1. Introduction
Fatigue, as one of the most common and complex
symptoms in clinical practice, has notable impacts on individual
health status, quality of life and socioeconomic development
(1-3).
According to epidemiological research data from 2020, 15-20% of the
global population often experience notable fatigue symptoms
(1), and this proportion may be
higher in certain populations, especially against the backdrop of
the coronavirus disease 2019 (COVID-19) pandemic: 40-60% of
recovered patients with COVID-19 experience persistent fatigue
symptoms (4).
The widespread application of next-generation
multi-omics technologies has revealed novel molecular mechanisms
and biomarkers (5,6), providing novel research directions
and technical support for early diagnosis, precise treatment and
prognosis assessment. Artificial intelligence (AI), machine
learning and big data analysis in fatigue diagnosis and management
have notably improved clinical practice efficiency and accuracy,
opening novel avenues for individualized diagnosis and treatment
(7-10).
Understanding of fatigue as a complex multisystem disease has
shifted treatment paradigms towards integrated multidisciplinary
approaches. The present review aimed to provide evidence-based
recommendations for clinical practice, ultimately promoting the
improvement of fatigue diagnosis and treatment outcomes.
2. Fatigue: Definition and classification
framework
Evolution of definitions
The conceptualization of fatigue has evolved from a
unidimensional symptom description to multidimensional syndrome
recognition. Traditional definitions emphasize a decline in
physical or mental vitality, whereas contemporary perspectives
integrate physiological, psychological and functional dimensions.
Historical physiological definitions described fatigue as a
temporary decrease in the ability of the body to work after
sustained or repeated activity (11). Psychology emphasizes the subjective
experience of fatigue, defining it as an unpleasant feeling
accompanied by decreased motivation, reduced vigor, cognitive
slowing and attention difficulties (12).
Research has redefined fatigue from a systems
biology and precision medicine as a complex biological process
involving energy metabolism, cognitive function and neuroimmune
regulation, with marked individual heterogeneity, whose occurrence
and development are influenced by genetic, environmental and
psychosocial factors (1). This
definition reflects a multi-level understanding of the nature of
fatigue.
Classification system
Modern medicine has a deeper understanding of
fatigue, and its classification system is constantly being
improved. Based on the latest research evidence, fatigue can be
classified along multiple dimensions (Tables I and II).
 | Table IClassification of fatigue. |
Table I
Classification of fatigue.
| Classification
dimension | Key
characteristics | (Refs.) |
|---|
| Etiology | | (143) |
|
Primary | Represented by
myalgic encephalomyelitis/chronic fatigue syndrome, lacking clear
organic etiology | (143) |
|
Secondary | Caused by specific
underlying diseases, including neurological disorder, autoimmune
disease and endocrine metabolic disorders | (143) |
| Clinical
presentation | | (3) |
|
Predominantly
physical | Decreased physical
strength and exercise endurance; delayed post-exertional
recovery | (3) |
|
Predominantly
cognitive | Difficulty
maintaining attention, working memory decline; executive function
impairment; ~40% of patients show marked cognitive dysfunction | (144) |
|
Mixed
type | Coexistence of
physical and cognitive symptoms; ~45% of patients experience
simultaneous decline in physical and cognitive function | (118) |
| Disease course | | (145) |
|
Acute | <1 month | (145) |
|
Subacute | 1-6 months | (145) |
|
Chronic | >6 months,
typically accompanied by marked neuroimmune functional changes and
energy metabolism abnormality | (145) |
| Functional
impact | | - |
|
Mild | Slight limitation
on daily activity | - |
|
Moderate | Marked impact on
work and social functioning | - |
|
Severe | Requires continual
bed rest | - |
| Molecular
phenotyping | | (146) |
|
Energy
metabolism dysfunction | Mitochondrial
dysfunction and decreased ATP synthesis | (146) |
|
Neuroendocrine
dysregulation |
Hypothalamus-pituitary-adrenal axis
regulates the stress response of the body | (146) |
|
Immune
dysfunction | Abnormal
inflammatory factors and altered immune cell function | (146) |
|
Mixed
type | Improvements in
autonomic nervous system function and cardiovascular
parameters | (146) |
 | Table IISubtypes of fatigue. |
Table II
Subtypes of fatigue.
| Subtype | Biomarkers | Potential
mechanism | Treatment | Research
priority |
|---|
| Metabolic | Decreased ATP and
coenzyme Q10; increased lactate | Mitochondrial
dysfunction | Energy support
therapy | High |
|
Neuroinflammatory | Increased IL-6,
TNF-α and CRP | Immune
activation | Anti-inflammatory
drugs | High |
| Neuroendocrine | Decreased cortisol
and growth hormone |
Hypothalamus-pituitary-adrenal axis
dysfunction | Hormone
replacement | Medium |
| Cognitive | Decreased BDNF;
abnormal functional connectivity between specific brain regions,
including the globus pallidus, left lateral occipital cortex and
cuneus | Neural network
disruption | Cognitive
enhancement | Medium |
| Mixed | Decreased HRV | Multi-system
involvement | Combination
therapy | High |
3. Common causes of secondary fatigue
Fatigue is an independent syndrome, but it is also
often secondary to various types of disease, such as rheumatoid
arthritis, Parkinson's disease, viral hepatitis and major
depressive disorder. Thus, identifying the causes of fatigue is key
for treatment.
Neurological disease
Nervous system diseases are one of the notable
causes of secondary fatigue. Manjaly et al (13) found that 60-90% of patients with
multiple sclerosis (MS) experience fatigue symptoms; in addition,
fatigue is not only one of the most common symptoms in patients
with MS but may also be an early manifestation. Central fatigue
(14) is associated with the
inflammatory response, demyelinating lesions and axonal damage in
the central nervous system (15).
Fatigue is related to factors such as
hypothalamus-pituitary-adrenal (HPA) axis dysfunction and increased
inflammatory factors (16). A
meta-analysis of 7,427 patients with Parkinson's disease showed
that the prevalence of fatigue is 50%, which may be related to the
hypoactivity of dopaminergic activity in the nigrostriatal pathway
(17). In addition, stroke,
epilepsy, brain trauma and spinal cord injury cause secondary
fatigue (15,18,19).
Endocrine system disease
Fatigue associated with endocrine system disorders
has been extensively studied (16,19-21).
Fatigue levels in patients with hypothyroidism are positively
associated with thyroid stimulating hormone levels, and their
symptoms improve with hormone replacement therapy (22). Thyroid hormones affect the
metabolism and neural activity of the body through multiple
mechanisms such as regulating mitochondrial function and
neurotransmitters, whereas hypopituitarism causes multiple hormone
deficiencies, resulting in common fatigue symptoms that are
difficult to relieve (23). In
terms of adrenal insufficiency, Husebye et al (24) found that patients with primary or
secondary adrenal insufficiency may experience severe fatigue,
decreased quality of life and ability to work and increased
mortality.
Immune disease
Fatigue is one of the main symptoms of autoimmune
diseases. Systemic lupus erythematosus (SLE) fatigue may be
associated with neuroendocrine immune regulation disorder mediated
by inflammatory factors such as autoantibodies. Previous studies
(25,26) have shown that the incidence of
fatigue in patients with SLE is 67-90%, and the degree of fatigue
is associated with disease activity (27). Data from a fatigue visual analogue
scale (VAS) study showed that 50% of patients with rheumatoid
arthritis have fatigue symptoms, which are mainly related to joint
inflammation and dysfunction, in which inflammatory mediators such
as TNF-α serve a notable role (28). Fatigue associated with autoimmune
disease has a complex pathogenesis, involving inflammation,
autoimmunity and neuroendocrine aspects. Immunomodulatory treatment
targeting the primary disease alleviates fatigue symptoms (29).
Chronic infectious disease
Infections such as viruses, bacteria and parasites
can cause chronic inflammatory responses, leading to secondary
fatigue. In patients with chronic viral hepatitis, symptoms such as
depression, anxiety, fatigue, neurocognitive disease and sleep
disorder are detected in 50% of cases, which markedly affect the
quality of life of patients (30).
Elevated levels of IgM and IgA antibodies to the exotoxin
lipopolysaccharide (LPS), a potent microbe-associated molecular
pattern, as well as elevated blood levels of bacterial LPS,
LPS-binding protein and soluble CD14, are observed in myalgic
encephalomyelitis/chronic fatigue syndrome (ME/CFS) (31). A systematic review of follow-up
studies of 45 patients with coronavirus 2019 found that the most
common symptoms are long-term, such as shortness of breath, fatigue
and sleep disturbance (32). In
addition, chronic infection, such as tuberculosis, brucellosis and
malaria, also causes notable fatigue (33-35).
The mechanism of infection-associated fatigue may involve direct
damage from pathogens, inflammatory factors and metabolic disorders
(21).
Malignant tumors
Tumor-associated fatigue is one of the most common
symptoms in patients with cancer, with the incidence of fatigue
being 60-80%, and the incidence of severe fatigue being ~40%
(36). Severe fatigue is defined
according to the Numeric Rating Scale recommended by the European
Society for Medical Oncology, with a cutoff score of ≥4 on a 0-10
scale (37). In a 2020
meta-analysis of 144,813 participants, the diagnostic rate of
cancer-related fatigue (CRF) was estimated to be 52% (38). In the treatment of patients with
cancer, chemotherapy and radiotherapy are the primary factors
leading to CRF. The mechanism of CRF has not been fully elucidated
and may be associated with (28,35,36)
tumor and host inflammatory response, such as increased IL-1, IL-6
and TNF-α; metabolic disorders, such as increased blood sugar and
insulin resistance; HPA axis dysfunction, such as abnormal cortisol
circadian rhythm; neurotransmitter imbalance, such as decreased
activity of serotonin (5-HT) and dopamine; genetic susceptibility;
and psychosocial factors, such as depression, anxiety and coping
styles (19,31,39).
Therefore, the management of CRF requires multidisciplinary
collaboration and comprehensive measures, including symptomatic
treatment, psychological intervention and exercise rehabilitation,
to improve the quality of life of patients.
Mental illness
Depression and anxiety are common psychological
factors of fatigue, with fatigue cited as one of the key items of
the Hospital Depressive Symptom Scale (40,41).
A study of 323 outpatients with MS showed that 83 patients had
scores indicating anxiety (25.7%) and 44 patients had depression
(13.6%), and fatigue was positively associated with depression and
anxiety (40). The mechanism of
depression-associated fatigue is not yet fully understood but may
be associated with decreased activity of neurotransmitters such as
5-HT and norepinephrine (19); HPA
axis dysfunction and decreased melatonin (16); changes in brain function and
structure, such as abnormalities in the frontal lobe and limbic
system (20); increased levels of
inflammatory factors, such as IL-6 and TNF-α (25); and metabolic disorders, such as
mitochondrial dysfunction and decreased energy metabolism (42). Anxiety may induce or aggravate
fatigue through mechanisms such as sympathetic nerve excitement and
HPA axis activation (16). The
association between fatigue, depression and anxiety is complex, and
there may be a bidirectional causal association.
4. Pathophysiological mechanisms of
fatigue
Energy metabolism dysfunction
Energy metabolism disorder is a key mechanism of
fatigue. A total of >90% of the energy for the human body is
derived from ATP produced by mitochondrial oxidative
phosphorylation (42).
Mitochondrial dysfunction and abnormal energy metabolism are common
in patients with chronic fatigue (42). Mitochondrial dysfunction, which
primarily manifests as decreased activity of respiratory chain
complexes, markedly decreases ATP synthesis efficiency, reduces the
mitochondrial DNA copy number, and causes mitochondrial metabolic
homeostasis imbalance as well as dynamic imbalance leading to
abnormal mitochondrial morphology (Fig. 1). Furthermore, contractile load is
a key determinant of fatigue resistance improvement induced by
isometric intermittent training, potentially via p38
MAPK/peroxisome proliferator-activated receptor γ
coactivator-1α-mediated mitochondrial content increase, even in
muscles lacking nutrients (43).
