Introduction
Use of nanomaterials in medicine began in the
mid-20th century and has increased steadily in previous years
(1,2). The ability to protect tissues from
ischemia, as well as antimicrobial properties that may help defend
the host against pathogens, has encouraged researchers to
investigate carbon-based nanomaterials (2). Fullerenes have become a central focus
of this research. The antioxidant properties of nanomaterials such
as fullerenes, along with their protective effects against ischemia
and antimicrobial activity, facilitate their potential use as
therapeutic agents (3-5).
Among fullerene derivatives, fullerenol C60 exhibits
higher aqueous solubility than other fullerene structures due to
the presence of hydroxyl groups on its surface (6). The water solubility of fullerenol
C60 makes it the most ideal nanomaterial among
fullerenes, which also simplifies its use in in vivo
(6,7).
The cecal ligation and puncture (CLP) method is a
widely used procedure in animal models to induce polymicrobial
sepsis and closely mimics the pathophysiology of clinical sepsis
(8). This procedure involves
surgically ligating a specific portion of the cecum and then
puncturing it. CLP leads to an increased microbial load in the
abdominal cavity and translocation of bacteria into the
bloodstream, thereby triggering multiple organ failure,
inflammation and immune responses that simulate clinical sepsis.
Due to the immunological and physiological responses resembling
clinical sepsis, this model is especially preferred in studies
investigating sepsis pathogenesis and treatment (8,9). The
CLP method is widely accepted as a reliable and reproducible
experimental model for investigating sepsis-related complications
and evaluating potential therapeutic interventions (8)
Sepsis is a life-threatening clinical condition
caused by infection (10,11); it is estimated to account for ~49
million cases and ~11 million deaths worldwide each year, and
remains one of the leading causes of mortality in Intensive Care
Units (12).
Therefore, preventing sepsis or, if prevention is
not possible, identifying effective treatment options is a primary
focus for researchers. Although various therapeutic approaches,
including early broad-spectrum antibiotics, fluid resuscitation,
vasopressor support and adjunctive corticosteroid therapy, are
routinely used in clinical practice, sepsis continues to be
associated with high mortality, and no definitive therapy has yet
demonstrated a consistent survival benefit (13). The development of multiple organ
failure, particularly involving vital organs such as the liver,
kidneys, lungs, brain and heart, presents significant challenges
for clinicians, as it necessitates supportive interventions (for
example, mechanical ventilation, vasopressor therapy and renal
replacement therapy), complicates hemodynamic and metabolic
stabilization, and is associated with increased morbidity and
mortality (14). Sepsis is a
significant global health problem; there are >30 million
patients with sepsis worldwide every year (15). Sepsis-associated encephalopathy
(SAE) is one of the symptoms of sepsis and has a clinical
presentation ranging from confusion to delirium and coma. The
symptom affects up to 70% of patients with sepsis (15) and increases the mortality rate
(12). In the study by Krzyżaniak
et al (12), which included
443 patients with sepsis, it was reported that 240 patients
developed SAE; the mortality rate in the SAE group (45.42%) was
significantly higher than that in the non-SAE group (7.88%). The
liver plays a key role in immune responses through the synthesis of
acute-phase proteins and complement components, cytokine production
and pathogen clearance via Kupffer cells, functioning as a critical
immunometabolic defense organ (16). The presence of chronic
comorbidities alongside sepsis markedly increases the risk of
end-organ failure (16,17). Therefore, in the treatment of
sepsis, it is important to implement therapeutic strategies not
only targeting the infectious agent, but also addressing underlying
chronic conditions (14,17).
With the rise of sedentary lifestyles and obesity,
diabetes has become a major global metabolic disease, currently
affecting ~537 million adults worldwide and projected to reach 783
million by 2045(18). Diabetes is
accompanied by chronic hyperglycemia, which increases the
production of reactive oxygen species (ROS) within the cell
(19). These radicals cause
oxidative stress, leading to damage to cell membranes, proteins and
DNA. Oxidative stress serves a role in the development of diabetic
complications such as neuropathy, retinopathy and nephropathy
(20,21). For this reason, antioxidant
therapies are being investigated to reduce the oxidative damage
associated with diabetes (20). In
previous years, nanomaterials exhibiting antioxidant properties,
particularly carbon-based nanomaterials, have emerged as potential
therapeutic agents in this context (21). In the management of diabetes,
reducing oxidative stress is considered as important as controlling
blood glucose levels (19-21).
In diabetes, mitochondrial dysfunction has been
observed; however, the underlying molecular mechanisms have not
been fully elucidated (22),
despite it contributing towards inflammatory processes by driving
increased anaerobic metabolism at the tissue level, thereby
increasing sepsis-related mortality (23). Sepsis-induced heart and kidney
damage is a frequent complication in intensive care units and leads
to high morbidity and mortality (24). The liver is an organ that serves a
vital role in functions such as detoxification, immune regulation
and the antioxidant system (including glutathione production) of
the body (25). Therefore,
protecting the liver during sepsis strengthens the antioxidant
capacity of the body. In sepsis, increased blood-brain barrier
permeability, overproduction of proinflammatory cytokines, such as
tumor necrosis factor-α (TNF-α), IL-1β and IL-6, their infiltration
into the brain parenchyma, activation of glial cells and
sepsis-related mitochondrial dysfunction lead to an enhanced
neuroimmune response, resulting in neuronal cell death (23).
In diabetic mice, fullerenol C60, with
its notable antioxidant capacity, has been shown to protect against
ischemia-reperfusion (I/R) injury in muscle, heart and lung tissues
(26,27). Yavuz et al (28) also observed the protective effects
of fullerenol C60 on liver tissue in a liver I/R injury
model. Although very few studies have demonstrated the
effectiveness of fullerenol C60 in sepsis, in our
previous study, its protective effects on the kidneys and lungs
were demonstrated in an abdominal sepsis model (29). Furthermore, the present study aimed
to prevent multiple organ failure that may develop due to sepsis in
diabetic rats by utilizing the antioxidant properties of fullerenol
C60, as well as investigate its protective effects on
the heart, brain and liver.
Materials and methods
In accordance with ARRIVE 2.0 guidelines (30), the present study was conducted at
The Gazi University Laboratory Animal Breeding and Experimental
Research Center (Ankara, Turkey) with approval from The Gazi
University Experimental Animal Ethics Committee (Ankara, Turkey;
approval no. G.Ü.ET-24.046). The present study was also conducted
in accordance with The Guide for the Care and Use of Laboratory
Animals (31).
A total of 30 Wistar albino male rats (6-7 weeks
old; body weight, 200-225 g), obtained from the Gazi University
Laboratory Animal Breeding and Experimental Research Center
(Ankara, Türkiye), were used, comprising 24 rats with
streptozotocin-induced diabetes and 6 healthy rats that served as
the control group. The number of rats was determined according to
the maximum number of rats permitted by the ethics committee. The
rats were housed under a 12 h light/dark cycle, with an
environmental temperature of 20-21˚C and humidity of 45-55%. The
rats had free access to food and water.
Induction of diabetes
To induce diabetes, the same procedures were applied
as outlined in our previous studies (32,33).
Experimental design
Rats were randomly assigned to four groups: Six in
the control group and eight in all other groups. Before laparotomy,
all rats were anesthetized through intramuscular administration of
ketamine (50 mg/kg; Ketalar®; Pfizer Türkiye) and
xylazine hydrochloride (10 mg/kg; Dogalazin® 2% xylazine
injectable solution; Doğa İlaç) Procedures were performed under a
heating lamp with the rats in the supine position to prevent heat
loss during the experiment. The rats were treated as follows: i) In
the control (S) group (n=6), after anesthesia, rats underwent
midline laparotomy and the abdomen was closed without further
intervention; ii) in the diabetic control (D) group (n=8), rats
with streptozotocin-induced diabetes underwent the same sham
laparotomy procedure as the S group without CLP induction; iii) in
the diabetes + CLP (DM/SEP) group (n=8), rats underwent midline
laparotomy followed by cecal ligation and puncture (CLP), performed
using a 21-gauge syringe needle at the antimesenteric border, 1-2
cm distal to the ileocecal valve. The cecum was repositioned, and
the abdomen was closed; iv) in the diabetes + CLP + fullerenol
C60 (FUL/C-60) group (n=8), rats underwent the same CLP
procedure as the DM/SEP group. At 30 min post-abdominal closure,
fullerenol C60 (100 mg/kg) was administered
intraperitoneally. The route of administration and dosage were
determined based on previous studies (26-29).
At 24 h post-laparotomy, the rats were anesthetized with ketamine
hydrochloride (50 mg/kg) and xylazine hydrochloride (10 mg/kg), and
sacrificed via intracardiac blood collection (5-10 ml). After
cessation of heartbeat and respiration, animals were monitored for
an additional 2 min. Liver, heart and brain tissues were carefully
harvested for histopathological and oxidative stress analyses, and
blood samples were collected for biochemical evaluation. No rats
were lost during the study. Pre-determined humane endpoints were
established in accordance with institutional animal care guidelines
and included severe lethargy, inability to access food or water,
persistent hypothermia, loss of >20% of body weight or signs of
severe distress; animals meeting any of these criteria were
humanely euthanized.
Histopathological evaluation
Liver, heart and brain tissues were fixed in 10%
neutral-buffered formalin at room temperature (20-25˚C) for 24 h
and embedded in paraffin. Paraffin blocks were sectioned at a 4-µm
thickness using a rotary microtome. After deparaffinization in
xylene and rehydration through graded ethanol, sections were
stained with hematoxylin (5 min) and eosin (2 min) at room
temperature. These sections were two experienced histopathologists,
blinded to the procedures, using a semiquantitative grading scale
(0, no change; +1, minimal change; +2, moderate change; +3, severe
change) (29).
Biochemical evaluation
Oxidative stress assessment in the brain, liver and
heart tissue samples was performed by measuring catalase (CAT)
activity and thiobarbituric acid reactive substance (TBARS) levels.
The TBARS assay for assessing lipid peroxidation was performed
using the TBA method. Tissue homogenates were mixed with
trichloroacetic acid and TBA solution and heated in a boiling water
bath (95-100˚C) for 15 min under acidic conditions. After cooling,
the mixture was centrifuged at 1,600 x g for 10 min at 4˚C, and the
absorbance of the supernatant was measured spectrophotometrically
at 532 nm. Malondialdehyde levels were calculated using
1,1,3,3-tetraethoxypropane as the standard (MilliporeSigma). CAT
activity was measured based on the decomposition of hydrogen
peroxide (H2O2). The decrease in absorbance
due to H2O2 consumption was monitored at 240
nm for 1 min at room temperature using a spectrophotometer, and
enzyme activity was expressed as U/mg protein. For serum
biochemical analysis, blood samples were centrifuged at 1,000 x g
for 10 min at room temperature to obtain serum. Serum AST (cat. no.
04490734190), ALT (cat. no. 04490718190), γ-GGT (cat. no.
03002728122), total bilirubin (cat. no. 05795393190), direct
bilirubin (cat. no. 05795423190) and albumin (cat. no. 05166861190)
levels were measured using commercially available assay kits (Roche
Diagnostics) on an automated biochemical analyzer according to the
manufacturer's instructions.
Statistical analysis
Statistical analysis was performed using SPSS for
Windows (version 26.0; IBM Corp.) employing the tests listed below.
Data distribution was analyzed using the Shapiro-Wilk test.
Comparisons of >2 groups were performed using the Kruskal-Wallis
test followed by Dunn's post hoc test or one-way ANOVA followed by
Tukey's post hoc test, as appropriate. P<0.05 was considered to
indicate a statistically significant difference. Data are expressed
as mean ± SD and median with interquartile range.
