Introduction
Invasive ductal carcinoma (IDC) is the most common
histological subtype of breast cancer, accounting for 70-75% of all
cases. Mortality in breast cancer is predominantly attributable to
distant metastases, with the most frequent sites including the
bones, lungs, liver and brain (1).
By contrast, peritoneal metastasis is a relatively rare occurrence
in the natural history of breast cancer, with contemporary data
continuing to show a higher propensity for invasive lobular
carcinoma (ILC) compared with IDC, attributable to its distinct
pattern of dissemination (2,3).
This disparity is marked, as one cohort study of metastatic breast
cancer reported peritoneal involvement in 68.8% of ILC cases
compared with only 1% of IDC cases (4). Diagnosis is often delayed due to its
insidious onset and non-specific clinical manifestations, which can
mimic a variety of benign abdominal disorders (such as functional
dyspepsia, peptic ulcer disease, gastroenteritis or pancreatitis)
and malignant abdominal conditions (such as primary ovarian,
gastric or colorectal cancer, lymphoma or primary peritoneal
carcinoma).
The introduction of CDK4/6 inhibitors has markedly
improved outcomes for patients with hormone receptor (HR)-positive
and HER2-negative metastatic breast cancer (5,6).
However, gastrointestinal adverse effects, such as nausea, vomiting
and diarrhea, commonly associated with this drug class may closely
mimic symptoms of peritoneal carcinomatosis or other forms of
disease progression, such as worsening peritoneal carcinomatosis
(malignant ascites), bowel obstruction/ileus from peritoneal
deposits or progression at other metastatic sites. This symptomatic
overlap introduces notable diagnostic ambiguity, potentially
delaying the recognition of metastatic spread.
The present study outlines an instructive case of a
patient with HR+/HER2- IDC who developed
extensive peritoneal metastases during treatment with the CDK4/6
inhibitor palbociclib, manifesting as symptomatic gastric wall
thickening and functional gastric outlet obstruction. The present
case highlights a key diagnostic challenge, underscores the
difficulty in distinguishing drug-related toxicity from true
disease progression and demonstrates how serosal-based metastases
can evade conventional diagnostic modalities such as endoscopy and
cross-sectional imaging. Furthermore, the multifactorial
pathophysiology, including neural and myogenic dysfunction, that
underlies the resulting delayed gastric emptying is explored. These
observations underscore the need for clinicians to maintain a high
index of suspicion in patients receiving similar therapies and to
pursue prompt additional diagnostic procedures to establish a
definitive diagnosis.
Case report
Patient presentation
Primary treatment, recurrence and the diagnostic
journey for peritoneal metastasis across the complex clinical
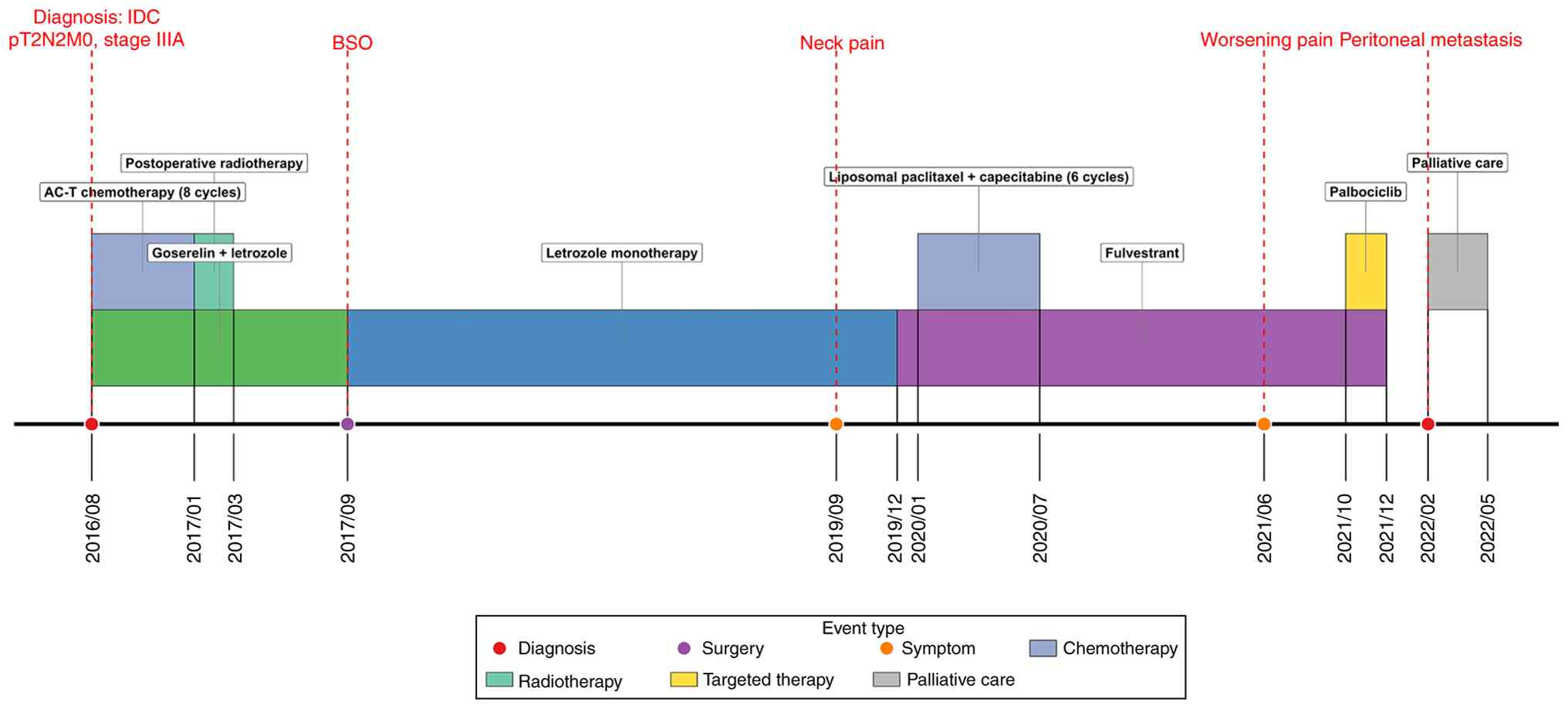
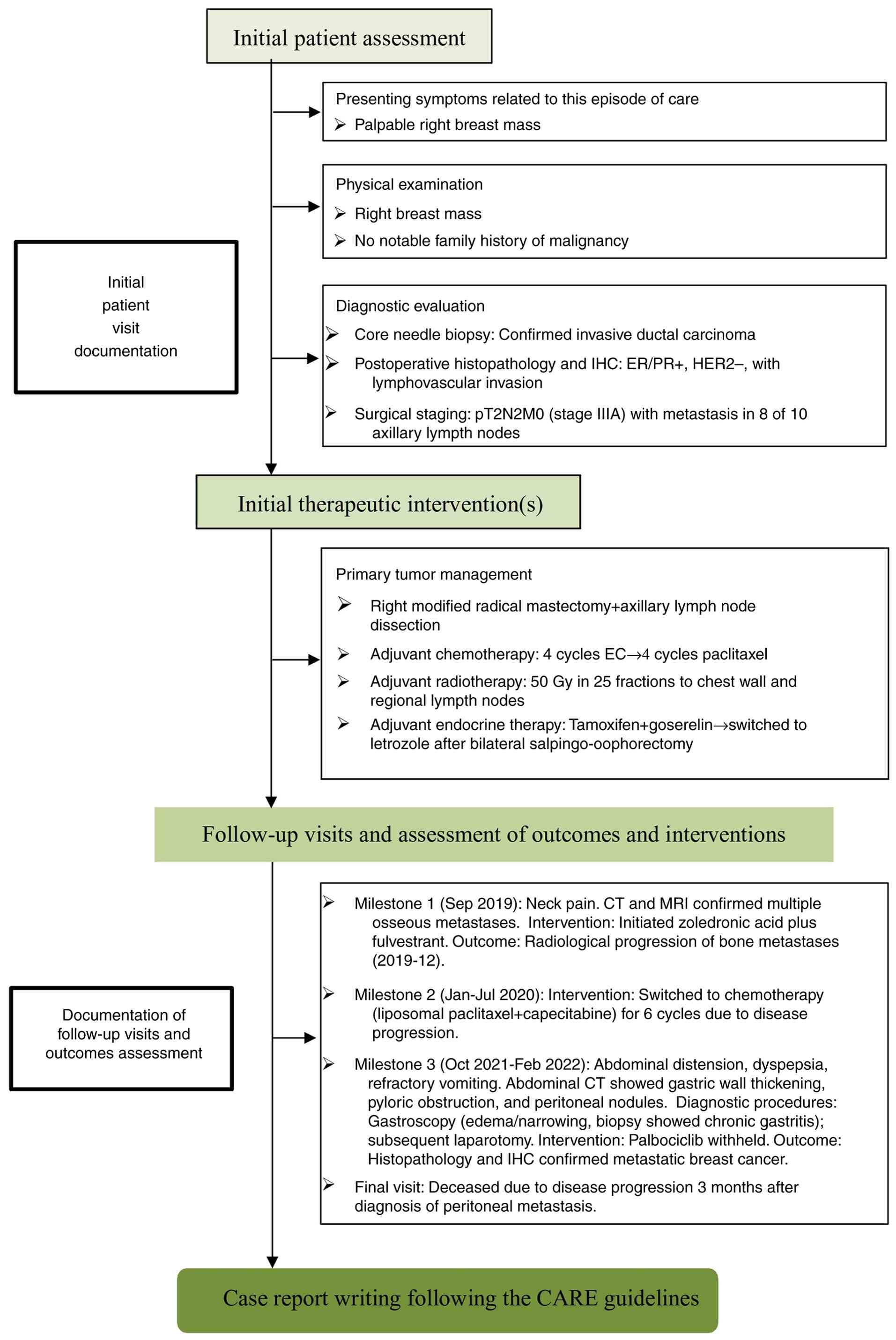
course, is summarized in the timeline provided in Fig. 1. In August 2016, a 48-year-old
woman with no notable family history of malignancy presented to
Ya'an People's Hospital (Sichuan, China) with a palpable right
breast mass. A core needle biopsy confirmed IDC, clinically staged
as cT2N2M0 (stage IIIA) according to the American Joint Committee
on Cancer 7th edition TNM staging system (7). Subsequently, in August 2016, the
patient underwent a right modified radical mastectomy with axillary
lymph node dissection at The General Hospital of Western Theater
Command (Sichuan, China). Subsequent adjuvant chemotherapy,
radiotherapy and regular follow-ups were all conducted at this
institution. Histopathological examination confirmed IDC, which was
positive for estrogen receptor (ER) and progesterone receptor (PR),
negative for HER2 and exhibited lymphovascular invasion (Fig. S1A-D). The Ki-67 proliferative
index was ~20% (Fig. S1E).
