Introduction
According to global cancer epidemiological data
(GLOBOCAN 2020 statistics), the incidence and mortality of lung
cancer rank first among all malignant tumors (1). The incidence of multiple pulmonary
nodules ranges 5-20% in studies of different sample sizes and
populations (2,3). With the increasingly common use of
low-dose CT, the detection rate of multiple primary lung cancer
(MPLC) has gradually increased. The number of cases diagnosed with
simultaneous multiple lung lesions is increasing annually, where
the majority of cases are finally confirmed as simultaneous MPLC.
Global data shows that the incidence of MPLC ranges from 0.8 to
8.4%, and is on a continuous upward trend (4). Epidemiological studies in China
report an incidence of MPLC ranging from 0.52 to 2.45% (5), while a cohort study of surgical
patients showed that 12% of patients with lung cancer were
pathologically diagnosed with MPLC (6). The diagnosis of MPLC requires a
combination of histopathology and molecular characteristics.
Clinically, 12-18% of patients with multiple pulmonary nodules are
ultimately confirmed to have synchronous MPLC (6,7).
MPLCs can be easily misdiagnosed as metastatic cancers when more
than two nodules appear in the lungs. The distinction of MPLC from
intrapulmonary metastases (IM) is critical but challenging. The
former is mainly treated by surgery and the prognosis is favorable,
whereas the latter involves systemic chemotherapy and exhibits a
poor prognosis (8). For patients
with synchronous MPLC undergoing bilateral staged surgery, if the
lesions are completely resected, the 5-year survival rate can reach
60-80% (9-11).
Multiple lung cancer types typically present as
anatomical pulmonary nodules on imaging (12,13).
This has persistently been a challenge for pathologists and
thoracic surgeons due to their important implications for treatment
and prognosis as a result of the accurate staging of these nodules.
However, current tumor staging systems mainly rely on histological
and pathological features, which lack definitive criteria for
diagnosing MPLC. This condition can lead to ambiguous cases, where
pulmonary nodules are histologically, pathologically identical or
highly similar (2,14). Next-generation sequencing (NGS) has
been garnering attention as a valuable adjunct to the existing
histopathological diagnostic workup, notably in lung cancer
(15). Molecular typing can aid in
treatment selection. In the present case, a patient with MPLC with
>20 lesions in both lungs is reported, highlighting the critical
role of the combination of imaging, pathological and molecular
features obtained from each tumor lesion in the diagnosis of MPLC.
The data can be shared with individualized treatment programs.
Case report
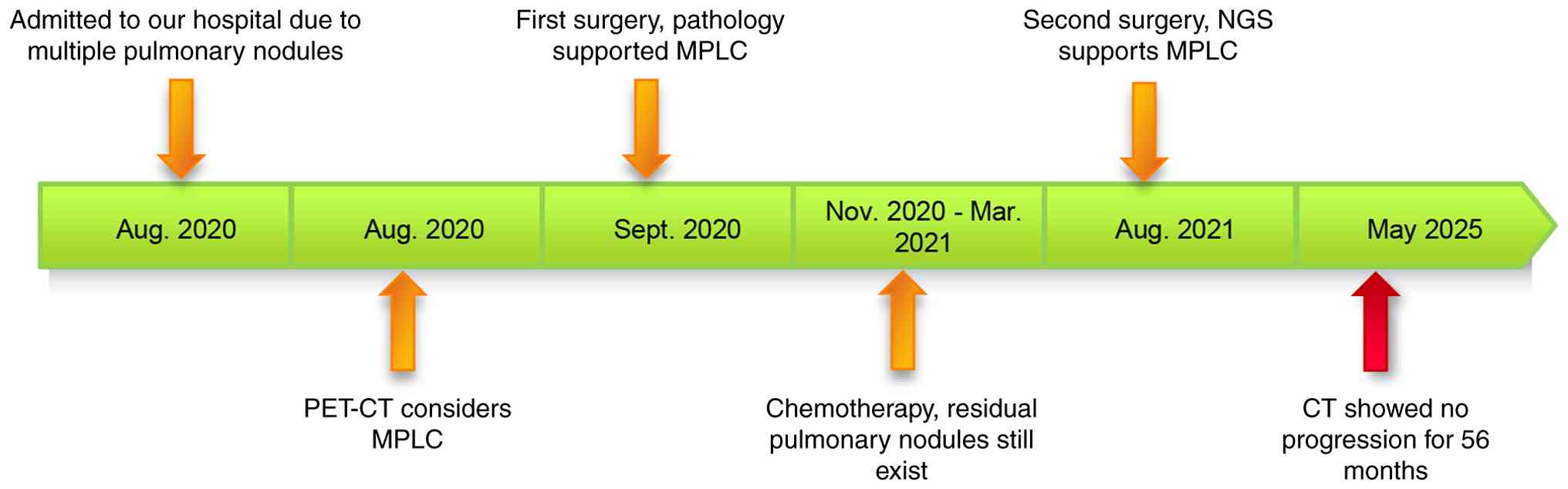
A 45-year-old female patient was admitted to the
Affiliated Hospital of Guangdong Medical University (Zhanjiang,
China) in August 2020 following detection of multiple nodules in
both lungs during a CT scan (Fig.
1). The purpose of the visit was to clarify the pathology of
lung lesions and provide adequate treatment. The patient had no
specific medical symptoms, such as cough, sputum or fever. The
