Introduction
Lung cancer represents a major global health burden
and remains one of the leading causes of cancer-associated
morbidity and mortality worldwide (1,2).
Contemporary treatment strategies have evolved into multimodal
approaches, with surgical resection for early-stage disease, and
combinations of platinum-based chemotherapy, radiotherapy and
immune checkpoint inhibitors (ICIs) administered for advanced or
unresectable cases (3). In
parallel with the development of these strategies, emerging studies
have explored novel adjuvant strategies, including bioactive
compounds derived from traditional Chinese herbal medicines and
nanomedicine-based drug delivery systems, which have offered new
insights into potential adjunctive therapies (4-6).
Even though ICIs have transformed the therapeutic
landscape of advanced malignancies, they have the disadvantage of
being associated with a broad spectrum of immune-related adverse
events (irAEs) involving multiple organ systems (7). Rhabdomyolysis (RM) is a clinical
syndrome characterized by skeletal muscle fiber necrosis and the
release of intracellular components, including myoglobin (Mb) and
creatine kinase (CK), into the circulation. Clinically, it
typically presents with myalgia, fatigue and dark-colored urine,
and is most commonly caused by trauma, strenuous exercise or drug
and toxin exposure (8). The
excessive immune activation associated with ICI therapy, in
combination with the hypometabolic state caused by hypothyroidism,
may serve to act as a synergistic ‘double-hit’ to the skeletal
muscle, contributing to the development of RM. Although isolated
cases of RM triggered by ICI-induced hypothyroidism or inflammatory
myopathy have been reported, the concurrent occurrence of these two
conditions acting synergistically to precipitate severe RM remains
relatively uncommon (9,10). The present case study reported on a
patient with squamous cell lung carcinoma who developed severe
synergistic immune-endocrine toxicity following sintilimab therapy,
manifesting as overt hypothyroidism and an overlap syndrome,
ultimately culminating in profound RM. With the administration of
supportive care, prompt levothyroxine replacement and a tapering
glucocorticoid regimen, the elevated cardiac enzyme levels were
found to decrease, the estimated glomerular filtration rate (eGFR)
improved and the RM-associated symptoms were largely resolved. This
case report underscores the importance of vigilant monitoring for
overlapping irAEs and also provides new insights into how endocrine
and immune dysfunction may converge to cause severe tissue
injury.
Case report
A 65-year-old male was admitted to the Department of
Cardiovascular Diseases at the Affiliated Hospital of Shandong
University of Traditional Chinese Medicine (Jinan, China) in
December 2024 with the chief complaint of bilateral hand and foot
swelling, accompanied by fatigue for 2 months. On admission, the
patient presented with prominent pitting edema of the hands and
feet. The affected skin appeared dark yellow, coarse and deeply
wrinkled. The patient also reported marked fatigue, a sensation of
distension and tightness in the limbs, and minimal response to
previous use of oral diuretics, including furosemide (20 mg qd) and
spironolactone (50 mg qd), which had been prescribed by the doctor
of Shandong Provincial Qianfoshan Hospital (Jinan, China) due to
oedema. Additional symptoms included cold extremities, profuse
sweating and rough skin over the swollen areas.
The patient had a history of old myocardial
infarction for >3 years, and dyslipidemia of a similar duration,
for which no lipid-lowering medications had been taken. The patient
also had a history of cerebral infarction for >3 years, which
had resulted in residual dysarthria and left lower limb motor
impairment. A diagnosis of right pulmonary squamous cell carcinoma
had been made >6 months prior to admission to the Affiliated
Hospital of Shandong University of Traditional Chinese Medicine
(Jinan, China). Between May and August 2024, the patient had
undergone four cycles of chemotherapy and immunotherapy at the
Provincial Hospital Affiliated to Shandong First Medical University
(Jinan, China). In September of the same year, the patient received
CT-guided implantation of radioactive iodine-125 seeds for local
control of the right lung tumor at Shandong Provincial Qianfoshan
Hospital (Jinan, China). The patient had a history of alcohol
consumption for >40 years and tobacco use for >50 years, but
had ceased consuming both since his lung cancer diagnosis. The
patient denied any history of drug allergies or other adverse
personal habits.
Cardiac examination on admission revealed a mildly
enlarged cardiac silhouette, with the point of maximal impulse
displaced inferolaterally. The patient's heart rate was 81 bpm,
which was within the normal range, with a regular rhythm and strong
heart sounds. Percussion indicated leftward and downward extension
of cardiac dullness, and auscultation revealed an ejective systolic
murmur in the aortic valve area. No notable positive findings were
identified in the examination of other systems.
The patient first presented in April 2024 with
hemoptysis at Jinan Traditional Chinese Medicine Hospital (Jinan,
China). Chest CT revealed a space-occupying lesion in the right
upper lobe of the lung. Bronchoscopy and biopsy of the right main
bronchus subsequently confirmed squamous cell carcinoma. According
to the 8th edition of the American Joint Committee on Cancer TNM
Staging System, the patient was diagnosed with right upper lobe
squamous cell carcinoma (stage cT2bN2M0; IIIA) (11). Based on the medical records
provided by the patient, considering his history of
arteriosclerosis, lobectomy was not recommended; instead, a
combination regimen was initiated, including albumin-bound
paclitaxel (on days 1 and 5), nedaplatin (on days 2 and 3) and
sintilimab (on day 4) for concurrent chemotherapy and immunotherapy
(information on the doses could not be retrieved).
Following the second treatment cycle, the patient
developed hypothyroidism, as detected by a thyroid function test,
which was suspected to be an irAE associated with sintilimab, as
indicated in the patient's medical records. As a result, sintilimab
was discontinued in subsequent cycles and the patient completed the
remaining two cycles of chemotherapy with albumin-bound paclitaxel
and nedaplatin alone. Approximately 2 months after having completed
the four-cycle regimen, follow-up imaging revealed inadequate tumor
control. The patient subsequently underwent the CT-guided
implantation of iodine-125 radioactive seeds at Shandong Provincial
Qianfoshan Hospital (Jinan, China) for localized brachytherapy, a
total of 14 particles, each containing 0.8 mCi, were implanted into
the tumor in an effort to reduce the size of the primary
lesion.
