1. Introduction
Breast cancer is a notable global public health
issue, ranking as the second most common cancer worldwide after
lung cancer and the most commonly diagnosed malignancy among women
(1). Recent epidemiological data
has reported ~2.31 million new cases annually, accounting for 11.6%
of all cancer diagnoses, resulting in 670,000 mortalities each
year. These figures underscore the profound impact of breast cancer
on global mortality rates and healthcare burden (1,2).
Despite advances in treatment and early detection, the pathogenesis
and progression of breast cancer remain incompletely understood,
necessitating further exploration of the underlying mechanisms to
inform prevention and therapeutic strategies.
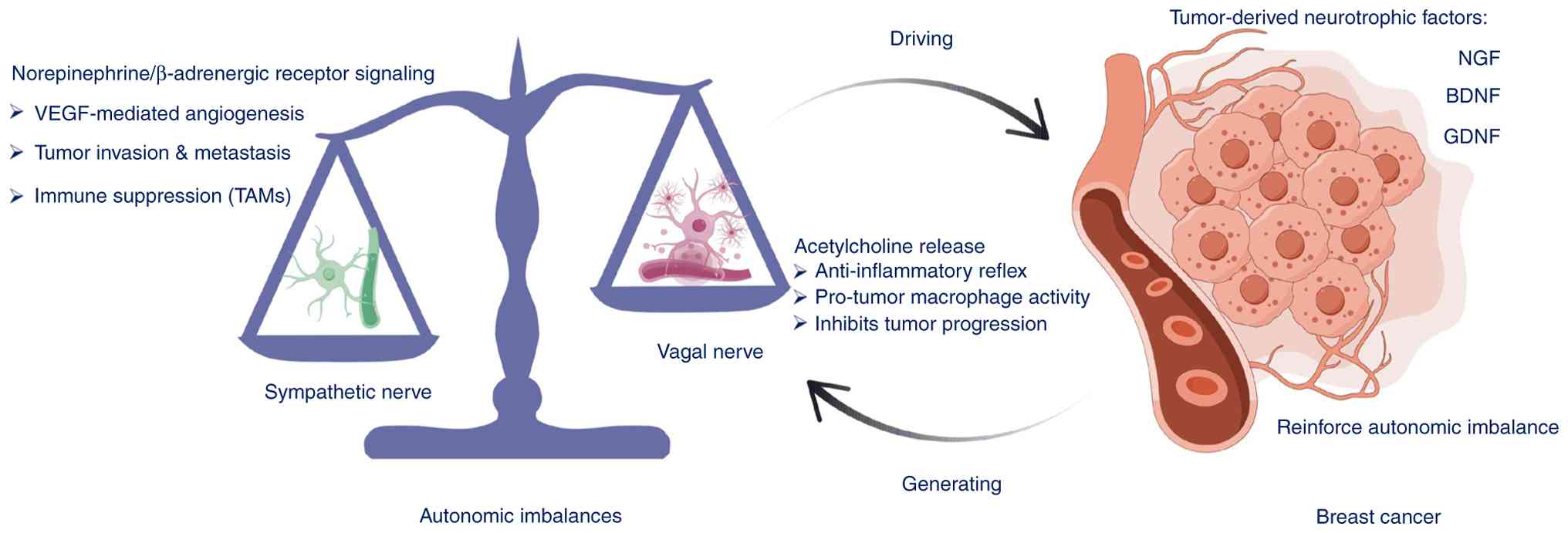
One emerging area of interest is the role of the
autonomic nervous system (ANS), which primarily consists of the
sympathetic and vagus nerves in the regulation of nearly all
tissues and organs, excluding cartilage and lens (3). Traditionally considered peripheral to
cancer progression, the ANS has previously been identified as
exhibiting a key role in the pathophysiology of breast cancer.
Dysregulation of the ANS, characterized by sympathetic
overactivation and reduced vagal tone, has been previously
implicated in both the onset and progression of breast cancer,
linking neuro-tumor crosstalk with unfavorable prognosis and
increased recurrence rates (4-7).
High nerve fiber density within tumors has also been associated
with poorer clinical outcomes, suggesting that the ANS serves a key
regulatory role in breast cancer development (8,9).
Mechanistically, overactivation of sympathetic
nerves facilitates tumor growth and metastasis through the release
of various neurotransmitters, such as norepinephrine, promoting
angiogenesis through upregulation of vascular endothelial growth
factor (VEGF) (7,10-12).
By contrast, increased vagal tone has been shown to exert
protective effects, potentially mitigating tumor progression
(6,7,10,13).
This bidirectional influence highlights the balance between
sympathetic and parasympathetic systems in breast cancer
pathogenesis. Furthermore, the ANS can modulate the tumor
microenvironment through secretion of neurotrophic factors (such as
nerve growth factor) and through regulation of immune cell activity
and angiogenesis, further contributing to tumor progression
(12,14). Therefore, investigating the
interactions between the ANS and breast cancer should deepen
understanding of the pathophysiological mechanisms underlying this
disease and offer novel anti-cancer strategies for enhancing
treatment (Fig. 1).
The present review summarizes and critically
evaluates current literature regarding the interactions between the
ANS and breast cancer. By highlighting the ANS as a key regulator
in breast cancer pathophysiology, the present review aims to
provide insight into the novel mechanistic pathways and therapeutic
targets. The present review is intended for researchers and
clinicians in the field of breast cancer and tumor-nervous system
interactions, including translational and clinical
investigators.
2. Survey methodology
To ensure a comprehensive and unbiased coverage of
the literature, a systematic search was conducted across multiple
databases, including PubMed (https://pubmed.ncbi.nlm.nih.gov/), Web of Science
(https://www.webofscience.com/) and China
National Knowledge Infrastructure (CNKI; https://www.cnki.net/). The primary search period
spanned from 2010-2024, with additional inclusion of earlier
seminal articles that remain relevant to the field. To identify
pertinent studies, a combination of the following keywords and
their Boolean operators were used: ‘Autonomic nervous system’,
‘nerves’, ‘sympathetic nervous system’, ‘vagus nerve’,
‘parasympathetic nervous system’, ‘breast cancer’, ‘cancer
neuroscience’ and ‘cancer neurotherapy’. Key word equivalents and
variations in Chinese were also included for searches conducted in
CNKI. Inclusion criteria focused on articles addressing the
interaction between the ANS and breast cancer, with particular
attention to mechanistic studies, clinical observations and
translational research. Articles were screened for quality,
relevance and originality. Low-quality studies, articles with
limited relevance to the topic and duplicate entries were excluded
following appraisal. Low-quality studies were defined as those
exhibiting substantial methodological limitations, such as unclear
study design, insufficient sample size, incomplete data reporting
or inability to assess study validity, and were excluded along with
articles of limited relevance and duplicate records.