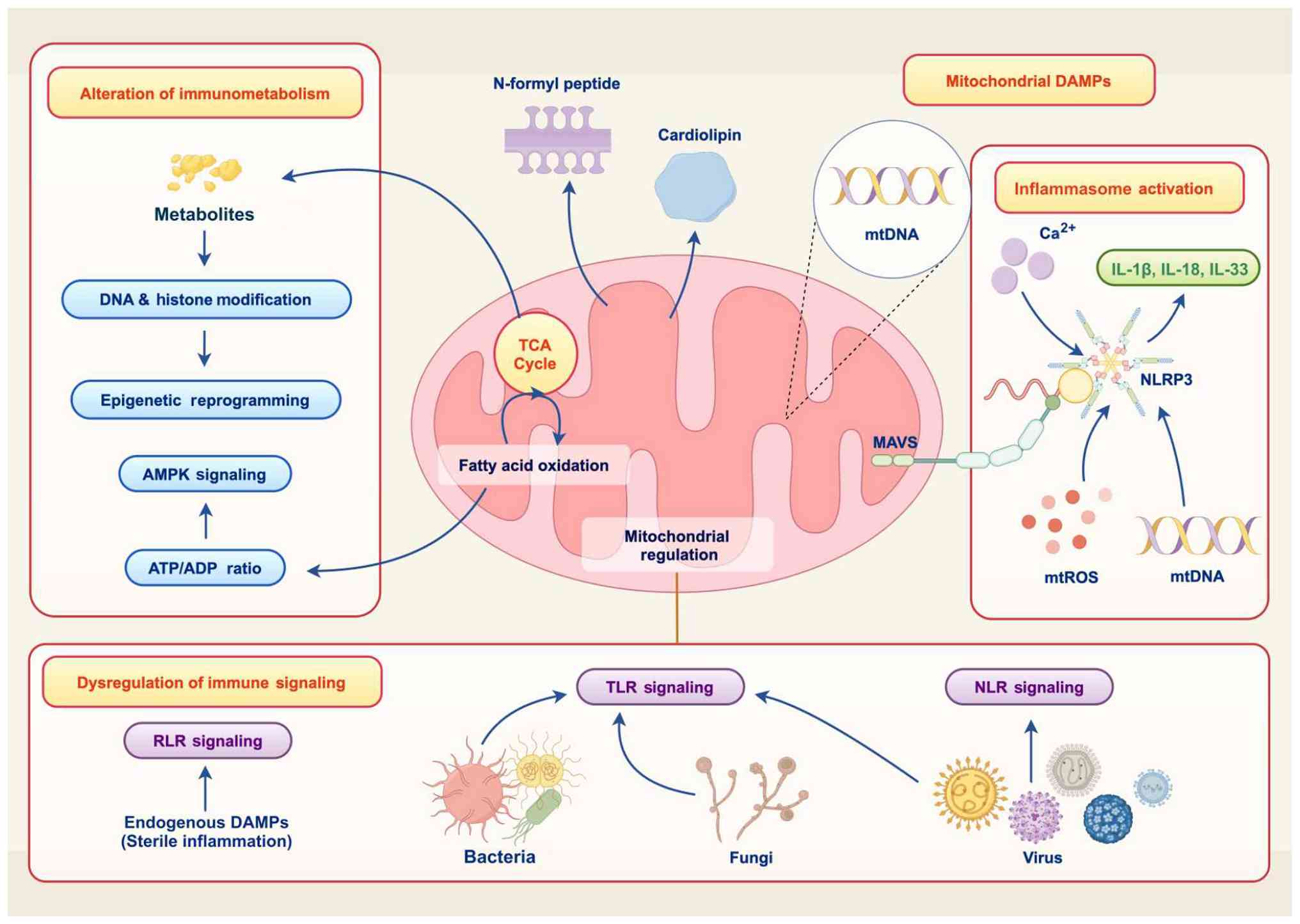 | Figure 1Mitochondria in fatigue.
Mitochondrial dysfunction causes cell damage and energy deficiency
through multiple pathways, leading to fatigue. These pathways
involve the release of mitochondrial DAMPs, including mtDNA,
cardiolipin and N-formyl peptide, which activate inflammasomes such
as NLRP3 and trigger the release of IL-1β, IL-18 and IL-33.
Additionally, mtROS participate in the dysregulation of immune
signaling. Meanwhile, disruptions in metabolic processes such as
the TCA cycle and fatty acid oxidation impact immunometabolism. For
example, metabolites such as acetyl-CoA are involved in DNA and
histone modification, epigenetic reprogramming, and the activation
of AMPK signaling via the ATP/ADP ratio. AMPK, AMP-activated
protein kinase; mtROS, mitochondrial reactive oxygen species; MAVS,
mitochondrial antiviral signaling protein; TCA, tricarboxylic acid;
RLR, RIG-I-like receptor; DAMP, damage-associated molecular
pattern; TLR, toll-like receptor; NLR, NOD-like receptor. |
In addition to mitochondrial function, other
metabolic pathways such as glycolysis and fatty acid β-oxidation
are also affected in chronic fatigue (44). Changes in glucose and lipid
metabolism primarily manifest as inhibition of the glycolysis
pathway, obstruction of fatty acid β-oxidation, decreased
efficiency of ketone body utilization and abnormal lactic acid
metabolism (42,45).
Oxidative stress and cell damage
Oxidative stress serves an important role in the
pathogenesis of fatigue. Proteomic analysis has found that patients
with fatigue typically have elevated levels of oxidative stress
(2), which manifests as increased
production of reactive oxygen species (ROS) (46), weakened function of the antioxidant
system, abnormal mitochondrial membrane potential and increased
cell apoptosis.
Sweetman et al (47) found that oxidative stress markers
are notably elevated in the peripheral blood of patients with
ME/CFS, while the function of the antioxidant defense system is
weakened. Specific manifestations included increased
malondialdehyde levels (reflecting lipid peroxidation), increased
8-hydroxydeoxyguanosine (indicating DNA oxidative damage) and
decreased glutathione peroxidase activity and superoxide dismutase
expression.
Genetic and epigenetic factors
T cells in patients with ME/CFS are epigenetically
predisposed to terminal exhaustion. The CD8+ T cells
from patients with ME/CFS exhibit specific epigenetic modification
patterns that make T cells more prone to exhaustion. Researchers
have used assay for transposase-accessible chromatin with
sequencing technology to compare chromatin accessibility between
memory T cells and naive T cells, identifying 67,189 chromatin
accessible regions in the same cohort of ME/CFS patients and
healthy controls (the same population enrolled for T cell
epigenetic and functional analyses). The authors observed
upregulation of key transcription factors associated with T cell
exhaustion in the CD8+ T cell effector memory subset,
and fatigue markers, including IL-6, TNF-α and CRP, were markedly
upregulated following exercise challenges (48).
Previous research analyzed peripheral blood
mononuclear cell (PBMC) composition changes through single-cell RNA
sequencing, demonstrating increased total T cell frequency in
patients with ME/CFS, with notable decreases in natural killer (NK)
cells, monocytes, conventional dendritic cells and plasmacytoid
dendritic cells (4). The
aforementioned study identified excessive communication initiated
by monocytes and transmitted to other immune cell components via
the estrogen related receptor α/amyloid β precursor protein/CD74
pathway, which serves as a potential biomarker (6).
Additionally, a multi-omics study has shown that
genetic variants in genes such as G protein-coupled receptor 180,
NOTCH3, supervillin, hydroxysteroid 17-β dehydrogenase 11 and
plexin A1 are associated with various fatigue dimensions, including
physical fatigue, cognitive fatigue and emotional fatigue. The
correlation coefficients ranged from -0.539 to 0.517 (P<0.05),
providing preliminary insights into potential involvement of lipid
metabolism changes, catecholamine biosynthesis disruption,
microbial imbalance and specific genetic variants in fatigue among
patients with non-communicable disease (5).
Neuroendocrine-immune network
The brain is the key organ for regulating fatigue
(18), and brain imaging studies
have found that the activity and connectivity patterns of brain
regions change when patients are fatigued (18,49-51).
The severity of fatigue is positively associated with functional
connectivity between the globus pallidus and occipital cortex, and
functional connectivity within the cortico-cerebellar network is
closely related to fatigue perception, suggesting that the basal
ganglia-occipital-cerebellar circuit serves an important role in
the neural mechanisms of fatigue (18). A previous two-sample Mendelian
randomization analysis confirmed that increased volume in the right
lateral orbitofrontal, left caudomedial frontal and right caudal
middle and orbitomedial frontal cortices is associated with
decreased fatigue susceptibility (10). A previous neuroimaging study has
shown similar findings in the prefrontal cortex and basal ganglia
regions involved in fatigue regulation in patients with ME/CFS and
MS (20).
Neurotransmitters are the basis of the activity of
the nervous system. Fatigue is associated with metabolic disorders
of neurotransmitters such as monoamines and amino acids (19). Positron emission tomography (PET)
shows decreased 5-HT transporter binding, suggesting decreased 5-HT
neuron function. Glutamate and γ-aminobutyric acid (GABA) are
notable excitatory and inhibitory neurotransmitters in the central
nervous system, respectively; a spectral study found that the GABA
levels in the brain of patients with CFS are decreased, whereas the
glutamate levels are increased, and the ratio of the two is
decreased, indicating an imbalance in neurotransmitters (20).
Fatigue involves multiple neuroendocrine axes, and
its abnormalities may originate from central nervous system
dysfunction, which affects peripheral organs. The HPA axis is a key
neuroendocrine system regulating the stress response of the body,
and chronic stress can induce dysfunction of the HPA axis, leading
to fatigue. Lee et al (16)
found through a meta-analysis and animal experiments that knocking
out glucocorticoid receptors in the mouse brain causes fatigue-like
behavior. Low cortisol levels may be the result of long-term stress
leading to HPA axis hypofunction, which may be associated with
immune dysfunction and inflammatory response.
Chronic fatigue is associated immune dysfunction.
Viral infection is considered a potential etiological factor of
ME/CFS, accompanied by immune disorders (21). CFS is often secondary to viral
infection, suggesting infection-induced immune dysregulation may be
involved in the pathogenesis of CFS (52). A systematic review showed that
patients with CFS have abnormal PBMC function, which typically
manifests as decreased NK cell killing activity and imbalance of T
cell subsets. In addition, the levels of inflammatory factors such
as IL-4, IL-5, IL-7, IL-12p70 and TNF-α are elevated in patients
with CFS (25,26), indicating a chronic low-grade
inflammatory state.
Immune inflammatory responses induce fatigue through
a variety of pathways (Fig. 2).
Inflammatory factors directly act on the central nervous system,
causing symptoms such as drowsiness, loss of appetite and social
withdrawal (21). Inflammatory
factors affect the metabolism of neurotransmitters, decreasing the
synthesis of 5-HT and dopamine (19,20).
Continuous inflammatory response consumes energy and aggravates
metabolic disorders (21,25,26).
Inflammation induces mitochondrial dysfunction, and increased ROS
causes oxidative stress (2,47).
Immune complex deposition and autoantibody production are involved
in the occurrence of muscle weakness (16,21).
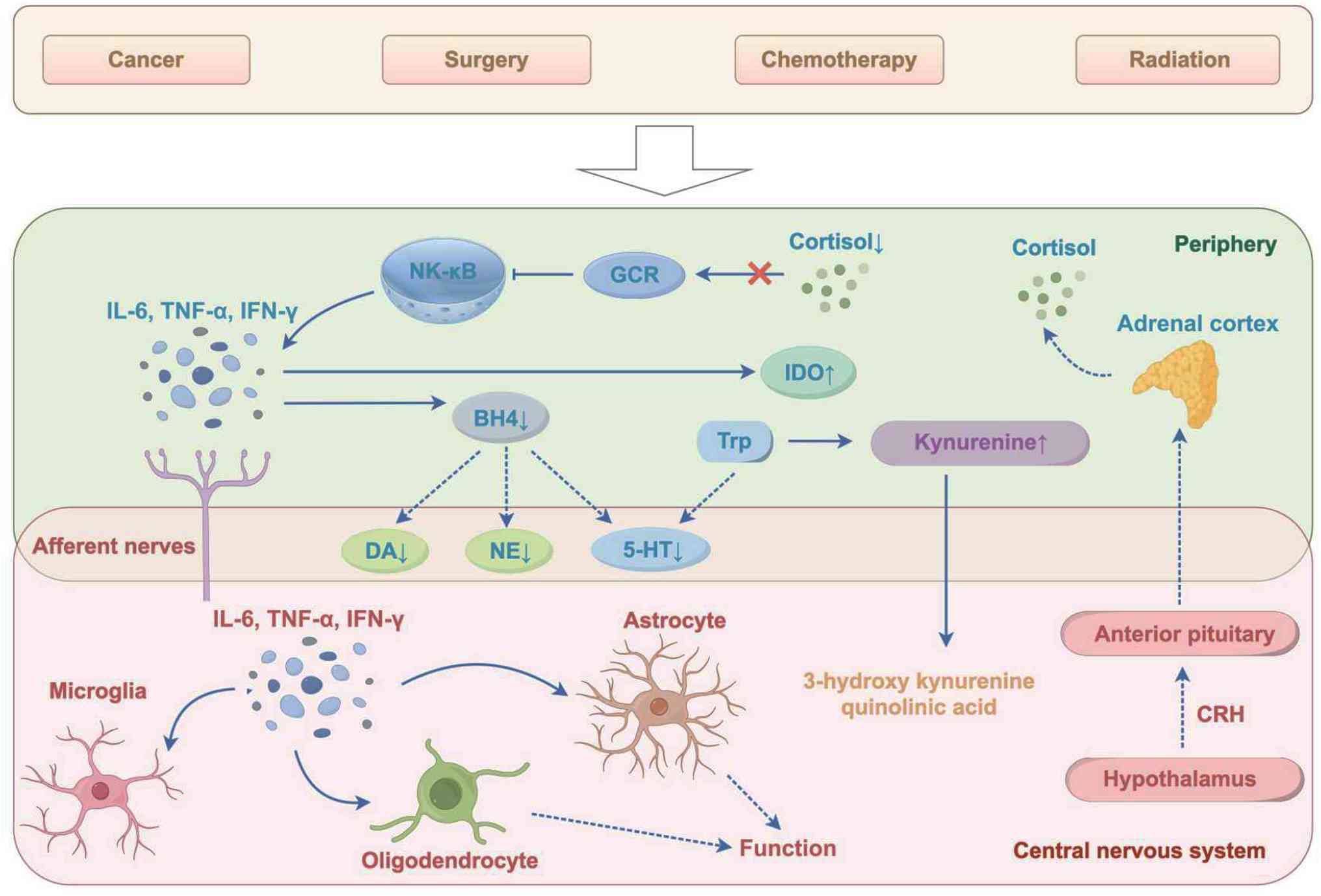 | Figure 2Role of abnormal immune inflammation
in fatigue. Cancer, surgery, chemotherapy or radiation activate
leukocytes, leading to increased expression of the transcription
factor NF-κB, which in turn promotes the release of inflammatory
factors such as IL-6, TNF-α and IFN-γ. These inflammatory factors
induce fatigue manifestations through multiple pathways: They
upregulate IDO and downregulate BH4. IDO promotes the conversion of
Trp to kynurenine, the metabolites (3-hydroxy kynurenine and
quinolinic acid) of which act on the central nervous system. BH4
deficiency leads to reduced levels of DA, NE and 5-HT. Meanwhile,
GCR signaling is impaired due to decreased cortisol, further
amplifying inflammatory responses. In the central nervous system,
microglia and astrocytes are activated by inflammatory factors,
affecting oligodendrocyte function, while the
hypothalamic-pituitary-adrenal axis (involving CRH, the anterior
pituitary and adrenal cortex) is dysregulated, collectively
contributing to fatigue via abnormal immune inflammation and
neurochemical imbalance. GCR, glucocorticoid receptor; BH4,
tetrahydrobiopterin; GABA, glutamate and γ-aminobutyric acid; DA,
dopamine; NE, norepinephrine; 5-HT, serotonin; CRH,
corticotropin-releasing hormone; IDO, indoleamine 2,3-dioxygenase;
Trp, tryptophan. |
Intestinal flora
The intestinal flora is the largest and most complex
microbial system in the human body and serves a key role in
maintaining health. Intestinal flora imbalance is involved in the
occurrence of chronic fatigue (31). Patients with ME/CFS typically
exhibit gastrointestinal symptoms, primarily including intestinal
inflammation and changes and disorders in the intestinal microbiota
(53). Abundance of Firmicutes in
the feces of patients with CFS is markedly reduced, whereas the
abundance of Bacteroidetes is increased, and the diversity
and stability of the bacterial community is decreased (53,54).