Upon admission to the hospital, the attending
physician ordered a series of laboratory tests. Key findings
included the following: Creatine kinase-myocardial brand (CK-MB),
37.44 ng/ml (reference range: 0-5 ng/ml); CK, 4,974 U/l (reference
range: 50-310 U/l); lactate dehydrogenase (LDH), 556 U/l (reference
range: 120-250 U/l); α-hydroxybutyrate dehydrogenase (α-HBDH), 361
U/l (reference range: 59-126.4 U/l); free tri-iodothyronine,
<0.6 pmol/l (reference range: 3.1-6.8 pmol/l); free thyroxine,
<0.5 pmol/l (reference range: 12.8-21.3 pmol/l); thyroid
stimulating hormone (TSH), >100 µIU/ml (reference range:
0.27-4.2 µIU/ml); thyroglobulin, <0.04 ng/ml (reference range:
3.5-77 ng/ml); thyroglobulin antibodies (TGAb), 533 IU/ml
(reference range: 0-115 IU/ml); and thyroid peroxidase antibody
(TPOAb), 63.8 IU/ml (reference range: 0-34 IU/ml). Additional test
results are shown in Table I.
 | Table IKey laboratory parameters at admission
and prior to discharge. |
Table I
Key laboratory parameters at admission
and prior to discharge.
| Day 40 after
discharge | - | 28.40 | 1.58 | 68.00 | 224.00 | 193.00 | - | 65.00 | 97.27 | 402.00 | - |
|---|
| Day 12 after
discharge | - | - | 4.89 | 269.00 | 265.00 | 181.00 | - | 111.00 | 60.11 | 483.00 | - |
| Day 17-18 | 61.00 | - | 6.58 | 795.00 | 352.00 | 240.00 | - | 87.00 | 80.70 | 449.00 | 89.80 |
| Day 12 | 54.00 | - | 11.70 | 1931.00 | 428.00 | 287.00 | - | 113.00 | 58.83 | 621.00 | >100 |
| Day 7 | - | 76.60 | 16.18 | 2256.00 | 433.00 | 304.00 | 171.28 | 89.00 | 78.51 | - | - |
| Day 5 | - | 68.37 | 18.66 | 3199.00 | 474.00 | 313.00 | 137.53 | 88.00 | 79.59 | - | - |
| Day 3-4 | 53.00 | 68.31 | 22.19 | 4311.00 | 474.00 | 288.00 | 153.95 | 99.00 | 69.03 | - | - |
| Day 2 | - | 80.82 | 25.60 | 5102.00 | 490.00 | 305.00 | - | 99.00 | 69.03 | - | - |
| Day 1 | - | 113.90 | 37.44 | 4974.00 | 556.00 | 361.00 | - | 134.00 | 47.87 | 553.00 | >100 |
| Normal reference
value range and units | 0-15 mm/h | 0-14 ng/l | 0-5 ng/ml | 50-310 U/l | 120-250 U/l | 59-126.4 U/l | 28-72 ng/ml | 57-111 µmol/l | 90-120 ml/min | 208-428 µmol/l | 0.27-4.2
µIU/ml |
| Test items | ESR | hs-cTnT | CK-MB | CK | LDH | α-HBDH | Mb | sCr | eGFR | UA | TSH |
The above abnormal findings indicated a pathological
state of irreversible damage to thyroid tissue, as suggested by the
patient's thyroid function tests revealing Hashimoto's thyroiditis
and hypothyroidism, combined with diffuse thyroid changes
demonstrated by thyroid color Doppler ultrasound (data not shown).
CK levels were found to 4,974 U/l (reference range: 50-310 U/l),
exceed the upper limit of normal by 15-fold, with a CK/CK-MB ratio
exceeding 100, suggesting that skeletal muscle injury predominated
over myocardial involvement. Given the patient's muscle weakness on
admission, RM was considered. Elevated cardiac enzyme levels,
including CK, CK-MB, LDH and α-HBDH, are indicative of potential
cardiac complications, such as myocardial infarction or coronary
artery disease. Renal function was also found to be impaired, with
an eGFR of 47.87 ml/min (chronic kidney disease, stage G3a; normal
reference range: 90-120 ml/min), along with elevated levels of
blood urea nitrogen at 10.56 mmol/l (reference range: 50-310
mmol/l), serum creatinine at 134 µmol/l (reference range: 57-111
µmol/l) and uric acid at 553 µmol/l (reference range: 208-428
µmol/l), which indicated glomerular filtration dysfunction and
tubular reabsorption impairment.
Based on the patient's clinical history and
ancillary investigations, the attending physician immediately
initiated symptomatic treatment, including diuresis (furosemide
injection 2 ml qd iv) and urine alkalinization (sodium bicarbonate
tablets 1 g tid p.o.), alongside fundamental supportive measures
such as antiplatelet therapy (clopidogrel hydrogen sulfate tablets
75 mg qd p.o.) and liver function protection (glutathione injection
1.2 g qd iv drip).
Given the presence of renal impairment and severe
hypothyroidism, consultations with the nephrology and endocrinology
departments were requested on the day of admission. The attending
physician, guided by the consultation findings, conducted
autoimmune marker tests, including anti-neutrophil cytoplasmic
antibodies (ANCA) and antinuclear antibodies (ANA). Subsequent
assessments of the levels of adrenocorticotropic hormone, 31.1
pg/ml (reference range: 7.2-63.3 pg/ml) and cortisol, 85.08 µg/l
(reference range: 48.2-195 µg/l) ruled out adrenal insufficiency,
and therefore, levothyroxine sodium tablets (25 mg qd p.o.) was
initiated.
On the third day of hospitalization, edema in the
hands and feet persisted, although joint pain had subsided and
muscle strength had partially recovered. Follow-up cardiac enzyme
marker tests showed improvements in their levels. Mb levels reached
twice the upper limit of normal, further supporting the diagnosis
of RM. Positive ANA results suggested the possibility of autoimmune
diseases. Accordingly, with informed consent from the patient and
his family, the patient was referred to the rheumatology department
for further specialized treatment. The rheumatology team conducted
additional limb electromyography (EMG) and myositis antibody
testing. The results obtained from the EMG indicated peripheral
nerve damage in the limbs, including the median, tibial and
peroneal nerves, presenting as multiple, asymmetric lesions
affecting both sensory and motor fibers. Specifically, the distal
motor latency of the median nerve was prolonged to 6.8 msec on the
right and 5.2 msec on the left. The compound muscle action
potential amplitude of the right common peroneal nerve recorded
over the extensor digitorum brevis was markedly reduced to 1.5 mV.
