Introduction
Postural orthostatic tachycardia syndrome (POTS) is
a disease defined by symptoms of orthostatic intolerance and a
heart rate increase of >30 bpm from a recumbent to a standing
position or >120 bpm in a standing position in the absence of
orthostatic hypotension (1,2). The
etiology and pathophysiology of POTS are unknown; however, signs of
autonomic neuropathy, such as orthostatic intolerance, pre-syncope
and palpitations, and small fiber neuropathy with intraepidermal
nerve fiber density less than the 5th percentile of normative
values, have previously been described (3).
Ehlers-Danlos syndrome (EDS) comprises 13 heritable
connective tissue disorders and is characterized by joint
hypermobility, skin hyperextensibility and tissue fragility, among
other clinical features (4). The
classification and diagnosis of EDS is based upon clinical
examination findings; however, a definite diagnosis is primarily
made based upon molecular determination, except in cases of
hypermobile EDS (hEDS), whereby the genetic variants remain unknown
(5). Furthermore, in patients with
symptomatic joint hypermobility who do not completely fulfill the
criteria for EDS or hEDS, the term hypermobility spectrum disorder
(HSD) is used (5,6). Genetically, EDS is heterogenous with
varying mutations of protein-coding genes, including genes coding
for collagen (5).
Histopathologically, EDS is characterized by disorganized collagen
fibers and fiber bundles (7,8). HSD
and hEDS are more frequent in patients with POTS compared with the
general population (9,10).
Gastrointestinal symptoms in POTS and HSD/hEDS occur
frequently, both as functional gastrointestinal disorders and as
more severe intestinal dysmotility disorders (10-12).
Dysmotility may be due to pathology in the nervous system, muscles
and/or connective tissue, (13)
and diagnosing severe gastrointestinal dysmotility is challenging,
as gastrointestinal motility varies widely among healthy
individuals, with full-thickness bowel biopsies only occasionally
being performed (11,14). Neuropathy is often generalized
(15) and thus, biopsy sampling
for diagnostic purposes should be prioritized in easily accessible
organs, such as the skin. Our previous studies have reported that
phase-contrast tomographic imaging with 3D illustration could
reveal disorganized dermal collagen fiber bundles in a patient with
POTS and hEDS and severe intestinal dysmotility, who also exhibited
a reduced intraepidermal nerve fiber density (16,17).
An additional patient with dysmotility exhibited an increased
amount of inter-fibrillar ground substance (17). Notably, Histomography®
GmbH has developed a laboratory-based micro-CT system, specifically
optimized for paraffin-embedded biopsy cores. This technique
enables detailed 3D structural analysis of soft tissue samples
(18,19).
Therefore, the primary aim of the present study was
to examine skin biopsies using virtual 3D histology to evaluate the
suitability of the method to describe the dermal architecture and
any presence of nerve fibers in a series of patients with POTS,
with or without hEDS/EDS, and in healthy controls. The secondary
aim was to relate the obtained dermal findings to clinical data and
gastrointestinal symptoms.
Materials and methods
Recruitment of participants and skin
biopsy procedure
A previously established cohort of 43 patients with
POTS and 61 healthy controls without gastrointestinal symptoms was
recruited from Skåne University Hospital (Lund, Sweden) between
October 2020 and January 2022(20). From this cohort, 38 patients and 13
healthy controls underwent skin biopsy sampling. POTS was diagnosed
based on cardiovascular autonomic tests with tilt testing and
continuous hemodynamic monitoring prior to study inclusion
(21). The inclusion criteria were
an age of 18-70 years with a diagnosis of POTS, ability to fully
understand the study information and a home base within a
reasonable proximity to Malmö, Sweden. Exclusion criteria were
severe somatic comorbidity or mental illness and alcohol and drug
abuse.
Among the patients with POTS, 12 patients also
exhibited concomitant hEDS/EDS, which was diagnosed based on
clinical examination by a physiotherapist or physician, without any
genetic characterization. After study inclusion, their medical
records were scrutinized and the types of diagnostic tests used,
classification of subtypes and concomitant diagnoses were
recorded.
A total of two skin biopsies were taken from
non-lesional skin 10 cm proximally to the lateral malleolus as a 3
mm punch biopsy during local subcutaneous anesthesia (0.5 ml
Carbocain®; 10 mg/ml). The skin defects were closed with
one 4-0 suture each and covered by a small dressing allowing for
free physical activities. Biopsy samples were immediately fixed in
4% buffered formaldehyde for ≥24 h at room temperature before
dehydration in alcohol and paraffin-embedding according to clinical
routines in the accredited hospital laboratory (16). Furthermore, one sample from each
participant was then harvested for 3D imaging with a 1.5 mm punch
using a dissection microscope and placed into a Kapton®
tube (Paramount Tube), with a biopsy length of 2-5 mm. All other
paraffin-embedded blocks were sectioned at 5 µm and
immunohistochemically stained with a rabbit polyclonal protein gene
product 9.5 (PGP9.5) antibody (cat. no. 318A-1; dilution, 1:3,000;
Cell Marque™), according to the manufacturer's protocol.
The biopsies were examined in a Sectra IDS7 workstation (Sectra
AB), where the scanned tissue sections could be viewed and examined
microscopically according to clinical routines (22).
Virtual 3D histology
For 3D imaging, the prepared tissue samples were
sent to Histomography® GmbH. The analysis was conducted
using X-rays in a laboratory micro-CT system specifically optimized
for small formalin-fixed, paraffin-embedded core biopsies with a
diameter of 1.5 mm. Imaging of soft tissue with X-rays presents a
marked challenge due to its low X-ray absorption, especially
compared with established applications in denser materials such as
bone or metal (23). Exploiting
the self-interference of partially coherent X-rays from laboratory
X-ray sources offers a viable approach to address this issue. As
the X-rays pass through the sample, minimal density variations
induce phase shifts in the radiation. These phase shifts create
interference patterns, which manifest on the detector image (as
enhanced edge contrasts). Phase-contrast imaging was combined with
advanced phase-retrieval algorithms and iterative tomographic
reconstruction methods to markedly enhance image contrast, making
it suitable for visualizing fine structures within soft tissues
(18,19). The resulting 3D volumes had an
isotropic voxel size of 840 nm. With a dynamic range of 16 bits,
each dataset was ~10 GB in size, enabling detailed inspection of 3D
tissue morphology. The datasets were subsequently provided through
a browser-based volumetric viewer solution developed by
Histomography® GmbH. The viewer enables interactive
visualization of large 3D datasets and allows users to stream and
interactively explore the data, including inspecting virtual slices
of the 3D volume from numerous orientations and measurement of
tissue structures in 3D, facilitating a comprehensive examination
of tissue architecture (www.histomography.com).