After this screening process, a total of 75
high-quality articles were selected to form the basis of the
present review. This curated selection ensured a balanced
representation of the literature, integrating foundational studies
and recent advancements to provide a holistic perspective on the
topic.
3. Impact of the nervous system on breast
cancer
Autonomic innervation of breast tissue
and its role in tumor development
Breast tissue primarily receives innervation from
sympathetic and sensory nerves originating from the chest wall,
whilst parasympathetic nerves indirectly influence breast
physiology through their effect on other organs, such as the
intestines and lungs (15). For
example, vagal signaling can regulate intestinal barrier integrity
and mucosal immune responses, thereby shaping gut microbial
metabolism; the gut microbiota (‘estrobolome’) may in turn
influence enterohepatic estrogen recycling and systemic immune
tone, which are both implicated in breast cancer biology (16). In addition, vagal activity can
modulate pulmonary inflammation through the cholinergic
anti-inflammatory pathway, and lung/systemic inflammatory states
have been shown to facilitate breast cancer lung metastatic
colonization and even awaken dormant disseminated tumor cells in
the lung (17). Under normal
conditions, these autonomic components maintain a dynamic
equilibrium, supporting both breast function and local
physiological processes. However, pathological conditions may
disrupt this balance, contributing to the development of breast
cancer. Pathological conditions such as chronic psychosocial stress
(sustained sympathetic activation/β-adrenergic signaling) and
obesity-associated chronic low-grade inflammation can disrupt
autonomic balance and thereby foster a tumor-promoting systemic
milieu (4-6).
A previous study has indicated that in patients with invasive
ductal carcinoma, >33% display neural fiber infiltration in the
breast tissue. Importantly, the density of these fibers is
positively associated with tumor aggressiveness (18). Sympathetic nerve activity has been
shown to promote metastasis of breast cancer to bone, but
β-blockers have been shown to inhibit such metastatic processes
(19,20).
Furthermore, sensory nerves serve a key role in
tumorigenesis and metastasis. The depletion of local sensory
neuromediators (such as substance P, calcitonin gene-related
peptide, vasoactive intestinal peptide and neurokinin A) changes
the phenotype of cancer cells within the primary tumor, encouraging
the proliferation of metastatic subsets. Activation of sensory
nerves using the transient receptor potential vanilloid 1 agonist
olvanil has been shown to markedly reduce lung and liver metastases
(21-23).
Therefore, the ANS is implicated in the
pathophysiology of breast cancer. This can be mediated by
regulating tumor behavior directly through neurotransmitters (such
as norepinephrine/epinephrine acting on β-adrenergic receptors or
acetylcholine acting on muscarinic/nicotinic receptors) (24). By contrast, indirectly this can be
achieved by skewing the immune-inflammatory tumor microenvironment
toward a pro-tumor state, such as that exhibited by β-adrenergic
signaling, which increases IL-6/IL-8 and VEGF levels to promote
M2-like tumor-associated macrophage polarization and regulatory
T-cell/myeloid-derived suppressor-cell expansion whilst diminishing
natural killer cell cytotoxicity (25,26).
All of the aforementioned processes contribute to facilitating
angiogenesis, invasion and immune evasion.
Additionally, breast cancer and its treatment
methods (including surgery, chemotherapy, radiotherapy and
endocrine therapy) can perturb the ANS, sustaining sympathetic
activation and attenuating vagal tone, thereby promoting a
feed-forward loop that aggravates the disease (27,28).
In accordance, patients with breast cancer frequently exhibit
autonomic dysfunction, operationalized as reduced heart rate
variability [HRV; for example, standard deviation of NN intervals
(SDNN) and root mean square of successive differences (RMSSD)],
together with an increased low frequency/high frequency (HF) ratio
(reflecting reduced HF power and a relative shift toward
sympathetic predominance), higher resting heart rate and blunted
baroreflex sensitivity (29).
These abnormalities are positively associated with clinical
severity, including greater tumor burden (for example, larger
size/advanced stage) and higher symptom load (for example, fatigue,
pain, sleep disturbance), and they are exacerbated during therapy
and linked to a higher risk/severity of treatment-related
toxicities (for example, cardiotoxicity).
Sympathetic nervous system and breast
cancer
Immunohistochemical analysis of the sympathetic
neuronal marker tyrosine hydroxylase (TH) has demonstrated that
TH-positive sympathetic nerve fibers can innervate the breast
cancer tumor microenvironment (20,30).
Overactivation of the sympathetic nervous system has been
demonstrated to promote breast cancer growth and metastasis,
whereas a higher density of sympathetic nerves within cancerous
tissues is associated with poorer clinical outcomes in patients
with breast cancer (20,30). Sympathetic signaling primarily
functions through the release of norepinephrine by peripheral
sympathetic nerve endings or systemic release from the adrenal
medulla, directly impacting the development and progression of
breast cancer (31-33).
Norepinephrine and adrenaline act by binding to adrenergic
receptors (ARs), present in breast cancer cell lines and patient
tumor samples (32). Liu et
al (34) previously showed
that β2-AR expression is elevated in several HER2-overexpressing
breast cancer subtypes, and that higher β2-AR levels are associated
with lymph node metastasis and poorer prognosis in patients with
HER2-positive breast cancer.
Similarly, Kurozumi et al (35) found that high β2-AR levels are
associated with poor prognosis in patients with estrogen
receptor-positive breast cancer, accompanied by low
tumor-infiltrating lymphocyte grades and lower expression levels of
programmed death ligand 1. In addition, high expression of α2-AR
has been shown to further breast cancer progression. Previous
studies suggest that selective activation of α2-adrenergic
receptors by dexmedetomidine can significantly enhance the
proliferation, migration and invasion of the breast cancer cell
line MCF-7 in vitro, potentially via activation of the
α2-AR/STAT3/ERK signaling pathway (36-39).
By contrast, administration of tramadol has been observed to
markedly inhibit the α2-AR/ERK signaling pathway, thereby
suppressing the proliferation, invasion and migration of breast
cancer cells (40). Therefore,
antagonizing the β2-AR or α2-AR pathways may represent potential
targets for the prevention and treatment of breast cancer.
Distant metastasis represents the predominant
pattern of breast-cancer recurrence and is the principal cause of
disease-specific mortality (41).