patient did not smoke and had no family history of cancer.
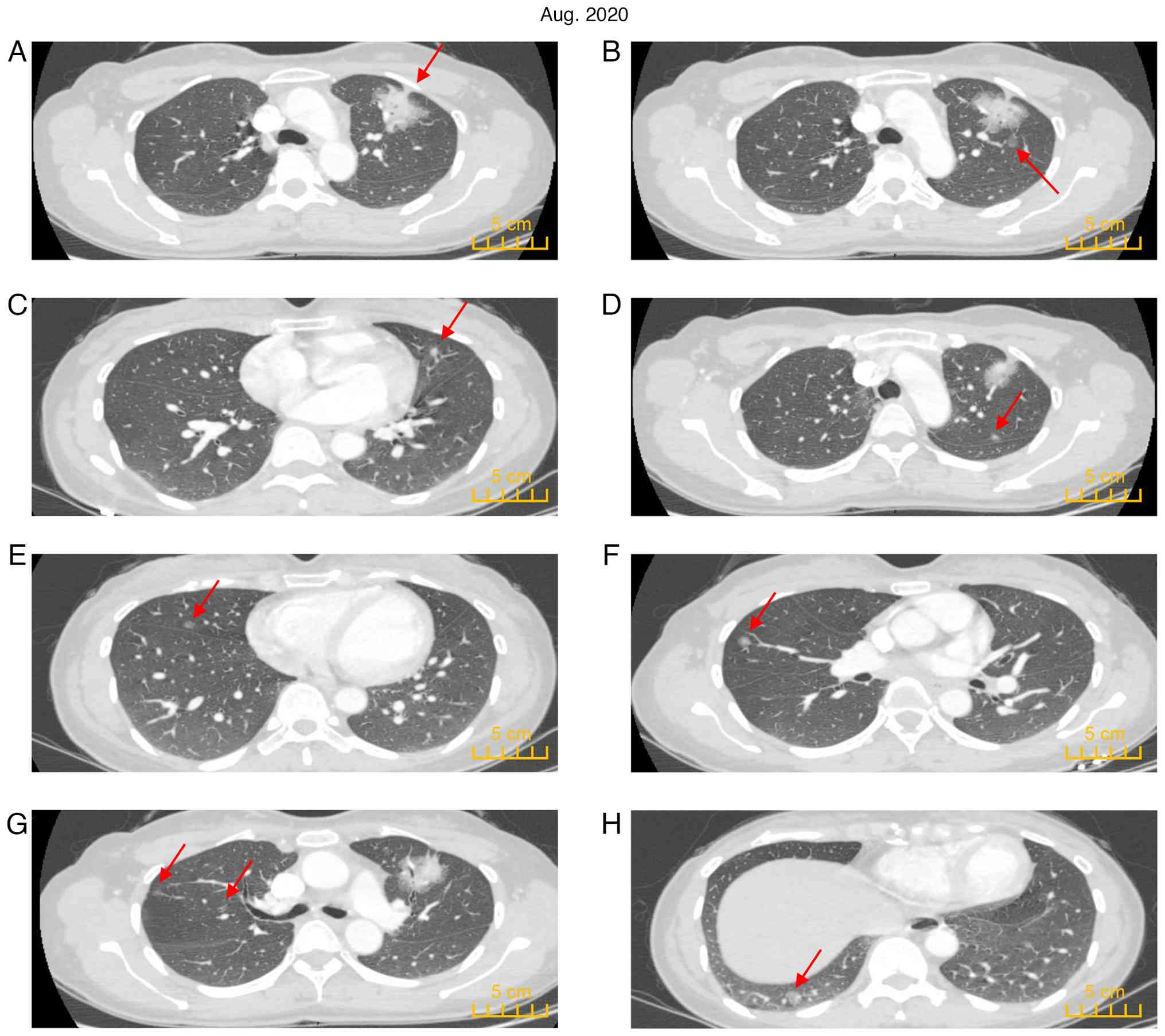
Chest-enhanced CT indicated a solid mass shadow (4.6x4.0 cm) in the
irregular part of the anterior segment of the left upper lobe with
blurred edges, visible lobulation, spiculation and cavitation
signs, local pleural traction signs, irregular bronchial stenosis
and occlusion in the lesion. Enhanced scanning indicated apparent
enhancement of the solid component of the lesion (Fig. 2A). Multiple ground-glass nodules
(GGN) and mixed ground-glass nodules were scattered in both lungs
(Fig. 2B-H). No apparent
abnormalities were noted by brain enhanced MRI. The levels of
carcinoembryonic antigen, squamous cell carcinoma antigen,
neuron-specific enolase and cytokeratin fragment were all within
the normal range. Pulmonary function retest indicated forced
expiratory volume in 1 sec (FEV1) of 2.37 l,
FEV1% (measured value/predicted value) of 91.62%, forced
vital capacity (FVC) of 2.88 l, FVC% (measured value/predicted
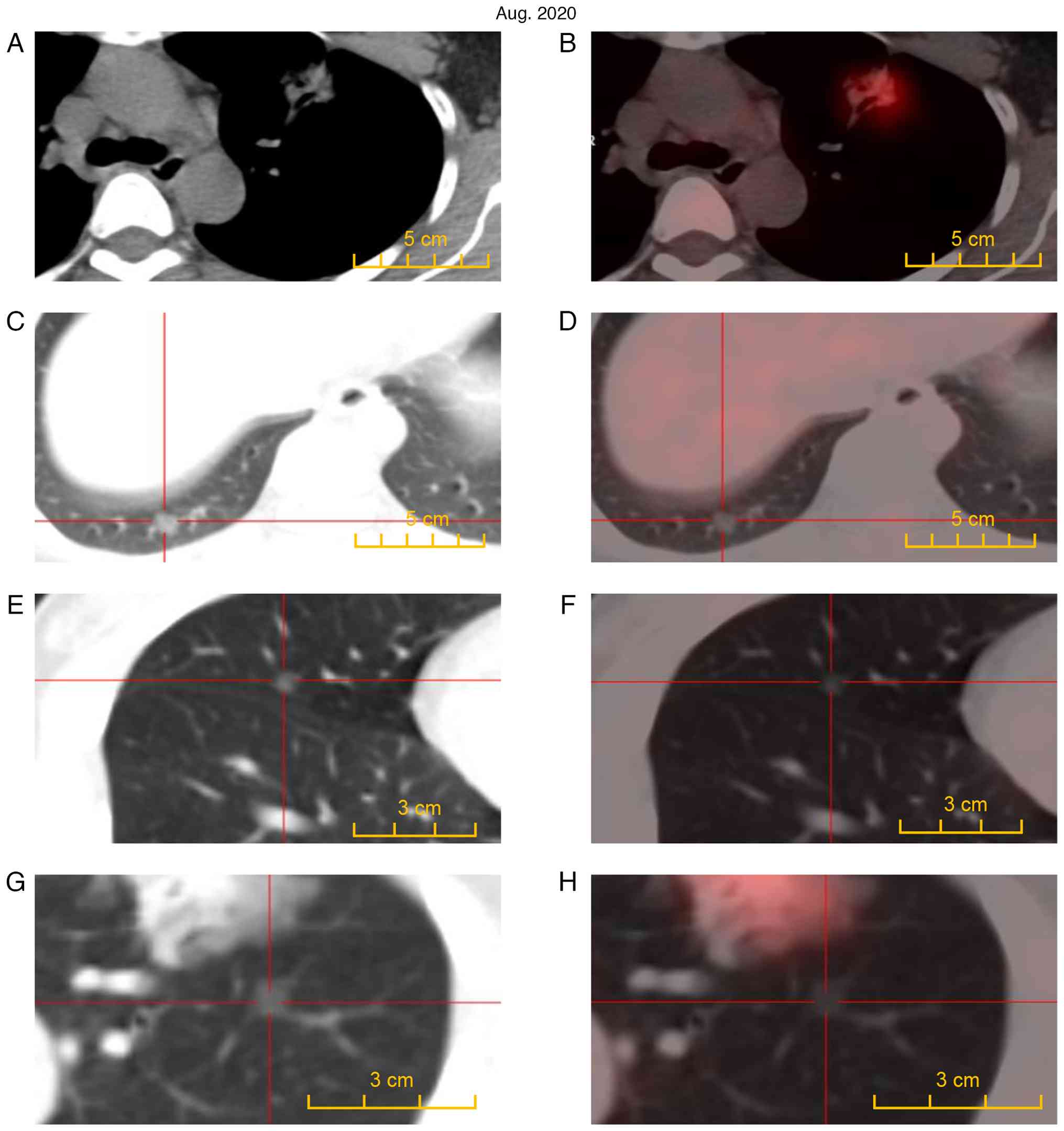
value) of 94.46% and FEV1/FVC 82.84%. Whole-body PET-CT
revealed an anterior left upper lobe lesion with increased
F18-fluorodeoxyglucose (FDG) metabolism [maximum
standardized uptake value (SUVmax)=7.3] (Fig. 3A and B). No abnormal increase in FDG metabolism
was found in the other multiple lesions (Fig. 3C-H). Peripheral lung cancer in the
anterior segment of the upper lobe of the left lung was considered.
The results suggested the presence of MPLC, and no distant
metastasis was noted. The patient was only 45 years old and their
son was young and required care. The patient was anxious after
learning that they may have late-stage lung cancer and urgently
sought diagnosis and treatment. Following multidisciplinary
discussions, a high possibility for a primary tumor was considered
and surgical treatment was recommended. The patient and their
family agreed to receive surgical treatment.
Due to the presence of >20 lesions in each lobe
of both lungs, it was impossible to remove all lesions completely.
Following multidisciplinary discussion, it was decided to perform
surgery in two stages.
As the main lesion was in the left upper lung, in
September 2020, the patient underwent a thoracoscopic left upper
lobectomy, left lower lung wedge resection and hilar-mediastinal
lymph node resection.
Frozen sections (-20˚C) of the lesions were prepared
by cutting 5-µm thick tissue sections. The sections were then
rapidly fixed with 95% ethanol and stained with hematoxylin-eosin.
Tissues were fixed in 10% formalin for 12 h at 20˚C, embedded in
paraffin and serially sectioned into 5-µm-thick sections. The
sections were then stained with hematoxylin for 10 min (at 20˚C)
and eosin for 20 sec (at 20˚C), before being observed under a light
microscope. Pathologically, adenocarcinoma in situ (AIS)
tumor cells would proliferate along the alveolar wall without
destructive interstitial invasion (16,17).
By contrast, minimally invasive adenocarcinoma (MIA) of the lung
primarily grows adherently but also has an invasive component of ≤5
mm (16-18).
Invasive adenocarcinoma (IA) has an invasive component of >5 mm
or involves lymphatic, vascular or pleural invasion (17,19).