Histopathological evaluation
Only one sample from a patient with POTS with hEDS
was excluded as the final punching step of the tissue sample
failed. All samples were pseudonymized and evaluated blinded to the
examinator in a semi-quantitative manner by two independent
examiners. After evaluation, biopsies that were differently
evaluated by the two examiners (n=5) were discussed and a consensus
was reached concerning the classification of the biopsies. The
overall histology of the samples was studied to identify the
different layers and structures. Each skin layer was clearly
visible, including the horn layer, squamous epithelium,
dermo-epidermal junction, adnexal structure in the dermal layer and
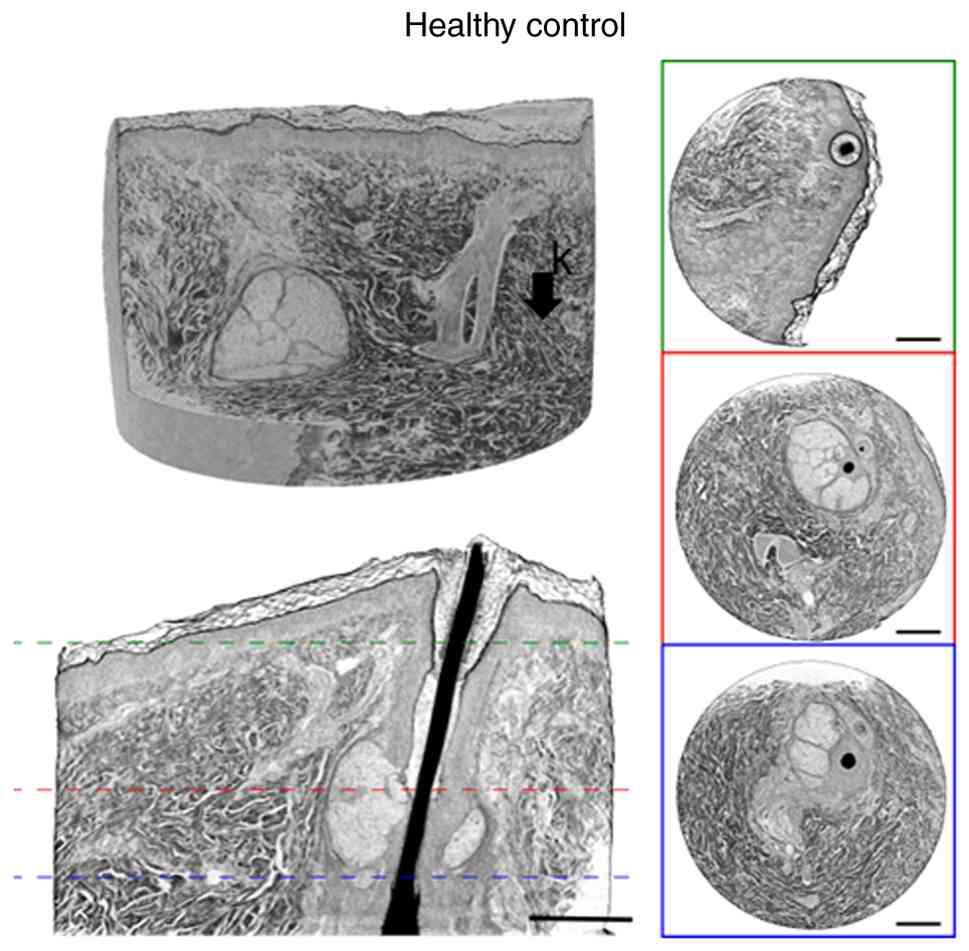
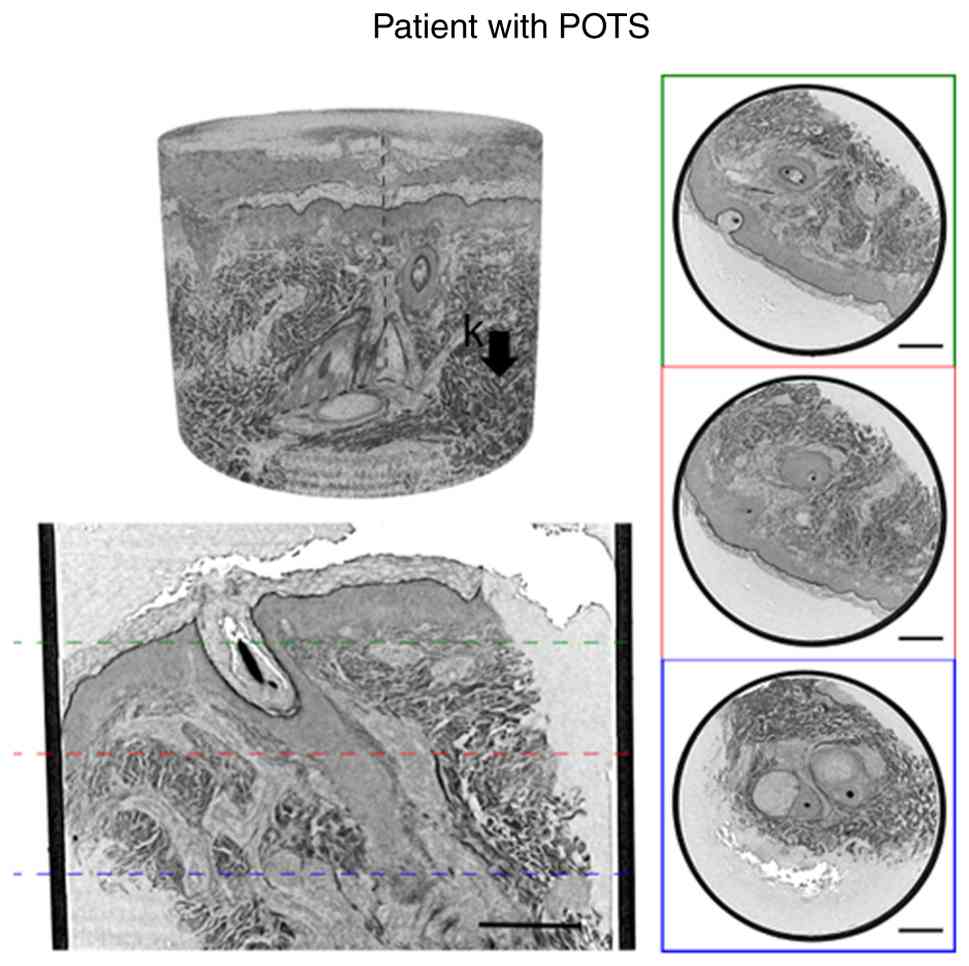
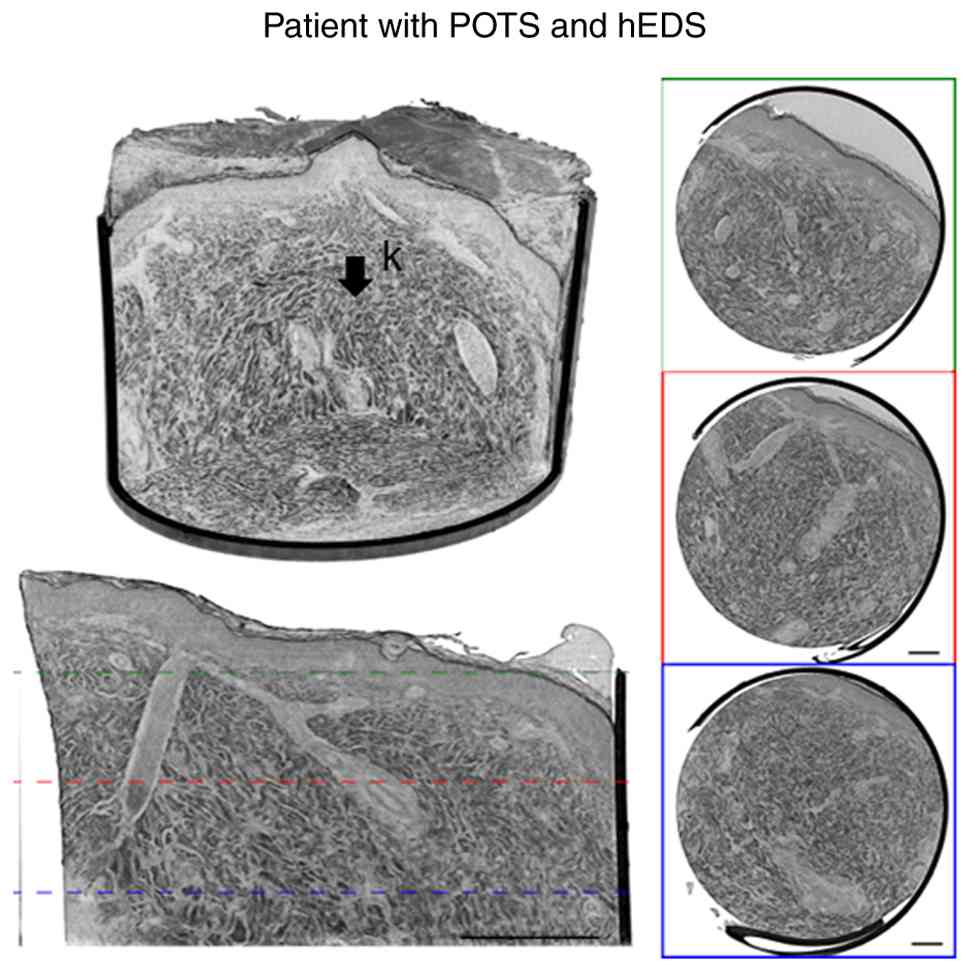
subcutaneous fat (Fig. 1). Any
presence of intraepidermal nerve fibers was examined.