Metastatic carcinoma was identified in 8 out of 10 resected
axillary lymph nodes, resulting in a final pathological stage of
pT2N2M0 (stage IIIA).
Postoperatively, the patient received adjuvant
chemotherapy starting in September 2016, consisting of four 21-day
cycles of epirubicin (100 mg/m2) and cyclophosphamide
(600 mg/m2), followed by four 21-day cycles of
paclitaxel (175 mg/m2). Upon completion of chemotherapy,
the patient received adjuvant radiotherapy to the right chest wall
and regional lymph nodes (axillary, supraclavicular and
infraclavicular regions) at a total dose of 50 Gy delivered in 25
fractions across 5 weeks, which was completed in March 2017.
Adjuvant endocrine therapy was initiated concurrently with
chemotherapy in September 2016, consisting of tamoxifen (20 mg
daily orally) and goserelin (3.6 mg subcutaneously every 28 days).
After undergoing bilateral salpingo-oophorectomy for ovarian
ablation in September 2017, the endocrine regimen of the patient
was switched to letrozole (2.5 mg daily orally) alone. The patient
was then followed up every 3 months in the Oncology Outpatient
Clinic for 2 years, with no evidence of recurrence.
In September 2019, ~3 years after surgery, the
patient developed neck pain. A chest CT scan with bone window
reconstruction was subsequently performed at The General Hospital
of Western Theatre Command in December 2019, which revealed
multiple punctate and patchy sclerotic densities involving the
bilateral humeral heads, scapulae, sternum, thoracic vertebral
bodies and portions of the ribs (Fig.
S2A and B). Concurrently, a
spinal MRI demonstrated scattered speckled and patchy abnormal
signals in multiple vertebrae (Fig.
S3A and B). These imaging
findings, in conjunction with the symptoms presented, were highly
suggestive of osseous metastases. Treatment was initiated at this
institution in December 2019, with monthly intravenous zoledronic
acid and intramuscular fulvestrant administered at a loading dose
of 500 mg on days 1, 15 and 29, followed by monthly maintenance
therapy (500 mg intramuscularly every 28 days). The imaging
findings were indicative of progressive metastatic disease at that
time. Accordingly, from January to July 2020, the patient received
six cycles of chemotherapy with liposomal paclitaxel (175
mg/m2) and capecitabine (1,000 mg/m2 twice
daily for 14 days, every 21 days).
In June 2021, the patient was readmitted due to
progressively worsening pain. A whole-body bone scan revealed
multiple foci of increased metabolic activity throughout the
skeleton, highly suggestive of metastatic disease (Fig. S2C). To obtain histological
confirmation, a CT-guided pelvic biopsy was performed on July 23,
2021. However, it revealed no malignant cells (Fig. S1F). Despite this negative biopsy
result, a subsequent spinal MRI in October 2021 demonstrated
unequivocal progression of the bony lesions, with an increase in
both the number and size of metastatic deposits compared with prior
imaging (Fig. S3C and D). Given the compelling radiological and
clinical evidence of progressive metastatic disease, treatment with
palbociclib (125 mg daily, 21 days on and 7 days off) and
fulvestrant (500 mg intramuscularly on days 1 and 15 of cycle 1,
followed by 500 mg every 28 days) was initiated in October
2021.
In December 2021, after completing two cycles of
palbociclib, the patient developed symptoms of abdominal
distension, dyspepsia and vomiting. The temporal association
initially suggested a potential adverse effect of palbociclib.
These symptoms began ~8 weeks after initiating the drug. The
symptoms of the patient persisted despite standard antiemetic
support. In an initial attempt to manage these symptoms, the dosing
schedule was modified to an alternate-day regimen for 1 week.
However, the gastrointestinal symptoms showed no improvement.
Furthermore, due to the worsening of their discomfort, the patient
declined to continue taking the medication. Laboratory tests showed
mild leukopenia (white blood cell count, 3.2x109/l;
reference range, 3.5-9.5x109/l) but no notable
neutropenia (absolute neutrophil count, 1.4x109/l;
reference range, 1.8-6.3x109/l). The leukopenia was
managed with subcutaneous recombinant human granulocyte
colony-stimulating factor (150 µg daily), which normalized the
white blood cell count. The drug was withheld after two cycles due
to the suspicion of drug-induced toxicity and the decision of the
patient. However, the symptoms exhibited by the patient persisted
despite standard antiemetic support and, notably, showed no
resolution for >4 weeks after palbociclib discontinuation. This
protracted course, despite drug withdrawal, raised the first strong
suspicion for an underlying malignant etiology, prompting further
investigation.
The patient was admitted to The General Hospital of
Western Theater Command in January 2022 due to persistent symptoms.
At admission, an Eastern Cooperative Oncology Group performance
status of 2 was reported (8). The
patient reported notable weight loss of ~6 kg across the preceding
2 months. Laboratory investigations revealed mild anemia (97 g/l;
reference range, 115-150 g/l), hypokalemia (3.29 mmol/l; reference
range, 3.5-5.3 mmol/l) and hypoalbuminemia (28 g/l; reference
range, 35-52 g/l). Tumor markers were notably elevated, with
carcinoembryonic antigen (CEA) at 219.74 ng/ml (reference value,
<5 ng/ml), carbohydrate antigen (CA)125 at 124.90 U/ml
(reference value, <35 U/ml) and CA15-3 at 146.30 U/ml (reference
value, <25 U/ml). A longitudinal assessment of tumor markers
(Fig. S4) demonstrated dynamic
changes in serum CEA, CA125 and CA15-3 levels from the diagnosis of
bone metastasis in December 2019 to the presentation of peritoneal
disease in January 2022, which was associated with disease
progression. A physical examination revealed notable abdominal
distension, with no palpable mass detected in the left breast.
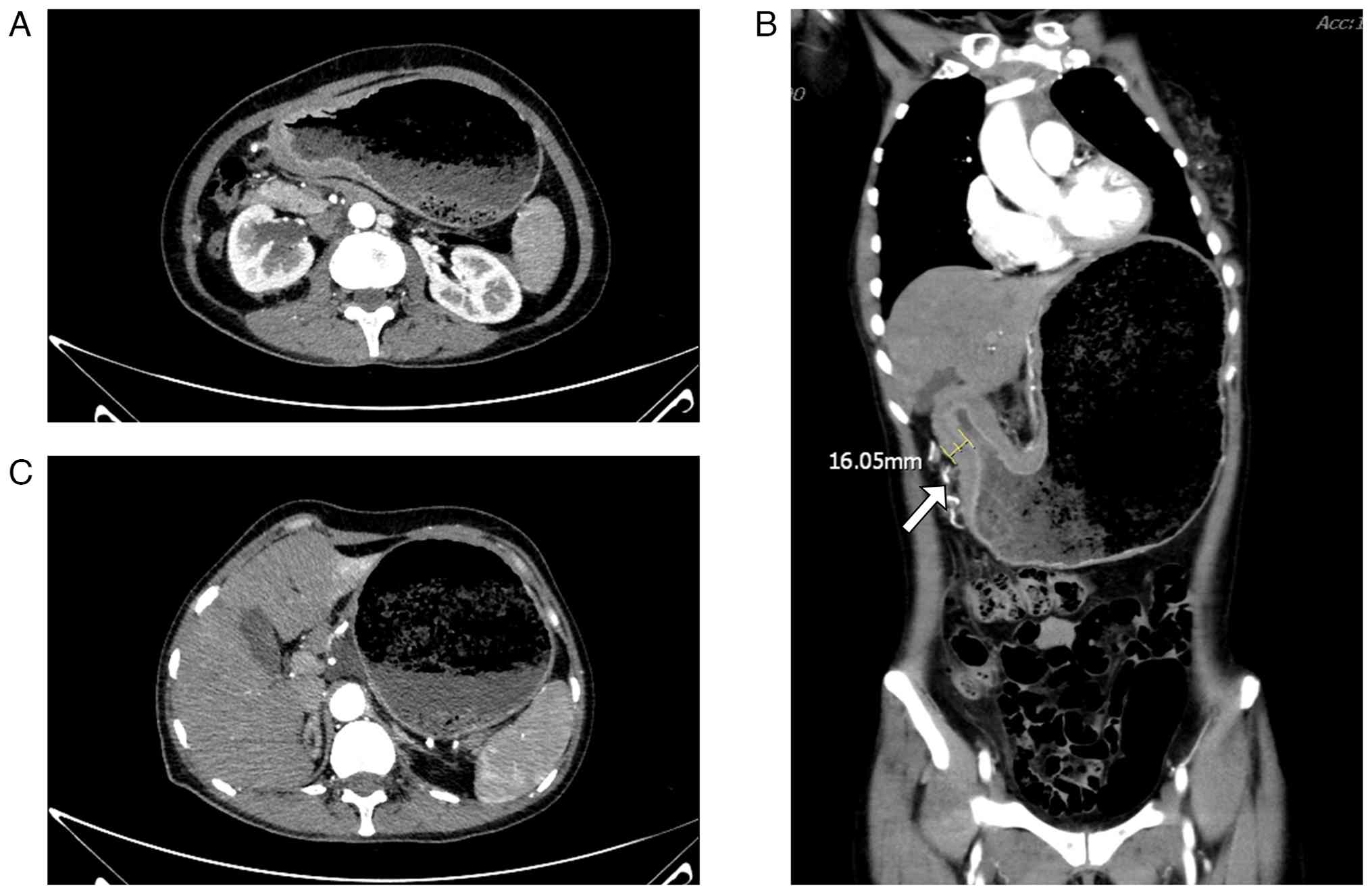
Contrast-enhanced abdominal CT performed in January 2022
demonstrated marked circumferential wall thickening (maximum
thickness 16.05 mm; Fig. 2C)
extending from the gastric antrum to the pylorus, with
heterogeneous attenuation, accompanied by notable gastric
distension and fluid retention, consistent with pyloric obstruction
and gastrostasis. Although these findings were consistent with
gastrostasis, the primary clinical presentation was dominated by
symptoms of functional gastric outlet obstruction, initially
suggesting a gastroparesis-like mechanism rather than a fixed
mechanical obstruction (Fig. 2).
