Introduction
Myopia is a type of refractive error marked by the
elongation of the eyeball; this anatomical change causes light rays
to focus in front of the retina rather than directly on it,
resulting in blurred vision when viewing distant objects.
Historically regarded as a harmless condition that can be easily
corrected, myopia has become a notable global public health issue.
Its growing severity stems from two key factors: A rise in its
prevalence worldwide, which is largely attributed to increased
educational pressures, prolonged near work and limited time spent
outdoors, and the link between high myopia (as well as pathological
myopia) and severe, vision-endangering complications such as
scleral thinning, chorioretinal atrophy and tractional maculopathy
(1). Both the World Health
Organization and the International Myopia Institute have identified
myopia as an expanding epidemic. These organizations forecast that
by 2050, nearly half of the global population will be myopic, and
~10% will suffer from high myopia (2,3).
Furthermore, parallel to the high incidence of myopia, astigmatism
also exhibits a notable prevalence profile, with an estimated
pooled prevalence of ~40% in adults across World Health
Organization regions and even higher in Asia (1,4,5).
Small-incision lenticule extraction (SMILE) is a
relatively new flapless laser refractive procedure for correcting
myopia and myopic astigmatism, which has been clinically practiced
worldwide for more than a decade (6,7). The
procedure involves two main steps: First, a femtosecond laser is
used to create an intrastromal lenticule (a small, lens-shaped
piece of corneal tissue), and second, this lenticule is removed
through a tiny corneal incision measuring 2-3 mm (8). SMILE has similar visual and
refractive outcomes compared with femtosecond laser-assisted in
situ keratomileusis (FS-LASIK) (9). This flapless approach with smaller
corneal wound could reduce the risk for dry eye and improve the
maintenance of the corneal biomechanical integrity (10); however, suction loss is a
complication experienced during SMILE which could affect the
surgery, with an incidence rate ranging from 0.17-5.06% (11).
Management strategies for suction loss include
immediate re-SMILE, delayed re-SMILE and conversion to other
surgical methods such as LASIK, Laser-Assisted Subepithelial
Keratectomy (LASEK) or Trans-Epithelial Photorefractive Keratectomy
(TPRK), among others, and the management approaches vary depending
on different suction loss stages. Most of the methods yield good
results and patients have a high level of satisfaction. The risk
factors for suction loss comprise a larger cap diameter, higher
astigmatism, anxiety-related eye movement and lack of surgical
experience (12). Numerous
investigations have reported the occurrence of suction loss during
SMILE and the corresponding treatment measures (13,14),
but to the best of our knowledge, reports on two consecutive
suction losses during the operation remain limited. The present
report is a case in which two consecutive suction losses occurred
in one eye during SMILE surgery, resulting in an incomplete
operation. Notably, the patient subsequently underwent elective
FS-LASIK surgery where the treating physicians were unaware of the
exact details of first procedure, and favorable visual acuity
outcomes were finally achieved.
Case report
The present report describes the case of a
24-year-old man who underwent SMILE at General Hospital of Ningxia
Medical University (Yinchuan, China) in January 2025. The surgery
was successfully completed in the right eye, however, the left eye
experienced two consecutive suction losses, resulting in incomplete
treatment. At ~1.5 months post operation, in March 2025, the
patient presented at The First Affiliated Hospital of Xi'an
Jiaotong University (Xi'an, China) for a pre-employment medical
examination. At that time, except for the preoperative examination
records prior to the first SMILE surgery that the patient had
brought with him, no records regarding the specific design
parameters and detailed procedures of the previous SMILE surgery
were received. However, the patient was eager to complete the
pre-employment medical examination to avoid affecting his job
application, so we directly performed preoperative examinations on
him.
Ophthalmic examination findings included: the
uncorrected distance visual acuity (UDVA), right eye 20/20 and left
eye 20/400; corrected distance visual acuity, left eye 20/16; and
intraocular pressure, right eye 13.3 and left eye 21.3 mmHg. The
bilateral eyelid opening and closing were normal, without
congestion or edema. In addition, the upper and lower lacrimal
puncta were properly positioned, with no secretion reflux observed
upon lacrimal sac compression, and lacrimal duct irrigation was
unobstructed. The tear break-up time was 6 sec, there was no
congestion or edema and both corneas were transparent, with no
keratic precipitates. The surgical incisions were visible, without
epithelial ingrowth or corneal haze. The sclera had no icterus and
the anterior chamber was moderately deep with clear aqueous humor;
the iris texture was clear, without iridodonesis or
anterior/posterior synechiae; and the bilateral pupils were round,
equal in size (3 mm in diameter), with brisk direct light reflexes.