Clinical evidence shows that nerve fibers are detected in 15% of
lymph node-negative tumors compared with 28% of lymph node-positive
tumors, indicating a substantially higher prevalence of
tumor-associated nerves in metastatic disease. This enrichment in
lymph node-positive cases supports an association between increased
nerve presence/density and lymph node involvement, rather than a
random distribution of nerve fibers (18). Initial research suggested that
activation of the sympathetic nervous system serves a key role in
the metastasis of breast cancer to distant sites, such as the lymph
nodes and lungs, primarily mediated by β-ARs. This activation
promotes the infiltration of CD11b(+) and F4/80(+) macrophages into
the primary tumor mass, leading to the upregulation of metastatic
genes associated with M2 macrophage differentiation, such as ARG1,
CD163, MRC1 (CD206), IL10, TGFB1 and VEGFA, thereby fostering a
pro-metastatic tumor microenvironment (20). Furthermore, the β-blocker
propranolol has been shown to inhibit sympathetic activation,
restrict macrophage infiltration and thereby reduce the
dissemination of breast cancer cells to distant tissues (20).
In clinical practice, β-adrenergic receptor blockers
are commonly used to inhibit sympathetic nervous system activation.
Numerous studies have shown that patients with breast cancer
treated with β-blockers exhibit markedly lower mortality rates
compared with those who do not use these medications (42-49).
Another meta-analysis involving patients with breast cancer
receiving cardiovascular drugs revealed that β-blockers can
substantially reduce both recurrence and mortality rates associated
with breast cancer (49).
Additionally, recent findings from a retrospective cohort study
conducted in Sweden by Strell et al (50) suggested that β-blockers may provide
a protective effect against invasive breast cancer, with cumulative
exposure associated with a dose-dependent decrease in breast cancer
risk (50).
Hiller et al (51) previously conducted a triple-blind,
placebo-controlled clinical trial, which demonstrated that
preoperative administration of β-blockers markedly reduced the
expression of pro-tumorigenic intratumoral stromal genes, including
IL6, CXCL8 (IL8), VEGFA, PTGS2 (COX-2) and MMP9, in early operable
breast cancer, while concomitantly enhancing immune cell
infiltration, such as macrophages and CD8⁺ T cells (51). By contrast, a cohort study
involving nearly 200,000 patients with breast cancer across Europe
conducted by Cardwell et al (52) revealed no association between the
use of propranolol before and after breast cancer diagnosis and
breast cancer-specific or all-cause mortality. Similar
ineffectiveness was observed with non-selective β-blockers,
indicating that neither propranolol nor non-selective β-blockers
are able to improve survival rates (52). Additionally, other meta-analyses
have shown no statistically significant association between
β-blocker use and breast cancer mortality [19 studies; hazard ratio
(HR), 0.90; 95% CI, 0.78-1.04] or recurrence (16 studies; HR, 0.87;
95% CI, 0.71-1.08) (53-56).
These findings underscore the need for future research,
particularly large-scale prospective randomized controlled trials,
to provide robust clinical evidence regarding the efficacy of
β-blockers in the prevention and treatment of breast cancer.
Overall, sympathetic activation, principally via β-
and α2-adrenergic signaling can drive pro-metastatic,
immunomodulatory programs in breast cancer and is associated with
adverse outcomes. Although preclinical data and a number of
observational studies are supportive, current clinical evidence
remains heterogeneous, since β-blockers are not indicated for
anticancer use outside of trials (53-56).
Adequately powered randomized controlled trials with standardized
autonomic phenotyping are needed.
Vagus nervous system and breast
cancer
Although studies have previously documented
promoting effects of the sympathetic nervous system on breast
cancer, the role of the vagus nervous system remains ambiguous.
Notable insights emerged in 2008 when a study demonstrated that
severing the cervical vagus nerve in a breast cancer mouse model
promoted the distant metastasis of tumor cells to the lungs, liver,
heart and kidneys (57).
Additionally, vagotomy was found to decrease adrenal metastasis and
reverse tumor-induced adrenal functional changes in the same model
(58). Immunohistochemical
analysis using the vesicular acetylcholine transporter (VAChT), a
marker for vagal neurons, revealed that VAChT-positive vagus nerve
fibers tend to innervate the tumor microenvironment in breast
cancer (59). However, vagal
fibers appear to exert a tumor-suppressive (inhibitory) influence
on breast-cancer progression compared with sympathetic fibers
(57,59). Consistent with this concept,
reduced intratumoral vagal fiber density and where measured, lower
vagal tone/HRV, has been associated with poorer clinical outcomes,
whereas higher vagal input has been associated with more favorable
prognosis in observational cohorts (60-62).
These associations warrant further investigation with the
standardized quantification of neural elements and prospective
clinical phenotyping.
It has been previously established that activation
of the vagus nerve can trigger the classic cholinergic
anti-inflammatory pathway, effectively suppressing inflammatory
immune responses (63-65).
This mechanism may underpin the vagus nerve's antitumor effects.
Inflammation is intricately associated with cancer progression,
since macrophages and various pro-inflammatory factors (such as
TNF-α, IL-6, IL-1β, CXCL8 and CCL2) within the tumor
microenvironment can stimulate tumor cell proliferation,
angiogenesis, invasion and metastasis (66,67).
Consequently, reducing macrophage activity and pro-inflammatory
factor levels may diminish tumor growth and metastasis (68,69).
Previous studies have increasingly associated higher vagal activity
with improved long-term survival rates in patients with breast
cancer, suggesting that vagal regulation of immune functions may
also restrict tumor occurrence and progression (60-62).
Clinical evidence has indicated that increased vagal
tone in patients with advanced breast cancer is positively
associated with long-term survival (60,61).
Furthermore, vagal activation may inhibit distant metastasis of
breast cancer by elevating substance P levels, subsequently
reducing inflammatory cytokine levels (70). Substance P is a neuropeptide found
in the afferent fibers of the vagus nerve that notably influences
immune-inflammatory regulation, thereby impeding tumor development.
Previous studies have suggested that vagus nerve stimulation (VNS)
can mitigate the development and distant metastasis of breast
cancer by correcting the cardiac autonomic imbalance and lessening
the cardiotoxicity induced by chemotherapy agents, such as
doxorubicin (71-73).
This dual therapeutic potential of VNS, which includes reducing
chemotherapy-induced cardiotoxicity, indicates its potential for
cancer treatment. VNS is already being utilized clinically for
managing epilepsy, depression and strokes (74-76),
with both preclinical and clinical studies preliminarily reporting
that vagal regulation can be effectively applied in preventing and
treating central nervous system disorders, autoimmune diseases and
cardiovascular diseases (77-79).
However, further investigations are required to explore the safety
and efficacy of VNS in treating breast cancer, thereby providing
novel insights and strategies for clinical application. Overall,
the vagus nerve appears to be tumor suppressive and
anti-inflammatory in breast cancer, since reduced vagal tone/fiber
density is associated with worse outcomes. Given the controversial
preclinical findings and observational human data, non-invasive VNS
may be confined to clinical studies, with future trials integrating
autonomic phenotyping, mechanistic readouts and cardio-oncology
endpoints.