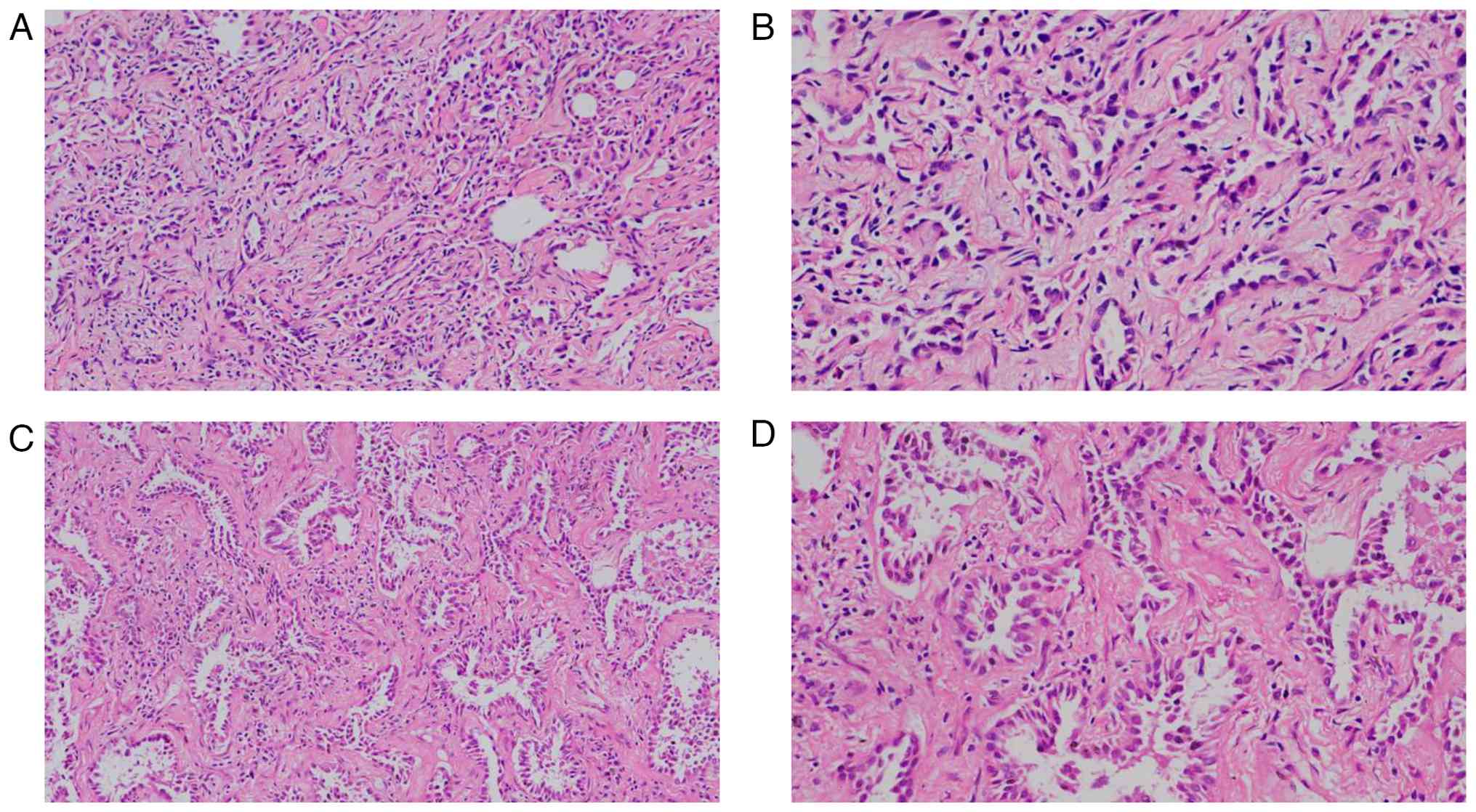
The pathological results of the frozen sections
indicated that the left upper lung (LUL) lesion, denoted as 1 (main
lesion), was IA whereas the LUL lesions 2, 3, 4, 5 and 6 were MIAs.
LUL lesion 7 and the left lower lung lesions were AIS. LUL lesion 8
was a benign fibrous nodule. Postoperative pathology indicated that
the LUL lesion 1 was IA (100% papillary subtype; T2bN0M0; IASLC 8th
edition (20); Fig. 4A and B). The results of the other lesions
(lesion 2-7) were consistent with the results of the frozen section
pathology analysis (Fig.
5A-F).
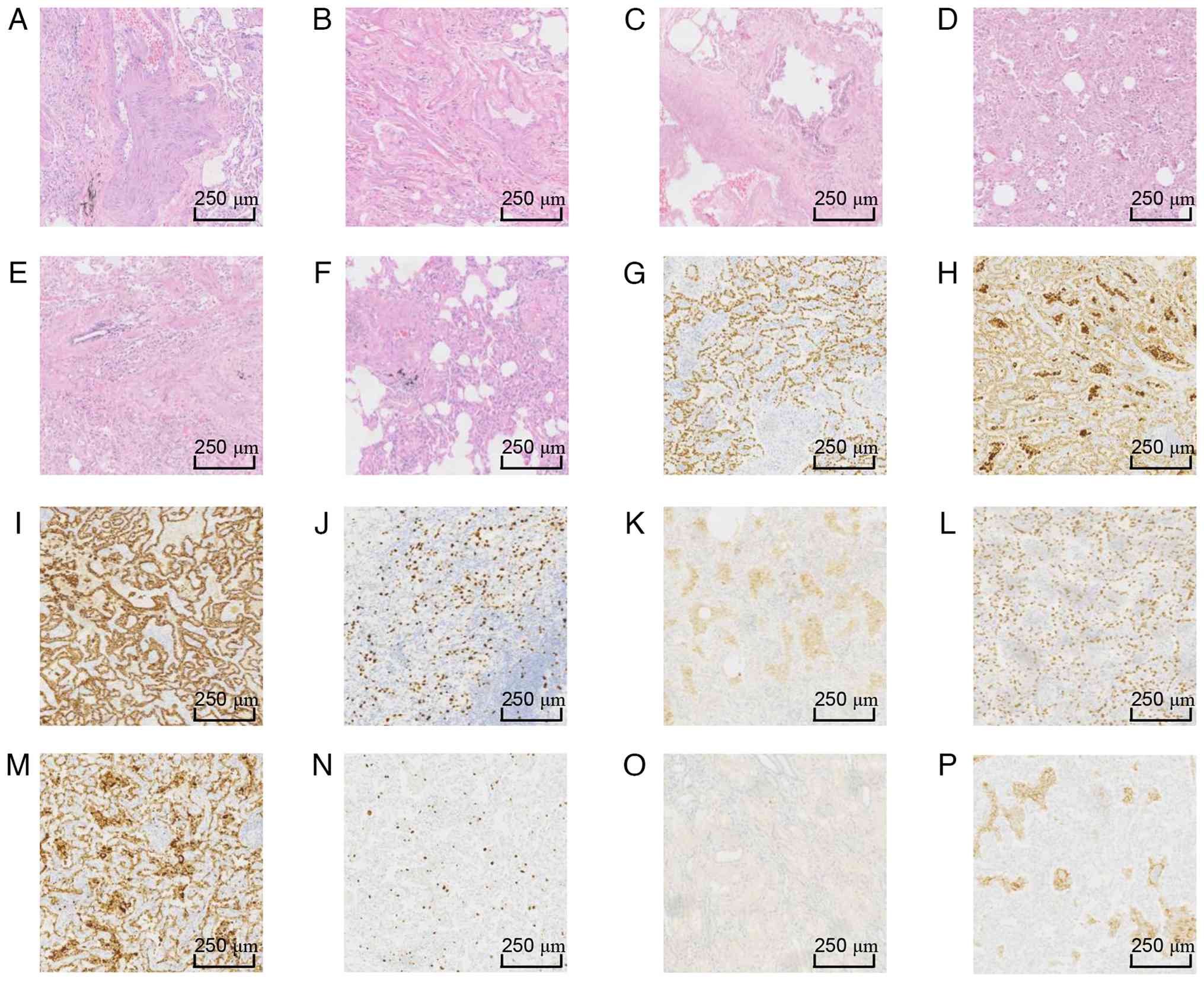 | Figure 5Pathological staining results for
samples from both lobes. H&E staining for (A) LUL2, (B) LUL3,
(C) LUL4, (D) LUL5 and (E) LUL6. Staining results indicated
minimally invasive adenocarcinoma. (F) H&E staining of LUL7
showed adenocarcinoma in situ. (G) LUL1 immunohistochemical
staining showed (G) TTF-1 (+), (H) Napsin A (+), (I) cytokeratin 7
(+), (J) Ki67 (10%) and (K) PD-L1 (tumor cells-, interstitial
macrophages+, 50%). Right lower lung lesion 1 immunohistochemical
staining showed (L) TTF-1 (+), (M) Napsin A (+), (N) Ki67 (3%), (O)
carcinoembryonic antigen (-) and (P) PD-L1 (tumor cells-, tissue
cells+, >50%). LUL, left upper lung; TTF-1, thyroid
transcription factor 1; PD-L1, programmed death-ligand 1. |
Immunohistochemistry was then performed using the
EnVision two-step assay according to standard protocols. Antibodies
used included Ki67 (cat. no. 8605580), carcinoembryonic antigen
(cat. no. IR051-5), cytokeratin 7 (cat. no. IM061-5), Napsin A
(cat. no. IM469-5), thyroid transcription factor 1 (TTF-1; cat. no.