Results
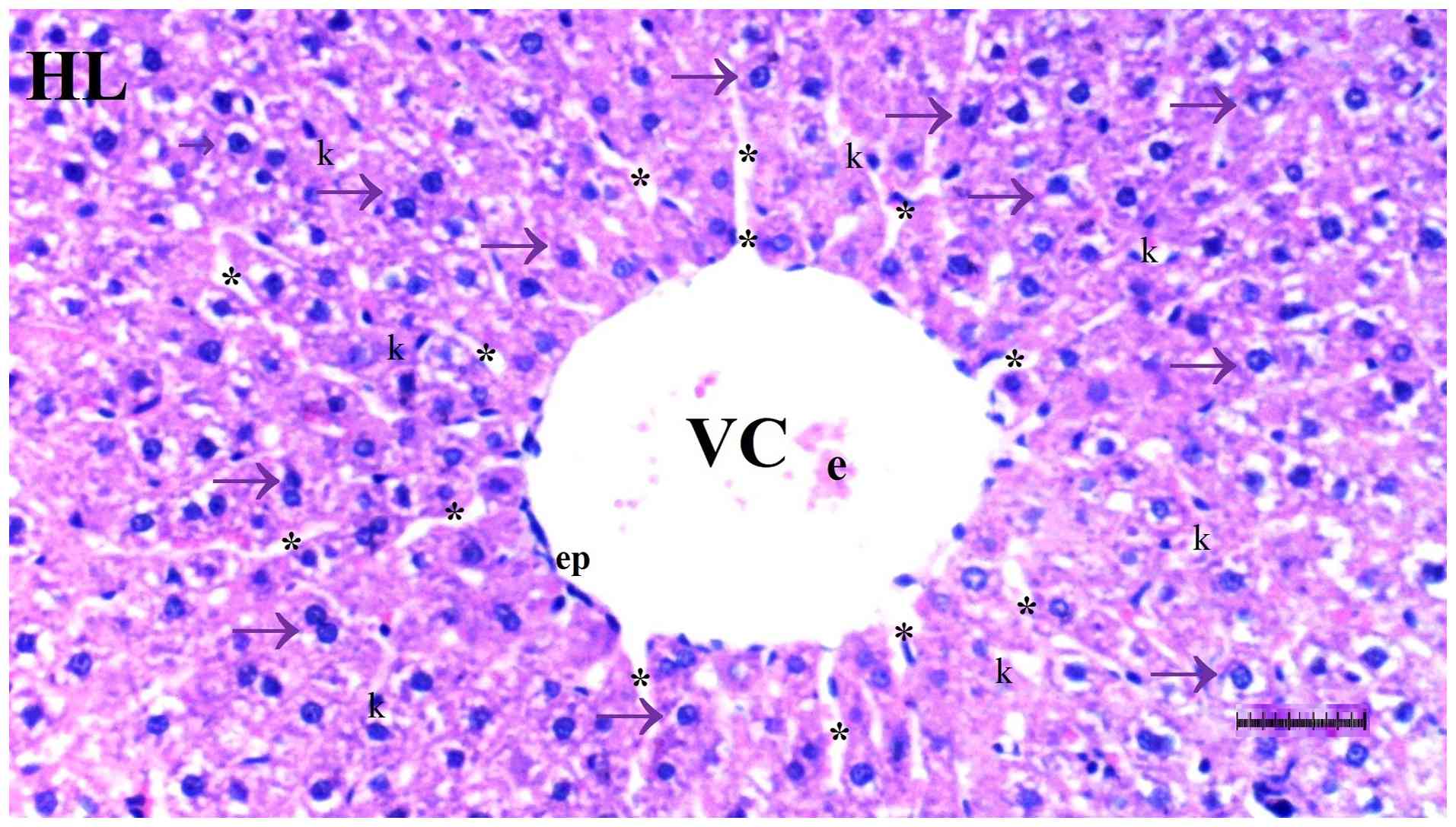
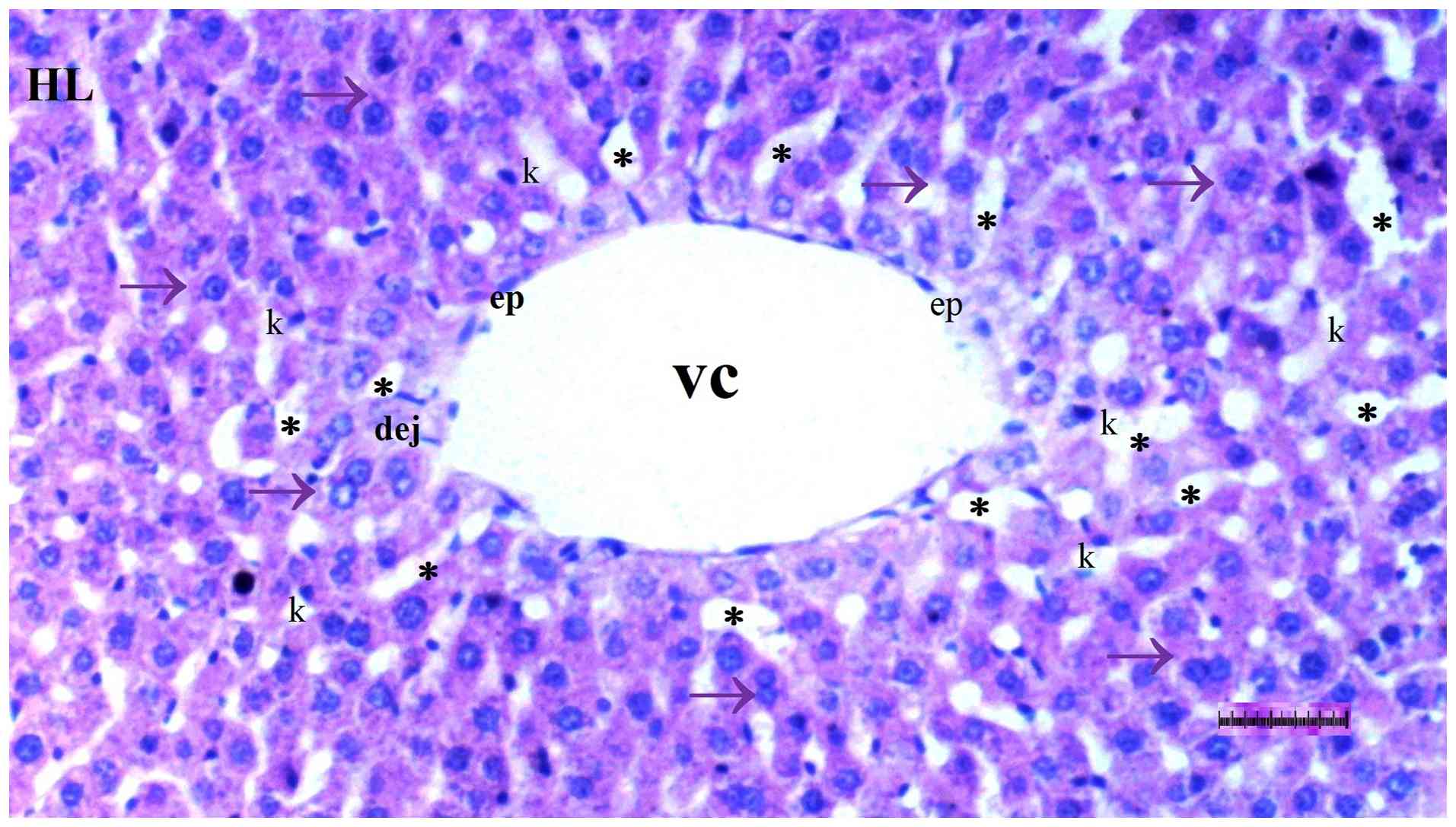
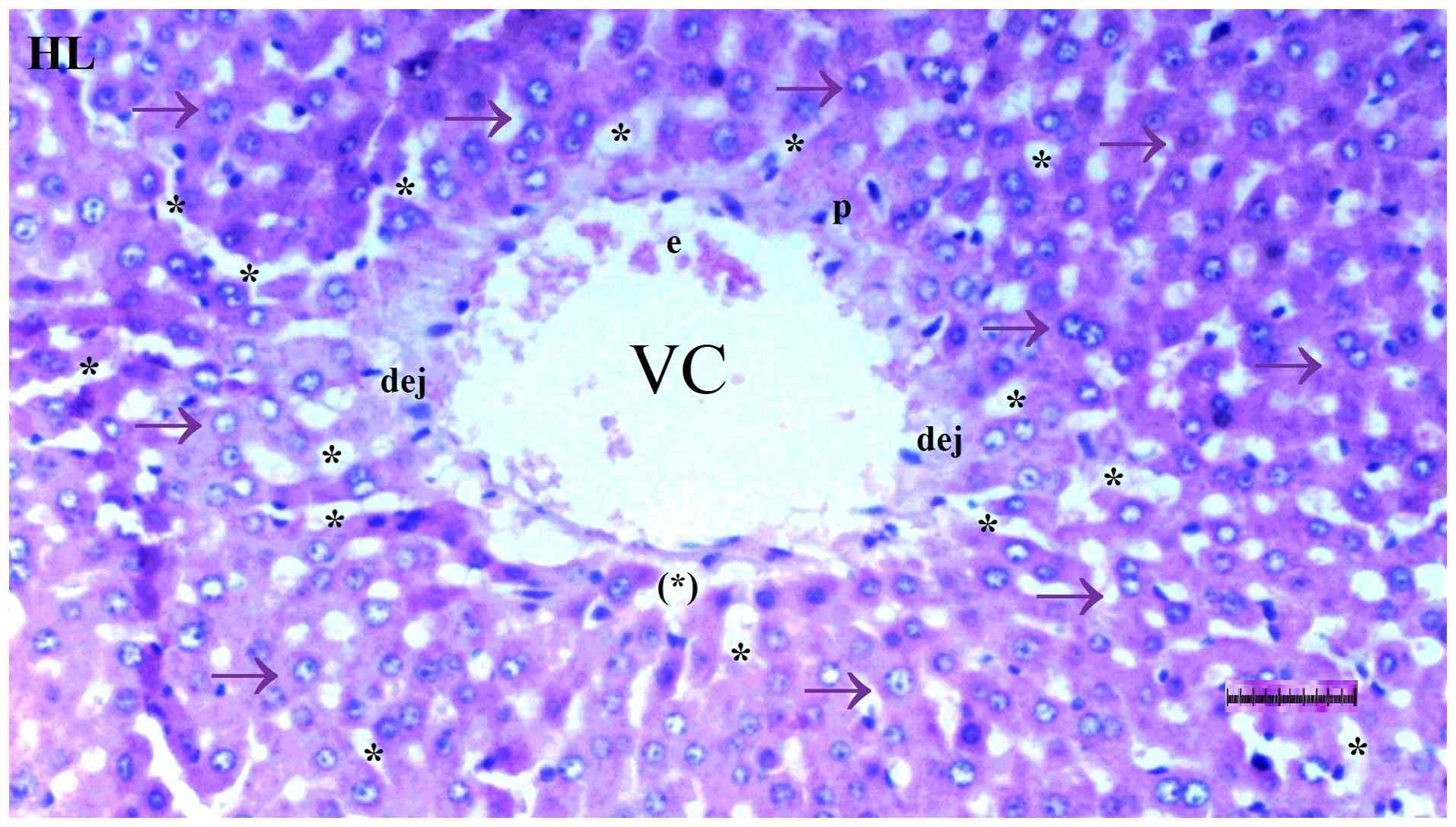
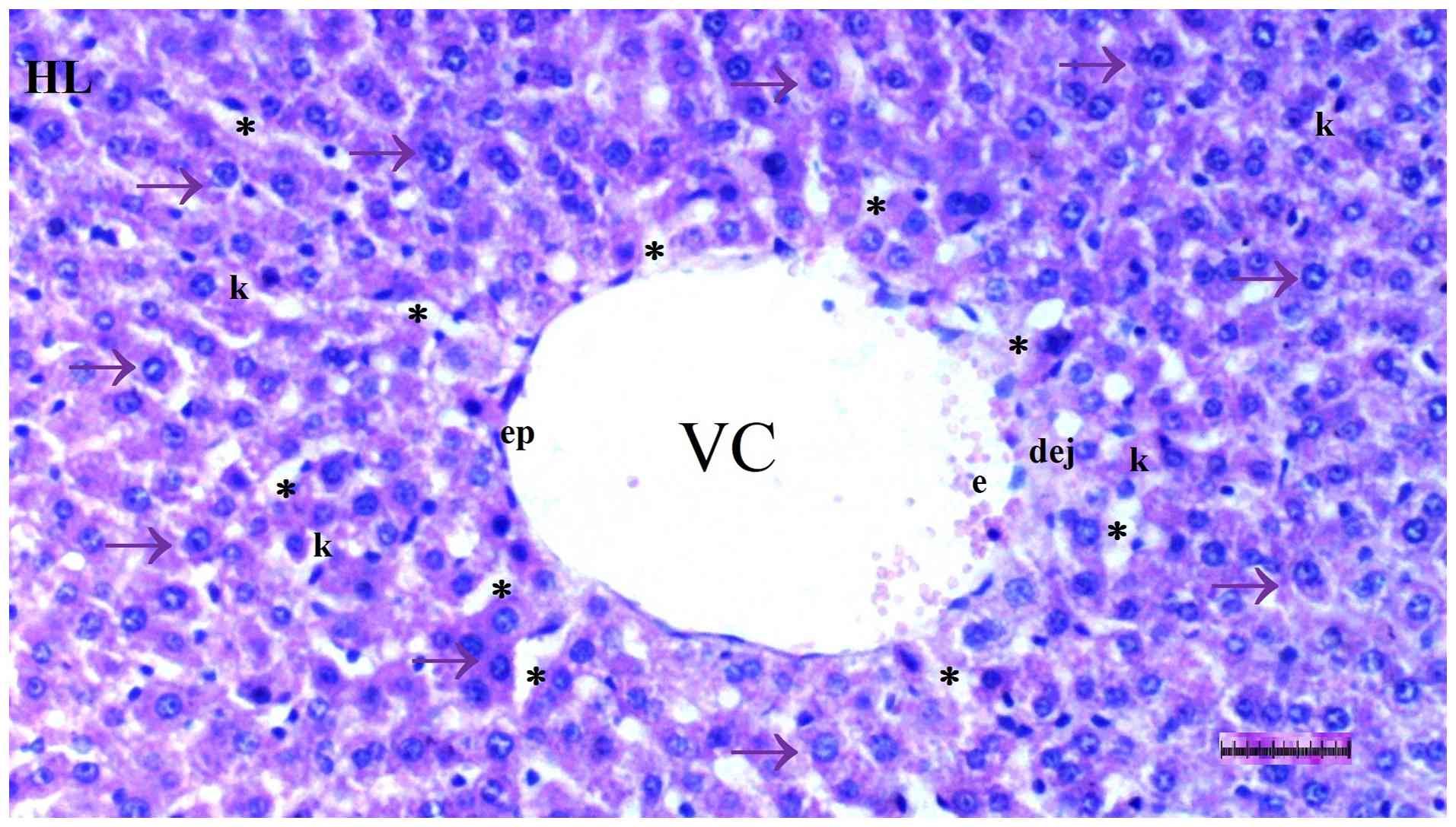
Histopathological examination of liver
tissue
Under light microscopy, hepatocyte degeneration
differed significantly among groups (P=0.017), being more frequent
in the DM/SEP group compared with the S and D groups (P=0.001 and
P=0.013, respectively). However, it was significantly reduced in
the FUL/C-60 group compared with that in the DM/SEP group (P=0.042)
(Table I; Fig. 1, Fig.