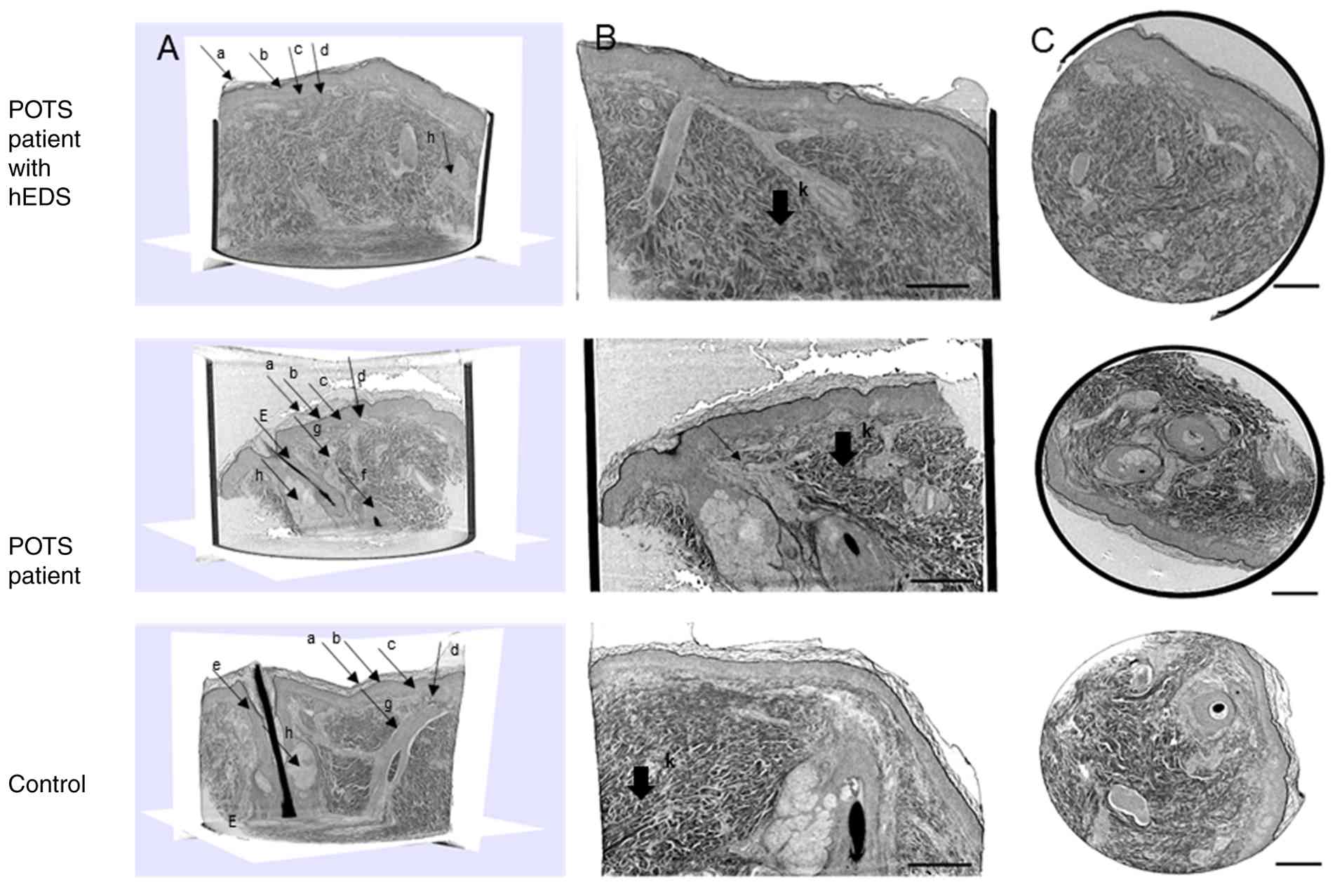 | Figure 1Skin tissue punch biopsy from patients
with POTS, with and without hEDS, and 1 control, showing a scanned
vertical overview. (A) Overview of skin biopsies. (B) The same
scans with higher magnification illustrating the disorganized
collagen fiber bundles throughout the biopsy in the patients with
and without hEDS and more organized parallel fiber bundles
throughout the biopsy in the control. (C) A transversal
cross-section of the data set. Scale bars, 250 µm. a, stratum
corneum; b, stratified granulosum; c, stratum spinosum; d, dermo
epidermal junction; e, hair follicle with hair shaft; f, follicle
bulb; g, arrector pili; h, sebaceous gland; k, collagen fiber
bundles. hEDS, hypermobile Ehler-Danlos syndrome; POTS, postural
orthostatic tachycardia syndrome. |
The collagen fiber bundles in the dermal skin tissue
were evaluated in 3D throughout the biopsy. The collagen
arrangement was assessed qualitatively by visual inspection by
trained pathologists and no quantitative metrics were applied. The
degree of parallel or disorganized collagen fiber bundles was
evaluated in the dermis and categorized into three groups: i)
Parallel bundles throughout the biopsy; ii) parallel bundles
superficially and disorganized bundles in deeper layers; or iii)
disorganized bundles throughout the biopsy. Furthermore, the
thickness of collagen fiber bundles and presence of loosely
organized fiber bundles with more ground substance between the
bundles was estimated. Fiber bundles were subjectively divided into
two categories: Thick or thin bundles; and loosely or tightly
organized bundles. After the examination was finished, the code
list of pseudo-anonymized samples was opened, and samples could be
related to the participant, to identify the correct group
identity.
Questionnaires
All study participants were asked to complete a
questionnaire regarding previous and current illnesses, family
history and current pharmacological treatment. Furthermore, the
validated visual analog scale for irritable bowel syndrome
(VAS-IBS) was used to estimate the influence of abdominal pain,
diarrhea, constipation, bloating and flatulence, vomiting and
nausea, psychological well-being and intestinal symptoms on daily
life using scales from 0-100 mm (whereby 0 mm represents no
symptoms, and 100 mm represents severe symptoms). The scales were
inverted from their original format (24). Reference values are available from
healthy controls (25). The
validated IBS-severity scoring system (IBS-SSS) was used to
estimate abdominal pain, abdominal distension, satisfaction with
bowel habits and the impact of bowel habits on daily life using
visual analog scales (VASs) ranging from absent (0 mm) to very
severe (100 mm) symptoms. The number of days with abdominal pain in
the last 10 days was reported. The overall maximum achievable score
was 500. Scores ranging between 75-174 indicated mild disease,
scores between 175-299 indicated moderate disease and scores ≥300
indicated severe disease. Extraintestinal symptoms (nausea,
difficulties eating a whole meal, headache, back pain, fatigue,
belching/excess wind, reflux, urinary urgency, leg pain and
muscle/joint pain) were also estimated on VASs with a maximal
achievable score of 500(26).
Statistical analysis
Statistical analyses were performed in SPSS (version
29; IBM Corp.). Differences between groups were compared using the
non-parametric Mann-Whitney U test and Fisher's exact tests.
P-values adjusted with Bonferroni correction due to multiple tests
were given B values and were taken as the main result. A one-sample
test was used to calculate differences in proportions of collagen
bundles within each group of participants. Values are presented as
the median (interquartile range) and number (percentage). P (or B
when applicable) <0.05 was considered to indicate a
statistically significant difference.
Results
Basal characteristics
All patients with POTS were divided into patients
without hEDS/EDS (n=26) and patients with concomitant hEDS/EDS
(n=11) groups for separate comparisons with the controls (n=13). Of
the patients with concomitant hEDS/EDS, 6 patients were diagnosed
with hEDS and 5 patients were diagnosed with unspecific EDS.
Healthy controls had a higher education level, had less sick leave,
drank more alcohol and were less physically active compared with
POTS patients without hEDS/EDS (Table
I).
 | Table IBasal characteristics and symptoms in
healthy controls compared with patients with POTS with or without
concomitant hEDS/EDS. |
Table I
Basal characteristics and symptoms in
healthy controls compared with patients with POTS with or without
concomitant hEDS/EDS.