4. Application of autonomic nervous function
monitoring in breast cancer
HRV, which measures minute fluctuations in heart
rate (R-R intervals), is the most widely used indicator for
monitoring the activity of sympathetic and vagus nerves in clinical
settings at present (80).
Time-domain and frequency-domain analyses of HRV can be used to
reflect the dynamic balance of the ANS, encompassing both the
sympathetic and vagus nerve systems (80,81).
Arab et al (82) found that
the overall standard deviation of HRV (SDNN) and the RMSSD are
inversely associated with patient staging in patients with breast
cancer. Notably, a lower SDNN is associated with a poorer long-term
prognosis in patients with advanced breast cancer (82).
In addition, previous studies have identified
impairments in autonomic function, such as reduced HRV, diminished
aerobic adaptability, altered metabolic indicators and increased
fatigue, as markers of clinical phenotype in breast cancer
survivors (60-62,83).
Previous clinical studies have demonstrated that the overall
autonomic function in breast cancer survivors is compromised and
closely associated with tumor staging, compared with patients with
early-stage breast cancer, since those with advanced disease
demonstrate decreased vagal tone and heightened sympathetic
activity (84-86).
A meta-analysis that included 12 studies evaluated the impact of
autonomic nervous function on staging, treatment efficacy and
long-term prognosis in patients with breast cancer by analyzing
their HRV. It was then revealed that higher HRV parameters were
closely associated with improved long-term survival rates (62). HRV was also found to be associated
with common effects caused by breast cancer, such as fatigue,
depression and stress. In clinical practice, HRV assessment can
assist in evaluating the side effects of chemotherapy in patients
with breast cancer (28).
Therefore, HRV can serve as a supplementary,
non-invasive tool for the early diagnosis of autonomic dysfunction
in patients with breast cancer and assist in assessing their
long-term survival rates. In addition, the levels of certain
neurohormones in bodily fluids, such as adrenaline, noradrenaline
and cortisol, can also indicate autonomic activity. A previous
study has indicated that in patients with breast cancer, elevated
plasma levels of adrenaline, adrenocorticotropic hormone,
noradrenaline and cortisol are positively associated with symptoms
of pain, depression and fatigue (87). These findings suggest that
monitoring serum neurohormone levels may serve as a novel biomarker
for assessing prognosis in patients with breast cancer.
5. ANS regulation strategies for preventing
and treating breast cancer
Overview of ANS modulation
strategies
ANS serves a key role in both preventing and
treating breast cancer by directly or indirectly regulating the
tumor microenvironment. Developing strategies to modulate the ANS
for the prevention and treatment of breast cancer whilst
translating these neuroregulatory concepts into clinical practice
represent notable future directions for breast cancer management.
This section will now outline the main methods of modulating the
ANS.
Pharmacological approaches
A number of the most extensively investigated drugs
are β-adrenergic receptor blockers. Numerous clinical studies have
previously demonstrated that β-blockers can markedly reduce the
recurrence and mortality rates of breast cancer (48,88-92).
However, retrospective designs are vulnerable to confounding by
indication and comorbidities, immortal-time bias (a ‘guaranteed’
event-free interval before exposure classification that can
spuriously favor the exposed group) and time-window biases,
exposure misclassification and variable adjustment for concomitant
therapies. In addition, effect estimates differed by tumor subtype
(estrogen receptor, progesterone receptor or HER2), stage, timing
of exposure (perioperative, adjuvant or chronic) and by agent class
(β1-selective vs. non-selective), with meta-analyses reaching
inconsistent conclusions (48,54,58).
Although several pilot perioperative trials (typically enrolling
20-100 participants) have reported modulation of stress-adrenergic
and inflammatory pathways, these studies were not powered to detect
differences in clinical endpoints such as disease-free or overall
survival (91,92). Taken together, definitive survival
benefit has not been established. Accordingly, β-blockers should be
used for cardiovascular indications only and oncologic use should
be limited to clinical trials that incorporate standardized
autonomic phenotyping and prespecified survival endpoints.
Vagus nerve intervention
One of the most researched surgical procedures for
this strategy is vagotomy. Previous studies have shown that
cervical vagus nerve resection can inhibit the growth and distant
metastasis of breast cancer to some extent (57,58).
However, this approach causes irreversible nerve damage, resulting
in loss of the inherent physiological regulatory and protective
function of the nerves. It also lacks tumor tissue specificity and
selective nerve fiber type resection, limiting its clinical
translation. Xie et al (93) demonstrated that auricular VNS can
regulate the local parasympathetic innervation of tumors, promoting
a shift in the tumor immune microenvironment from inflammatory to
cytotoxic, thereby enhancing antitumor immune responses. This
approach is synergistic with the antitumor effects of doxorubicin,
slowing tumor tissue growth, reducing local infiltration of breast
cancer and inhibiting distant pulmonary metastasis and alveolar
infiltration.
Genetic neuro-manipulation techniques
based on adenoviral vectors
These techniques address the inherent limitations in
pharmacological, surgical and electrical neural stimulation
methods. By targeting specific nerve fibers for gene delivery,
these methods facilitate precise and reversible manipulation of
nerve fiber types, thereby enabling diverse forms of neural
regulation, including stimulation, inhibition or denervation
(94-96).
Viral-vector approaches (most commonly AAV) have already been
applied in breast-cancer mouse models to enable circuit-specific
chemogenetic/optogenetic neuromodulation that reshapes intratumoral
autonomic activity and tumor growth (97,98).
The application of these neuroregulatory strategies across
different tumor types may represent a novel approach for cancer
treatment. However, the use of adenoviral vectors raises potential
biosafety concerns. Consequently, additional research is imperative
to verify the safety and efficacy of these techniques.
6. Conclusions
The accumulated evidence indicates that autonomic
imbalance, characterized by sympathetic overactivation and reduced
vagal tone, interfaces with breast-cancer biology in ways that are
clinically observable and potentially targetable. For current
practice, the present review recommends the following: i) Where
feasible, incorporating simple autonomic phenotyping (resting heart
rate and HRV indices such as SDNN and RMSSD) into baseline
assessment and survivorship follow-up to flag patients at higher
risk of symptom burden or treatment toxicity; ii) optimizing
perioperative and on-treatment stress, anxiety, sleep and pain
management, alongside exercise and cardiometabolic risk control, to
mitigate excessive sympathetic drive; iii) using β-blockers only
for established cardiovascular indications and not for anticancer
intent outside a trial, whilst considering clinical-trial
enrollment when ANS-modulating strategies are contemplated; iv)
coordinating early with cardio-oncology (especially for
anthracycline-based regimens) to individualize cardioprotection in
patients with autonomic imbalance; and v) reserving non-invasive
VNS and other neuromodulatory interventions for ethically approved
investigations rather than routine care. Investigational
approaches, such as adenoviral vector-based local
neuro-manipulation, remain preclinical and are not recommended for
clinical use at this time. Priority research should include
adequately powered randomized trials that incorporate standardized
ANS phenotyping, perioperative and adjuvant β-adrenergic modulation
strategies and non-invasive vagal neuromodulation, with metastasis,
survival, toxicity and quality-of-life endpoints, alongside
harmonized methods for quantifying neural elements in tumor
tissue.