A metabolomic analysis has suggested that patients with CFS have
decreased intestinal bacteria production of tyrosine and tryptophan
metabolism, and decreased levels of short-chain fatty acids (SCFAs)
such as butyrate (54).
The intestinal flora affects the occurrence of
fatigue through pathways such as the gut-brain axis (Fig. 3). Intestinal flora imbalance leads
to a decrease in the 5-HT precursor tryptophan. Dysbiosis induces
the production of pro-inflammatory factors and aggravates chronic
inflammation. Inflammatory factors and oxidative stress damage the
intestinal mucosal barrier, leading to reduced expression and
impaired function of tight junction proteins, as well as increased
intestinal permeability. Decreased levels of SCFAs affect the
energy supply of host cells.
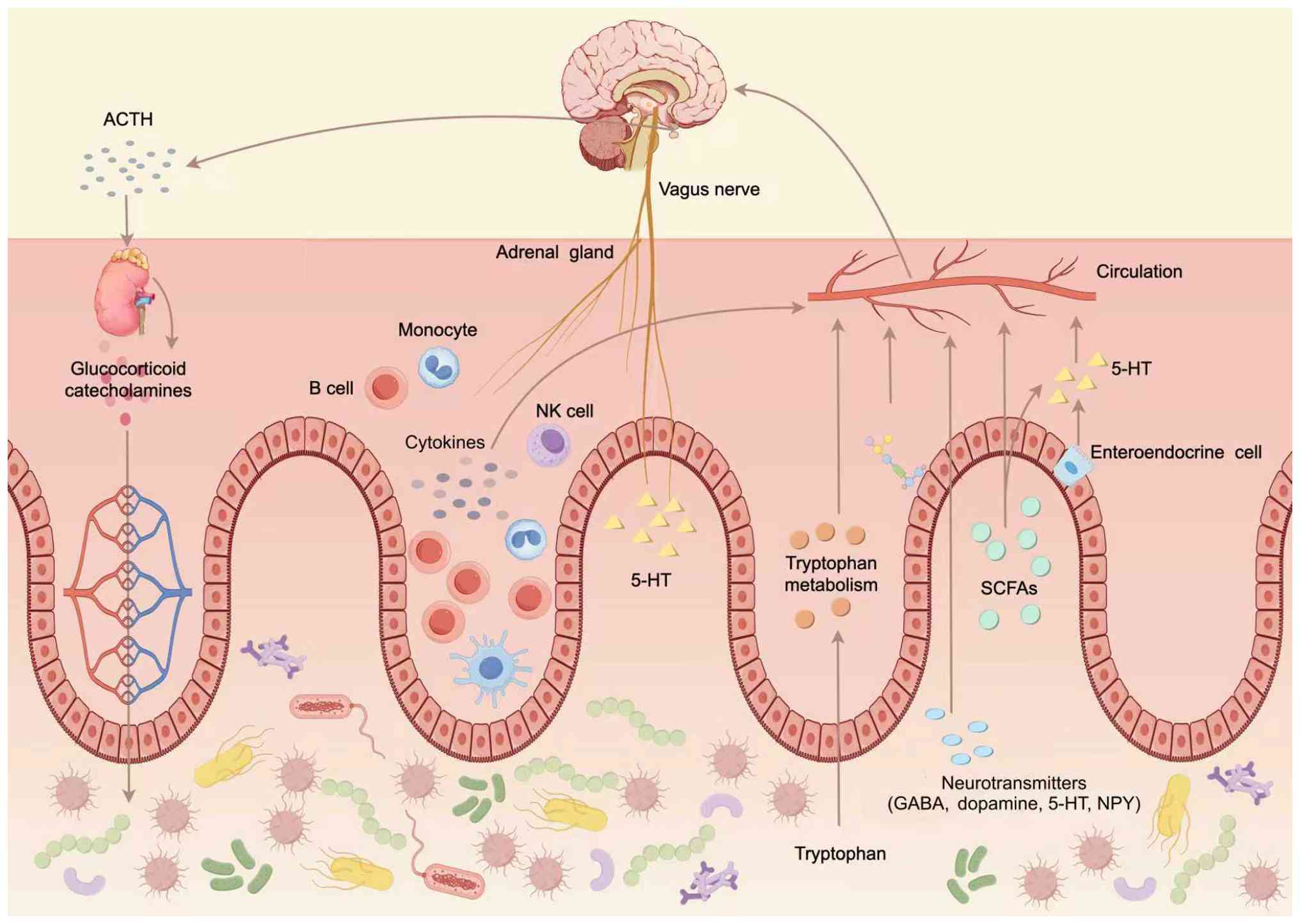 | Figure 3Intestinal flora is involved in the
mechanism of fatigue. Dysbiosis causes adverse consequences by
affecting host metabolism and immunity. Specifically, gut microbes
influence the production of SCFAs and neurotransmitters (GABA,
dopamine, serotonin and NPY), as well as tryptophan metabolism.
These molecules enter the circulation and act on the brain via the
vagus nerve or systemic transport. Meanwhile, the intestinal flora
modulates immune cells (monocytes, B cells and NK cells) and
cytokines, which interact with the hypothalamic-pituitary-adrenal
axis (involving ACTH and glucocorticoid/catecholamine release from
the adrenal gland). Additionally, enteroendocrine cells secrete
5-HT, which participates in this gut-brain communication network,
collectively contributing to fatigue when the intestinal flora is
imbalanced. ACTH, adrenocorticotropic hormone; NK, natural killer;
5-HT, serotonin; SCFA, short chain fatty acid; NPY, neuropeptide
Y. |
5. Clinical manifestations and assessment of
fatigue
Clinical manifestations and subtype
analysis
The clinical manifestations of fatigue are diverse
and complex, involving multiple systems. A large-scale prospective
study found that the primary manifestations of fatigue include
(55) persistent fatigue, which
can manifest as physical weakness or mental exhaustion, ranging
from mild discomfort to complete loss of ability to move; cognitive
dysfunction, primarily manifesting as lack of attention, memory
loss and slow reaction (56);
sleep disorders, mainly manifesting as difficulty falling asleep,
poor sleep quality, frequent dreams and fatigue after waking
(57); autonomic nervous system
dysfunction, such as palpitations, blood pressure regulation
disorder (58), sweating, dry
mouth, constipation or diarrhea; musculoskeletal symptoms, such as
muscle soreness and weakness and joint pain; discomfort in the
throat and neck, swollen and tender lymph nodes in the neck or
armpits (59); endocrine and
metabolic abnormality, fear of cold and heat and menstrual disorder
and mental and psychological problems, such as depression, anxiety
and low mood.
The clinical manifestations of patients with fatigue
vary and may be related to factors such as the cause, course of
disease and comorbidities of fatigue. Vaes et al (56) conducted a large-scale
cross-sectional study and cluster analysis of patients with ME/CFS
based on symptom manifestations. The aforementioned study
identified five symptom clusters, as follows: i) Mild symptoms and
less limitation in daily function; ii) moderate fatigue and
cognitive dysfunction, and limited exercise tolerance; iii) severe
symptoms, notable autonomic nervous system symptoms and immune
dysfunction; iv) cognitive dysfunction and marked brain fog and v)
aggravated symptoms following exercise and notably reduced exercise
tolerance.
The study by Vaes et al (56) emphasizes the heterogeneous nature
of CFS, and that different subtypes may reflect differences in the
mechanisms of fatigue development, which may guide the formulation
of individualized treatment plans, accuracy of prognostic
assessment and clinical stratified management (Tables III and IV).
 | Table IIIClinical comparison of fatigue
subtypes. |
Table III
Clinical comparison of fatigue
subtypes.
| Characteristic | ME/CFS | MS-associated |
Cancer-associated |
Depression-associated |
|---|
| Primary
mechanism |
Immune/metabolic |
Neuroinflammation |
Cytokine-mediated | Neurotransmitter
dysfunction |
| Onset pattern | Post-infectious
acute |
Progressive/recurrent |
Treatment-associated |
Episodic/chronic |
| PEM response | Severe (>24 h
delay) | Variable | Mild | None |
| Exercise
tolerance | Severely
impaired | Moderately
impaired | Temporarily
decreased | Variable |
| Cognitive
features | Brain fog | Decreased
processing speed | Attention
deficit | Executive function
impairment |
| Biomarker
profile | Decreased NK cells;
increased cytokines | MRI lesions;
increased NFL | Increased IL-6 and
CRP | Decreased 5-HT and
cortisol |
 | Table IVDifferential diagnosis of fatigue
syndrome. |
Table IV
Differential diagnosis of fatigue
syndrome.
| A,
Neurological |
|---|
| Medical
condition | Differentiating
features | Diagnostic
tests | Warning signs | (Refs.) |
|---|
| Multiple
sclerosis | Progressive, focal
deficit | MRI, cerebrospinal
fluid oligoclonal bands | Neurological
symptoms | (13,16,17,56,144,147) |
| Parkinson's
disease | Motor symptoms;
bradykinesia | Clinical
evaluation | Tremor,
rigidity | (1,15-17,19,20,50,51,56,86,111,121) |
| B, Endocrine |
| Medical
condition | Differentiating
features | Diagnostic
tests | Warning signs | (Refs.) |
| Hypothyroidism | Cold intolerance,
weight gain | TSH, T4, T3 | Goiter, hair
loss | (21-24,52) |
| Adrenal
insufficiency | Hypotension,
electrolyte abnormalities | Cortisol, ACTH
stimulation test |
Hyperpigmentation | (5,16,23,24,46) |
| C, Autoimmune |
| Medical
condition | Differentiating
features | Diagnostic
tests | Warning signs | (Refs.) |
| Systemic lupus
erythematosus | Arthritis, rash,
organ involvement | ANA,
anti-dsDNA | Malar rash,
nephritis | (1,16,21,25,27-29,52,91) |
| Rheumatoid
arthritis | Joint inflammation,
morning stiffness | RF, anti-CCP | Synovitis,
erosions | (2,21,27-29,46,52,59,90) |
| D, Infectious |
| Medical
condition | Differentiating
features | Diagnostic
tests | Warning signs | (Refs.) |
| Chronic viral
hepatitis | Abnormal liver
function | Serology, liver
enzymes | Jaundice,
hepatomegaly | (21,30,31,33-35,43,52,75) |
| Post-viral
syndrome | Recent infection
history | Virial titer,
inflammatory markers | Acute onset | (21,30,31,33-35,43,52,75) |
| E, Neoplastic |
| Medical
condition | Differentiating
features | Diagnostic
tests | Warning signs | (Refs.) |
| Active
malignancy | Weight loss,
organ-specific symptoms | Imaging, tumor
markers | Systemic
symptoms | (2,6,19,21,31,38,39,47,52,148) |
|
Treatment-associated | History of
chemotherapy/radiotherapy | - | - | - |
| F, Psychiatric |
| Medical
condition | Differentiating
features | Diagnostic
tests | Warning signs | (Refs.) |
| Major depressive
disorder | Mood symptoms,
anhedonia | Clinical
assessment | Suicidal
ideation | (16,19,20,25,42,71,72,75,85-88,107,109,110) |
| Anxiety
disorder | Worry, panic,
avoidance | Clinical
assessment | Panic attacks | (16,19,20,25,42,71,72,75,85-88,107,109,110) |
Table III
clinically differentiates four fatigue subtypes (ME/CFS,
MS-related, cancer-related and depression-related) by primary
mechanism, onset pattern, post-exertional malaise, exercise
tolerance, cognitive features and biomarkers, enabling precise
differential diagnosis and tailored management.
Table IV outlines
the differential diagnosis of fatigue syndrome across disease
categories such as neurological, endocrine, autoimmune, infectious,
neoplastic and psychiatric. Table
IV details specific diseases, key differentiating features,
diagnostic tests, warning signs and references to guide accurate
identification.
