kidneys at
therapeutic concentrations in mice showed no toxic effects.
Therefore, we suggest that myrrh is a relatively safe drug for the
treatment of endometriosis. However, further safety studies,
including a Good Laboratory Practice-level toxicity experiment, may
be required before myrrh can be used in human clinical trials.
In conclusion, here we provide the first
experimental evidence of the potential effects of myrrh on
endometriosis. Results from in vivo and in vitro
models showed that myrrh suppressed the growth of endometriotic
cells, thereby reducing the size of endometriotic foci. RNA
sequencing and bioinformatics analyses suggested ER
stress-associated apoptosis as the mechanism of myrrh's
anti-endometriosis action. Taken together, these results suggest
that myrrh aqueous extract may be an effective and safe option for
the treatment of endometriosis.
Supplementary Material
In silico prediction of
endometriosis-related pathways that may represent preliminary
targets of myrrh. (A) Potential target genes of myrrh were obtained
from the Traditional Chinese Medicine Integrative Database and
analyzed with the JEPPETO plugin in Cytoscape. KEGG, (B) Biocarta,
and (C) GO_MF enrichment analyses indicate that these genes are
enriched in pathways related to apoptosis, mitochondrial
dysfunction, proteolytic regulation and stress responses, which are
dysregulated in endometriosis and may represent putative targets
for myrrh.
Histological analysis of kidney and
liver tissues after myrrh treatment in endometriosis-induced mice.
H&E staining results of (A) kidney and (B) liver tissue. The
mice were treated indicated dose of myrrh (0.7 or 3.5 mg/kg/day)
after induction of endometriosis. The microphotographic images of
H&E-stained kidney and liver sections were presented
(magnification, x100). Con, control.
Effects of myrrh on TP53 expression
and p53 protein levels (A) TP53 expression levels were visualized
using RNA sequencing data. (B) Analysis of alterations in p53
protein by myrrh. Immunoblot analysis revealed that p53 was
detected after 24 h of treatment with each concentration of myrrh.
GAPDH was used as a internal control. CON, control.
Possible targets of myrrh were
collected from TCMID and HIT database.
KEGG pathway analysis was conducted by
Cytoscape with JEPPETO plugin using possible target genes of
myrrh.
Biocarta pathway analysis was
conducted by Cytoscape with JEPPETO plugin using possible target
genes of myrrh.
GO biological pathway analysis was
conducted by Cytoscape with JEPPETO plugin using possible target
genes of myrrh.
GO molecular function analysis was
conducted by Cytoscape with JEPPETO plugin using possible target
genes of myrrh.
GO cellular components analysis was
conducted by Cytoscape with JEPPETO plugin using possible target
genes of myrrh.
Genes related with endometriosis in
DisGeNet database.
Acknowledgements
Not applicable.
Funding
Funding: This study was supported by a Biomedical Research
Institute Grant (grant no. 20230192) from Pusan National University
Hospital and a grant from Kosin University College of Medicine
(2023).
Availability of data and materials
The RNA sequencing dataset generated in this study
has been deposited in the NCBI Gene Expression Omnibus (GEO) under
accession number GSE246172 (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE246172).
The raw and processed LC-MS datasets are publicly available in the
Harvard Dataverse repository at https://dataverse.harvard.edu/dataset.xhtml?persistentId=doi:10.7910/DVN/UUSTOQ.
All other data supporting the findings of this study are available
from the corresponding authors upon reasonable request.
Authors' contribution
BSK and MC conducted and performed most of the
experiments. JHH, JSJ and SJB performed and analyzed animal
experiments and tissue staining. YuJ, DR and JJ performed
bioinformatic analysis. SBK and YeJ conducted and analyzed the
LC-MS data. BSK and IK wrote the draft of the paper. KTH reviewed
and revised the paper. IK and KTH conceived and supervised
throughout the study. BSK and KTH confirm the authenticity of all
the raw data. All authors have read and approved the final
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Parasar P, Ozcan P and Terry KL:
Endometriosis: Epidemiology, diagnosis and clinical management.
