Introduction
Primary angiitis of the central nervous system
(PACNS) is a rare condition characterized by inflammation of the
blood vessels in the CNS, without systemic vascular involvement
(1). The incidence rate of PACNS
has been reported to be 2.4 cases per million individuals per year.
PACNS primarily affects adults aged 30-60 years, with no clear sex
predominance (58 women and 43 men in a 101-patient cohort), and is
further associated with a number of neurological symptoms including
headaches, cognitive impairment, seizures and hemiparesis (1,2).
Serum inflammatory markers are typically normal (for example,
acute-phase reactants and autoantibodies, including antinuclear
antibodies, antineutrophil cytoplasm antibodies and
antiphospholipid antibodies); however cerebrospinal fluid
abnormalities are present in 80-90% of cases, typically showing a
mildly increased leucocyte count and total protein concentration
(1). Diagnosis can be made through
magnetic resonance angiography, conventional angiography or tissue
biopsy, revealing characteristic histopathological patterns that
are commonly classified as granulomatous, lymphocytic or
necrotizing vasculitis (1,3). Given the severity of the disease and
its associated high mortality rate (reported to range from 6 to 17%
across cohorts), early detection and timely initiation of treatment
are important for reducing morbidity and mortality, as they help
prevent irreversible damage and improve long-term outcomes
(1,3).
Spinal cord vasculitis is a rare subtype of PACNS
that primarily affects the blood vessels of the spinal cord,
leading to localized inflammation and ischemic damage. Spinal cord
vasculitis is typically characterized by symptoms such as limb
weakness, numbness and urinary or bowel dysfunction (4). Due to its rarity and the overlap of
clinical and imaging features with other conditions (for example,
intramedullary tumors, inflammatory myelitis and spinal cord
infarction), spinal cord vasculitis is often misdiagnosed or
overlooked. The present report outlines a case of spinal cord
vasculitis diagnosed through biopsy, with the aim to elucidate the
clinical diagnostic challenges and underlying pathophysiology of
this subtype and to enhance the understanding of this rare disease
for improved clinical recognition and early diagnosis.
Case report
In March 2023, a 31-year-old male patient presented
to the Department of Neurosurgery, Beijing Tiantan Hospital
(Beijing, China) with a 4-month history of progressive right-hand
numbness and weakness. Initially, the numbness and weakness were
localized to the right hand, along with muscle atrophy and pain in
the right shoulder, back and upper arm. The symptoms progressed to
involve the right lower limb, with a walking-on-cotton sensation. A
cervical spine MRI revealed a longitudinally extensive
intramedullary lesion with cord swelling, showing T1 hypointensity
and T2 hyperintensity from C3 to T1 with heterogeneous enhancement
(Fig. 1A a-1). These findings,
combined with the clinical presentation, led to the suspicion of an
intramedullary spinal cord tumor, given the expansile, mass-like
appearance on MRI and the relatively mild clinical deficits despite
the marked imaging abnormalities.
