Introduction
Mesenteric venous thrombosis (MVT) is a rare but
life-threatening gastrointestinal (GI) vascular disorder, with an
incidence rate of 1 in 5,000-15,000 hospital admissions and 1 in
1,000 Emergency Department visits worldwide (1). The mortality rate of acute MVT ranges
from 20 to 50%, with delayed diagnosis and treatment markedly
increasing the risk of bowel infarction, sepsis and multiorgan
failure (2). As a notable cause of
acute mesenteric ischemia, MVT differs from arterial mesenteric
ischemia in its more insidious onset and non-specific clinical
presentation, which often contribute to diagnostic delays (3). In addition, hematemesis is a rare
manifestation of MVT, reported in <15% of cases in the existing
literature, which further complicates the diagnostic process and
may lead to misdiagnosis as common upper GI bleeding etiologies,
such as peptic ulcers or esophageal varices (2).
Pathophysiologically, thrombus formation in the
mesenteric venous system impairs intestinal venous return, causing
venous engorgement and reduced arterial perfusion, and potentially
transmural bowel infarction, bacterial translocation and
life-threatening complications if untreated. MVT is categorized
into primary (associated with obesity, diabetes and tobacco use,
among others), secondary (associated with protein C, protein S or
antithrombin deficiency, malignancy and oral contraceptives, among
others) and idiopathic etiologies. Contrast-enhanced abdominal CT
remains a standard procedure in diagnosis, while anticoagulation is
central to all treatment options, with surgery being reserved for
bowel necrosis, perforation or peritonitis (1-3).
The present report aims to enhance clinicians' awareness by
reporting a rare case of MVT complicated with hematemesis and small
bowel necrosis, which was successfully managed through
laparotomy.
Case report
A 29-year-old man presented to Emergency Department
of Jilin Central Hospital (Jilin, China) in August 2018, with a
7-day history of intermittent upper abdominal pain. The pain was
described as distended and non-radiating. The medical history of
the patient included a left lower limb deep venous thrombosis and a
stent implantation conducted in March 2008, at which time the
patient was 175 cm in height, weighed 95 kg and exhibited a BMI of
>30 kg/m2. The patient had no history of liver
cirrhosis, portal hypertension, peptic ulcer disease or
hematochezia. An initial abdominal CT scan revealed gas-fluid
levels, leading to a diagnosis of intestinal obstruction.
Conservative treatment was administered, which included symptomatic
measures such as nil per os, gastrointestinal decompression,
acid suppression (40 mg omeprazole per dose, intravenously, every
12 h for 5 days), anti-inflammatory therapy (1.5 g cefuroxime
sodium per dose, intravenously, every 12 h for 5 days) and an
enema, which temporarily alleviated the abdominal pain. However, 6
days later, the pain worsened and became persistent, prompting
referral of the patient to Department of Gastrointestinal and
Colorectal Surgery of China-Japan Union Hospital of Jilin
University (Jilin, China).
Upon admission, the vital signs of the patient were
stable (blood pressure, 140/80 mmHg; heart rate, 80 bpm). Physical
examination revealed a flat, soft abdomen with mild tenderness in
the upper quadrants, with no rebound tenderness or muscle rigidity.
Shortly after transfer (~20 min after admission), the patient
experienced nausea followed by hematemesis, vomiting ~1,200 ml
blood. The blood pressure of the individual subsequently dropped to
80/40 mmHg, necessitating an immediate blood transfusion (4 units
of packed red blood cells) and intravenous fluid resuscitation.