Assessment of fatigue
Fatigue is subjective and multidimensional, and
fatigue assessment has undergone a development process from a
single subjective score to a multidimensional integrated
assessment. Single indicators do not fully reflect its severity.
Modern fatigue assessment emphasizes combining subjective and
objective indicators, and static and dynamic assessments, forming a
systematic assessment framework.
Subjective assessment scale
The subjective assessment scale is the most commonly
used fatigue assessment tool in clinical practice and obtains
information on multiple dimensions of fatigue through patient
self-assessment. Commonly used scales include: i) Fatigue Severity
Scale, which consists of nine items to assess the impact of fatigue
on daily activity. Each item is scored from 1-7 points. The higher
the score, the more severe the fatigue. This scale is simple and
easy to use and has been validated for the assessment of fatigue
caused by numerous diseases (60);
ii) the multidimensional fatigue inventory, which consists of 20
items and assesses fatigue from five dimensions, namely general,
physical and mental fatigue and decreased activity and motivation.
This scale has good reliability and validity, and is widely used in
chronic fatigue research (61,62);
the fatigue impact scale (FIS), which consists of 40 items and
assesses the impact of fatigue on cognitive, physical and social
function. This scale has high sensitivity in assessing the
functional status and quality of life of patients with fatigue
(63); and iv) Fatigue Assessment
Inventory, which consists of 29 items and assesses the impact of
fatigue on daily and social activity, including the severity of
fatigue, situational specificity and consequences of fatigue
(64).
These scales have been rigorously validated for
reliability and validity, and are suitable for fatigue assessment
in different clinical scenarios (62). In addition, the VAS and Chalder
fatigue scale (65,66) are used in clinical practice and
research. Choice of assessment basis should be individualized
according to clinical purposes and patient characteristics
(67,68).
Objective evaluation indicators.
Exercise physiology indicators
Exercise physiology indicators objectively reflect
the energy metabolism state of the body and cardiopulmonary
function. Maximum oxygen uptake (VO2 max) is the gold
standard for evaluating aerobic exercise capacity, but is markedly
lower in patients with ME/CFS than in healthy controls. Nelson
et al (69) found that
patients with ME/CFS show notable abnormality in exercise capacity
assessment through 2-day continuous cardiopulmonary exercise
testing. The specific manifestations include marked decrease in
VO2 max, shortened exercise tolerance time and an
earlier time to reach the anaerobic threshold during exercise. This
serves as an important reference indicator for diagnosis. In the
assessment of autonomic nervous function, heart rate variability
(HRV) is a key objective indicator. Escorihuela et al
(70) found that the HRV
indicators of patients with ME/CFS are markedly decreased, among
which, the root mean square of successive differences parameter
value, reflecting parasympathetic nerve activity, is associated
with the severity of fatigue symptoms.
Cognitive function tests. Fatigue is
associated with cognitive dysfunction. Commonly used tests include
the Stroop color-word, paced auditory serial addition, VAS and
trail making test cue production test (71). Computerized cognitive testing
systems such as the Cambridge Neuropsychological Test Automated
Battery provide standardized and sensitive measurements of spatial
working memory, rapid visual information processing, paired
associate learning and emotion recognition task of patients with
ME/CFS, thus providing tools for the assessment of cognitive
function in CFS (72).
Imaging examination. Brain imaging technology
plays an important role in revealing the central nervous
system-mediated mechanisms of fatigue. Using advanced neuroimaging
techniques such as PET and diffusion tensor imaging, previous
research has identified specific changes in brain structure and
function in patients with ME/CFS, such as reduced serotonin neuron
function (15,18,20,49).
Functional magnetic resonance imaging shows alterations in the
functional connectivity of brain regions such as the default and
dorsal attention network in patients with CFS, as well as
abnormalities in prefrontal and limbic system activity and
neurovascular coupling in ME/CFS; these changes are associated with
cognitive dysfunction (50,51).
In addition, magnetic resonance spectroscopy quantitatively
analyzes brain tissue metabolites; patients with ME/CFS have
altered N-acetylaspartate levels, and abnormal lactate metabolism
and choline compound levels (2).
Laboratory tests. Although ME/CFS lacks
specific diagnostic biomarkers, previous multi-omics studies
(2,14,21,45,47)
have found that patients with CFS have abnormal PBMC function
(25), increased lactate and
decreased glutathione peroxidase levels (47), suggesting energy metabolism
disorder and increased oxidative stress (2). Serological examinations reveal
manifestations of immune dysfunction, such as increased
immunoglobulin and decreased complement levels and decreased number
and activity of NK cells (21,26,52).
Endocrine function tests reveal HPA axis hypofunction, and changes
in growth hormones and estrogen (16). Genomics has found that patients
with CFS have decreased mitochondrial DNA copy number (42) and ATP synthesis efficiency
(73), suggesting increased
genomic instability. Transcriptomics shows that the expression of
immune and metabolic-associated genes is dysregulated in patients
with CFS (47).
Emerging digital health
technology
Traditional fatigue assessment methods rely on
surveys or physiological signal measurements, which often fail to
provide real-time monitoring and are limited by patient discomfort.
However, multi-omics approaches and AI offers transformative
potential for fatigue research. For example, a novel non-contact
fatigue level diagnosis system that uses multimodal sensor data,
including video, thermal imaging and audio, minimizes physical
discomfort while enabling precise, real-time data collection and
analysis for fatigue evaluation (46,74,75).
Classification of the progressive stages of
fatigue-induced physiological tremor is achieved using a hybrid
bidirectional long short-term memory-gated recurrent unit neural
network (76,77). Cross-sectional area (CSA) is
measured from muscle volume changes during limb movement, and
different feature combinations are fed into the network to evaluate
performance metrics for CSA-informed tremor classification
(78).
BioMapAI, a supervised deep neural network (75), provides systems-level insights into
ME/CFS, refining existing hypotheses and proposing unique
mechanisms. It simultaneously models diverse data types to predict
clinical severity, identify disease- and symptom-specific
biomarkers, and classify ME/CFS in both retained and independent
external cohorts, thereby increasing the effective sample size
(75).
In recent years (79-81),
surface electromyography (sEMG) has emerged as a novel technology
for quantitative assessment of exercise-induced fatigue. A study
proposed (10) a multi-attention
convolutional network (MACNet) for three-tiered evaluation of
muscle fatigue based on sEMG signals. MACNet achieved the highest
average classification accuracy and F1 score. The F1 score refers
to the harmonic mean of precision and recall, balancing the
trade-off between false positives and false negatives, which helps
comprehensively assess the model's classification performance. This
network enhances the extraction of exercise fatigue-related
features from sEMG channels and time-domain characteristics
(10).
6. Management and treatment of fatigue
General treatment principles
The priority is to establish a clear diagnosis of
fatigue, which requires systematic evaluation to exclude secondary
causes of fatigue, including endocrine, autoimmune, infection and
other disease. The importance of multidisciplinary consultation is
emphasized during the diagnostic process to ensure no causes are
missed. An individualized treatment plan is developed based on the
severity of fatigue, specific clinical manifestations and
concurrent symptoms. The choice of treatment regimen should be
based on the latest evidence and adjusted dynamically as the
disease progresses. At the same time, biological, psychological and
social factors should be considered to adopt a multi-pronged
approach, including drug treatment, psychotherapy and lifestyle
intervention. Clinically, it is necessary to explain the natural
course of the disease, treatment plan and prognosis to patients to
enhance treatment compliance. Chronic fatigue requires long-term
follow-up and rehabilitation plans, regular evaluation of treatment
effects and timely adjustment of treatment plans to prevent
recurrence.
Drug treatment
Although there are currently no US Food and Drug
Administration-approved anti-fatigue medications, certain
medications can be used to relieve fatigue-associated symptoms.
Central stimulants. Stimulants relieve
fatigue by increasing the excitability of the central nervous
system. Commonly used drugs include modafinil [high-quality
evidence; Grading of Recommendations Assessment, Development and
Evaluation (GRADE)] (82). In a
previous study of 141 patients with relapsing-remitting MS
accompanied by fatigue, the subjects were randomly assigned to
receive four treatment regimens in a cross-over manner. Each
treatment phase lasted 6 weeks. Modafinil relieved MS-related
fatigue and was well-tolerated (83). Its mechanism may be associated with
enhancing noradrenergic, dopaminergic and histaminergic
neurotransmission, as well as regulating inflammatory responses
(84). Methylphenidate
(low-quality evidence, GRADE), which is primarily used in the
clinical treatment of attention deficit hyperactivity disorder, has
also been used in fatigue management in previous years.
Methylphenidate improves fatigue severity, concentration and memory
in patients with CFS (85). Its
mechanism is primarily associated with dopamine reuptake inhibition
(85). Pemoline (low-quality
evidence, GRADE) is a non-amphetamine central nervous system
stimulant with dopamine-enhancing effects. A previous small-sample
study showed that pemoline has a certain effect on relieving
symptoms related to depression, such as disturbances in
concentration, memory, tension and fatigue in depressed patients,
but there is a lack of evidence from large-sample randomized
controlled studies (86).
Antidepressants. Antidepressants are used to
treat fatigue in patients with comorbid depressive symptoms.
Selective 5-HT reuptake inhibitors (SSRIs; high-quality evidence,
GRADE) are the most widely used antidepressant drugs in clinical
practice, and effectively improve symptoms of depression and
fatigue. For example, fluoxetine, a SSRI commonly used for
psychiatric disorder, is considered to have neuroprotective
effects, thereby decreasing fatigue symptoms in patients with MS
(87). However, certain studies
have shown that SSRIs are not effective for patients with CFS
without depressive symptoms (86,88).
Therefore, antidepressants are primarily suitable for patients with
CFS with notable depressive symptoms.
Immunosuppressants. Patients with CFS
typically have immune dysfunction, suggesting immune regulation may
have potential value in improving CFS symptoms. Fluge et al
(89) conducted a randomized
double-blind trial to explore the efficacy and safety of the B cell
depleting drug rituximab (moderate-quality evidence, GRADE) in the
treatment of CFS. The study included 151 patients with CFS, who
were randomly divided into a rituximab (n=77) and a placebo control
group (n=74). The fatigue severity scores of patients in the
treatment group markedly decreased compared with baseline and were
markedly improved compared with those in the placebo group
(moderate-quality evidence, GRADE), with therapeutic effects
lasting several months. It is hypothesized that rituximab may
relieve CFS symptoms by clearing autoreactive B cells and
decreasing the production of inflammatory factors. A previous small
open-label trial (90) showed that
low-dose naltrexone (low-quality evidence, GRADE) could regulate
immune function and decrease neuroinflammation. Naltrexone relieves
fatigue in patients with CFS, but this effect needs to be confirmed
in large-sample studies (90).
Non-steroidal anti-inflammatory drugs
(NSAIDs). NSAIDs (low-quality evidence, GRADE) primarily exert
their antipyretic, analgesic and anti-inflammatory effects by
inhibiting cyclooxygenase activity and decreasing prostaglandin
synthesis. They can relieve symptoms such as muscle soreness and
joint pain in patients with CFS but have limited efficacy on core
fatigue symptoms (91).
Coenzyme Q10. Coenzyme Q10 (moderate-quality
evidence, GRADE), also known as ubiquinone, serves as an electron
carrier in the mitochondrial electron transport chain, participates
in ATP synthesis, scavenges free radicals and exerts an antioxidant
effect. CFS is related to mitochondrial dysfunction (92,93)
therefore, coenzyme Q10 may alleviate fatigue by improving
mitochondrial function and enhancing the metabolic and antioxidant
capability of the body (94).
Calcium channel blockers. Patients with CFS
typically have symptoms of autonomic dysfunction such as
palpitations and tachycardia. It is hypothesized that increased
sympathetic nerve excitability may be a potential mechanism
(95). Calcium ion blockers
(low-quality evidence, GRADE) such as amlodipine block L-type
calcium channels, inhibit the release of norepinephrine from
sympathetic nerve endings and decrease sympathetic nerve
excitability, thus relieving CFS-related cardiovascular symptoms
(7). To the best of our knowledge,
there are few studies on calcium antagonists for the treatment of
CFS, most of which are small-sample observational studies or case
reports, and there is a lack of evidence-based medicine (96-99).
Vitamins. Methylcobalamin (low-quality
evidence, GRADE) is a cobalt-containing vitamin B12 derivative that
serves as a coenzyme in the synthesis and metabolism of proteins,
nucleic acids, FA and other substances in the body. Previous
studies have found that methylcobalamin may have beneficial effects
on CFS-associated fatigue (100,101) and cognitive dysfunction by
improving cell energy metabolism and enhancing neural repair and
neuroprotection (94).
Non-pharmacological treatment.
Exercise therapy
Exercise therapy (moderate-quality evidence, GRADE)
is a key methods for treating CFS. A Cochrane systematic review
included eight randomized controlled trials involving a total of
1,518 patients with CFS (102).