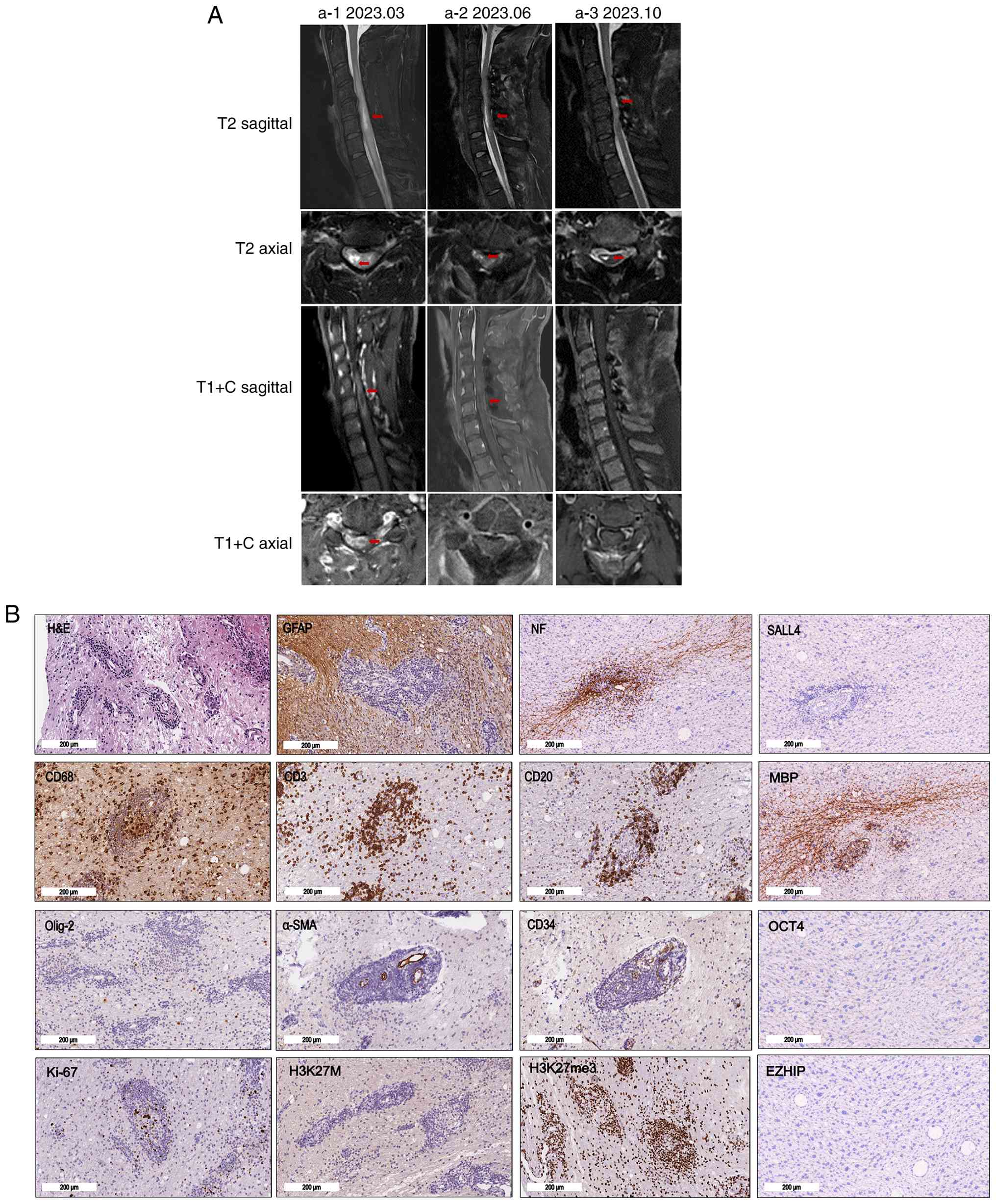 | Figure 1Cervical lesions detected through MRI
scans and pathological findings. (A) Serial cervical spine MRI
findings. (a-1) Baseline MRI demonstrated a longitudinally
extensive intramedullary lesion with cord swelling on T2-weighted
images and heterogeneous enhancement on post-contrast T1-weighted
images (arrows). (a-2) Immediate postoperative MRI showed
persistent intramedullary signal abnormality with residual
enhancement (arrows). (a-3) Follow-up MRI after high-dose
corticosteroid therapy showed interval reduction of the lesion and
no residual enhancement (arrows). (B) H&E staining showing
vasculocentric lymphocytic infiltration involving small vessel
walls, consistent with small-vessel vasculitis.