An emergency upper GI endoscopy was performed within
1 h of hematemesis, which revealed no source of bleeding in the
esophagus, stomach or duodenum. Furthermore, an abdominal
ultrasonography revealed moderate ascites, which was drained under
ultrasound guidance, yielding ~500 ml of bloody fluid. An abdominal
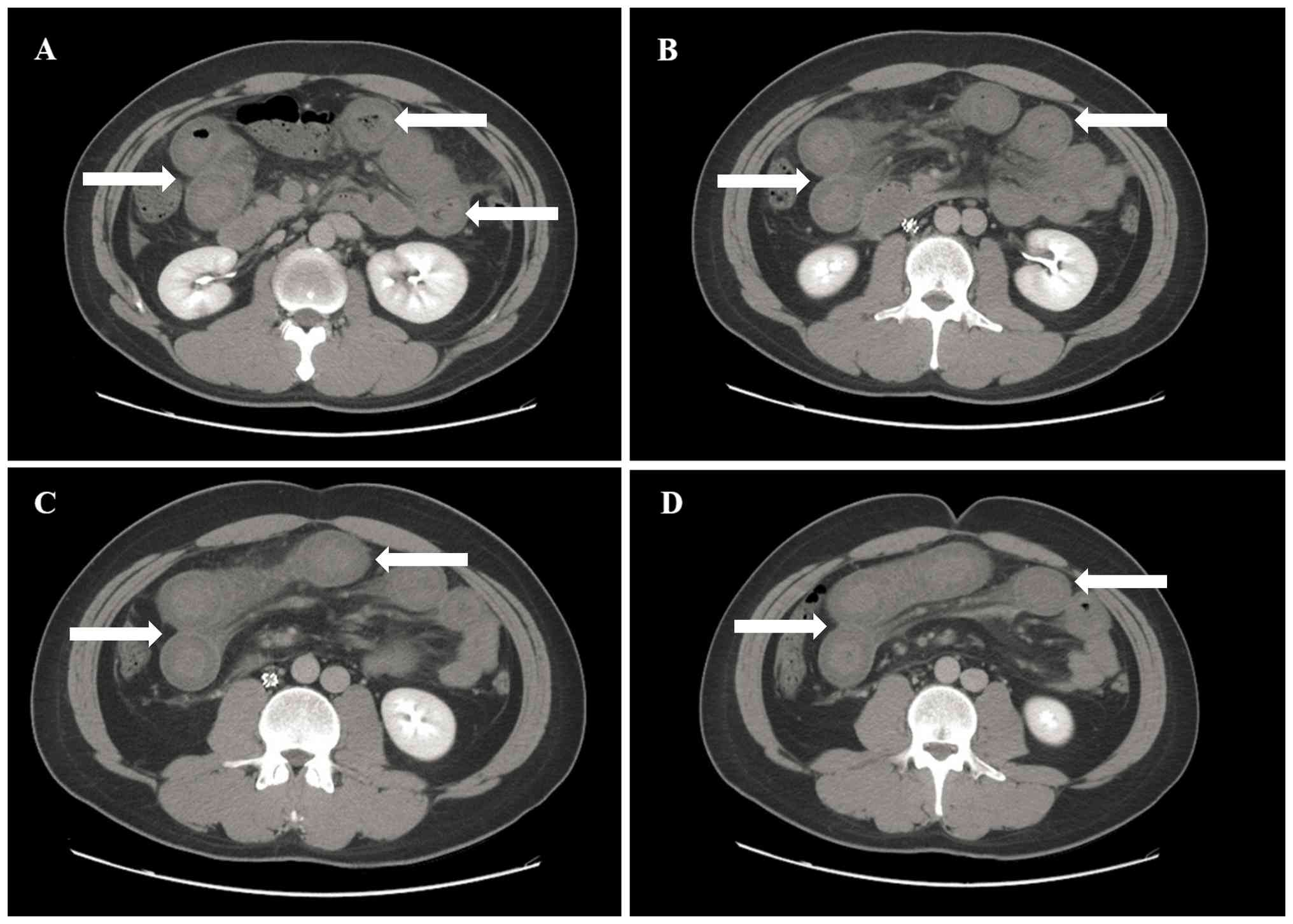
CT scan exhibited extensive dilation of the small intestine in the
upper abdomen, with thickened intestinal walls also observed
(maximum thickness, 0.9 cm; Fig.
1).
Laboratory findings upon admission included a blood
leukocyte count of 23.87x109/l (normal range,
4-10x109/l), a neutrophil percentage of 92.8% (normal
range, 50-70%), a platelet count of 171x109/l (normal
range, 100-300x109/l), a red blood cell count of
5.05x1012/l (normal range, 4.00-5.00x1012/l),
a hemoglobin level of 146 g/l (normal range, 120-160 g/l), a
hematocrit level of 0.436 (normal range, 0.400-0.500) and a mean
corpuscular volume of 86.3 fl (normal range, 80.0-100.0 fl). A
plasma D-dimer level of 39.6 µg/ml (normal range, 0-0.5 µg/ml) and
a serum amylase level of 42 U/l (normal range, 25-125 U/l) were
noted. Prothrombin time was 15.5 sec (normal range, 11.0-15.0 sec)
and activated partial thromboplastin time was 35.3 sec (normal
range, 28.0-43.5 sec). The fibrinogen level was 4.57 g/l (normal
range, 2.00-4.00 g/l), the serum level of alanine aminotransferase
was 42.63 g/l (normal range, 5.00-40.00 g/l), the serum level of
aspartate aminotransferase was 26.08 g/l (normal range, 8.00-40.00
g/l) and the serum level of LDH was 507.50 g/l (normal range,
80.00-248.00 g/l). Based on these findings, MVT with intestinal
ischemia was highly suspected; accordingly, emergency transfusion
of 4 units of packed red blood cells and 400 ml of plasma was
administered, and an emergency laparotomy was performed 3 h after
admission.