Acknowledgements
Not applicable.
Funding
Funding: The present review was supported by the Foundation for
Innovative Research of the Third People's Hospital of Honghe
Prefecture (grant no. HHSYKYLX14-2022-03-10-21).
Availability of data and materials
Not applicable.
Authors' contributions
MW and XC conceptualized the review framework and
supervised revision of the final manuscript. MW was also
responsible for drafting the manuscript. ZZ and LZ were primarily
responsible for the literature search and data analysis pertaining
to the role of the autonomic nervous system in breast cancer. SL,
QC, CG and SZ contributed to drafting and revising the content
critically for important intellectual content. XC also coordinated
the team efforts and integrated feedback from all authors. All
authors read and approved the final version of the manuscript for
publication. Data authentication not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2024.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Kunkler IH, Williams LJ, Jack WJL, Cameron
DA and Dixon JM: Breast-conserving surgery with or without
irradiation in early breast cancer. N Engl J Med. 388:585–594.
2023.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Wehrwein EA, Orer HS and Barman SM:
Overview of the anatomy, physiology, and pharmacology of the
autonomic nervous system. Compr Physiol. 6:1239–1278.
2016.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Hu J, Chen W, Shen L, Chen Z and Huang J:
Crosstalk between the peripheral nervous system and breast cancer
influences tumor progression. Biochim Biophys Acta Rev Cancer.
1877(188828)2022.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Cui Q, Jiang D, Zhang Y and Chen C: The
tumor-nerve circuit in breast cancer. Cancer Metastasis Rev.
42:543–574. 2023.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Faulkner S, Jobling P, March B, Jiang CC
and Hondermarck H: Tumor neurobiology and the war of nerves in
cancer. Cancer Discov. 9:702–710. 2019.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Silverman DA, Martinez VK, Dougherty PM,
Myers JN, Calin GA and Amit M: Cancer-associated neurogenesis and
nerve-cancer cross-talk. Cancer Res. 81:1431–1440. 2021.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Monje M, Borniger JC, D'silva NJ, Deneen
B, Dirks PB, Fattahi F, Frenette PS, Garzia L, Gutmann DH, Hanahan
D, et al: Roadmap for the emerging field of cancer neuroscience.
Cell. 181:219–222. 2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Mancusi R and Monje M: The neuroscience of
cancer. Nature. 618:467–479. 2023.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Yaman I, Agac Cobanoglu D, Xie T, Ye Y and
Amit M: Advances in understanding cancer-associated neurogenesis
and its implications on the neuroimmune axis in cancer. Pharmacol
Ther. 239(108199)2022.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Shi DD, Guo JA, Hoffman HI, Su J,
Mino-Kenudson M, Barth JL, Schenkel JM, Loeffler JS, Shih HA, Hong
TS, et al: Therapeutic avenues for cancer neuroscience:
Translational frontiers and clinical opportunities. Lancet Oncol.
23:e62–e74. 2022.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Li RQ, Zhao XH, Zhu Q, Liu T, Hondermarck
H, Thorne RF, Zhang XD and Gao JN: Exploring neurotransmitters and
their receptors for breast cancer prevention and treatment.
Theranostics. 13:1109–1129. 2023.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Huang Y, Xiang B, Manyande A, Xiang H and
Xiong J: Neurogenesis manifestations of solid tumor and tracer
imaging studies: A narrative review. Am J Cancer Res. 13:713–726.
2023.PubMed/NCBI
|
|
14
|
Cole SW, Nagaraja AS, Lutgendorf SK, Green
PA and Sood AK: Sympathetic nervous system regulation of the tumour
microenvironment. Nat Rev Cancer. 15:563–572. 2015.PubMed/NCBI View
Article : Google Scholar
|
|
15
|
Sarhadi NS, Shaw-Dunn J and Soutar DS:
Nerve supply of the breast with special reference to the nipple and
areola: Sir Astley Cooper revisited. Clin Anat. 10:283–288.
1997.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Bonaz B: Anti-inflammatory effects of
vagal nerve stimulation with a special attention to intestinal
barrier dysfunction. Neurogastroenterol Motil.
34(e14456)2022.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Kox M, Vaneker M, van der Hoeven JG,
Scheffer GJ, Hoedemaekers CW and Pickkers P: Effects of vagus nerve
stimulation and vagotomy on systemic and pulmonary inflammation in
a two-hit model in rats. PLoS One. 7(e34431)2012.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Pundavela J, Roselli S, Faulkner S, Attia
J, Scott RJ, Thorne RF, Forbes JF, Bradshaw RA, Walker MM, Jobling
P and Hondermarck H: Nerve fibers infiltrate the tumor
microenvironment and are associated with nerve growth factor
production and lymph node invasion in breast cancer. Mol Oncol.
9:1626–1635. 2015.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Campbell JP, Karolak MR, Ma Y, Perrien DS,
Masood-Campbell SK, Penner NL, Munoz SA, Zijlstra A, Yang X,
Sterling JA and Elefteriou F: Stimulation of host bone marrow
stromal cells by sympathetic nerves promotes breast cancer bone
metastasis in mice. PLoS Biol. 10(e1001363)2012.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Sloan EK, Priceman SJ, Cox BF, Yu S,
Pimentel MA, Tangkanangnukul V, Arevalo JM, Morizono K, Karanikolas
BD, Wu L, et al: The sympathetic nervous system induces a
metastatic switch in primary breast cancer. Cancer Res.
70:7042–7052. 2010.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Erin N: Role of sensory neurons,
neuroimmune pathways, and transient receptor potential vanilloid 1
(TRPV1) channels in a murine model of breast cancer metastasis.
Cancer Immunol Immunother. 69:307–314. 2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Erin N, Akman M, Aliyev E, Tanrıöver G and
Korcum AF: Olvanil activates sensory nerve fibers, increases T cell
response and decreases metastasis of breast carcinoma. Life Sci.
291(120305)2022.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Erin N and Szallasi A: Carcinogenesis and
metastasis: Focus on TRPV1-positive neurons and immune cells.