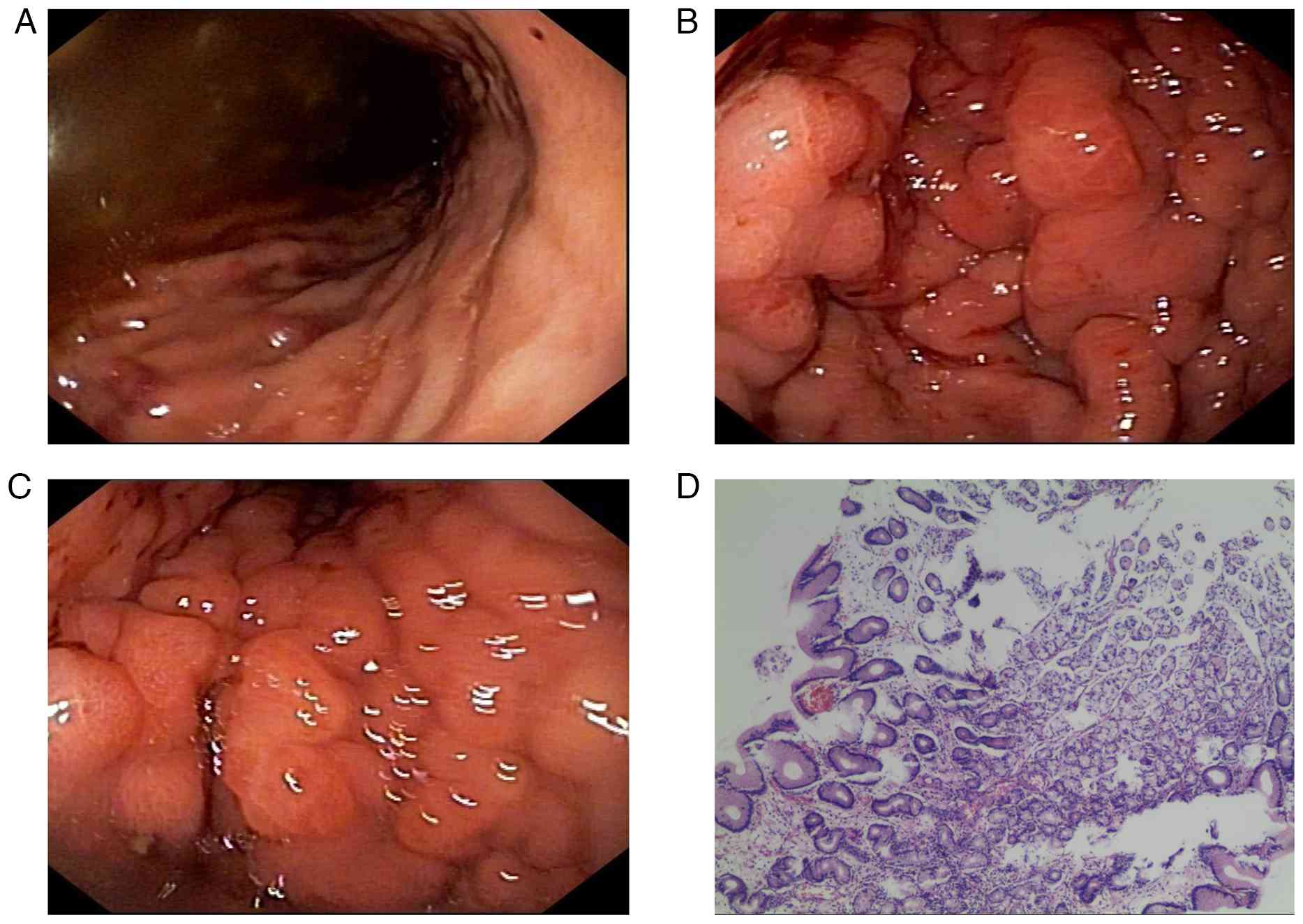
Gastroscopy performed in January 2022 revealed edema and narrowing
in the gastric body and antrum (Fig.
3A-C). Given the poor nutritional status of the patient, a
nasojejunal tube was placed under ultrathin gastroscopic guidance
and biopsies were obtained. Histopathological examination of the
gastric mucosal biopsies on routine sections showed only chronic
superficial gastritis (Fig. 3D).
The initial endoscopic biopsy was therefore considered
non-diagnostic for malignancy.
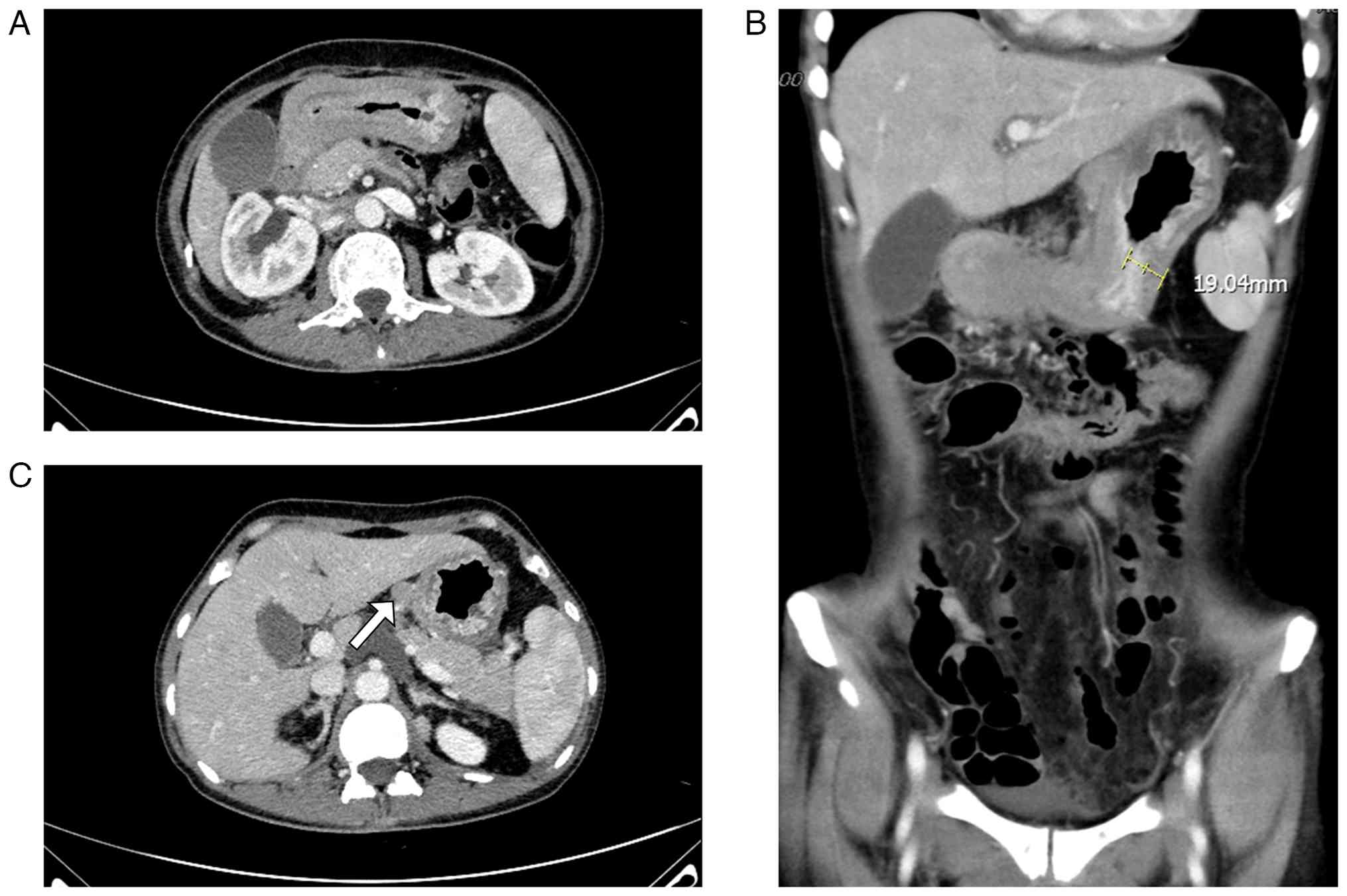
A total of 1 month later (36 days after the last
conducted CT), in February 2022, the patient was readmitted due to
persistent abdominal distension. Follow-up contrast-enhanced CT
performed in February 2022 revealed progressive diffuse and
irregular gastric wall thickening (maximum thickness 19.04 mm;
Fig. 4C) extending from the fundus
and body to the pyloric region, exhibiting heterogeneous
enhancement and focal luminal narrowing (Fig. 4A-C). Additionally, newly enlarged
nodules were detected within the hepatogastric ligament (Fig. 4B). The mesentery, omentum and
peritoneal surfaces exhibited thickening, stranding and nodular
changes, with newly identified implant-like nodules (Fig. S5). These findings were highly
suggestive of metastatic disease. The persistent and progressive
symptoms, coupled with the new radiographic evidence of peritoneal
disease and the ongoing disparity between profound functional
gastric outlet obstruction and the lack of a fixed luminal lesion,
strengthened the suspicion of a motility disorder secondary to
metastatic infiltration. Endoscopic ultrasound (EUS) and gallium-68
fibroblast activation protein inhibitor positron emission
tomography/CT (68Ga-FAPI PET/CT) were considered but not
performed owing to concerns regarding the tolerance and safety of
the patient
Subsequently, an exploratory laparotomy was
performed at The General Hospital of Western Theater Command in
February 2022, whereby ~300 ml of clear yellowish ascitic fluid was
drained intraoperatively. The surgical examination showed a notably
thickened and indurated gastric wall, severe pyloric stenosis,
enlarged perigastric lymph nodes and multiple firm nodules
involving the serosal surfaces of the stomach, bowel and
peritoneum. Representative intraoperative images documenting the
nodular changes on the mesentery, omentum and peritoneal surfaces
were not systematically archived during the exploratory laparotomy.
