Introduction
Small cell lung cancer (SCLC) is an aggressive
subtype of lung cancer, accounting for 10-15% of all lung cancer
cases (1). Compared with non-SCLC,
SCLC exhibits a more malignant biological behavior, characterized
by rapid proliferation and a high propensity for widespread distant
metastasis (2), and as a result,
patients are often diagnosed at an advanced stage. Although
targeted therapies and immunotherapies, such as poly (ADP-ribose)
polymerase (PARP) inhibitors and programmed cell death
protein-1/programmed death-ligand 1 (PD-1/PD-L1) inhibitors have
shown promising efficacy in certain patients with lung cancer in
previous years, chemotherapy remains the mainstay of treatment for
SCLC, especially in advanced-stage SCLC (3). However, the side effects of
chemotherapy are notable, with cardiotoxicity being a crucial
factor affecting long-term survival (4,5).
Chemotherapeutic agents, particularly anthracyclines
(such as doxorubicin) and platinum-based drugs (such as cisplatin),
can induce marked cardiotoxicity while treating tumors (6). This toxicity primarily manifests as
myocardial injury and left ventricular dysfunction, ultimately
leading to heart failure. Heart failure not only markedly impacts
the quality of life of patients but also increases the rates of
rehospitalization and mortality (7,8). One
study showed that SCLC patients with heart failure have an ~75%
higher risk of death compared with those without heart failure
(8). Therefore, early
identification of high-risk patients and predicting the occurrence
and severity of heart failure is of clinical importance (9).
Cardiac troponin I (cTnI) and cardiac troponin T
(cTnT) are currently the most commonly used cardiac biomarkers,
released into the bloodstream during myocardial cell injury
(10,11), with elevated levels of cTnI and
cTnT being widely used for the early diagnosis of myocardial
infarction and monitoring of cardiotoxicity. B-type natriuretic
peptide (BNP) and N-terminal pro-BNP (NT-proBNP) are another class
of biomarkers commonly used in the diagnosis of heart failure, with
elevated levels typically associated with increased ventricular
pressure load and wall stress (12). Additionally, left ventricular
ejection fraction (LVEF) is a crucial indicator of left ventricular
function that is commonly used to assess the severity of heart
failure, and a decrease in LVEF is a marker of impaired cardiac
systolic function and often indicates a poor prognosis (13).
However, the predictive ability of a single
biomarker may be insufficient to address the complex cardiotoxicity
that may arise after chemotherapy. Therefore, a comprehensive
assessment combining multiple cardiac biomarkers and functional
indicators may improve the accuracy of early detection and
prediction of the severity of heart failure (14,15).
Moreover, different tumor biological characteristics, treatment
regimens and individual patient differences may also influence the
risk of heart failure. Thus, a systematic analysis of these
potential influencing factors is necessary to identify independent
prognostic factors and develop an effective comprehensive
predictive model.
The present study aimed to evaluate various clinical
and laboratory indicators in patients with SCLC during the early
phase of chemotherapy, in order to predict the occurrence and
severity of heart failure. The goal was to provide a reliable risk
assessment tool for clinical practice, guide intervention
strategies and ultimately improve the long-term prognosis of
patients with SCLC.
Materials and methods
Study subjects and data
collection
The present study retrospectively included patients
diagnosed with SCLC receiving chemotherapy at Hebei Provincial
Hospital of Traditional Chinese Medicine (Shijiazhuang, China) from
January 2016 to January 2018. The inclusion criteria were as
follows: i) Patients diagnosed with SCLC through pathology; ii)
patients receiving standard chemotherapy of etoposide combined with
carboplatin, with the standard chemotherapy regimen dose and
treatment cycle recommended by the current National Comprehensive
Cancer Network guidelines for SCLC (16); and iii) patients with complete
follow-up data, including the occurrence and severity of heart
failure during the chemotherapy course and follow-up period.
Exclusion criteria included: i) Patients with a history of heart
failure or other serious cardiovascular diseases (such as
congenital heart disease and pulmonary hypertension); ii) patients
who were lost to follow-up during the follow-up process; and iii)
patients with severe mental illness or poor compliance (poor
treatment adherence), including those with schizophrenia, major
depression or bipolar disorder. It is noteworthy that the use of
specific medications was not employed as an exclusion criterion in
the present study. The patient screening process is shown in
Fig. S1. All data in the present
study were collected at the end of the first cycle of chemotherapy
and follow-up period.
Data collection included the following aspects: i)
Baseline demographic information: Age, sex, body mass index (BMI),
smoking history and alcohol consumption history; ii) tumor
characteristics: Clinical staging of the tumor, primary tumor
staging (according to the TNM classification) (17) and gene mutation status; iii)
chemotherapy-related information, such as the cumulative dose of
chemotherapy drugs received by the patients; iv) cardiac function
and biomarkers: LVEF measured by echocardiography (Philips iE33;
Philips Medical Systems B.V.), diastolic function assessed for
normalcy using echocardiography (Philips iE33), cTnI and cTnT
measured by immunoassay (ARCHITECT i2000SR; Abbott), BNP and
NT-proBNP measured by immunoassay (Roche Cobas e411; Roche
Diagnostics GmbH) and creatine kinase-myocardial band (CK-MB)
measured by enzymatic assay (AU5800; Beckman Coulter, Inc.). The
specific procedures for BNP and NT-proBNP were as follows: First,
peripheral venous blood was collected using
ethylenediaminetetraacetic acid (EDTA)-anticoagulated tubes, and
after centrifugation (2,000 x g, 10 min, 4˚C), plasma was
separated, labeled, registered and loaded onto the analyzer (Roche
Cobas e411; Roche Diagnostics GmbH). The samples were then loaded
onto an automated immunoassay analyzer (Roche Cobas e411; Roche
Diagnostics GmbH) for analysis, and the instrument automatically
completed the immunoreaction, signal detection and quantitative
determination. The collection method for CK-MB involved peripheral
venous blood sampling followed by centrifugation (2,000 x g, 10
min, room temperature) to obtain serum; the samples were
subsequently labeled, registered, and loaded onto the analyzer, and
CK-MB was measured on an automated biochemical analyzer (AU5800;
Beckman Coulter, Inc.), with the instrument automatically
completing the detection and calculating CK-MB levels. Heart
failure was monitored in all patients, who were followed up for 5
years. Throughout the follow-up period, heart failure occurrence
was recorded and graded according to the Common Terminology
Criteria for Adverse Events (CTCAE) classification (18) into three levels: Mild heart failure
(asymptomatic or mild), moderate-to-severe heart failure and
life-threatening heart failure or death. Mild heart failure
(asymptomatic or mild) includes CTCAE grade 1 (asymptomatic, with
only detected cardiac dysfunction) and grade 2 (symptomatic but
with minimal impact on daily activities, possibly requiring
medication). Moderate-to-severe heart failure includes CTCAE grade
3 (notable symptoms, impacting daily activities, requiring
hospitalization). Life-threatening heart failure or death includes
CTCAE grade 4 (life-threatening heart failure, usually requiring
emergency treatment or life support) and grade 5 (death related to
heart failure).