In addition, the sensory nerve action potential (SNAP) of the right
median nerve (digit-wrist segment) was absent, and the SNAP
amplitudes of the bilateral sural and superficial peroneal nerves
were all <5 µV. Antibody testing revealed positive results for
anti-PM-Scl75 antibodies (++), anti-proteinase 3 antibodies (+) and
anti-mitochondrial M2 antibodies (+). Following several days of
diuretic therapy, the patient's edema had significantly subsided,
revealing palmar hyperkeratosis with fissures (‘mechanic's hands’)
and purple papules (Gottron's papules) (12). These findings further suggested
that the patient likely presented with an overlapping syndrome
dominated by inflammatory myopathy, superimposed by ANCA-associated
vasculitis and subclinical primary biliary cholangitis. However,
after the rheumatology attending physician proposed a muscle
biopsy, the patient declined this invasive procedure, and a
definitive diagnosis could not be established from a pathological
perspective.
On the seventh day of hospitalization, with the
aforementioned investigations completed, the rheumatology team
prepared to initiate glucocorticoid pulse therapy to treat the
inflammatory myopathy. Considering the patient's poor glycemic
control, blindly adding corticosteroids might have had the effect
of exacerbating the hyperglycemia risk (13). Therefore, treatment commenced with
methylprednisolone sodium succinate (20 mg qd iv drip), while
diuretic therapy and levothyroxine supplementation continued
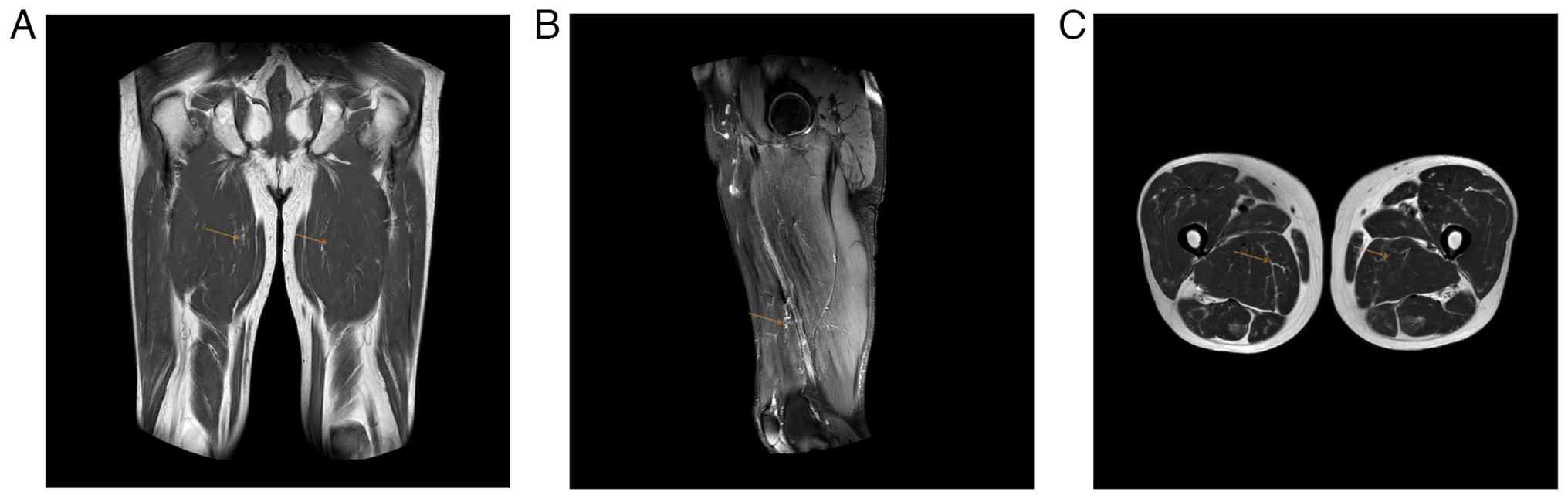
concurrently. An MRI of the lower limbs revealed persistent muscle
edema with localized hematomas in multiple areas, including the
biceps femoris and semitendinosus muscles, further supporting the
radiological diagnosis of RM (Fig.
1). However, glucocorticoid use did induce significant
hyperglycemia, with a randomly assessed blood glucose level
reaching 15.9 mmol/l (reference range: 3.9-11.1 mmol/l); therefore,
dapagliflozin (10 mg qd p.o.) and acarbose tablets (50 mg tid p.o.)
were added to the regimen, and thereby, glycemic control was
satisfactorily achieved.
The patient's condition gradually stabilized and
improved, and plans were prepared for discharge and continued
medication. Given the limited feasibility of intravenous
administration outside the hospital, glucocorticoid therapy was to
be switched to oral methylprednisolone tablets. In addition, the
diuretic combination of drugs (furosemide plus spironolactone),
blood glucose control medications (dapagliflozin plus acarbose) and
hypothyroidism treatment (sodium levothyroxine) were all to remain
unchanged.
