1. Introduction
Due to the outbreak of the 2019 coronavirus disease
(COVID-19) pandemic and its accompanying public health measures,
which were implemented to curb the rapid spread of the virus, daily
life across all age groups has been markedly disrupted. From the
outset, the COVID-19 pandemic was recognized as a potential threat
to both physical and mental health (1-3),
particularly among vulnerable populations, including young children
and their families (4,5).
Early childhood (0-5 years) is a period of rapid
brain development and heightened sensitivity to environmental
conditions. During this developmental period, stable routines,
responsive caregiving and access to early learning experiences
support the emergence of socio-emotional skills, language,
executive functions and foundational neural circuits (6). The COVID-19 pandemic disrupted these
developmental supports through lockdowns, social-distancing
measures and widespread closures of early childhood education and
care settings (7). These
disruptions introduce unique challenges, especially for young
children, whose developmental growth depends on consistent
routines, rich social interactions (such as responsive
caregiver-child exchanges, peer play and opportunities for shared
attention), and core educational experiences (including
language-rich activities, guided play, early numeracy and literacy
exposure, and structured opportunities for exploration and
problem-solving) (8-10).
Although multiple studies (11-13)
are examining the developmental consequences of the pandemic,
previous reviews (14,15) provide fragmented overviews. These
reviews typically focus on single domains such as socio-emotional
or behavioral outcomes (11),
language development (12) or
executive function (13),
combining preschoolers with older children in their analyses
(14,15), or summarizing findings from the
early phase of the pandemic (1,4,7,8). As
a result, integrated evidence describing how pandemic-related
disruptions affect multiple developmental systems specifically in
children aged 0-5 years, whose developmental needs and
vulnerabilities differ markedly from those of school-aged children,
is yet to be elucidated.
The present narrative review aimed to address the
aforementioned gaps by summarizing empirical studies published from
2020-2025 across three interconnected domains: i) Socio-emotional
and behavioral development; ii) cognitive, language and
executive-function (EF) outcomes; and iii) biological and
physiological markers of stress. By examining these domains
together, the present study aimed to clarify the converging and
diverging patterns across studies, identify contextual factors that
affected the responses of children to pandemic-related stressors,
and highlight the methodological limitations that constrain
interpretation. Therefore, the present integrated,
early-childhood-specific review provided an understanding of how
pandemic disruptions influenced developmental pathways and where
evidence remains preliminary or inconsistent.
2. Methods
PubMed (https://pubmed.ncbi.nlm.nih.gov/), Embase (https://www.embase.com/), PsycINFO (https://www.apa.org/pubs/databases/psycinfo) and Web
of Science (https://www.webofscience.com/) were searched for
empirical studies published between January 2020 and June 2025 (the
full search strings are outlined in Table SI). Empirical studies
(cross-sectional, cohort and longitudinal), systematic reviews and
meta-analyses reporting data for children aged 0-5 years (or
separate analyses for this age group) were included. Eligible
studies examined pandemic-related restrictions (such as lockdowns
and early childhood education and care settings) and used validated
outcome measures in socio-emotional, cognitive/language or
biological domains. Non-empirical commentary, studies that combined
preschoolers with older children without separate reporting and
studies that lacked validated outcomes were excluded. Only
peer-reviewed studies published in English were included; studies
published in other languages or in non-peer-reviewed sources were
excluded.
Titles and abstracts were screened independently by
two reviewers and full texts were retrieved if studies were judged
to be potentially eligible. Disagreements regarding eligibility
were resolved by consensus. To aid interpretation, each included
study received a simple quality rating (high, moderate or low)
based on sample size and representativeness, study design,
biomarker assay validity, and control for confounding (the ratings
and brief justifications are reported in Table SII). Subsequently, studies were
organized into three interconnected areas: i) Socio-emotional and
behavioral development; ii) cognitive, language and executive
function outcomes; and iii) epigenetics and biological markers of
stress.
3. Integrated summary of results
Across studies, a number of consistent patterns
emerged in socio-emotional, cognitive, language and executive
function, and biological domains. Table I summarizes converging and
diverging findings across these domains and highlights key
moderators as well as the strength of the evidence.
 | Table ICross-domain synthesis of evidence on
early childhood development during the 2019 coronavirus disease
pandemic (2020-2025). |
Table I
Cross-domain synthesis of evidence on
early childhood development during the 2019 coronavirus disease
pandemic (2020-2025).
| Domain | Consistent
findings | Divergent or mixed
findings | Key moderators | Evidence
strength |
|---|
| Socioemotional and
behavioral | Small to modest
increases in internalizing and externalizing symptoms across the
majority of cohorts; higher parental stress, anxiety and depression
predicted greater child emotional and behavioral difficulties;
prenatal and postpartum maternal mental health problems associated
with higher infant negative affect; greater pandemic-related family
disruption associated with more sleep problems, anxiety and
behavioral difficulties; higher quality teacher contact during
remote learning associated with improved social and behavioral
outcomes. | Minimal
socioemotional differences when parental mental health was stable
or when family routines remained intact; peer problems increased
primarily in lower-education families, not universally;
meta-analytic estimates showed high heterogeneity, with varied
effect sizes across regions, tools and timing. | Caregiver mental
health (stress, anxiety and depression); parenting practices (harsh
or inconsistent vs. supportive parenting); SES and caregiver
education; household chaos, routine disruption and daily stability;
parent-child relationship quality (bonding, attachment and positive
parenting); child biological sensitivity (such as basal cortisol
moderating effects of harsh parenting). | Moderate-high
(large-pooled samples and multiple longitudinal cohorts; consistent
patterns despite heterogeneity; heavy reliance on parent-report
measures). |
| Cognitive and
language | Small reductions in
communication and problem-solving; increased probability of
screening positive for delay inpandemic-born infants; stronger
effects where ECEC access was reduced. | No differences when
maternal infection was mild/asymptomatic; motor outcomes largely
stable. | Access to ECEC;
home learning environment and language-rich interactions;
prematurity and VLBW status (amplifying developmental
vulnerability); SES and digital access; severity of educational
disruption. | Moderate (large
population-based samples; evidence drawn from both parent-report
screening and standardized assessments across studies). |
| Executive function
and regulatory capacity | Maternal stress
associated with poorer infant regulatory capacity; working memory
and attentional processes sensitive to environmental instability;
working memory, attention and cognitive flexibility sensitive to
environmental instability; reduced social support associated with
poorer early regulatory behaviors. | Minimal differences
in cohorts with stable routines and strong family structure; a
number of EF domains, such as inhibitory control, exhibited
relative stability in numerous cohorts. | Parenting stress;
social support; household chaos and routine disruption; quality of
early caregiving interactions. | Moderate
(longitudinal evidence across multiple cohorts, though samples are
generally small and EF often measured indirectly). |
| Biological and
physiological markers (epigenetics, stress physiology and
neurobiology) | Prenatal
pandemic-related stress associated with altered methylation in
NR3C1 and SLC6A4; severe maternal SARS-CoV-2 infection associated
with widespread infant differential methylation and lower ASQ-3
scores at 12 months; prenatal maternal distress associated with
altered infant brain structure and connectivity (such as smaller
amygdala volumes and altered amygdala-PFC connectivity); pandemic
exposure associated with broad epigenetic signatures in neonates
vs. pre pandemic controls. | No methylation
differences associated with mild/asymptomatic maternal SARS-CoV-2
infection; hair cortisol findings were largely null, as no
pandemic-associated changes were reported, and prenatal stress was
not consistently associated with maternal or infant cortisol; a
number of stress biomarkers (such as hair cortisol or DHEA) were
moderated by SES as opposed to pandemic exposure alone. | Severity of
maternal infection; SES (moderating cortisol); timing of prenatal
stress (trimester-specific effects); maternal inflammation (such as
IL-6 and IL-17A) predicting infant neurodevelopment; maternal
anxiety and depression (predicting infant amygdala structure and
temperament). |
Preliminary-moderate (small to moderate
samples; heterogeneous biomarkers; strong mechanistic designs but
limited replication). |
Socioemotional and behavioral
development
Across studies of socio-emotional and behavioral
development (16-28),
there was a consistent pattern of elevated difficulties among
children exposed to the COVID-19 pandemic period, with evidence
from meta-analyses indicating a higher prevalence of emotional and
behavioral problems compared with pre-pandemic estimates (11,14).