Treatment regimen
All patients received antiemetic therapy, gastric
protection and nutritional support during chemotherapy. The
standard triple antiemetic regimen was used to adequately prevent
both acute and delayed nausea and vomiting. Approximately 30 min
before the start of chemotherapy, ondansetron (8-16 mg, IV) was
administered once, together with dexamethasone 12 mg orally;
aprepitant 125 mg orally was given 1 h before chemotherapy. To
cover the risk of delayed nausea and vomiting, aprepitant 80 mg and
dexamethasone 8 mg were continued orally each morning on days 2-4
after chemotherapy. The same regimen was applied in each
chemotherapy cycle. Oral high-protein supplements providing ~15-25
g of protein per day were administered throughout the entire
chemotherapy cycle. On the first day of each treatment cycle,
carboplatin (Furen Pharmaceutical Group Co., Ltd.) was administered
at 200-400 mg/m2 via intravenous infusion once;
simultaneously, etoposide injection (Jiangsu Hengrui
Pharmaceuticals Co., Ltd.) at 120 mg/m2 was administered
via intravenous infusion on days 1-3, once daily. Each cycle lasted
21 days, the remaining days of the cycle served as a rest period,
with a total of 4-6 cycles followed.
Statistical analysis
All statistical analyses in this study were
performed using R software version 4.4.0(19). Continuous variables are presented
as median (minimum-maximum), and categorical variables are
presented as frequency (percentage). Fisher's exact test or χ² test
was used to analyze the association between categorical variables
(such as smoking history, alcohol consumption history and tumor
staging) and the occurrence and severity of heart failure. For
continuous variables (such as LVEF, cTnI and NT-proBNP), Unpaired
Student's t-test or Mann-Whitney U test was used to compare
differences between the heart failure group and the non-heart
failure group. One-way ANOVA or Kruskal-Wallis test was used to
analyze the expression differences of continuous variables among
the three heart failure groups (mild, moderate-to-severe and
life-threatening or death). No post-hoc tests were performed, and
the reported P-values represent the overall comparison among the
three groups. P<0.05 was considered to indicate a statistically
significant difference.
Multivariate logistic regression analysis.
Variables with P<0.05 from the aforementioned statistical
analysis were included in a multivariate logistic regression model
to identify independent prognostic factors associated with heart
failure. Logistic regression analysis was used to calculate the
odds ratio (OR) and 95% confidence interval for each variable.
Multinomial logistic regression analysis. A
multinomial logistic regression analysis was used to assess the
factors influencing the severity of heart failure. ‘No symptoms or
mild heart failure’ and ‘moderate-to-severe heart failure’ were set
as the reference categories. The regression coefficients, OR and
P-values for each independent variable were calculated.
Combined indicator construction and receiver
operating characteristic (ROC) curve analysis. Because
these two variables differ in their units and numerical ranges,
Z-score standardization was applied to transform them into
distributions with a mean of 0 and a standard deviation of 1,
allowing for direct comparison within the same model. After
standardizing cTnI and LVEF, a combined predictive index was
constructed using the formula Combined indicator=-LVEF_std +
cTnI_std (where ‘std’ indicates the Z-score standardized value of
the variable). The performance of cTnI and LVEF individually and
their combination in predicting the occurrence and severity of
heart failure was analyzed using ROC curves (AUC value, 95%
confidence interval, threshold, youden index, sensitivity and
specificity). In the analysis of heart failure severity, ‘no
symptoms or mild heart failure’ was compared with other heart
failure categories and ‘life-threatening heart failure or death’
was compared with the other two categories.
Results
Baseline characteristics
A total of 550 eligible patients with SCLC were
included in the present study, with a mean age of 47 years (ranging
from 19-75±14.2 years), and 51.09% of the patients were male
(n=281). The average BMI of the patients was 21.4 kg/m2,
with 36.73% having a history of smoking and 43.09% having a history
of alcohol consumption. Regarding tumor staging, 49.64% of the
patients were in stage I, 25.27% were in stage II, 16.91% in stage
III and 8.18% in stage IV. Analysis of the genetic mutation status
showed that 18.36% of the patients had TP53 mutations and 29.64%
had RB1 mutations. All patients received standard chemotherapy
regimens, with a mean cumulative chemotherapy dose of 354 (range,
114-595) mg/m2. Tumor staging (P<0.001) and primary
tumor staging (P<0.001) were significantly associated with the
occurrence of heart failure (Table
I). During the follow-up period, a total of 93 patients
(16.91%) developed heart failure; according to the CTCAE
classification, 68 patients (73.12%) experienced asymptomatic or
mild heart failure, 19 patients (20.43%) developed
moderate-to-severe heart failure and 6 patients (6.45%) experienced
life-threatening heart failure or death. The mean age of patients
who developed heart failure was 48±11.8 years, with 54.84% being
male, 27.96% having a history of smoking and 48.39% having a
history of alcohol consumption. The differences in tumor staging
(P<0.001) and primary tumor staging (P=0.008) among the groups
were statistically significant, suggesting that these factors may
be related to the severity of heart failure. Other variables such
as age, sex, BMI, smoking, alcohol consumption and genetic mutation
status did not show statistically significant differences among the
groups (Table II).
 | Table IBaseline data analysis of patients
with and without heart failure after chemotherapy. |
Table I
Baseline data analysis of patients
with and without heart failure after chemotherapy.
| | | Heart failure | |
|---|
| Characteristic | All patients
(n=550) | Yes (n=93) | No (n=457) | P-value |
|---|
| Age, years | 47 (19-75) | 48 (27-74) | 47 (19-75) | 0.138 |
| Sex, n (%) | | | | 0.495 |
|
Male | 281 (51.09) | 51 (54.84) | 230 (50.33) | |
|
Female | 269 (48.91) | 42 (45.16) | 227 (49.67) | |
| BMI,
kg/m2 | 21.4 (18.3-24.9) | 22 (18.4-24.9) | 21.4 (18.3-24.9) | 0.195 |
| Smoking, n (%) | | | | 0.059 |
|
Yes | 202 (36.73) | 26 (27.96) | 176 (38.51) | |
|
No | 348 (63.27) | 67 (72.04) | 281 (61.49) | |
| Alcohol
consumption, n (%) | | | | 0.301 |
|
Yes | 237 (43.09) | 45 (48.39) | 192 (42.01) | |
|
No | 313 (56.91) | 48 (51.61) | 265 (57.99) | |
| Tumor stage, n
(%) | | | | <0.001 |
|
I | 273 (49.64) | 39 (41.94) | 234 (51.2) | |
|
II | 139 (25.27) | 21 (22.58) | 118 (25.82) | |
|
III | 93 (16.91) | 12 (12.9) | 81 (17.72) | |
|
IV | 45 (8.18) | 21 (22.58) | 24 (5.25) | |
| Gene mutation
status, n (%) | | | | 0.169 |
|
TP53 | 101 (18.36) | 11 (11.83) | 90 (19.69) | |
|
RB1 | 163 (29.64) | 30 (32.26) | 133 (29.1) | |
|
WYC | 120 (21.82) | 26 (27.96) | 94 (20.57) | |
|
Others | 166 (30.18) | 26 (27.96) | 140 (30.63) | |
| Primary tumor
stage, n (%) | | | | <0.001 |
|
T1 | 217 (39.45) | 34 (36.56) | 183 (40.04) | |
|
T2 | 150 (27.27) | 32 (34.41) | 118 (25.82) | |
|
T3 | 117 (21.27) | 7 (7.53) | 110 (24.07) | |
|
T4 | 66(12) | 20 (21.51) | 46 (10.07) | |
 | Table IIBaseline data analysis of patients
with cardiac toxicity after chemotherapy. |
Table II
Baseline data analysis of patients
with cardiac toxicity after chemotherapy.