2, Fig. 3 and Fig. 4).
 | Table IHistopathological findings of the S
group (n=6), D group (n=8), DM/SEP group (n=8) and FUL/C-60 group
(n=8) in liver tissue. |
Table I
Histopathological findings of the S
group (n=6), D group (n=8), DM/SEP group (n=8) and FUL/C-60 group
(n=8) in liver tissue.
| Feature | S group | D group | DM/SEP | FUL/C-60 | P-value |
|---|
| Hepatocyte
degeneration | 0.50
(0.00-1.00) | 1.00
(0.75-1.00) | 1.50
(1.00-2.25)a,b | 1.00
(1.00-1.00)c | 0.017 |
| Sinusoidal
dilatation | 0.00
(0.00-1.00) | 1.00
(0.75-1.25) | 1.50
(1.00-2.25)a | 1.00
(0.00-1.00)c | 0.022 |
| Pyknotic cells | 0.00
(0.00-1.00) | 0.50
(0.00-1.00) | 1.00
(1.00-2.00)a,b | 0.50
(0.00-1.00)c | 0.044 |
| Cells undergoing
necrosis | 0.00
(0.00-0.25) | 0.50
(0.00-1.00) | 1.50
(1.00-2.25)a,b | 1.00
(1.00-1.00)a,c | <0.001 |
| Parenchymal MN cell
infiltration | 0.00
(0.00-1.00) | 1.00
(0.00-1.25) | 1.00
(1.00-2.00)a | 1.00
(1.00-1.00)a | 0.035 |
Sinusoidal dilatation also differed significantly
among groups (P=0.022), being more prominent in the DM/SEP group
compared with the S group (P=0.002) and significantly lower in the
FUL/C-60 group compared with the DM/SEP group (P=0.013) (Table I; Fig.
1, Fig. 2, Fig. 3 and Fig. 4).
The presence of pyknotic cells differed
significantly among groups (P=0.044). Pyknotic cells were more
commonly observed in the DM/SEP group compared with the S and D
groups (P=0.003 and P=0.037, respectively), whereas pyknotic cells
were significantly fewer in the FUL/C-60 group compared with the
DM/SEP group (P=0.037) (Table I;
Fig. 1, Fig. 2, Fig.
3 and Fig. 4).
The number of cells undergoing necrosis differed
significantly among groups (P<0.001). More necrotic cells were