| Characteristic | Healthy controls
(n=13) | POTS (n=26) | POTS with hEDS/EDS
(n=11) |
P/B-valuea |
P/B-valueb |
|---|
| Age, years | 39 (36-41) | 31 (27-44) | 28 (28-40) | 0.216/0.432 | 0.045/0.090 |
| Sex, n (%) | | | | 1.000/1.000 | 0.482/0.964 |
|
Female | 11 (84.6) | 23 (88.5) | 11 (100.0) | | |
|
Male | 2 (15.4) | 3 (11.5) | 0 (0.0) | | |
| BMI,
kg/m2 | 21.7
(20.6-24.0) | 24.6
(21.3-26.6) | 24.1
(21.6-29.6) | 0.188/0.376 | 0.167/0.334 |
| Education, n
(%) | | | | 0.009/0.018 | 0.018/0.036 |
|
Primary
school | | 2 (7.7) | 1 (9.1) | | |
|
College | | 8 (30.8) | 4 (36.4) | | |
|
≥1 more year
of education | 1 (7.7) | 6 (23.1) | 1 (9.1) | | |
|
University | 12 (92.3) | 10 (38.5) | 5 (45.5) | | |
| Occupation, n
(%) | | | |
<0.001/0.002 | 0.002/0.004 |
|
Working
100% | 8 (61.5) | 4 (15.4) | 1 (9.1) | | |
|
Working
99-51% | 5 (38.5) | 3 (11.5) | 2 (18.2) | | |
|
Sick
leave | | 13 (50.0) | 5 (45.5) | | |
|
Unemployment | | 2 (7.7) | 1 (9.1) | | |
|
Studying | | 4 (15.4) | 2 (18.2) | | |
| Marital status, n
(%) | | | | 0.276/0.552 | 0.033/0.066 |
|
Living
alone | 2 (15.4) | 9 (34.6) | 7 (63.6) | | |
|
Married/living
together | 11 (84.6) | 17 (65.4) | 4 (36.4) | | |
| Smoking, n (%) | | | | 0.557/1.000 | 0.845/1.000 |
|
Regularly | 1 (7.7) | 3 (11.5) | 2 (18.2) | | |
|
Former
smoker | 3 (23.1) | 2 (7.7) | 2 (18.2) | | |
|
Never
smoker | 9 (69.2) | 21 (80.8) | 7 (63.6) | | |
| Glasses of
alcohol/weekc, n
(%) | | | |
<0.001/0.002 |
<0.001/0.002 |
|
<1
glass | 1 (7.7) | 22 (84.6) | 10 (90.9) | | |
|
1-4
glasses | 10 (76.9) | 4 (15.4) | | | |
|
5-9
glasses | 2 (15.4) | | 1 (9.1) | | |
| Physical
activity/weekd, n
(%) | | | | 0.010/0.020 | 0.927/1.000 |
|
No time at
all | | 6 (23.1) | 1 (9.1) | | |
|
<30
min | 2 (15.4) | 2 (7.7) | 3 (27.3) | | |
|
30-60
min | 6 (46.2) | 3 (11.5) | 4 (36.4) | | |
|
60-90
min | 2 (15.4) | 10 (38.5) | 1 (9.1) | | |
|
90-120
min | 2 (15.4) | | 2 (18.2) | | |
|
>120
min | 1 (7.7) | 5 (19.2) | | | |
| Gastrointestinal
symptome | | | | | |
|
Abdominal
pain | 0 (0-0) | 30 (6-54) | 63 (17-77) |
<0.001/0.002 |
<0.001/0.002 |
|
Diarrhea | 0 (0-0) | 10 (0-61) | 51 (0-72) |
<0.001/0.002 | 0.003/0.006 |
|
Constipation | 0 (0-16) | 24 (2-72) | 73 (34-91) | 0.006/0.012 |
<0.001/0.002 |
|
Bloating and
flatulence | 3 (0-16) | 73 (15-88) | 62 (25-90) |
<0.001/0.002 | 0.009/0.018 |
|
Vomiting and
nausea | 0 (0-6) | 36 (12-69) | 48 (30-70) |
<0.001/0.002 |
<0.001/0.002 |
|
Intestinal
symptoms' influence on daily life | 0 (0-2) | 49 (18-74) | 74 (19-85) |
<0.001/0.002 |
<0.001/0.002 |
|
Psychological
well-being | 0 (0-32) | 48 (12-60) | 57 (35-79) | 0.008/0.016 | 0.001/0.002 |
|
Difficulties
eating a meal | 0 (0-0) | 25 (0-52) | 55 (5-79) |
<0.001/0.002 |
<0.001/0.002 |
|
Headache | 7 (2-24) | 78 (44-94) | 67 (48-80) |
<0.001/0.002 |
<0.001/0.002 |
|
Back
pain | 7 (0-28) | 50 (13-77) | 50 (35-83) | 0.003/0.006 |
<0.001/0.002 |
|
Fatigue | 10 (0-22) | 94 (79-100) | 92 (85-100) |
<0.001/0.002 |
<0.001/0.002 |
|
Belching/excess
wind | 0 (0-2) | 55 (22-89) | 71 (36-81) |
<0.001/0.002 |
<0.001/0.002 |
|
Reflux | 0 (0-0) | 17 (2-58) | 59 (23-71) |
<0.001/0.002 |
<0.001/0.002 |
|
Urinary
urgency | 0 (0-3) | 54 (8-85) | 63 (23-82) |
<0.001/0.002 |
<0.001/0.002 |
|
Leg
pain | 0 (0-0) | 8 (2-61) | 44 (0-51) |
<0.001/0.002 |
<0.001/0.002 |
|
Muscle/joint
pain | 0 (0-6) | 68 (15-84) | 90 (65-97) |
<0.001/0.002 |
<0.001/0.002 |
| Total IBS-SSS | 8 (0-41) | 204 (96-297) | 288 (139-409) |
<0.001/0.002 |
<0.001/0.002 |
| Total
extraintestinal IBS-SSS | 38 (32-86) | 485 (344-633) | 675 (473-708) |
<0.001/0.002 |
<0.001/0.002 |
Comorbidities were frequent among these patients
with POTS, whereby irritable bowel syndrome was most frequently
reported (n=7), followed by asthma (n=6), thyroid disorders (n=5),
migraine (n=4), myalgic encephalomyelitis (n=3), dyspepsia (n=2),
gastroparesis (n=2), fibromyalgia (n=2), post-coronavirus disease
19 (n=2), mast cell activation syndrome (n=2) and inappropriate
sinus tachycardia (n=2) (data not shown). All specific
gastrointestinal symptoms and extraintestinal symptoms, as well as
total scores, were higher in patients with POTS without hEDS/EDS
compared with controls, with B=0.002, except for constipation
(B=0.012), psychological well-being (B=0.016) and back pain
(B=0.006; Table I).
Upon comparison of healthy controls and patients
with POTS with concomitant hEDS/EDS, control patients tended to be
older and more often married or living together, with a higher
education level and less sick leave taken. The controls also drank
more alcohol compared with all other patients (Table I). Apart from hEDS/EDS, the
patients also suffered from one or more autism spectrum disorders
(n=4), myalgic encephalomyelitis (n=3), anxiety (n=2), depression
(n=2), endometriosis (n=2), fibromyalgia (n=2), gastrointestinal
dysmotility (n=2), sleeping disturbances (n=2), migraine (n=1) and
mast cell activation syndrome (n=1) (data not shown). All specific
gastrointestinal symptoms and extraintestinal symptoms, as well as
total scores, were higher in patients with hEDS/EDS compared with
controls, with B=0.002, except for diarrhea (B=0.006) and bloating
and flatulence (B=0.018; Table I).
There were no statistically significant differences in symptoms
between patients with POTS with and without concomitant hEDS/EDS
(data not shown).
Histopathology
Through the user-friendly and easy to navigate
browser-based tool, provided by Histomography® GmbH,
skin layers in 3D were scrutinized. The magnification gave a clear
overview of the various tissue components in the skin biopsy
(Fig. 1). The three layers
epidermis, dermis and subcutis were studied in a vertical view. In
the epidermis, the different layers stratum corneum, stratum
granulosum, stratum spinosum and stratum basales could be
separated. The dermo-epidermal junction appeared rather distinct.
In the dermal layer, collagen bundles and hair follicles with hair
shaft together with adnexal structures such as arrector pili and
sebaceous glands were visible (Fig.
1A). There were no differences in adnexal structures between
the patients and controls. Disorganized collagen fiber bundles were
visualized in 2 patients with and without hEDS, in contrast to the
organized parallel fiber bundles in a healthy participant (Fig. 1A-C). The same samples were also
visualized at different transversal depths (Fig. 2, Fig.
3 and Fig. 4). The
magnification was not sufficient to separate varying cell types and
for evaluation of intracellular changes in the skin. Neither was it
possible to identify any intraepidermal nerve fibers.