| | Degree of heart
failure | |
|---|
| Characteristic | All patients
n=93 | Asymptomatic or
mild symptomatic (n=68) | Moderate-to-severe
symptomatic (n=19) | Life-threatening or
death (n=6) | P-value |
|---|
| Age, years | 48 (27-74) | 45.5 (41-64) | 52 (38-71) | 52.5 (27-74) | 0.708 |
| Sex, n (%) | | | | | 0.676 |
|
Male | 51 (54.84) | 36 (48.53) | 12 (52.63) | 3(50) | |
|
Female | 42 (45.16) | 33 (51.47) | 7 (47.37) | 2(50) | |
| BMI,
kg/m2 | 22 (18.4-24.9) | 21.7
(18.5-24.8) | 21.6
(19.5-24.6) | 24 (18.4-24.9) | 0.304 |
| Smoking, n (%) | | | | | 0.068 |
|
Yes | 26 (27.96) | 15 (22.06) | 9 (47.37) | 2 (33.33) | |
|
No | 67 (72.04) | 53 (77.94) | 10 (52.63) | 4 (66.67) | |
| Alcohol
consumption, n (%) | | | | | 0.051 |
|
Yes | 45 (48.39) | 35 (51.47) | 10 (52.63) | 0 (0) | |
|
No | 48 (51.61) | 33 (48.53) | 9 (47.37) | 6(100) | |
| Tumor stage, n
(%) | | | | | <0.001 |
|
I | 39 (41.94) | 37 (54.41) | 2 (10.53) | 0 (0.00) | |
|
II | 21 (22.58) | 17 (25.00) | 4 (21.05) | 0 (0.00) | |
|
III | 12 (12.9) | 4 (5.88) | 6 (31.58) | 2 (33.33) | |
|
IV | 21 (22.58) | 10 (14.71) | 7 (36.84) | 4 (66.67) | |
| Gene mutation
status, n (%) | | | | | 0.512 |
|
TP53 | 11 (11.83) | 7 (10.29) | 3 (15.79) | 1 (16.67) | |
|
RB1 | 30 (32.26) | 20 (29.41) | 8 (42.11) | 2 (33.33) | |
|
WYC | 26 (27.96) | 20 (29.41) | 3 (15.79) | 3 (50.00) | |
|
Others | 26 (27.96) | 21 (30.88) | 5 (26.32) | 0 (0.00) | |
| Primary tumor
stage, n (%) | | | | | 0.008 |
|
T1 | 34 (36.56) | 30 (44.12) | 2 (10.53) | 2 (33.33) | |
|
T2 | 32 (34.41) | 18 (26.47) | 12 (63.16) | 2 (33.33) | |
|
T3 | 7 (7.53) | 4 (5.88) | 1 (5.26) | 2 (33.33) | |
|
T4 | 20 (21.51) | 16 (23.53) | 4 (21.05) | 0 (0) | |
Cardiac function and biomarkers
Among the 550 patients with SCLC, the cumulative
chemotherapy dose was significantly higher in the heart failure
group compared with the dose in the non-heart failure group (398
vs. 346 mg/m2; P=0.007), and the levels of cTnI and cTnT
were also significantly elevated in the heart failure group
compared with those in the non-heart failure group (0.06 vs. 0.05
ng/ml, P=0.038; 0.013 vs. 0.014 ng/ml, P=0.036, respectively).
Additionally, the LVEF was significantly reduced (38.0 vs. 43.6%;
P=0.004), the proportion of patients with abnormal diastolic
dysfunction was higher (39.78 vs. 28.01%; P=0.026) and CK-MB levels
were significantly elevated (7.63 vs. 6.96 ng/ml; P=0.017) in the
heart failure group. Conversely, BNP and NT-proBNP levels were
higher in the heart failure group but the differences did not reach
statistical significance. These results suggested that cumulative
chemotherapy dose, cTnI, cTnT, LVEF, diastolic function and CK-MB
potentially play important roles in predicting the risk of heart
failure after chemotherapy in patients with SCLC (Table III). Among the 93 patients with
heart failure, as the severity of heart failure increased, the
cumulative chemotherapy dose (P=0.040) and cTnI levels (P=0.023)
significantly increased, whereas LVEF significantly decreased
(P=0.027). Although cTnT levels were slightly higher in the most
severe heart failure group, the difference was not statistically
significant (P=0.254). Additionally, BNP and CK-MB levels were
significantly elevated in patients with more severe heart failure
(P=0.031 and P=0.020, respectively), and the proportion of patients
with abnormal diastolic dysfunction increased with the severity of
heart failure (P=0.013). However, this did not occur between the
moderate/severe and life-threatening groups. These results
indicated that cumulative chemotherapy dose, cTnI, BNP, LVEF,
diastolic function and CK-MB may be closely related to and are
potential indicators for predicting the severity of heart failure
(Table IV).
 | Table IIIComparison of clinical indicators
between patients with and without heart failure. |
Table III
Comparison of clinical indicators
between patients with and without heart failure.
| | | Heart failure | |
|---|
| Clinical
indicator | Reference
range | All patients
(n=550) | Yes (n=93) | No (n=457) | P-value |
|---|
| Cumulative dose,
mg/m² | - | 354 (114-595) | 398 (120-588) | 346 (114-595) | 0.007 |
| cTnI, ng/ml | <0.0400 | 0.0548
(0.0303-0.0799) | 0.0557
(0.0306-0.0788) | 0.05
(0.0303-0.0799) | 0.038 |
| cTnT, ng/ml | <0.0140 | 0.0135
(0.00801-0.0200) | 0.013
(0.00801-0.0200) | 0.014
(0.00826-0.0198) | 0.036 |
| BNP, pg/ml | <100 | 304 (81.1-546) | 319 (81.1-546) | 267 (81.8-546) | 0.242 |
| NT-proBNP,
pg/ml | <125 | 311 (101-499) | 296 (115-499) | 276 (101-493) | 0.098 |
| LVEF, % | 50.0-70.0 | 41.8
(25.1-52.6) | 38.0
(25.1-49.5) | 43.6
(37.3-52.6) | 0.004 |
| Diastolic function,
n (%) | - | | | | 0.026 |
|
Normal | | 385 (70.00) | 56 (60.22) | 329 (71.99) | |
|
Abnormal | | 165 (30.00) | 37 (39.78) | 128 (28.01) | |
| CK-MB, ng/ml | 0.00-5.00 | 7.04
(4.00-9.99) | 7.63
(4.01-9.9) | 6.96
(4.00-9.99) | 0.017 |
 | Table IVComparison of clinical indicators
among patients with different degrees of heart failure. |
Table IV
Comparison of clinical indicators
among patients with different degrees of heart failure.