Additionally, longitudinal cohorts documented increases in
emotional and peer problems over time, with changes observed from
the pre-pandemic period (2018-2019) through the early pandemic
phase (2020) and into later phases (2021-2022) (15). A subset of children followed
persistent high-symptom trajectories across the pandemic period,
particularly in emotional and oppositional-defiant domains
(16). These trajectories were
strongly shaped by family-level stressors, including elevated
parental psychological distress, job or income loss, increased
caregiving burden and disruptions to daily routines. Such
family-level stressors were central to the observed patterns as
they reduced the emotional and regulatory resources available
within the family environment, thereby heightening children's
vulnerability to socio-emotional difficulties during periods of
prolonged uncertainty and disruption. Broader caregiver stressors,
such as financial strain, household chaos, reduced childcare access
and pandemic-related disruptions, were consistently associated with
increases in children's internalizing symptoms (for example,
anxiety and withdrawal) and externalizing symptoms (for example,
irritability and aggression), sleep problems and behavioral
dysregulation (17-19),
with longitudinal evidence indicating stronger associations in
families with less socioeconomic resources (15).
Among infants and toddlers, higher maternal
depression and anxiety, increased parenting stress, reduced social
support, and disrupted daily routines were associated with
increased socio-emotional difficulties (20,29,30).
Peer difficulties increased in preschoolers during the pandemic,
with positive parent-child interactions buffering these effects,
although these supports did not consistently mitigate emotional
symptoms, which remained associated with caregiver stress and
broader household disruptions (15,16,29).
Despite these patterns, findings were not uniform. A number of
studies reported relatively consistent socio-emotional functioning
when family routines were preserved or when parental mental health
remained stable (31,32). In addition, the magnitude of
socio-emotional difficulties varied across cultural and policy
contexts, where strict lockdowns, extended preschool closures and
culturally specific caregiving patterns, such as reliance on
structured early education in Germany and prolonged home
confinement with limited outdoor play in China, were associated
with more pronounced effects in settings experiencing prolonged
lockdowns or greater household disruption (17,19).
In these studies, household disruption referred to increased
parental stress, job or income loss, crowded or unstable living
arrangements and significant disturbances to daily routines. These
variations highlight the importance of contextual factors in
shaping the socio-emotional responses of children to
pandemic-related stress. However, interpretation of these findings
was constrained by numerous methodological limitations, including
reliance on parent-reported measures, convenience sampling,
heterogeneous exposure definitions and notable attrition in
longitudinal cohorts. Therefore, caution is warranted when
interpreting the magnitude of socio-emotional differences, even
though the overall pattern of increased vulnerability remained
consistent.
Cognition, language and executive
function
Across studies examining cognition, language and EF
development (12,13,33-45),
a number of points of convergence emerged regarding the
developmental consequences of the COVID-19 pandemic for infants and
young children. Firstly, modest but consistent reductions across
communication, problem-solving and personal-social domains were
observed across numerous large-scale and international cohorts
(33,46), with communication and
problem-solving demonstrating the clearest disruptions (14,33,46).
Furthermore, a number of studies reported increased probabilities
of screening positive for developmental delay, which was driven in
part by increased rates of language-specific delays (34-36).
However, these patterns were not uniform across all children, as
preterm (gestational age <32 weeks) or very low birth weight
(<1,500 g) infants exhibited the most pronounced differences,
with notably lower cognitive and language scores compared with
pre-pandemic peers (37). Although
these infants represented a subgroup that was biologically at-risk
due to immature brain development, higher rates of neonatal
complications and increased vulnerability to environmental
stressors, their developmental profiles were consistent with
broader international findings, which indicated small but reliable
reductions in early problem-solving and communication abilities
among pandemic-born infants (14),
as well as early delays in social communication, including reduced
meaningful word use, pointing and waving (38). Converging findings from cohort
studies and a comprehensive review of international research
documented consistent patterns of lower performance in
morphosyntax, expressive vocabulary, phonological awareness and
early literacy skills, with instructionally dependent language
skills having the largest disruptions and children from lower
socioeconomic backgrounds having the longest delays (12,39,40).
Teacher-reported longitudinal data, analyzed in a study by Bub
et al (47), indicated that
early learning and social-emotional skills were shaped by the
quality of remote learning supports, with marginalized children
receiving a reduced number of resources and less frequent teacher
contact. These population-level shifts were moderated by parental
mental health and early-education environments, in which maternal
depression amplified developmental delays, whereas higher-quality
early care settings were associated with more favorable outcomes,
including enhanced expressive vocabulary, better early literacy
skills and more advanced socio-emotional competencies.
A second area of consistent convergence was the role
of caregiver psychological functioning. Across diverse cohorts,
prenatal psychological distress, measured through mental health
symptoms, pandemic-related stress or reduced social support,
consistently predicted less optimal infant socio-emotional,
regulatory and socio-cognitive outcomes, often operating indirectly
through postnatal maternal mental health and parenting stress
(20-22,29,30).