The results of the narrative review showed that compared with
conventional treatment such as pharmacotherapy and cognitive
behavioral therapy, sport and exercise therapy could improve
fatigue symptoms in patients with CFS, but had no significant
advantages in terms of depression and sleep quality. This suggests
that the benefits of exercise therapy are primarily concentrated on
physical fitness (103), whereas
mood and sleep problems may require coordinated treatment. Another
study conducted network meta-analysis on 56 studies, and showed
that combing aerobic and resistance exercise, yoga and regular
physical activity markedly alleviates CRF (104). The mechanism by which exercise
therapy relieves CFS is not fully understood but may include
improving cardiopulmonary function and muscle endurance, reversing
deconditioning adaptive changes in patients with CFS, improving
autonomic dysfunction and reducing sympathetic nerve tone,
increasing the levels of β-endorphins and monoamine
neurotransmitters in the brain, improving pain and mood, regulating
cytokines and immune function, decreasing chronic inflammatory
responses and enhancing self-efficacy and sense of control
(3,105,106).
Psychotherapy. Psychotherapy (high-quality
evidence, GRADE) is a key component of the comprehensive management
of CFS and can effectively improve fatigue symptoms, emotional
state and coping style. Commonly used psychological treatments
include cognitive behavioral therapy (CBT) and mindfulness-based
stress reduction (MBSR) (107-110).
CBT helps patients develop positive disease management strategies
by amending cognition and coping styles. A meta-analysis of eight
randomized controlled trials involving 1,298 patients with CFS
showed that CBT causes marked improvement in fatigue severity,
physical function and emotional symptoms compared with standard
treatment. The standard treatment in this analysis was defined as
supportive care, including symptom management, psychoeducation and
general lifestyle advice without structured psychological or
behavioral intervention. This beneficial effect was consistent
across key patient characteristics, with no significant
heterogeneity observed (107). A
randomized controlled trial involving 240 patients with CFS found
that internet CBT is as effective as face-to-face CBT in reducing
fatigue and improving daily function and has higher compliance
(108).
MBSR is a meditation-based stress reduction method.
A randomized controlled trial compared patients who underwent MBSR
with untreated patients, and the results showed that patients with
CFS in the MBSR group exhibit marked improvements in fatigue
severity and sleep quality, with therapeutic effects maintained for
3 months after the end of the intervention (109). MBSR may alleviate CFS symptoms by
regulating autonomic nervous function and alleviating the chronic
stress response (110).
Nutriregulation technology. Transcranial
magnetic stimulation (TMS; low-quality evidence, GRADE) is a
non-invasive neuromodulation technology that generates local
currents in the cerebral cortex through electromagnetic induction,
affecting neuronal membrane potential and cortical function
(43). A previous study used a
multimodal approach, combining peripheral neuromuscular
electrophysiological assessment and TMS-electroencephalography
technology (74), and found that,
following fatigue induction, the oscillation energy of the g
frequency band (30-45 Hz) of the motor cortex in the MS group
decreases markedly, while the functional connectivity within and
between the default and the frontoparietal control networks is
weakened, which is negatively associated with the severity of
fatigue (43,74,111).
Cryotherapy. There are a number of
preliminary studies exploring the effects of cryotherapy
(low-quality evidence, GRADE) on symptoms and function in patients
with CFS (112-114).
A small controlled study involving 24 patients with CFS found that
a combined intervention of whole-body cryotherapy (-110˚C; 3 min
each; 3 times/week for 4 weeks) plus static stretching could
markedly improve fatigue severity (FIS), with beneficial effects
sustained for at least 4 weeks post-intervention. No notable
adverse reactions were reported, and the improvements were
associated with enhanced autonomic nervous system function, a key
mechanism underlying symptom relief in patients with CFS (115-117).
Another randomized controlled trial compared conventional care
combined with local cryotherapy (freezing the limbs for 30 sec
each; twice/day for 15 days) with conventional care alone; daily
living ability and quality of life scores of patients in the
combined group were markedly higher than those in the control
group, suggesting cryotherapy can be a beneficial supplement to
conventional therapy (118).
Physiotherapy. Certain physical factors
(low-quality evidence, GRADE) such as low-intensity laser and
static magnetic field may alleviate CFS symptoms. Transcranial
low-level laser therapy (LLLT) uses near-infrared light to perform
photo biomodulation of specific brain regions (119). It improves mitochondrial
respiratory chain function by upregulating the expression of key
enzymes such as nitric oxide synthase and cytochrome c oxidase, and
enhances ATP synthesis, thereby exerting a role in cell protection
and repair (120). This suggests
LLLT may alleviate core symptoms such as fatigue and cognitive
decline by improving mitochondrial dysfunction in patients with CFS
and alleviating neuroinflammation. Extremely low frequency
electromagnetic field therapy uses a constant magnetic field of
specific intensity to produce a similar electromagnetic induction
effect in the human body, regulates the cell membrane potential and
ion channel function and enhances the antioxidant activity of
enzymes in patients, and improves their functional and
psychological status (121). In
addition, transcranial electrical stimulation (tES) technology has
recently been applied for CFS, tES exerts specific effects in
patients with CFS, including alleviating core symptoms such as
persistent fatigue, improving cognitive functions and reducing
associated emotional distress. These effects are considered to be
mediated by its ability to regulate cerebral cortex excitability
and induce plasticity changes in neural circuits involved in
fatigue perception and cognitive processing. Notably, tES has shown
efficacy in diseases such as depression and chronic pain by
applying a weak constant or an alternating current to specific
parts of the scalp (122).
Traditional Chinese medicine. Traditional
Chinese medicine (low-quality evidence, GRADE) of CFS has
advantages of overall regulation and multi-target effects (123-125).
In terms of traditional Chinese medicine treatment, modern
pharmacological studies have shown that tonic herbs can improve
fatigue symptoms through various mechanisms. For example, herbs
such as ginseng, astragalus and codonopsis can enhance immune
function and improve energy metabolism (126). In addition, acupuncture notably
improves fatigue severity, depression and anxiety levels and
quality of life, while having few adverse reactions and being safe
(127). The mechanism by which
acupuncture exerts its efficacy may be associated with regulating
the neuro-endocrine-immune network, improving autonomic function
and decreasing oxidative stress and inflammatory responses
(128).
Self-management. Self-management
(low-quality evidence, GRADE) is a key part of the comprehensive
treatment of CFS. Prognosis is notably improved by imparting
disease knowledge, coping skills and behavioral strategies to
patients and improving their awareness and ability to actively
participate in disease management. A comprehensive review of
fatigue self-management education in individuals with
disease-related fatigue included 26 randomized controlled trials
involving eight disease groups (129). At follow-up, 46% of the included
studies reported statistically significant improvements in fatigue,
with positive effects observed particularly in patients with cancer
and multiple sclerosis. However, the overall evidence for the
effectiveness of fatigue self-management education on fatigue and
quality of life remains limited and inconsistent (129).
Weight management. Healthy weight loss (very
low-quality evidence, GRADE) is a common dietary management goal
for patients with CFS. Previous studies have found that obesity
aggravates the symptoms of CFS, while moderate weight loss can help
improve fatigue, sleep and mood (130-133).
However, due to limited physical activity in patients with CFS,
weight loss should primarily focus on dietary adjustments,
supplemented by exercise within tolerable limits, and should be
progressed gradually.
7. Future prospects and research
priorities
Despite notable progress in fatigue research,
numerous challenges and practical limitations remain. One of the
primary challenges in CFS clinical research is the lack of
standardized assessment tools. Existing diagnostic tools primarily
rely on self-reported multidimensional fatigue scales and
contact-based sensor measurements (such as electromyography or
respiratory sensors). These methods are cumbersome, time-consuming
and impractical, lacking reliable biomarker support. Additionally,
due to the complexity of symptoms, self-reported fatigue is not
accurate, and psychological, physical and environmental factors
affect diagnostic accuracy and treatment timeliness.
Existing clinical studies generally exhibit
limitations, including small sample size, insufficient follow-up
duration, lack of data on risk factors for fatigue and fatigue
severity, and the use of different fatigue assessment tools without
standardization, which make comparative studies and result
interpretation difficult. These factors compromise the reliability,
reproducibility and generalizability of research findings. There
are no established standard treatment methods for chronic severe
fatigue associated with specific diseases, and the development of
individualized treatment plans lacks sufficient theoretical basis
and practical guidance.
The development of polygenic risk score (PRS)
models is an important direction in precision medicine for CFS. By
integrating gene variants associated with CFS, PRS can predict
individual susceptibility to fatigue and provide a basis for early
intervention (134-136).
Future research should focus on developing more accurate PRS models
and verifying their applicability in different populations.
Epigenetic profiling provides a method
understanding the environment-genetic interactions in CFS. Patients
with CFS typically exhibit abnormal DNA methylation patterns, which
may be associated with viral infection, psychosocial stress and
other factors (5,6,48).
Future research should explore the dynamic changes of these
epigenetic markers and their association with symptom severity.
The regulatory roles of long non-coding RNA and
microRNA in CFS have gained increasing attention (52,137-138). Single-cell RNA sequencing
technology has revealed cellular heterogeneity in CFS (139). By analyzing expression
differences of different cell types in patients with CFS, specific
cell subpopulations and their functional disorder can be
identified. Spatial transcriptomics demonstrates tissue-specific
gene expression patterns, particularly changes in the brain and
immune tissue. Although multi-omics technologies have made progress
in CFS research, data standardization and computational
infrastructure remain challenges (5,48).
There are differences in data formats and analytical methods across
different omics platforms (such as genomics, transcriptomics and
proteomics), and unified standards need to be established to
facilitate data integration. Additionally, processing large-scale
omics data requires efficient computational platforms and
algorithms to support joint analysis of multi-omics data (75).
The application of AI in CFS diagnosis is
developing rapidly: The BioMapAI deep learning model, developed in
2025, has successfully integrated gut microbiome, plasma metabolome
and immune cell profiling data from 249 participants, and
constructed a microbiome-immune-metabolic interaction network. This
model reveals the association between key molecules such as SCFAs,
γδT cells, IFN-γ and symptom heterogeneity, providing novel
strategies for precision diagnosis and treatment (75,140). Natural language processing
technology has also shown potential in CFS research; by extracting
key information from electronic health records, AI systems identify
clinical features and subtypes of CFS. The application of AI in
optimizing CFS treatment primarily focuses on predictive modeling;
however, by analyzing multi-omics data, symptom characteristics and
treatment responses, AI models can predict the efficacy of specific
treatment plans. Additionally, AI guides drug optimization, such as
predicting patient responses to specific drugs and decreasing the
risk of adverse events.
In terms of identifying therapeutic targets and
drug development, the application of respiratory chain modulators
such as coenzyme Q10 in CFS treatment remains controversial.
Although certain studies have reported improvements in other
symptoms of patients with CFS with coenzyme Q10 supplementation,
notable efficacy in fatigue relief is lacking (141,142). Future research should explore
more effective respiratory chain modulators, such as specific
complex enhancers.
Future research on CFS should focus on multi-omics
integration, AI applications, development of novel therapeutic
targets and improvement of clinical research methodologies.
Multi-omics integration may provide biomarkers and mechanistic
basis for precise diagnosis and treatment of CFS. AI applications
may enhance diagnostic accuracy and treatment efficiency. The
development of novel therapeutic targets will offer more effective
treatment options for patients with CFS, and improvement of
clinical research methodologies may ensure the reliability of
research findings and their clinical translational value.
With the advancement of research, CFS may transform
from an unexplained syndrome into a disease with precise biological
basis and targeted treatment methods. However, this faces numerous
challenges, including data standardization, computational
infrastructure, cost-effectiveness and patient engagement. Future
research needs to overcome these challenges to promote the
development of precise diagnosis and treatment for CFS.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by the National Natural
Science Foundation of China (grant no. 31900853) and the Natural
Science Foundation of Hubei Province (grant no. 2024AFC057).
Availability of data and materials
Not applicable.
Authors' contributions
LL conceived the study. HZ, WY and JL performed the
literature review and wrote the manuscript. WY drew and modified
figures. RC, YC and GW conceived the study and provided key
discussions for the images. RC, YC and GW constructed the figures.
LL and WY revised the manuscript. All authors have read and
approved the final the manuscript. Data authentication is not
applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Lim EJ and Son CG: Review of case
definitions for myalgic encephalomyelitis/chronic fatigue syndrome
(ME/CFS). J Transl Med. 18(289)2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Mueller C, Lin JC, Sheriff S, Maudsley AA
and Younger JW: Evidence of widespread metabolite abnormalities in
Myalgic encephalomyelitis/chronic fatigue syndrome: Assessment with
whole-brain magnetic resonance spectroscopy. Brain Imaging Behav.
14:562–572. 2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Bateman L, Bested AC, Bonilla HF, Chheda
BV, Chu L, Curtin JM, Dempsey TT, Dimmock ME, Dowell TG,
Felsenstein D, et al: Myalgic encephalomyelitis/chronic fatigue
syndrome: Essentials of diagnosis and management. Mayo Clin Proc.
96:2861–2878. 2021.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Nalbandian A, Desai AD and Wan EY:
Post-COVID-19 condition. Annu Rev Med. 74:55–64. 2023.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Kobayashi Y, Fujiwara N, Murakami Y,
Ishida S, Kinguchi S, Haze T, Azushima K, Fujiwara A, Wakui H,
Sakakura M, et al: Visualizing fatigue mechanisms in
non-communicable diseases: An integrative approach with multi-omics
and machine learning. BMC Med Inform Decis Mak.
25(204)2025.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Sun Y, Zhang Z, Qiao Q, Zou Y, Wang L,
Wang T, Lou B, Li G, Xu M, Wang Y, et al: Immunometabolic changes
and potential biomarkers in CFS peripheral immune cells revealed by
single-cell RNA sequencing. J Transl Med. 22(925)2024.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Lizyness K and Dewald O: BRASH Syndrome.