| | | Degree of heart
failure | |
|---|
| Clinical
indicator | Reference
range | All patients
(n=93) | Asymptomatic or
mild symptomatic (n=68) | Moderate-to-severe
symptomatic (n=19) | Life-threatening or
death (n=6) | P-value |
|---|
| Cumulative dose,
mg/m² | - | 398 (120-588) | 282 (134-450) | 298 (200-426) | 362 (120-588) | 0.040 |
| cTnI, ng/ml | <0.0400 | 0.0557
(0.0306-0.0788) | 0.0469
(0.0323-0.0624) | 0.0502
(0.0311-0.0773) | 0.0608
(0.0306-0.0788) | 0.023 |
| cTnT, ng/ml | <0.0140 | 0.013
(0.00801-0.0199) | 0.013
(0.00894-0.0143) | 0.0132
(0.00821-0.0197) | 0.0144
(0.00801-0.0199) | 0.254 |
| BNP, pg/ml | <100 | 319 (81.1-546) | 178 (117-333) | 262 (81.1-546) | 354 (129-537) | 0.031 |
| NT-proBNP,
pg/ml | <125 | 296 (115-498) | 276 (138-408) | 341 (115-458) | 347 (81.1-546) | 0.413 |
| LVEF, % | 50.0-70.0 | 38.0
(25.1-49.5) | 39.9
(34.1-48.7) | 37.3
(30.4-49.5) | 34.4
(25.1-41.3) | 0.027 |
| Diastolic function,
n (%) | - | | | | | 0.013 |
|
Normal | | 56 (60.22) | 46 (68.66) | 7 (35.00) | 3 (50.00) | |
|
Abnormal | | 37 (39.78) | 21 (31.34) | 13 (65.00) | 3 (50.00) | |
| CK-MB, ng/ml | 0.00-5.00 | 7.63
(4.01-9.90) | 4.65
(4.01-6.14) | 7.05
(4.10-9.47) | 7.16
(4.04-9.90) | 0.020 |
Multivariate logistic regression
analysis
Based on the aforementioned factors that were
significantly associated with heart failure, further logistic
regression analysis showed that cumulative chemotherapy dose, cTnI,
BNP, LVEF and CK-MB were independent prognostic factors for heart
failure. Cumulative chemotherapy dose (OR=1.002; P=0.016), cTnI
(OR=12.953; P=0.032), BNP (OR=1.002; P=0.020) and CK-MB (OR=1.150;
P=0.041) were significantly associated with an increased risk of
heart failure, whereas low levels of LVEF (OR=0.929; P=0.012) were
significantly associated with a risk of heart failure (Table V).
 | Table VMultivariate logistic regression
analysis of factors affecting heart failure in patients with small
cell lung cancer undergoing chemotherapy. |
Table V
Multivariate logistic regression
analysis of factors affecting heart failure in patients with small
cell lung cancer undergoing chemotherapy.
| Indicator | B | SE | P-value | OR | CI lower | CI upper |
|---|
| Cumulative dose,
mg/m² | 0.002 | 0.001 | 0.016 | 1.002 | 1.000 | 1.004 |
| cTnI, ng/ml | 2.561 | 0.291 | 0.032 | 12.953 | 7.314 | 22.954 |
| BNP, pg/ml | 0.002 | 0.001 | 0.020 | 1.002 | 1.000 | 1.005 |
| LVEF, % | -0.073 | 0.029 | 0.012 | 0.929 | 0.878 | 0.984 |
| CK-MB, ng/ml | 0.140 | 0.068 | 0.041 | 1.150 | 1.006 | 1.315 |
Multinomial logistic regression
analysis
Based on the aforementioned factors influencing
significantly the severity of heart failure, further multinomial
logistic regression analysis was conducted. The results showed that
cumulative chemotherapy dose exhibited significant differences
between asymptomatic vs. moderate and life-threatening heart
failure (both P<0.010) but not between moderate and
life-threatening heart failure (P>0.050). cTnI exhibited