At follow-up visits in January and February 2025,
laboratory tests revealed that the myocardial enzyme levels had
returned to near-normal levels and the eGFR had increased to 97.27
ml/min, indicating significant renal function recovery. The
randomly assessed blood glucose level was 7.83 mmol/l and the
hemoglobin A1c count was 8.6% (reference range: 4.0-6.0%),
reflecting relatively stable glycemic control. The patient
continued on the prescribed regimen, with sustained symptom relief
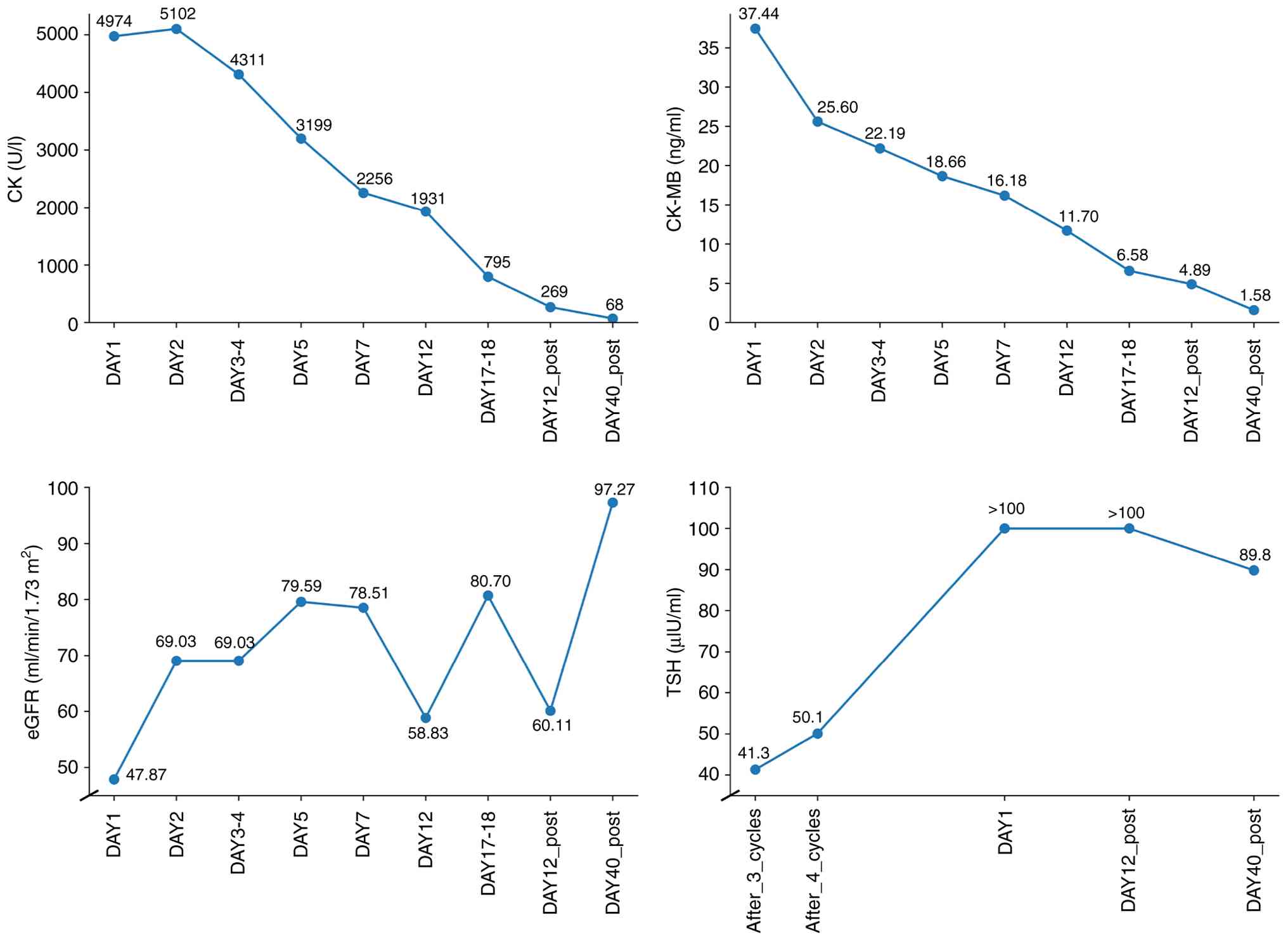
and a favorable therapeutic response. Changes in the levels of
cardiac enzymes, eGFR and thyroid function during the patient's
hospitalization are shown in Fig.
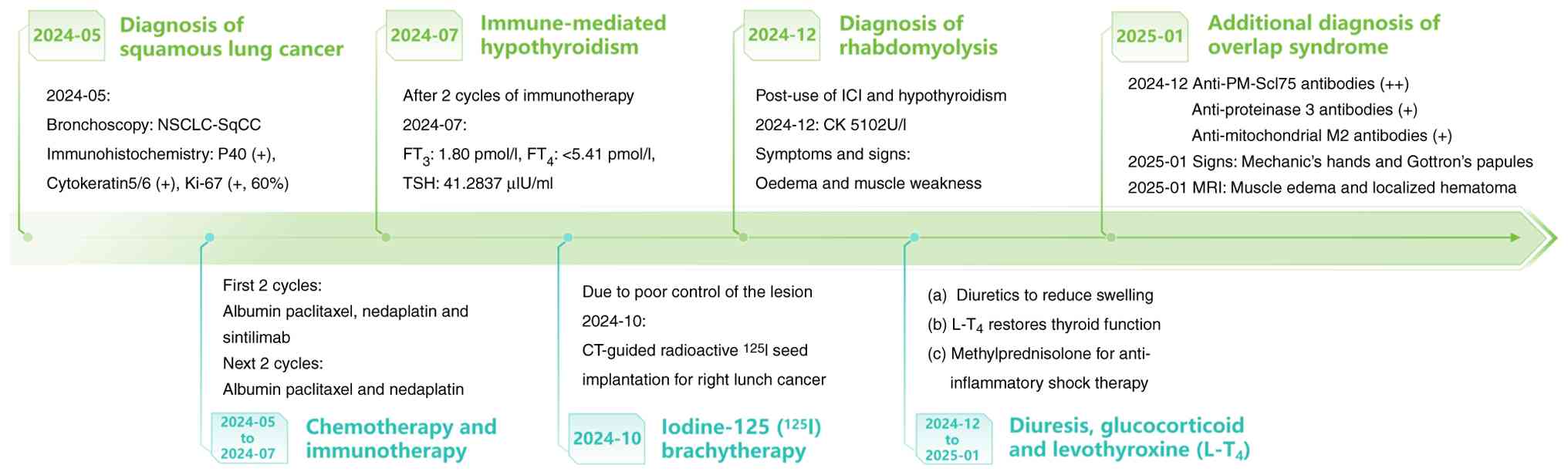
2. The timeline of the patient's diagnosis and treatment
process is shown in Fig. 3.
Discussion
This case report presents a highly complex
multisystem toxicity event triggered by an ICI during the treatment
of a patient with lung cancer. Following the administration of
sintilimab, the patient sequentially developed severe
hypothyroidism and overlap syndrome, which ultimately
synergistically led to life-threatening RM. In reviewing the
diagnostic and therapeutic course, it was observed that this
sequence of events was not a simple superposition of single
diseases; rather, it revealed that immune-endocrine axis
dysregulation in the context of administering ICIs can produce
synergistic effects, jointly attacking different target organs
(14).
Cases where ICI-induced thyroiditis and myopathy
sequentially occur in the same patient, and synergistically lead to
severe RM, are relatively rare. In the present case, the elevation
in the level of CK reached 4,974 U/l, ~15 times the upper limit of
normal, far exceeding the levels commonly seen in myopathy
associated with isolated hypothyroidism, which typically causes
only a mild-to-moderate CK elevation (namely, <10 times the
upper limit of normal) (15).
CK=4,974 U/l supports the presence of dual pathological processes
involving both immune-related inflammatory myopathy and
hypothyroidism-associated endocrine myopathy. In addition, thyroid
function testing revealed a TSH level >100 µIU/ml accompanied by
a markedly reduced level of FT4, consistent with severe
primary hypothyroidism, a pattern that is commonly associated with
destructive thyroiditis.