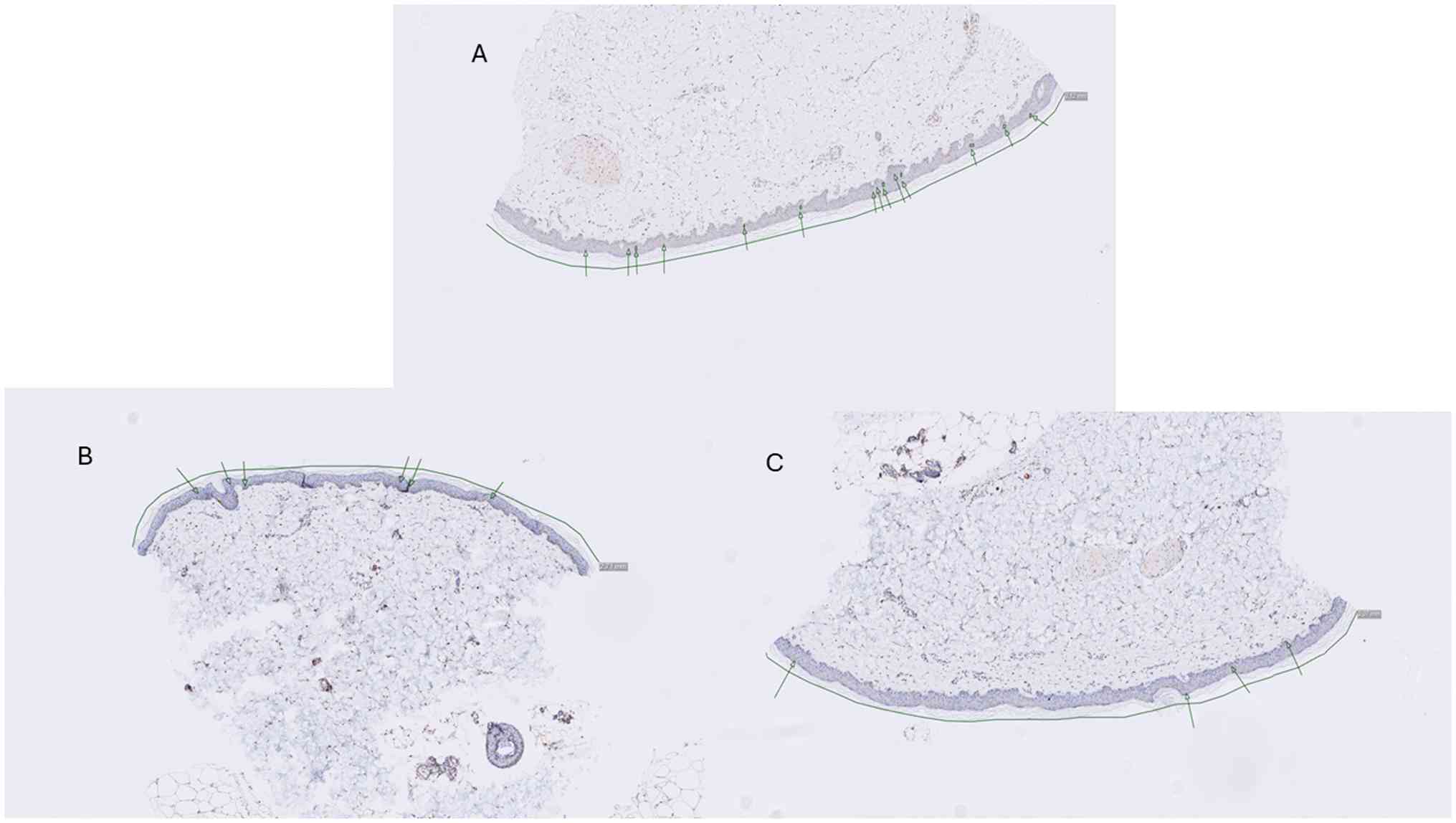
Conventional immunohistochemistry using a PGP9.5
antibody was conducted and a representative image is shown for
comparison with the micro-CT from 2 patients with POTS, 1 with hEDS
and 1 without hEDs, and 1 healthy control, which demonstrated the
different layers of the skin and adnexal structures (Fig. 5).
Collagen fiber bundles
Table II shows the
distribution of the three categories of collagen fiber bundles: i)
Parallel collagen fiber bundles throughout the biopsy; ii)
superficial parallel fiber bundles with disorganized fiber bundles
in deeper layers; and iii) disorganized fiber bundles throughout
the biopsy. The number of participants with disorganized bundles
tended to differ among the three groups of participants (controls,
patients with POTS and patients with POTS and hEDS/EDS; P=0.056),
due to primarily disorganized bundles exhibited by the hEDS/EDS
group. The number of patients in the three fiber organization
categories tended to differ between patients with only POTS and
patients with POTS and hEDS/EDS (B=0.057).
 | Table IIPrevalence of parallel or
disorganized collagen fiber bundles in healthy controls compared
with POTS patients with or without hEDS/EDS. |
Table II
Prevalence of parallel or
disorganized collagen fiber bundles in healthy controls compared
with POTS patients with or without hEDS/EDS.
| Collagen
fibers | Healthy controls
(n=13) | POTS (n=26) | hEDS/EDS
(n=11) | P-value |
P/B-valuea |
P/B-valueb |
P/B-valuec |
|---|
| Bundles divided
into three categories | | | | 0.056 | 0.461/1.000 | 0.144/0.432 | 0.019/0.057 |
|
Category 1,
parallel bundles throughout the biopsy | 4 (30.8) | 4 (15.4) | 1 (9.1) | | | | |
|
Category 2,
parallel bundles superficially and disorganized deeply | 4 (30.8) | 13 (50.0) | 1 (9.1) | | | | |
|
Category 3,
disorganized bundles throughout the biopsy | 5 (38.5) | 9 (34.6) | 9 (81.8) | | | | |
| Bundles divided
into two categories | | | | 0.030 | 1.000/1.000 | 0.047/0.141 | 0.013/0.039 |
|
Parallel
bundles, at least superficially (categories 1 + 2) | 8 (61.5) | 17 (65.4) | 2 (18.2) | | | | |
|
Disorganized
bundles throughout the biopsy (category 3) | 5 (38.5) | 9 (34.6) | 9 (81.8) | | | | |
| Bundles divided
into two categories | | | | 0.384 | | | |
|
Parallel
bundles throughout the biopsy (category 1) | 4 (30.8) | 4 (15.4) | 1 (9.1) | | | | |
|
Disorganized
bundles, at least deeply (categories 2 and 3) | 9 (69.2) | 22 (84.6) | 10 (90.9) | | | | |
There was a significant difference among the three
groups in the number of patients with parallel bundles throughout
the biopsy or at least superficially (categories 1 and 2) compared
with complete disorganized bundles (category 3) (P=0.030). Healthy
controls and patients with POTS exhibited a more similar bundle
distribution compared with patients with hEDS/EDS, with a
significant difference between patients with only POTS and patients
with POTS and hEDS/EDS (B=0.039; Table II).
One-sample test was used to calculate differences in
proportions of different collagen bundles within each study group.
The proportion of controls with complete disorganized bundles
(category 3) did not differ compared with the proportion of
controls with partly or complete parallel bundles (38.5 vs. 61.5%;
P=0.427). Neither was there any difference between the proportion
of any disorganized bundles (categories 2 and 3) and parallel
bundles throughout the biopsy (69.2 vs. 30.8%; P=0.175). Within the
POTS group without hEDS/EDS, the proportion of patients with
complete disorganized bundles did not differ from the proportion of
patients with parallel bundles at least superficially (34.6 vs.
65.4%; P=0.118), however proportions differed between patients with
any disorganized bundles and patients with parallel bundles
throughout the biopsy (84.6 vs. 15.4%; P<0.001). In patients
with POTS and hEDS/EDS, the proportion of patients with complete
disorganized bundles differed significantly from the proportion of
patients with parallel bundles at least superficially (81.8 vs.
18.2%; P=0.026). The difference was more pronounced when comparing
the proportions of patients with any disorganized bundles and
patients with parallel bundles throughout the biopsy (90.9 vs.
9.1%; P<0.001). There were no differences in terms of the
thickness of the bundles (P=1.000) or the structure of tight or
loose bundles (P=0.872) between the groups (data not shown).
Clinical features stratified by fiber
bundle structure
Participants with parallel fiber bundles at least in
the superficial layers (n=27) did not differ in sociodemographic
factors or lifestyle habits compared with those with disorganized
fiber bundles (n=23). Although participants with disorganized
bundles exhibited more symptoms of, fpr example, abdominal pain,
constipation, headache, muscle/joint pain, total IBS-SSS and total
extraintestinal IBS-SSS, the difference between groups did not
reach statistical significance (Table III).
 | Table IIIBasal characteristics and symptoms
depending on parallel, at least superficially or disorganized fiber
bundles. |
Table III
Basal characteristics and symptoms
depending on parallel, at least superficially or disorganized fiber
bundles.