During surgery, a 110-cm segment of necrotic small
intestine (180 cm distal to the ligament of Treitz), with
thromboembolism in the corresponding mesenteric veins, was
identified. The necrotic segment was resected and an end-to-end
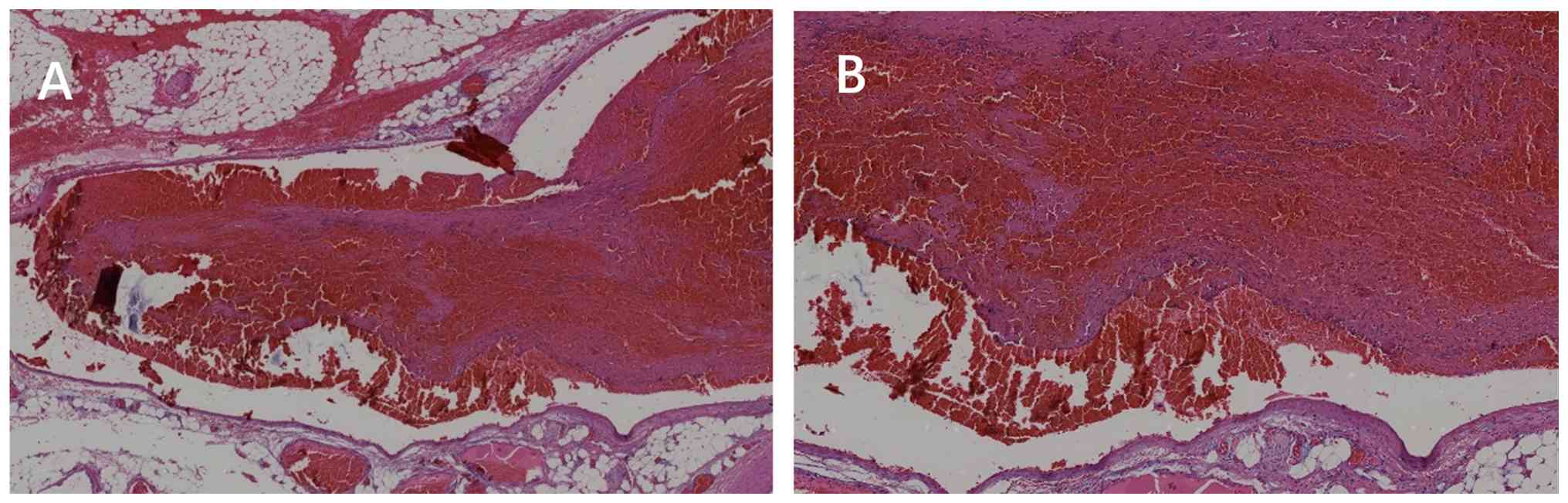
anastomosis was performed. Pathological examination demonstrated
intestinal hemorrhagic infarction (Fig. 2).
To determine the diagnosis of intestinal hemorrhagic
infarction in the present patient, the necrotic small intestinal
tissue resected during laparotomy was subjected to H&E
staining. Under sterile conditions, lesional intestinal tissue and
adjacent normal intestinal tissue (serving as internal controls)
were harvested immediately after sample acquisition. Sterile
surgical blades were used to trim tissues into uniform cubic blocks
(0.5x0.5x0.2 cm) to ensure complete and uniform fixative
penetration. Trimmed tissues were immersed in 10% neutral buffered
formalin (pH 7.2-7.4) at a fixative-to-tissue ratio ≥10:1 to
prevent autolysis and incubated at 4˚C for 12-24 h. After fixation,
tissues were rinsed with PBS (pH 7.4) for 5 min, then dehydrated
with ethanol [70% (1 h), 80% (1 h), 95% (1 h) and 100% (twice, 1 h
each)]. Dehydrated tissues were then subjected to xylene (twice, 30
min each) until completely transparent. Cleared tissues were
immersed in molten paraffin (56-58˚C) twice for 1 h each, then
embedded in fresh paraffin. Tissues were oriented to cover lesional
and marginal normal areas, and paraffin blocks were solidified at
room temperature and stored at 4˚C until sectioning.