Recent evidence suggested that hypothyroidism
significantly impairs muscle regenerative capacity and also alters
metabolic homeostasis, which potentially diminishes myocyte
resilience to additional injury (16). In hypothyroidism, the disruption of
local thyroid hormone signaling pathways mediated by deiodinases
(namely, D2 and D3) and thyroid hormone receptors (especially
thyroid hormone receptor alpha) directly impairs satellite cell
survival, myogenic differentiation and effective repair capacity
(17,18). Under immune checkpoint inhibition,
muscle cells may become more susceptible to T cell-mediated
inflammatory attacks, leading to more extensive muscle
necrosis.
The first-line chemotherapeutic regimen used
(namely, nab-paclitaxel combined with nedaplatin administered to
the patient) is representative of platinum-based concurrent
chemoradiotherapy protocols for unresectable stage IIIB non-small
cell lung cancer (19).
Additionally, consolidation immunotherapy with sintilimab has been
validated in clinical settings (20). However, clinical trials of
sintilimab have reported that >10% of participants experienced
immune-associated endocrine dysfunction, including hypothyroidism
(21). Programmed death-1 (PD-1)
inhibitors are considered to cause destructive thyroiditis via
activating T cells that infiltrate thyroid tissue and damage
follicular cells, with this adverse reaction typically emerging
after 1-3 treatment cycles. Concurrently, elevated levels of TPOAb
and TGAb suggest the involvement of B cells, synergizing with T
cell-mediated cytotoxicity to produce autoimmune thyroiditis, which
typically precedes hypothyroidism (22). Regarding muscle damage, the
presence of anti-PM-Scl75 antibodies similarly indicates B-cell
involvement, suggesting an overlapping phenotype that ultimately
manifests as an overlap syndrome characterized by inflammatory
myopathy (23).
In the present case, both the thyroid and skeletal
muscle became targets of immune attack, suggesting a systemic
autoimmune storm at the cellular level driven by the core
underlying mechanism of ICIs. PD-1 acts as a central ‘brake’ on
peripheral T-cell activation, contributing to the maintenance of
self-tolerance through restraining effector T-cells and supporting
the function of regulatory T-cells (Tregs) (24). As a PD-1 inhibitor, sintilimab not
only promotes the expansion of CD8+ T cells and tissue
migration via releasing the inhibitory effect on T-cell activation,
but it also exerts direct cytotoxic effects on parenchymal tissues,
including thyroid follicular cells and skeletal muscle cells.
Concurrently, it enhances CD4+ T-cell-mediated support
for B-cell responses and epitope spreading, thereby amplifying
autoimmune reactions (25). This
disrupts Treg-mediated autoimmune tolerance, thereby providing a
common pathological basis for the simultaneous or sequential attack
on multiple organs, clinically manifesting as clustered irAEs.
Under the influence of low thyroid hormone levels in
severe hypothyroidism, mitochondrial oxidative phosphorylation in
muscle cells is impaired, which leads to insufficient ATP
production. Consequently, the activities of energy-dependent
Na+/K+-ATPase and sarcoplasmic reticulum
Ca²+-ATPase are decreased, thereby impairing
sarcoplasmic reticulum Ca²+ reuptake. This is
accompanied by increased Ca²+ leakage mediated by
rhabdomyolysis receptors, ultimately triggering cytoplasmic
Ca²+ accumulation. This cytoplasmic Ca²+
overload further activates Ca²+-dependent proteases,
which both leads to an exacerbation of mitochondrial dysfunction
and accelerates the disintegration of sarcolemma and myofibrils
(26,27).
By contrast, immune-mediated myopathies primarily
mediate muscle damage through immune effector mechanisms. Key
pathways feature the activation and infiltration of CD8+
T cells and macrophages, an abnormal upregulation of major
histocompatibility complex class I molecules on muscle fiber
surfaces, and the collaborative involvement of multiple
autoantibody-dependent pathways. This process recruits and deposits
the complement terminal complex (C5b-9) on to muscle capillary
endothelium and myofibrillar structures, ultimately leading to
focal myofibrillar necrosis and microvascular injury (28,29).
These dual pathways of metabolic dysfunction and immune-mediated
injury exhibit synergistic amplification effects at the skeletal
muscle level, leading to extensive damage to myocyte membrane
structures and significantly impaired repair capacity, ultimately
precipitating myofibrillar destruction dominated by cell necrosis.
The result is a massive release of intracellular components, which
is manifested in laboratory tests as highly elevated CK levels
(30). This provides mechanistic
support for the critical role of the PD-1 inhibitor-associated
dual-strike pathway in the occurrence of fulminant RM.
The mechanism of action of the aforementioned
‘synergistic toxicity’ has been well demonstrated in this case;
however, although myositis-specific antibodies and
myositis-associated autoantibodies provide important clues for
diagnosing inflammatory myopathies, antibody positivity alone is
insufficient to reliably distinguish between classic polymyositis,
endocrine-associated myopathies, paraneoplastic inflammatory
myopathies and ICI-associated myositis (31). The finding of neuropathic damage on
EMG examination is not specific to idiopathic inflammatory
myopathies (IIMs) and may also be observed in other neuromuscular
disorders, such as chemotherapy-related or paraneoplastic
peripheral neuropathies or chronic inflammatory demyelinating
polyradiculoneuropathy. The patient's history of lung malignancy
treated with radiotherapy, chemotherapy and immunotherapy suggests
two non-mutually exclusive possibilities, either tumor-associated
paraneoplastic myopathy or ICI-induced immune-mediated myopathy.
Previous case reports and case series have confirmed that nivolumab
and pembrolizumab may be associated with dermatomyositis/myositis
and other myositis subtypes, suggesting that ICI therapy may either
reveal underlying paraneoplastic autoimmune phenomena or
independently induce new-onset autoimmune myopathies (32-34).
A review of the patient's hospitalization records
during antitumor therapy revealed that hypothyroidism had already
developed in the course of immunotherapy. At that time, the
attending physician diagnosed immune-related hypothyroidism and
temporarily suspended the third and fourth cycles of ICI treatment.