Postnatal maternal depression, anxiety and perceived stress had
similar associations, and a systematic review identified
associations between postnatal depression and poorer mother-infant
bonding, alongside difficulties in infant motor, self-regulation
and socioemotional development (23). Furthermore, individual cohorts
demonstrated that higher maternal stress predicted increased infant
crying, fussiness and regulatory challenges even after accounting
for prenatal symptoms (24,29).
In addition, early motor development had a similar sensitivity to
maternal mental health; higher prenatal and postpartum depressive
symptoms were associated with poorer neonatal fine and gross motor
abilities, with the strongest effects observed among preterm and
low-birth-weight infants (25,33).
Population-level data in studies by Giesbrecht et al
(48) and Perrigo et al
(46) also demonstrated these
patterns as large municipal and national datasets showed that
children exposed to the pandemic exhibited developmental lags by
preschool age, with widening variability in developmental outcomes.
These population-level shifts were moderated by family mental
health and early-education environments (26,47,49).
Taken together, these findings suggested that pandemic-related
disruptions influenced not only mean levels of development but also
the distribution and heterogeneity of developmental functioning
across cohorts.
Despite these areas of convergence, a number of
divergences were noted. Studies examining the impact of maternal
severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)
infection during pregnancy yielded mixed results. Certain studies
reported elevated developmental risk, particularly when infection
occurred in the first or second trimester, was severe or
co-occurred with prematurity (34,35,37).
However, other studies found no association between prenatal
infection and developmental outcomes (41-43).
These discrepancies may reflect differences in illness severity,
timing of exposure, sample characteristics and measurement tools, a
pattern consistent with broader heterogeneity across pandemic-era
cohorts. Additionally, findings regarding motor development varied
across studies, as although a number identified delays,
particularly among high-risk infants or in the context of maternal
mental health difficulties (25,37,44),
a large population-based study reported no pandemic-related
differences in gross motor (namely, walking without assistance and
climbing stairs with assistance) and fine motor (namely, building a
tower of cubes) milestones at 2 years of age (45).
EF-related processes (for example working memory,
inhibitory control, cognitive flexibility) also appeared
particularly sensitive to these stress-related and environmental
disruptions (13,24,50-53).
A quasi-experimental study from Russia found that preschoolers
assessed during lockdown showed slower gains in cognitive
flexibility and working memory, while inhibitory control remained
relatively stable; girls had increased attenuation in
working-memory development, suggesting potential sex-based
differences in sensitivity to disrupted routines (13). Other studies also documented
EF-related disruptions. In preschoolers, working memory mediated
associations between environmental routines and emotional
dysregulation (51) and studies of
infant regulatory behaviors showed that maternal stress and reduced
social support were associated with poorer early regulatory
capacity (24,52), an EF-precursor skill (53). Additional studies reported
increased difficulties regarding attentiveness and distractibility
in the context of family stress, disrupted routines or harsh
parenting, defined as punitive, irritable or overreactive
disciplinary practices measured using the Parenting Scale and the
inconsistent-discipline items of the Alabama Parenting
Questionnaire (18,27), reinforcing the role of early stress
and environmental instability in shaping EF-related pathways.
Collectively, these findings indicated that while cognitive and
language disruptions during the pandemic were modest, EF processes
had an increased sensitivity to stress-related and environmental
instability, highlighting the role of caregiver wellbeing and daily
routines in shaping early developmental trajectories.
Taken together, this evidence indicated that the
COVID-19 pandemic exerted broad but heterogeneous influences on
early development. The most consistent patterns suggested modest
population-level shifts in communication and socio-cognitive
domains (33,46,54),
increased vulnerabilities among biologically or socioeconomically
at-risk infants (12,37), and robust pathways that associate
caregiver psychological functioning with infant outcomes (39,47).
Divergences across studies, particularly regarding prenatal
infection and motor development, highlights the need for continued
longitudinal follow-up and harmonized assessment approaches.
However, the interpretation of these findings is
affected by a number of methodological limitations across these
cognition, language and EF studies. The majority of cohorts relied
on parent-reported screening tools, such as the Ages and Stages
Questionnaire-3, which are sensitive to parental stress and
reporting bias (55) and may not
detect subtle cognitive or language differences (56). Numerous samples were
convenience-based, highly educated or lacked socioeconomic and
racial diversity, limiting generalizability. Exposure definitions
also varied widely, from broad pandemic-period indicators to
specific measures of maternal infection or family stress,
complicating comparisons across studies. Studies investigating
maternal SARS-CoV-2 infection often included mild or asymptomatic
cases, reducing the ability to detect the effects of more severe
illness. Follow-up periods were frequently short, particularly for
infants assessed in the first year of life, leaving uncertainty
regarding whether early differences persist or resolve over time.
Taken together, due to these limitations, caution in interpreting
the magnitude of cognitive and language differences is suggested,
even though the overall pattern indicated modest disruptions
associated with contextual, as opposed to viral, exposures.
Epigenetics and biological
markers
Across studies examining biological embedding during
the COVID-19 pandemic, findings indicate subtle yet marked
alterations in infant and child stress-related biology, though
effects vary across biomarkers, tissues and exposure definitions
(31,54,57-61).
An epigenetic study reported pandemic-related differences in DNA
methylation of stress-regulatory genes. Infants exposed in
utero exhibit altered methylation in nuclear receptor subfamily
3 group C member 1 and solute carrier family 6 member 4 (SLC6A4),
with trimester-specific patterns suggesting that first-trimester
exposure may represent a particularly sensitive window of
vulnerability (57). Genome-wide
comparisons of pandemic-exposed neonates with pre-pandemic controls
identifies hundreds of differentially methylated sites enriched in
neurodevelopmental pathways (synapse organization, neurogenesis and
axon guidance) and immune pathways (cytokine-mediated signaling,
interferon responses and leukocyte activation), whereas maternal
SARS-CoV-2 infection itself is not associated with methylation
differences (58). By contrast,
severe maternal infection is associated with widespread
differential DNA methylation in infants and elevated maternal IL-6
concentrations, reflecting a heightened inflammatory milieu during
pregnancy. These biological alterations show modest,
domain-specific associations with developmental scores at 12
months, suggesting that maternal immune activation may exert subtle
but measurable influences on early neurodevelopment (59,61).
Additional research shows that higher prenatal pandemic-related
stress predicts increases in infant SLC6A4 methylation, which is
associated with altered temperament at 3 months (60).