IR301-5) and programmed death-ligand 1 (PD-L1; cat. no. 22C3; Dako;
Agilent Technologies, Inc.). The aforementioned primary antibodies
were used at a dilution of 1:100 and incubated at 4˚C for 12 h.
The results of the immunohistochemical detection
(Fig. 5G-K) indicated detection of
Napsin A (+), cytokeratin 7 (+), Ki-67 (10%), PD-L1 (tumor cells-,
interstitial macrophages+, 50%) and TTF-1 (+). Co-expression of
Napsin A and TTF-1 is a typical characteristic of lung
adenocarcinoma, while CK7 positivity rules out the possibility of
adenocarcinoma from other sites. These immunohistochemical stains
supported the diagnosis of lung adenocarcinoma.
The 9-gene test utilizes the clinically validated
LungCore® panel, encompassing the following nine common
lung cancer gene mutations: EGFR, ALK, MET,
KRAS, ERBB2, BRAF, ROS1, PIK3CA
and RET. For LUL lesion 1, Library preparation and
hybridization capture were performed using the LungCore®
Next-Generation Sequencing Kit (Guangzhou Burning Rock Medical
Laboratory Co., Ltd.) and sequencing was performed on the Illumina
MiSeqDX platform (Ilumina, Inc.). All experimental procedures
strictly followed the standard operating procedures certified by
Burning Rock Biotech under ISO 15189, including DNA extraction,
hybridization capture, sequencing and bioinformatics analysis. An
overview of Burning Rock Biotech's LungCore® 9-gene
testing panel is available online at https://www.brbiotech.com/service/c4 (accessed on
2026-1-10).
The detection results of these nine target genes
indicated that KRAS gene exon 2 mutation was detected in
both LUL lesions 1 and 5. Except for the main lesion in the LUL,
the remaining lesions were not tested for PD-L1 because they did
not reach IA staging. Combined with the analysis of imaging and
pathological results, the patient was considered for the diagnosis
of MPLC. The lesion stage was IIA (IASLC 8th edition) (20) and postoperative chemotherapy was
indicated. The patient received four cycles of pemetrexed 700 mg
combined with lobaplatin 45 mg adjuvant therapy (every 21 days)
from November 2020 to March 2021. No adverse reactions were noted
during this period.
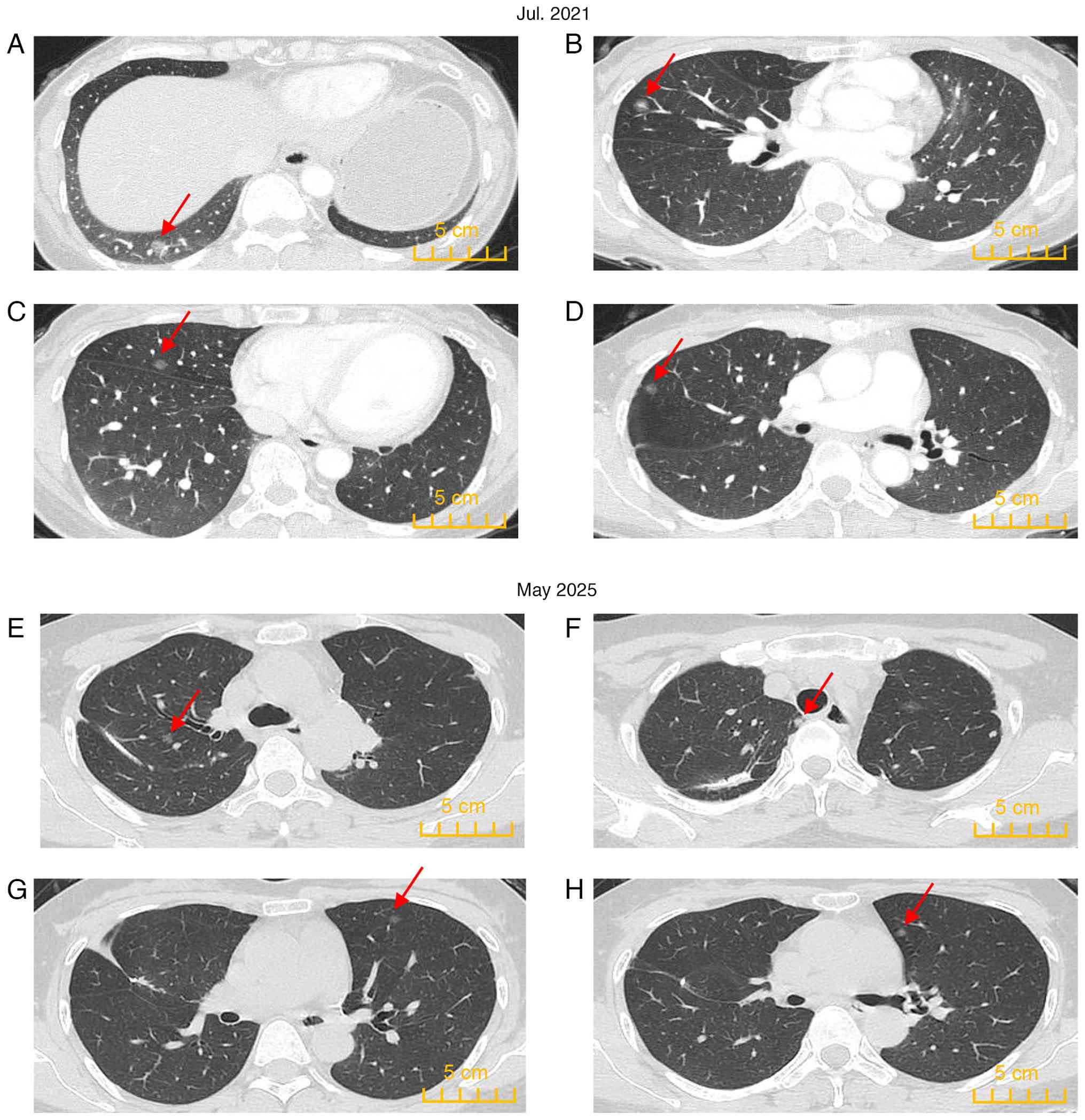
Following chemotherapy, the patient still exhibited
multiple high-risk nodules in the lungs in July 2021 (Fig. 6A-D) and the desired pulmonary
nodule regression was not achieved. At this time, the pulmonary
function retest values were as follows: FEV1, 1.41 l;
FEV1%, 54.15%; FVC, 2.05 l; FVC%, 67.08%; and
FEV1/FVC, 68.97%. The patient exhibited no asthma in
their daily life. High-risk residual lesions were recommended for
treatment and both surgery and thermal ablation were considered
options. However, the patient elected to undergo surgery. As a
result, the patient underwent a second surgery in August 2021.
Considering that excessive resection of lung tissue may lead to
insufficient pulmonary functional reserve, wedge resection was
performed on each lobe of the right lung.
The pathological results of the frozen sections
indicated that the right upper lung (RUL) lesion 1, right middle
lung lesions 1, 2 and right lower lung (RLL) lesion 1 were MIAs and
that RLL lesion 2 was AIS. RUL lesions 2 and 3 were benign fibrous
nodules. Postoperative routine pathological results indicated that
RLL lesion 1 was IA (predominantly adherent type; T1bN0M0 IASLC 8th
edition (20); Fig. 4C and D). The results of the other lesions were
consistent with those of the frozen section pathology.
Immunohistochemical analysis (Fig.
5L-P) of RLL lesion 1 indicated the following results: Napsin A
(+), PD-L1 (tumor cells-, tissue cells+, >50%), Ki-67 (3%),
carcinoembryonic antigen (-) and TTF-1 (+). Except for RLL lesion
1, the remaining lesions were not tested for PD-L1 because they did
not reach IA stage.
To further analyze the relationship between the
lesions, 425 gene-NGS detection was performed in the lesions
derived from the two surgical resections of the patient. A
pathologist confirmed that all samples contained ≥10% tumor. The
tissue was fixed with 10% formaldehyde, embedded in paraffin at
20˚C for 24 h, and then sectioned. After dewaxing with xylene,
genomic DNA was extracted. The genomic DNA was extracted using the
QIAamp DNA FFPE Tissue Kit (Qiagen GmbH). DNA fragments library
preparation was made using the KAPA hyper library preparation kit
(KAPA Biosystems; Roche Diagnostics GmbH). The resulting libraries
were sequenced using the Illumina HiSeq 4000 platform (Illumina,
Inc.). Sequencing data were analyzed using a validated automated
pipeline from Gene Biogene. The bioinformatics analysis platform
from Nanjing Shihe Gene Biotechnology Co., Ltd. (https://zh.geneseeq.com/220801144116.html) was used to
identify genetic variants, perform variant annotation, variant
screening and comprehensive analysis of variant information,
including mutations, fusions, amplifications and deletions at 425
loci (21).