| Characteristic | Parallel bundles
(n=27) | Disorganized
bundles (n=23) | P-value |
|---|
| Age, years | 39 (28-45) | 32 (28-40) | 0.259 |
| Sex, n (%) | | | 0.167 |
| Female | 26 (96.3) | 19 (82.6) | |
| Male | 1 (3.7) | 4 (17.4) | |
| BMI,
kg/m2 | 21.7
(20.7-26.1) | 23.5
(21.6-26.5) | 0.396 |
| Education, n
(%) | | | 0.785 |
|
Primary
school | 2 (7.4) | 1 (4.3) | |
|
College | 5 (18.5) | 7 (30.4) | |
|
≥1 more year
of education | 5 (1.5) | 3 (13.0) | |
|
University | 15 (55.6) | 12 (52.2) | |
| Occupation, n
(%) | | | 0.244 |
|
Working
100% | 7 (25.9) | 6 (26.1) | |
|
Working
99-51% | 6 (22.2) | 4 (17.4) | |
|
Sick
leave | 9 (33.3) | 9 (39.1) | |
|
Unemployment | | 3 (13.0) | |
|
Studying | 5 (18.5) | 1 (4.3) | |
| Marital status, n
(%) | | | 0.559 |
|
Living
alone | 11 (40.7) | 7 (30.4) | |
|
Married/living
together | 16 (59.3) | 16 (69.6) | |
| Smoking, n (%) | | | 0.788 |
|
Regularly | 3 (11.1) | 3 (13.0) | |
|
Former
smoker | 3 (11.1) | 4 (17.4) | |
|
Never
smoker | 21 (77.8) | 16 (69.6) | |
| Glasses of
alcohol/weeka, n
(%) | | | 0.571 |
|
<1
glass | 17 (63.0) | 16 (69.6) | |
|
1-4
glasses | 9 (33.3) | 5 (21.7) | |
|
5-9
glasses | 1 (3.7) | 2 (8.7) | |
| Physical
activity/weekb, n
(%) | | | 0.147 |
|
No time at
all | 3 (11.1) | 4 (17.4) | |
|
<30
min | 2 (7.4) | 5 (21.7) | |
|
30-60
min | 6 (22.2) | 7 (30.4) | |
|
60-90
min | 8 (29.6) | 5 (21.7) | |
|
90-120
min | 2 (7.4) | 2 (8.7) | |
|
>120
min | 6 (22.2) | | |
| Gastrointestinal
symptomc | | | |
|
Abdominal
pain | 11 (0-52) | 30 (0-62) | 0.450 |
|
Diarrhea | 0 (0-54) | 10 (0-64) | 0.325 |
|
Constipation | 15 (0-65) | 51 (2-76) | 0.160 |
|
Bloating and
flatulence | 50 (1-83) | 62 (13-81) | 0.435 |
|
Vomiting and
nausea | 31 (0-64) | 29 (6-57) | 0.797 |
|
Intestinal
symptoms' influence on daily life | 30 (0-74) | 38 (13-77) | 0.322 |
|
Psychological
well-being | 37 (2-61) | 35 (10-57) | 0.843 |
|
Difficulties
eating a meal | 5 (0-52) | 12 (0-54) | 0.722 |
|
Headache | 38 (9-81) | 69 (34-80) | 0.285 |
|
Back
pain | 32 (0-71) | 40 (9-70) | 0.724 |
|
Fatigue | 84 (20-95) | 90 (49-100) | 0.190 |
|
Belching/excess
wind | 40 (0-87) | 36 (2-76) | 0.796 |
|
Reflux | 6 (0-38) | 22 (0-70) | 0.461 |
|
Urinary
urgency | 19 (0-82) | 23 (6-76) | 0.649 |
|
Leg
pain | 4 (0-51) | 2 (0-50) | 0.807 |
|
Muscle/joint
pain | 22 (0-84) | 56 (19-90) | 0.304 |
| Total IBS-SSS | 96 (4-286) | 188 (42-315) | 0.272 |
| Total
extraintestinal IBS-SSS | 361 (40-629) | 410 (141-636) | 0.496 |
Participants with parallel fiber bundles throughout
the biopsy were significantly older compared with the remaining
participants with disorganized fiber bundles at least in deeper
layers (P=0.006). Furthermore, participants with disorganized
bundles exhibited significantly more pronounced symptoms of muscle
and joint pain compared with those with parallel bundles throughout
the biopsy (P=0.031). No other parameters reached statistical
significance (Table IV).
 | Table IVBasal characteristics and symptoms
depending on parallel bundles throughout the biopsy or disorganized
fiber bundles. |
Table IV
Basal characteristics and symptoms
depending on parallel bundles throughout the biopsy or disorganized
fiber bundles.
| Characteristic | Parallel bundles
(n=9) | Disorganized
bundles (n=41) | P-value |
|---|
| Age, years | 41 (39-44) | 31 (27-40) | 0.006 |
| Sex, n (%) | | | 0.570 |
|
Female | 9 (100.0) | 36 (87.8) | |
|
Male | 0 (0.0) | 5 (12.2) | |
| BMI,
kg/m2 | 21.3
(20.5-27.6) | 23.5
(21.4-26.0) | 0.567 |
| Education, n
(%) | | | 0.209 |
|
Primary
school | | 3 (7.3) | |
|
College | 1 (11.1) | 11 (26.8) | |
|
≥1 more year
of education | | 8 (19.5) | |
|
University | 8 (88.9) | 19 (46.3) | |
| Occupation, n
(%) | | | 0.652 |
|
Working
100% | 2 (22.2) | 11 (26.8) | |
|
Working
99-51% | 3 (33.3) | 7 (17.1) | |
|
Sick
leave | 4 (44.4) | 14 (34.1) | |
|
Unemployment | | 3 (7.3) | |
|
Studying | | 6 (14.6) | |
| Marital status, n
(%) | | | 1.000 |
|
Living
alone | 3 (33.3) | 15 (36.6) | |
|
Married/living
together | 6 (66.7) | 26 (63.4) | |
| Smoking, n (%) | | | 0.856 |
|
Regularly | | 6 (14.6) | |
|
Former
smoker | 1 (11.1) | 6 (14.6) | |
|
Never
smoker | 8 (88.9) | 29 (70.7) | |
| Glasses of
alcohol/weeka, n
(%) | | | 0.167 |
|
<1
glass | 4 (44.4) | 29 (70.7) | |
|
1-4
glasses | 5 (55.6) | 9 (22.0) | |
|
5-9
glasses | | 3 (7.3) | |
| Physical
activity/weekb, n
(%) | | | 0.708 |
|
No time at
all | | 7 (17.1) | |
|
<30
min | 1 (11.1) | 6 (14.6) | |
|
30-60
min | 4 (44.4) | 9 (22.0) | |
|
60-90
min | 2 (22.2) | 11 (26.8) | |
|
90-120
min | 1 (11.1) | 3 (7.3) | |
|
>120
min | 1 (11.1) | 5 (12.2) | |
| Gastrointestinal
symptomc | | | |
|
Abdominal
pain | 0 (0-37) | 25 (0-62) | 0.107 |
|
Diarrhea | 0 (0-56) | 5 (0-59) | 0.369 |
|
Constipation | 5 (0-42) | 25 (2-74) | 0.136 |
|
Bloating and
flatulence | 16 (0-82) | 56 (5-82) | 0.496 |
|
Vomiting and
nausea | 11 (0-72) | 30 (5-57) | 0.595 |
|
Intestinal
symptoms' influence on daily life | 0 (0-78) | 38 (4-74) | 0.311 |
|
Psychological
well-being | 50 (0-68) | 35 (10-59) | 0.926 |
|
Difficulties
eating a meal | 0 (0-41) | 12 (0-54) | 0.210 |
|
Headache | 15 (5-74) | 69 (24-82) | 0.085 |
|
Back
pain | 27 (0-48) | 43 (8-74) | 0.226 |
|
Fatigue | 79 (6-91) | 89 (48-99) | 0.149 |
|
Belching/excess
wind | 0 (0-86) | 40 (2-78) | 0.485 |
|
Reflux | 0 (0-34) | 15 (0-56) | 0.231 |
|
Urinary
urgency | 0 (0-46) | 38 (2-82) | 0.168 |
|
Leg
pain | 0 (0-28) | 4 (0-50) | 0.159 |
|
Muscle/joint
pain | 0 (0-51) | 65 (40-297) | 0.031 |
| Total IBS-SSS | 54 (4-204) | 177 (40-297) | 0.170 |
| Total
extraintestinal IBS-SSS | 137 (38-623) | 469 (134-634) | 0.238 |
Discussion
Within the present study, the main findings
indicated that the structures of the different layers in the skin
could be visualized by 3D virtual histology using X-ray
phase-contrast micro-CT. No intraepidermal nerve fibers could be
visualized. The percentage of disorganized collagen fiber bundles
was similar between healthy controls and patients with POTS but
different in POTS with hEDS/EDS and the percentage of disorganized
fibers in patients with concomitant hEDS/EDS differed from that in
patients with only POTS. Furthermore, the proportion of
participants with any disorganized collagen bundles and parallel
bundles throughout the biopsy differed within both patient groups
but not within the control group. However, a number of clinical
differences were observed between participants with parallel and
disorganized collagen fiber bundles.
The first EDS classification in 1998 assumed that
all six EDS subtypes (or the majority of) were a consequence of
alterations in fibrillar collagen genes or genes encoding collagen
modifiers (27). Since then,
numerous new EDS subtypes have been described (4,5).
Using next-generation sequencing technologies, further mutations
have been identified in genes not directly involved in collagen
biosynthesis and/or structure, leading to the revised
classification system with 13 EDS subtypes (5). Due to the vast genetic heterogeneity
and phenotype variability of EDS subtypes, the definite diagnosis
must rely on molecular determination of causative variants to
optimize treatment and research purposes (5). The present patients were not examined
by genotyping, as it cannot currently be used to diagnose hEDS,
since genetic variants are unknown (5). Theoretically, the present patients
with POTS with EDS and normal collagen bundles may have also had
subtypes of EDS involving molecular components other than collagen
or collagen-regulating factors.