observed in the DM/SEP group compared with the S and D groups
(P<0.001 and P=0.004, respectively). Furthermore, the number of
necrotic cells was higher in the FUL/C-60 group compared with the S
group (P=0.012), but significantly lower in the FUL/C-60 group
compared with the DM/SEP group (P=0.040) (Table I; Fig.
1, Fig. 2, Fig. 3 and Fig. 4).
Mononuclear (MN) cell infiltration in the parenchyma
was significantly different among groups (P=0.035). MN cell
infiltration was more pronounced in the DM/SEP and FUL/C-60 groups
compared with the S group (P=0.004 and P=0.040, respectively)
(Table I; Fig. 1, Fig.
2, Fig. 3 and Fig. 4).
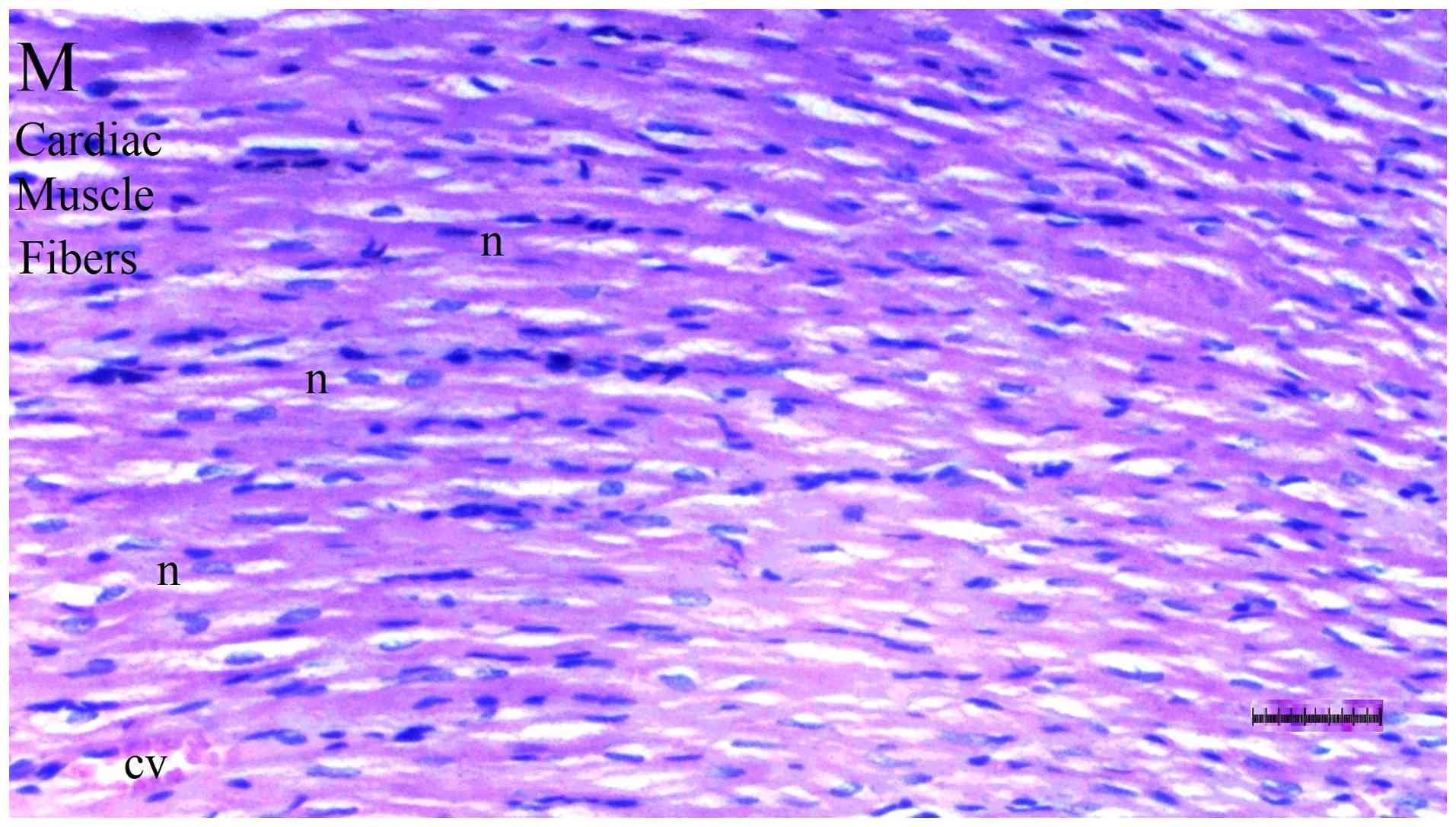
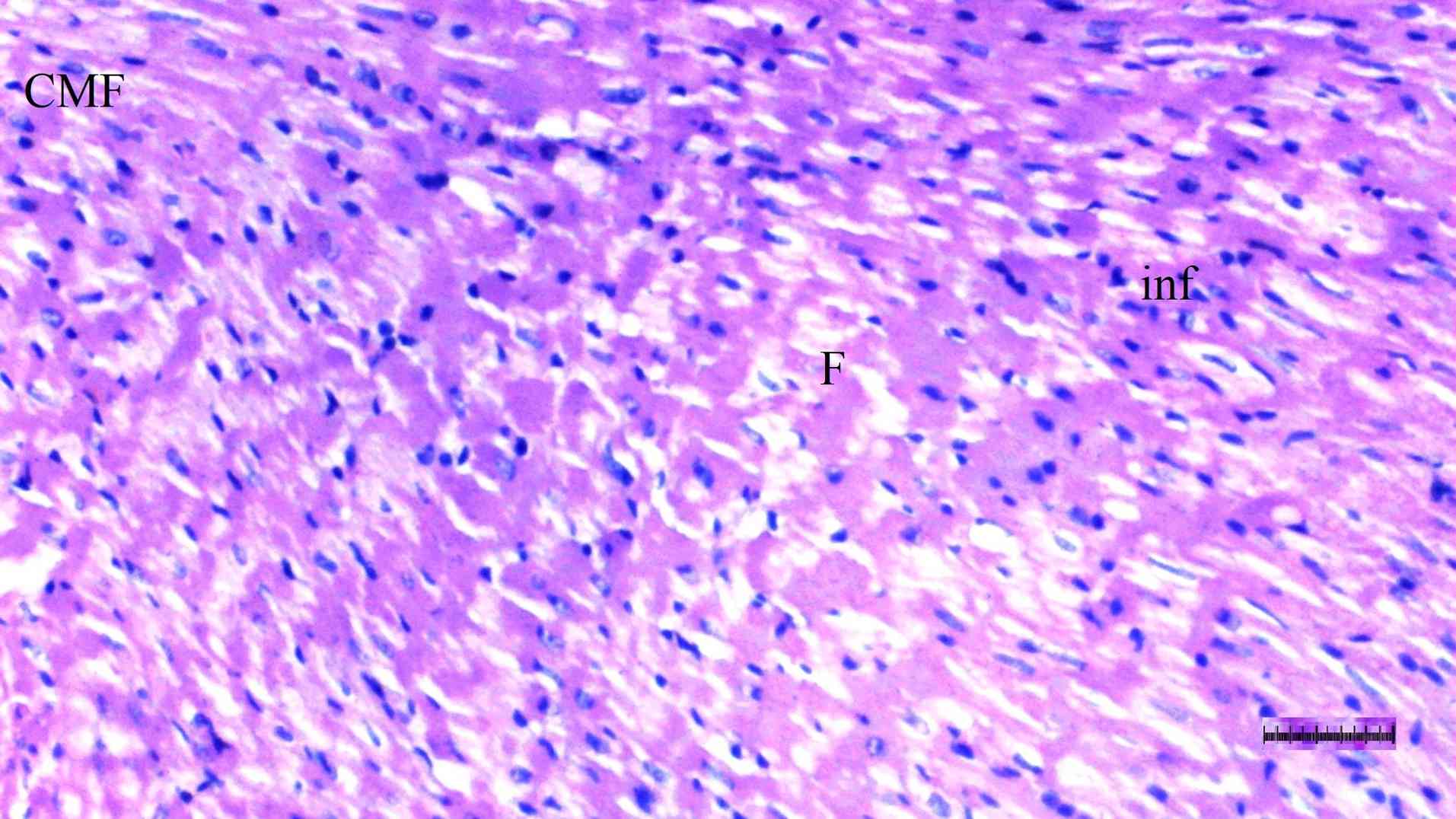
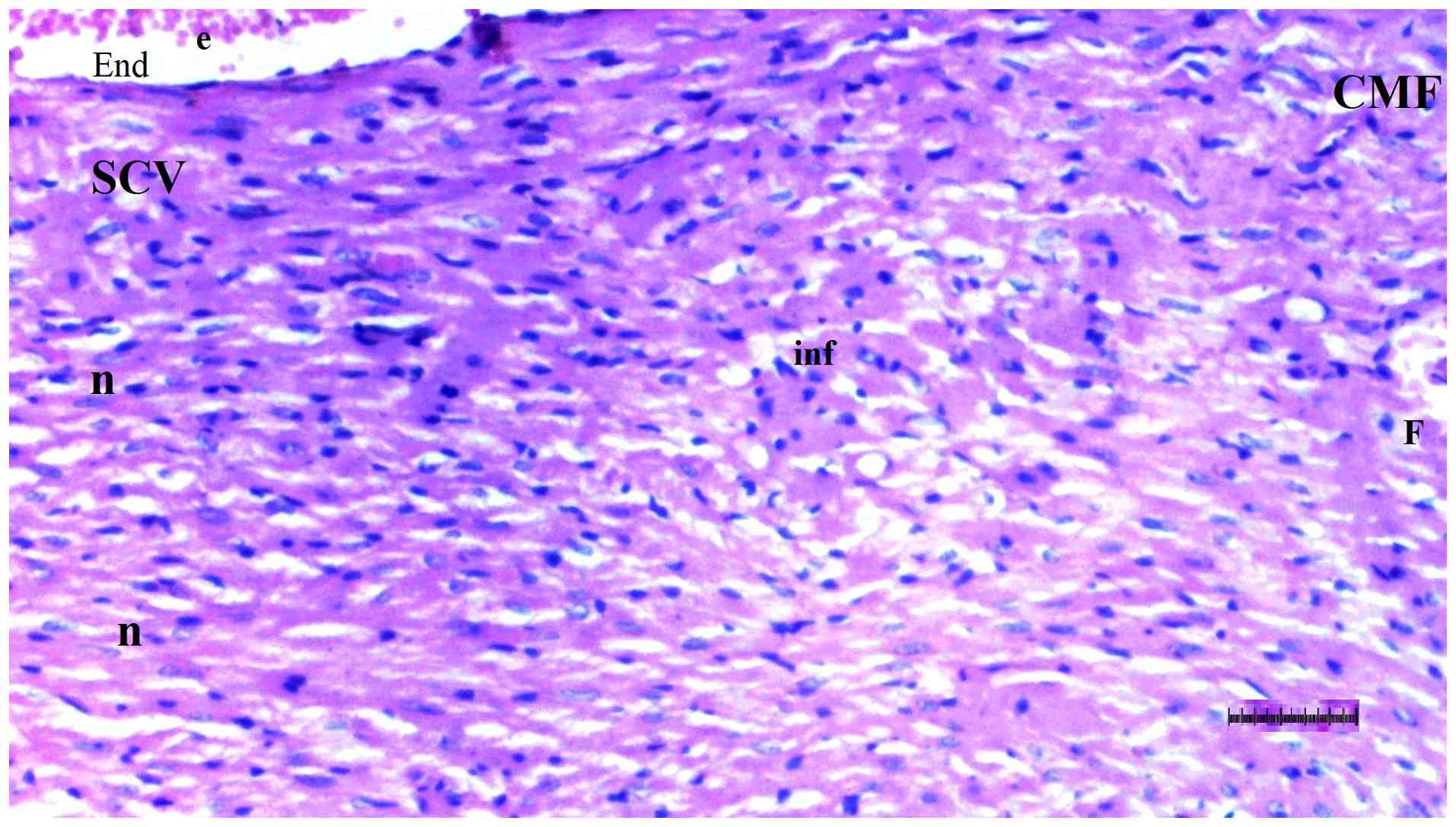
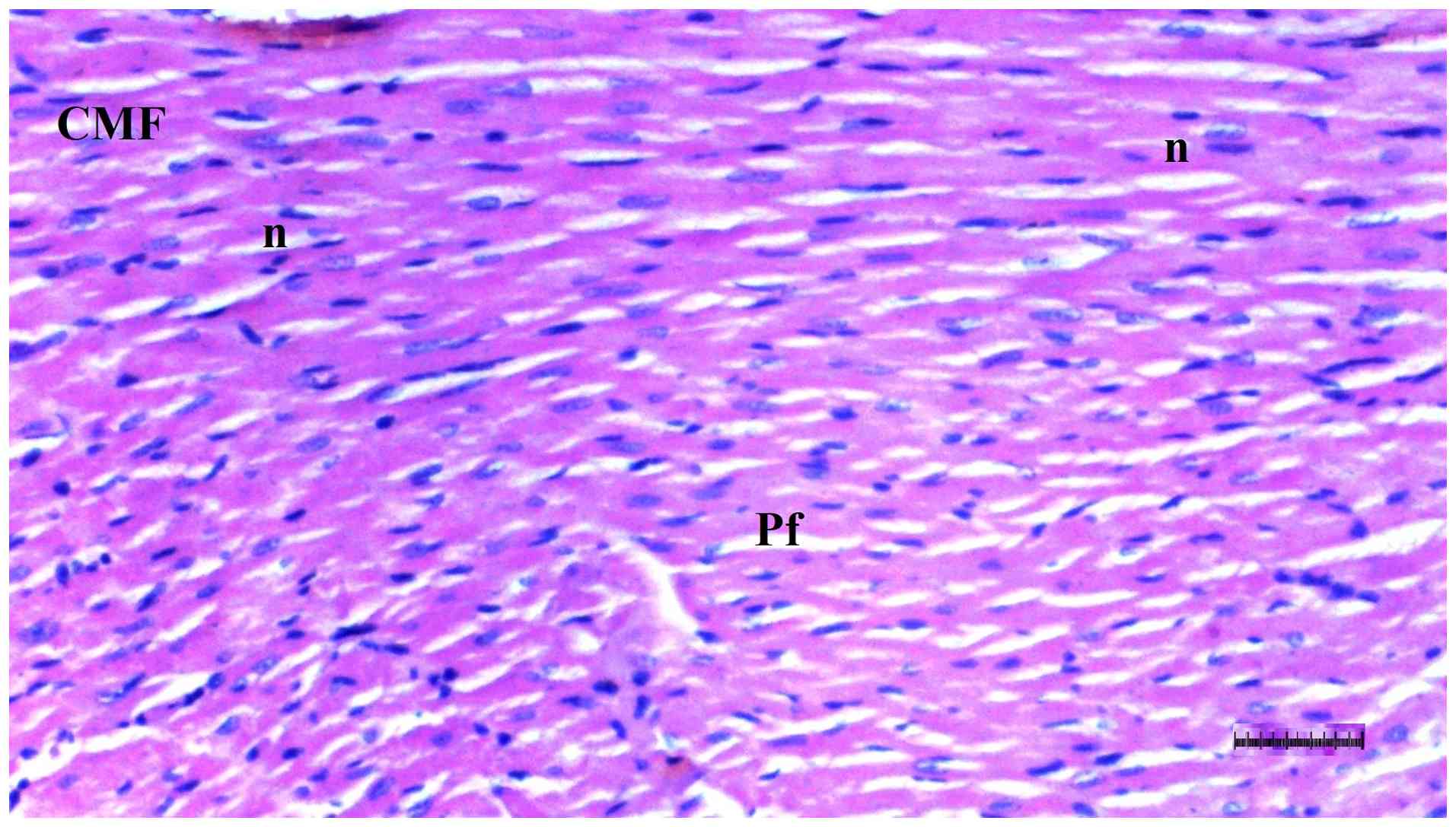
Histopathological examination of heart
tissue
Inflammation varied significantly among groups
(P=0.036). The DM/SEP group exhibited a significantly higher degree
of inflammation compared with the S group (P=0.013) (Table II; Fig. 5, Fig.
6, Fig. 7 and Fig. 8). Interstitial fibrosis differed
significantly among groups (P=0.040); it was significantly greater
in the DM/SEP group compared with the S and D groups (P=0.004 and
P=0.012, respectively); however, it was significantly reduced in
the FUL/C-60 group compared with the DM/SEP group (P=0.040)
(Table II; Fig. 5, Fig.
6, Fig. 7 and Fig. 8).
 | Table IIHistopathological findings of the S
group (n=6), D group (n=8), DM/SEP group (n=8) and FUL/C-60 group
(n=8) in heart tissue. |
Table II
Histopathological findings of the S
group (n=6), D group (n=8), DM/SEP group (n=8) and FUL/C-60 group
(n=8) in heart tissue.