Immunohistochemistry showed CD3+, CD20+,
CD68+, α-SMA+, CD34+,
GFAP+, NF+, MBP+,
Olig-2-, OCT4-, H3K27me3+,
EZHIP-, SALL4-, H3K27M- and
Ki-67+ (scattered) staining, supporting small-vessel
vasculitis and arguing against a neoplastic process. Scale bar, 200
µm. α-SMA, α-smooth muscle actin; GFAP, glial fibrillary acidic
protein; NF, neurofilament; MBP, myelin basic protein; Olig2,
oligodendrocyte transcription factor 2; EZHIP, enhancer of zeste
inhibitory protein; SALL4, Spalt-like transcription factor 4;
H3K27M, histone H3 (mutated K27M); H3K27me3, histone H3 (tri methyl
K27); C, contrast-enhanced. |
In June 2023, after a waiting period for hospital
admission, the patient underwent microsurgical biopsy of the
intramedullary spinal cord lesion, which was preoperatively
suspected to be neoplastic based on the clinical presentation and
MRI findings. No corticosteroids were administered prior to the
biopsy. In the immediate postoperative period, the patient
experienced neurological worsening, with a new onset of bilateral
lower limb numbness, particularly on the right, along with
stiffness and gait instability. Although there was no bladder or
bowel dysfunction, a postoperative MRI revealed persistent
intramedullary signal abnormality with T1 hypointensity and T2
hyperintensity, along with residual linear enhancement, raising
further diagnostic uncertainty, as it was difficult to determine
whether these postoperative findings reflected surgical changes,
ongoing inflammation or residual neoplastic pathology (Fig. 1A a-2).
A neurological examination revealed atrophy of the
right thenar and hypothenar muscles, decreased strength in wrist
flexion and extension, as well as finger adduction/abduction on the
right side. Reflexes were diminished in the right upper limb but
hyperactive in both lower limbs. A sensory examination demonstrated
decreased pinprick and vibratory sensations in the right hand and
both lower limbs. The Babinski sign (extensor plantar response) was
positive on the right side. These findings pointed to a severe,
possibly inflammatory spinal cord lesion, yet differentiation from
a neoplasm remained challenging based on the clinical and imaging
data alone.
Laboratory tests were unremarkable, including
routine blood, urine and stool tests, serum biochemistry,
coagulation profile, infectious disease screening and tumor
markers. The immunological work-up was negative, including an
autoantibody panel, inflammatory markers (C-reactive protein and
erythrocyte sedimentation rate), antistreptolysin O, rheumatoid
factor, anticardiolipin antibody, antineutrophil cytoplasmic
antibody, thyroid autoantibodies, complement component 3 and
complement component 4, and angiotensin-converting enzyme.
Cerebrospinal fluid analysis showed no abnormalities except for
mildly elevated total protein levels. Notably, serological and
cerebrospinal fluid antibody tests against aquaporin-4, myelin
oligodendrocyte glycoprotein, glial fibrillary acidic protein
(GFAP) and myelin basic protein (MBP), as well as paraneoplastic
antibody tests (including anti-Hu, anti-Yo, anti-Ri,
anti-CV2/CRMP5, anti-Ma2 and anti-amphiphysin) were negative. The
absence of serological and cerebrospinal fluid markers added to the
difficulty in establishing a clear diagnosis.
The development in diagnosis came with the
histopathological analysis of the spinal cord biopsy in June 2023
conducted by the Department of Pathology, Beijing Tiantan Hospital
(Beijing, China), which revealed lymphocytic infiltration
surrounding the small blood vessels of spinal cord and occasional
epithelioid cell nodules, suggestive of small vessel vasculitis. No
well-formed granulomas or multinucleated giant cells were
identified. Immunohistochemical staining demonstrated inflammatory
cell markers (CD3, CD20 and CD68), supporting a lymphocytic
inflammatory infiltrate. Markers of vascular structures (CD34 and
α-smooth muscle actin) highlighted small vessels, whereas
neural/glial markers (GFAP, neurofilament, MBP and oligodendrocyte
transcription factor 2) characterized the surrounding spinal cord