The results indicated that two different KRAS
exon 2 mutations, p.G12D and p.G12C, were noted in the two IA
lesions. The other lesion-mutated genes are shown in Table I; no significant common mutated
genes were found. All lesions indicated low tumor mutation burden
(TMB). NGS results showed no significant common mutated genes
across the different lesions in this patient, which supports the
diagnosis of MPLC.
 | Table IGenomic alterations detected by NGS
profiling of pulmonary resections from the present
casea. |
Table I
Genomic alterations detected by NGS
profiling of pulmonary resections from the present
casea.
|
Characteristics | LUL 1 | LUL 2 | LUL 3 | LUL 4 | LUL 5 | LLL | RLL 1 | RUL 1 | RUL 3 | RML 1 | RML 2 |
|---|
| Histology | IA | MIA | MIA | MIA | MIA | AIS | IA | MIA | Benign | MIA | MIA |
| TMB | 4.2 | 1.1 | 0 | 4.2 | 0 | 0 | 1.1 | 3.2 | 0 | 2.1 | 2.1 |
| PD-L1 (tumor
proportion score) | - (<1%) | / | / | / | / | / | - (<1%) | / | / | / | / |
| KRAS: Exon
2, p.G12D | + | - | - | - | + | - | - | - | - | - | - |
| KRAS: Exon
2, p.G12C | - | - | - | - | - | - | + | - | - | - | - |
| ERBB2: Exon
20, p.Y772_A775dup | - | - | - | - | - | - | - | - | + | - | + |
| PRKCI: Exon
3, p.L95Qfs*8 | + | - | - | - | - | - | - | - | - | - | - |
| MYC | + | + | - | - | - | - | - | - | - | - | - |
| TERT | + | - | - | - | - | - | - | - | - | - | - |
| NKX2-1 | - | + | - | + | - | - | - | - | - | - | - |
| TERC | - | + | - | - | - | - | - | - | - | - | - |
| ARID1B | - | - | + | - | - | - | - | - | - | - | - |
| TSC1: Exon
7, p.V172Wfs*38 | - | - | - | - | - | + | - | - | - | - | - |
| ZNF703: Exon
2, p.G224S | - | - | - | - | - | - | - | + | - | - | - |
| MAP2K1: Exon
3, p.L98_I103del | - | - | - | - | - | - | - | + | - | - | - |
| EXT2: Exon
2, p.R94H | - | - | - | - | - | - | - | + | - | - | - |
| MAP3K1: Exon
5, p.R364W | - | - | - | - | - | - | - | - | + | - | - |
| NF1: Exon
14, p.Q514Rfs*43 | - | - | - | - | - | - | - | - | - | + | - |
| NF1: Exon
16, p.W599Cfs*9 | - | - | - | - | - | - | - | - | - | + | - |
| RHOA: Exon
2, p.G17A | - | - | - | - | - | - | - | - | - | - | + |
Subsequently, low-risk pulmonary residual lesions
were regularly reviewed every 6 months and were found to be stable
with no progression (Fig. 6E-H).
This case did not receive chemotherapy or radiotherapy after the
second surgery. The patient remained in recovery without recurrence
or progression until the latest follow-up in May 2025
(progression-free survival >56 months continuing). Currently,
after the second surgery, the patient experiences mild
breathlessness following physical activity, which is relieved by
rest. However, their daily life has not been affected.
Discussion
At present, the diagnosis of MPLC has been primarily
based on the diagnostic criteria of Martini-Melamed (22). In 2011, the International
Association for the Study of Lung Cancer recommended a new
classification of lung adenocarcinoma as a factor to be considered
in identifying MPLC (23,24). Lung cancer was classified into AIS,
MIA and IA. Subsequently, in 2013 the American College of Chest
Physicians updated the diagnostic criteria of MPLC to improve the
Martini criteria (22) as follows:
i) Same histological type, primary in different lung lobes; ii)
lack of N2 and N3 lymph node metastases; iii) lack of systemic
metastases; iv) different histological types; v) different genetic
and molecular biological characteristics, AIS foci with different
origins; vi) the same histological type; vii) different onset at
the same time; and viii) a >4 year interval between the two
onsets.
There has been considerable uncertainty in
distinguishing between MPLC and IM (25). It is difficult to obtain all the
lesion tissues for pathological examination and genetic testing in
patients with multifocal bilateral lungs prior to surgery.
Comprehensive imaging analysis is therefore key for the
preoperative judgment of these two conditions. At present, the
preoperative diagnosis and identification of MPLC mainly rely on
chest CT (26). The majority of
the cancer lesions in MPLC tend to exhibit the typical CT
manifestations of primary lung cancer (27), such as marginal burrs, pleural
traction, lobulation, vascular bundle sign, mixed density (such as
ground glass or solid), enhancement, lack of lymph node metastasis
and distant metastasis. In addition, it is typically difficult to
characterize the initial diagnosis and the follow-up observation of
CT is also an essential means of differential diagnosis. Untreated
primary lung cancer (following treatment of the main lesion)
frequently develops slowly, whilst patients with metastases
progress rapidly and the general condition of the patients is poor
(28,29). Compared with these aforementioned
observations, single or multiple lung metastases are mostly round
and oval, generally have smooth edges and rarely have burrs and
pleural traction signs (30,31).
CT imaging features provide the core value in the
differential diagnosis of MPLC, whilst the metabolic parameters of
PET-CT can provide supplementary evidence. Liu et al
(32) previously demonstrated that
the SUVmax among MPLC lesions was significantly
different (ΔSUV ≥-3.0), whilst the metabolic consistency of
intrapulmonary metastatic lesions was high (sensitivity 78.9%).
However, it should be noted that the low uptake characteristics of
pure GGN may result in false negative results. Following
combination of these two examinations, the study by Liu et
al (32) suggested the
diagnosis was biased towards MPLC. According to the recommendations
of previous studies, MPLC is considered a localized pathology and
should be treated with radical surgery, contributing to optimal
outcomes (7,33). It should not be arbitrarily
assessed as an advanced metastatic disease, which can deprive the
patients from selecting surgery as a treatment method. Surgical
treatment can confer optimal survival benefits to patients with
MPLC.
At present, no unified standard exists for the
surgical methods of MPLC and the selection of the methods currently
used in clinical practice depends on various aspects. The two
principles of surgery are to remove as much tumor as possible
whilst preserving as much normal lung tissue as possible. Tie et
al (34) proposed the
application of anatomic resection when the lung reserve is
sufficient, suggesting the following treatment methods: Lobectomy,
double lobectomy, pneumonectomy and lymph node dissection. When the
patient lung function is limited, lobectomy and sublobar resection
or sublobar resection alone can be performed. Among them,
anatomical segmental resection is the first option for sublobar
resection (35). The study by Yang
et al (36) demonstrated
the lack of significant differences in the 5-year survival between
patients with MPLC who underwent bilateral lobectomy and lobectomy
+ sublobar resection. Therefore, it is suggested that bilateral
MPLC can be treated with main lesion lobectomy combined with
contralateral sublobar resection.
In patients with bilateral lung multiple tumors,
single or delayed resection is safer compared with one-stage
surgery (7). Accurate
identification of MPLC and IM following surgery is essential, since
it can affect disease staging, treatment decisions and patient
outcomes. Ichinokawa et al (37) demonstrated that the third or
subsequent surgery on the same individual was expected to increase
the risk of surgical complications, such as prolonged operation
time, increased bleeding and prolonged air leakage. In the present
case, the patient exhibited >20 lesions in both lungs, prompting
an individualized treatment plan. Pathological diagnosis and
genetic testing of the tumor aided the further disease diagnosis,
stage and identification of the follow-up treatment plan. In a
study of 26 patients with MPLC, a multivariate analysis previously
indicated that adjuvant chemotherapy positively improved patient
survival (38). The present
patient achieved optimal surgical results and high quality of life
following undergoing second-stage surgery combined with
chemotherapy where no tumor recurrence or progression was noted
during the 56-month follow-up period.
Clinically, the differential diagnosis of MPLC and
IM is complex. In addition to imaging examinations, the diagnosis
should be combined with a comprehensive analysis of pathological
and molecular biological features (39). With the development of molecular
pathology, it was first proposed to use molecular genetics to
diagnose multiple lung lesions of the same pathological type
(40). A previous study has shown
that sequencing of ~50 genes can be used as an indicator of
multiple lung tumors containing different driver mutations. These
tumors were characterized as MPLC, where in case only one driver
mutation is common, the tumors would be characterized as IM
(41). In cases with no clear
histopathological distinction or similar histological subtypes, the
consistency of genomic alteration profiles among multiple nodules
would then provide additional insights into their clonal
relationships and therefore guides the diagnosis of MPLC (42,43).