significant differences across all comparisons made between
different levels of heart failure severity (all P<0.001). BNP
also showed significant differences between asymptomatic and
life-threatening heart failure (P=0.014), while LVEF exhibited
significant differences between asymptomatic and life-threatening
and between moderate and life-threatening heart failure (P=0.012
and P=0.015, respectively). However, the difference in diastolic
function did not show statistical significance (P>0.050). CK-MB
showed a significant difference between asymptomatic and moderate
heart failure (P=0.031). These results indicated that cTnI and LVEF
may be key factors influencing the severity of heart failure
(Table VI).
 | Table VIFactors affecting the degree of heart
failure in patients with small cell lung cancer undergoing
chemotherapy. |
Table VI
Factors affecting the degree of heart
failure in patients with small cell lung cancer undergoing
chemotherapy.
| Comparison | P-value for
cumulative dose | P-value for
cTnI | P-value for
BNP | P-value for
LVEF | P-value for
diastolic function | P-value for
CK-MB |
|---|
| Asymptomatic or
mild symptomatic vs. moderate-to-severe symptomatic heart
failure | 0.006 | <0.001 | 0.223 | 0.091 | 0.131 | 0.031 |
| Asymptomatic or
mild symptomatic vs. life-threatening heart failure or death | 0.008 | <0.001 | 0.014 | 0.012 | 0.126 | 0.201 |
| Moderate-to-severe
symptomatic heart failure vs. life-threatening heart failure or
death | 0.072 | <0.001 | 0.048 | 0.015 | 0.493 | 0.853 |
ROC curve analysis of independent
prognostic factors and combined indicators
In summary, cTnI and LVEF were identified as
independent prognostic factors for the occurrence and severity of
heart failure. ROC curves were plotted, and the area under the
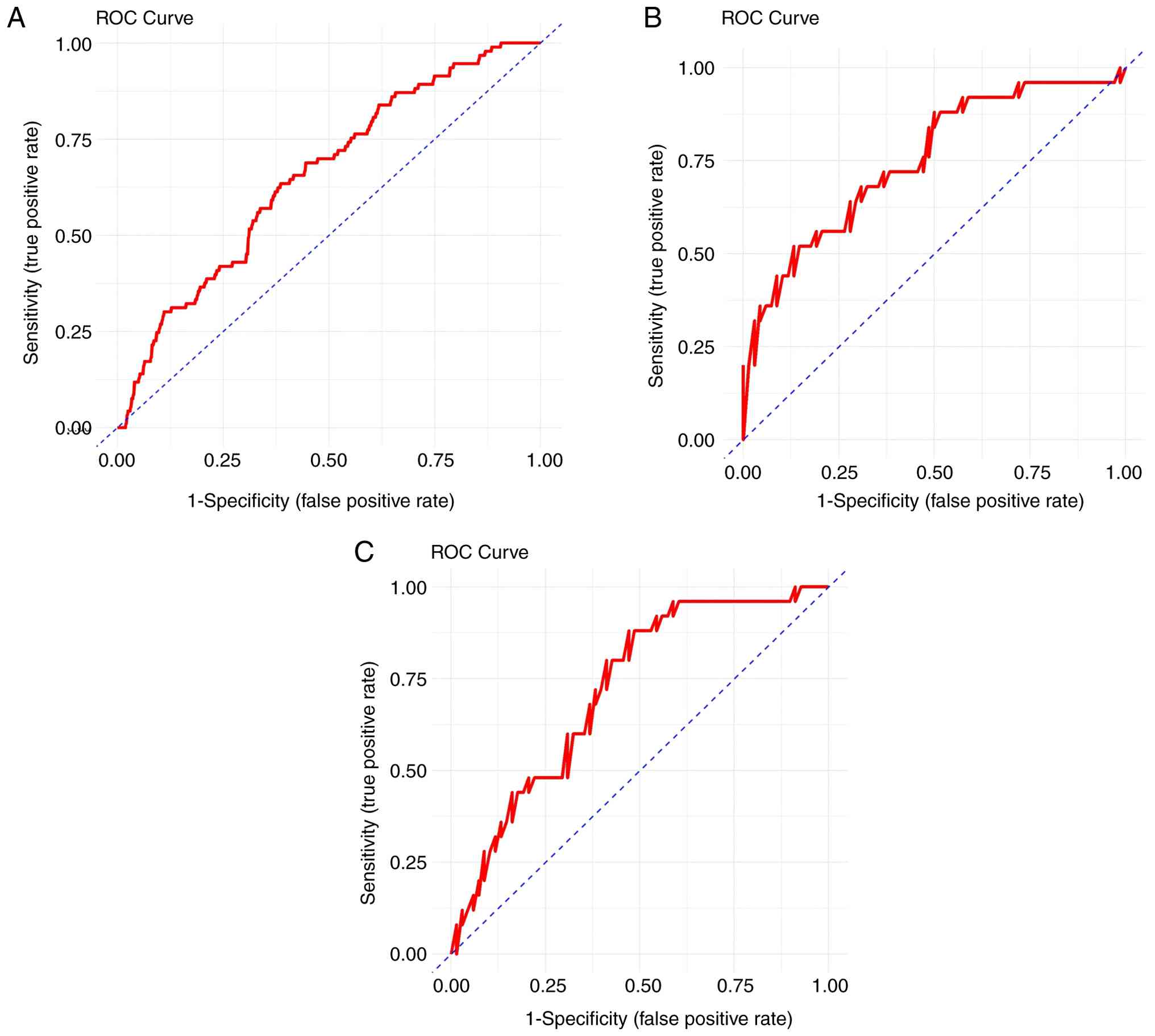
curve (AUC) values for cTnI were 0.651, 0.753 and 0.711 for
predicting mild, moderate and life-threatening heart failure
respectively, with optimal thresholds of 0.044, 0.059 and 0.063
ng/ml, respectively. These findings indicated that when cTnI is
<0.044 ng/ml, heart failure is not expected to occur; however,
when cTnI is ≥0.044 ng/ml and <0.059 ng/ml, asymptomatic or mild
heart failure is likely; when cTnI is ≥0.059 ng/ml and <0.063
ng/ml, moderate-to-severe heart failure is likely; and when cTnI is
≥0.063 ng/ml, life-threatening heart failure or death is expected
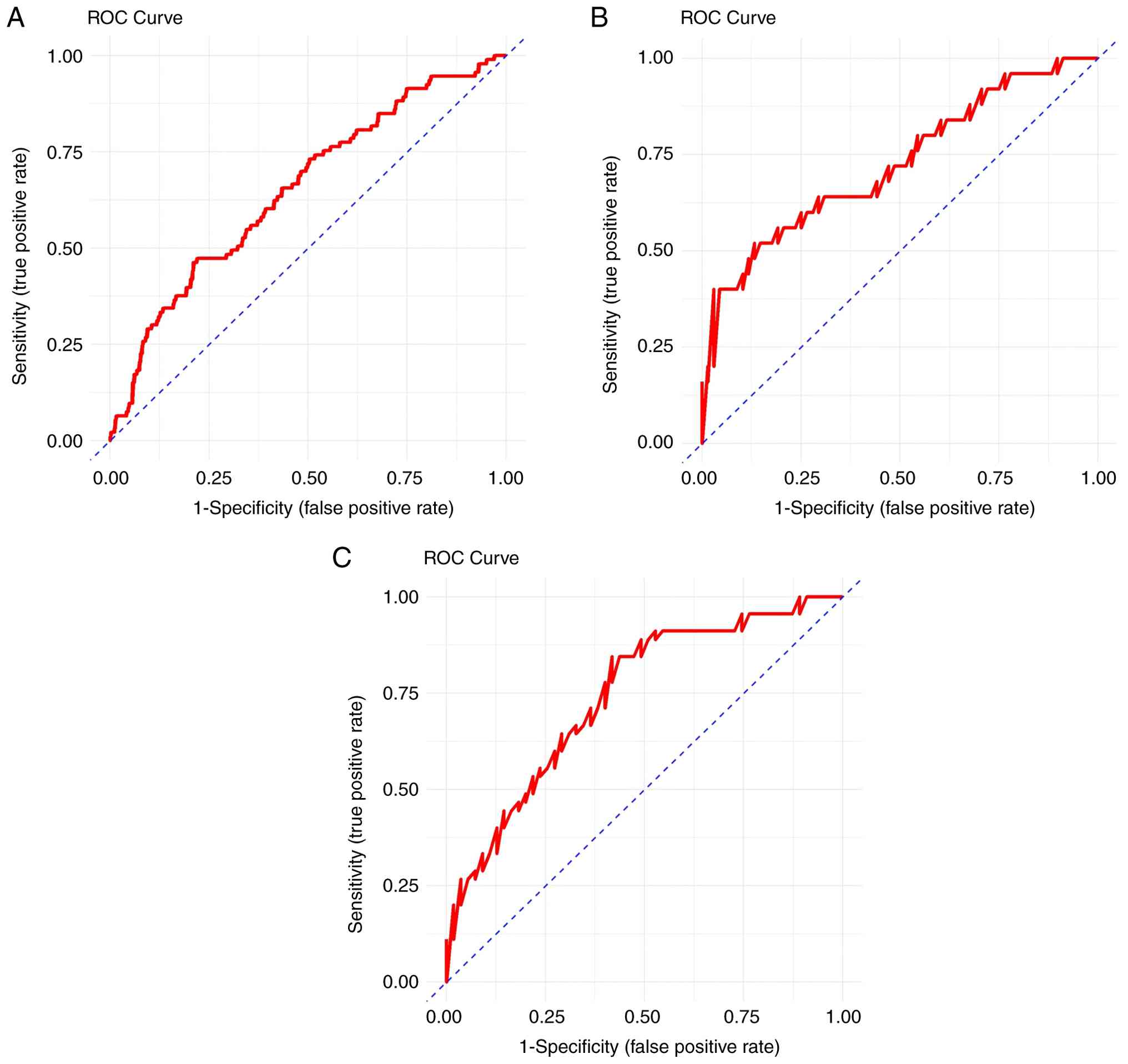
(Fig. 1). For LVEF, the AUC values
were 0.671, 0.724 and 0.749 for predicting mild, moderate and
life-threatening heart failure respectively, with optimal
thresholds of 39.083, 37.216 and 35.532%, respectively. These
findings indicated that when LVEF is >39.083%, heart failure is
not expected to occur; when LVEF is ≤39.083% and >37.216%,
asymptomatic or mild heart failure is likely; when LVEF is ≤37.216%
and >35.532%, moderate-to-severe heart failure is likely; and
when LVEF is ≤35.532%, life-threatening heart failure or death is
expected (Fig. 2).