tissue; tumor-associated markers [OCT4, enhancer of zeste
inhibitory protein (EZHIP), histone H3K27M, histone H3K27me3 and
Spalt-like transcription factor 4 (SALL4)] and the proliferation
marker Ki-67 were included to evaluate for a neoplastic process
(Fig. 1B). The biopsy tissue was
fixed in 10% neutral buffered formalin at room temperature
(overnight) and then paraffin-embedded according to routine
diagnostic procedures in the Department of Pathology, Beijing
Tiantan Hospital. The blocks were sectioned at a thickness of 4 µm,
deparaffinized in xylene and rehydrated through a descending
ethanol series, and then stained with H&E using a commercial
H&E staining kit (cat. no. G1120; Beijing Solarbio Science
& Technology Co., Ltd.), according to the manufacturer's
instructions. Immunohistochemistry was performed on the sections
following standard diagnostic protocols, including antigen
retrieval and blocking with 3% bovine serum albumin at room
temperature for 30 min, followed by HRP-based visualization with
3,3'-diaminobenzidine, with hematoxylin counterstaining. Primary
antibody incubation was performed at 4˚C overnight. The primary
antibodies used were CD3 (ab16669; 1:150 dilution; Abcam), CD20
(ab78237; 1:250 dilution; Abcam), CD68 (ab955; 1:3,000 dilution;
Abcam), α-smooth muscle actin (ab124964; 1:400 dilution; Abcam),
CD34 (ab81289; 1:50 dilution; Abcam), GFAP (ab4674; 1:6,000
dilution; Abcam), neurofilament (ab7794; 1:1,000 dilution; Abcam),
MBP (ab7349; 1:4,000 dilution; Abcam), oligodendrocyte
transcription factor 2 (Olig2) (ab109186; 1:100 dilution; Abcam),
OCT4 (ab181557; 1:1,000 dilution; Abcam), Ki-67 (ab16667; 1:200
dilution; Abcam), EZHIP (ab316856; 1:500 dilution; Abcam), histone
H3 (mutated K27M) (ab190631; 1:1,000 dilution; Abcam), histone H3
(tri methyl K27) (ab192985; 1:500 dilution; Abcam) and SALL4
(ab29112; 1:100 dilution; Abcam), which were used according to the
manufacturer's instructions. Signal detection was performed using
the EnVision™ + System-HRP (Dako; Agilent Technologies,
Inc.), i.e., species-specific HRP-labeled polymer reagents selected
according to the host species of the primary antibodies, followed
by DAB visualization, according to the manufacturer's instructions.
Slides were examined and imaged using a bright-field light
microscope. The final pathology interpretation supporting
small-vessel vasculitis became available 2 weeks after the biopsy
(after completion of the histopathological review and ancillary
immunohistochemistry analysis of the specimen). With these
findings, a diagnosis of PACNS was established. Therefore, the
present case report underscores the diagnostic complexity, as the
longitudinally extensive spinal cord lesion with persistent
enhancement mimicked a tumor.
In July 2023, 4 weeks after the biopsy, the patient
was admitted to the Department of Neurology, Beijing Tiantan
Hospital and treated with high-dose intravenous methylprednisolone,
followed by rehabilitation. During this hospitalization,
intravenous methylprednisolone was administered for 9 days (500 mg
for 3 days, 250 mg for 3 days and 120 mg for 3 days), followed by
oral prednisone acetate (60 mg/day), initiated after completion of
intravenous methylprednisolone, and tapered by 5 mg (one 5-mg
tablet) each week until discontinuation, for a total duration of 12
weeks. Concomitant medications during corticosteroid therapy
included potassium chloride sustained-release (1 g twice daily),
calcitriol (0.25 µg twice daily), calcium carbonate (0.75 g three
times daily) and omeprazole (20 mg once daily) to prevent
steroid-induced side effects, with supportive therapy using
mecobalamin (0.5 mg three times daily) and vitamin B1 (10 mg three
times daily) for nerve nutrition. At the 4-month follow-up, the
patient exhibited an improvement in limb weakness and gait
stability. MRI scans at the follow-up in October 2023 demonstrated
a reduction in lesion size and absence of enhancement (Fig. 1A a-3). Cyclophosphamide or other
immunosuppressive agents were not administered during this period.