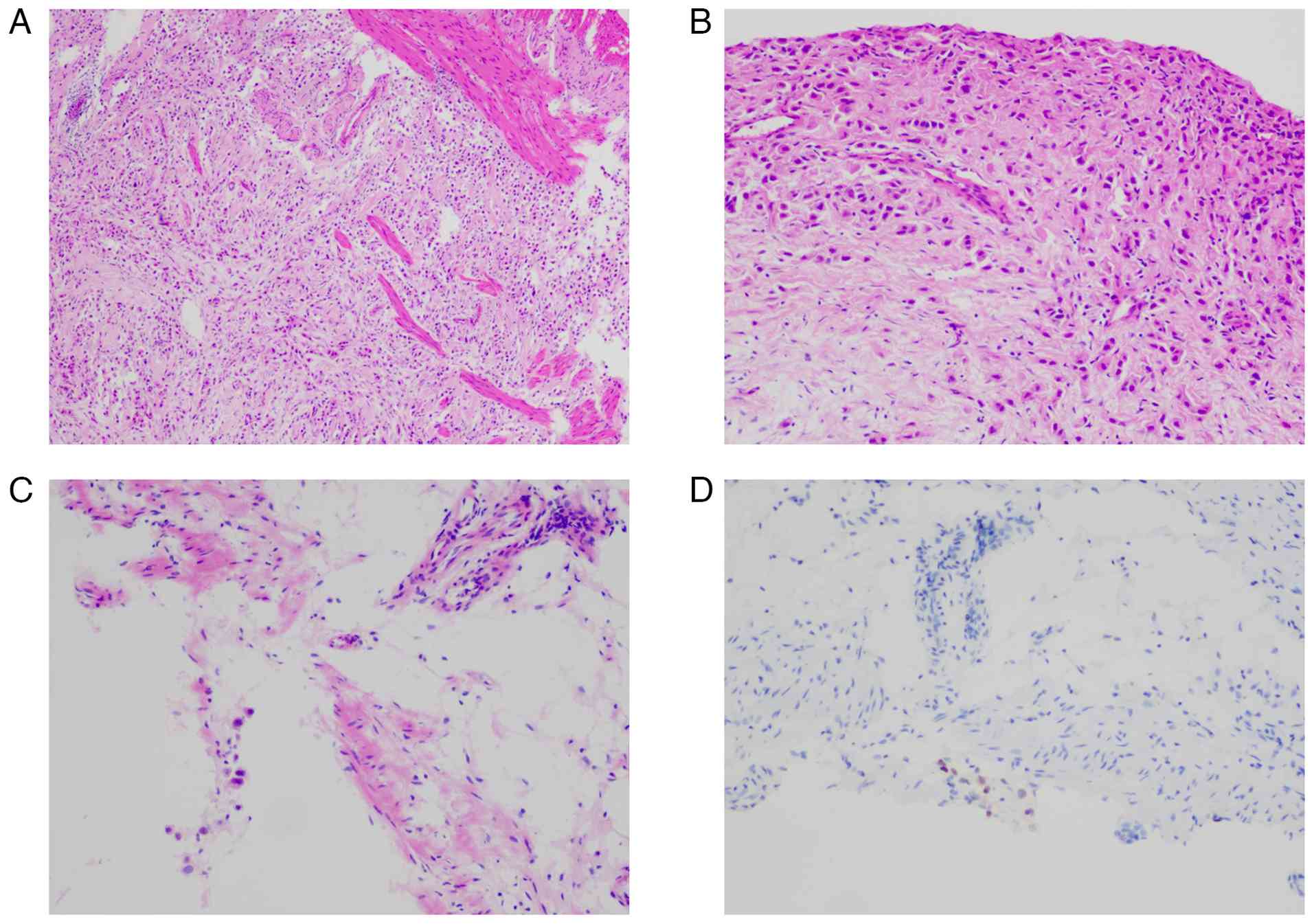
Histopathological examination of the surgical specimens confirmed
the extensive involvement of the intestinal wall, gastric wall and
antrum with features suggesting poorly differentiated
adenocarcinoma (Fig. 5). Omental
biopsy specimens collected during the procedure confirmed a
diagnosis of poorly differentiated adenocarcinoma (Fig. 6A). Immunohistochemical (IHC)
analysis supported the diagnosis of metastatic breast carcinoma
(Fig. 6B-L), demonstrating strong
positive results for estrogen receptor (ER; 80%), GATA binding
protein 3 (GATA3), gross cystic disease fluid protein 15
(GCDFP-15), cytokeratin (CK)-7 and CK8/18. The tumor was negative
for CK20, CK5/6, p63, thyroid transcription factor 1 (TTF-1),
Villin and Wilms tumor 1 (WT-1). The Ki-67 proliferative index was
40% (Fig. S6).
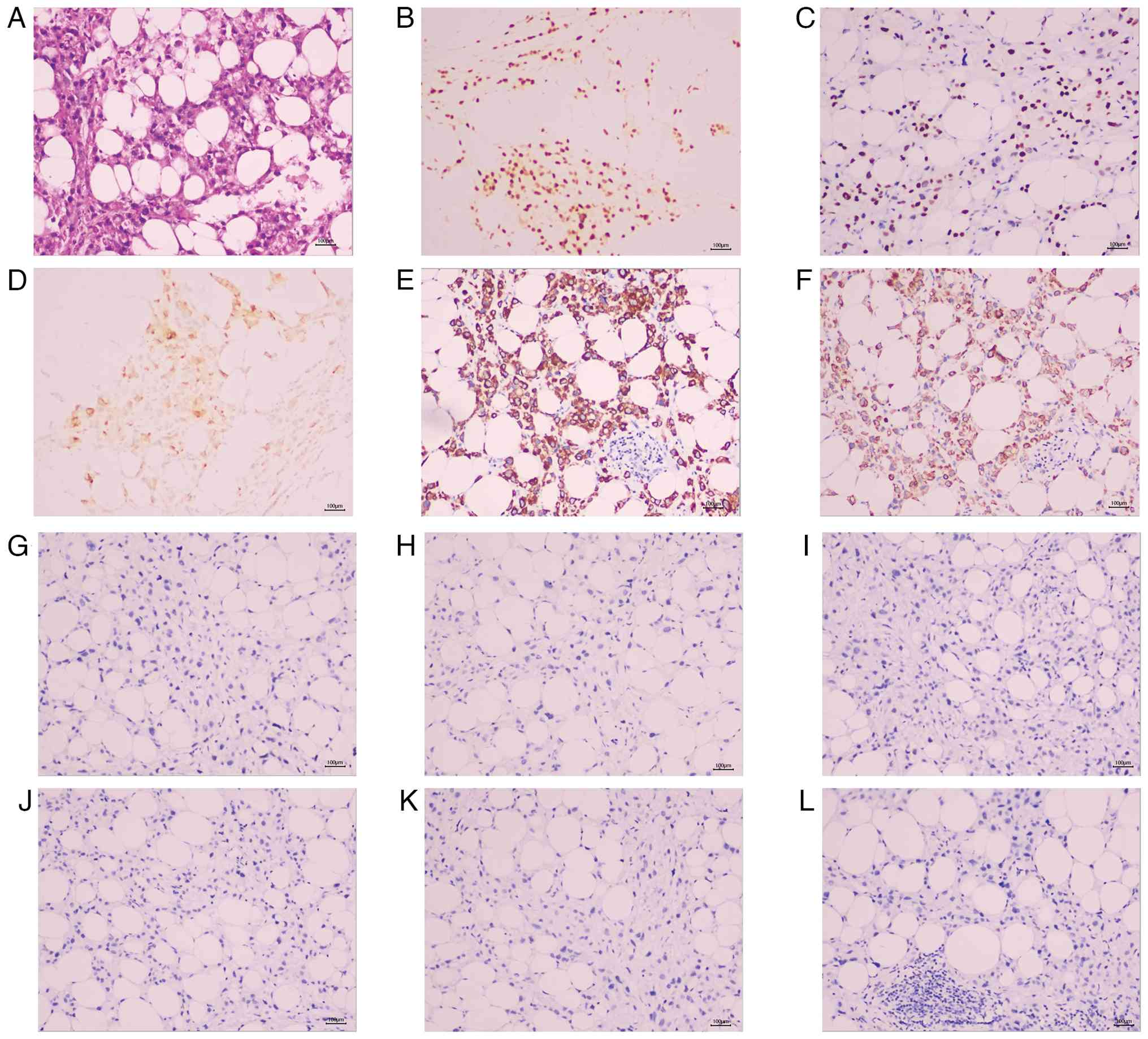 | Figure 6Histopathological and IHC findings of
the omental biopsy. (A) Photomicrograph (H&E stain;
magnification, x100) shows infiltration of poorly differentiated
adenocarcinoma composed of atypical cells with eosinophilic
cytoplasm within the fibroadipose tissue. IHC staining
(magnification, x100) demonstrates strong positivity for (B)
estrogen receptor (80%), (C) GATA binding protein 3, (D) gross
cystic disease fluid protein 15, (E) CK7 and (F) CK8/18. (G-L) The
tumor cells are negative for (G) CK20, (H) CK5/6, (I) p63, (J)
thyroid transcription factor 1, (K) Villin and (L) Wilms tumor 1.