In: StatPearls [Internet]. StatPearls Publishing, Treasure Island,
FL, 2025.
|
|
8
|
Kakhi K, Jagatheesaperumal SK, Khosravi A,
Alizadehsani R and Acharya UR: Fatigue monitoring using wearables
and AI: Trends, challenges, and future opportunities. Comput Biol
Med. 195(110461)2025.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Toljan K, Aboseif A and Amin M: Efficacy
of pharmacologic treatments for fatigue in multiple sclerosis: A
systematic review and meta-analysis. Mult Scler Relat Disord.
96(106352)2025.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Zhang Y, Zhang Z, Yu Q, Jiang Y, Fei C, Wu
F and Li F: Mapping fatigue: Discovering brain regions and genes
linked to fatigue susceptibility. J Transl Med.
23(293)2025.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Gandevia SC: Spinal and supraspinal
factors in human muscle fatigue. Physiol Rev. 81:1725–1789.
2001.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Chaudhuri A and Behan PO: Fatigue in
neurological disorders. Lancet. 363:978–988. 2004.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Manjaly ZM, Harrison NA, Critchley HD, Do
CT, Stefanics G, Wenderoth N, Lutterotti A, Müller A and Stephan
KE: Pathophysiological and cognitive mechanisms of fatigue in
multiple sclerosis. J Neurol Neurosurg Psychiatry. 90:642–651.
2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Rupp TL, Garbarino S, Guglielmi O and
Lanteri P: Concepts of fatigue, sleepiness, and Alertness. In:
Reference Module in Neuroscience and Biobehavioral Psychology.
Elsevier, 2017.
|
|
15
|
Palotai M, Nazeri A, Cavallari M, Healy
BC, Glanz B, Gold SM, Weiner HL, Chitnis T and Guttmann CRG:
History of fatigue in multiple sclerosis is associated with grey
matter atrophy. Sci Rep. 9(14781)2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Lee JS, Jeon YJ, Park SY and Son CG: An
adrenalectomy mouse model reflecting clinical features for chronic
fatigue syndrome. Biomolecules. 10(71)2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Siciliano M, Trojano L, Santangelo G, De
Micco R, Tedeschi G and Tessitore A: Fatigue in Parkinson's
disease: A systematic review and meta-analysis. Mov Disord.
33:1712–1723. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Boissoneault J, Sevel L, Robinson ME and
Staud R: Functional brain connectivity of remembered fatigue or
happiness in healthy adults: Use of arterial spin labeling. J Clin
Exp Neuropsychol. 40:224–233. 2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Tschopp R, König RS, Rejmer P and Paris
DH: Health system support among patients with ME/CFS in
Switzerland. J Taibah Univ Med Sci. 18:876–885. 2023.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Prajjwal P, Kalluru PKR, Marsool MD, Inban
P, Gadam S, Al-Ezzi SMS, Marsool AD, Al-Ibraheem AMT, Al-Tuaama
AZH, Amir O and Arunachalam SP: Association of multiple sclerosis
with chronic fatigue syndrome, restless legs syndrome, and various
sleep disorders, along with the recent updates. Ann Med Surg
(Lond). 85:2821–2832. 2023.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Rasa-Dzelzkaleja S, Krumina A, Capenko S,
Nora-Krukle Z, Gravelsina S, Vilmane A, Ievina L, Shoenfeld Y and
Murovska M: VirA project. The persistent viral infections in the
development and severity of myalgic encephalomyelitis/chronic
fatigue syndrome. J Transl Med. 21(33)2023.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Clarke SA, Abbara A and Dhillo WS: Impact
of COVID-19 on the endocrine system: A mini-review. Endocrinology.
163(bqab203)2022.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Paragliola RM and Corsello SM: Secondary
adrenal insufficiency: From the physiopathology to the possible
role of modified-release hydrocortisone treatment. Minerva
Endocrinol. 43:183–197. 2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Husebye ES, Pearce SH, Krone NP and Kämpe
O: Adrenal insufficiency. Lancet. 397:613–629. 2021.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Mandarano AH, Maya J, Giloteaux L,
Peterson DL, Maynard M, Gottschalk CG and Hanson MR: Myalgic
encephalomyelitis/chronic fatigue syndrome patients exhibit altered
T cell metabolism and cytokine associations. J Clin Invest.
130:1491–1505. 2020.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Chu L, Valencia IJ, Garvert DW and Montoya
JG: Onset patterns and course of myalgic encephalomyelitis/chronic
fatigue syndrome. Front Pediatr. 7(12)2019.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Mertz P, Schlencker A, Schneider M, Gavand
PE, Martin T and Arnaud L: Towards a practical management of
fatigue in systemic lupus erythematosus. Lupus Sci Med.
7(e000441)2020.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Pope JE: Management of fatigue in
rheumatoid arthritis. RMD Open. 6(e001084)2020.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Prasad P, Verma S, Surbhi Ganguly NK,
Chaturvedi V and Mittal SA: Rheumatoid arthritis: Advances in
treatment strategies. Mol Cell Biochem. 478:69–88. 2023.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Polukchi TV, Abuova GN and Slavko YA: The
neuropsychiatric aspect of the chronic viral hepatitis. Prague Med
Rep. 124:94–107. 2023.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Arron HE, Marsh BD, Kell DB, Khan MA,
Jaeger BR and Pretorius E: Myalgic encephalomyelitis/chronic
fatigue syndrome: The biology of a neglected disease. Front
Immunol. 15(1386607)2024.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Nasserie T, Hittle M and Goodman SN:
Assessment of the frequency and variety of persistent symptoms
among patients with COVID-19. JAMA Netw Open.
4(e2111417)2021.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Zheng R, Xie S, Lu X, Sun L, Zhou Y, Zhang
Y and Wang K: A systematic review and meta-analysis of epidemiology
and clinical manifestations of human brucellosis in China. Biomed
Res Int. 2018:1–10. 2018.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Long B, MacDonald A, Liang SY, Brady WJ,
Koyfman A, Gottlieb M and Chavez S: Malaria: A focused review for
the emergency medicine clinician. Am J Emerg Med. 77:7–16.
2024.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Yang TY, Lin CL, Yao WC, Lio CF, Chiang
WP, Lin K, Kuo CF and Tsai SY: How mycobacterium tuberculosis
infection could lead to the increasing risks of chronic fatigue
syndrome and the potential immunological effects: A
population-based retrospective cohort study. J Transl Med.
20(99)2022.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Thong MSY, van Noorden CJF, Steindorf K
and Arndt V: Correction to: Cancer-related fatigue: Causes and
current treatment options. Curr Treat Options Oncol. 23:450–451.
2022.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Fabi A, Bhargava R, Fatigoni S, Guglielmo
M, Horneber M, Roila F, Weis J, Jordan K and Ripamonti CI: ESMO
Guidelines Committee. Electronic address:
Clinicalguidelines@esmo.org. Cancer-related fatigue: ESMO clinical
practice guidelines for diagnosis and treatment. Ann Oncol.
31:713–723. 2020.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Ma Y, He B, Jiang M, Yang Y, Wang C, Huang
C and Han L: Prevalence and risk factors of cancer-related fatigue:
A systematic review and meta-analysis. Int J Nurs Stud.
111(103707)2020.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Kang YE, Yoon JH, Park N, Ahn YC, Lee EJ
and Son CG: Prevalence of cancer-related fatigue based on severity:
A systematic review and meta-analysis. Sci Rep.
13(12815)2023.PubMed/NCBI View Article : Google Scholar
|
|
40
|
AlSaeed S, Aljouee T, Alkhawajah NM,
Alarieh R, AlGarni H, Aljarallah S, Ayyash M and Abu-Shaheen A:
Fatigue, depression, and anxiety among ambulating multiple
sclerosis patients. Front Immunol. 13(844461)2022.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Zigmond AS and Snaith RP: The hospital
anxiety and depression scale. Acta Psychiatr Scand. 67:361–370.
1983.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Holden S, Maksoud R, Eaton-Fitch N,
Cabanas H, Staines D and Marshall-Gradisnik S: Correction to: A
systematic review of mitochondrial abnormalities in myalgic
encephalomyelitis/chronic fatigue syndrome/systemic exertion
intolerance disease. J Transl Med. 18(407)2020.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Yamauchi N, Ashida Y, Naito A, Tokuda N,
Niibori A, Motohashi N, Aoki Y and Yamada T: Fatigue resistance and
mitochondrial adaptations to isometric interval training in
dystrophin-deficient muscle: Role of contractile load. FASEB J.
39(e70631)2025.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Toogood PL, Clauw DJ, Phadke S and Hoffman
D: Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS):
Where will the drugs come from? Pharmacol Res.
165(105465)2021.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Wehling-Henricks M, Oltmann M, Rinaldi C,
Myung KH and Tidball JG: Loss of positive allosteric interactions
between neuronal nitric oxide synthase and phosphofructokinase
contributes to defects in glycolysis and increased fatigability in
muscular dystrophy. Hum Mol Genet. 18:3439–3451. 2009.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Shankar V, Wilhelmy J, Curtis EJ, Michael
B, Cervantes L, Mallajosyula V, Davis RW, Snyder M, Younis S,
Robinson WH, et al: Oxidative stress is a shared characteristic of
ME/CFS and Long COVID. Proc Natl Acad Sci USA.
122(e2426564122)2025.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Sweetman E, Noble A, Edgar C, Mackay A,
Helliwell A, Vallings R, Ryan M and Tate W: Current research
provides insight into the biological basis and diagnostic potential
for myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS).
Diagnostics (Basel). 9(73)2019.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Iu DS, Maya J, Vu LT, Fogarty EA, McNairn
AJ, Ahmed F, Franconi CJ, Munn PR, Grenier JK, Hanson MR and
Grimson A: Transcriptional reprogramming primes CD8+ T cells toward
exhaustion in Myalgic encephalomyelitis/chronic fatigue syndrome.
Proc Natl Acad Sci USA. 121(e2415119121)2024.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Provenzano D, Washington SD and Baraniuk
JN: A machine learning approach to the differentiation of
functional magnetic resonance imaging data of chronic fatigue
syndrome (CFS) from a sedentary control. Front Comput Neurosci.
14(2)2020.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Maksoud R, du Preez S, Eaton-Fitch N,
Thapaliya K, Barnden L, Cabanas H, Staines D and Marshall-Gradisnik
S: A systematic review of neurological impairments in myalgic
encephalomyelitis/chronic fatigue syndrome using neuroimaging
techniques. PLoS One. 15(e0232475)2020.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Shan ZY, Barnden LR, Kwiatek RA, Bhuta S,
Hermens DF and Lagopoulos J: Neuroimaging characteristics of
myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS): A
systematic review. J Transl Med. 18(335)2020.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Soffritti I, Gravelsina S, D'Accolti M,
Bini F, Mazziga E, Vilmane A, Rasa-Dzelzkaleja S, Nora-Krukle Z,
Krumina A, Murovska M and Caselli E: Circulating miRNAs expression
in myalgic encephalomyelitis/chronic fatigue syndrome. Int J Mol
Sci. 24(10582)2023.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Luo C, Wei X, Song J, Xu X, Huang H, Fan
S, Zhang D, Han L and Lin J: Interactions between gut microbiota
and polyphenols: New insights into the treatment of fatigue.
Molecules. 27(7377)2022.PubMed/NCBI View Article : Google Scholar
|
|
54
|
König RS, Albrich WC, Kahlert CR, Bahr LS,
Löber U, Vernazza P, Scheibenbogen C and Forslund SK: The gut
microbiome in myalgic encephalomyelitis (ME)/chronic fatigue
syndrome (CFS). Front Immunol. 12(628741)2022.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Jason LA, Islam MF, Conroy K, Cotler J,
Torres C, Johnson M and Mabie B: COVID-19 symptoms over time:
comparing long-haulers to ME/CFS. Fatigue. 9:59–68. 2021.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Vaes AW, Van Herck M, Deng Q, Delbressine
JM, Jason LA and Spruit MA: Symptom-based clusters in people with
ME/CFS: An illustration of clinical variety in a cross-sectional
cohort. J Transl Med. 21(112)2023.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Zerón-Rugerio MF, Zaragozá MC, Domingo JC,
Sanmartín-Sentañes R, Alegre-Martin J, Castro-Marrero J and Cambras
T: Sleep and circadian rhythm alterations in myalgic
encephalomyelitis/chronic fatigue syndrome and post-COVID fatigue
syndrome and its association with cardiovascular risk factors: A
prospective cohort study. Chronobiol Int. 41:1104–1115.
2024.PubMed/NCBI View Article : Google Scholar
|
|
58
|
van Campen CL, Rowe PC and Visser FC:
Worsening symptoms is associated with larger cerebral blood flow
abnormalities during tilt-testing in myalgic
encephalomyelitis/chronic fatigue syndrome (ME/CFS). Medicina
(Kaunas). 59(2153)2023.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Montoya JG, Holmes TH, Anderson JN,
Maecker HT, Rosenberg-Hasson Y, Valencia IJ, Chu L, Younger JW,
Tato CM and Davis MM: Cytokine signature associated with disease
severity in chronic fatigue syndrome patients. Proc Natl Acad Sci
USA. 114:E7150–E7158. 2017.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Krupp LB, LaRocca NG, Muir-Nash J and
Steinberg AD: The fatigue severity scale. Arch Neurol.
46:1121–1123. 1989.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Smets EMA, Garssen B, Bonke B and De Haes
JCJM: The multidimensional fatigue inventory (MFI) psychometric
qualities of an instrument to assess fatigue. J Psychosom Res.