The lens was properly positioned and transparent and the vitreous
showed mild liquefaction. Furthermore, at the fundus the bilateral
optic discs had clear margins and a pale red color (cup-to-disc
ratio=0.2); macular light reflex was present, leopard spot fundus
was observed and the retina was flat without obvious abnormalities.
The patient had no reported history of ocular trauma or systemic
diseases such as diabetes, heart disease or hypertension. The
patient disclosed that he underwent bilateral SMILE surgery at a
local hospital 1.5 months previously; the right eye surgery was
uneventful with satisfactory postoperative recovery, whereas the
left eye surgery was terminated prematurely due to intraoperative
suction loss. No other ocular complications were identified
preoperatively.
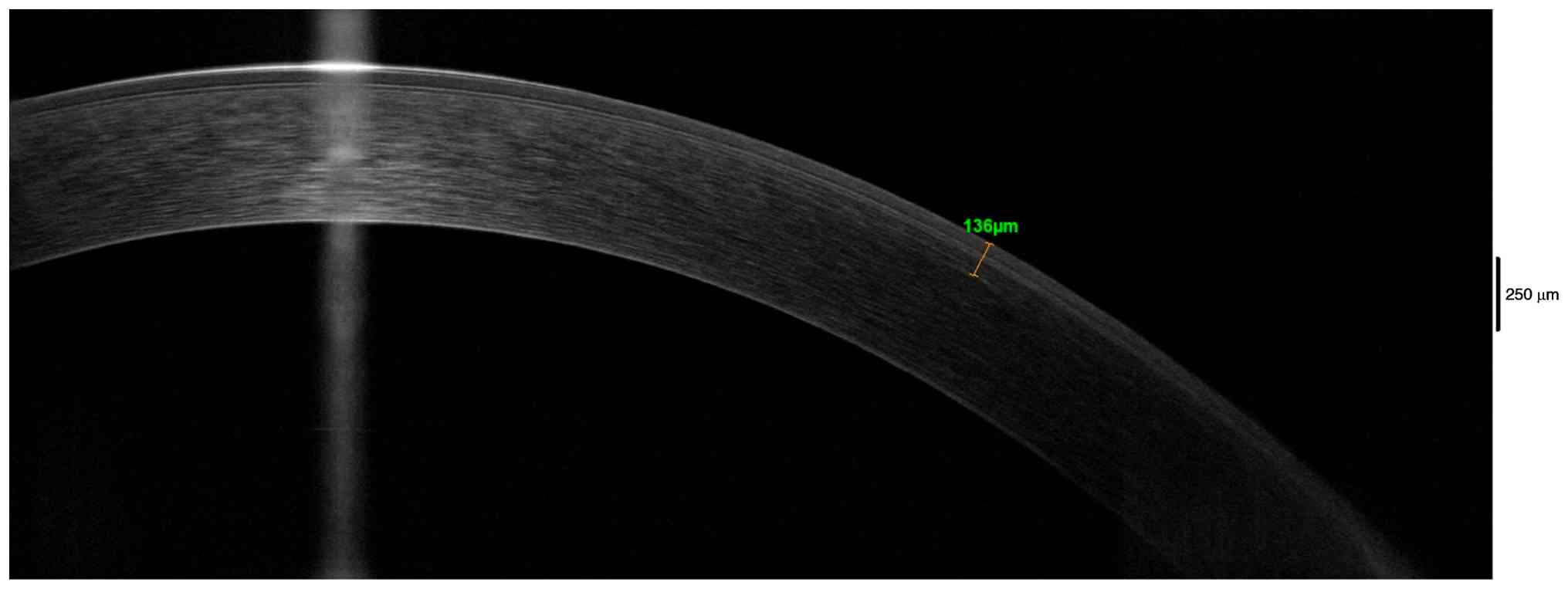
Corneal topography system Sirius (software version
phoenix.1; Costruzione Strumenti Oftalmici Srl) showed the left eye
had a thinnest corneal thickness of 511 µm, flat keratometry of
43.4 diopter (D) and steep keratometry of 44.1 D. Anterior segment
optical coherence tomography (AS-OCT; Optovue, Inc.) revealed a
corneal cap thickness of 136 µm in the left eye from the previous
SMILE surgery (Fig. 1).