This immunoprofile confirms metastatic breast carcinoma. Scale
bars, 100 µm. IHC, immunohistochemical; CK, cytokeratin. |
This immunoprofile confirmed the breast origin of
the metastasis, consistent with the known primary carcinoma the
patient exhibited. The intraoperative findings thus confirmed
extensive serosal and transmural tumor infiltration, which was
associated with severe gastric dysmotility and functional gastric
outlet obstruction. Given the confirmed diagnosis and the poor
nutritional status of the patient due to functional gastric outlet
obstruction, a jejunostomy was established through an open surgical
technique, with the distal limb placed 20 cm beyond the ligament of
Treitz, for enteral feeding. Owing to the overall poor condition of
the patient, active antitumor therapy was not pursued and
palliative care was initiated, including opioid analgesia (10 mg
morphine subcutaneously every 4 h), antiemetics (8 mg ondansetron
intravenously every 8 h) and total parenteral nutrition (2,000
kcal/day). The patient succumbed to disease progression in May
2022, with malignant ascites as a contributing complication.
To reconcile the initial non-diagnostic gastroscopic
biopsy with the eventual confirmation of extensive peritoneal
metastasis. Retrospective deeper sectioning of the same
paraffin-embedded biopsy block was performed in February 2025
during the preparation of the present case report. This was
prompted by the need to reconcile the initial non-diagnostic
histopathology with the notable evidence of peritoneal metastasis,
which included refractory symptoms (abdominal distension,
dyspepsia, vomiting and marked weight loss) and radiological
findings (marked diffuse gastric wall thickening, gastric
distension and ascites). This later analysis revealed occasional
atypical cells with eosinophilic cytoplasm at the edge of the
submucosa (Fig. 5C), consistent
with submucosal or muscular involvement and explaining the initial
false-negative result. This finding supports the hypothesis of a
mucosa-sparing, serosa-origin metastasis pattern. The absence of
mucosal malignancy, coupled with the radiographic evidence of
impaired gastric emptying despite anatomical patency, raised strong
suspicion for a functional motility disorder consistent with
functional gastric outlet obstruction.
Histopathological and
immunohistochemical analysis
Tissue samples were fixed in 4% neutral buffered
formalin at room temperature for 24-48 h, embedded in paraffin and
sectioned at a thickness of 4 µm. Hematoxylin and eosin (H&E)
staining was performed at room temperature: Sections were stained
with hematoxylin for 5-7 min, followed by eosin for 1-2 min.
For immunohistochemical analysis, 4-µm-thick
paraffin-embedded sections were deparaffinized and rehydrated.
Heat-induced antigen retrieval was performed using citrate buffer
(pH 6.0) at 95-100˚C for 20 min. Staining was performed using the
Titan S automated immunohistochemistry system (Fuzhou Maixin
Biotechnology Development Co., Ltd.). Endogenous peroxidase
activity was blocked with 3% hydrogen peroxide for 15 min at room
temperature. Non-specific binding was blocked using 5% normal
mouse/rabbit serum (Fuzhou Maixin Biotechnology Development Co.,
Ltd.) for 30 min at room temperature. Sections were then incubated
with primary antibodies (dilution 1:100; detailed in Table I) overnight at 4˚C. After washing,
sections were incubated with an HRP-conjugated anti-mouse/rabbit
IgG secondary antibody (ready-to-use; cat. no. KIT-5005; Fuzhou
Maixin Biotechnology Development Co., Ltd.) for 1 h at room
temperature. Signal detection was performed using
3,3'-diaminobenzidine (cat. no. TT-0805; Fuzhou Maixin
Biotechnology Development Co., Ltd.). Sections were counterstained
with hematoxylin for 1-2 min at room temperature. All slides were
examined and imaged under a light microscope (Olympus BX43; Olympus
Corporation) at a magnification of x100 (scale bar, 100 µm).
Positive and negative controls were run concurrently and showed
appropriate staining.
 | Table IAntibodies and interpretation
criteria used for immunohistochemical analysis. |
Table I
Antibodies and interpretation
criteria used for immunohistochemical analysis.
| Antibody | Clone | Cat. no. | Cellular
localization | Positivity
threshold/interpretation | Result (Fig.) |
|---|
| ER | MXR030 | RMA-1065 | Nuclear | ≥1% nuclear
staining (ASCO/CAP guidelines) | Positive, 80%
(6B) |
| GATA3 | EP368 | RMA-1067 | Nuclear | Any distinct
nuclear staining | Positive (6C) |
| GCDFP-15 | MX120 | MAB-1035 | Cytoplasmic | Cytoplasmic
staining | Positive (6D) |
| CK7 | MX053 | MAB-0828 | Cytoplasmic | Cytoplasmic
staining | Positive (6E) |
| CK8/18 | MX004+ MX035 | MAB-1002 | Cytoplasmic | Cytoplasmic
staining | Positive (6F) |
| CK20 | MX059 | MAB-0834 | Cytoplasmic | Cytoplasmic
staining | Negative (6G) |
| CK5/6 | MX040 | MAB-0744 | Cytoplasmic | Cytoplasmic
staining | Negative (6H) |
| p63 | MX013 | MAB-0694 | Nuclear | Nuclear
staining | Negative (6I) |
| TTF-1 | MX011 | MAB-0677 | Nuclear | Nuclear
staining | Negative (6J) |
| Villin | MXR040 | RMA-1090 | Cytoplasmic | Cytoplasmic
staining | Negative (6K) |
| WT-1 | MX012 | MAB-0678 | Nuclear | Nuclear
staining | Negative (6L) |
| Ki-67 | MX006 | MAB-0672 | Nuclear | Percentage of
positive tumor nuclei | 40% (S6) |
Discussion
A guide to the diagnostic workflow of the current
study, from initial presentation to final diagnosis, is outlined in
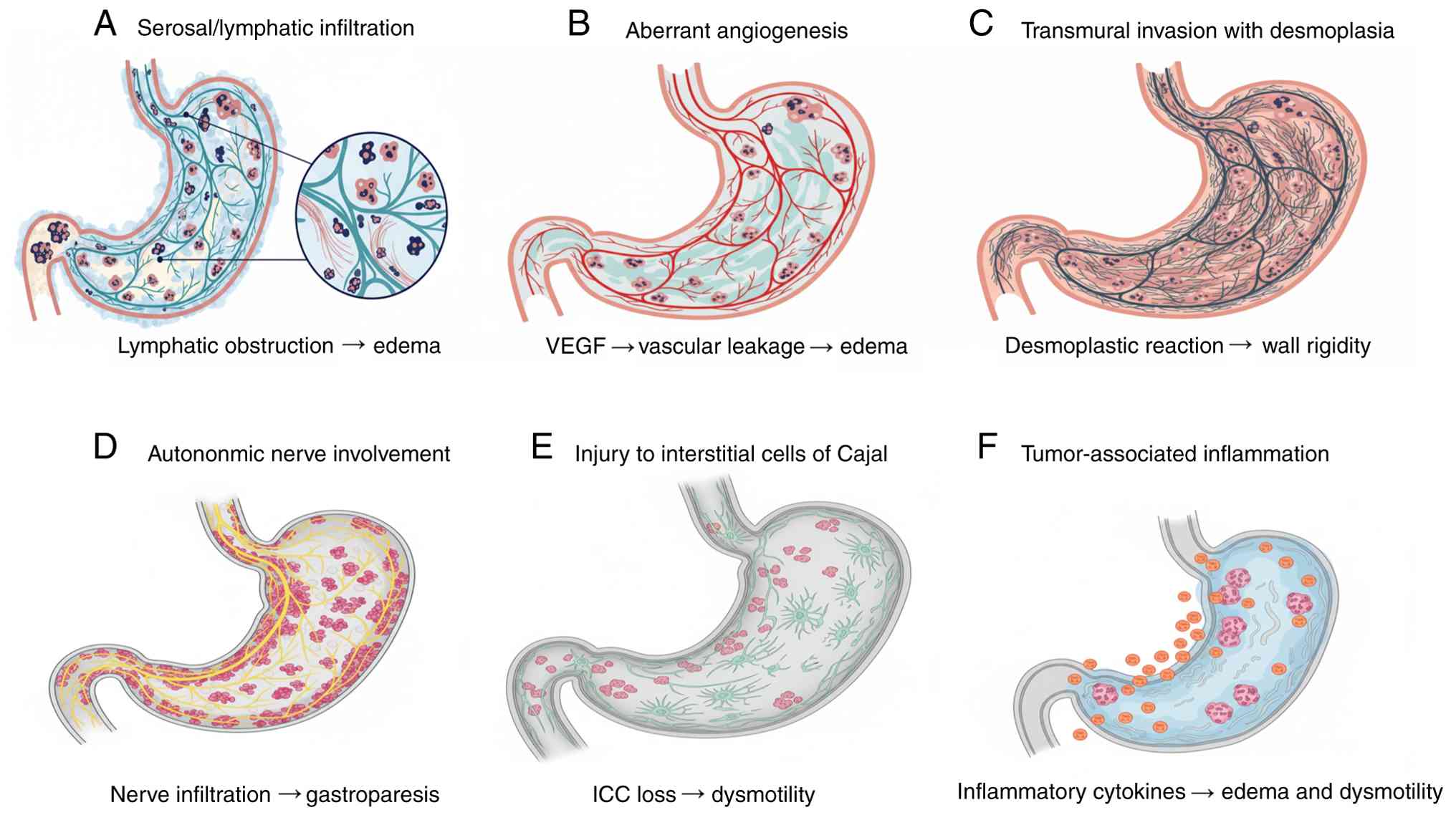
Fig. 7. To visually summarize the
complex and multifactorial pathophysiology proposed, a schematic of
the key interconnected mechanisms is provided in Fig. 8. The present case outlines a rare
but notable manifestation of advanced breast cancer, namely marked
diffuse gastric wall thickening leading to functional gastric
outlet obstruction. While peritoneal metastasis from breast cancer
is itself uncommon, its presentation as predominant gastric
involvement with such notable wall thickening and functional
impairment warrants a detailed exploration of the underlying
pathophysiology. The present study proposes several interconnected
mechanisms that may explain this clinical presentation, beyond the
more common mucosal metastases.