Neuroimaging studies provide converging evidence
that prenatal stress is associated with early alterations in infant
brain structure and connectivity (54,62).
Increased prenatal maternal anxiety predicts reduced left amygdala
volumes at 3 months, which are subsequently associated with
increased negative affectivity at 6 months (54). Complementary work shows that
greater prenatal maternal distress is associated with altered
infant white-matter microstructure and altered amygdala-prefrontal
connectivity, suggesting early deviations in neural circuits
supporting emotional regulation (62). Taken together, these findings offer
preliminary evidence that prenatal stress during the pandemic may
be associated with early structural and connectivity in neural
systems implicated in emotion processing and regulation.
Physiological biomarkers showed a heterogeneous
pattern as opposed to a uniform rise in stress physiology during
the pandemic. In a small prenatal cohort, prenatal COVID-19-related
stress was not associated with maternal hair cortisol
concentrations (HCC), although socioeconomic status moderated this
pattern, with a stress-cortisol association emerging only among
women with a higher socioeconomic status; maternal HCC did not
predict infant temperament (31).
In a separate pre- to post-pandemic cohort with repeated
hair-steroid assessments, mean levels of cortisol, cortisone and
related adrenal steroids, such as dehydroepiandrosterone and
dehydroepiandrosterone sulfate, did not change across the pandemic
onset. However, family income and parent-reported family
functioning (conflict resolution, cohesion and COVID-19-related
stress) moderated these patterns, with increases in child HCC
occurring primarily in households with lower income or greater
family stress, with social support buffering these rises (61). Children with higher pre-pandemic
morning basal salivary cortisol showed greater physiological
reactivity, exhibiting steeper increases in anger when exposed to
harsh or inconsistent parenting, but the lowest anger level when
exposure to such parenting was low. Harsh or inconsistent parenting
was defined as more punitive, irritable or unpredictable
disciplinary practices measured using the Parenting Scale and the
inconsistent-discipline subscale of the Alabama Parenting
Questionnaire, both validated parent-report instruments (27). Collectively, these findings suggest
a possible biological-sensitivity-to-context interpretation, in
which individual differences stress physiology heighten
vulnerability in adverse environments but are buffered in
supportive ones, rather than reflecting a population-wide increase
in cortisol.
Taken together, these findings suggest preliminary
evidence of possible biological embedding associated with prenatal
and early-life stress during the pandemic. However, interpretation
of this evidence is constrained by numerous methodological
limitations. The majority of studies relied on peripheral tissues
(buccal cells, saliva and hair), which may not capture central
nervous system processes. Sample sizes were small to modest,
reducing the statistical power to detect subtle effects and
increasing the likelihood of false positives in genome-wide
analyses. Furthermore, the majority of cohorts lacked pre-pandemic
biological baselines, making it difficult to isolate
pandemic-specific effects. Additionally, exposure definitions
varied widely between studies, from broad pandemic timing to
specific measures of maternal stress or infection, which
complicates comparability across studies. Samples were also often
highly educated and socioeconomically advantaged, limiting
generalizability. In addition, neuroimaging studies were restricted
to single time points, preventing assessment of developmental
trajectories. Finally, the observational nature of all studies
suggests the possibility of residual confounding from unmeasured
prenatal or postnatal factors. These limitations highlight the need
for cautious interpretation, even though the emerging evidence
indicates potential biological embedded effects of prenatal stress
during the pandemic.
4. Discussion and conclusion
Results of the present narrative review showed that
pandemic-related disruptions acted as a multisystem stressor that
altered caregiving (21,28), reduced access to early learning
(15,33) and disrupted the daily routines of
children (18,24). These disruptions also extended to
sleep (31) which is suggested to
be a multidimensional health process involving quality, timing and
circadian regularity as opposed to duration alone (63). Socioemotional outcomes showed the
most consistent evidence and were associated with caregiver mental
health (29,30). By comparison, developmental
outcomes were more strongly associated with pandemic-related
psychosocial stressors such as caregiver distress, routine
instability and reduced stimulation compared with direct viral
exposure. Evidence regarding motor development and musculoskeletal
outcomes remains limited, representing an important area of future
research, particularly given concerns regarding potential motor
delays associated with reduced physical activity during lockdowns
(64). Cognitive and language
effects appeared more variable, emerging in settings where
stimulation or early-education access was curtailed (15,33).
Biological measures provided preliminary, context-dependent signals
that requires validation in larger, harmonized cohorts (54,61).
These biological findings should be interpreted as early,
hypothesis-generating biomarkers as opposed to established
mechanistic pathways. Overall, the evidence indicated
context-dependent impacts as opposed to uniform population-level
effects, with children in socioeconomically disadvantaged or
high-stress households disproportionately affected (18,19).
This pattern is consistent with broader evidence that indicated
that children living in shelters experienced increased psychosocial
stress and reduced access to supportive services during the
pandemic (65). Biomedical
complications, vaccine-related effects and rare post-infectious
conditions fell outside the scope of the present developmental
review, which focused on psychosocial and neurodevelopmental
pathways; however, they are important areas for future clinical
research.
The pattern of results supported a
developmental-systems view in which caregiver functioning,
biological stress pathways and environmental resources together
moderate how children respond to acute societal stressors (Fig. 1).