All H&E staining steps were performed at 22-25˚C
using analytical grade reagents and double-distilled water.
Paraffin blocks were sectioned into 4- to 5-µm continuous serial
sections using a rotary microtome. Sections were floated on 40-42˚C
double-distilled water to flatten, mounted on poly-L-lysine-coated
slides, baked at 60˚C for 1-2 h and cooled to room temperature.
Slides were deparaffinized in xylene (twice, 10 min each),
rehydrated using an alcohol gradient (100, 95, 80 and 70% ethanol;
5 min each) and rinsed with double-distilled water for 5 min.
Rehydrated slides were stained with Harris hematoxylin for 5-10
min. Excess hematoxylin was rinsed off and slides were
differentiated in 1% hydrochloric acid-ethanol for 30-60 sec, then
blued with running tap water (10-15 min) or 1% ammonia water (1-2
min). Blued slides were stained with 0.5% eosin Y for 2-5 min.
Slides were dehydrated (70, 80, 95 and 100% ethanol), cleared in
xylene (twice, 5 min each), mounted with neutral balsam and
air-dried for 24 h before observation.
Dried H&E-stained slides were examined with a
compound light microscope equipped with 20X and 40X objective
lenses (corresponding to x200 and x400 final magnifications).
Digital images were captured at x200 and x400 magnifications using
a charge-coupled device imaging system, with standardized
parameters (exposure time, white balance, light intensity and
resolution) for consistency. Numerous non-overlapping fields were
captured per sample. Images were saved as uncompressed TIFF files
(≥300 dpi) to preserve histological details for further analysis.
The images obtained by H&E staining were captured as shown in
Fig. 2, illustrating the typical
histological features of intestinal hemorrhagic infarction.
The patient was discharged in late August 2018 (on
the 10th postoperative day) without complications. Throughout the
entire treatment process, cefuroxime sodium (1.5 g per dose,
intravenously, every 12 h for 7 days), omeprazole (40 mg per dose,
intravenously, every 12 h for 7 days) and low-molecular-weight
heparin (4,000 IU per dose, subcutaneously, once a day for 10 days)
were administered, along with parenteral nutrient solution (25-30
kcal/kg body weight per day, continuous administration via central
venous catheter for 5 days). All drug dosages were based on adult
clinical guidelines (4), with
adjustments made according to the renal and hepatic function of the
patient, as well as inflammatory indicators and improvements in
clinical symptoms.
Since discharge, the patient has undergone intensive
follow-up (each month for 3 months, then quarterly) from 2018-2019,
6-monthly follow-ups from 2019-2020 and annual follow-ups from
2020-2026. Follow-up data reported a height of 180 cm, a stable
weight of 71-75 kg and a normal BMI (21.9-23.1 kg/m2),
with no abdominal pain/distension recurrence, regular 10 mg daily
oral rivaroxaban (no missed doses or adverse reactions), normal
routine tests (complete blood count, liver function and coagulation
function) and a healthy lifestyle. The patient has had no
recurrence of symptoms, such as abdominal pain or abdominal
distension since discharge, so abdominal CT and gastrointestinal
endoscopy have not been performed.
No dosage adjustments or emergencies have occurred
and the patient remains stable without recurrence or complications,
with effective follow-up and good medication compliance.
Discussion
MVT is a rare but potentially lethal condition
characterized by non-specific clinical and laboratory findings,
resulting in diagnostic challenges and delayed intervention
(5). Acute MVT typically presents
with sudden, cramping abdominal pain disproportionate to physical
examination findings. Bowel infarction, a severe complication, may
manifest with peritoneal signs such as rebound tenderness (6). A delayed diagnosis is associated with
mortality rates of 19-23% (5).
Thrombosis typically originates from the superior mesenteric vein
in 95% of cases, with the inferior mesenteric vein involved in only
4-6% of cases (1).