During the same period, ANA screening was negative and no further
myositis-specific antibody testing was performed. Baseline thyroid
function and rheumatologic autoantibody data prior to the
initiation of immunotherapy were not available, which makes it
difficult to definitively establish a causal relationship between
immunotherapy and hypothyroidism. Notably, prior to the onset of
endocrine dysfunction, the patient did not exhibit typical clinical
manifestations suggestive of a paraneoplastic syndrome, such as
characteristic rash or soft-tissue edema. Overall, while the
possibility of a paraneoplastic process cannot be entirely ruled
out, the temporal sequence and existing laboratory findings are
more consistent with an immune-mediated mechanism.
Given the patient's refusal to undergo muscle biopsy
during hospitalization, a definitive pathological diagnosis could
not be obtained. Therefore, the classification of this case was
primarily based on a comprehensive assessment of indirect evidence,
including clinical symptoms and signs, laboratory findings and
imaging data. According to the 2017 European League Against
Rheumatism/American College of Rheumatology (EULAR/ACR)
classification criteria for adult IIMs, the patient presented with
late-onset disease, which was clinically characterized by symmetric
proximal muscle weakness and was accompanied by Gottron's papules,
markedly elevated serum levels of CK and muscle edema, as revealed
by muscle MRI imaging. In the 2017 EULAR/ACR scoring system without
muscle biopsy, the cumulative score from these indicators reached
7.9 points, exceeding the threshold for a high probability of IIM.
This suggested that the patient's comprehensive clinical
presentation was highly consistent within the inflammatory myopathy
spectrum (35). The specific
points for each item in this scoring system are summarized in
Table II.
 | Table IIEULAR/ACR score sheet for the present
patient. |
Table II
EULAR/ACR score sheet for the present
patient.
| EULAR/ACR item | Points | Evidence for the
present patient |
|---|
| Onset age | 2.1 | Adult onset (age
≥40 years) |
| Symmetric proximal
muscle weakness | 2.4 | Symmetrical
proximal muscle weakness (affecting both upper and lower limbs),
with proximal muscle weakness in the lower limbs being more
pronounced than distal weakness. |
| Dysphagia | 0 | Absent |
| Gottron's papules /
Gottron sign | 2.1 | Clinical exam:
Gottron's papules become visible |
| CK elevation | 1.3 | Peak CK=5,102 U/l
(document date: 2024.12) |
| EMG showing
myopathic abnormalities | 0 | EMG did not
demonstrate definite myopathic abnormalities, predominantly
neuropathic features with spontaneous activity were noted. |
| Muscle biopsy
consistent with IIM | 0 | Absent (patient
declined) |
| Total score
(without biopsy) | 7.9 | Total score=7.9
(without biopsy), exceeding the EULAR/ACR threshold for definite
IIM classification. |
The patient's complex antibody profile provided
deeper diagnostic and differential diagnostic clues. In addition to
anti-PM-Scl75 antibodies, positive anti-proteinase 3 and
anti-mitochondrial M2 antibodies were detected, suggesting the
potential for broader, or more latent, autoimmune activation
following PD-1 blockade. During the clinical differential diagnosis
process for the present case, a clear temporal correlation was
noted between the onset of myopathic symptoms and serological
abnormalities and the administration of sintilimab. Concurrently,
the patient exhibited irAEs, including thyroid involvement and
potential primary biliary cholangitis, and also demonstrated a
marked response to glucocorticoid therapy. Further integration with
the previously described history of sintilimab use, extensive
autoantibody profile and multisystem involvement reveals pronounced
overlapping features. Based on a comprehensive analysis of the
above evidence, it was possible to classify this case as an
ICI-associated overlap syndrome, primarily manifesting as
inflammatory myopathy.
In the present case, during the course of diagnosis
and treatment, it should be acknowledged that the absence of muscle
biopsy imposed an inherent limitation on obtaining definitive
histopathological confirmation. Although biopsy remains the gold
standard for differentiating subtypes of immune-mediated myopathies
and excluding non-inflammatory myopathies, the patient's refusal to
undergo this invasive procedure reflects a common decision-making
constraint encountered in real-world clinical practice. The term
‘overlap syndrome’ accurately reflects the complex clinical
phenotype, and provides a coherent conceptual framework for
understanding the unique pathophysiology of synergistic
immune-endocrine toxicity.
In recent years, given the widespread use of ICIs,
research on irAEs has increasingly shifted towards mechanistic
elucidation and precision management. Distinct irAEs exhibit
organ-specific immunological characteristics. In
myositis-associated irAEs, large-scale transcriptomic analyses have
demonstrated that different myositis subtypes harbor unique
cytokine signatures and immune checkpoint gene expression profiles,
thereby offering new insights into the biological heterogeneity of
these conditions (36). In the
context of thyroid toxicity, pre-existing antithyroid antibodies
have been identified as important risk factors for the development
of thyroid irAEs. For destructive thyroiditis, current management
strategies have emphasized regular monitoring at monthly intervals
during ICI therapy and the timely initiation of thyroid hormone
replacement once hypothyroidism develops (37,38).
Collectively, these advances have shown that contemporary irAE
management is evolving towards a paradigm centered on risk
stratification, early intervention and organ-specific therapeutic
strategies. In parallel, efforts to identify the molecular pathways
and predictive biomarkers for irAEs remain an active area of
research (39). From a clinical
practice perspective, current guidelines support a comprehensive
approach: Patients with suspected ICI-associated inflammatory
myopathy should undergo early evaluation, including cardiac enzyme
assessment, muscle MRI, EMG and myositis-specific autoantibodies,
with muscle biopsy also considered when feasible. For confirmed
cases, multidisciplinary management involving oncology,
rheumatology and endocrinology is recommended, together with timely
initiation of stepwise immunosuppressive therapy, which is
typically initiated with systemic corticosteroids (9,38,40).
In conclusion, through the diagnosis and management
of this case, an irAE was used as the entry point to supplement
existing knowledge regarding underlying mechanisms and real-world
clinical decision-making. Building on prior descriptions of
multisystem irAEs caused by ICIs, the concept of ‘immune-endocrine
synergy’ may be proposed, whereby severe hypothyroidism and
immune-mediated overlap myositis may act together to produce
unusually marked creatine kinase elevations and RM. Future studies
should further elucidate the shared genetic and immunological
mechanisms underlying multisystem irAEs and prioritize the
development of predictive biomarkers, thereby facilitating a
transition from reactive management to precision prevention. From a
clinical perspective, increased vigilance for irAEs during cancer
immunotherapy is warranted. From a diagnostic standpoint, timely
serologic testing is crucial, and particularly when muscle biopsy
is not feasible, a composite strategy that integrates targeted
autoantibody panels, EMG, muscle MRI and early specialist
consultation provides important practical guidance. Regarding
prevention and treatment, the clinical course of this case
underscores the need for proactive monitoring of cardiac enzymes
together with thyroid function during ICI therapy, and for the
timely initiation of immunomodulatory therapy and/or endocrine
replacement when an irAE is identified. The occurrence of a single
irAE should prompt consideration of potential overlapping events
and the early initiation of coordinated multi-organ monitoring and
multidisciplinary team-based management; such an approach may
reduce the risk of severe complications and enable timely,
pathophysiology-informed interventions that improve clinical
outcomes.