| Feature | S group | D group | DM/SEP | FUL/C-60 | P-value |
|---|
| Inflammation | 0.00
(0.00-0.25) | 0.50
(0.00-1.00) | 1.00
(1.00-1.00)a | 0.50
(0.00-1.00) | 0.036 |
| Myocardial
disorganization | 0.50
(0.00-1.00) | 1.00
(0.75-1.00) | 1.00
(1.00-2.00) | 1.00
(0.75-1.00) | 0.068 |
| Interstitial
fibrosis | 0.00
(0.00-1.00) | 0.50
(0.00-1.00) | 1.00
(1.00-2.00)a,b | 0.50
(0.00-1.00)c | 0.040 |
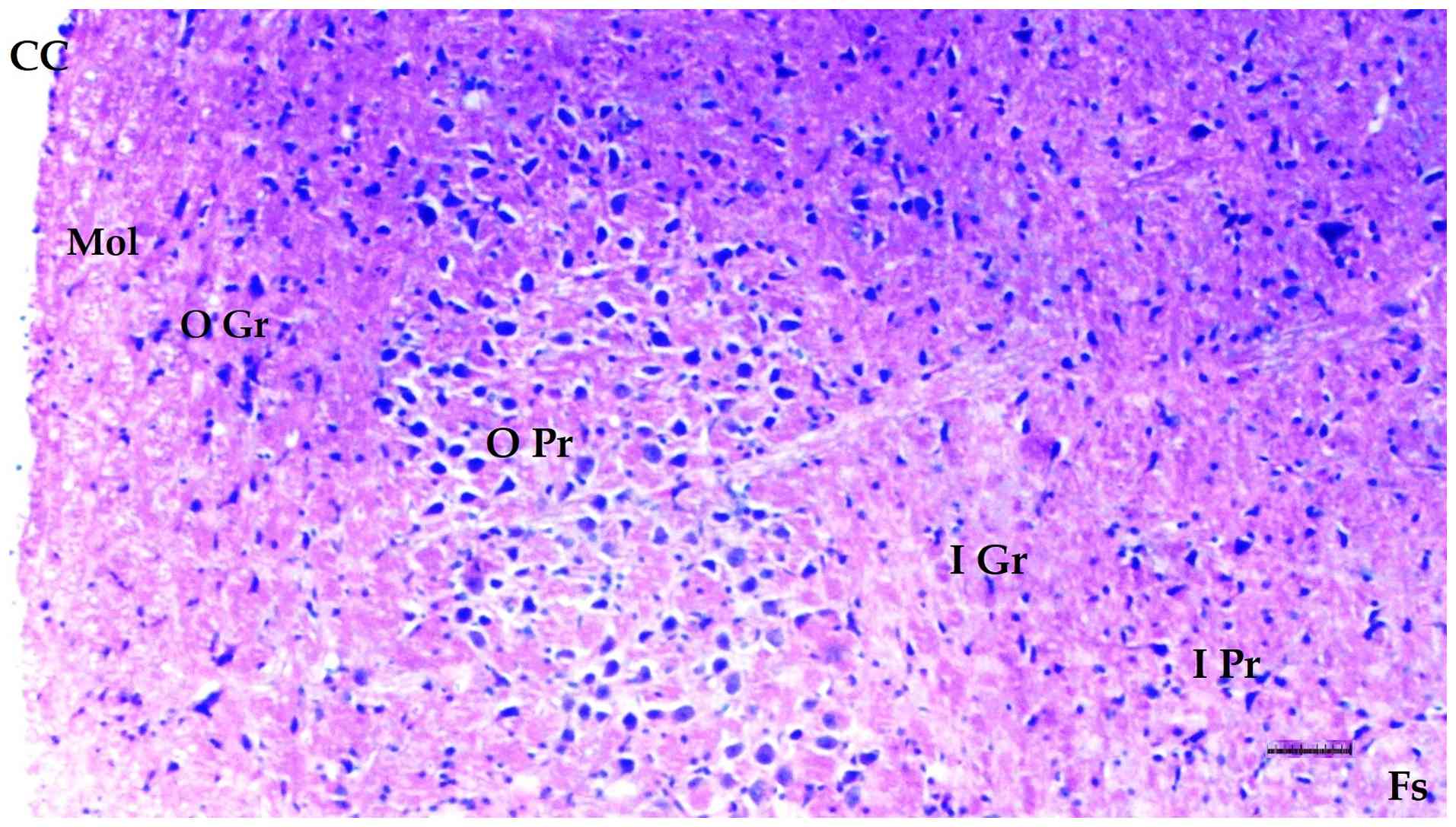
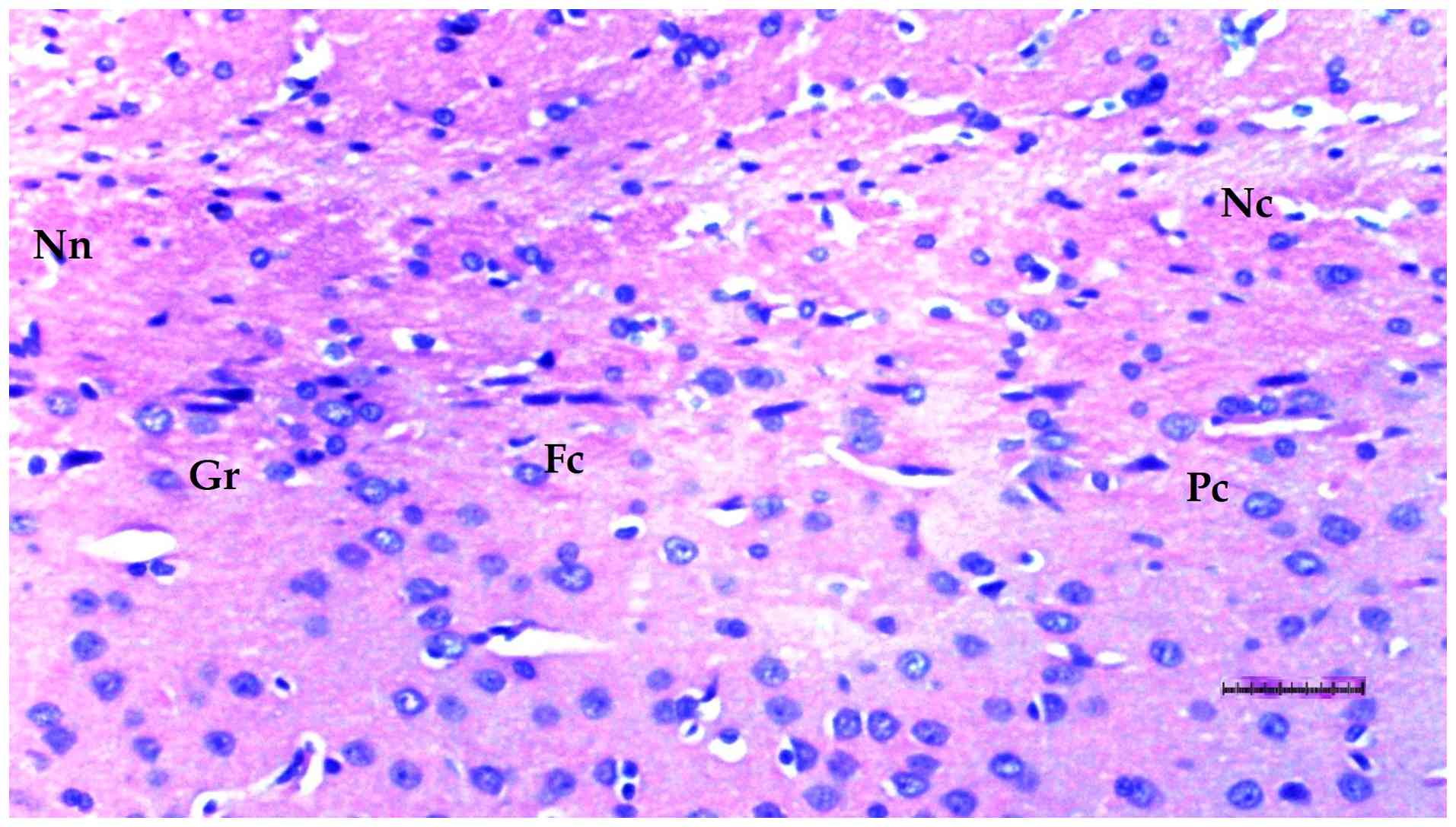
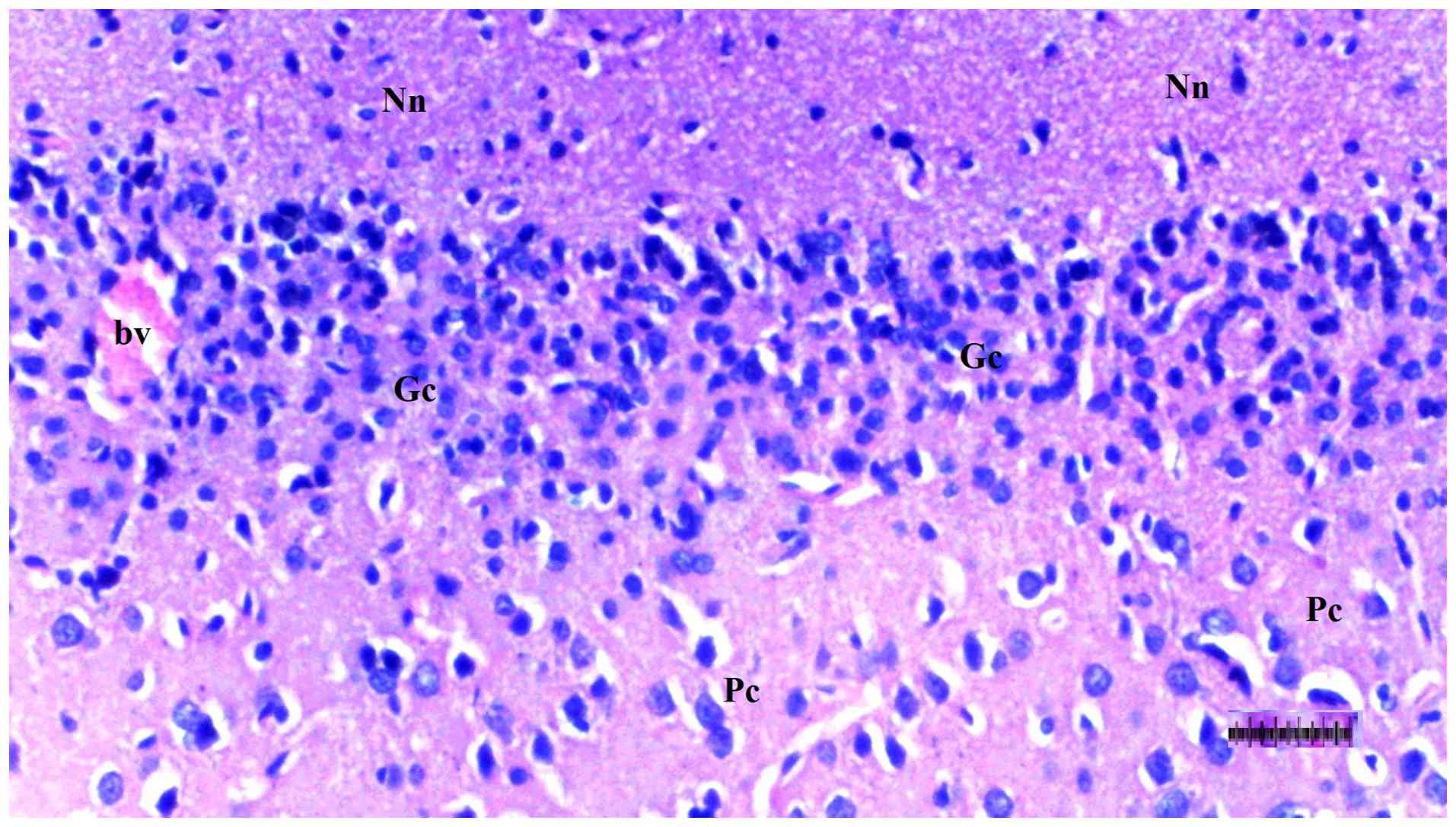
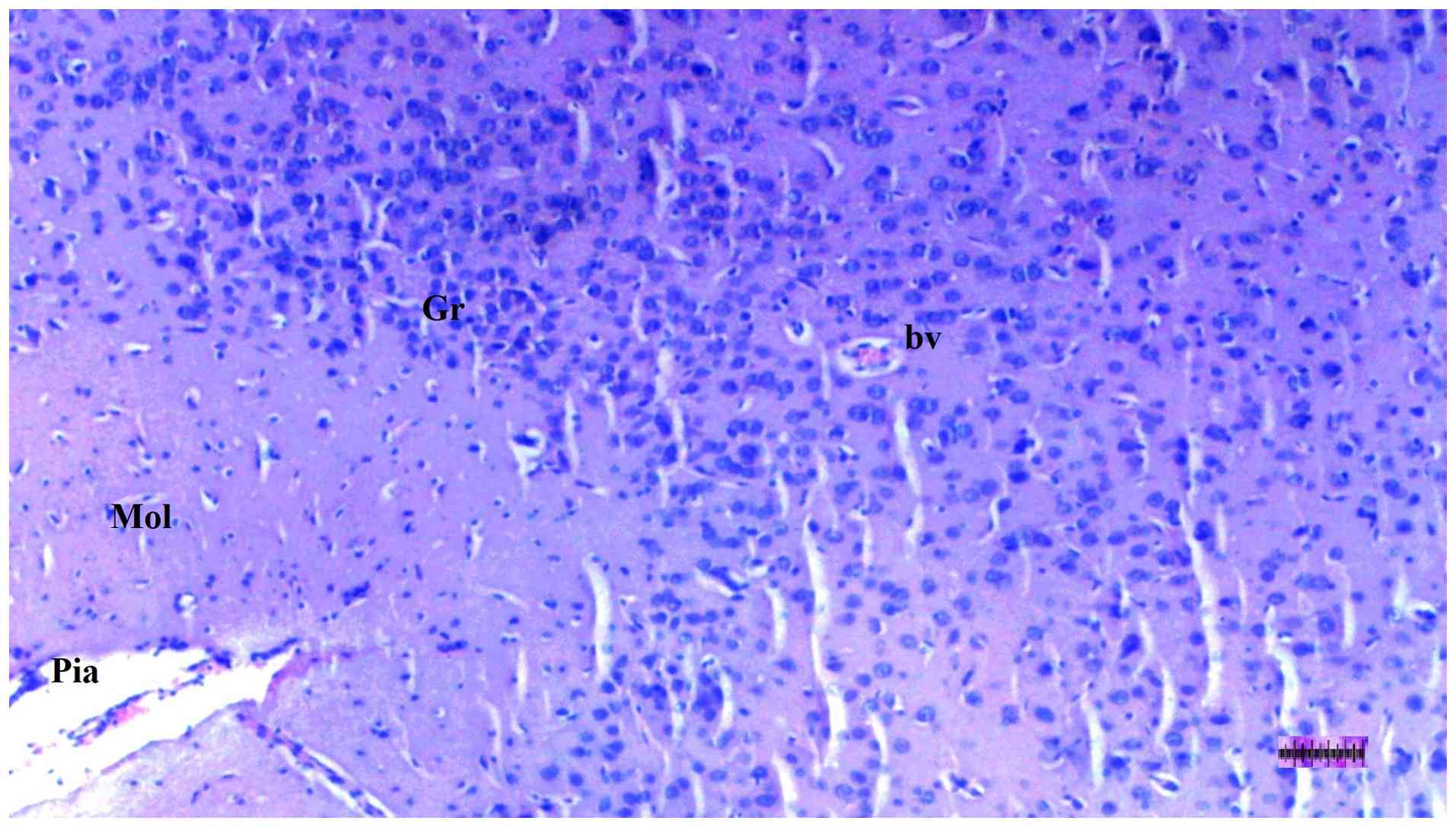
Histopathological examination of brain
tissue
An increase in cellularity (macrophages and
astrocytes) was found to be significantly different among groups
(P=0.014). The DM/SEP group showed a greater cellularity compared
with the S and D groups (P<0.001 and P=0.003, respectively),
while the FUL/C-60 group exhibited significantly lower cellularity
(macrophages and astrocytes) compared with the DM/SEP group
(P=0.034) (Table III; Fig. 9, Fig.