Biomolecules. 13(983)2023.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Chang A, Le CP, Walker AK, Creed SJ, Pon
CK, Albold S, Carroll D, Halls ML, Lane JR, Riedel B, et al:
β2-Adrenoceptors on tumor cells play a critical role in
stress-enhanced metastasis in a mouse model of breast cancer. Brain
Behav Immun. 57:106–115. 2016.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Qiao G, Chen M, Bucsek MJ, Repasky EA and
Hylander BL: Adrenergic signaling: A targetable checkpoint limiting
development of the antitumor immune response. Front Immunol.
9(164)2018.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Qin JF, Jin FJ, Li N, Guan HT, Lan L, Ni H
and Wang Y: Adrenergic receptor β2 activation by stress promotes
breast cancer progression through macrophages M2 polarization in
tumor microenvironment. BMB Rep. 48:295–300. 2015.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Scott JM, Jones LW, Hornsby WE, Koelwyn
GJ, Khouri MG, Joy AA, Douglas PS and Lakoski SG: Cancer
therapy-induced autonomic dysfunction in early breast cancer:
Implications for aerobic exercise training. Int J Cardiol.
171:e50–e501. 2014.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Arab C, Dias DP, Barbosa RT, Carvalho TD,
Valenti VE, Crocetta TB, Ferreira M, Abreu LC and Ferreira C: Heart
rate variability measure in breast cancer patients and survivors: A
systematic review. Psychoneuroendocrinology. 68:57–68.
2016.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Winkler F, Venkatesh HS, Amit M, Batchelor
T, Demir IE, Deneen B, Gutmann DH, Hervey-Jumper S, Kuner T,
Mabbott D, et al: Cancer neuroscience: State of the field, emerging
directions. Cell. 186:1689–1707. 2023.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Li D, Hu LN, Zheng SM, La T, Wei LY, Zhang
XJ, Zhang ZH, Xing J, Wang L, Li RQ, et al: High nerve density in
breast cancer is associated with poor patient outcome. FASEB
Bioadv. 4:391–401. 2022.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Szpunar MJ, Belcher EK, Dawes RP and
Madden KS: Sympathetic innervation, norepinephrine content, and
norepinephrine turnover in orthotopic and spontaneous models of
breast cancer. Brain Behav Immun. 53:223–233. 2016.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Conceicao F, Sousa DM, Paredes J and
Lamghari M: Sympathetic activity in breast cancer and metastasis:
Partners in crime. Bone Res. 9(9)2021.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Jayachandran P, Battaglin F, Strelez C,
Lenz A, Algaze S, Soni S, Lo JH, Yang Y, Millstein J, Zhang W, et
al: Breast cancer and neurotransmitters: Emerging insights on
mechanisms and therapeutic directions. Oncogene. 42:627–637.
2023.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Liu D, Deng Q, Sun L, Wang T, Yang Z, Chen
H, Guo L, Liu Y, Ma Y, Guo N and Shi M: A Her2-let-7-β2-AR circuit
affects prognosis in patients with Her2-positive breast cancer. BMC
Cancer. 15(832)2015.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Kurozumi S, Kaira K, Matsumoto H, Hirakata
T, Yokobori T, Inoue K, Horiguchi J, Katayama A, Koshi H, Shimizu
A, et al: β2-Adrenergic receptor expression is
associated with biomarkers of tumor immunity and predicts poor
prognosis in estrogen receptor-negative breast cancer. Breast
Cancer Res Treat. 177:603–610. 2019.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Liu Y, Sun J, Wu T, Lu X, Du Y, Duan H, Yu
W, Su D, Lu J and Tian J: Effects of serum from breast cancer
surgery patients receiving perioperative dexmedetomidine on breast
cancer cell malignancy: A prospective randomized controlled trial.
Cancer Med. 8:7603–7612. 2019.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Chi M, Shi X, Huo X, Wu X, Zhang P and
Wang G: Dexmedetomidine promotes breast cancer cell migration
through Rab11-mediated secretion of exosomal TMPRSS2. Ann Transl
Med. 8(531)2020.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Xia M, Ji NN, Duan ML, Tong JH, Xu JG,
Zhang YM and Wang SH: Dexmedetomidine regulate the malignancy of
breast cancer cells by activating α2-adrenoceptor/ERK signaling
pathway. Eur Rev Med Pharmacol Sci. 20:3500–3506. 2016.PubMed/NCBI
|
|
39
|
Carnet Le Provost K, Kepp O, Kroemer G and
Bezu L: Trial watch: Dexmedetomidine in cancer therapy.
Oncoimmunology,. 2024, 13: 2327143, 2024.
|
|
40
|
Xia M, Tong JH, Zhou ZQ, Duan ML, Xu JG,
Zeng HJ and Wang SH: Tramadol inhibits proliferation, migration and
invasion via α2-adrenoceptor signaling in breast cancer cells. Eur
Rev Med Pharmacol Sci. 20:157–165. 2016.PubMed/NCBI
|
|
41
|
Scully OJ, Bay BH, Yip G and Yu Y: Breast
cancer metastasis. Cancer Genomics Proteomics. 9:311–320.
2012.PubMed/NCBI
|
|
42
|
Barron TI, Connolly RM, Sharp L, Bennett K
and Visvanathan K: Beta blockers and breast cancer mortality: A
population-based study. J Clin Oncol. 29:2635–2644. 2011.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Phadke S and Clamon G: Beta blockade as
adjunctive breast cancer therapy: A review. Crit Rev Oncol Hematol.
138:173–177. 2019.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Wang T, Li Y, Lu HL, Meng QW, Cai L and
Chen XS: β-Adrenergic receptors : New target in breast cancer.
Asian Pac J Cancer Prev. 16:8031–8039. 2015.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Montoya A, Amaya CN, Belmont A, Diab N,
Trevino R, Villanueva G, Rains S, Sanchez LA, Badri N, Otoukesh S,
et al: Use of non-selective β-blockers is associated with decreased
tumor proliferative indices in early stage breast cancer.
Oncotarget. 8:6446–6460. 2017.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Montoya A, Varela-Ramirez A, Dickerson E,
Pasquier E, Torabi A, Aguilera R, Nahleh Z and Bryan B: The beta
adrenergic receptor antagonist propranolol alters mitogenic and
apoptotic signaling in late stage breast cancer. Biomed J.
42:155–165. 2019.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Zhao Y, Wang Q, Zhao X, Meng H and Yu J:
Effect of antihypertensive drugs on breast cancer risk in female
hypertensive patients: Evidence from observational studies. Clin
Exp Hypertens. 40:22–27. 2018.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Caparica R, Bruzzone M, Agostinetto E, De
Angelis C, Fêde Â, Ceppi M and de Azambuja E: Β-blockers in
early-stage breast cancer: A systematic review and meta-analysis.