MVT is primarily divided into three etiologies:
Primary, secondary and idiopathic (7). The leading causes of secondary MVT
include protein C or S deficiency, antithrombin deficiency,
myelofibrosis, malignancy, oral contraceptives, pregnancy,
inflammatory bowel disease, peritonitis, abdominal trauma and
cirrhosis (8). The risk factors
for primary MVT include obesity (BMI >30 kg/m2),
diabetes, tobacco use and thrombophilia (9,10).
The present patient exhibited a heightened risk for primary
thrombotic disorder due to a BMI >30 kg/m2 and a
medical history of left lower limb deep venous thrombosis.
A diagnosis of MVT is often delayed due to its
non-specific symptoms, which may mimic other acute abdominal
conditions such as pancreatitis, intestinal perforation,
cholecystitis or appendicitis. Patients with colonic ischemia often
present with lower GI bleeding, whereas intestinal lesions proximal
to the ligament of Treitz typically result in upper GI bleeding. A
history of deep venous thrombosis or the presence of ascites in a
patient with acute abdominal pain should therefore raise suspicion
of MVT (11,12). Delayed treatment may lead to
peritonitis or sepsis, resulting in hemodynamic instability and
multiorgan system failure (13).
In order to understand the consequences of undiagnosed and
untreated MVT, the pathophysiology of this condition should be
acknowledged. MVT impairs venous return from the bowel, resulting
in venous engorgement and ischemia. Due to the rapid and complete
occlusion of mesenteric veins, transmural bowel infarction may
occur. In addition, venous engorgement may cause arterial spasm,
with resulting irreversible bowel ischemia. With a transmural
infarction, there is loss of integrity of the bowel mucosa,
allowing bacterial translocation and potential for occurrence of
the fatal consequence of lactic acidosis, sepsis, multiorgan
failure and mortality (8). The
present patient was initially incorrectly diagnosed with intestinal
obstruction. Due to the lack of timely and effective treatment, the
MVT of the patient progressed, resulting in jejunal necrosis 180 cm
distal to the ligament of Treitz and subsequent upper GI
bleeding.
Abdominal contrast-enhanced CT scans remain the
standard for diagnosis for MVT, with an accuracy of 95-100%
(14,15). Key findings from CT scans include
filling defects in mesenteric veins, thickened bowel walls, dilated
bowel loops, indistinct bowel margins, thickened mesentery and
ascites (16). However, for the
present patient, neither obvious MVT nor venous occlusion was
recognized on admission. During surgery, a number of small venous
occlusions were identified within the mesentery associated with the
necrotic segment of the intestine. It was speculated that the small
diameter of the embolized vein may have obscured the visualization
of the thrombus. Other imaging techniques, such as Doppler
ultrasound, may detect large thrombi but are unable to visualize
small thrombi. Furthermore, nuclear angiography is rarely used in
the contemporary evaluation of MVT due to its poor sensitivity and
limited availability.
The primary goals of treatment for acute MVT are to
prevent intestinal infarction and reduce the risk of thrombosis
recurrence. Anticoagulation is a key therapy, alongside heparin
initiation upon diagnosis (8).
When the international normalized ratio reaches the target range of
2-3, heparin is discontinued and warfarin alone is continued
(7). Long-term anticoagulation
with warfarin is recommended for at least 3-6 months, with extended
therapy further advised if residual thrombus or hypercoagulable
disorders are present (17,18).
For patients refractory to anticoagulation, catheter-directed
therapy may be considered, although it is associated with a higher
risk of complications, including bleeding (8). Surgical intervention remains reserved
for patients with bowel infarction, perforation or peritonitis.
Resection and anastomosis are the standard procedures, with the aim
of preserving as much bowel as possible. A second-look operation
may be necessary within 12 to 48 h to address additional necrotic
segments (1,8).
In conclusion, the present case underscores the
critical challenge of diagnosing MVT when it presents with the rare
symptom of hematemesis. Diagnostic delay led to bowel necrosis,
highlighting the need for a high index of suspicion in individuals
with thrombophilic risk factors, even when initial tests are
inconclusive. Optimal outcomes depend on prompt anticoagulation,
emergent surgery for necrosis and structured long-term follow-up.
This report contributes to the literature by emphasizing that an
integrated clinical approach is essential to mitigate the high
morbidity and mortality rates associated with atypical MVT
presentations.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by The Science and
Technology Development Program of Jilin Province (grant no.