Given the spinal cord-limited, biopsy-proven small-vessel
vasculitis and the excellent response of the patient to
corticosteroids, careful long-term clinical and MRI follow-up were
opted for, rather than immediate additional immunosuppression. At
the last follow-up, 2 years after the initiation of corticosteroid
therapy (June 2025), the modified Rankin scale value (5) of the patient was 2 (slight
disability; unable to carry out all previous activities but able to
look after own affairs without assistance), without relapse. The
clinical treatment timeline of the patient is summarized in
Fig. S1.
Discussion
PACNS is a rare condition characterized by
inflammatory lesions confined to the blood vessels of the brain and
spinal cord (1). The present case
report highlights two key challenges, namely the difficulty in
distinguishing isolated spinal vasculitis from spinal cord tumors,
given their similar radiographic presentations, such as marked
swelling and persistent enhancement, as well as the uncertainty
regarding the prevention of recurrence. While corticosteroid
treatment led to clinical improvement, the long-term strategy for
preventing recurrence without immunosuppression remains unclear. In
a number of cases, immunosuppressive agents such as
cyclophosphamide may be needed, but the timing and choice of such
treatments must be individualized based on patient response (for
example, relapse or radiological/clinical progression despite
glucocorticoid treatment) and risk factors (for example, disease
severity). In the present report, ‘isolated spinal vasculitis’
referred to spinal cord-limited PACNS. Given that the Calabrese and
Mallek diagnostic criteria were developed for cerebral disease and
have not been specifically validated for spinal cord-limited
vasculitis, histopathological analysis remains key in diagnosis
(6,7). Histopathologically, PACNS is commonly
classified into granulomatous, lymphocytic and necrotizing pattern
categories. The biopsy conducted in the present study showed
vasculocentric lymphocytic inflammation involving small vessel
walls with occasional epithelioid cell nodules, consistent with
predominantly lymphocytic small-vessel vasculitis with possible
focal granulomatous features (1,8).
These nodules likely represent focal epithelioid histiocyte
aggregates, as well-formed granulomas or multinucleated giant cells
were not identified. CD4 and CD8 immunostaining was not performed
in the present case, therefore information on the CD4 and CD8
staining pattern or the CD4/CD8 ratio is not available. Future
studies with expanded T-cell subset profiling may further
characterize the inflammatory microenvironment. IgG4-related
disease is a recognized mimic of inflammatory CNS disorders and
should be considered in the differential diagnosis of suspected CNS
vasculitis (9). In the present
case, IgG4 immunostaining was not performed as it was not included
in the initial immunohistochemistry panel at the time of biopsy,
and IgG4-related disease was not specifically considered given the
absence of typical clinicoradiologic features, such as dural
thickening/enhancement or systemic manifestations (9). Examples of such clinical
manifestations of IgG4-RD include swelling of salivary glands
(especially the parotid glands), fever, weight loss and multi-organ
involvement. Radiologically, IgG4-RD is often associated with
uniform organ enlargement and enhancement, which can be detected
using MRI or CT scans. However, IgG4 staining may be useful when
feasible in similar cases. Advanced imaging may provide
complementary information beyond conventional spinal MRI when CNS
vasculitis is suspected. While a high-resolution contrast-enhanced
vessel wall MRI may add diagnostic specificity in cerebral
vasculitis (10), its application
in spinal cord-limited disease remains technically challenging and
is not routinely available due to the small size and complex
anatomy of spinal cord vessels, as well as the influence of
surrounding bone structures and artifacts. Therefore, this was not
performed on the present patient. Neurological deterioration after
primary spinal cord biopsy has previously been reported. For
example, in a multicenter series of 61 primary spinal cord
biopsies, postoperative neurological worsening (for example, new or
worsened motor and/or sensory deficits) occurred in 47.5% (29/61)
of patients; of those who worsened, 48.3% (14/29) recovering within
3 weeks (11).
For severe, rapidly progressive or generalized
PACNS, induction therapy with high-dose corticosteroid combined
with cyclophosphamide is commonly used in clinical practice and
supported by retrospective cohort experience and contemporary
reviews (3,12,13).