10, Fig. 11 and Fig. 12). Inflammation, both macrophagic
and astroglial, was significantly different among groups (P=0.036),
being significantly more pronounced in the DM/SEP group compared
with the S group (P=0.003).
 | Table IIIHistopathological findings of the S
group (n=6), D group (n=8), DM/SEP group (n=8) and FUL/C-60 group
(n=8) in in brain-cerebellar tissue. |
Table III
Histopathological findings of the S
group (n=6), D group (n=8), DM/SEP group (n=8) and FUL/C-60 group
(n=8) in in brain-cerebellar tissue.
| Feature | S group | D group | DM/SEP | FUL/C-60 | P-value |
|---|
| Ischemic neuronal
change (cortex/medulla) | 0.00
(0.00-0.25) | 0.00
(0.00-1.00) | 0.50
(0.00-1.25) | 0.00
(0.00-1.00) | 0.614 |
| Increased
cellularity (macrophages and astrocytes) | 0.00
(0.00-1.00) | 0.50
(0.00-1.00) | 1.50
(1.00-2.00)a,b | 1.00
(0.00-1.00)c | 0.014 |
| Focal necrosis | 0.00
(0.00-1.00) | 0.00
(0.00-1.00) | 0.50
(0.00-1.25) | 0.50
(0.00-1.00) | 0.819 |
| Inflammation
(macrophagic and astroglial) | 0.00
(0.00-0.25) | 0.50
(0.00-1.00) | 1.00
(1.00-1.00)a | 0.50
(0.00-1.00) | 0.036 |
Liver tissue oxidant status parameter
assessment
When liver tissue was compared in terms of TBARS
levels, a significant difference was observed among groups
(P<0.001). TBARS levels were significantly higher in all
groups compared with the S group (all P<0.001) and in the DM/SEP
and FUL/C-60 groups compared with the D group (P<0.001 and
P=0.002, respectively). These levels were significantly lower in
the FUL/C-60 group compared with the DM/SEP group (P<0.001)
(Table IV).
 | Table IVOxidative status parameters of the S
group (n=6), D group (n=8), DM/SEP group (n=8) and FUL/C-60 group
(n=8) in liver tissue. |
Table IV
Oxidative status parameters of the S
group (n=6), D group (n=8), DM/SEP group (n=8) and FUL/C-60 group
(n=8) in liver tissue.
| Parameter | S group | D group | DM/SEP | FUL/C-60 | P-value |
|---|
| TBARS, nmol/mg | 1.20±0.08 |
2.02±0.09a |
4.71±0.39a,b |
2.63±0.44a,b,c | <0.001 |
| CAT, IU/mg | 422.88±38.12 |
312.88±106.76a |
98.28±9.17a,b |
175.70±29.62a,b,c | <0.001 |
With regard to CAT enzyme activity in liver tissue,
a significant difference was observed among groups (P<0.001).
CAT enzyme activity was significantly lower in all experimental
groups compared with the S group (P=0.004, P<0.001, and
P<0.001, respectively) and in the DM/SEP and FUL/C-60 groups
compared with the D group (both P<0.001). CAT activity was
significantly higher in the FUL/C-60 group compared with the DM/SEP
group (P=0.034) (Table IV).
Brain tissue oxidant status parameter
assessment
When brain tissue was compared in terms of TBARS
levels, a significant difference was identified (P<0.001). TBARS
levels were significantly higher in all groups compared with the S
group (all, P<0.001). In addition, TBARS levels were
significantly higher in the DM/SEP group compared with the D group
(P<0.001) and significantly lower in the FUL/C-60 group compared
with the DM/SEP group (P<0.001) (Table V). With regard to CAT enzyme
activity in brain tissue, a significant difference was observed
among groups (P<0.001). Levels were significantly lower in the
DM/SEP and FUL/C-60 groups compared with the D group (both,
P<0.001) and significantly higher in the FUL/C-60 group compared
with the DM/SEP group (P=0.009; Table
V).
 | Table VOxidative status parameters of the S
group (n=6), D group (n=8), DM/SEP group (n=8) and FUL/C-60 group
(n=8) in brain tissue. |
Table V
Oxidative status parameters of the S
group (n=6), D group (n=8), DM/SEP group (n=8) and FUL/C-60 group
(n=8) in brain tissue.
| Parameter | S group | D group | DM/SEP | FUL/C-60 | P-value |
|---|
| TBARS, nmol/mg | 1.57±0.30 |
2.41±0.28a |
4.29±0.38a,b |
2.67±0.37a,b,c | <0.001 |
| CAT, IU/mg | 403.28±35.09 |
278.28±61.88a |
91.25±13.36a,b |
156.75±30.60a,b,c | <0.001 |
Serum AST, ALT, GGT, total bilirubin,
direct bilirubin and albumin results
Serum AST, ALT, total bilirubin, direct bilirubin
and albumin levels differed significantly among groups (all
P<0.001; Table VI). AST and
ALT levels were significantly elevated in the DM/SEP and FUL/C-60
groups compared with the S (all P<0.001) and D groups (all
P<0.001). Levels were significantly lower in the FUL/C-60 group
compared with the DM/SEP group (both P<0.001) (Table VI). GGT levels were significantly
higher in the DM/SEP and FUL/C-60 groups compared with the S group
(P<0.001 and P=0.004, respectively) and the D group (P<0.001
and P=0.032, respectively). Levels were significantly lower in the
FUL/C-60 group compared with the DM/SEP group (P=0.008) (Table VI). Total and direct bilirubin
levels were significantly higher in the DM/SEP and FUL/C-60 groups
compared with the S group (all P<0.001) but significantly lower
in the FUL/C-60 group compared with the DM/SEP group (both
P<0.001) (Table VI). Albumin
levels were significantly lower in the DM/SEP and FUL/C-60 groups
compared with the S group (P<0.001 and P=0.005, respectively)
and the D group (P=0.004) (Table
VI).
 | Table VISerum levels of AST, ALT, GGT,
bilirubin and albumin in the S group (n=6), D group (n=8), DM/SEP
group (n=8) and FUL/C-60 group (n=8). |
Table VI
Serum levels of AST, ALT, GGT,
bilirubin and albumin in the S group (n=6), D group (n=8), DM/SEP
group (n=8) and FUL/C-60 group (n=8).
| Parameter | S group | D group | DM/SEP | FUL/C-60 | P-value |
|---|
| AST, IU/l | 141.17±8.90 | 171.00±25.04 |
1,944.50±313.69a,b |
1,133.17±34.36a,b,c | <0.001 |
| ALT, IU/l | 65.33±13.87 | 66.83±12.92 |
305.83±55.28a,b |
210.66±44.98a,b,c | <0.001 |
| GGT, U/l | 6.50±0.55 | 7.67±1.63 |
14.50±2.88a,b |
10.67±3.01a,b,c | <0.001 |
| Total bilirubin,
mg/dl | 0.11±0.01 | 0.12±0.03 |
0.34±0.09a,b |
0.14±0.04c | <0.001 |
| Direct bilirubin,
mg/dl | 0.10±0.00 | 0.12±0.04 |
0.29±0.09a,b |
0.12±0.03c | <0.001 |
| Albumin, g/dl | 3.80±0.41 | 3.53±0.49 |
2.85±0.22a,b |
3.15±0.24a | <0.001 |
Discussion
In the present study, the histopathological and
biochemical effects of fullerenol C60 on sepsis-induced
multiple organ failure were evaluated in a diabetic rat model of
sepsis. The findings obtained through light microscopy and
biochemical analyses demonstrated that pathological alterations
triggered by sepsis were markedly increased, particularly in the
liver, heart and brain tissues and that the administration of
fullerenol C60 significantly attenuated these
changes.
A number of antioxidant agents have been
investigated in diabetic or septic models with varying degrees of
success, including melatonin. Taher et al (34) reported that high-dose melatonin (50
mg for 5 consecutive nights) administered to patients with early
septic shock was associated with a reduction in the Sequential
Organ Failure Assessment (SOFA) score (11), a commonly used measure of organ
dysfunction severity in critically ill patients, with higher scores
being associated with increased mortality. Compared with placebo,
melatonin treatment also resulted in a greater proportion of
patients achieving SOFA scores ≤6 by day 28, along with markedly
increased ventilator-free and vasopressor-free days, suggesting a
potential role for melatonin as an adjunctive therapy in septic
shock.
Rosengrave et al (35) evaluated the effects of intravenous
vitamin C (25 mg/kg every 6 h for up to 96 h) on vasopressor
requirements in 40 patients with septic shock. The study found no
significant differences between the vitamin C and placebo groups in
terms of vasopressor dose or duration, SOFA score, intensive care
unit or hospital length of stay, or mortality, and stated that
further studies are needed in this area. In their study evaluating
the effects of curcumin supplementation (1,000 mg daily for 12
weeks) on psychological status and markers of inflammation and
oxidative stress in patients with type 2 diabetes and coronary
heart disease, Shafabakhsh et al (36) observed that curcumin intake
markedly improved sleep quality and reduced oxidative damage, with
a decrease in malondialdehyde levels and an increase in total
antioxidant capacity and glutathione levels. These findings
demonstrated that curcumin provides systemic antioxidant and
anti-inflammatory benefits in patients with diabetes and
cardiovascular disease. In line with the literature, the present
results showed that fullerenol C60, reported to possess potent free
radical scavenging and antioxidant properties (37), significantly reduced TBARS levels
in liver and brain tissues and increased CAT activity. Although
oxidative stress markers (TBARS and CAT) were evaluated only in
liver and brain tissues, not in cardiac tissue, in the present
study, significant histopathological improvement was observed in
the liver, brain and heart.