39:315–325. 1995.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Bakalidou D, Krommydas G, Abdimioti T,
Theodorou P, Doskas T and Fillopoulos E: The dimensionality of the
multidimensional fatigue inventory (MFI-20) derived from healthy
adults and patient subpopulations: A challenge for clinicians.
Cureus. 14(e26344)2022.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Fisk JD, Pontefract A, Ritvo PG, Archibald
CJ and Murray TJ: The impact of fatigue on patients with multiple
sclerosis. Can J Neurol Sci. 21:9–14. 1994.PubMed/NCBI
|
|
64
|
Schwartz JE, Jandorf L and Krupp LB: The
measurement of fatigue: A new instrument. J Psychosom Res.
37:753–762. 1993.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Świątczak M, Raczak A, Świątczak A,
Młodziński K, Sikorska K, Jaźwińska A, Kaufmann D and
Daniłowicz-Szymanowicz L: Fatigue assessment in patients with
hereditary hemochromatosis: First use of the popular diagnostic
tools. J Clin Med. 13(5544)2024.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Broadbent DE, Cooper PF, FitzGerald P and
Parkes KR: The cognitive failures questionnaire (CFQ) and its
correlates. Br J Clin Psychol. 21:1–16. 1982.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Lee KA, Hicks G and Nino-Murcia G:
Validity and reliability of a scale to assess fatigue. Psychiatry
Res. 36:291–298. 1991.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Hewlett S, Dures E and Almeida C: Measures
of fatigue: Bristol rheumatoid arthritis fatigue multi-dimensional
questionnaire (BRAF MDQ), bristol rheumatoid arthritis fatigue
numerical rating scales (BRAF NRS) for severity, effect, and
coping, chalder fatigue questionnaire (CFQ), checklist individual
strength (CIS20R and CIS8R), fatigue severity scale (FSS),
functional assessment chronic illness therapy (Fatigue) (FACIT-F),
multi-dimensional assessment of fatigue (MAF), multi-dimensional
fatigue inventory (MFI). Arthritis Care Res (Hoboken).
63:S263–S286. 2011.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Nelson MJ, Buckley JD, Thomson RL, Clark
D, Kwiatek R and Davison K: Diagnostic sensitivity of 2-day
cardiopulmonary exercise testing in Myalgic
Encephalomyelitis/Chronic fatigue syndrome. J Transl Med.
17(80)2019.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Escorihuela RM, Capdevila L, Castro JR,
Zaragozà MC, Maurel S, Alegre J and Castro-Marrero J: Reduced heart
rate variability predicts fatigue severity in individuals with
chronic fatigue syndrome/myalgic encephalomyelitis. J Transl Med.
18(4)2020.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Sebaiti MA, Hainselin M, Gounden Y, Sirbu
CA, Sekulic S, Lorusso L, Nacul L and Authier FJ: Systematic review
and meta-analysis of cognitive impairment in myalgic
encephalomyelitis/chronic fatigue syndrome (ME/CFS). Sci Rep.
12(2157)2022.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Sabahi Z, Farhoudi M, Naseri A and Talebi
M: Working memory assessment using cambridge neuropsychological
test automated battery can help in the diagnosis of mild cognitive
impairment: A systematic review and meta-analysis. Dement
Neuropsychol. 16:444–456. 2022.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Missailidis D, Sanislav O, Allan CY,
Annesley SJ and Fisher PR: Cell-based blood biomarkers for myalgic
encephalomyelitis/chronic fatigue syndrome. Int J Mol Sci.
21(1142)2020.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Lee Y, Lee Y, Kim S, Kim S and Yoo S:
contactless fatigue level diagnosis system through multimodal
sensor data. Bioengineering (Basel). 12(116)2025.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Xiong R, Aiken E, Caldwell R, Vernon SD,
Kozhaya L, Gunter C, Bateman L, Unutmaz D and Oh J: AI-driven
multi-omics modeling of myalgic encephalomyelitis/chronic fatigue
syndrome. Nat Med. 31:2991–3001. 2025.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Schuster M and Paliwal KK: Bidirectional
recurrent neural networks. IEEE Transactions on Signal Processing.
45:2673–2681. 1997.
|
|
77
|
Ahmadzadeh E, Kim H, Jeong O, Kim N and
Moon I: A deep bidirectional LSTM-GRU network model for automated
ciphertext classification. IEEE Access. 10:3228–3237. 2022.
|
|
78
|
Palani P, Panigrahi S, Renganathan G,
Kurita Y and Thondiyath A: Empirical classification of
fatigue-induced physiological tremor in robot-assisted manipulation
tasks using BiLSTM-GRU network. Front Rehab Sci.
6(1474203)2025.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Tang X, Qi Y, Zhang J, Liu K, Tian Y and
Gao X: Enhancing EEG and sEMG fusion decoding using a multi-scale
parallel convolutional network with attention mechanism. IEEE Trans
Neural Syst Rehabil Eng. 32:212–222. 2024.PubMed/NCBI View Article : Google Scholar
|
|
80
|
He L, Zhang L, Sun Q and Lin X: A
generative adaptive convolutional neural network with attention
mechanism for driver fatigue detection with class-imbalanced and
insufficient data. Behav Brain Res. 464(114898)2024.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Mu D, Wang J, Li F, Hu W and Chen R:
Multilevel attention mechanism for motion fatigue recognition based
on sEMG and ACC signal fusion. PLoS One.
19(e0310035)2024.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Guyatt GH, Oxman AD, Vist GE, Kunz R,
Falck-Ytter Y, Alonso-Coello P and Schünemann HJ: GRADE Working
Group. GRADE: An emerging consensus on rating quality of evidence
and strength of recommendations. BMJ. 336:924–926. 2008.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Nourbakhsh B, Revirajan N, Morris B,
Cordano C, Creasman J, Manguinao M, Krysko K, Rutatangwa A, Auvray
C, Aljarallah S, et al: Safety and efficacy of amantadine,
modafinil, and methylphenidate for fatigue in multiple sclerosis: A
randomised, placebo-controlled, crossover, double-blind trial.
Lancet Neurol. 20:38–48. 2021.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Wisor J: Modafinil as a Catecholaminergic
agent: Empirical evidence and unanswered questions. Front Neurol.
4(139)2013.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Shellenberg TP, Stoops WW, Lile JA and
Rush CR: An update on the clinical pharmacology of methylphenidate:
Therapeutic efficacy, abuse potential and future considerations.
Expert Rev Clin Pharmacol. 13:825–833. 2020.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Bahji A and Mesbah-Oskui L: Comparative
efficacy and safety of stimulant-type medications for depression: A
systematic review and network meta-analysis. J Affect Disord.
292:416–423. 2021.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Yousefi F, Kamyab P, Fakhraei B, Farjam M,
Rezaei S, Mahmoudi SS, Karimimoghadam Z, Tabrizi R and Jaafari N:
The effects of selective serotonin reuptake inhibitors on
neurological and depressive symptoms in multiple sclerosis: A
systematic review and meta-analysis of randomized controlled
trials. Galen Med J. 12(e3153)2023.PubMed/NCBI View Article : Google Scholar
|
|
88
|
Gil-Sanchez A, Canudes M, Valcheva P,
Nogueras L, González-Mingot C, Hervás JV, Peralta S, Solana M and
Brieva L: Effects of vortioxetine on cognition and fatigue in
patients with multiple sclerosis and depression: A case series
study. CNS Neurol Disord Drug Targets. 23:395–401. 2024.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Fluge Ø, Rekeland IG, Lien K, Thürmer H,
Borchgrevink PC, Schäfer C, Sørland K, Aßmus J, Ktoridou-Valen I,
Herder I, et al: B-lymphocyte depletion in patients with myalgic
encephalomyelitis/chronic fatigue syndrome: A randomized,
double-blind, placebo-controlled trial. Ann Intern Med.
170:585–593. 2019.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Bolton MJ, Chapman BP and Van Marwijk H:
Low-dose naltrexone as a treatment for chronic fatigue syndrome.
BMJ Case Rep. 13(e232502)2020.PubMed/NCBI View Article : Google Scholar
|
|
91
|
Soomro M, Lyons S, Bravo R, McBeth J, Lunt
M, Dixon WG and Jani M: Use of over-the-counter supplements, sleep
aids and analgesic medicines in rheumatology: Results of a
cross-sectional survey. Rheumatol Adv Pract.
8(rkae129)2024.PubMed/NCBI View Article : Google Scholar
|
|
92
|
Bian Z and Wei L: The role of coenzyme Q10
in exercise tolerance and muscle strength. Arch Physiol Biochem.
131:1–20. 2025.PubMed/NCBI View Article : Google Scholar
|
|
93
|
Salekzamani S, Pakkhesal S, Babaei M,
Mirzaaghazadeh E, Mosaddeghi-Heris R, Talebi M, Sanaie S and Naseri
A: Coenzyme Q10 supplementation in multiple sclerosis; A systematic
review. Mult Scler Relat Disord. 93(106212)2025.PubMed/NCBI View Article : Google Scholar
|
|
94
|
Castro-Marrero J, Sáez-Francàs N, Santillo
D and Alegre J: Treatment and management of chronic fatigue
syndrome/myalgic encephalomyelitis: All roads lead to Rome. Br J
Pharmacol. 174:345–369. 2017.PubMed/NCBI View Article : Google Scholar
|
|
95
|
Bozzini S, Albergati A, Capelli E, Lorusso
L, Gazzaruso C, Pelissero G and Falcone C: Cardiovascular
characteristics of chronic fatigue syndrome. Biomed Rep. 8:26–30.
2017.PubMed/NCBI View Article : Google Scholar
|
|
96
|
Scheibenbogen C and Wirth KJ: Key
Pathophysiological role of skeletal muscle disturbance in post
COVID and myalgic encephalomyelitis/chronic fatigue syndrome
(ME/CFS): Accumulated evidence. J Cachexia Sarcopenia Muscle.
16(e13669)2025.PubMed/NCBI View Article : Google Scholar
|
|
97
|
Woods PC, Swank DM and Miller MS: Stretch
activation combats force loss from fatigue in fast-contracting
mouse skeletal muscle fibers. J Gen Physiol.
157(e202413679)2025.PubMed/NCBI View Article : Google Scholar
|
|
98
|
Allen DG, Lee JA and Westerblad H:
Intracellular calcium and tension during fatigue in isolated single
muscle fibres from Xenopus laevis. J Physiol. 415:433–458.
1989.PubMed/NCBI View Article : Google Scholar
|
|
99
|
Zhang N, Jiang P, Fan S, Wang W and Liu F:
Current status and determinants of fatigue in Chinese older adults
receiving maintenance hemodialysis: A multicentre cross-sectional
study. Clin Interv Aging Volume. 20:1847–1860. 2025.PubMed/NCBI View Article : Google Scholar
|
|
100
|
Tardy AL, Bois De Fer B, Cañigueral S,
Kennedy D, Scholey A, Hitier S, Aran A and Pouteau E: Reduced
self-perception of fatigue after intake of panax ginseng root
extract (G115®) formulated with vitamins and minerals-an
open-label study. Int J Environ Res Public Health.
18(6257)2021.PubMed/NCBI View Article : Google Scholar
|
|
101
|
Tardy AL, Pouteau E, Marquez D, Yilmaz C
and Scholey A: Vitamins and minerals for energy, fatigue and
cognition: A narrative review of the biochemical and clinical
evidence. Nutrients. 12(228)2020.PubMed/NCBI View Article : Google Scholar
|
|
102
|
Larun L, Brurberg KG, Odgaard-Jensen J and
Price JR: Exercise therapy for chronic fatigue syndrome. Cochrane
Database Syst Rev. 12(CD003200)2017.PubMed/NCBI View Article : Google Scholar
|
|
103
|
Gerland L and Baumann FT: Sport and
exercise therapy for burnout and fatigue-a narrative review.
Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz.
67:1288–1294. 2024.PubMed/NCBI View Article : Google Scholar : (In German).
|
|
104
|
Dong B, Qi Y, Lin L, Liu T, Wang S, Zhang
Y, Yuan Y, Cheng H, Chen Q, Fang Q, et al: Which exercise
approaches work for relieving cancer-related fatigue? a network
meta-analysis. J Orthop Sports Phys Ther. 53:343–352.
2023.PubMed/NCBI View Article : Google Scholar
|
|
105
|
Corbitt M, Eaton-Fitch N, Staines D,
Cabanas H and Marshall-Gradisnik S: A systematic review of
cytokines in chronic fatigue syndrome/myalgic
encephalomyelitis/systemic exertion intolerance disease
(CFS/ME/SEID). BMC Neurol. 19(207)2019.PubMed/NCBI View Article : Google Scholar
|
|
106
|
Loy BD, O'Connor PJ and Dishman RK: Effect
of acute exercise on fatigue in people with ME/CFS/SEID. Med Sci
Sports Exerc. 48:2003–2012. 2016.PubMed/NCBI View Article : Google Scholar
|
|
107
|
Kuut TA, Buffart LM, Braamse AMJ, Csorba
I, Bleijenberg G, Nieuwkerk P, Moss-Morris R, Müller F and Knoop H:
Does the effect of cognitive behavior therapy for chronic fatigue
syndrome (ME/CFS) vary by patient characteristics? A systematic
review and individual patient data meta-analysis. Psychol Med.