Despite the absence of immunosuppressants, the patient described in
the present study has remained free from recurrence for 2 years,
raising an important therapeutic question, namely under what
circumstances immunotherapy should be initiated. Recurrence in
PACNS, particularly when limited to the spinal cord, is a
recognized risk and cases in the literature report that relapses
may occur after prolonged intervals of ≥1 year (for example, 13
months later, or cerebral involvement emerging 5 years later)
(4,13,14).
In large cohorts of patients with PACNS, relapses have been
observed in 27% of cases. Accordingly, long-term surveillance is
warranted even after apparent clinical stability. The optimal
duration of therapy remains uncertain and maintenance strategies
using steroid-sparing agents (such as azathioprine, mycophenolate
mofetil, methotrexate or other immunosuppressants), are often
considered (3,12,13).
Previous studies appear to advocate combination induction and/or
maintenance immunosuppression to mitigate relapse risk in PACNS
(3,12,13).
Furthermore, although agents such as cyclophosphamide are commonly
used as part of induction and relapse-prevention strategies in
PACNS, their associated toxicities, particularly in younger
patients, complicate this decision, given the risks of bladder
toxicity (hemorrhagic cystitis reported in 12-41% in high
cumulative-dose cohorts), gonadal toxicity (ovarian failure
reported in 26% in a classic SLE cohort), and late malignancies
(e.g., increased risks of bladder cancer and acute myeloid leukemia
reported after exposure in vasculitis cohorts) (15,16).
In addition, late-onset brain involvement (namely, delayed
intracranial extension after an initially spinal cord-limited
presentation), further complicates therapeutic decisions and
underscores the need for long-term clinical/MRI surveillance and
individualized consideration of treatment duration.
PACNS involving the spinal cord is rare. In the Mayo
Clinic 40-year cohort, spinal cord involvement occurred in 10/216
cases (4.6%), whereas truly spinal cord-limited disease was
exceptionally rare (1/216, 0.5%) (6). These occurrence rates add to the
diagnostic complexity, as spinal cord tumors and other inflammatory
conditions (such as inflammatory myelitis, demyelinating diseases
such as NMOSD or MS, and spinal cord infarction) are often more
strongly considered. The course of the present case report suggests
that isolated spinal vasculitis may have a favorable prognosis,
particularly when diagnosed early and managed appropriately.
However, further studies are required to clarify the long-term
outcomes and role of immunosuppressive therapy in preventing
recurrence. From a mechanistic perspective, emerging work in
neurovascular inflammation highlights the role of endothelial
dysfunction and inflammatory mediators in regulating CNS vascular
integrity (1,17). Although not examined in the present
case, future studies integrating immunophenotyping and molecular
profiling may help clarify the pathophysiology of spinal
cord-limited vasculitis and identify potential therapeutic targets,
consistent with the evolving perspective that CNS tissues are not
absolutely immune-privileged.
Supplementary Material
Clinical timeline summary. Timeline of
symptom onset, neurosurgical biopsy, pathology interpretation,
initiation of high-dose corticosteroid therapy and follow-up
outcomes in the present case of spinal cord-limited primary central
nervous system vasculitis. IV, intravenous; mRS, modified Rankin
scale.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by the Youth Talent
Development Program of Tiantan Hospital-Chinese Institute for Brain
Research, Beijing (grant no. 202310) and the Beijing Municipal
Administration of Hospitals Incubating Program (grant no.
PX2025018).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
DCT contributed to the conception and design of the
study, supervised the clinical management of the patient and
critically revised the manuscript. YY contributed to data
collection, clinical data review and analysis, interpretation of
imaging findings, and drafting of the manuscript. GHD contributed
to the histopathological analysis and interpretation, and
preparation of the pathological figures. All authors have read and
approved the final version of the manuscript. YY and DCT confirm
the authenticity of all the raw data.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Written informed consent for publication of the
clinical information and images was obtained from the patient.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Salvarani C, Brown RD Jr and Hunder GG:
Adult primary central nervous system vasculitis. Lancet.