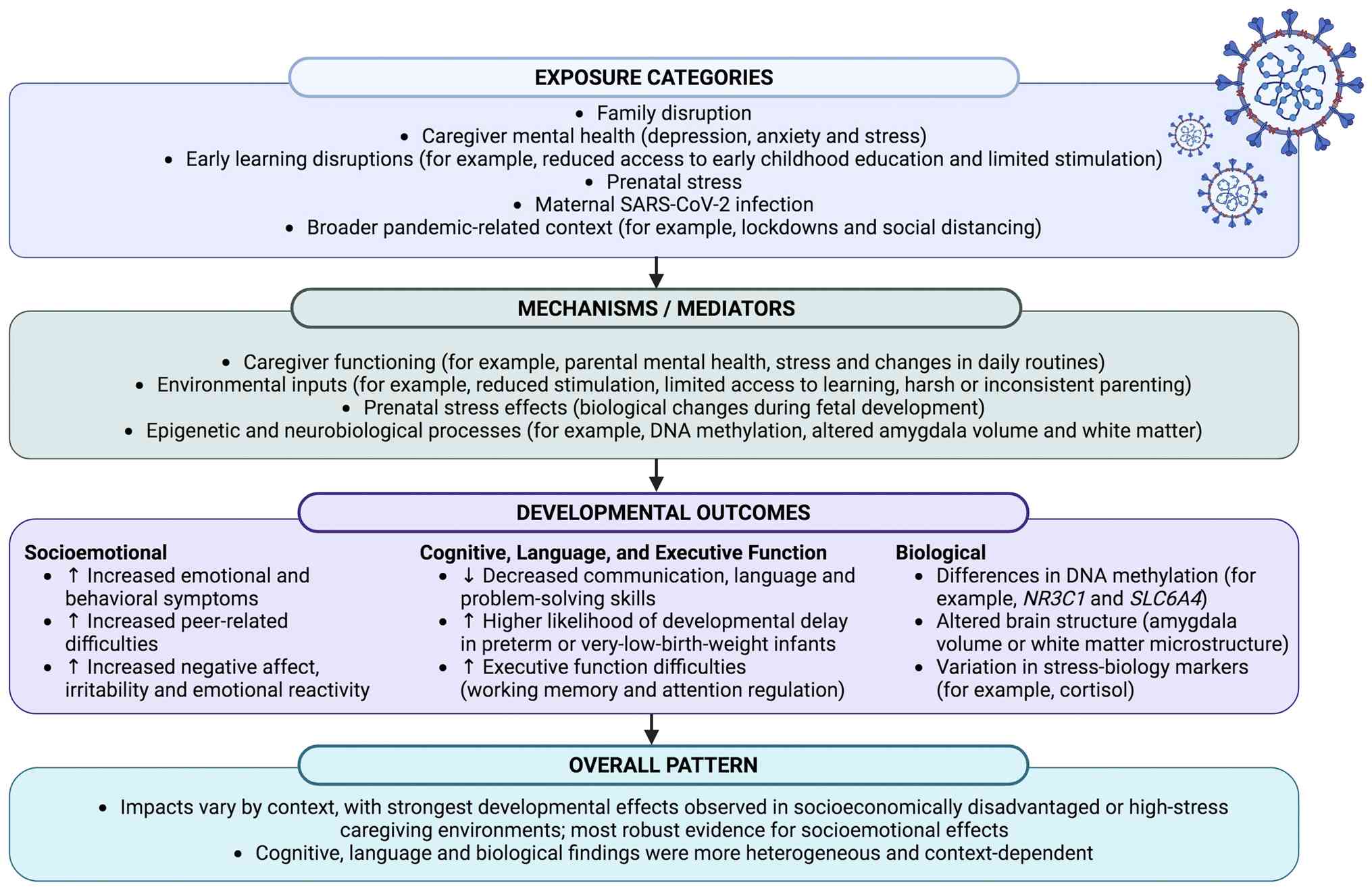 | Figure 1Conceptual pathway model integrating
caregiver mental health, biological stress pathways and
environmental inputs as mediators associating pandemic-related
exposures with early childhood developmental outcomes. The present
model summarizes the evidence regarding how pandemic-related
exposures may have influenced socioemotional, cognitive and
biological development. Key exposure categories included family
disruption, caregiver mental health challenges, disruptions to
learning in early childhood (such as reduced access to early
childhood education and limited stimulation), prenatal stress and
maternal SARS-CoV-2 infection. These exposures may influence
developmental outcomes through interconnected pathways involving
caregiver functioning, environmental inputs, prenatal stress
effects and biological embedding (including epigenetic and
neurobiological changes). Observed developmental outcomes included
elevated socioemotional difficulties (such as emotional and
behavioral symptoms or peer problems), modest cognitive,
communication and language delays (particularly among preterm or
high-risk infants) and biological alterations (such as DNA
methylation differences or altered brain structure/connectivity).
The overall pattern indicated possible context-dependent effects,
with the strongest developmental disruptions observed in
socioeconomically disadvantaged or high-stress caregiving
environments. Mild or asymptomatic maternal infection was not
consistently associated with developmental differences. Although
interpretation was limited by methodological variability across
studies, the present model summarizes converging evidence on early
developmental vulnerabilities during the 2019 coronavirus disease
pandemic. Images were generated using BioRender (https://www.biorender.com/). SARS-CoV-2, severe acute
respiratory syndrome coronavirus 2; NR3C1, nuclear receptor
subfamily 3 group C member 1; SLC6A4, solute carrier family 6
member 4. |
These findings were consistent with mechanistic
models of early life stress, showing that alterations in caregiver
availability, predictability and emotional climate shape neural,
endocrine and behavioral development through a number of
interacting pathways (66). These
pathways encompass alterations in corticolimbic circuitry involved
in emotion regulation, changes in hypothalamic-pituitary-adrenal
axis functioning, shifts in immune and inflammatory signaling, and
disruptions in learning and predictability processes that guide
children's emerging behavioral and socioemotional regulation.
Socioemotional outcomes had the strongest and most consistent
associations with caregiver stress (29,30),
suggesting that interventions targeting caregiver mental health
could yield rapid benefits for child well-being. Evidence further
highlighted the notable mental-health burden experienced by parents
of young children during the pandemic, with elevated anxiety and
depression associated with contextual stressors such as childcare
and school closures, financial strain, social isolation,
work-family conflict and concerns about healthcare access (5). Cognitive and language outcomes
reflect differential exposure to stimulation and early learning
supports (15,33), which highlighted structural
inequalities in access to protective environments. Biological
findings, including cortisol, DNA methylation and infant MRI,
provided early, hypothesis-generating signals in a number of
high-risk samples (54,61); however, these were limited by
modest sample sizes, assay heterogeneity and limited longitudinal
follow-up. These emerging biological patterns were consistent with
developmental-evolutionary models that proposed that heightened
stress reactivity reflected a biologically sensitive and
conditionally adaptive calibration to early environments, with
effects that varied depending on the balance of adversity and
support (67). Given these
methodological constraints, the possibility of publication bias in
pandemic-era research should also be acknowledged.
Small or non-representative samples,
single-informant designs, heterogeneous measures and limited
cross-cultural coverage reduced comparability and increased bias
risk, making causal claims premature and highlighting the need for
more rigorous, harmonized research. These limitations may also
explain why socioemotional outcomes showed clearer patterns
compared with cognitive or biological measures, which rely on more
variable or resource-intensive assessments. Therefore, these
methodological constraints indicated that the current evidence base
supports possible associations as opposed to causal conclusions.
However, despite these constraints, the present findings suggested
numerous implications for practice, policy and future research,
particularly in areas where evidence converged across domains.