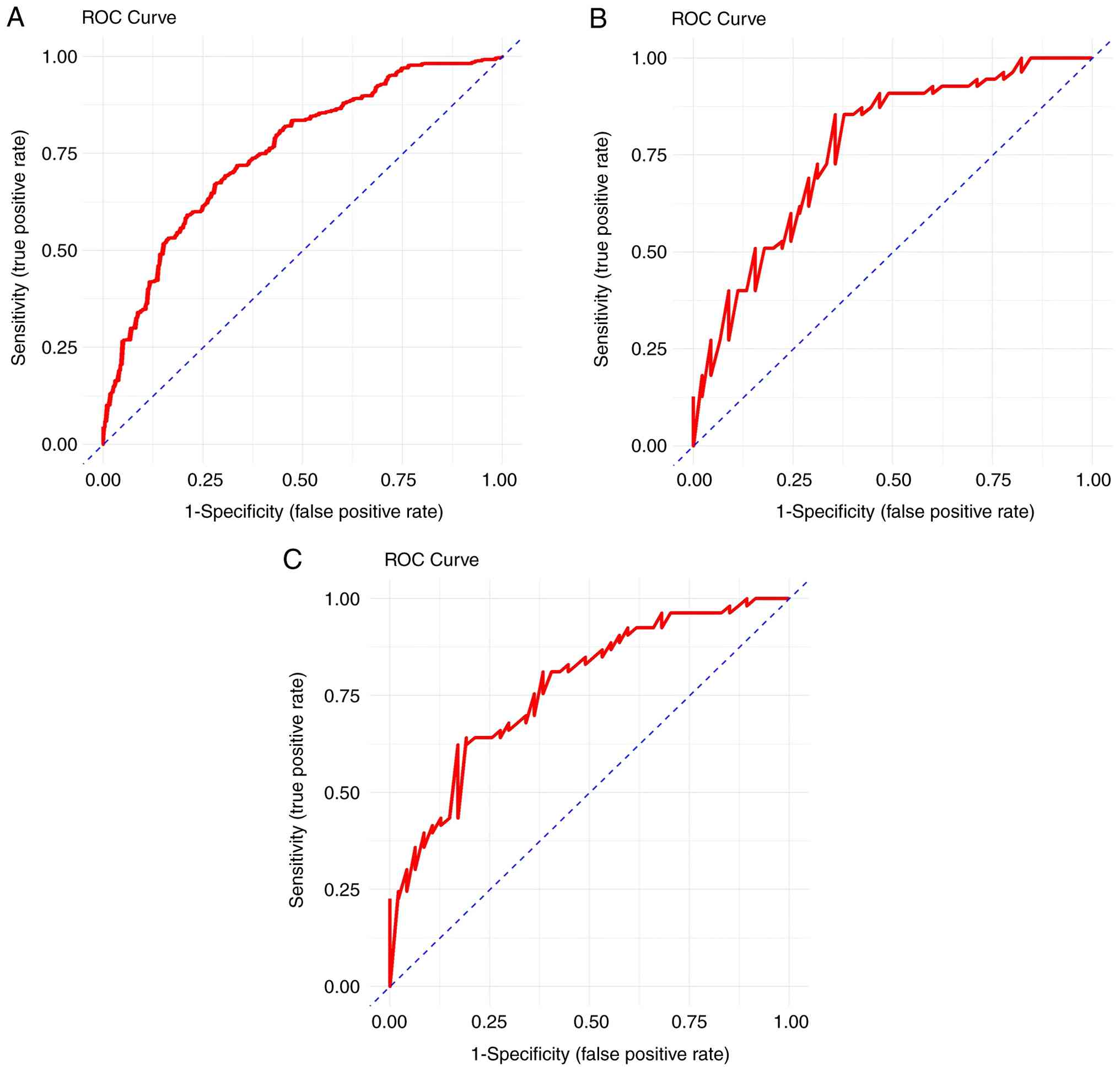
For the combined cTnI and LVEF indicator, the AUC
values were 0.763, 0.775 and 0.777 for predicting mild, moderate
and life-threatening heart failure, respectively, with optimal
thresholds of -0.035, 1.317 and 1.682, respectively. These findings
indicated that when the combined indicator is <-0.035, heart
failure is not expected to occur; when the combined indicator is
≥-0.035 and <1.317, asymptomatic or mild heart failure is
likely; when the combined indicator is ≥1.317 and <1.682,
moderate-to-severe heart failure is likely; and when the combined
indicator is ≥1.682, life-threatening heart failure or death is
expected (Fig. 3). The AUC values
of the combined indicator showed higher diagnostic efficacy,
especially in predicting life-threatening heart failure, with a
sensitivity of 0.831 and a specificity of 0.645. These results
suggested that the combination of cTnI and LVEF may have higher
diagnostic value in predicting heart failure compared with
individual indicators (Table
VII).
 | Table VIIReceiver operating characteristic
curve parameters for independent risk factors. |
Table VII
Receiver operating characteristic
curve parameters for independent risk factors.
| A, cTnI |
|---|
| Prediction | AUC | AUC CI lower | AUC CI upper | Threshold | Youden index | Sensitivity | Specificity |
|---|
| Occurrence of mild
heart failure | 0.651 | 0.591 | 0.710 | 0.044 | 0.279 | 0.785 | 0.495 |
| Occurrence of
moderate-to-severe heart failure | 0.753 | 0.647 | 0.859 | 0.059 | 0.428 | 0.840 | 0.588 |
| Occurrence of
life-threatening heart failure or death | 0.711 | 0.467 | 0.954 | 0.063 | 0.408 | 0.833 | 0.575 |
| B, LVEF |
| Prediction | AUC | AUC CI lower | AUC CI upper | Threshold | Youden index | Sensitivity | Specificity |
| Occurrence of mild
heart failure | 0.671 | 0.611 | 0.731 | 39.083 | 0.302 | 0.634 | 0.667 |
| Occurrence of
moderate-to-severe heart failure | 0.724 | 0.604 | 0.843 | 37.216 | 0.458 | 0.840 | 0.618 |
| Occurrence of
life-threatening heart failure or death | 0.749 | 0.615 | 0.883 | 35.532 | 0.586 | 1.000 | 0.586 |
| C, cTnI and
LVEF |
| Prediction | AUC | AUC CI lower | AUC CI upper | Threshold | Youden index | Sensitivity | Specificity |
| Occurrence of mild
heart failure | 0.763 | 0.661 | 0.861 | -0.035 | 0.463 | 0.646 | 0.817 |
| Occurrence of
moderate-to-severe heart failure | 0.775 | 0.704 | 0.835 | 1.317 | 0.352 | 0.742 | 0.610 |
| Occurrence of
life-threatening heart failure or death | 0.777 | 0.697 | 0.835 | 1.682 | 0.476 | 0.831 | 0.645 |
Discussion
The present study systematically collected
demographic characteristics and cardiac function biomarkers of
patients with SCLC in the early stage of chemotherapy. Through
intergroup comparisons, multivariate logistic regression and
multinomial logistic regression analyses, risk factors related to
heart failure were identified, and the predictive performance of
risk factors was evaluated through ROC curve analysis.
The key indicators identified in the present study
as independent prognostic factors for heart failure include the
cumulative dose of chemotherapy drugs, cTnI, NT-proBNP, LVEF and
CK-MB. Previous research has found that the platinum-based
chemotherapy may be associated with cardiotoxicity. Carboplatin has
been shown to promote the generation of reactive oxygen species,
leading to mitochondrial dysfunction in cardiomyocytes, which in
turn induces cell apoptosis and impairs myocardial contractile
function, thereby increasing the risk of heart failure (20). The elevation of cTnI and BNP are
considered a reliable biomarker for myocardial stress and injury
(21), and a decrease in LVEF
typically indicates a weakened pumping function of the heart, thus
increasing the likelihood of developing heart failure in the future
(22,23). In addition, CK-MB serves as a
specific marker of myocardial injury, and its elevation indicates
damage to myocardial cells, thus increasing the risk of future
heart failure (24).
ROC curve analysis showed that combining cTnI and
LVEF provides a more accurate prediction of heart failure
occurrence and severity in patients with SCLC following
chemotherapy when compared against using only one of the two
biomarkers. The present study identified several key thresholds
that can help clinicians monitor and manage the cardiac health of
patients more effectively in daily practice. For example, when the
level of cTnI is ≥0.044 ng/ml or LVEF is ≤39.083%, this indicates
that the patient may develop heart failure in the future, and this
information is crucial for the timely adjustment of treatment
plans. When cTnI levels rise to 0.063 ng/ml or LVEF drops ≤35.532%,
this indicates a risk of life-threatening heart failure,
necessitating more aggressive treatment measures to prevent further
deterioration of the cardiac function, such as early initiation of
angiotensin-converting enzyme inhibitors (25). Additionally, the present study
established a standardized combined indicator, optimizing the
combined use of cTnI and LVEF to enhance the predictive ability for
heart failure occurrence. Specifically, the results demonstrated
that when the value of this combined indicator is ≥-0.035, it can
predict the occurrence of heart failure; when it is ≥1.682, it
strongly suggests that the patient may be at risk of
life-threatening heart failure. This standardized indicator may
provide clinicians with a more intuitive and easy-to-apply tool
that can serve as a reference in the routine monitoring of
patients.