380:767–777. 2012.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Salvarani C, Brown RD Jr, Calamia KT,
Christianson TJ, Weigand SD, Miller DV, Giannini C, Meschia JF,
Huston J III and Hunder GG: Primary central nervous system
vasculitis: Analysis of 101 patients. Ann Neurol. 62:442–451.
2007.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Beuker C, Schmidt A, Strunk D, Sporns PB,
Wiendl H, Meuth SG and Minnerup J: Primary angiitis of the central
nervous system: Diagnosis and treatment. Ther Adv Neurol Disord.
11(1756286418785071)2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Salvarani C, Brown RD Jr, Calamia KT,
Christianson TJ, Huston J III, Meschia JF, Giannini C, Miller DV
and Hunder GG: Primary CNS vasculitis with spinal cord involvement.
Neurology. 70:2394–2400. 2008.PubMed/NCBI View Article : Google Scholar
|
|
5
|
van Swieten JC, Koudstaal PJ, Visser MC,
Schouten HJ and van Gijn J: Interobserver agreement for the
assessment of handicap in stroke patients. Stroke. 19:604–607.
1988.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Salvarani C, Brown RD Jr, Christianson
TJH, Huston J III, Giannini C and Hunder GG: PCNSV with spinal cord
involvement: A 40-year single-center study. Stroke. 56:1149–1158.
2025.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Calabrese LH and Mallek JA: Primary
angiitis of the central nervous system. Report of 8 new cases,
review of the literature, and proposal for diagnostic criteria.
Medicine (Baltimore). 67:20–39. 1988.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Giannini C, Salvarani C, Hunder G and
Brown RD: Primary central nervous system vasculitis: Pathology and
mechanisms. Acta neuropathologica. 123:759–772. 2012.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Lanzillotta M, Mancuso G and Della-Torre
E: Advances in the diagnosis and management of IgG4 related
disease. BMJ. 369(m1067)2020.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Kang N, Qiao Y and Wasserman BA:
Essentials for interpreting intracranial vessel wall MRI results:
State of the art. Radiology. 300:492–505. 2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Mallereau CH, Dannhoff G, Todeschi J,
Severac F, Aghakhani N, Parker F, Benali A, Ganau M, Hamdan N, Le
Van T, et al: Tips and tricks of spinal cord biopsy: Insights from
a multicenter series of 61 patients. J Neurooncol. 173:719–729.
2025.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Birnbaum J and Hellmann DB: Primary
angiitis of the central nervous system. Arch Neurol. 66:704–709.
2009.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Salvarani C, Brown RD Jr, Christianson TJ,
Huston J III, Giannini C, Miller DV and Hunder GG: Adult primary
central nervous system vasculitis treatment and course: Analysis of
one hundred sixty-three patients. Arthritis Rheumatol.
67:1637–1645. 2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Bhatele P, Vibha D, Singh RK, Elavarasi A
and Tripathi M: Primary CNS vasculitis presenting with spinal cord
involvement: A rare presentation. Neurol Sci. 43:727–730.
2022.PubMed/NCBI View Article : Google Scholar
|
|
15
|
de Souza AWS, Dantas JG, Montandon ACOES,
Calich AL, Mont' Alverne ARS, Gasparin AA, Bianchi D, Yuki EFN,
Sacilotto N, Dos Reis Neto ET, et al: Position statement of the
Brazilian society of Rheumatology on mesna use as a preventive
therapy for bladder disease in patients with systemic autoimmune
diseases and systemic vasculitis under cyclophosphamide treatment.
Adv Rheumatol. 64(41)2024.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Faurschou M, Sorensen IJ, Mellemkjaer L,
Loft AGR, Thomsen BS, Tvede N and Baslund B: Malignancies in
Wegener's granulomatosis: Incidence and relation to
cyclophosphamide therapy in a cohort of 293 patients. J Rheumatol.
35:100–105. 2008.PubMed/NCBI
|
|
17
|
Abbott NJ: Inflammatory mediators and
modulation of blood-brain barrier permeability. Cell Mol Neurobiol.
20:131–147. 2000.PubMed/NCBI View Article : Google Scholar
|