Practical implications may include strengthening
caregiver mental-health supports in pediatric and early-childhood
settings and promoting programs that help families maintain
predictable routines and positive parent-child interactions during
periods of disruption. Policy implications suggested the importance
of ensuring continuity of early-education services during
emergencies, through prioritized reopening, remote supports
tailored to low-resource families and proactive outreach, as well
as providing economic supports to families with young children to
mitigate cascading stressors. Furthermore, research implications
highlighted the need for multisite, longitudinal cohorts with
harmonized measures, integration of multi-modal biomarkers with
behavioral and contextual data, and transparent reporting practices
that include null findings and quality metrics.
Overall, pandemic-related disruptions may have
amplified existing inequities in early childhood development, with
the most notable impacts concentrated among children facing
socioeconomic disadvantage and caregiver distress. Therefore,
moving from preliminary associations to actionable knowledge may
require coordinated, longitudinal and multi-modal research, coupled
with policies that protect early-education access and support
caregiver wellbeing.
Supplementary Material
Full database search strings used in
the literature search (January 2020-June 2025).
Quality appraisal criteria for
narrative review evidence.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
IG and VE wrote the original draft, edited and
revised the manuscript. MS, PK, JNT, DAS and ER revised and edited
the manuscript. IG, VE, MS, PK, JNT, DAS and ER contributed to the
conception, writing and revision of the work. All authors read and
approved the final version of the manuscript. Data authentication
is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
IG, VE, MS, PK, JNT and ER declare that they have no
competing interests. DAS is the Editor-in-Chief for the journal,
but had no personal involvement in the reviewing process, or any
influence in terms of adjudicating on the final decision, for this
article.
References
|
1
|
Tsamakis K, Tsiptsios D, Ouranidis A,
Mueller C, Schizas D, Terniotis C, Nikolakakis N, Tyros G,
Kympouropoulos S, Lazaris A, et al: COVID-19 and its consequences
on mental health (Review). Exp Ther Med. 21(244)2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Efstathiou V, Stefanou MI, Demetriou M,
Siafakas N, Katsantoni E, Makris M, Tsivgoulis G, Zoumpourlis V,
Kympouropoulos SP, Tsoporis JN, et al: New-onset neuropsychiatric
sequelae and ‘long-COVID’ syndrome (Review). Exp Ther Med.
24(705)2022.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Efstathiou V, Stefanou MI, Demetriou M,
Siafakas N, Makris M, Tsivgoulis G, Zoumpourlis V, Kympouropoulos
SP, Tsoporis JN, Spandidos DA, et al: Long COVID and
neuropsychiatric manifestations (Review). Exp Ther Med.
23(363)2022.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Giannopoulou I, Galinaki S, Kollintza E,
Adamaki M, Kympouropoulos S, Alevyzakis E, Tsamakis K, Tsangaris I,
Spandidos DA, Siafakas N, et al: COVID-19 and post-traumatic stress
disorder: The perfect ‘storm’ for mental health (Review). Exp Ther
Med. 22(1162)2021.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Rodríguez-Toscano E, Schleicher D, Ecker
A, Kandsperger S, Brunner R and Jarvers I: Exploring the mental
health impact of COVID-19 on parents of young children: Anxiety,
depression, and contributing factors. BMC Public Health.
25(2174)2025.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Gabard-Durnam L and McLaughlin KA:
Sensitive periods in human development: Charting a course for the
future. Curr Res Behav Sci. 36:120–128. 2020.
|
|
7
|
Danese A and Smith P: Debate: Recognising
and responding to the mental health needs of young people in the
era of COVID-19. Child Adolesc Ment Health. 25:169–170.
2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Yoshikawa H, Wuermli AJ, Britto PR, Dreyer
B, Leckman JF, Lye SJ, Ponguta LA, Richter LM and Stein A: Effects
of the global coronavirus disease-2019 pandemic on early childhood
development: Short- and long-term risks and mitigating program and
policy actions. J Pediatr. 223:188–193. 2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Bondi BC, Tassone VK, Bucsea O, Desrocher
M and Pepler DJ: A systematic review of neurodevelopmental
assessments in infancy and early childhood: Developing a conceptual
framework, repository of measures, and clinical recommendations.
Neuropsychol Rev. 35:337–353. 2025.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Bronfenbrenner U and Morris PA: The
Bioecological Model of Human Development. In: Handbook of Child
Psychology. Damon W, Lerner LM and Lerner RM (eds). John Wiley
& Sons, Ltd., 2007.
|
|
11
|
Jing JQ, Yang CJ, Wang Y, Su XY and Du YJ:
Impact of COVID-19 on emotional and behavioral problems among
preschool children: A meta-analysis. BMC Pediatr.
24(455)2024.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Zuniga-Montanez C, Davies C, Ligoxygakis
L, Kašćelan D and Gonzalez-Gomez N: Annual research review: How did
COVID-19 affect young children's language environment and language
development? A scoping review. J Child Psychol Psychiatry.
66:569–587. 2025.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Chichinina EA and Gavrilova MN: Growth of
executive functions in preschool-age children during the COVID-19
lockdown: Empirical evidence. Psychol Russ. 15:124–136.
2022.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Alcon S, Shen S, Wong HN, Rovnaghi CR,
Truong L, Vedelli JKH and Anand KJS: Effects of the COVID-19
pandemic on early childhood development and mental health: A
systematic review and meta-analysis of comparative studies. Psychol
Int. 6:986–1012. 2024.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Oppermann E, Blaurock S, Zander L and
Anders Y: Children's social-emotional development during the
COVID-19 pandemic: Protective effects of the quality of children's
home and preschool learning environments. Early Educ Dev.
35:1432–1460. 2024.
|
|
16
|
Landolt MA, Ruther N, Strebel NL and
Schmidt SJ: Trajectories of emotional and behavioral problems in
young children during the COVID-19 pandemic: A longitudinal study.
PLOS Ment Health. 2(e0000265)2025.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Jarvers I, Ecker A, Schleicher D, Brunner
R and Kandsperger S: Impact of preschool attendance, parental
stress, and parental mental health on internalizing and
externalizing problems during COVID-19 lockdown measures in
preschool children. PLoS One. 18(e0281627)2023.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Ding X, Liu H, Wang H, Song Q, Su W, Li N,
Liang M, Guo X, Qu G, Ma S, et al: COVID-19 pandemic impact on
family life and exacerbated emotional and behavioral health among
preschool children: A longitudinal study. Int J Clin Health
Psychol. 22(100327)2022.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Weng W, Liu M, Wang S, Li X, Zhang J, Fu
Y, Dong C and Li Y: The impact of lockdown on child adjustment: A
propensity score matched analysis. BMC Psychol.
12(409)2024.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Duguay G, Garon-Bissonnette J, Lemieux R,
Dubois-Comtois K, Mayrand K and Berthelot N: Socioemotional
development in infants of pregnant women during the COVID-19
pandemic: The role of prenatal and postnatal maternal distress.