ESMO Open. 6(100066)2021.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Raimondi S, Botteri E, Munzone E, Cipolla
C, Rotmensz N, DeCensi A and Gandini S: Use of β-blockers,
angiotensin-converting enzyme inhibitors and angiotensin receptor
blockers and breast cancer survival: Systematic review and
meta-analysis. Int J Cancer. 139:212–219. 2016.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Strell C, Smith DR, Valachis A, Woldeyesus
H, Wadsten C, Micke P, Fredriksson I and Schiza A: Use of
beta-blockers in patients with ductal carcinoma in situ and risk of
invasive breast cancer recurrence: A Swedish retrospective cohort
study. Breast Cancer Res Treat. 207:293–299. 2024.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Hiller JG, Cole SW, Crone EM, Byrne DJ,
Shackleford DM, Pang JB, Henderson MA, Nightingale SS, Ho KM, Myles
PS, et al: Preoperative β-Blockade with propranolol reduces
biomarkers of metastasis in breast cancer: A phase II Randomized
trial. Clin Cancer Res. 26:1803–1811. 2020.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Cardwell CR, Pottegard A, Vaes E, Garmo H,
Murray LJ, Brown C, Vissers PA, O'Rorke M, Visvanathan K,
Cronin-Fenton D, et al: Propranolol and survival from breast
cancer: A pooled analysis of European breast cancer cohorts. Breast
Cancer Res. 18(119)2016.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Scott OW, TinTin S, Cavadino A and Elwood
JM: Beta-blocker use and breast cancer outcomes: A meta-analysis.
Breast Cancer Res Treat. 206:443–463. 2024.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Li C, Li T, Tang R, Yuan S and Zhang W:
β-Blocker use is not associated with improved clinical outcomes in
women with breast cancer: A meta-analysis. Biosci Rep.
40(BSR20200721)2020.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Kim HY, Jung YJ, Lee SH, Jung HJ and Pak
K: Is beta-blocker use beneficial in breast cancer? A
meta-analysis. Oncology. 92:264–268. 2017.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Løfling LL, Støer NC, Sloan EK, Chang A,
Gandini S, Ursin G and Botteri E: β-blockers and breast cancer
survival by molecular subtypes: A population-based cohort study and
meta-analysis. Br J Cancer. 127:1086–1096. 2022.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Erin N, Akdas Barkan G, Harms JF and
Clawson GA: Vagotomy enhances experimental metastases of 4THMpc
breast cancer cells and alters substance P. level. Regul Pept.
151:35–42. 2008.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Erin N, Barkan GA and Clawson GA: Vagus
nerve regulates breast cancer metastasis to the adrenal gland.
Anticancer Res. 33:3675–3682. 2013.PubMed/NCBI
|
|
59
|
Han H, Yang C, Zhang Y, Han C and Zhang G:
Vascular endothelial growth factor mediates the sprouted
axonogenesis of breast cancer in rat. Am J Pathol. 191:515–526.
2021.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Giese-Davis J, Wilhelm FH, Tamagawa R,
Palesh O, Neri E, Taylor CB, Kraemer HC and Spiegel D: Higher vagal
activity as related to survival in patients with advanced breast
cancer: An analysis of autonomic dysregulation. Psychosom Med.
77:346–355. 2015.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Huang WB, Lai HZ, Long J, Ma Q, Fu X, You
FM and Xiao C: Vagal nerve activity and cancer prognosis: A
systematic review and meta-analysis. BMC Cancer.
25(579)2025.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Kloter E, Barrueto K, Klein SD, Scholkmann
F and Wolf U: Heart rate variability as a prognostic factor for
cancer survival-a systematic review. Front Physiol.
9(623)2018.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Borovikova LV, Ivanova S, Zhang M, Yang H,
Botchkina GI, Watkins LR, Wang H, Abumrad N, Eaton JW and Tracey
KJ: Vagus nerve stimulation attenuates the systemic inflammatory
response to endotoxin. Nature. 405:458–462. 2000.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Rosas-Ballina M, Olofsson PS, Ochani M,
Valdés-Ferrer SI, Levine YA, Reardon C, Tusche MW, Pavlov VA,
Andersson U, Chavan S, et al: Acetylcholine-synthesizing T cells
relay neural signals in a vagus nerve circuit. Science. 334:98–101.
2011.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Bonaz B, Picq C, Sinniger V, Mayol JF and
Clarençon D: Vagus nerve stimulation: from epilepsy to the
cholinergic anti-inflammatory pathway. Neurogastroenterol Motil.
25:208–221. 2013.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Mantovani A, Allavena P, Sica A and
Balkwill F: Cancer-related inflammation. Nature. 454:436–444.
2008.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Coussens LM and Werb Z: Inflammation and
cancer. Nature. 420:860–867. 2002.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Bogeska R, Mikecin AM, Kaschutnig P, Fawaz
M, Büchler-Schäff M, Le D, Ganuza M, Vollmer A, Paffenholz SV,
Asada N, et al: Inflammatory exposure drives long-lived impairment
of hematopoietic stem cell self-renewal activity and accelerated
aging. Cell Stem Cell. 29:1273–1284 e8. 2022.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Wu S, Xie S, Yuan C, Yang Z, Liu S, Zhang
Q, Sun F, Wu J, Zhan S, Zhu S and Zhang S: Inflammatory bowel
disease and long-term risk of cancer: A prospective cohort study
among half a million adults in UK Biobank. Inflamm Bowel Dis.
29:384–395. 2023.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Erin N, Duymus O, Ozturk S and Demir N:
Activation of vagus nerve by semapimod alters substance P. levels
and decreases breast cancer metastasis. Regul Pept. 179:101–108.
2012.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Lai Y, Zhou X, Guo F, Jin X, Meng G, Zhou
L, Chen H, Liu Z, Yu L and Jiang H: Non-invasive transcutaneous
vagus nerve stimulation improves myocardial performance in
doxorubicin-induced cardiotoxicity. Cardiovasc Res. 118:1821–1834.
2022.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Prathumsap N, Ongnok B, Khuanjing T,
Arinno A, Maneechote C, Apaijai N, Chunchai T, Arunsak B, Kerdphoo
S, Janjek S, et al: Vagus nerve stimulation exerts cardioprotection
against doxorubicin-induced cardiotoxicity through inhibition of
programmed cell death pathways. Cell Mol Life Sci.
80(21)2022.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Guo F, Wang Y, Wang J, Liu Z, Lai Y, Zhou
Z, Liu Z, Zhou Y, Xu X, Li Z, et al: Choline protects the heart
from doxorubicin-induced cardiotoxicity through vagal activation
and Nrf2/HO-1 pathway. Oxid Med Cell Longev.
2022(4740931)2022.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Panebianco M, Rigby A and Marson AG: Vagus
nerve stimulation for focal seizures. Cochrane Database Syst Rev.