A previous study has reported that metastatic lung lesions rarely
exhibit discordant mutational patterns (44). Another study of 120 patients by
Mansuet-Lupo et al (45)
indicated that molecular typing could increase the sensitivity of
the detection of MPLC compared with histopathological features and
proposed an integrated tissue-molecular algorithm for MPLC. When
multiple tumors share a frequent hotspot mutation (such as
EGFR exon 19 deletions, EGFR p.L858R or KRAS
p.G12X), histological algorithms can aid the confirmation of the
diagnosis.
However, IM and MPLC are similar in genetic and
immune characteristics, such that genomics alone may not be able to
effectively distinguish IM from MPLC (46). Therefore, a comprehensive
evaluation of the present case was conducted through imaging,
pathological and genetic analyses. Han et al (47) indicated that all three nodules in
an MPLC case expressed RET mutations. However, there was
significant heterogeneity in the gene mutations (differences in the
number of cellular mutations, substitution composition levels and
clustering analysis of the three nodules). Thomas et al
(48) further indicated that whole
genome sequencing can be used to distinguish whether the nodules
possess a definite origin. Saab et al (49) in another report documented that 65%
patients with MPLC could be identified based on clinical
manifestations, imaging and morphology. In addition, 94% of
patients can be identified by combining patient morphological
characteristics and genomics.
For unresected residual nodules, the current
consensus recommends individualized monitoring based on their
biological characteristics. A prospective study by Shimada et
al (50) has indicated that
the progression rate of residual GGN following resection of the
main lesion was only 8% (median follow-up, 58 months). The
incidence of new lesions (23%) and the growth of residual lesions
did not affect the patient overall survival [overall survival (OS);
P=0.82]. This supports the rationale of the ‘main lesion first’
strategy. When the residual GGN is pure ground glass density and
the diameter is <8 mm, then annual CT follow-up was recommended
to be sufficient (51). By
contrast, in case of a partially solid nodule or a solid component
≥6 mm, then review should be shortened to 6 months (52).
The indications for surgical resection of all
lesions should be strictly limited to the following: i) Progression
of residual GGN during follow-up (diameter increase >2 mm or new
solid components); ii) lesions located in the ‘advantageous site’
for sublobar resection; and iii) patients with severe anxiety
symptoms. For deep small nodules, thermal ablation can be used as
an alternative. However, the 5-year local control rate of thermal
ablation (42-55%) is still lower compared with that of surgical
resection (80-94%) (53).
Regarding thermal ablation, several retrospective
studies have demonstrated that for patients with high-risk stage I
lung cancer who are not suitable for surgery, thermal ablation
treatments, such as radiofrequency ablation and microwave ablation,
can achieve a prognosis similar to that of lobectomy (53-55).
Currently, the safety and efficacy of thermal ablation for subsolid
nodules have been reported and preliminary results comparable to
those of surgical resection have been achieved, with a 5-year OS
and tumor-specific survival rates of ~95 and 100%, respectively
(55-57).
Thermal ablation has become one of the treatment options or a
supplemental treatment to surgery for multiple ground-glass nodules
in the lungs and a consensus has been reached (58,59).
In the present case, the patient exhibited residual high-risk
lesions in the right lung, which were at the edge of the lobe. When
lung function was still acceptable, high-risk residual lesions were
recommended for treatment, and both surgery and thermal ablation
were options. However, the patient elected to undergo surgery.
NGS-driven comprehensive genomic analysis is
reshaping the diagnostic standards for MPLC. Chang et al
(60) previously reported that NGS
could reduce the misdiagnosis rate of MPLC and IM by 22% compared
with traditional histological evaluation techniques. Notably, the
accuracy of identifying intrapulmonary metastases was improved by
44%. NGS can be used to accurately trace the origin of multiple
primary lesions by detecting a number of driver gene mutations,
such as HER2 and chromosomal rearrangements (61). In the field of treatment, in
addition to the thermal ablation, immunotherapy combined with
radiotherapy has also shown potential. A phase II trial by Chang
et al (62) indicated that
PD-1 inhibitors combined with stereotactic radiotherapy increased
the 4-year event-free survival rate of early non-small cell lung
cancer to 77%, resulting to an increase of 24% compared with
radiotherapy alone. However, targeted therapy is not without
limitations. Cheng et al (63) reported that the response rate of
EGFR-tyrosine kinase inhibitors to multiple GGN residual lesions
was only 23.9%, mainly due to the heterogeneity of the mutation
spectrum of each lesion (only 7.9% of secondary lesions carry the
same driver mutation as the primary lesion).
To the best of our knowledge, the present case
report presents the highest number of resections of multiple
primary lung cancers (13 lesions) and the highest number of lung
cancer lesions (10 lesions) detected by NGS in a single patient. It
was found that all lesions exhibited low TMB, which was consistent
with the existing research data on multiple primary lung cancers
(21,64). Among them, the LUL lesion 1 and the
RLL lesion 1 were both IAs. However, their histological subtypes
differed. In addition, KRAS gene exon 2 p.G12D mutation was
detected in LUL lesion 1, whilst KRAS gene exon 2 p.G12C
mutation was detected in RLL lesion 1. Combined with pathological
results and genetic testing results, MPLC diagnosis was supported.
The multiplexed nature of NGS technology results in high throughput
and sensitivity, providing an effective complement to current
diagnostic efforts. With the development of molecular diagnostic
technology, the absorption of higher number of genes and molecular
features will aid the development of a more objective basis for
identifying MPLC and metastatic cancer, providing scientific and
rational standardized diagnosis and treatment for patients with
MPLC.
In conclusion, due to the development of novel
technologies, the diagnosis and treatment of MPLC have developed
rapidly. Using only imaging, pathology and genetic testing to
diagnose multiple lung lesions can readily lead to the misdiagnosis
of MPLC as IM, which may in turn result in the lack of surgical
treatment for the patients. Broad-spectrum NGS can differentiate
between MPLC and IM and serves a vital role in the diagnosis and
subsequent treatment (identification of driver oncogenes) of MPLC.
Integrating radiology, histopathology and integrated genomic
features in clinical practice by a multidisciplinary team
facilitates a more accurate diagnosis of MPLC, and is expected to
become a trend in the differential diagnosis of MPLC in the future.
Furthermore, an individualized treatment design is more beneficial
to patients with MPLC containing a large number of lesions in both
lungs. A case of the diagnosis and individualized treatment of MPLC
was provided with ultra-multiple lesions in both lungs, which can
be used as a reference for the diagnosis and treatment of similar
patients.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The sequencing data generated in the present study
can be found in the Genome Sequence Archive (GSA) database of the
China National Center for Bioinformation (https://ngdc.cncb.ac.cn/gsa-human) under accession
number HRA003776. Further inquiries can be directed to the
corresponding author. The individual name of this patient in the
dataset is P24, and the individual accession number is HRI328653.
After the application is reviewed by the GSA database, the genetic
data of this patient can be downloaded. Unrestricted public access
to the NGS data is not possible due to national legal requirements
(Regulations on the Management of Human Genetic Resources of the
People's Republic of China, articles 7 and 28; https://www.most.gov.cn/xxgk/xinxifenlei/fdzdgknr/fgzc/flfg/201906/t20190612_147044.html).
The other data generated in the present report may be requested
from the corresponding author.
Authors' contributions
GZ and ZL conceived the study. WW and BD extracted
and organized the original data. GZ and YZ wrote the main part of
the original manuscript. WW and CC developed the treatment plan and
wrote a literature review of the progress in the discussion
section. YZ analyzed and interpreted the patient's imaging results,
BD and ZL interpreted the patient's pathology results. GZ and ZL
confirm the authenticity of all the raw data. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
The present report was reviewed and approved by the
Ethics Committee of the Affiliated Hospital of Guangdong Medical
University (approval no. YJLW2022007; Zhanjiang, China).
Patient consent for publication
Written informed consent was obtained from the
individual for the publication of any potentially identifiable
images or data included in the present case report.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249.