Child Adolesc Psychiatry Ment Health. 16(28)2022.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Provenzi L, Grumi S, Altieri L, Bensi G,
Bertazzoli E, Biasucci G, Cavallini A, Decembrino L, Falcone R,
Freddi A, et al: Prenatal maternal stress during the COVID-19
pandemic and infant regulatory capacity at 3 months: A longitudinal
study. Dev Psychopathol. 35:35–43. 2023.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Nazzari S, Grumi S, Biasucci G, Decembrino
L, Fazzi E, Giacchero R, Magnani ML, Nacinovich R, Scelsa B,
Spinillo A, et al: Maternal pandemic-related stress during
pregnancy associates with infants' socio-cognitive development at
12 months: A longitudinal multi-centric study. PLoS One.
18(e0284578)2023.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Federica G, Renata T and Marzilli E:
Parental postnatal depression in the time of the COVID-19 Pandemic:
A systematic review of its effects on the parent-child relationship
and the child's developmental outcomes. Int J Environ Res Public
Health. 20(2018)2023.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Bradley H, Fine D, Minai Y, Gilabert L,
Gregory K, Smith L, Gao W, Giase G, Krogh-Jespersen S, Zhang Y, et
al: Maternal perceived stress and infant behavior during the
COVID-19 pandemic. Pediatr Res. 94:2098–2104. 2023.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Papadopoulos A, Nichols ES, Mohsenzadeh Y,
Giroux I, Mottola MF, Van Lieshout RJ and Duerden EG: Prenatal and
postpartum maternal mental health and neonatal motor outcomes
during the COVID-19 pandemic. J Affect Disord Rep.
10(100387)2022.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Jeličić L, Sovilj M, Bogavac I, Drobnjak
AE, Gouni O, Kazmierczak M and Subotić M: The impact of maternal
anxiety on early child development during the COVID-19 pandemic.
Front Psychol. 12(792053)2021.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Ostrov JM, Murray-Close D, Perry KJ,
Perhamus GR, Memba GV, Rice DR and Nowalis S: Parenting and
adjustment problems among preschoolers during COVID-19. J Child Fam
Stud. 32:93–109. 2023.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Joo YS and Lee WK: Impact of
COVID-19-related stress on preschool children's internalizing and
externalizing problem behaviors: The indirect effect of mother's
depression and parenting behavior. Child Indic Res. 15:2093–2113.
2022.PubMed/NCBI View Article : Google Scholar
|
|
29
|
López-Morales H, Canet-Juric L, Del-Valle
MV, Sosa JM, López MC and Urquijo S: Prenatal anxiety during the
pandemic context is related to neurodevelopment of 6-month-old
babies. Eur J Pediatr. 182:4213–4226. 2023.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Sacchi C, De Carli P, Gregorini C, Monk C
and Simonelli A: In the pandemic from the womb. Prenatal exposure,
maternal psychological stress and mental health in association with
infant negative affect at 6 months of life. Dev Psychopathol.
36:810–820. 2024.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Bruinhof N, Vacaru SV, van den Heuvel MI,
de Weerth C and Beijers R: Prenatal hair cortisol concentrations
during the COVID-19 outbreak: Associations with maternal
psychological stress and infant temperament.
Psychoneuroendocrinology. 144(105863)2022.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Vrantsidis DM, van de Wouw M, Hall ERM,
Kuret V, Rioux C, Conrad ML, Mesa C, Harris A, Lebel C,
Tomfohr-Madsen L and Giesbrecht GF: Neurodevelopment in the first 2
years of life following prenatal exposure to maternal SARS-CoV-2
infection. JAMA Netw Open. 7(e2443697)2024.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Johnson SB, Kuehn M, Lambert JO, Spin JP,
Klein LM, Howard B, Sturner R and Perrin EM: Developmental
milestone attainment in US children before and during the COVID-19
pandemic. JAMA Pediatr. 178:586–594. 2024.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Ayed M, Embaireeg A, Kartam M, More K,
Alqallaf M, AlNafisi A, Alsaffar Z, Bahzad Z, Buhamad Y, Alsayegh
H, et al: Neurodevelopmental outcomes of infants born to mothers
with SARS-CoV-2 infections during pregnancy: A national prospective
study in Kuwait. BMC Pediatr. 22(319)2022.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Santos CAD, Paula AP, Filho GGF, Alves MM,
Nery AF, Pontes MGA, Macedo EYL, Oliveira RM Jr, Freitas SM, Lima
S, et al: Developmental impairment in children exposed during
pregnancy to maternal SARS-COV2: A Brazilian cohort study. Int J
Infect Dis. 139:146–152. 2024.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Fajardo-Martinez V, Ferreira F, Fuller T,
Cambou MC, Kerin T, Paiola S, Mok T, Rao R, Mohole J, Paravastu R,
et al: Neurodevelopmental delay in children exposed to maternal
SARS-CoV-2 in-utero. Sci Rep. 14(11851)2024.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Kim H, Jang YH, Lee JY, Lee GY, Sung JY,
Kim MJ, Lee BG, Yang S, Kim J, Yoon KS, et al: Impact of COVID-19
pandemic on neurodevelopmental outcome in very low birth weight
infants: A nationwide cohort study. Front Pediatr.
12(1368677)2024.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Byrne S, Sledge H, Franklin R, Boland F,
Murray DM and Hourihane J: CORAL Study group. Social communication
skill attainment in babies born during the COVID-19 pandemic: A
birth cohort study. Arch Dis Child. 108:20–24. 2023.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Nevo E: The Effect of the COVID-19
pandemic on low SES kindergarteners' language abilities. Early
Child Educ J. 1-11:2023.PubMed/NCBI View Article : Google Scholar : (Epub ahead of
print).
|
|
40
|
Cadime I, Santos AL, Ribeiro I, Viana FL
and Martín-Aragoneses MT: Living the first years in a pandemic:
children's linguistic development and related factors in and out of
the COVID-19 lockdowns. J Child Lang. 52:1157–1183. 2025.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Jaswa EG, Huddleston HG, Lindquist KJ, Wu
AHB, Bishop SL, Kim YS, Kaing A, Prahl M, Gaw SL, Corley J, et al:
In utero exposure to maternal COVID-19 and offspring
neurodevelopment through age 24 months. JAMA Netw Open.