Curr Obstet Gynecol Rep. 6:34–41. 2017.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Horne AW and Missmer SA: Pathophysiology,
diagnosis, and management of endometriosis. BMJ.
379(e070750)2022.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Koninckx PR, Ussia A, Adamyan L, Wattiez
A, Gomel V and Martin DC: Pathogenesis of endometriosis: The
genetic/epigenetic theory. Fertil Steril. 111:327–340.
2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Anglesio MS, Papadopoulos N, Ayhan A,
Nazeran TM, Noë M, Horlings HM, Lum A, Jones S, Senz J, Seckin T,
et al: Cancer-associated mutations in endometriosis without cancer.
New Engl J Med. 376:1835–1848. 2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Leap K, Yotova I, Horvath S and
Martinez-Agosto JA: Epigenetic age provides insight into tissue
origin in endometriosis. Sci Rep. 12(21281)2022.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Saunders PTK and Horne AW: Endometriosis:
Etiology, pathobiology, and therapeutic prospects. Cell.
184:2807–2824. 2021.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Labbadia J and Morimoto RI: The biology of
proteostasis in aging and disease. Annu Rev Biochem. 84:435–464.
2015.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Sala AJ, Bott LC and Morimoto RI: Shaping
proteostasis at the cellular, tissue, and organismal level. J Cell
Biol. 216:1231–1241. 2017.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Bhattarai KR, Riaz TA, Kim HR and Chae HJ:
The aftermath of the interplay between the endoplasmic reticulum
stress response and redox signaling. Exp Mol Med. 53:151–167.
2021.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Hetz C, Chevet E and Oakes SA:
Proteostasis control by the unfolded protein response. Nat Cell
Biol. 17:829–838. 2015.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Yeo SG, Lee SJ, Lee JW, Oh S and Park DC:
Levels of endoplasmic reticulum stress-related mRNA in peritoneal
fluid of patients with endometriosis or gynaecological cancer. J
Int Med Res. 49(3000605211065376)2021.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Koga H, Kaushik S and Cuervo AM: Protein
homeostasis and aging: The importance of exquisite quality control.
Ageing Res Rev. 10:205–215. 2011.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Sethuram R, Bukowski M, Hernandez F, You
Y, Puscheck E, Mor G, Jeyasuria P and Condon JC: Endoplasmic
reticulum stress response and the regulation of endometrial
interferon-beta production. F S Sci. 4:151–162. 2023.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Choi J, Jo M, Lee E, Lee DY and Choi D:
Involvement of endoplasmic reticulum stress in regulation of
endometrial stromal cell invasiveness: Possible role in
pathogenesis of endometriosis. Mol Hum Reprod. 25:101–110.
2019.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Choi JY, Jo MW, Lee EY, Lee DY and Choi
DS: Ovarian steroid dependence of endoplasmic reticulum stress
involvement in endometrial cell apoptosis during the human
endometrial cycle. Reproduction. 155:493–503. 2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Al-Hetty H, Jabbar AD, Eremin VF, Jabbar
AM, Jalil AT, Al-Dulimi AG, Gharban HAJ, Khan MUF and Saleh MM: The
role of endoplasmic reticulum stress in endometriosis. Cell Stress
Chaperones. 28:145–150. 2023.PubMed/NCBI View Article : Google Scholar
|
|
17
|
The Plant List. 2010. Available at:
http://www.theplantlist.org/tpl/record/kew-2733595.
|
|
18
|
Shen T, Li GH, Wang XN and Lou HX: The
genus Commiphora: A review of its traditional uses, phytochemistry
and pharmacology. J Ethnopharmacol. 142:319–330. 2012.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Batiha GE, Wasef L, Teibo JO, Shaheen HM,
Zakariya AM, Akinfe OA, Teibo TKA, Al-Kuraishy HM, Al-Garbee AI,
Alexiou A and Papadakis M: Commiphora myrrh: A phytochemical and
pharmacological update. Naunyn Schmiedebergs Arch Pharmacol.