First, extensive serosal and lymphatic infiltration
is likely a primary contributor. Breast cancer cells seeding the
peritoneum can extensively infiltrate the gastric serosa and
subserosal lymphatic network. This widespread infiltration can
obstruct lymphatic drainage, leading to notable edema and
consequent uniform thickening of the gastric wall, a process
occasionally termed ‘lymphangitic carcinomatosis’ of the stomach
wall (a condition characterized by tumor cell infiltration and
obstruction of the gastric wall lymphatic vessels), as described in
cases of metastatic breast cancer to the gastrointestinal tract
(9). This ‘lymphangitic
carcinomatosis’ of the stomach wall primarily affects the outer
layers, explaining the initial normal mucosal appearance on
endoscopy.
Second, aberrant angiogenesis and increased vascular
permeability represent another notable factor. Tumor cells induce
angiogenesis, forming new but structurally abnormal and
hyperpermeable vessels. Furthermore, both tumor-derived and host
inflammatory cells release vasoactive factors, including VEGF,
histamine and bradykinin. This process, which is part of the
peritoneal metastatic microenvironment, leads to a notable increase
in vascular permeability and extensive leakage of plasma proteins
and fluid into the interstitium, causing severe interstitial edema
(10). This process acts
synergistically with lymphatic obstruction to exacerbate the
diffuse, edematous gastric wall thickening observed radiologically.
The resultant interstitial edema may also impair smooth muscle cell
function, further contributing to the gastroparesis-like
symptoms.
Third, transmural tumor invasion and a profound
desmoplastic reaction serve a key role. Although the mucosa was
initially spared, progressive invasion from the serosa through the
muscularis propria can lead to transmural replacement by tumor
cells. From a molecular pathological perspective, certain breast
cancer subtypes, when metastasizing to the peritoneum, may adopt a
growth pattern reminiscent of gastric signet-ring cell carcinoma or
diffuse-type gastric cancer (linitis plastica). This pattern of
infiltrative growth with desmoplastic stromal reaction has been
observed in breast cancer metastases masquerading as primary
gastric malignancies (11).
Furthermore, tumor-associated inflammation, potentially involving
cytokines such as IL-17, can drive fibroblast activation and
collagen deposition, contributing to fibrotic remodeling of the
gastric wall (12). Although IL-17
was not assessed through IHC in the present case, future studies in
similar presentations should consider evaluating IL-17 and related
cytokines to further elucidate their role in the desmoplastic
reaction and fibrotic microenvironment associated with peritoneal
metastases from breast cancer. This further contributes to wall
thickening, rigidity and loss of peristaltic function, culminating
in functional gastric outlet obstruction. This specific
infiltrative pattern provides a molecular and histomorphological
basis for the ‘leather bottle’ stomach appearance, explaining the
marked thickening and functional impairment even in the absence of
a large tumor mass.
Fourth, possible autonomic nerve plexus involvement
provides a plausible explanation for the gastroparesis-like
symptoms. The celiac plexus and other autonomic nerves regulating
gastric motility are located in the retroperitoneum. Extensive
peritoneal carcinomatosis, as seen in the present patient, can
directly invade or exert perineural pressure on these structures.
Perineural invasion is a recognized mechanism of cancer progression
that can disrupt autonomic innervation (13), leading to impaired gastric motility
and emptying, functionally presenting as obstruction (functional
gastric outlet obstruction) even in the absence of a grossly
occluded lumen. The extensive retroperitoneal disease observed on
CT imaging in the present patient supports the potential for such
neural compromise.
Fifth, potential injury to the interstitial cells of
Cajal (ICC) may represent a key, underlying myogenic mechanism for
the profound dysmotility. While not directly proven in the present
case report, the marked functional impairment observed clinically
and radiologically (marked gastric distension despite anatomical
patency) is consistent with the known pathophysiology of ICC loss.
ICCs are the pacemaker cells of the gastrointestinal tract, key in
initiating and coordinating smooth muscle contractions. Tumor
infiltration, local inflammation and ischemia secondary to vascular
compromise can all lead to ICC network damage or depletion. The
loss of functional ICCs has been shown to directly disrupt the
rhythmicity and propagation of gastric contractions, resulting in
severe gastroparesis and pseudo-obstruction, which aligns with the
clinical presentation of functional gastric outlet obstruction
(14). This ICC-centric mechanism,
alongside autonomic nerve dysfunction, comprehensively explains the
neuro-myogenic failure underlying the gastric outlet obstruction
exhibited by the patient.
Finally, a paraneoplastic or inflammatory-mediated
mechanism, though more speculative, cannot be entirely ruled out.
Tumor-associated inflammatory cytokines, as part of the
cancer-related inflammatory response, can affect smooth muscle
function and interstitial fluid dynamics (15), contributing to both dysmotility and
wall edema. This mechanism has also been proposed in cases of
gastric metastasis from breast cancer (16).
In the present patient, the profound gastric wall
thickening and obstructive symptoms may have resulted from a
synergistic combination of these mechanisms. The dominant processes
included lymphatic obstruction and increased vascular permeability
causing marked mural edema, coupled with transmural tumor invasion
and a powerful desmoplastic reaction leading to gastric wall
rigidity. This structural pathology may have been compounded by a
profound functional impairment potentially resulting from both
neural dysfunction (autonomic plexus involvement) and myogenic
failure (ICC injury), which collectively manifested clinically as
severe, refractory symptoms suggestive of functional gastric outlet
obstruction.
Table II
summarizes the key differentiating features between primary linitis
plastica and metastatic breast carcinoma to the stomach,
highlighting the importance of clinical history and IHC profiling
in forming an accurate diagnosis.
 | Table IIContrasting features of primary
linitis plastica and metastatic breast carcinoma to the
stomach. |
Table II
Contrasting features of primary
linitis plastica and metastatic breast carcinoma to the
stomach.
| Feature | Primary linitis
plastica | Metastatic breast
carcinoma to the stomach |
|---|
| Primary tumor
history | Usually absent or
synchronous | Known history of
breast cancer |
| Endoscopic
mucosa | Often abnormal, may
show erosions, rigidity | Frequently normal
initially (mucosa-sparing) |
| Histology
(H&E) | Signet-ring cells
typical | Ductal or lobular
morphology; signet-ring cells rare |
|
Immunohistochemistry |
CK7+/CK20+
(variable) |
CK7+/CK20- |
| | CDX2+
(often) |
CDX2- |
| | E-cadherin loss (in
diffuse-type) | E-cadherin retained
(in ductal carcinoma) |
| |
ER-/PR-
(typically) |
ER+/PR+
(frequently) |
| | GATA3-
(typically) |
GATA3+ |
| |
GCDFP-15- (typically) |
GCDFP-15+ (often) |
| | HER2+
(subset) | HER2 status matches
primary breast cancer |
| Common clinical
context | Primary gastric
cancer symptoms | Often preceded by
widespread metastases |
| Radiological
findings | Gastric wall
thickening, limits plastica | Gastric wall
thickening + peritoneal deposits, ascites |
The present case exemplifies a diagnostic challenge
in terms of contemporary breast oncology, with the struggle to
distinguish CDK4/6 inhibitor-induced gastrointestinal toxicity from
peritoneal carcinomatosis. The temporal onset of abdominal symptoms
within 2 months of palbociclib initiation initially suggested a
class-effect adverse event, which occurs in a notable proportion of
patients. However, key discriminatory features emerged that pointed
decisively toward a malignant etiology, including the unremitting
nature of symptoms for >4 weeks despite drug cessation, their
progression to refractory vomiting and the eventual development of
unequivocal signs of functional gastric outlet obstruction.
Although gastrointestinal symptoms are a well-documented class
effect of CDK4/6 inhibitors (17-19),
their persistence beyond a reasonable washout period must raise
immediate concern for underlying disease progression. This
underscores the necessity for a symptom-based risk stratification
strategy. Low-risk features, such as symptom onset during the first
treatment cycle, responsiveness to antiemetics and prompt
resolution after drug holding, may be managed expectantly. By
contrast, high-risk features, including symptom onset after
multiple cycles, progression despite maximal supportive care,
persistence beyond 2 weeks of drug interruption and accompanying
constitutional symptoms, warrant prompt and comprehensive
diagnostic investigation for disease progression (20). The course of the present patient is
a good example of the latter scenario.
The diagnostic trajectory of the present case
highlights the limitations of conventional techniques in detecting
serosal-based metastases. Primary gastrointestinal malignancies
typically present with mucosal abnormalities on endoscopy. By
contrast, metastatic involvement often manifests as submucosal and
muscular infiltration, frequently yielding normal endoscopic
findings (21), as demonstrated in
the present case, where gastric mucosal biopsies revealed only
chronic superficial gastritis. This ‘mucosal-sparing’
pathophysiology is characteristic of peritoneal metastases, where
malignant cells implant on the outer serosal surface and spread
inward, initially preserving the mucosal lining. This explains both
the initial futility of endoscopy and the delayed appearance of
radiological signs on cross-sectional imaging, which only become
evident after notable transmural infiltration or lymphatic
obstruction develops (9). The
interval between symptom onset and the development of definitive CT
findings (diffuse gastric wall thickening with heterogeneous
enhancement) in the present patient underscores the progressive
nature of this process and mandates a low threshold for serial
imaging in high-risk patients. Studies indicate that incidentally
detected gastrointestinal wall thickening on CT often suggests
underlying pathology, with malignancies accounting for ~30% of
cases (22-25).
Emerging functional imaging techniques may offer
enhanced diagnostic sensitivity in this challenging scenario. For
example, Li et al (21)
reported that 68Ga-FAPI PET/CT showed markedly higher
tracer uptake in thickened gastric lining and peritoneum compared
with fluorine-18 fluorodeoxyglucose PET/CT, and these findings may
greatly aid in diagnosing metastatic, gastric, peritoneal
involvement (21,26). However, imaging studies
specifically focused on indirect, infiltrative gastric metastases
remain limited. With regard to gastroscopy, metastatic deposits
typically present in one of three patterns: i) A volcanic ulcer,
ii) single or multiple localized nodules or polypoid lesions or
iii) diffusely involved, rigid gastric walls with luminal
narrowing. Notably, as the majority of gastric metastases originate
from submucosal and muscular infiltration, endoscopic findings can
often be deceptively normal, further underscoring the necessity for
deep-wall biopsies or advanced imaging when clinical suspicion is
high (27,28).