7(CD002896)2022.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Marwaha S, Palmer E, Suppes T, Cons E,
Young AH and Upthegrove R: Novel and emerging treatments for major
depression. Lancet. 401:141–153. 2023.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Liu YL, Wang SR, Ma JX, Yu LH and Jia GW:
Vagus nerve stimulation is a potential treatment for ischemic
stroke. Neural Regen Res. 18:825–831. 2023.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Johnson RL and Wilson CG: A review of
vagus nerve stimulation as a therapeutic intervention. J Inflamm
Res. 11:203–213. 2018.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Capilupi MJ, Kerath SM and Becker LB:
Vagus nerve stimulation and the cardiovascular system. Cold Spring
Harb Perspect Med. 10(a034173)2020.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Liu L, Lou S, Fu D, Ji P, Xia P, Shuang S,
Dong W, Yuan X, Wang J, Xie K, et al: Neuro-immune interactions:
Exploring the anti-inflammatory role of the vagus nerve. Int
Immunopharmacol. 159(114941)2025.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Santos-De-Araujo AD, Shida-Marinho R and
Pontes-Silva A: Heart rate variability (HRV): Checklist for
observational and experimental studies. Autoimmun Rev.
21(103190)2022.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Gregoire JM, Gilon C, Carlier S and
Bersini H: Autonomic nervous system assessment using heart rate
variability. Acta Cardiol. 78:648–662. 2023.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Arab C, Vanderlei LCM, Da Silva Paiva L,
Fulghum KL, Fristachi CE, Nazario ACP, Elias S, Gebrim LH, Ferreira
Filho C, Gidron Y and Ferreira C: Cardiac autonomic modulation
impairments in advanced breast cancer patients. Clin Res Cardiol.
107:924–936. 2018.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Vigo C, Gatzemeier W, Sala R, Malacarne M,
Santoro A, Pagani M and Lucini D: Evidence of altered autonomic
cardiac regulation in breast cancer survivors. J Cancer Surviv.
9:699–706. 2015.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Wu S, Chen M, Wang J, Shi B and Zhou Y:
Association of short-term heart rate variability with breast tumor
stage. Front Physiol. 12(678428)2021.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Majerova K, Zvarik M, Ricon-Becker I,
Hanalis-Miller T, Mikolaskova I, Bella V, Mravec B and Hunakova L:
Increased sympathetic modulation in breast cancer survivors
determined by measurement of heart rate variability. Sci Rep.
12(14666)2022.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Caro-Morán E, Fernández-Lao C,
Galiano-Castillo N, Cantarero-Villanueva I, Arroyo-Morales M and
Díaz-Rodríguez L: Heart rate variability in breast cancer survivors
after the first year of treatments: A case-controlled study. Biol
Res Nurs. 18:43–49. 2016.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Thornton LM, Andersen BL and Blakely WP:
The pain, depression, and fatigue symptom cluster in advanced
breast cancer: covariation with the hypothalamic-pituitary-adrenal
axis and the sympathetic nervous system. Health Psychol.
29:333–337. 2010.PubMed/NCBI View Article : Google Scholar
|
|
88
|
Childers WK, Hollenbeak CS and Cheriyath
P: β-blockers reduce breast cancer recurrence and breast cancer
death: A meta-analysis. Clin Breast Cancer. 15:426–431.
2015.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Lewinter C, Nielsen TH, Edfors LR, Linde
C, Bland JM, LeWinter M, Cleland JGF, Køber L, Braunschweig F and
Mansson-Broberg A: A systematic review and meta-analysis of
beta-blockers and renin-angiotensin system inhibitors for
preventing left ventricular dysfunction due to anthracyclines or
trastuzumab in patients with breast cancer. Eur Heart J.
43:2562–2569. 2022.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Mravec B: Βeta-blockers and breast
cancer-letter. Cancer Epidemiol Biomarkers Prev.
30(1765)2021.PubMed/NCBI View Article : Google Scholar
|
|
91
|
Shaashua L, Shabat-Simon M, Haldar R,
Matzner P, Zmora O, Shabtai M, Sharon E, Allweis T, Barshack I,
Hayman L, et al: Perioperative COX-2 and β-Adrenergic blockade
improves metastatic biomarkers in breast cancer patients in a
phase-II Randomized trial. Clin Cancer Res. 23:4651–4661.
2017.PubMed/NCBI View Article : Google Scholar
|
|
92
|
Haldar R, Shaashua L, Lavon H, Lyons YA,
Zmora O, Sharon E, Birnbaum Y, Allweis T, Sood AK, Barshack I, et
al: Perioperative inhibition of β-adrenergic and COX2 signaling in
a clinical trial in breast cancer patients improves tumor Ki-67
expression, serum cytokine levels, and PBMCs transcriptome. Brain
Behav Immun. 73:294–309. 2018.PubMed/NCBI View Article : Google Scholar
|
|
93
|
Xie M, Guo F, Song L, Tan W, Han X, Xu S,
Li X, Wang Y, Wang Y, Zhou L, et al: Noninvasive neuromodulation
protects against doxorubicin-induced cardiotoxicity and inhibits
tumor growth. iScience. 27(109163)2024.PubMed/NCBI View Article : Google Scholar
|
|
94
|
Nectow AR and Nestler EJ: Viral tools for
neuroscience. Nat Rev Neurosci. 21:669–681. 2020.PubMed/NCBI View Article : Google Scholar
|
|
95
|
Hui Y, Zheng X, Zhang H, Li F, Yu G, Li J,
Zhang J, Gong X and Guo G: Strategies for targeting neural
circuits: How to manipulate neurons using virus vehicles. Front
Neural Circuits. 16(882366)2022.PubMed/NCBI View Article : Google Scholar
|
|
96
|
Betley JN and Sternson SM:
Adeno-associated viral vectors for mapping, monitoring, and
manipulating neural circuits. Hum Gene Ther. 22:669–677.
2011.PubMed/NCBI View Article : Google Scholar
|
|
97
|
Wen HZ, Xiong SY, Lou YX, Yang SZ, Sun L,
Yi YL, Tang BQ, Shu Q, Wang ZQ, Yan XJ, et al: Social interaction
in mice suppresses breast cancer progression via a corticoamygdala
neural circuit. Neuron. 113:3374–3389.e9. 2025.PubMed/NCBI View Article : Google Scholar
|
|
98
|
Xiong SY, Wen HZ, Dai LM, Lou YX, Wang ZQ,
Yi YL, Yan XJ, Wu YR, Sun W, Chen PH, et al: A brain-tumor neural
circuit controls breast cancer progression in mice. J Clin Invest.
133(e167725)2023.PubMed/NCBI View Article : Google Scholar
|