54:447–456. 2024.PubMed/NCBI View Article : Google Scholar
|
|
108
|
Janse A, Worm-Smeitink M, Bleijenberg G,
Donders R and Knoop H: Efficacy of web-based cognitive-behavioural
therapy for chronic fatigue syndrome: Randomised controlled trial.
Br J Psychiatry. 212:112–118. 2018.PubMed/NCBI View Article : Google Scholar
|
|
109
|
Chayadi E, Baes N and Kiropoulos L: The
effects of mindfulness-based interventions on symptoms of
depression, anxiety, and cancer-related fatigue in oncology
patients: A systematic review and meta-analysis. PLoS One.
17(e0269519)2022.PubMed/NCBI View Article : Google Scholar
|
|
110
|
Chen Y, Wang R, Yu J, Zhu L, Lu Y and Deng
X: Effects of MBSR therapy on negative emotions, fatigue, and sleep
quality in ‘post-ICU patients’. Medicine (Baltimore).
101(e28331)2022.PubMed/NCBI View Article : Google Scholar
|
|
111
|
Leodori G, Mancuso M, Maccarrone D,
Tartaglia M, Ianniello A, Certo F, Baione V, Ferrazzano G,
Malimpensa L, Belvisi D, et al: Neural bases of motor fatigue in
multiple sclerosis: A multimodal approach using neuromuscular
assessment and TMS-EEG. Neurobiol Dis. 180(106073)2023.PubMed/NCBI View Article : Google Scholar
|
|
112
|
Hirata K, Shiozaki D, Yamada K, Miyokawa
Y, Yajima Y and Akagi R: Cryotherapy with carbon dioxide hydrate
enhances immediate recovery of muscle function from neuromuscular
fatigue. J Sports Sci. 42:2103–2114. 2024.PubMed/NCBI View Article : Google Scholar
|
|
113
|
Dauksaite G, Eimantas N, Solianik R,
Daniuseviciute-Brazaite L, Malciene L and Brazaitis M: Head-neck
cooling effects on central and peripheral fatigue, motor accuracy,
and blood markers of stress in men with multiple sclerosis and
healthy men: A randomized crossover study. Mult Scler Relat Disord.
90(105840)2024.PubMed/NCBI View Article : Google Scholar
|
|
114
|
Arc-Chagnaud C, Dupuy O, Garcia M, Bosquet
L, Bouzigon R and Pla R: D-Day Consortium. Dugué B: Effects of
repeated cryostimulation exposures on sleep and wellness in healthy
young adults. Cryobiology. 117(104948)2024.PubMed/NCBI View Article : Google Scholar
|
|
115
|
Vitenet M, Tubez F, Marreiro A, Polidori
G, Taiar R, Legrand F and Boyer FC: Corrigendum to ‘Effect of whole
body cryotherapy interventions on health-related quality of life in
fibromyalgia patients: A randomized controlled trial’. Complement
Ther Med 36 (2018) 6-8]. Complement Ther Med. 38:92–93.
2018.PubMed/NCBI View Article : Google Scholar
|
|
116
|
Kujawski S, Słomko J, Godlewska BR,
Cudnoch-Jędrzejewska A, Murovska M, Newton JL, Sokołowski Ł and
Zalewski P: Combination of whole body cryotherapy with static
stretching exercises reduces fatigue and improves functioning of
the autonomic nervous system in chronic fatigue syndrome. J Transl
Med. 20(273)2022.PubMed/NCBI View Article : Google Scholar
|
|
117
|
Kujawski S, Zalewski P, Godlewska BR,
Cudnoch-Jędrzejewska A, Murovska M, Newton JL, Sokołowski Ł and
Słomko J: Effects of whole-body cryotherapy and static stretching
are maintained 4 weeks after treatment in most patients with
chronic fatigue syndrome. Cryobiology. 112(104546)2023.PubMed/NCBI View Article : Google Scholar
|
|
118
|
Rivera J, Tercero MJ, Salas JS, Gimeno JH
and Alejo JS: The effect of cryotherapy on fibromyalgia: A
randomised clinical trial carried out in a cryosauna cabin.
Rheumatol Int. 38:2243–2250. 2018.PubMed/NCBI View Article : Google Scholar
|
|
119
|
Yeh SW, Hong CH, Shih MC, Tam KW, Huang YH
and Kuan YC: Low-level laser therapy for fibromyalgia: A systematic
review and meta-analysis. Pain Physician. 22:241–254.
2019.PubMed/NCBI
|
|
120
|
Couturaud V, Le Fur M, Pelletier M and
Granotier F: Reverse skin aging signs by red light
photobiomodulation. Skin Res Technol. 29(e13391)2023.PubMed/NCBI View Article : Google Scholar
|
|
121
|
Cichoń N, Bijak M, Miller E and Saluk J:
Extremely low frequency electromagnetic field (ELF-EMF) reduces
oxidative stress and improves functional and psychological status
in ischemic stroke patients. Bioelectromagnetics. 38:386–396.
2017.PubMed/NCBI View Article : Google Scholar
|
|
122
|
Tedeschi R: ‘Transcranial direct current
stimulation for chronic foot pain: A comprehensive review.’.
eNeurologicalSci. 35(100498)2024.PubMed/NCBI View Article : Google Scholar
|
|
123
|
Zeng J, Wu Q, Meng XD and Wang J:
Systematic review of Buzhong Yiqi method in alleviating
cancer-related fatigue: A meta-analysis and exploratory network
pharmacology approach. Front Pharmacol. 15(1451773)2024.PubMed/NCBI View Article : Google Scholar
|
|
124
|
Liu T, Sun W, Guo S, Chen T, Zhu M, Yuan
Z, Li B, Lu J, Shao Y, Qu Y, et al: Research progress on
pathogenesis of chronic fatigue syndrome and treatment of
traditional Chinese and Western medicine. Auton Neurosci.
255(103198)2024.PubMed/NCBI View Article : Google Scholar
|
|
125
|
Wang R, Liu Y, Jiang Y, Zhang Y, Zhang Y,
Wang B, Lu H, Su H, Liao W, Liu L, et al: Shenling Baizhu San
alleviates central fatigue through SIRT1-PGC-1α-Mediated
mitochondrial biogenesis. J Ethnopharmacol.
339(119110)2025.PubMed/NCBI View Article : Google Scholar
|
|
126
|
Yang J, Li Y, Chau CI, Shi J, Chen X, Hu H
and Ung COL: Efficacy and safety of traditional Chinese medicine
for cancer-related fatigue: A systematic literature review of
randomized controlled trials. Chin Med. 18(142)2023.PubMed/NCBI View Article : Google Scholar
|
|
127
|
Lu G, Chen F, Guo C and Wu J: Acupuncture
for senile insomnia: A systematic review of acupuncture point. Arch
Gerontol Geriatr. 127(105586)2024.PubMed/NCBI View Article : Google Scholar
|
|
128
|
Zhang Q, Gong J, Dong H, Xu S, Wang W and
Huang G: Acupuncture for chronic fatigue syndrome: A systematic
review and meta-analysis. Acupunct Med. 37:211–222. 2019.PubMed/NCBI View Article : Google Scholar
|
|
129
|
Hersche R, Roser K, Weise A, Michel G and
Barbero M: Fatigue self-management education in persons with
disease-related fatigue: A comprehensive review of the
effectiveness on fatigue and quality of life. Patient Educ Couns.
105:1362–1378. 2022.PubMed/NCBI View Article : Google Scholar
|
|
130
|
Rozich JJ, Holmer A and Singh S: Effect of
lifestyle factors on outcomes in patients with inflammatory bowel
diseases. Am J Gastroenterol. 115:832–840. 2020.PubMed/NCBI View Article : Google Scholar
|
|
131
|
Jafarnezhadgero A, Moradzadeh N, Mirzang
EF, Sajedi H, Dixon S and Akrami M: Influence of a fatiguing
exercise on lower limb electromyographic activities and
co-contraction in overweight females during running. PLoS One.
20(e0322167)2025.PubMed/NCBI View Article : Google Scholar
|
|
132
|
Al-Naamani Z, Gormley K, Noble H, Santin
O, Al Omari O, Al-Noumani H and Madkhali N: Navigating strategies
used to manage fatigue by patients undergoing hemodialysis: A
qualitative exploratory design. BMC Nephrol. 26(226)2025.PubMed/NCBI View Article : Google Scholar
|
|
133
|
Lim S and Chia S: The prevalence of
fatigue and associated health and safety risk factors among taxi
drivers in Singapore. Singapore Med J. 56:92–97. 2015.PubMed/NCBI View Article : Google Scholar
|
|
134
|
Huang K, Lidbury BA, Thomas N, Gooley PR
and Armstrong CW: Machine learning and multi-omics in precision
medicine for ME/CFS. J Transl Med. 23(68)2025.PubMed/NCBI View Article : Google Scholar
|
|
135
|
Kendler KS, Rosmalen JGM, Ohlsson H,
Sundquist J and Sundquist K: A distinctive profile of family
genetic risk scores in a Swedish national sample of cases of
fibromyalgia, irritable bowel syndrome, and chronic fatigue
syndrome compared to rheumatoid arthritis and major depression.
Psychol Med. 53:3879–3886. 2023.PubMed/NCBI View Article : Google Scholar
|
|
136
|
Das S, Taylor K, Kozubek J, Sardell J and
Gardner S: Genetic risk factors for ME/CFS identified using
combinatorial analysis. J Transl Med. 20(598)2022.PubMed/NCBI View Article : Google Scholar
|
|
137
|
Yang CA, Bauer S, Ho YC, Sotzny F, Chang
JG and Scheibenbogen C: The expression signature of very long
non-coding RNA in myalgic encephalomyelitis/chronic fatigue
syndrome. J Transl Med. 16(231)2018.PubMed/NCBI View Article : Google Scholar
|
|
138
|
Yang X, Li F, Ma J, Liu Y, Wang X, Wang R,
Zhang Y, Zhang W, He Q, Song D and Yu J: Study on the relationship
between the miRNA-centered ceRNA regulatory network and fatigue. J
Mol Neurosci. 71:1967–1974. 2021.PubMed/NCBI View Article : Google Scholar
|
|
139
|
Vu LT, Ahmed F, Zhu H, Iu DSH, Fogarty EA,
Kwak Y, Chen W, Franconi CJ, Munn PR, Tate AE, et al: Single-cell
transcriptomics of the immune system in ME/CFS at baseline and
following symptom provocation. Cell Rep Med.
5(101373)2024.PubMed/NCBI View Article : Google Scholar
|
|
140
|
Xiong R, Aiken E, Caldwell R, Vernon SD,
Kozhaya L, Gunter C, Bateman L, Unutmaz D and Oh J: BioMapAI:
Artificial intelligence multi-omics modeling of myalgic
encephalomyelitis/chronic fatigue syndrome. bioRxiv: doi:
10.1101/2024.06.24.600378.
|
|
141
|
Castro-Marrero J, Cordero MD, Segundo MJ,
Sáez-Francàs N, Calvo N, Román-Malo L, Aliste L, Fernández de
Sevilla T and Alegre J: Does oral coenzyme Q 10 Plus
NADH supplementation improve fatigue and biochemical parameters in
chronic fatigue syndrome? Antioxid Redox Signal. 22:679–685.
2015.PubMed/NCBI View Article : Google Scholar
|
|
142
|
Lee YJ, Cho WJ, Kim JK and Lee DC: Effects
of coenzyme Q 10 on arterial stiffness, metabolic
parameters, and fatigue in obese subjects: A double-blind
randomized controlled study. J Med Food. 14:386–390.
2011.PubMed/NCBI View Article : Google Scholar
|
|
143
|
Townsend L, Dyer AH, Jones K, Dunne J,
Mooney A, Gaffney F, O'Connor L, Leavy D, O'Brien K, Dowds J, et
al: Persistent fatigue following SARS-CoV-2 infection is common and
independent of severity of initial infection. PLoS One.
15(e0240784)2020.PubMed/NCBI View Article : Google Scholar
|
|
144
|
Bileviciute-Ljungar I, Norrefalk JR and
Borg K: Pain burden in post-COVID-19 syndrome following mild
COVID-19 infection. J Clin Med. 11(771)2022.PubMed/NCBI View Article : Google Scholar
|
|
145
|
Wong TL and Weitzer DJ: Long COVID and
myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS)-A
systemic review and comparison of clinical presentation and
symptomatology. Medicina (B Aires). 57(418)2021.PubMed/NCBI View Article : Google Scholar
|
|
146
|
Morris G, Maes M, Berk M and Puri BK:
Myalgic encephalomyelitis or chronic fatigue syndrome: How could
the illness develop? Metab Brain Dis. 34:385–415. 2019.PubMed/NCBI View Article : Google Scholar
|
|
147
|
Braley TJ, Ehde DM, Alschuler KN, Little
R, Ng YT, Zhai Y, von Geldern G, Chervin RD, Conroy D, Valentine
TR, et al: Comparative effectiveness of cognitive behavioural
therapy, modafinil, and their combination for treating fatigue in
multiple sclerosis (COMBO-MS): A randomised, statistician-blinded,
parallel-arm trial. Lancet Neurol. 23:1108–1118. 2024.PubMed/NCBI View Article : Google Scholar
|
|
148
|
Zhang G, Yang B, Zan P and Zhang D:
Multilevel assessment of exercise fatigue utilizing multiple
attention and convolution network (MACNet) based on surface
electromyography. IEEE Trans Neural Syst Rehabil Eng. 33:243–254.
2025.PubMed/NCBI View Article : Google Scholar
|

