Therefore, in patients with a history of breast
cancer and high clinical suspicion of peritoneal metastasis,
proactive diagnostic measures, such as EUS, repeated
cross-sectional imaging, advanced PET/CT techniques and, when
clinical concern persists, diagnostic laparoscopy, should be
pursued to establish a definitive diagnosis, even when initial
biopsies are negative. In the present case, EUS was not performed
due to the severe luminal narrowing and patient instability and
68Ga-FAPI PET/CT, although considered, was not performed
as it was unavailable at the institution and was deemed impractical
given concerns regarding tolerability and safety.
Histopathological features of peritoneal metastatic
carcinoma are often non-specific, making determination of the tumor
origin based solely on conventional H&E staining challenging.
The comprehensive IHC analysis proved decisive in confirming the
breast origin of the peritoneal metastases and exemplifies the
systematic approach required for accurate diagnosis of unknown
peritoneal malignancies. The strong nuclear expression of ER (80%)
provided the first definitive indicator of a HR-positive breast
primary, as gastrointestinal adenocarcinomas rarely express ER.
GATA3, a transcription factor with high specificity for breast
carcinoma, served as a notable marker in this context (29,30),
while GCDFP-15 maintains high specificity for breast tissue and
provided additional confirmatory evidence. The CK profile
(CK7+/CK20-) effectively excluded colorectal
adenocarcinoma (typically CK7-/CK20+) and
supported an origin from the breast, lung or gynecological tract.
The subsequent negation of TTF-1 (lung), WT-1 (ovarian serous
carcinoma) and Villin (intestinal differentiation) completed this
diagnostic algorithm, creating an immunophenotypic profile
pathognomonic for breast origin. This systematic IHC approach,
beginning with ER status, then employing tissue-specific markers
(GATA3) and finally utilizing exclusion markers, provided a robust
framework for determining the origin of peritoneal metastases,
which is particularly important in patients with a history of
multiple potential primaries.
It is uncommon for breast cancer to spread to the
peritoneum, instead metastases more frequently originate from
ovarian, colorectal or gastric primary tumors (10). The clinical presentation of
peritoneal metastasis is highly variable and non-specific. Symptoms
such as dyspepsia, anorexia, early satiety, epigastric pain,
vomiting and hematemesis often mimic those of benign
gastrointestinal disorders or primary gastric cancer, frequently
leading to diagnostic delays. Among these manifestations, gastric
outlet obstruction represents a particularly severe complication.
Previous studies have documented that gastric metastases from
breast cancer can result in pyloric stenosis (31,32).
Although the symptoms of metastatic gastric involvement are
non-specific and may resemble those of primary gastric cancer,
malignant gastric outlet obstruction (MGOO) due to pyloric or
duodenal obstruction markedly compromises both patient survival and
quality of life. Primary treatment options for MGOO include
surgical gastrojejunostomy, endoscopic placement of self-expanding
metal stents and endoscopic ultrasound-guided gastroenterostomy
using lumen-apposing metal stents (33). In the present case, the severity of
pyloric stenosis precluded stent placement, necessitating
jejunostomy.
Owing to the rarity of peritoneal metastasis
originating from breast cancer, no consensus exists regarding its
management and no large-scale studies have compared treatment
strategies (34). Nevertheless,
palliative surgery remains necessary to address symptomatic
obstruction, bleeding or perforation, even in the absence of a
demonstrated survival benefit (35). However, a number of retrospective
studies have described the combined use of cytoreductive surgery
and hyperthermic intraperitoneal chemotherapy in patients with
secondary peritoneal carcinomatosis from breast cancer and other
primaries, reporting improvements in both morbidity and mortality
rates (36-38).
However, these studies are limited by their inclusion of only
patients with recurrent disease, their retrospective design and
small sample sizes. Consequently, robust evidence regarding optimal
management and accurate prognostic assessment remains scarce.
Treatment should be individualized based on patient-specific
factors, anticipated performance status and quality-of-life
considerations. The role of surgery warrants further investigation
in prospective studies focusing on patients with limited metastatic
burden, no extraperitoneal disease and a high likelihood of
complete cytoreduction.
Within the present case report, the limitations
inherent to single-case reports must be acknowledged. However, the
detailed diagnostic process and clinicopathological associations
presented offer valuable insights for clinicians encountering
similar diagnostic challenges. Future prospective studies are
needed to establish optimal diagnostic algorithms and management
strategies for this distinct clinical population. Numerous
limitations within the present study must be acknowledged. Firstly,
as a single-case report, the findings cannot be generalized.
Secondly, the diagnostic evaluation was constrained by the critical
condition of the patient and available resources. Specifically,
objective gastric motility studies (such as scintigraphy or
manometry) were not performed and advanced diagnostic modalities
such as EUS and 68Ga-FAPI PET/CT were not available at
the institution at the time. These factors preclude definitive
conclusions on the functional nature of the obstruction and may
have delayed the diagnosis. Finally, the quality of IHC staining,
as presented in Fig. 6B-L, is
suboptimal, which is attributed to fading of the original slides
over time.
Overall, the present case underscores the importance
of maintaining vigilant suspicion for peritoneal metastasis in
patients with breast cancer receiving CDK4/6 inhibitors who develop
persistent gastrointestinal symptoms. The present study advocates
for a structured diagnostic approach that recognizes the
limitations of conventional imaging and endoscopy for detecting
serosal-based disease, utilizes IHC strategically for tissue
confirmation and employs a low threshold for surgical evaluation
when clinical suspicion persists despite initially negative
investigations. Timely diagnosis remains key in implementing
appropriate palliative interventions that can alleviate functional
gastric outlet obstruction and preserve quality of life, and for
identifying patients who may be candidates for more aggressive
cytoreductive approaches where appropriate.
Supplementary Material
Histopathological and IHC findings of
the primary breast tumor and CT-guided pelvic biopsy. (A-F)
Photomicrographs of the primary breast tumor (invasive ductal
carcinoma): (A) H&E staining (x100 magnification); (B) Estrogen
receptor-positive (IHC; x100 magnification); (C) progesterone
receptor-positive (IHC; x100 magnification); (D) HER2-negative
(IHC; x100 magnification); (E) Ki-67 proliferative index ~20% (IHC;
x100 magnification); (F) Photomicrograph (H&E; x100
magnification) of the CT-guided pelvic biopsy performed in July
2021, showing fragments of cortical bone, inflammatory exudate and
necrotic bone, with no malignant cells identified. IHC,
immunohistochemistry.
Chest CT and bone scintigraphy
findings suggestive of osseous metastases. (A and B) Representative
images from a chest CT scan (bone window) performed in December
2019, showing multiple punctate and patchy sclerotic densities
involving the bilateral humeral heads, scapulae, sternum, thoracic
vertebral bodies and portions of the ribs, suggestive of osseous
metastases. (C) Whole-body bone scan revealing multiple foci of
increased metabolic activity throughout the skeleton, highly
indicative of metastatic disease.
Spinal MRI demonstrating progression
of bony metastases. (A and B) Spinal MRI performed in December
2019: (A) T1WI showing scattered speckled and patchy abnormal
signals in multiple vertebrae; (B) T2WI with corresponding signal
abnormalities. (C and D) Follow-up spinal MRI performed in October
2021: (C) T1WI and (D) T2WI demonstrating unequivocal progression
of bony lesions. WI, weighted image.
Longitudinal trends of serum tumor
markers. Graph showing the dynamic changes in serum levels of CEA,
CA125 and CA15-3 from the diagnosis of bone metastasis (December
2019) to the diagnosis of peritoneal disease (January 2022). CEA,
carcinoembryonic antigen; CA, carbohydrate antigen.
Contrast-enhanced abdominal CT imaging
demonstrating peritoneal involvement. (A) Axial CT image shows
thick-ening and stranding of the mesentery (arrow). (B) Axial CT
image reveals nodular changes and thickening of the omentum. (C)
Coronal CT image depicts newly identified implant-like nodules on
the peritoneal surface (arrow). These findings are consistent with
peritoneal carcinomatosis.
IHC staining for Ki-67.
Photomicrograph (IHC; magnification, x100) shows a Ki-67
proliferative index of ~40% in the tumor cells from the omental
biopsy. IHC, immunohistochemistry.
Acknowledgements
The authors would like to thank Dr Fangyuan Kong
(Department of Oncology, The General Hospital of Western Theater
Command, Chengdu, China) for their insightful perspective on the
diagnostic challenge of differentiating CDK4/6 inhibitor toxicity
from disease progression in metastatic breast cancer. Their
clinical expertise contributed to enriching the discussion on
patient management strategies.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
BH and LLI contributed equally to this work. BH
contributed to patient management, clinical data collection from
the medical records (including treatments, laboratory results,
imaging findings and follow-up information), data curation, figure
preparation and initial manuscript drafting. LLI contributed to
clinical and imaging data collection, ultrasonographic evaluation,
image acquisition and interpretation, and manuscript review. TY was
responsible for pathological diagnosis, immunohistochemical
analysis and critical revision of the manuscript for important
intellectual content. YT contributed to clinical data collection,
literature review and manuscript editing. LZ was responsible for
study conception and design, supervision, manuscript revision and
final approval. All authors have read and approved the final
version of the manuscript. BH and LZ confirm the authenticity of
all the raw data.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Written informed consent for the publication of
anonymized clinical details and images was obtained from the
patient's husband following the death of the patient.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Riihimäki M, Thomsen H, Sundquist K,
Sundquist J and Hemminki K: Clinical landscape of cancer
metastases. Cancer Med. 7:5534–5542. 2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Yu B, Yan L, Wang H and Yang J and Yang J:
Invasive lobular carcinoma of the breast: Metastatic patterns and
treatment modalities-a review. Front Oncol.