Diagnoses based on subjective symptom reports and
clinical examination may be difficult to evaluate and only those
clinicians with specialist knowledge of in HSD, including EDS, may
be able to correctly diagnose EDS according to the established
guidelines (5). Therefore,
histopathological findings may improve the ability to make a
diagnosis and separate different entities, such as functional
disorders and EDS. Full-thickness biopsies of the bowel to diagnose
dysmotility are only chosen in selected cases, since they are
performed under anesthesia which may be complicated by bowel
rupture with severe complications (11). Histopathologically, EDS is
characterized by insufficient contexture of the collagen elements
in the cutis, subcutis and joint capsules (7). Despite disorderly arranged collagen
bundles being a typical characteristic, there may be large
interindividual differences in the magnitude of the ultrastructural
changes (28). Electron microscopy
(EM) is needed to study the structure of collagen fibrils, however
the irregularity of collagen fibers and bundles is also observed
using light microscopy (7,8). A number of stains may be used to
visualize collagen and fibroblasts (8), yet in EM, there is no specific stain
for this application. The validation of dermal structures using
micro-CT compared with conventional staining was performed and
described in our previous study, whereby a PGP9.5 image was used to
determine the presence of nerve fibers (17). In the present study of POTS, an
antibody to PGP9.5 was used to stain neural fibers, but other
dermal structures were also visible. There were differences in the
structure of collagen bundles between the different study groups,
but nothing that may currently be used diagnostically due to the
small group sizes.
The present results were in alignment with the
previous observation in a patient with POTS and concomitant hEDS
(17). Results indicated that 3D
scanning without any staining is suitable to visualize and describe
collagen fiber bundles. The image quality of the laboratory
micro-CT was comparable to previously published data obtained at a
synchrotron radiation facility in parallel beam geometry at a voxel
size of 650 nm (17). This
demonstrated that laboratory setups provided a viable alternative
to synchrotron facilities for studying skin structure. Due to their
accessibility and ease of use, laboratory-based systems may enable
broader availability and facilitate long-term, multi-site studies
in the future. Since the connective tissue is present in the whole
body, 3D scanning of a greater area in unstained skin biopsies may
give a relevant illustration of the tissue and could improve the
diagnostic accuracy in patients with suspicious connective tissue
disorders (17). Skin biopsy is
relatively safe, with few mild complications, such as a small risk
of local infection. The advantage of the present X-ray technique is
the possibility to scan large and unstained preparations without
physical sectioning, preserving sample integrity. However, this may
also be a disadvantage, as it is not possible to differentiate a
number of cell types from each other, such as fibroblasts and
immune cells, due to absence of staining. Depending on the clinical
question, alternative methods are needed. Through further
development of 3D virtual histology, whole paraffin samples may be
scanned, which may lead to even greater areas and more
representative materials being examined, overcoming obstacles of
histopathological dependency on 2D limitations and slice
orientation. The primary contribution of the present study is as a
feasibility analysis of laboratory-based phase-contrast
micro-CT.
The alignment of structures in the data may be
quantified using gray value-based algorithms which are primarily
based upon a structure tensor analysis and describe the local
orientation in the neighborhood (sigma) of each voxel. However,
this algorithm works on the entire dataset and analyzes the
surrounding paraffin, air and sample holder (tube). Furthermore,
the results of structure tensor analysis highly depend on the sigma
of the analysis. Thus, this cannot be used in clinical settings
yet.
It has been established that severe gastrointestinal
symptoms are frequent in POTS and HSD/hEDS (10,12,29).
However, only a number of gastrointestinal or extraintestinal
symptoms differed between participants with or without disorganized
fibers in the present study. Furthermore, a number of healthy
controls also exhibited disorganized collagen fiber bundles.
Although the percentage of disorganized collagen fiber bundles
differed between POTS patients with and without hEDS/EDS,
gastrointestinal symptoms did not differ. Thus, the presence of
disorganized collagen fiber bundles in the skin may support a
diagnosis of hEDS/EDS and gastrointestinal dysmotility in a patient
with clinical features of disease; however this cannot replace
bowel biopsies.
Examination of the nervous system in varying organs
has revealed similar findings represented by the same
pathophysiology in the autonomic nervous system, the enteric
nervous system and the peripheral nervous system in skin (16). Thus, utilization of skin biopsies
may improve the diagnostic accuracy in a simpler way, exhibiting
fewer risks and complications for patients. However, laboratory
X-ray phase-contrast CT is not yet applicable for examining
peripheral intraepidermal nerve fibers.
Micro-CT has recently been described as a
complementary tool for histopathological diagnosis of oral soft
tissue (30,31). To the best of our knowledge, since
the present study was the first to describe skin biopsy analysis
using this technique and a large dataset of 50 participants was
evaluated, it is important to consider how the technique may be
used in the future. Notably, a promising future application of this
technique for skin examination could be to determine infiltration
and the radicality of surgery in basal cell carcinoma and to be
able to separate different types of alopecia. The current 3D
virtual histology using X-rays is an alternative option to evaluate
the radicality of surgery in whole tissues, scanned from the bottom
of the resected tissue. Scalp biopsy is currently the most reliable
diagnostic technique to diagnose and classify different types of
alopecia (32). Micro-CT is an
alternative to multiple sectioning, offering a 3D view of the whole
hair follicle and surrounding tissue.
The strength of the present study lies in the
comparison of skin biopsies from patients with POTS with healthy
controls, since a number of previous studies have not examined
controls (33-35).
However, one limitation was that the patients with hEDS/EDS were
recruited from a POTS cohort, which may have led to different
symptoms and basal characteristics than if patients with only EDS
had been included. No patient had any molecular diagnostic tests of
EDS, however all diagnoses were made after a clinical examination
by a specialist in the field. For ~50% of patients, a diagnosis of
EDS only was made, without any specification. This means that
numerous patients may also have been suffering from hEDS or
potentially the less specified diagnosis HSD, whereby not all
criteria for hEDS were fulfilled (5,6).
Upon study inclusion, the patients were not examined clinically
regarding muscle/joint function and the Beighton score evaluating
each joint with a goniometer was not calculated (5). Therefore, in future studies, patients
with hEDS/EDS should be included based on their primary diagnosis,
determined by clinical and molecular genetic testing for diagnosis.
The markedly high prevalence of disorganized collagen fiber bundles
in the POTS group without known hEDS/EDS may be explained by
undiagnosed cases of hEDS/EDS in the present cohort. In addition,
although there was a relatively large dataset of 50 participants,
its division into three groups led to rather small groups, which
made statistical calculations uncertain. If the groupings had been
larger, a number of the present findings may have been
statistically significant. Thus, future examination of larger
groups is important to elucidate out the role of the skin
architecture in this entity and investigate the role of skin
sampling for diagnostic accuracy. Systematic benchmarking against
conventional histological stains was also not performed and
therefore the present findings require further validation in the
diagnostic characterization of dermal components.
In conclusion, 3D virtual histology may be used for
visualization of the architecture and structure in the skin but not
for examination of intraepidermal nerve fibers. Furthermore, 3D
virtual histology may contribute to the diagnostic workup in
connective tissue disorders; however, further larger studies are
needed to evaluate the use of 3D skin biopsies as a diagnostic tool
in numerous skin disorders.
Acknowledgements
Thanks also go to Karolina Palmér, statistician, for
assistance with statistical advice.