396:405–420. 2023.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Suliman RS, Alghamdi SS, Ali R, Aljatli D,
Aljammaz NA, Huwaizi S, Suliman R, Kahtani KM, Albadrani GM,
Barhoumi T, et al: The role of myrrh metabolites in cancer,
inflammation, and wound healing: Prospects for a multi-targeted
drug therapy. Pharmaceuticals (Basel). 15(944)2022.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Sun MX, Hua J, Liu GS, Huang PY, Liu NS
and He XP: Myrrh induces the apoptosis and inhibits the
proliferation and migration of gastric cancer cells through
down-regulating cyclooxygenase-2 expression. Bioscience Rep.
40(BSR20192372)2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Khalil N, Fikry S and Salama O:
Bactericidal activity of Myrrh extracts and two dosage forms
against standard bacterial strains and multidrug-resistant clinical
isolates with GC/MS profiling. Amb Express. 10(21)2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
De Leo V, Cagnacci A, Cappelli V, Biasioli
A, Leonardi D and Seracchioli R: Role of a natural integrator based
on lipoic acid, palmitoiletanolamide and myrrh in the treatment of
chronic pelvic pain and endometriosis. Minerva Ginecol. 71:191–195.
2019.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Alfonso FN: Experience with nutraceutical
supplements in the treatment of pelvic pain in gynaecology: Case
reports. Drugs Context. 11(2021-10-8)2022.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Xue RC, Fang Z, Zhang MX, Yi ZH, Wen CP
and Shi TL: TCMID: Traditional Chinese medicine integrative
database for herb molecular mechanism analysis. Nucleic Acids Res.
41:D1089–D1095. 2013.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Yan DY, Zheng GH, Wang CC, Chen Z, Mao T,
Gao J, Yan Y, Chen X, Ji X, Yu J, et al: HIT 2.0: An enhanced
platform for herbal ingredients' targets. Nucleic Acids Res.
50:D1238–D1243. 2022.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Winterhalter C, Widera P and Krasnogor N:
JEPETTO: A Cytoscape plugin for gene set enrichment and topological
analysis based on interaction networks. Bioinformatics.
30:1029–1030. 2014.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Cho MK, Jin JS, Jo Y, Han JH, Shin S, Bae
SJ, Ryu D, Joo J, Park JK and Ha KT: Frankincense ameliorates
endometriosis via inducing apoptosis and reducing adhesion. Integr
Med Res. 12(100947)2023.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Pinero J, Ramirez-Anguita JM,
Sauch-Pitarch J, Ronzano F, Centeno E, Sanz F and Furlong L: The
DisGeNET knowledge platform for disease genomics: 2019 update.
Nucleic Acids Res. 48:D845–D855. 2020.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Szklarczyk D, Morris JH, Cook H, Kuhn M,
Wyder S, Simonovic M, Santos A, Doncheva NT, Roth A, Bork P, et al:
The STRING database in 2017: Quality-controlled protein-protein
association networks, made broadly accessible. Nucleic Acids Res.
45:D362–D368. 2017.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Somigliana E, Vigano P, Rossi G, Carinelli
S, Vignali M and Panina-Bordignon P: Endometrial ability to implant
in ectopic sites can be prevented by interleukin-12 in a murine
model of endometriosis. Human Reprod. 14:2944–2950. 1999.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Saadh MJ, Haddad M, Dababneh MF, Bayan MF
and Al-Jaidi BA: A guide for estimating the maximum safe starting
dose and conversion it between animals and humans. Syst Rev Pharm.
11:98–101. 2020.
|
|
33
|
Kim BY, Lim HS, Kim YJ, Sohn E, Kim YH,
Koo I and Jeong SJ: Similarity of therapeutic networks induced by a
multi-component herbal remedy, Ukgansan, in neurovascular unit
cells. Sci Rep. 10(2658)2020.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Kim EY, Jin BR, Chung TW, Bae SJ, Park H,
Ryu D, Jin L, An HJ and Ha KT: 6-sialyllactose ameliorates
dihydrotestosterone-induced benign prostatic hyperplasia through
suppressing VEGF-mediated angiogenesis. BMB Rep. 52:560–565.
2019.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Kim J, Lee H, Jin EJ, Kang BE, Ryu D and
Kim G: A Microfluidic device to fabricate one-step cell bead-laden
hydrogel struts for tissue engineering. Small.