Based on these preoperative examination results,
FS-LASIK was designed for the left eye of the patient, with a flap
thickness of 105 µm, a flap diameter of 8.6 mm and a customized
Q-value of -0.24. The patient was admitted to the present hospital
in March 2025, and the FS-LASIK-Q operation proceeded after 1 day
with no complications; the UDVA was 20/20. The last follow-up was
performed at the 1-month postoperative visit in April 2025; at that
time, the UDVA was 20/13, the flat keratometry was 39.8 D and steep
keratometry was 40.0 D, the cornea was transparent and the incision
was well-healed. No complications were observed, and the patient
remained in a stable condition with satisfactory surgical
outcomes.
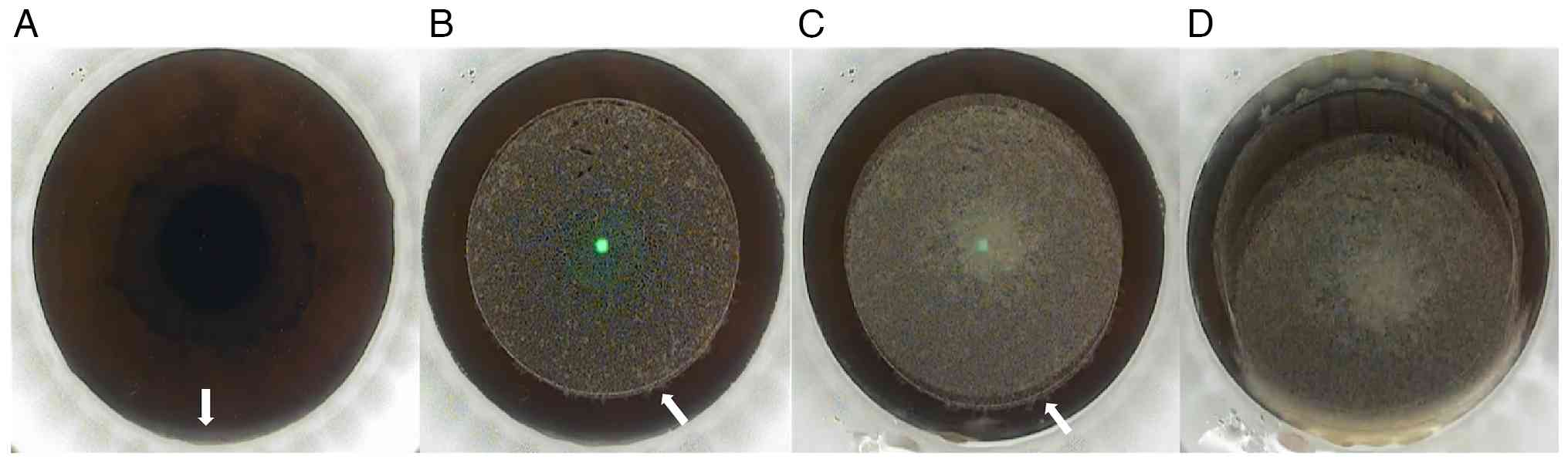
At ~2 weeks after the FS-LASIK-Q operation, the
videos of the first SMILE surgery of the patient were received from
the local hospital and were analyzed to gain experience. During the
first suction loss, a small part of the bulbar conjunctiva in the
upper area could be observed entering the negative pressure area
when establishing negative pressure before laser scanning (Fig. 2A). Subsequently, during the cutting
of the lens edge, there was a slight upward displacement (<0.3
mm) of the eye position (Fig. 2B).
At the instant before suction loss during cap cutting, the center
of the cap was separated from the center of the lens and the eye
position shifted upward ~1 mm (Fig.
2C). These sequential positional deviations disrupted the
alignment between the laser scanning trajectory and the predesigned
surgical zone, which directly contributed to the progressive first
suction loss that occurred when 90% of the cap scanning was
completed, and the range was approaching to cover the lens. The
bubbles dispersed to the limbus within the upper one-third range of
the cornea at that time, which turned the cornea white (Fig. 2D). Consequently, the surgeons
decided to continue with SMILE without making any modifications to
the original treatment plan and re-establish negative pressure for
cap scanning.
When establishing negative pressure for the second
time and before conducting laser scanning, the cutting line of the
lens edge could be observed, the upper peripheral corneal area was
in a state of bubble dispersion and the bulbar conjunctiva in the
upper part entered the negative pressure surface (Fig. 3A). Then the surgeon restarted cap
scanning, the center of the cap basically coincided with that of
the lens, everything went smoothly until the cap scanning was
completed; however, when making the cap incision, the eye position
started to move upward again (Fig.