Fullerenol C60, reported to possess
potent antioxidant and free radical scavenging properties (37), can readily react with radical
species through conjugated carbon-carbon double bonds, neutralizing
oxygen-free radicals and mimicking superoxide dismutase (SOD)
activity. The transcription factor nuclear factor erythroid 2
p45-associated factors 2 (Nrf2), regulates the endogenous
antioxidant response pathway by binding to antioxidant response
elements (AREs) in the promoter regions of antioxidants. Nrf2
regulates glutathione (GSH) recycling by controlling the expression
of γ-GCL, a key enzyme in GSH biosynthesis. The matrix GSH redox
cycle, coordinated with SOD-mediated clearance of superoxide anion,
is key in preventing excessive ROS accumulation in the
mitochondria. C60 has also been shown to modulate the
activity and expression of antioxidant enzymes such as SOD and GSH
through Nrf2/ARE-dependent pathways (37).
Sepsis is a major contributor to multiple organ
failure, particularly through structural and functional impairments
in the liver. Systemic inflammatory response-induced hypoperfusion
and impaired oxygenation result in mitochondrial dysfunction and
cellular injury in hepatocytes (38). This process increases ROS, which,
in turn, leads to an increase in oxidative stress. Increased
oxidative stress leads to lipid peroxidation in hepatocyte
membranes, accelerating the hepatocyte injury and cell death. These
alterations impair hepatic detoxification and bile excretory
function, potentially leading to cholestasis and elevated bilirubin
levels (38,39).
Furthermore, under the influence of pro-inflammatory
cytokines, such as TNF-α, interleukin (IL)-1β and IL-6, hepatic
microcirculation is disrupted, leading to a marked increase in
hepatic enzyme levels. Consequently, sepsis-related liver
dysfunction exacerbates disease severity and worsens prognosis
(39). In addition, the systemic
inflammatory response may induce notable dysfunction in the
cardiovascular system. Increased vasodilation and capillary
permeability lead to hypotension and impaired tissue perfusion.
Mitochondrial dysfunction and cellular damage in cardiomyocytes,
driven by inflammatory mediators, result in myocardial depression
and reduced cardiac output. In addition, endothelial dysfunction
and coagulopathy impair microcirculation by increasing vascular
permeability and promoting microthrombus formation, thereby
reducing capillary blood flow and leading to tissue hypoperfusion.
These mechanisms form the basis of the high mortality rates
associated with cardiovascular complications in sepsis (40).
Furthermore, sepsis can affect the central nervous
system and lead to the development of sepsis-associated
encephalopathy, a marked neurological complication that contributes
to increased morbidity and mortality. Systemic inflammatory
response and cerebral hypoperfusion induce metabolic stress in
neurons, characterized by mitochondrial dysfunction and reduced ATP
levels. During this pathophysiological process, excessive ROS
production triggers oxidative damage, leading to lipid
peroxidation, protein denaturation and disruption of DNA integrity.
Furthermore, increased blood-brain barrier permeability facilitates
the passage of peripheral pro-inflammatory cytokines, such as
TNF-α, interleukin (IL)-1β and IL-6, into the brain parenchyma,
enhancing glial activation and amplifying the neuroinflammatory
response (15). Consequently,
neuronal dysfunction and cellular injury provide the groundwork for
a clinical setting characterized by cognitive impairment, confusion
and decreased levels of consciousness.
In the present study, histopathological findings in
line with those reported by Elshater et al (41) were observed. This study
investigated the effects of fullerenes on the liver in rats. In
particular, the notably increased level of hydropic degeneration,
sinusoidal dilatation, pyknotic cells and necrotic cells in the
DM/SEP group reflected the extent of liver damage induced by sepsis
in diabetic rats. Sepsis-induced oxidative stress leads to the
generation of free radicals that disrupt cellular functions and
contribute to cell death. The reduction of these pathological
changes in the FUL/C-60 group highlights the ability of fullerenol
C60 to counteract free radical-induced cellular damage
through its antioxidant properties.
The effect of fullerenol C60 on hepatic
oxidative stress was also evident in the present biochemical
analyses. TBARS levels, an indicator of lipid peroxidation and
oxidative stress, are of particular importance. In the present
study, findings similar to those in previous work by Kubat et
al (29) were observed, a
study in which the impact of fullerenol C60 on TBARS
levels in kidney and lung tissues was reported. Similarly, Yavuz
et al (28) demonstrated
marked changes in TBARS levels associated with the effect of
fullerenol C60 on the liver. In the present study, the
significantly elevated TBARS levels in the DM/SEP group reflected
the oxidative stress caused by sepsis, while the reduction of this
parameter in the FUL/C-60 group suggested that fullerenol
C60 mitigated lipid peroxidation and limited oxidative
tissue damage. Furthermore, the increase in CAT enzyme activity
indicated the potential of fullerenol C60 to support
endogenous antioxidant defense mechanisms. This finding implied
that fullerenol C60 modulated endogenous antioxidant
enzyme activity and reduced the impact of free radicals.
As previously reported (41-46),
the beneficial effects of fullerenes on the liver are well
established. In the present study, significantly elevated levels of
liver injury markers such as AST, ALT, GGT and bilirubin in the
DM/SEP group indicated hepatic damage and leakage of intracellular
enzymes into serum due to sepsis. However, the reduction in these
enzyme levels in the FUL/C-60 group suggested that fullerenol
C60 may exert hepatoprotective effects by maintaining
hepatocyte stability and limiting enzyme release, findings that
align with previous studies (41-46).
This also indicates the potential anti-inflammatory properties of
fullerenol C60, as suppression of sepsis-related
inflammatory processes is key in preventing multiple organ
failure.
Sepsis not only affects the liver but also
negatively impacts cardiac tissue. In the present study, cardiac
oxidative stress markers (such as TBARS and CAT) and inflammatory
mediators were not assessed in heart tissue. However,
histopathological findings showed that myocardial disorganization
and interstitial fibrosis were more pronounced in the DM/SEP group
compared with the other groups, indicating the detrimental cardiac
effects of sepsis in patients with diabetic. Sepsis triggers a
complex interplay of immune activation, oxidative stress,
mitochondrial failure and cardiomyocyte apoptosis, resulting in
myocardial disorganization and disruption of cardiomyocyte
architecture and functional impairment. By contrast, interstitial
fibrosis stiffens the tissue, thereby compromising cardiac
performance (47). Similar to the
findings of Injac et al (42), the present study showed a decrease
in pathological changes in heart tissue in the FUL/C-60 group,
suggesting that fullerenol C60 exhibited
cardioprotective effects and alleviated sepsis-induced
cardiotoxicity. The antioxidant and anti-inflammatory actions of
fullerenol C60 may represent the underlying mechanisms
of this protective effect.
Carbon-based antioxidants with neuroprotective and
neuroregenerative properties may help limit the effects of sepsis
on the brain (48,49). In parallel with our previous study,
which showed the effects of fullerenol C60 on kidney and
lung tissues (29), the changes
observed in the levels of TBARS and CAT enzymes, indicators of
oxidative stress processes in brain tissue and histopathological
findings also support the neuroprotective effect of fullerenol
C60. The elevated TBARS level observed in the brain
tissue of the DM/SEP group indicated that oxidative stress
triggered neurodegenerative processes. However, the reduction in
TBARS levels in the FUL/C-60 group suggested that fullerenol
C60 attenuated free radical-induced damage. In addition,
the increase in CAT enzyme activity indicated the potential of
fullerenol C60 to support antioxidant defense mechanisms
in the brain.
Based on the findings of the present study,
fullerenol C60 may be considered a potential
prophylactic agent for preventing sepsis-induced organ tissue
damage in diabetic rats. Given its ability to suppress
sepsis-induced oxidative stress and inflammation, thereby reducing
multiple organ failure, further experimental research is needed to
determine its therapeutic efficacy and safety, supporting the
clinical application of fullerenol C60.
However, the present study exhibits certain
limitations. Markers of oxidative stress in the heart, systemic
inflammatory markers (including cytokines) and the molecular
mechanisms of action of fullerenol C60 were not studied.
In the present study, based on previous research, fullerenol
C60 was administered as a single dose. Samples were
taken from rats 24 h after laparotomy, therefore the long-term
effects of fullerenol C60 were not studied. Further
research is required to understand the effects and mechanisms of
action on dose-dependent response. Another limitation of the
present study is the absence of a non-diabetic sepsis (CLP-only)
group. Inclusion of such a group would have allowed more precise
differentiation between the effects of diabetes and sepsis on organ
injury and oxidative stress parameters. Due to ethical restrictions
on the maximum number of animals permitted, the present
experimental design focused primarily on the diabetic sepsis model.
Future studies incorporating both diabetic and non-diabetic sepsis
groups are warranted to further clarify the interaction between
diabetes and sepsis in the context of fullerenol C60
treatment.
In conclusion, histopathologically, the present
study found that fullerenol C60 reduced oxidative stress
and inflammation in a streptozotocin-induced diabetic rat sepsis
model, thereby reducing sepsis-induced damage in liver, heart and
brain tissues. The structural and biochemical improvements observed
in liver, heart and brain tissues in the present study suggested
that fullerenol C60, with its antioxidant and
anti-inflammatory properties, may be beneficial in the preventive
treatment of multiple organ failure in sepsis. The precise
molecular mechanisms underlying these protective effects of
fullerenol C60 were not directly investigated in the
present study. Future studies incorporating molecular analyses such
as the evaluation of Nrf2/ARE pathway activation, NF-κB signaling
modulation, mitochondrial function markers and apoptosis-related
proteins will therefore contribute to an improved understanding of
the safety and therapeutic efficacy fullerenol C60 and
its organ-protective effects in diabetic sepsis, as well as provide
a promising basis for new approaches to sepsis treatment in the
diabetic population.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
MAr, ÖK, KD, NŞ, LF and HB conceived and designed
the study, and contributed to data analysis and interpretation. ÖK,
HB and KD performed the experimental procedures. MK, FE, MAt and ŞS
conducted the biochemical and histopathological analyses. ÖK and FE
collected the samples. AB and IG contributed to data analysis and
interpretation, and participated in the evaluation of the findings.
AB, IG, KD, ÖK and MK confirm the authenticity of all the raw data.
ÖK, MK, NŞ, LF, AB, IG, MAr, and ŞS critically revised the
manuscript for important intellectual content. All authors have
read and approved the final version of the manuscript, and agree to
be accountable for all aspects of the work.
Ethics approval and consent to
participate
Ethical approval for the present study was obtained
from The Animal Research Committee of Gazi University (Ankara,
Turkey; approval no. G.Ü.ET-24-046).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Afreen S, Muthoosamy K, Manickam S and
Hashim U: Functionalized fullerene (C60) as a potential
nanomediator in the fabrication of highly sensitive biosensors.
Biosens Bioelectron. 63:354–364. 2015.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Díez-Pascual AM: Carbon-based
nanomaterials. Int J Mol Sci. 22(7726)2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Bakry R, Vallant RM, Najam-ul-Haq M,
Rainer M, Szabo Z, Huck CW and Bonn GK: Medicinal applications of
fullerenes. Int J Nanomedicine. 2:639–649. 2007.PubMed/NCBI
|
|
4
|
Anilkumar P, Lu F, Cao L, Luo PG, Liu JH,
Sahu S, Tackett KN, Wang Y and Sun YP: Fullerenes for applications
in biology and medicine. Curr Med Chem. 18:2045–2059.