Acknowledgements
The authors acknowledge Dr Jianguo Xu from the
Department of Geriatric Medicine, Affiliated Hospital of Shandong
University of Traditional Chinese Medicine (Jinan, China) for his
expert guidance during manuscript revision and valuable discussions
on diagnostic and mechanistic issues.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
BC was involved in the conception and design of the
study and drafted the manuscript. BC QZe and QZh were involved in
the collection of data and clinical observations. BC and YL
analyzed and interpreted the data. BC and QZe created and revised
figures based on collected data. YL and SY acquired the medical
images. BC, XL and QZh provided medical advice on patient
treatment. YL and XL assisted in editing and revisions to the
article. BC and QZh were involved in the follow-up of the patient
after discharge from the hospital. BC, YL and QZh participated in
the follow-up of the patient after discharge. BC and XL checked and
confirmed the authenticity of the raw data. All authors confirmed
the authenticity of all original data, and have read and approved
the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Written informed consent was obtained from the
patient for the publication of this case report, including the
images, data and treatment protocols generated during the course of
treatment.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, artificial
intelligence tools (ChatGPT version 4.0; https://chatgpt.com/) were used to improve the
readability and language of the manuscript, and subsequently, the
authors revised and edited the content produced by the artificial
intelligence tools as necessary, taking full responsibility for the
ultimate content of the present manuscript.
References
|
1
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2024.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Haggstrom L, Chan WY, Nagrial A, Chantrill
LA, Sim HW, Yip D and Chin V: Chemotherapy and radiotherapy for
advanced pancreatic cancer. Cochrane Database Syst Rev.
12(CD011044)2024.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Riely GJ, Wood DE, Aisner DL, Loo BW Jr,
Axtell AL, Bauman JR, Bharat A, Chang JY, Desai A, Dilling TJ, et
al: NCCN guidelines® insights: Non-small cell lung
cancer, version 7.2025. J Natl Compr Canc Netw. 23:354–362.
2025.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Gao S, Ye K, Zhang Z, Wang W, Li J, Xu T
and Tan H: Polysaccharides of Pseudostellaria heterophylla (Miq.)
Pax ex Pax et Hoffm: Extraction, purification, structural
characteristics, pharmacological activities, and structure-activity
relationships: A review. Int J Biol Macromol.
330(148082)2025.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Gao S, Zhang Z, Ye K, Wang W, Li J, Xu T
and Tan H: Comprehensive characterization of Rubus idaeus L.
Polysaccharides: Extraction, purification, structural diversity,
biological efficacy, and structure-activity relationships. J
Ethnopharmacol. 355(120677)2026.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Jiang Y, Wang C, Zu C, Rong X, Yu Q and
Jiang J: Synergistic potential of nanomedicine in prostate cancer
immunotherapy: Breakthroughs and prospects. Int J Nanomedicine.
19:9459–9486. 2024.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Singh N, Hocking AM and Buckner JH:
Immune-related adverse events after immune check point inhibitors:
Understanding the intersection with autoimmunity. Immunol Rev.
318:81–88. 2023.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Zimmerman JL and Shen MC: Rhabdomyolysis.
Chest. 144:1058–1065. 2013.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Jayan A, Mammen AL and Suarez-Almazor ME:
Immune checkpoint inhibitor-induced myositis. Rheum Dis Clin North
Am. 50:281–290. 2024.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Plomp L, Chassepot H, Psimaras D,
Maisonobe T, Mensi E, Leonard-Louis S, Plu I, Rozes A, Tubach F,
Touat M, et al: Features of myositis and myasthenia gravis in
patients treated with immune checkpoint inhibitors: A multicentric,
retrospective cohort study. Lancet Reg Health Eur.
50(101192)2025.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Neppl C, Keller MD, Scherz A, Dorn P,
Schmid RA, Zlobec I and Berezowska S: Comparison of the 7 and 8th
edition of the UICC/AJCC TNM staging system in primary resected
squamous cell carcinomas of the lung-A single center analysis of
354 cases. Front Med (Lausanne). 6(196)2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Weng C, Ding Z, Zhou Y, Yang Q, Xue L,
Zhang L, Wang G and Liu Z: Clinical characteristics of
dermatomyositis with interstitial lung disease: A retrospective
case-control study. Rheumatol Ther. 10:635–648. 2023.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Li JX and Cummins CL: Fresh insights into
glucocorticoid-induced diabetes mellitus and new therapeutic
directions. Nat Rev Endocrinol. 18:540–557. 2022.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Klecha AJ, Barreiro Arcos ML, Frick L,
Genaro AM and Cremaschi G: Immune-endocrine interactions in
autoimmune thyroid diseases. Neuroimmunomodulation. 15:68–75.
2008.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Baghi MA, Sirajudeen J, Naushad VA, Alarbi
KS and Benshaban N: Severe hypothyroidism-induced rhabdomyolysis: A
case report. Clin Case Rep. 9(e05107)2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Bloise FF, Cordeiro A and Ortiga-Carvalho
TM: Role of thyroid hormone in skeletal muscle physiology. J
Endocrinol. 236:R57–R68. 2018.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Dentice M, Ambrosio R, Damiano V, Sibilio
A, Luongo C, Guardiola O, Yennek S, Zordan P, Minchiotti G, Colao
A, et al: Intracellular inactivation of thyroid hormone is a
survival mechanism for muscle stem cell proliferation and lineage
progression. Cell Metab. 20:1038–1048. 2014.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Aguiari P, Villani V, Liu KY, Brent GA,
Perin L and Milanesi A: 8670 Hypothyroidism impairs skeletal muscle
regeneration after injury. J Endocr Soc. 8 (Suppl
1)(bvae163.1812)2024.
|
|
19
|
Govindan R, Bogart J, Stinchcombe T, Wang
X, Hodgson L, Kratzke R, Garst J, Brotherton T and Vokes EE:
Randomized phase II study of pemetrexed, carboplatin, and thoracic
radiation with or without cetuximab in patients with locally
advanced unresectable non-small-cell lung cancer: Cancer and
Leukemia Group B trial 30407. J Clin Oncol. 29:3120–3125.