3B). Therefore, the scanning of the cap incision was carried
out during the displacement, and in the end the cap incision was
not completed, it was in a misaligned position (Fig. 3C). Later the surgeons attempted to
separate the incision manually, but it was unsuccessful. Due to the
dispersed bubbles above the cornea affecting the surgeon's
observation of the scanning status and completion degree of the cap
incision, the surgeon did not try to make a mechanical
incision.
Discussion
In the present case, the reasons why the patient
suffered two consecutive suction losses and the SMILE surgery was
incomplete mainly include three aspects: First, anxiety-related eye
movement, where the patient showed uncontrolled upward eyeball
rotation; second, the patient had a small corneal longitudinal
diameter, with conjunctivalization of the superior corneal limbus,
which caused the negative suction unstable; and third, due to the
lack of surgical experience, the surgeon did not choose to make a
manual cap incision after the failure of creating a cap incision
during the second suction loss.
When the process and outcome of the first SMILE
surgery remain unknown, some preoperative preparations are required
for designing the second surgery; for example, the visual acuity,
intraocular pressure, ocular axis, computerized optometry, corneal
thickness and corneal topography parameters. This can be measured
using AS-OCT to assess the thickness of epithelial, cap and
lenticule and using a slit lamp microscope to exam the status of
the cornea and other ocular surface structures are necessary.
There are three appropriate elective surgical
approaches after the suction losses in this case to consider.
First, for FS-LASIK, it is necessary to precisely design the
corneal flap thickness and diameter, ensuring the flap thickness is
thinner than the corneal cap depth from the initial SMILE surgery
and does not overlap with the first laser scan to avoid
misalignment. The second approach is LASEK or TPRK; the advantage
of this choice is that there is no risk of misalignment. However,
the complications include slow recovery, infections, pain and
corneal haze (15,16). The third approach is making a
manual cap incision. If the status of cap thickness, diameter and
incision completion degree are clear, this choice is recommended.
This recommendation is based on the fact that manual incision is
consistent with the original surgical procedure of SMILE,
eliminating the need for switching to an alternative surgical plan.
More importantly, it aligns with the core feature of SMILE
surgery-no corneal flap creation- and thus minimizes corneal
damage; however, there is still a risk of failing to remove the
lenticule, which comprises the following circumstances: i) Failure
to identify the upper and lower layers of the lenticule; ii)
incomplete lenticule dissection due to progressive displacement;
and iii) inconsistency between the location and depth of the
incision. Furthermore, the surgical timing needs to be considered;
if elective FS-LASIK, LASEK or TPRK is selected, preoperative
re-evaluation is required. The elective surgery can be performed
only when parameters such as corneal morphology and refractive
power are consistent with those examination results before the
first SMILE surgery. If a cap incision is manually created to
remove the lenticule, surgical intervention should be performed as
soon as possible.
Ultimately, the present team decided to perform
FS-LASIK taking into consideration the overall circumstances, and
the details were as follows: On the one hand, the specific eye
condition after SMILE was unclear before the operation videos were
received, particularly the status of the laser scanning during the
aspiration process. On the other hand, the preoperative examination
results cannot show exact lens and cap scanning lines and their
thickness. To ensure safety as well as considering the patient's
need for rapid visual recovery, FS-LASIK was selected as the
surgical approach in the end. Furthermore, there are some key
points for intraoperative procedures: First, the central alignment
should be precise; second, the patient should be reassured to
alleviate anxiety and pre-operative fixation training should be
conducted to improve cooperation; third, it is necessary to design
a thinner flap to prevent layer misalignment; and finally, the
corneal flap should be dissected gently and slowly to avoid
involving the underlying completed lenticule.
The present study has some limitations: i) The
present study is a single-case report with a small sample size, and
the generalizability of the research conclusions needs to be
further verified by multi-center studies with large sample sizes;
and ii) the follow-up period is relatively short, and the long-term
efficacy (such as corneal stability >1 year after surgery) still
requires extended follow-up for further observation. In response to
the aforementioned limitations, it is the plan to perform
multi-center retrospective studies in the future and to expand the
sample size and extend the follow-up period, to provide more
sufficient clinical evidence for the management of intraoperative
suction loss during SMILE surgery.