However, compared with cTnI and LVEF, the predictive
value of other traditional myocardial markers (such as CK-MB) is
relatively limited. This may be because CK-MB has a short half-life
and is typically used to reflect acute and short-term myocardial
injury (26) . Therefore, its
ability to predict long-term heart failure is inferior to that of
LVEF and cTnI. This may also be one of the reasons why CK-MB
demonstrated lower significance compared with cTnI and LVEF in the
present study.
In the baseline data analysis performed in the
present study, there was no significant association between the
occurrence of gene mutations and heart failure after chemotherapy.
Overall, the relationship between gene mutations and
chemotherapy-induced HF remains uncertain, the evidence is mixed,
and further studies are needed. However, specific gene mutations,
such as TP53 and RB1, have been found to be closely associated with
the efficacy and prognosis of patients with SCLC after chemotherapy
(27). Patients carrying these
mutations may have different reactions to chemotherapy and exhibit
a higher risk of disease recurrence. It has been suggested that
specific gene mutations, such as those related to cardiac function,
such as TTN truncating variants (28), may affect the structure and
function of the heart and be associated with changes in the levels
of cardiac markers, such as BNP and cTnI (29). In the future, large-scale
longitudinal cohort studies should be considered to determine which
specific gene mutations are associated with changes in cardiac
markers and explore whether these factors can predict the
occurrence of heart failure alone or in combination.
As a retrospective study, the present research
provided a foundation for future validation of the prediction model
in different centers and populations. Through comprehensive
analysis, the importance of the combined indicator of cTnI and LVEF
was confirmed in predicting chemotherapy-related heart failure in
patients with SCLC. Existing models, such as the cardiotoxicity
risk score (CRS) (30), primarily
assess clinical risk factors such as age, history of cardiovascular
disease, hypertension and chemotherapy dosage; however, these
static variables mainly reflect the baseline condition of the
patient and do not directly capture the extent of myocardial injury
or the actual status of cardiac function, which creates certain
limitations in predicting heart failure. By contrast, the present
study identified cTnI and LVEF as core indicators that directly
reflect the cardiac status, and their combined use may enable a
more accurate prediction of the occurrence and severity of heart
failure. Even when based solely on early data before chemotherapy,
these indicators may potentially compensate for the limitations of
the CRS model in assessing myocardial damage or changes in cardiac
function; however, this is merely a hypothesis proposed in the
present study and requires validation in future research, cTnI and
LVEF are important indicators for assessing myocardial injury and
cardiac function, and numerous studies have applied them to the
evaluation of heart failure and its prognosis (31). For example, in pediatric patients
with heart failure, cTnI levels at admission have marked clinical
value in assessing the severity and progression of heart failure
(32), and continuous measurement
of cTnI is closely associated with the prognosis of acute heart
failure (33). However, to the
best of our knowledge, studies focusing on chemotherapy-related
heart failure in patients with SCLC remain relatively limited, and
research on the combined use of these two indicators for predicting
heart failure is even rarer. The present study demonstrated that
cTnI and LVEF can predict the occurrence and severity of heart
failure in patients with SCLC undergoing chemotherapy, providing a
reference for clinical monitoring and intervention.
The present study also has certain limitations. As a
retrospective analysis, the current study is subject to selection
bias and information bias, and some important confounding factors,
such as nutritional status, hypertension and diabetes, may not have
been fully controlled. The lack of dynamic monitoring of cardiac
biomarkers (such as cTnI, CK-MB and NT-proBNP) during chemotherapy
and the reliance solely on baseline values for analysis, may
underestimate the true risk of heart failure and the predictive
capability of these biomarkers. Future prospective studies should
incorporate dynamic monitoring to more comprehensively evaluate the
predictive value of temporal changes in cardiac biomarkers for
heart failure and provide a basis for early intervention. Although
the present study explored the association between
chemotherapy-related factors and heart failure, other potential
risk factors (such as hypertension and diabetes) were not
monitored. Furthermore, due to the retrospective nature of the
present study, causal relationships cannot be established. Lastly,
the data may be affected by time-related changes, as certain
chemotherapy regimens and cardiac function evaluation methods may
have evolved. Therefore, future multicenter, large-sample,
prospective studies are needed to validate these findings.
In conclusion, the present study showed that cTnI
and LVEF are independent prognostic factors for predicting heart
failure and its severity in patients with SCLC following
chemotherapy, and the combined indicator of cTnI with LVEF
demonstrated higher accuracy. The key thresholds and standardized
combined indicator identified in the present study provide
clinicians with effective tools for the early identification of
high-risk patients. This offers important scientific evidence for
improving treatment outcomes in patients with SCLC.
Supplementary Material
Flowchart of patient selection.
Acknowledgements
Not applicable.
Funding
Funding: This study is funded by Administration of Traditional
Chinese Medicine of Hebei Province [The 2024 Annual Research
Project Plan for Traditional Chinese Medicine (project no.
2024022)].
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
JZ and XW were responsible for the conceptualization
of the present study, data curation, creation of the study design
and analysis plan, software tools used for data analysis and
visualization, and writing the original draft. XLi, WZ, XLu and RL
were involved in the statistical analysis of the collected data,
project administration and creation of figures and charts. XS was
responsible for the collection of data, performing experiments or
assessments related to the study, checking the accuracy and
reliability of the collected data and analyses, supervision, and
reviewing and editing the manuscript. All authors read and approved
the final manuscript. JZ and XS confirm the authenticity of all the
raw data.
Ethics approval and consent to
participate
The present study was conducted in accordance with
The Declaration of Helsinki and was approved by the Ethics
Committee of Hebei Provincial Hospital of Traditional Chinese
Medicine (approval no. 20240910; Shijiazhuang, China). The
requirement for informed consent was waived due to the
retrospective nature of the study, which involved anonymized
clinical data and posed no more than minimal risk to the
participants.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Saltos A, Shafique M and Chiappori A:
Update on the biology, management, and treatment of small cell lung
cancer (SCLC). Front Oncol. 10(1074)2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Rous FA, Singhi EK, Sridhar A, Faisal MS
and Desai A: Lung cancer treatment advances in 2022. Cancer Invest.
41:12–24. 2023.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Yang S, Zhang Z and Wang Q: Emerging
therapies for small cell lung cancer. J Hematol Oncol.
12(47)2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Curigliano G, Cardinale D, Suter T,
Plataniotis G, de Azambuja E, Sandri MT, Criscitiello C, Goldhirsch
A, Cipolla C and Roila F: ESMO Guidelines Working Group.
Cardiovascular toxicity induced by chemotherapy, targeted agents
and radiotherapy: ESMO clinical practice guidelines. Ann Oncol. 23
(Suppl 7):vii155–vii166. 2012.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Lyon AR, Dent S, Stanway S, Earl H,
Brezden-Masley C, Cohen-Solal A, Tocchetti CG, Moslehi JJ, Groarke
JD, Bergler-Klein J, et al: Baseline cardiovascular risk assessment
in cancer patients scheduled to receive cardiotoxic cancer
therapies: A position statement and new risk assessment tools from
the Cardio-Oncology Study Group of the Heart failure association of
the European Society of cardiology in collaboration with the
international cardio-oncology society. Eur J Heart Fail.
22:1945–1960. 2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Zhang S, Liu X, Bawa-Khalfe T, Lu LS, Lyu
YL, Liu LF and Yeh ET: Identification of the molecular basis of
doxorubicin-induced cardiotoxicity. Nat Med. 18:1639–1642.