2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Chen C, Huang X, Peng M, Liu W, Yu F and
Wang X: Multiple primary lung cancer: A rising challenge. J Thorac
Dis. 11 (Suppl 4):S523–S536. 2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Gazdar AF and Minna JD: Multifocal lung
cancers-clonality vs field cancerization and does it matter? J Natl
Cancer Inst. 101:541–543. 2009.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Samadzadeh Tabrizi N, Gallant B, Harris E,
Arnold BN and Fabian T: Contemporary incidence of synchronous
multiple primary lung cancers and survival in the era of lung
cancer screening. Innovations (Phila). 19:23–29. 2024.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Zhang DY, Liu J, Zhang Y, Ye JY, Hu S,
Zhang WX, Yu DL and Wei YP: One-stage resection of four genotypes
of bilateral multiple primary lung adenocarcinoma: A case report.
World J Clin Cases. 10:10301–10309. 2022.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Cheng H, Li WH, Li XJ, Zhong HC, Wang XJ,
Lin YJ, Liu XG, Wu XW and Cao QD: Small nodules (≤6 mm in diameter)
of multiple primary lung cancers: Prevalence and management. J
Cardiothorac Surg. 17(278)2022.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Zhang Z, Gao S, Mao Y, Mu J, Xue Q, Feng X
and He J: Surgical outcomes of synchronous multiple primary
non-small cell lung cancers. Sci Rep. 6(23252)2016.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Kawai H, Iguchi K, Takayashiki N, Okauchi
S and Satoh H: Metachronous isolated contralateral lung metastasis
from pulmonary adenosquamous carcinoma with EGFR mutation. Acta
Medica (Hradec Kralove). 63:141–144. 2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Nie Y, Wang X, Yang F, Zhou Z, Wang J and
Chen K: Surgical prognosis of synchronous multiple primary lung
cancer: Systematic review and meta-analysis. Clin Lung Cancer.
22:341–350.e3. 2021.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Zhang H, Liu Q, Chen L, Song L, Mao F,
Zhou W, Li J, Song Z, Miao W and Shentu Y: Identification of the
prognostic factors for synchronous multiple primary lung cancer
treated with staged bilateral surgery. Clin Respir J.
18(e70017)2024.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Liu M, He W, Yang J and Jiang G: Surgical
treatment of synchronous multiple primary lung cancers: A
retrospective analysis of 122 patients. J Thorac Dis. 8:1197–1204.
2016.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Wang L, Yu J, Luo Y, Nie J, Ge X, Li Y,
Hua B and Liu R: The intersection of artificial intelligence and
lung nodule research: Current applications and future prospects.
Int J Surg: January 13, 2026 (Epub ahead of print).
|
|
13
|
Astaraki M, Zakko Y, Toma Dasu I, Smedby Ö
and Wang C: Benign-malignant pulmonary nodule classification in
low-dose CT with convolutional features. Phys Med. 83:146–153.
2021.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Schneider F and Dacic S: Histopathologic
and molecular approach to staging of multiple lung nodules. Transl
Lung Cancer Res. 6:540–549. 2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Zheng R, Shen Q, Mardekian S, Solomides C,
Wang ZX and Evans NR III: Molecular profiling of key driver genes
improves staging accuracy in multifocal non-small cell lung cancer.
J Thorac Cardiovasc Surg. 160:e71–e79. 2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Motono N, Matsui T, Machida Y, Usuda K and
Uramoto H: Prognostic significance of histologic subtype in pStage
I lung adenocarcinoma. Med Oncol. 34(100)2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Travis WD, Asamura H, Bankier AA, Beasley
MB, Detterbeck F, Flieder DB, Goo JM, MacMahon H, Naidich D,
Nicholson AG, et al: The IASLC lung cancer staging project:
Proposals for coding T categories for subsolid nodules and
assessment of tumor size in part-solid tumors in the forthcoming
eighth edition of the TNM classification of lung cancer. J Thorac
Oncol. 11:1204–1223. 2016.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Shih AR, Uruga H, Bozkurtlar E, Chung JH,
Hariri LP, Minami Y, Wang H, Yoshizawa A, Muzikansky A, Moreira AL
and Mino-Kenudson M: Problems in the reproducibility of
classification of small lung adenocarcinoma: An international
interobserver study. Histopathology. 75:649–659. 2019.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Cohen JG, Reymond E, Jankowski A,
Brambilla E, Arbib F, Lantuejoul S and Ferretti GR: Lung
adenocarcinomas: Correlation of computed tomography and pathology
findings. Diagn Interv Imaging. 97:955–963. 2016.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Feng SH and Yang ST: The new 8th TNM
staging system of lung cancer and its potential imaging
interpretation pitfalls and limitations with CT image
demonstrations. Diagn Interv Radiol. 25:270–279. 2019.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Liang Z, Zeng G, Wan W, Deng B, Chen C, Li
F, Lin G, Lin Y, Lin H, Mo G and Miao H: The unique genetic
mutation characteristics based on large panel next-generation
sequencing (NGS) detection in multiple primary lung cancers (MPLC)
patients. Discov Med. 35:131–143. 2023.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Kozower BD, Larner JM, Detterbeck FC and
Jones DR: Special treatment issues in non-small cell lung cancer:
Diagnosis and management of lung cancer, 3rd ed: American college
of chest physicians evidence-based clinical practice guidelines.
Chest. 143 (5 Suppl):e369S–e399S. 2013.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Travis WD, Brambilla E, Noguchi M,
Nicholson AG, Geisinger KR, Yatabe Y, Beer DG, Powell CA, Riely GJ,
Van Schil PE, et al: International association for the study of
lung cancer/american thoracic society/european respiratory society
international multidisciplinary classification of lung
adenocarcinoma. J Thorac Oncol. 6:244–285. 2011.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Takamochi K, Oh S, Matsuoka J and Suzuki
K: Clonality status of multifocal lung adenocarcinomas based on the
mutation patterns of EGFR and K-ras. Lung Cancer. 75:313–320.
2012.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Homer RJ: Pathologists' staging of
multiple foci of lung cancer: Poor concordance in absence of
dramatic histologic or molecular differences. Am J Clin Pathol.
143:701–706. 2015.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Stefanidis K, Konstantellou E, Yusuf G,
Moser J, Tan C and Vlahos I: The evolving landscape of lung cancer
surgical resection: An update for radiologists with focus on key
chest ct findings. AJR Am J Roentgenol. 218:52–65. 2022.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Jiang L, Zheng X, Wu S, Zhang J, Ru G and
Li Y: A rare case of synchronous multiple primary lung cancer:
Squamous cell cancer and small cell lung cancer. Onco Targets Ther.
12:8801–8806. 2019.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Román A, Perez-Rozos A, Otero A, Jodar C,
García-Ríos I, Lupiañez-Perez Y, Antonio Medina J and Gomez-Millan
J: Efficacy and safety of a simplified SBRT regimen for central and
peripheral lung tumours. Clin Transl Oncol. 22:144–150.
2020.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Patrini D, Panagiotopoulos N, Bedetti B,
Mitsos S, Crisci R, Solli P, Bertolaccini L and Scarci M: Surgical
approach in oligometastatic non-small cell lung cancer. Ann Transl
Med. 6(93)2018.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Kawaguchi T, Takeda M, Yoshikawa D, Taiji
R, Yamada A, Miyata R, Hamaji M, Hosono M and Sawabata N: Pulmonary
metastases from pancreatic cancer have different
clinico-radiological features compared with those from colorectal
cancer. Jpn J Clin Oncol. 54:1314–1320. 2024.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Yu Y, Zhu J, Zhou Y, Sang S, Zhu Y and
Zhang X: Imaging characteristics and prognostic value of isolated
pulmonary metastasis from colorectal cancer demonstrated
with18F-FDG PET/CT. Biomed Res Int.
2022(2230079)2022.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Liu Y, Tang Y, Xue Z, Jin X, Ma G, Zhao P
and Chu X: SUVmax ratio on PET/CT may differentiate between lung
metastases and synchronous multiple primary lung cancer. Acad
Radiol. 27:618–623. 2020.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Dai L, Yang HL, Yan WP, Liang Z, Xiong HC,
Kang XZ, Yang YB, Fu H, Fan MY and Chen KN: The equivalent efficacy
of multiple operations for multiple primary lung cancer and a
single operation for single primary lung cancer. J Thorac Dis.
8:855–861. 2016.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Tie H, Luo J, Shi R, Li Z, Chen D and Wu
Q: Characteristics and prognosis of synchronous multiple primary
lung cancer after surgical treatment: A systematic review and
meta-analysis of current evidence. Cancer Med. 10:507–520.
2021.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Liu XD, Qu Y and Lu SS: Synchronous double
primary lung cancer: A report of three cases. Chin J Cancer Res.