In conclusion, the present case demonstrates that
suction loss carries a certain probability (0.17-5.06%) during
SMILE, with two consecutive intraoperative suction losses being
even more uncommon. However, with remedial measures, favorable
postoperative visual outcomes can be secured.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
LW, QS and CP designed the study and coordinated the
clinical evaluation. TQ, QS and SM participated in the diagnosis
and treatment of the patient. LW, TQ, NL and QS were responsible
for data collection and clinical evaluation of corneal conditions
before and after surgery. All authors contributed to the analysis
and interpretation of the laboratory and imaging data and
participated in the writing of the original draft and figure
preparation. QS, SM and CP supervised the study. LW and TQ
critically reviewed and revised the manuscript. SM and CP confirm
the authenticity of all the raw data. All authors have read and
approved the final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Written informed consent was obtained from the
patient for publication of this case report and any accompanying
images.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, artificial
intelligence tool Doubao (ByteDance, Version 11.9.1, based on
Doubao-Seed-1.8) was used to improve the readability and language
of the manuscript, and subsequently, the authors revised and edited
the content produced by Doubao as necessary, taking full
responsibility for the ultimate content of the present
manuscript.
References
|
1
|
Maulvi FA, Desai DT, Kalaiselvan P, Shah
DO and Willcox MDP: Current and emerging strategies for myopia
control: A narrative review of optical, pharmacological,
behavioural, and adjunctive therapies. Eye (Lond). 39:2635–2644.
2025.PubMed/NCBI View Article : Google Scholar
|
|
2
|
World Health Organization (WHO): World
report on vision. World Health Organization, Geneva, 2019.
|
|
3
|
Wolffsohn JS, Whayeb Y, Logan NS and Weng
R: International Myopia Institute Ambassador Group*. IMI-global
trends in myopia management attitudes and strategies in clinical
practice-2022 update. Invest Ophthalmol Vis Sci.
64(6)2023.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Luensmann D, Schaeffer JL, Rumney NJ,
Stanberry A and Fonn D: Magnitude of astigmatism-A comparison
between eyes. Cont Lens Anterior Eye. 45(101510)2022.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Song J, Cao H, Chen X, Zhao X, Zhang J, Wu
G and Wang Y: Small incision lenticule extraction (SMILE) versus
laser assisted stromal in situ keratomileusis (LASIK) for
astigmatism corrections: A systematic review and meta-analysis. Am
J Ophthalmol. 247:181–199. 2023.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Kim TI, Alió Del Barrio JL, Wilkins M,
Cochener B and Ang M: Refractive surgery. Lancet. 393:2085–2098.
2019.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Wang M, Chen Y and Zhang Y: Laser-assisted
subepithelial keratomileusis substituted for an aborted
small-incision lenticule extraction due to large black area
formation. J Cataract Refract Surg. 46:913–917. 2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Yao L, Zhang M, Wang D, Zhao Q, Wang S and
Bai H: Small incision lenticule extraction (SMILE) and laser in
situ keratomileusis (LASIK) used to treat myopia and myopic
astigmatism: A systematic review and meta-analysis of randomized
clinical trials. Semin Ophthalmol. 38:283–293. 2023.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Chiam NPY and Mehta JS: Comparing
patient-reported outcomes of laser in situ keratomileusis and
small-incision lenticule extraction: A review. Asia Pac J
Ophthalmol (Phila). 8:377–384. 2019.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Wong AHY, Cheung RKY, Kua WN, Shih KC,
Chan TCY and Wan KH: Dry eyes after SMILE. Asia Pac J Ophthalmol
(Phila). 8:397–405. 2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Huang Q, Liu L, Ma P, Sun Y, Wang Z, Bai J
and Liu T: Grading for suction loss in small incision lenticule
extraction. Int Ophthalmol. 43:665–675. 2023.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Wan KH, Lin TPH, Lai KHW, Liu S and Lam
DSC: Options and results in managing suction loss during
small-incision lenticule extraction. J Cataract Refract Surg.
47:933–941. 2021.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wang Y, Ma J, Zhang J, Dou R, Zhang H, Li
L, Zhao W and Wei P: Incidence and management of intraoperative
complications during small-incision lenticule extraction in 3004
cases. J Cataract Refract Surg. 43:796–802. 2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Krueger RR and Meister CS: A review of
small incision lenticule extraction complications. Curr Opin
Ophthalmol. 29:292–298. 2018.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Li SM, Zhan S, Li SY, Peng XX, Hu J, Law
HA and Wang NL: Laser-assisted subepithelial keratectomy (LASEK)
versus photorefractive keratectomy (PRK) for correction of myopia.
Cochrane Database Syst Rev. 2(CD009799)2016.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Wilson SE: Biology of keratorefractive
surgery-PRK, PTK, LASIK, SMILE, inlays and other refractive
procedures. Exp Eye Res. 198(108136)2020.PubMed/NCBI View Article : Google Scholar
|