2012.PubMed/NCBI View
Article : Google Scholar
|
|
7
|
Herrmann J, Lerman A, Sandhu NP,
Villarraga HR, Mulvagh SL and Kohli M: Evaluation and management of
patients with heart disease and cancer: Cardio-oncology. Mayo Clin
Proc. 89:1287–1306. 2014.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Liang H, Wang T, Liu D, Wang H, Ba Z, Xiao
Y, Liu Y, Yuan J and Yang W: Cardiovascular comorbidities and their
prognostic value in small cell lung cancer patients with
chemoradiotherapy. Clin Transl Oncol. 26:1348–1356. 2024.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Minotti G, Menna P, Salvatorelli E, Cairo
G and Gianni L: Anthracyclines: Molecular advances and
pharmacologic developments in antitumor activity and
cardiotoxicity. Pharmacol Rev. 56:185–229. 2004.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Cardinale D, Colombo A, Torrisi R, Sandri
MT, Civelli M, Salvatici M, Lamantia G, Colombo N, Cortinovis S,
Dessanai MA, et al: Trastuzumab-induced cardiotoxicity: Clinical
and prognostic implications of troponin I evaluation. J Clin Oncol.
28:3910–3916. 2010.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Sawaya H, Sebag IA, Plana JC, Januzzi JL,
Ky B, Cohen V, Gosavi S, Carver JR, Wiegers SE, Martin RP, et al:
Early detection and prediction of cardiotoxicity in
chemotherapy-treated patients. Am J Cardiol. 107:1375–1380.
2011.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Maisel A, Mueller C, Adams K Jr, Anker SD,
Aspromonte N, Cleland JG, Cohen-Solal A, Dahlstrom U, DeMaria A, Di
Somma S, et al: State of the art: Using natriuretic peptide levels
in clinical practice. Eur J Heart Fail. 10:824–839. 2008.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Neilan TG, Coelho-Filho OR, Shah RV, Feng
JH, Pena-Herrera D, Mandry D, Pierre-Mongeon F, Heydari B, Francis
SA, Moslehi J, et al: Myocardial extracellular volume by cardiac
magnetic resonance imaging in patients treated with
anthracycline-based chemotherapy. Am J Cardiol. 111:717–722.
2013.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Yu AF and Ky B: Roadmap for biomarkers of
cancer therapy cardiotoxicity. Heart. 102:425–430. 2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Cartas-Espinel I, Telechea-Fernández M,
Delgado CM, Barrera AA, Cuevas NS and Riffo-Campos AL: Novel
molecular biomarkers of cancer therapy-induced cardiotoxicity in
adult population: A scoping review. ESC Heart Fail. 9:1651–1665.
2022.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Ganti AKP, Loo BW, Bassetti M, Blakely C,
Chiang A, D'Amico TA, D'Avella C, Dowlati A, Downey RJ, Edelman M,
et al: Small cell lung cancer, version 2.2022, NCCN clinical
practice guidelines in oncology. J Natl Compr Canc Netw.
19:1441–1464. 2021.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Vallières E, Shepherd FA, Crowley J, Van
Houtte P, Postmus PE, Carney D, Chansky K, Shaikh Z and Goldstraw
P: International Association for the Study of Lung Cancer
International Staging Committee and Participating Institutions. The
IASLC lung cancer staging project: Proposals regarding the
relevance of TNM in the pathologic staging of small cell lung
cancer in the forthcoming (seventh) edition of the TNM
classification for lung cancer. J Thorac Oncol. 4:1049–1059.
2009.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Zhang S, Liang F and Tannock I: Use and
misuse of common terminology criteria for adverse events in cancer
clinical trials. BMC Cancer. 16(392)2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
a19. R Core Team (2021). R: A language and
environment for statistical computing. R Foundation for Statistical
Computing, Vienna, Austria. URL https://www.R-project.org/.
|
|
20
|
Chan SHY, Khatib Y, Webley S, Layton D and
Salek S: Identification of cardiotoxicity related to non-small cell
lung cancer (NSCLC) treatments: A systematic review. Front
Pharmacol. 14(1137983)2023.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Cardinale D, Sandri MT, Colombo A, Colombo
N, Boeri M, Lamantia G, Civelli M, Peccatori F, Martinelli G,
Fiorentini C and Cipolla CM: Prognostic value of troponin I in
cardiac risk stratification of cancer patients undergoing high-dose
chemotherapy. Circulation. 109:2749–2754. 2004.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Gevaert AB, Kataria R, Zannad F, Sauer AJ,
Damman K, Sharma K, Shah SJ and Van Spall HGC: Heart failure with
preserved ejection fraction: Recent concepts in diagnosis,
mechanisms and management. Heart. 108:1342–1350. 2022.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Reding KW, O'Connell NS, D'Agostino RB Jr,
Hundley W, Lucas AR, Ladd AC, Jordan JH, Heiston EM, Ge Y and
Hundley WG: Both intermuscular fat and LVEF decline promote heart
failure symptoms in cancer survivors. Cardiooncology.
7(16)2021.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Gupta V, Singh SK, Agrawal V and Singh TB:
Role of ACE inhibitors in anthracycline-induced cardiotoxicity: A
randomized, double-blind, placebo-controlled trial. Pediatr Blood
Cancer. 65(e27308)2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Rydzek J, Gąsior ZT, Dąbek J, Wojnar J and
Skrzypek M: Assessment of risk factors for mortality in patients
with cardiovascular disease and a history of treatment for
malignancy. Kardiol Pol. 73:730–739. 2015.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Katsioupa M, Kourampi I, Oikonomou E,
Tsigkou V, Theofilis P, Charalambous G, Marinos G, Gialamas I,
Zisimos K, Anastasiou A, et al: Novel biomarkers and their role in
the diagnosis and prognosis of acute coronary syndrome. Life
(Basel). 13(1992)2023.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Chen D, Xu J, Qiao R, Zhao Y, Chu T, Han B
and Zhong R: Detection of genetic mutations by next-generation
sequencing for predicting prognosis of extensive-stage small-cell
lung cancer. J Oncol. 2020(8811487)2020.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Linschoten M, Teske AJ, Baas AF, Vink A,
Dooijes D, Baars HF and Asselbergs FW: Truncating Titin (TTN)
variants in chemotherapy-induced cardiomyopathy. J Card Fail.
23:476–479. 2017.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Tham YK, Bernardo BC, Ooi JY, Weeks KL and
McMullen JR: Pathophysiology of cardiac hypertrophy and heart
failure: Signaling pathways and novel therapeutic targets. Arch
Toxicol. 89:1401–1438. 2015.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Öztürk C, Validyev D, Becher UM, Weber M,
Nickenig G and Tiyerili V: A novel scoring system to estimate
chemotherapy-induced myocardial toxicity: Risk assessment prior to
non-anthracycline chemotherapy regimens. Int J Cardiol Heart Vasc.
33(100751)2021.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Savarese G, Stolfo D, Sinagra G and Lund
LH: Heart failure with mid-range or mildly reduced ejection
fraction. Nat Rev Cardiol. 19:100–116. 2022.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Salem SS, Saleh NY, Soliman SE and
Abo-Haded HM: On-admission plasma levels of BNP, MR-proADM, and
cTnI in pediatric heart failure: Contributions to diagnosis,
prognosis, and outcome. Ir J Med Sci. 191:263–270. 2022.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Gürgöze MT, van Vark LC, Baart SJ, Kardys
I, Akkerhuis KM, Manintveld OC, Postmus D, Hillege HL,
Lesman-Leegte I, Asselbergs FW, et al: Multimarker analysis of
serially measured GDF-15, NT-proBNP, ST2, GAL-3, cTnI, creatinine,
and prognosis in acute heart failure. Circ Heart Fail.
16(e009526)2023.PubMed/NCBI View Article : Google Scholar
|