2011.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Bogdanović G and Djordjević A: Carbon
nanomaterials: Biologically active fullerene derivatives. Srp Arh
Celok Lek. 144:222–231. 2016.PubMed/NCBI
|
|
6
|
Nakamura S and Mashino T: Water-soluble
fullerene derivatives for drug discovery. J Nippon Med Sch.
79:248–254. 2012.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Biswas R, Batista Da Rocha C, Bennick RA
and Zhang J: Water-soluble fullerene monoderivatives for biomedical
applications. Chem Med Chem. 18(e202300296)2023.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Dejager L, Pinheiro I, Dejonckheere E and
Libert C: Cecal ligation and puncture: The gold standard model for
polymicrobial sepsis? Trends Microbiol. 19:198–208. 2011.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Drechsler S and Osuchowski M: Cecal
ligation and puncture. Methods Mol Biol. 2321:1–8. 2021.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Cecconi M, Evans L, Levy M and Rhodes A:
sepsis and septic shock. Lancet. 392:75–87. 2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Singer M, Deutschman CS, Seymour CW,
Shankar-Hari M, Annane D, Bauer M, Bellomo R, Bernard GR, Chiche
JD, Coopersmith CM, et al: The third international consensus
definitions for sepsis and septic shock (Sepsis-3). JAMA.
315:801–810. 2016.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Krzyżaniak K, Szymczyk A, Wściślak A,
Szczygieł N, Zembrzuska S, Abramczyk-Suszek A, Krion R, Remisiewicz
M and Siemiński M: Prevalence, symptoms, risk factors and impact of
sepsis-associated encephalopathy in emergency department patients:
A case-control study. BMC Emerg Med. 25(262)2025.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Gauer R, Forbes D and Boyer N: Sepsis:
Diagnosis and management. Am Fam Physician. 101:409–418.
2020.PubMed/NCBI
|
|
14
|
Baddam S and Burns B: Systemic
inflammatory response syndrome. StatPearls [Internet] Treasure
Island (FL): StatPearls Publishing, 2025.
|
|
15
|
Pan S, Lv Z, Wang R, Shu H, Yuan S, Yu Y
and Shang Y: Sepsis-induced brain dysfunction: Pathogenesis,
diagnosis, and treatment. Oxid Med Cell Longev.
2022(1328729)2022.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Beyer D, Hoff J, Sommerfeld O, Zipprich A,
Gaßler N and Press AT: The liver in sepsis: Molecular mechanism of
liver failure and their potential for clinical translation. Mol
Med. 28(84)2022.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Trevelin SC, Carlos D, Beretta M, da Silva
JS and Cunha FQ: Diabetes mellitus and sepsis: A challenging
association. Shock. 47:276–287. 2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Guariguata L, Whiting DR, Hambleton I,
Beagley J, Linnenkamp U and Shaw JE: Global estimates of diabetes
prevalence for 2013 and projections for 2035. Diabetes Res Clin
Pract. 103:137–149. 2014.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Dilworth L, Stennett D, Facey A, Omoruyi
F, Mohansingh S and Omoruyi FO: Diabetes and the associated
complications: The role of antioxidants in diabetes therapy and
care. Biomed Pharmacother. 181(117641)2024.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Darenskaya MA, Kolesnikova LI and
Kolesnikov SI: Oxidative stress: pathogenetic role in diabetes
mellitus and ıts complications and therapeutic approaches to
correction. Bull Exp Biol Med. 171:179–189. 2021.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Jiang H, Xia C, Lin J, Garalleh HA,
Alalawi A and Pugazhendhi A: Carbon nanomaterials: A growing tool
for the diagnosis and treatment of diabetes mellitus. Environ Res.
221(115250)2023.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Pham L, Arroum T, Wan J, Pavelich L, Bell
J, Morse PT, Lee I, Grossman LI, Sanderson TH, Malek MH and
Hüttemann M: Regulation of mitochondrial oxidative phosphorylation
through tight control of cytochrome c oxidase in health and disease
- Implications for ischemia/reperfusion injury, inflammatory
diseases, diabetes, and cancer. Redox Biol.
78(103426)2024.PubMed/NCBI View Article : Google Scholar
|
|
23
|
de Souza Stork S, Hübner M, Biehl E,
Danielski LG, Bonfante S, Joaquim L, Denicol T, Cidreira T, Pacheco
A, Bagio E, et al: Diabetes exacerbates sepsis-ınduced
neuroinflammation and brain mitochondrial dysfunction.
Inflammation. 45:2352–2367. 2022.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Üstündağ H, Doğanay S, Kalındemirtaş FD,
Demir Ö, Huyut MT, Kurt N, Özgeriş FB and Akbaba Ö: A new treatment
approach: Melatonin and ascorbic acid synergy shields against
sepsis-induced heart and kidney damage in male rats. Life Sci.
329(121875)2023.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Shang Y, Hewage SM, Wijerathne CUB, Siow
YL, Isaak CK and Karmin O: Kidney ischemia-reperfusion elicits
acute liver injury and inflammatory response. Front Med.
7(201)2020.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Polat Y, Şengel N, Küçük A, Özdemir Ç,
Yığman Z, Balcı AB, Ergörün Aİ, Kavutçu M and Arslan M: Effects of
sevoflurane and fullerenol C60 on lower limb ischemia-reperfusion
injury in streptozocin-induced diabetic mice. Sci Prog.
107(368504241239444)2024.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Örnek E, Alkan M, Erel S, Yığman Z, Dursun
AD, Dağlı A, Sarıkaya B, Kip G, Polat Y and Arslan M: Effects of
sevoflurane and fullerenol C60 on the heart and lung in
lower-extremity ıschemia-reperfusion ınjury in
streptozotocin-ınduced diabetes mice. Medicina (Kaunas).
60(1232)2024.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Yavuz A, Tuna AT, Ozdemir C, Mortas T,
Küçük A, Kasapbaşı E, Arslan M, Kavutçu M and Kurtipek Ö: Effects
of fullerene C60 on liver tissue in liver ischemia reperfusion
injury in rats undergoing sevoflurane anesthesia. Libyan J Med.
18(2281116)2023.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Kubat Ö, Dіkmen K, Bostanci H, Arslan M,
Er F, Kavutçu M, Sezen ŞC and Atli M: Investigating the protective
effects of fullerenol-C60 on kidney and lung tissues in
streptozotocin-induced diabetic rats with cecal sepsis. Sci Rep.
15(19096)2025.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Percie du Sert N, Hurst V, Ahluwalia A,
Alam S, Avey MT, Baker M, Browne WJ, Clark A, Cuthill IC, Dirnagl
U, et al: The ARRIVE guidelines 2.0: Updated guidelines for
reporting animal research. Br J Pharmacol. 177:3617–3624.
2020.PubMed/NCBI View Article : Google Scholar
|
|
31
|
National Research Council Committee for
the Update of the Guide for the, C. and A. Use of Laboratory, The
National Academies Collection: Reports funded by National
Institutes of Health, in Guide for the Care and Use of Laboratory
Animals. 2011, National Academies Press (US) Copyright ©. 2011,
National Academy of Sciences.: Washington (DC).
|
|
32
|
Arslan M, Poyraz F, Kiraz HA, Alkan M, Kip
G, Erdem OA, Ozer A, Şıvgın V and Comu FM: The effect of
dexmedetomidine on myocardial ischemia reperfusion injury in
streptozotocin induced diabetic rats. Anaesth Pain İntensive Care.
19:444–451. 2015.
|
|
33
|
Özer A, Şengel N, Küçük A, Yığman Z,
Özdemir Ç, Kılıç Y, Dursun AD, Bostancı H, Kip G and Arslan M: The
effect of cerium oxide (CeO2) on ıschemia-reperfusion ınjury in
skeletal muscle in mice with streptozocin-ınduced diabetes.
Medicina (Kaunas). 60(752)2024.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Taher A, Shokoohmand F, Abdoli E,
Mohammadi Y and Mehrpooya M: A pilot study on the melatonin
treatment in patients with early septic shock: Results of a
single-center randomized controlled trial. Ir J Med Sci.
191:1913–1924. 2022.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Rosengrave P, Spencer E, Williman J,
Mehrtens J, Morgan S, Doyle T, Van Der Heyden K, Morris A, Shaw G
and Carr AC: Intravenous vitamin C administration to patients with
septic shock: A pilot randomised controlled trial. Crit Care.
26(26)2022.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Shafabakhsh R, Mobini M, Raygan F,
Aghadavod E, Ostadmohammadi V, Amirani E, Mansournia MA and Asemi
Z: Curcumin administration and the effects on psychological status
and markers of inflammation and oxidative damage in patients with
type 2 diabetes and coronary heart disease. Clin Nutr ESPEN.
40:77–82. 2020.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Gonchar OO, Maznychenko AV, Klyuchko OM,
Mankovska IM, Butowska K, Borowik A, Piosik J and Sokolowska I: C60
fullerene reduces 3-nitropropionic acid-ınduced oxidative stress
disorders and mitochondrial dysfunction in rats by modulation of
p53, Bcl-2 and Nrf2 targeted proteins. Int J Mol Sci.
22(5444)2021.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Kasper P, Tacke F, Steffen HM and Michels
G: Hepatic dysfunction in sepsis. Med Klin Intensivmed Notfmed.
115:609–619. 2020.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Sun J, Zhang J, Wang X, Ji F, Ronco C,
Tian J and Yin Y: Gut-liver crosstalk in sepsis-induced liver
injury. Crit Care. 24(614)2020.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Merx MW and Weber C: Sepsis and the heart.
Circulation. 116:793–802. 2007.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Elshater AA, Haridy MAM, Salman MMA,
Fayyad AS and Hammad S: Fullerene C60 nanoparticles ameliorated
cyclophosphamide-induced acute hepatotoxicity in rats. Biomed
Pharmacother. 97:53–59. 2018.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Injac R, Perse M, Cerne M, Potocnik N,
Radic N, Govedarica B, Djordjevic A, Cerar A and Strukelj B:
Protective effects of fullerenol C60(OH)24 against
doxorubicin-induced cardiotoxicity and hepatotoxicity in rats with
colorectal cancer. Biomaterials. 30:1184–1196. 2009.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Kuznietsova H, Dziubenko N, Herheliuk T,
Prylutskyy Y, Tauscher E, Ritter U and Scharff P: Water-soluble
pristine C60 fullerene ınhibits liver alterations associated with
hepatocellular carcinoma in rat. Pharmaceutics.
12(794)2020.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Gharbi N, Pressac M, Hadchouel M, Szwarc
H, Wilson SR and Moussa F: [60]Fullerene is a powerful antioxidant
in vivo with no acute or subacute toxicity. Nano Letters.
5:2578–2585. 2005.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Injac R, Perse M, Obermajer N,
Djordjevic-Milic V, Prijatelj M, Djordjevic A, Cerar A and Strukelj
B: Potential hepatoprotective effects of fullerenol C60(OH)24 in
doxorubicin-induced hepatotoxicity in rats with mammary carcinomas.
Biomaterials. 29:3451–3460. 2008.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Kuznietsova H, Lynchak O, Dziubenko N,
Herheliuk T, Prylutskyy Y, Rybalchenko V and Ritter U:
Water-soluble pristine C60 fullerene attenuates
acetaminophen-induced liver injury. Bioimpacts. 9:227–237.
2019.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Lukić I, Mihić D, Varžić SC, Relatić KS,
Zibar L, Loinjak D, Ćurić ŽB, Klobučar L and Maričić L: Septic
cardiomyopathy. Rev Cardiovasc Med. 25(23)2024.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Henna TK, Raphey VR, Sankar R, Ameena
Shirin VK, Gangadharappa HV and Pramod K: Carbon nanostructures:
The drug and the delivery system for brain disorders. Int J Pharm.
587(119701)2020.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Bolshakova OI, Borisenkova AA, Golomidov
IM, Komissarov AE, Slobodina AD, Ryabova EV, Ryabokon IS, Latypova
EM, Slepneva EE and Sarantseva SV and Sarantseva SV: Fullerenols
prevent neuron death and reduce oxidative stress in drosophila
huntington's disease model. Cells. 12(170)2022.PubMed/NCBI View Article : Google Scholar
|