Funding
Funding: The present study was funded by The Ingrid and Sverker
Persson's Foundation, The Development Foundation of Region Skåne
(grant nos. 2022-Projekt0067 and 2021-01942), Skåne University
Hospital (grant nos. 2022-882 and 2022-974) and The Swedish
Research Foundation (grant no. 2021-01942).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
KL, HT, LD, MR and BO designed and performed the
present study. HT recruited the participants. LD performed the skin
biopsy. MR performed the scanning. KL and BO evaluated the
histopathology and analyzed the data. KL and BO confirm the
authenticity of all the raw data. BO wrote the manuscript. All
authors contributed to the intellectual process during the writing,
and all authors have read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
The present study was performed in accordance with
the Declaration of Helsinki and approved by The Swedish Ethical
Review Authority, with review occurring at The Uppsala University
Board (approval nos. 2020-02432 and 2021-00049; Dates of approval,
26/08/2020 and 11/02/2021). All subjects gave their oral and
written, informed consent to participate prior to inclusion and
study start.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Brignole M, Moya A, de Lange FJ, Deharo
JC, Elliott PM, Fanciulli A, Fedorowski A, Furlan R, Kenny RA,
Martín A, et al: 2018 ESC guidelines for the diagnosis and
management of syncope. Eur Heart J. 39:1883–1948. 2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Fedorowski A: Postural orthostatic
tachycardia syndrome: Clinical presentation, aetiology and
management. J Intern Med. 285:352–366. 2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Moak JP, Ramwell CB, Gordish-Dressman H,
Sule SD and Bettini E: Small fiber neuropathy in children,
adolescents, and young adults with chronic orthostatic intolerance
and postural orthostatic tachycardia syndrome: A retrospective
study. Auton Neurosci. 253(103163)2024.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Van Damme T, Colman M, Syx D and Malfait
F: The ehlers-danlos syndromes against the backdrop of inborn
errors of metabolism. Genes (Basel). 13(265)2022.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Malfait F, Francomano C, Byers P, Belmont
J, Berglund B, Black J, Bloom L, Bowen JM, Brady AF, Burrows NP, et
al: The 2017 international classification of the Ehlers-Danlos
syndromes. Am J Med Genet C Semin Med Genet. 175:8–26.
2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Castori M, Tinkle B, Levy H, Grahame R,
Malfait F and Hakim A: A framework for the classification of joint
hypermobility and related conditions. Am J Med Genet C Semin Med
Genet. 175:148–157. 2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Jansen LH: The structure of the connective
tissue, an explanation of the symptoms of the Ehlers-Danlos
syndrome. Dermatologica. 110:108–120. 1955.PubMed/NCBI View Article : Google Scholar
|
|
8
|
de Almeida HL Jr, Bicca E, Rocha NM and de
Castro LAS: Light and electron microscopy of classical
Ehlers-Danlos syndrome. Am J Dermatopathol. 35:102–105.
2013.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Miller AJ, Stiles LE, Sheehan T, Bascom R,
Levy HP, Francomano CA and Arnold AC: Prevalence of hypermobile
Ehlers-Danlos syndrome in postural orthostatic tachycardia
syndrome. Auton Neurosci. 224(102637)2020.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Tai FWD, Palsson OS, Lam CY, Whitehead WE,
Sperber AD, Tornblom H, Simren M and Aziz I: Functional
gastrointestinal disorders are increased in joint
hypermobility-related disorders with concomitant postural
orthostatic tachycardia syndrome. Neurogastroenterol Motil.
32(e13975)2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Lindberg G: Pseudo-obstruction, enteric
dysmotility and irritable bowel syndrome. Best Pract Res Clin
Gastroenterol. 40-41(101635)2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Alomari M, Hitawala A, Chadalavada P,
Covut F, Al Momani L, Khazaaleh S, Gosai F, Al Ashi S, Abushahin A
and Schneider A: Prevalence and predictors of gastrointestinal
dysmotility in patients with hypermobile Ehlers-Danlos syndrome: A
tertiary care center experience. Cureus. 12(e7881)2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Knowles CH, De Giorgio R, Kapur RP, Bruder
E, Farrugia G, Geboes K, Lindberg G, Martin JE, Meier-Ruge WA,
Milla PJ, et al: The London classification of gastrointestinal
neuromuscular pathology: Report on behalf of the gastro 2009
international working group. Gut. 59:882–887. 2010.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Törnblom H, Lindberg G, Nyberg B and
Veress B: Full-thickness biopsy of the jejunum reveals inflammation
and enteric neuropathy in irritable bowel syndrome.
Gastroenterology. 123:1972–1979. 2002.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Roth B, Schiro DB and Ohlsson B: Diseases
which cause generalized peripheral neuropathy: A systematic review.
Scand J Gastroenterol. 56:1000–1010. 2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Ohlsson B, Dahlin LB, Englund E and Veress
B: Autonomic and peripheral neuropathy with reduced intraepidermal
nerve fiber density can be observed in patients with
gastrointestinal dysmotility. Clin Case Rep. 8:142–148.
2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Eckermann M, Peruzzi N, Frohn J, Bech M,
Englund E, Veress B, Salditt T, Dahlin LB and Ohlsson B: 3d
phase-contrast nanotomography of unstained human skin biopsies may
identify morphological differences in the dermis and epidermis
between subjects. Skin Res Technol. 27:316–323. 2021.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Gutiérrez Y, Ott D, Töpperwien M, Salditt
T and Scherber C: X-ray computed tomography and its potential in
ecological research: A review of studies and optimization of
specimen preparation. Ecol Evol. 8:7717–7732. 2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Reichmann J, Verleden SE, Kühnel M, Kamp
JC, Werlein C, Neubert L, Müller JH, Bui TQ, Ackermann M, Jonigk D
and Salditt T: Human lung virtual histology by multi-scale x-ray
phase-contrast computed tomography. Phys Med Biol.
68(115014)2023.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Tufvesson H, Hamrefors V, Roth B,
Fedorowski A and Ohlsson B: Sociodemographic factors, nutritional
status, and inflammatory markers in patients with postural
orthostatic tachycardia syndrome. Acta Neurol Scand.
2023(3992718)2023.
|
|
21
|
Johansson M, Ricci F, Schulte J, Persson
M, Melander O, Sutton R, Hamrefors V and Fedorowski A: Circulating
levels of growth hormone in postural orthostatic tachycardia
syndrome. Sci Rep. 11(8575)2021.PubMed/NCBI View Article : Google Scholar
|
|
22
|
PGP 9.5 Rabbit Polyclonal Antibody,
2026.
|
|
23
|
Tao S, He C, Hao X, Kuang C and Liu X:
Principles of different X-ray phase-contrast imaging: A review.
Appl Sci. 11(2971)2021.
|
|
24
|
Bengtsson M, Ohlsson B and Ulander K:
Development and psychometric testing of the visual analogue scale
for irritable bowel syndrome (VAS-IBS). BMC Gastroenterol.
7(16)2007.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Bengtsson M, Persson J, Sjölund K and
Ohlsson B: Further validation of the visual analogue scale for
irritable bowel syndrome after use in clinical practice.
Gastroenterol Nurs. 36:188–198. 2013.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Francis CY, Morris J and Whorwell PJ: The
irritable bowel severity scoring system: A simple method of
monitoring irritable bowel syndrome and its progress. Aliment
Pharmacol Ther. 11:395–402. 1997.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Beighton P, De Paepe A, Steinmann B,
Tsipouras P and Wenstrup RJ: Ehlers-Danlos syndromes: Revised
nosology, villefranche, 1997. Ehlers-Danlos national foundation
(USA) and Ehlers-Danlos support group (UK). Am J Med Genet.
77:31–37. 1998.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Hermanns-Lê T and Piérard GE: Collagen
fibril arabesques in connective tissue disorders. Am J Clin
Dermatol. 7:323–326. 2006.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Tufvesson H, Hamrefors V, Fedorowski A,
Hansson M and Ohlsson B: Gastrointestinal symptoms in patients with
postural orthostatic tachycardia syndrome in relation to
hemodynamic findings and immunological factors. Front Physiol.
15(1342351)2024.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Yu J, Gu Z, Wang L, Zhang Q, Pu Y, Hu Q,
Xia C and Wang Y: Three-dimensional measurement of the depth of
invasion in oral squamous cell carcinoma samples using Lugol's
iodine-enhanced micro-computed tomography: An original study. J
Appl Oral Sci. 33(e20240304)2025.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Kats L, Gabet Y, Shpunt U and Vered M:
Micro-computed tomography as a complementary tool for
histopathological diagnosis of oral soft tissue lesions-proof of
concept. PLoS One. 20(e0335602)2025.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Kallipolitis A, Moutselos K, Zafeiriou A,
Andreadis S, Matonaki A, Stavropoulos TG and Maglogiannis I: Skin
image analysis for detection and quantitative assessment of
dermatitis, vitiligo and alopecia areata lesions: A systematic
literature review. BMC Med Inform Decis Mak. 25(10)2025.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Haensch CA, Tosch M, Katona I, Weis J and
Isenmann S: Small-fiber neuropathy with cardiac denervation in
postural tachycardia syndrome. Muscle Nerve. 50:956–961.
2014.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Billig SCI, Schauermann JC, Rolke R,
Katona I, Schulz BJ and Maier A: Quantitative sensory testing
predicts histological small fiber neuropathy in postural
tachycardia syndrome. Neurol Clin Pract. 10:428–434.
2020.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Zhang R, Mayuga K, Shields R, Cantrell C
and Wilson R: Skin biopsy and quantitative sudomotor axon reflex
testing in patients with postural orthostatic tachycardia syndrome.
Cureus. 14(e31021)2022.PubMed/NCBI View Article : Google Scholar
|