18(e2106487)2022.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Li B, Wang S, Duan H, Wang Y and Guo Z:
Discovery of gene module acting on ubiquitin-mediated proteolysis
pathway by co-expression network analysis for endometriosis. Reprod
Biomed Online. 42:429–441. 2021.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Beliard A, Noel A and Foidart JM:
Reduction of apoptosis and proliferation in endometriosis. Fertil
Steril. 82:80–85. 2004.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Kobayashi H, Matsubara S, Yoshimoto C,
Shigetomi H and Imanaka S: The role of mitochondrial dynamics in
the pathophysiology of endometriosis. J Obstet Gynaecol Res.
49:2783–2791. 2023.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Assimopoulou A, Zlatanos S and
Papageorgiou V: Antioxidant activity of natural resins and
bioactive triterpenes in oil substrates. Food Chem. 92:721–727.
2005.
|
|
40
|
Guo Y, Liu FY, Shen Y, Xu JY, Xie LZ, Li
SY, Ding DN, Zhang DQ and Han FJ: Complementary and alternative
medicine for dysmenorrhea caused by endometriosis: A review of
utilization and mechanism. Evid Based Complement Alternat Med.
2021(6663602)2021.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Su S, Hua Y, Wang Y, Gu W, Zhou W, Duan
JA, Jiang H, Chen T and Tang Y: Evaluation of the anti-inflammatory
and analgesic properties of individual and combined extracts from
Commiphora myrrha, and Boswellia carterii. J Ethnopharmacol.
139:649–656. 2012.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Liao Y, Guo C, Wen A, Bai M, Ran Z, Hu J,
Wang J, Yang J and Ding Y: Frankincense-Myrrh treatment alleviates
neuropathic pain via the inhibition of neuroglia activation
mediated by the TLR4/MyD88 pathway and TRPV1 signaling.
Phytomedicine. 108(154540)2023.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Hu D, Wang C, Li F, Su S, Yang N, Yang Y,
Zhu C, Shi H, Yu L, Geng X, et al: A combined water extract of
frankincense and myrrh alleviates neuropathic pain in mice via
modulation of TRPV1. Neural Plast. 2017(3710821)2017.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Cho MK, Jin L, Han JH, Jin JS, Cheon SY,
Shin S, Bae SJ, Park JK and Ha KT: Water-extracted prunella
vulgaris alleviates endometriosis by reducing aerobic glycolysis.
Front Pharmacol. 13(872810)2022.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Huang XC, Su SL, Duan JA, Sha X, Zhu KY,
Guo J, Yu L, Liu P, Shang E and Qian D: Effects and mechanisms of
shaofu-zhuyu decoction and its major bioactive component for
cold-stagnation and blood-stasis primary dysmenorrhea rats. J
Ethnopharmacol. 186:234–243. 2016.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Hetz C and Papa FR: The unfolded protein
response and cell fate control. Mol Cell. 69:169–181.
2018.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Wiseman RL, Mesgarzadeh JS and Hendershot
LM: Reshaping endoplasmic reticulum quality control through the
unfolded protein response. Mol Cell. 82:1477–1491. 2022.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Hetz C, Zhang K and Kaufman RJ:
Mechanisms, regulation and functions of the unfolded protein
response. Nat Rev Mol Cell Biol. 21:421–438. 2020.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Caffrey DR, Zhao J, Song Z, Schaffer ME,
Haney SA, Subramanian RR, Seymour AB and Hughes JD: siRNA
off-target effects can be reduced at concentrations that match
their individual potency. PLoS One. 6(e21503)2011.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Koller E, Propp S, Murray H, Lima W, Bhat
B, Prakash TP, Allerson CR, Swayze EE, Marcusson EG and Dean NM:
Competition for RISC binding predicts in vitro potency of siRNA.
Nucleic Acids Res. 34:4467–4476. 2006.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Zhou Y, Jin Y, Wang Y and Wu R: Hypoxia
activates the unfolded protein response signaling network: An
adaptive mechanism for endometriosis. Front Endocrinol (Lausanne).