YDZJ202501ZYTS054).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
CS, ZL, LP and BS designed the present study. CS,
ZL, LP and BS collected and analyzed the clinical data. CS and BS
reviewed previous cases. CS and BS wrote and revised the
manuscript. CS and BS confirm the authenticity of all the raw data.
All authors have read and approved the final version of the
manuscript.
Ethics approval and consent to
participate
All procedures were performed in accordance with the
1964 Helsinki Declaration and its later amendments or comparable
ethical standards.
Patient consent for publication
Written informed consent was obtained from the
patient for the publication of the present manuscript and any
accompanying images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Harnik IG and Brandt LJ: Mesenteric venous
thrombosis. Vasc Med. 15:407–418. 2010.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Kumar S, Sarr MG and Kamath PS: Mesenteric
venous thrombosis. N Engl J Med. 345:1683–1688. 2001.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Russell CE, Wadhera RK and Piazza G:
Mesenteric venous thrombosis. Circulation. 131:1599–1603.
2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Simonetto DA, Singal AK, Garcia-Tsao G,
Caldwell SH, Ahn J and Kamath PS: ACG clinical guideline: Disorders
of the hepatic and mesenteric circulation. Am J Gastroenterol.
115:18–40. 2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Acosta S, Ögren M, Sternby N-H, Bergqvist
D and Björck M: Mesenteric venous thrombosis with transmural
intestinal infarction: A population-based study. J Vasc Surg.
41:59–63. 2005.PubMed/NCBI View Article : Google Scholar
|
|
6
|
García-Botella A, Asenjo S, De la
Morena-Barrio ME, Corral J, Bolaños E, Carlin PS, López ES and
García AJ: First case with antithrombin deficiency, mesenteric vein
thrombosis and pregnancy: Multidisciplinary diagnosis and
successful management. Thromb Res. 144:72–75. 2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Lim KH, Jang J, Yoon HY and Park J: Acute
superior mesenteric vein thrombosis associated with abdominal
trauma: A rare case report and literature review. Medicine
(Baltimore). 96(e8863)2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Singal AK, Kamath PS and Tefferi A:
Mesenteric venous thrombosis. Mayo Clin Proc. 88:285–294.
2013.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Walter G, Richert Q, Ponnampalam A and
Sharma A: Acute superior mesenteric vein thrombosis in the setting
of cytomegalovirus mononucleosis: A case report and review of the
literature. Lancet Infect Dis. 21:e202–e207. 2021.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Gupta A, Sharma O, Srikanth K, Mishra R,
Tandon A and Rajput D: Review of mesenteric ischemia in COVID-19
patients. Indian J Surg. 85 (Suppl 1):S313–S321. 2023.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Blumberg SN and Maldonado TS: Mesenteric
venous thrombosis. J Vasc Surg Venous Lymphat Disord. 4:501–507.
2016.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Brandt LJ and Boley SJ: AGA technical
review on intestinal ischemia. American gastrointestinal
association. Gastroenterology. 118:954–968. 2000.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Klar E, Rahmanian PB, Bücker A, Hauenstein
K, Jauch KW and Luther B: Acute mesenteric ischemia: A vascular
emergency. Dtsch Arztebl Int. 109:249–256. 2012.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Hagspiel KD, Flors L, Hanley M and Norton
PT: Computed tomography angiography and magnetic resonance
angiography imaging of the mesenteric vasculature. Tech Vasc Interv
Radiol. 18:2–13. 2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Acosta S, Alhadad A and Ekberg O: Findings
in multi-detector row CT with portal phase enhancement in patients
with mesenteric venous thrombosis. Emerg Radiol. 16:477–482.
2009.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Guan X, Huang L and Li L: Acute mesenteric
venous thrombosis in a pregnant woman at 35 weeks of gestation: A
case report and review of the literature. BMC Pregnancy Childbirth.
18(487)2018.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Malec L and Young G: Treatment of venous
thromboembolism in pediatric patients. Front Pediatr.
5(26)2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Kim HS, Patra A, Khan J, Arepally A and
Streiff MB: Transhepatic catheter-directed thrombectomy and
thrombolysis of acute superior mesenteric venous thrombosis. J Vasc
Interv Radiol. 16:1685–1691. 2005.PubMed/NCBI View Article : Google Scholar
|