26:E17–E21. 2014.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Yang H, Sun Y, Yao F, Yu K, Gu H, Han B
and Zhao H: Surgical therapy for bilateral multiple primary lung
cancer. Ann Thorac Surg. 101:1145–1152. 2016.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Ichinokawa H, Sowa T, Yaguchi T, Oizumi H,
Takamochi K and Suzuki K: Six radical surgeries for metachronous
multiple primary lung cancer: A case report. Int J Surg Case Rep.
127(110860)2025.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Kocaturk CI, Gunluoglu MZ, Cansever L,
Demir A, Cinar U, Dincer SI and Bedirhan MA: Survival and
prognostic factors in surgically resected synchronous multiple
primary lung cancers. Eur J Cardiothorac Surg. 39:160–166.
2011.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Zhao L, Liu C, Xie G, Wu F and Hu C:
Multiple primary lung cancers: A new challenge in the era of
precision medicine. Cancer Manag Res. 12:10361–10374.
2020.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Detterbeck FC, Jones DR, Kernstine KH and
Naunheim KS: American College of Physicians. Lung cancer. Special
treatment issues. Chest. 123 (1 Suppl):244S–258S. 2003.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Donfrancesco E, Yvorel V, Casteillo F,
Stachowicz ML, Patoir A, Tiffet O, Péoc'h M and Forest F:
Histopathological and molecular study for synchronous lung
adenocarcinoma staging. Virchows Arch. 476:835–842. 2020.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Takahashi K, Kohno T, Matsumoto S,
Nakanishi Y, Arai Y, Yamamoto S, Fujiwara T, Tanaka N and Yokota J:
Clonal and parallel evolution of primary lung cancers and their
metastases revealed by molecular dissection of cancer cells. Clin
Cancer Res. 13:111–120. 2007.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Lu X, Xu Q, Wang J, Bi J, Wang Z and Li Y:
Allele frequency of somatic mutations in individuals reveals
signatures of cancer-related genes. Acta Biochim Biophys Sin
(Shanghai). 47:657–660. 2015.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Yatabe Y, Matsuo K and Mitsudomi T:
Heterogeneous distribution of EGFR mutations is extremely rare in
lung adenocarcinoma. J Clin Oncol. 29:2972–2977. 2011.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Mansuet-Lupo A, Barritault M, Alifano M,
Janet-Vendroux A, Zarmaev M, Biton J, Velut Y, Le Hay C, Cremer I,
Régnard JF, et al: Proposal for a combined histomolecular algorithm
to distinguish multiple primary adenocarcinomas from intrapulmonary
metastasis in patients with multiple lung tumors. J Thorac Oncol.
14:844–856. 2019.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Yang R, Li P, Wang D, Wang L, Yin J, Yu B,
Li M, Wang S and Wang Y: Genetic and immune characteristics of
multiple primary lung cancers and lung metastases. Thorac Cancer.
12:2544–2550. 2021.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Han Y, Geng Y, Sui Q, Liu Y, Xie S, Gao M,
Liu Q, Liu G and Wang S: When multiple primary lung cancers express
the same rare mutation: A case report. Front Oncol.
14(1475193)2024.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Thomas V, Rashed A, Faul C, Nicholson S,
Young V, Hanson J, Hennessy BT, Toomey S and Furney SJ: Genome
sequencing of multiple primary lung cancers harbouring mixed
histology and spontaneously regressing small-cell lung cancer. J
Pers Med. 14(257)2024.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Saab J, Zia H, Mathew S, Kluk M, Narula N
and Fernandes H: Utility of genomic analysis in differentiating
synchronous and metachronous lung adenocarcinomas from primary
adenocarcinomas with intrapulmonary metastasis. Transl Oncol.
10:442–449. 2017.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Shimada Y, Saji H, Otani K, Maehara S,
Maeda J, Yoshida K, Kato Y, Hagiwara M, Kakihana M, Kajiwara N, et
al: Survival of a surgical series of lung cancer patients with
synchronous multiple ground-glass opacities, and the management of
their residual lesions. Lung Cancer. 88:174–180. 2015.PubMed/NCBI View Article : Google Scholar
|
|
51
|
MacMahon H, Naidich DP, Goo JM, Lee KS,
Leung ANC, Mayo JR, Mehta AC, Ohno Y, Powell CA, Prokop M, et al:
Guidelines for management of incidental pulmonary nodules detected
on CT images: From the fleischner society 2017. Radiology.
284:228–243. 2017.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Jiang G, Chen C, Zhu Y, Xie D, Dai J, Jin
K, Shen Y, Wang H, Li H, Zhang L, et al: Shanghai pulmonary
hospital experts consensus on the management of ground-glass
nodules suspected as lung adenocarcinoma (version 1). Zhongguo Fei
Ai Za Zhi. 21:147–159. 2018.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
53
|
Genshaft SJ, Suh RD, Abtin F, Baerlocher
MO, Dariushnia SR, Devane AM, Himes E, Lisberg A, Padia S, Patel S
and Yanagawa J: Society of interventional radiology quality
improvement standards on percutaneous ablation of non-small cell
lung cancer and metastatic disease to the lungs. J Vasc Interv
Radiol. 32:1242.e1–1242.e10. 2021.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Ambrogi MC, Fanucchi O, Dini P, Melfi F,
Davini F, Lucchi M, Massimetti G and Mussi A: Wedge resection and
radiofrequency ablation for stage I nonsmall cell lung cancer. Eur
Respir J. 45:1089–1097. 2015.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Yang X, Ye X, Lin Z, Jin Y, Zhang K, Dong
Y, Yu G, Ren H, Fan W, Chen J, et al: Computed tomography-guided
percutaneous microwave ablation for treatment of peripheral
ground-glass opacity-lung adenocarcinoma: A pilot study. J Cancer
Res Ther. 14:764–771. 2018.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Kodama H, Yamakado K, Hasegawa T, Takao M,
Taguchi O, Fukai I and Sakuma H: Radiofrequency ablation for
ground-glass opacity-dominant lung adenocarcinoma. J Vasc Interv
Radiol. 25:333–339. 2014.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Iguchi T, Hiraki T, Gobara H, Fujiwara H,
Matsui Y, Soh J, Toyooka S, Kiura K and Kanazawa S: Percutaneous
radiofrequency ablation of lung cancer presenting as ground-glass
opacity. Cardiovasc Intervent Radiol. 38:409–415. 2015.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Huang G, Yang X, Li W, Wang J, Han X, Wei
Z, Meng M, Ni Y, Zou Z, Wen Q, et al: A feasibility and safety
study of computed tomography-guided percutaneous microwave
ablation: A novel therapy for multiple synchronous ground-glass
opacities of the lung. Int J Hyperthermia. 37:414–422.
2020.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Liu B and Ye X: Computed tomography-guided
percutaneous microwave ablation: A novel perspective to treat
multiple pulmonary ground-glass opacities. Thorac Cancer.
11:2385–2388. 2020.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Chang JC, Alex D, Bott M, Tan KS, Seshan
V, Golden A, Sauter JL, Buonocore DJ, Vanderbilt CM, Gupta S, et
al: Comprehensive next-generation sequencing unambiguously
distinguishes separate primary lung carcinomas from intrapulmonary
metastases: comparison with standard histopathologic approach. Clin
Cancer Res. 25:7113–7125. 2019.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Heeke AL, Elliott A, O'Keefe K, Livasy C,
Symanowski JT, Steiner MR, Kang IM, Hoon DSB, Walker P, Sledge GW,
et al: Human epidermal growth factor receptor 2 alterations and
prognostic implications in all subtypes of breast cancers. JCO
Precis Oncol. 9(e2300719)2025.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Chang JY, Lin SH, Dong W, Liao Z, Gandhi
SJ, Gay CM, Zhang J, Chun SG, Elamin YY, Fossella FV, et al:
Stereotactic ablative radiotherapy with or without immunotherapy
for early-stage or isolated lung parenchymal recurrent
node-negative non-small-cell lung cancer: An open-label,
randomised, phase 2 trial. Lancet. 402:871–881. 2023.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Cheng B, Li C, Zhao Y, Li J, Xiong S,
Liang H, Liu Z, Zeng W, Liang W and He J: The impact of
postoperative EGFR-TKIs treatment on residual GGO lesions after
resection for lung cancer. Signal Transduct Target Ther.
6(73)2021.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Hu C, Zhao L, Liu W, Fan S, Liu J, Liu Y,
Liu X, Shu L, Liu X, Liu P, et al: Genomic profiles and their
associations with TMB, PD-L1 expression, and immune cell
infiltration landscapes in synchronous multiple primary lung
cancers. J Immunother Cancer. 9(e003773)2021.PubMed/NCBI View Article : Google Scholar
|