13(945578)2022.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Bonsignore G, Martinotti S and Ranzato E:
Endoplasmic reticulum stress and cancer: Could unfolded protein
response be a druggable target for cancer therapy? Int J Mol Sci.
24(1566)2023.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Avril T, Vauleon E and Chevet E:
Endoplasmic reticulum stress signaling and chemotherapy resistance
in solid cancers. Oncogenesis. 6(e373)2017.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Choi J, Jo M, Lee E, Lee DY and Choi D:
Dienogest regulates apoptosis, proliferation, and invasiveness of
endometriotic cyst stromal cells via endoplasmic reticulum stress
induction. Mol Hum Reprod. 26:30–39. 2020.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Hasegawa A, Osuga Y, Hirota Y, Hamasaki K,
Kodama A, Harada M, Tajima T, Takemura Y, Hirata T, Yoshino O, et
al: Tunicamycin enhances the apoptosis induced by tumor necrosis
factor-related apoptosis-inducing ligand in endometriotic stromal
cells. Hum Reprod. 24:408–414. 2009.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Zhang L, Mohankumar K, Martin G, Mariyam
F, Park Y, Han SJ and Safe S: Flavonoids quercetin and kaempferol
are NR4A1 antagonists and suppress endometriosis in female mice.
Endocrinology. 164(bqad133)2023.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Ryu S, Bazer FW, Lim W and Song G: Chrysin
leads to cell death in endometriosis by regulation of endoplasmic
reticulum stress and cytosolic calcium level. J Cell Physiol.
234:2480–2490. 2019.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Park S, Lim W, Bazer FW and Song G:
Apigenin induces ROS-dependent apoptosis and ER stress in human
endometriosis cells. J Cell Physiol. 233:3055–3065. 2018.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Ramadan WS, Sait KH and Anfinan NM:
Anticancer activity of aqueous myrrh extract alone and in
combination with cisplatin in HeLa cells. Trop J Pharm Res.
16:889–896. 2017.
|
|
60
|
Das I, Png CW, Oancea I, Hasnain SZ,
Lourie R, Proctor M, Eri RD, Sheng Y, Crane DI, Florin TH and
McGuckin MA: Glucocorticoids alleviate intestinal ER stress by
enhancing protein folding and degradation of misfolded proteins. J
Exp Med. 210:1201–1216. 2013.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Beukes N, Levendal RA and Frost CL:
Selected terpenoids from medicinal plants modulate endoplasmic
reticulum stress in metabolic disorders. J Pharm Pharmacol.
66:1505–1525. 2014.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Huang C, Wang J, Lu X, Hu W, Wu F, Jiang
B, Ling Y, Yang R and Zhang W: Z-guggulsterone negatively controls
microglia-mediated neuroinflammation via blocking
IkappaB-alpha-NF-κB signals. Neurosci Lett. 619:34–42.
2016.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Zhu X, Huang L, Gong J, Shi C, Wang Z, Ye
B, Xuan A, He X, Long D, Zhu X, et al: NF-κB pathway link with ER
stress-induced autophagy and apoptosis in cervical tumor cells.
Cell Death Discov. 3(17059)2017.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Hwang YJ, Song J, Kim HR and Hwang KA:
Oleanolic acid regulates NF-κB signaling by suppressing MafK
expression in RAW 264.7 cells. BMB Rep. 47:524–529. 2014.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Lee ES, Kim HM, Kang JS, Lee EY, Yadav D,
Kwon MH, Kim YM, Kim HS and Chung CH: Oleanolic acid and
N-acetylcysteine ameliorate diabetic nephropathy through reduction
of oxidative stress and endoplasmic reticulum stress in a type 2
diabetic rat model. Nephrol Dial Transplant. 31:391–400.
2016.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Gao H, Liu H, Tang T, Huang X, Wang D, Li
Y, Huang P and Peng Y: Oleanonic acid ameliorates pressure
overload-induced cardiac hypertrophy in rats: The role of
PKCζ-NF-κB pathway. Mol Cell Endocrinol. 470:259–268.
2018.PubMed/NCBI View Article : Google Scholar
|