2011.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Zhang M, Zhang G, Niu Y, Zhang G, Ji Y,
Yan X, Zhang X, Wang Q, Jing X, Wang J, et al: Sintilimab with two
cycles of chemotherapy for the treatment of advanced squamous
non-small cell lung cancer: A phase 2 clinical trial. Nat Commun.
15(1512)2024.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Zhou C, Wu L, Fan Y, Wang Z, Liu L, Chen
G, Zhang L, Huang D, Cang S, Yang Z, et al: Sintilimab plus
platinum and gemcitabine as first-line treatment for advanced or
metastatic squamous NSCLC: Results from a randomized, double-blind,
phase 3 trial (ORIENT-12). J Thorac Oncol. 16:1501–1511.
2021.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Yasuda Y, Iwama S, Sugiyama D, Okuji T,
Kobayashi T, Ito M, Okada N, Enomoto A, Ito S, Yan Y, et al:
CD4+ T cells are essential for the development of
destructive thyroiditis induced by anti-PD-1 antibody in
thyroglobulin-immunized mice. Sci Transl Med.
13(eabb7495)2021.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Merino-Vico A, Kocyigit M, Frazzei G,
Landman L, Boon L, van Leeuwen EM, Lundberg IE, van der Kooi AJ,
Raaphorst J, van Hamburg JP and Tas SW: Modulating IL-21-driven B
cell responses in idiopathic inflammatory myopathies via inhibition
of the JAK/STAT pathway. Arthritis Res Ther. 27(76)2025.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Kuchroo JR, Hafler DA, Sharpe AH and Lucca
LE: The double-edged sword: Harnessing PD-1 blockade in tumor and
autoimmunity. Sci Immunol. 6(eabf4034)2021.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Francisco LM, Sage PT and Sharpe AH: The
PD-1 pathway in tolerance and autoimmunity. Immunol Rev.
236:219–242. 2010.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Pirkmajer S and Chibalin AV: Na,K-ATPase
regulation in skeletal muscle. Am J Physiol Endocrinol Metab.
311:E1–E31. 2016.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Nappi A, Moriello C, Morgante M, Fusco F,
Crocetto F and Miro C: Effects of thyroid hormones in skeletal
muscle protein turnover. J Basic Clin Physiol Pharmacol.
35:253–264. 2024.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Beecher G, Pinal-Fernandez I, Mammen AL
and Liewluck T: Immune checkpoint inhibitor myopathy: The
double-edged sword of cancer immunotherapy. Neurology.
103(e210031)2024.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Matas-García A, Milisenda JC,
Selva-O'Callaghan A, Prieto-González S, Padrosa J, Cabrera C,
Reguart N, Castrejón N, Solé M, Ros J, et al: Emerging PD-1 and
PD-1L inhibitors-associated myopathy with a characteristic
histopathological pattern. Autoimmun Rev. 19(102455)2020.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Hebert JF, Burfeind KG, Malinoski D and
Hutchens MP: Molecular mechanisms of rhabdomyolysis-induced kidney
injury: From bench to bedside. Kidney Int Rep. 8:17–29.
2022.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Sundarrajan C, Bhai S and Dimachkie MM:
Immune checkpoint inhibitor-related myositis: From pathophysiology
to treatment. Clin Exp Rheumatol. 41:379–385. 2023.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Shikano K, Kaneko K, Kaburaki K, Isobe K,
Kawabe K, Homma S, Kawai S and Nanki T: Nivolumab-induced
anti-aminoacyl-tRNA synthetase antibody-positive polymyositis
complicated by interstitial pneumonia in a patient with lung
adenocarcinoma. Scand J Rheumatol. 49:82–83. 2020.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Osaki M, Tachikawa R, Ohira J, Hara S and
Tomii K: Anti-transcriptional intermediary factor 1-γ
antibody-positive dermatomyositis induced by nivolumab for lung
adenocarcinoma: A case report. Invest New Drugs. 39:251–255.
2021.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Pilia AM, Salvati L, Guidolin A, Mazzoni
F, Antonuzzo L, Parronchi P and Liotta F: Pembrolizumab-associated
anti-MDA5 dermatomyositis in a patient with lung cancer: A first
case report. Swiss Med Wkly. 154(3513)2024.PubMed/NCBI View
Article : Google Scholar
|
|
35
|
Lundberg IE, Tjärnlund A, Bottai M, Werth
VP, Pilkington C, de Visser M, Alfredsson L, Amato AA, Barohn RJ,
Liang MH, et al: 2017 European league against rheumatism/American
college of rheumatology classification criteria for adult and
juvenile idiopathic inflammatory myopathies and their major
subgroups. Ann Rheum Dis. 76:1955–1964. 2017.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Kirou RA, Pinal-Fernandez I,
Casal-Dominguez M, Pak K, Preusse C, Dari D, Del Orso S, Naz F,
Islam S, Gutierrez-Cruz G, et al: Distinct cytokine and cytokine
receptor expression patterns characterize different forms of
myositis. medRxiv [Preprint]: Feb 21, 2025 (Epub ahead of
print).
|
|
37
|
Iwama S, Kobayashi T and Arima H:
Management, biomarkers and prognosis in people developing
endocrinopathies associated with immune checkpoint inhibitors. Nat
Rev Endocrinol. 21:289–300. 2025.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Yamauchi I and Yabe D: Best practices in
the management of thyroid dysfunction induced by immune checkpoint
inhibitors. Eur Thyroid J. 14(e240328)2025.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Sánchez-Camacho A, Torres-Zurita A,
Gallego-López L, Hernández-Pacheco R, Silva-Romeiro S, Álamo de la
Gala MDC, Peral-Gutiérrez de Ceballos E and de la Cruz-Merino L:
Management of immune-related myocarditis, myositis and myasthenia
gravis (MMM) overlap syndrome: A single institution case series and
literature review. Front Immunol. 16(1597259)2025.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Schneider BJ, Naidoo J, Santomasso BD,
Lacchetti C, Adkins S, Anadkat M, Atkins MB, Brassil KJ, Caterino
JM, Chau I, et al: Management of immune-related adverse events in
patients treated with immune checkpoint inhibitor therapy: ASCO
guideline update. J Clin Oncol. 39:4073–4126. 2021.PubMed/NCBI View Article : Google Scholar
|