15(1631670)2025.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Bhaludin BN, Tunariu N, Koh DM, Messiou C,
Okines AF, McGrath SE, Ring AE, Parton MM, Sharma B, Gagliardi T,
et al: A review on the added value of whole-body MRI in metastatic
lobular breast cancer. Eur Radiol. 32:6514–6525. 2022.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Inoue M, Nakagomi H, Nakada H, Furuya K,
Ikegame K, Watanabe H, Omata M and Oyama T: Specific sites of
metastases in invasive lobular carcinoma: A retrospective cohort
study of metastatic breast cancer. Breast Cancer. 24:667–672.
2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Sledge GW Jr, Toi M, Neven P, Sohn J,
Inoue K, Pivot X, Burdaeva O, Okera M, Masuda N, Kaufman PA, et al:
MONARCH 2: Abemaciclib in combination with fulvestrant in women
with HR+/HER2- advanced breast cancer who had progressed while
receiving endocrine therapy. J Clin Oncol. 35:2875–2884.
2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Hortobagyi GN, Stemmer SM, Burris HA, Yap
YS, Sonke GS, Paluch-Shimon S, Campone M, Blackwell KL, André F,
Winer EP, et al: Ribociclib as first-line therapy for HR-positive,
advanced breast cancer. N Engl J Med. 375:1738–1748.
2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Edge SB and Compton CC: The American joint
committee on cancer: The 7th edition of the AJCC cancer staging
manual and the future of TNM. Ann Surg Oncol. 17:1471–1474.
2010.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Oken MM, Creech RH, Tormey DC, Horton J,
Davis TE, McFadden ET and Carbone PP: Toxicity and response
criteria of the Eastern cooperative oncology group. Am J Clin
Oncol. 5:649–655. 1982.PubMed/NCBI
|
|
9
|
Nazareno J, Taves D and Preiksaitis HG:
Metastatic breast cancer to the gastrointestinal tract: A case
series and review of the literature. World J Gastroenterol.
12:6219–6224. 2006.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Mikuła-Pietrasik J, Uruski P, Tykarski A
and Książek K: The peritoneal ‘soil’ for a cancerous ‘seed’: A
comprehensive review of the pathogenesis of intraperitoneal cancer
metastases. Cell Mol Life Sci. 75:509–525. 2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Schwarz RE, Klimstra DS and Turnbull AD:
Metastatic breast cancer masquerading as gastrointestinal primary.
Am J Gastroenterol. 93:111–114. 1998.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Zhou G, Li A and Wang R: The role of IL-17
family in the process of pulmonary fibrosis. Life Conflux: Jun 30,
2025 (Epub ahead of print).
|
|
13
|
Liebig C, Ayala G, Wilks JA, Berger DH and
Albo D: Perineural invasion in cancer: A review of the literature.
Cancer. 115:3379–3391. 2009.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Wang TH, Angeli TR, Ishida S, Du P,
Gharibans A, Paskaranandavadivel N, Imai Y, Miyagawa T, Abell TL,
Farrugia G, et al: The influence of interstitial cells of Cajal
loss and aging on slow wave conduction velocity in the human
stomach. Physiol Rep. 8(e14659)2021.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Mantovani A, Allavena P, Sica A and
Balkwill F: Cancer-related inflammation. Nature. 454:436–444.
2008.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Villa Guzmán JC, Espinosa J, Cervera R,
Delgado M, Patón R and Cordero García JM: Gastric and colon
metastasis from breast cancer: Case report, review of the
literature, and possible underlying mechanisms. Breast Cancer (Dove
Med Press). 9:1–7. 2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Thill M and Schmidt M: Management of
adverse events during cyclin-dependent kinase 4/6 (CDK4/6)
inhibitor-based treatment in breast cancer. Ther Adv Med Oncol: Sep
3, 2018 (Epub ahead of print).
|
|
18
|
Onesti CE and Jerusalem G: CDK4/6
inhibitors in breast cancer: Differences in toxicity profiles and
impact on agent choice. A systematic review and meta-analysis.
Expert Rev Anticancer Ther. 21:283–298. 2021.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Braal CL, Jongbloed EM, Wilting SM,
Mathijssen RHJ, Koolen SLW and Jager A: Inhibiting CDK4/6 in breast
cancer with palbociclib, ribociclib, and abemaciclib: Similarities
and differences. Drugs. 81:317–331. 2021.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Cardoso F, Paluch-Shimon S, Senkus E,
Curigliano G, Aapro MS, André F, Barrios CH, Bergh J, Bhattacharyya
GS, Biganzoli L, et al: 5th ESO-ESMO international consensus
guidelines for advanced breast cancer (ABC 5). Ann Oncol.
31:1623–1649. 2020.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Li T, Jiang X, Zhang Z, Chen X, Wang J,
Zhao X and Zhang J: Case report: 68Ga-FAPI PET/CT, a
more advantageous detection mean of gastric, peritoneal, and
ovarian metastases from breast cancer. Front Oncol 12:1013066,
2022.
|
|
22
|
Somuncu E, Topal Ü, Sönmez S, Kara Y,
Bozdağ E, Özcan A, Başaran C, Özkan C, Tatlıdil YE and Kalaycı MU:
Incidentally detected gastrointestinal wall thickness on abdominal
computed tomography; What does it mean for endoscopy? Arch Iran
Med. 24:296–300. 2021.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Cai Q, Baumgarten DA, Affronti JP and
Waring JP: Incidental findings of thickening luminal
gastrointestinal organs on computed tomography: An absolute
indication for endoscopy. Am J Gastroenterol. 98:1734–1737.
2003.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Bleibel W, Guerrero JE, Kim S, Leao L,
Ghosh T and Kenney TJ Jr: The clinical significance of incidental
computer tomography finding of gastrointestinal luminal wall
thickening as evaluated by endoscopy. Dig Dis Sci. 52:1709–1712.
2007.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Tellez-Avila FI, García-Osogobio S,
Chavez-Tapia NC, Ramirez-Luna MA, Franco-Guzman A, Sosa-Lozano A
and Giovannini M: Utility of endoscopy in patients with incidental
gastrointestinal luminal wall thickening detected with CT. Surg
Endosc. 23:2191–2196. 2009.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Hathi DK and Jones EF: 68Ga
FAPI PET/CT: Tracer uptake in 28 different kinds of cancer. Radiol
Imaging Cancer. 1(e194003)2019.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Taal BG, Peterse H and Boot H: Clinical
presentation, endoscopic features, and treatment of gastric
metastases from breast carcinoma. Cancer. 89:2214–2221.
2000.PubMed/NCBI
|
|
28
|
De Palma GD, Masone S, Rega M, Simeoli I,
Donisi M, Addeo P, Iannone L, Pilone V and Persico G: Metastatic
tumors to the stomach: Clinical and endoscopic features. World J
Gastroenterol. 12:7326–7328. 2006.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Miettinen M, McCue PA, Sarlomo-Rikala M,
Rys J, Czapiewski P, Wazny K, Langfort R, Waloszczyk P, Biernat W,
Lasota J and Wang Z: GATA3: A multispecific but potentially useful
marker in surgical pathology: A systematic analysis of 2500
epithelial and nonepithelial tumors. Am J Surg Pathol. 38:13–22.
2014.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Shield PW, Papadimos DJ and Walsh MD:
GATA3: A promising marker for metastatic breast carcinoma in serous
effusion specimens. Cancer Cytopathol. 122:307–312. 2014.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Kusunoki C, Hamakawa T, Mano M, Nishikawa
K, Toshiyama R, Miyo M, Fujiwara A, Miyake M, Hama N, Miyamoto A,
et al: A case of advanced gastric cancer with umbilical metastasis,
pyloric stenosis, and peritoneal dissemination underwent palliative
surgery after chemotherapy. Gan To Kagaku Ryoho. 47:513–515.
2020.PubMed/NCBI(In Japanese).
|
|
32
|
Satoh E, Yatabe Y, Uehira D, Yonekura K,
Murakata A, Toyofuku Y, Tanami H, Osanai T, Sugano N, Sakoma T and
Maruyama S: A case of pyloric stenosis due to gastric metastasis of
breast cancer. Gan To Kagaku Ryoho. 48:2103–2105. 2021.PubMed/NCBI(In Japanese).
|
|
33
|
Troncone E, Fugazza A, Cappello A, Del
Vecchio Blanco G, Monteleone G, Repici A, Teoh AYB and Anderloni A:
Malignant gastric outlet obstruction: Which is the best therapeutic
option? World J Gastroenterol. 26:1847–1860. 2020.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Beniey M: Peritoneal metastases from
breast cancer: A scoping review. Cureus. 11(e5367)2019.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Tsujimura K, Teruya T, Kiyuna M, Higa K,
Higa J, Iha K, Chinen K, Asato M, Takushi Y, Ota M, et al: Colonic
metastasis from breast carcinoma: A case report. World J Surg
Oncol. 15(124)2017.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Cardi M, Sammartino P, Framarino ML,
Biacchi D, Cortesi E, Sibio S, Accarpio F, Luciani C, Palazzo A and
di Giorgio A: Treatment of peritoneal carcinomatosis from breast
cancer by maximal cytoreduction and HIPEC: A preliminary report on
5 cases. Breast. Oktober. 22:845–849. 2013.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Cardi M, Sammartino P, Mingarelli V, Sibio
S, Accarpio F, Biacchi D, Musio D, Sollazzo B and Di Giorgio A:
Cytoreduction and HIPEC in the treatment of ‘unconventional’
secondary peritoneal carcinomatosis. World J Surg Oncol.
13(305)2015.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Yu JH, Feng Y, Li XB, Zhang CY, Shi F, An
SL, Liu G, Zhang YB, Zhang K, Ji ZH, et al: Cytoreductive surgery
and hyperthermic intraperitoneal chemotherapy for peritoneal
metastasis from breast cancer: A preliminary report of 4 cases.
Gland Surg. 10:1315–1324. 2021.PubMed/NCBI View Article : Google Scholar
|