7(e2439792)2024.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Firestein MR, Shuffrey LC, Hu Y, Kyle M,
Hussain M, Bianco C, Hott V, Hyman SP, Kyler M, Rodriguez C, et al:
Assessment of neurodevelopment in infants with and without exposure
to asymptomatic or mild maternal SARS-CoV-2 infection during
pregnancy. JAMA Netw Open. 6(e237396)2023.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Berg J, Linden K, Zaigham M, Domellöf M,
Ahlsson F, Elfvin A, Åden U, Abrahamsson T, Ohlin A, Berg J, et al:
The association between antenatal SARS-CoV-2 exposure and infant
neurodevelopment at four months of age: A prospective multicenter
cohort survey within the COPE study. Int J Infect Dis.
158(107973)2025.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Tseng TC, Wang TM, Hsu YC, Hsu CT, Lin YH
and Lin MC: Taiwan Premature Infant Follow-up Network. Impact of
COVID-19 pandemic on neurodevelopmental outcomes of premature
infants: a retrospective national cohort study. BMJ Paediatr Open.
8(e002493)2024.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Eisen L, Vinograd A, Sheffer R, Kaliner E,
Zimmerman DR, Alroy-Preis S, Eisenberg VH, Lerner Geva L and Mor Z:
Nationwide analysis of child development amid the COVID-19
pandemic. Pediatrics Open Sci. 1:1–7. 2025.
|
|
46
|
Perrigo JL, Morales J, Jackson N, Janus M,
Stanley L, Wong M and Halfon N: COVID-19 pandemic and the
developmental health of kindergarteners. JAMA Pediatr. 179:550–558.
2025.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Bub KL, Neuharth-Pritchett S, Smolinski E
and Hirt S: Teacher-child contact and pre-kindergartener's academic
and social-emotional skills following COVID-19 Disruptions. Early
Childhood Educ J, 2025.
|
|
48
|
Giesbrecht GF, Lebel C, Dennis CL, Silang
K, Xie EB, Tough S, McDonald S and Tomfohr-Madsen L: Risk for
developmental delay among infants born during the COVID-19
pandemic. J Dev Behav Pediatr. 44:e412–e420. 2023.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Sato K, Fukai T, Fujisawa KK and Nakamuro
M: Association between the COVID-19 pandemic and early childhood
development. JAMA Pediatr. 177:930–938. 2023.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Zelazo PD and Carlson SM: Hot and cool
executive function in childhood and adolescence: Development and
plasticity. Child Dev Perspect. 6:354–360. 2012.
|
|
51
|
Lee JJ, Flouri E and Jackson Y: The role
of timing and amount of outdoor play in emotional dysregulation in
preschool children. Child Care Health Dev.
51(e70020)2025.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Alonso-Martínez AM, Ramírez-Vélez R,
García-Alonso Y, Izquierdo M and García-Hermoso A: Physical
activity, sedentary behavior, sleep and self-regulation in spanish
preschoolers during the COVID-19 lockdown. Int J Environ Res Public
Health. 18(693)2021.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Diamond A: Executive functions. Annu Rev
Psychol. 64:135–168. 2013.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Di Paolo AL, Nichols ES, Tomfohr-Madsen L,
Giesbrecht GF, Manning KY, Lebel CA and Duerden EG: The association
between prenatal maternal anxiety, infant brain volumes, and
temperament during the COVID-19 pandemic. Transl Psychiatry.
15(283)2025.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Lockhart M, Chaux R, Chevin M, Celle M,
Raia-Barjat T, Patural H, Chabrier S and Giraud A: Classification
performance of the ages and stages questionnaire: Influence of
maternal education level. Children (Basel). 10(449)2023.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Mackin R, Ben Fadel N, Feberova J, Murray
L, Nair A, Kuehn S, Barrowman N and Daboval T: ASQ3 and/or the
Bayley-III to support clinicians' decision making. PLoS One.
12(e0170171)2017.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Nazzari S, Grumi S, Mambretti F, Villa M,
Giorda R and Provenzi L: MOM-COPE Study Group. Maternal and infant
NR3C1 and SLC6A4 epigenetic signatures of the COVID-19 pandemic
lockdown: when timing matters. Transl Psychiatry.
12(386)2022.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Kocher K, Bhattacharya S,
Niforatos-Andescavage N, Almalvez M, Henderson D, Vilain E,
Limperopoulos C and Délot EC: Genome-wide neonatal epigenetic
changes associated with maternal exposure to the COVID-19 pandemic.
BMC Med Genomics. 16(268)2023.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Hill RA, Gibbons A, Suwakulsiri W, Taseska
A, Darke H, Malhotra A, Yee H, Fahey M, Hunt RW, Lim I, et al:
Investigating the impact of severe maternal SARS-CoV-2 infection on
infant DNA methylation and neurodevelopment. Mol Psychiatry.
30:1976–1984. 2025.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Provenzi L, Mambretti F, Villa M, Grumi S,
Citterio A, Bertazzoli E, Biasucci G, Decembrino L, Falcone R,
Gardella B, et al: Hidden pandemic: COVID-19-related stress, SLC6A4
methylation, and infants' temperament at 3 months. Sci Rep.
11(15658)2021.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Berger É, Larose MP, Capuano F, Letarte
MJ, Geoffroy MC, Lupien S, Brendgen M, Boivin M, Vitaro F, Tremblay
R, et al: Hair steroid before and after COVID-19 in preschoolers:
the moderation of family characteristics. Psychoneuroendocrinology.
166(107072)2024.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Manning KY, Long X, Watts D,
Tomfohr-Madsen L, Giesbrecht GF and Lebel C: Prenatal maternal
distress during the COVID-19 pandemic and associations with infant
brain connectivity. Biol Psychiatry. 92:701–708. 2022.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Dijk DJ: Sleep and health: Beyond sleep
duration and sleepiness? J Sleep Res. 21:355–356. 2012.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Quenzer-Alfred C: Transition at a
standstill: Preschoolers' motor development during the COVID-19
pandemic. Early Years. 45:456–471. 2025.
|
|
65
|
Damaskopoulou E, Papakonstantinou E,
Bacopoulou F, Eliopoulos E, Chrousos GP and Vlachakis D: Impact of
the COVID-19 pandemic on children living in shelters and children
in the community (Review). World Acad Sci J. 5(5)2023.
|
|
66
|
Smith KE and Pollak SD: Early life stress
and development: Potential mechanisms for adverse outcomes. J
Neurodev Disord. 12(34)2020.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Boyce WT and Ellis BJ: Biological
sensitivity to context: I. An evolutionary-developmental theory of
the origins and functions of stress reactivity. Dev Psychopathol.
17:271–301. 2005.PubMed/NCBI View Article : Google Scholar
|















